Rapid Identification of Phospholipase A2 Transcripts from Snake Venoms
Abstract
:1. Introduction
2. Results and Discussion
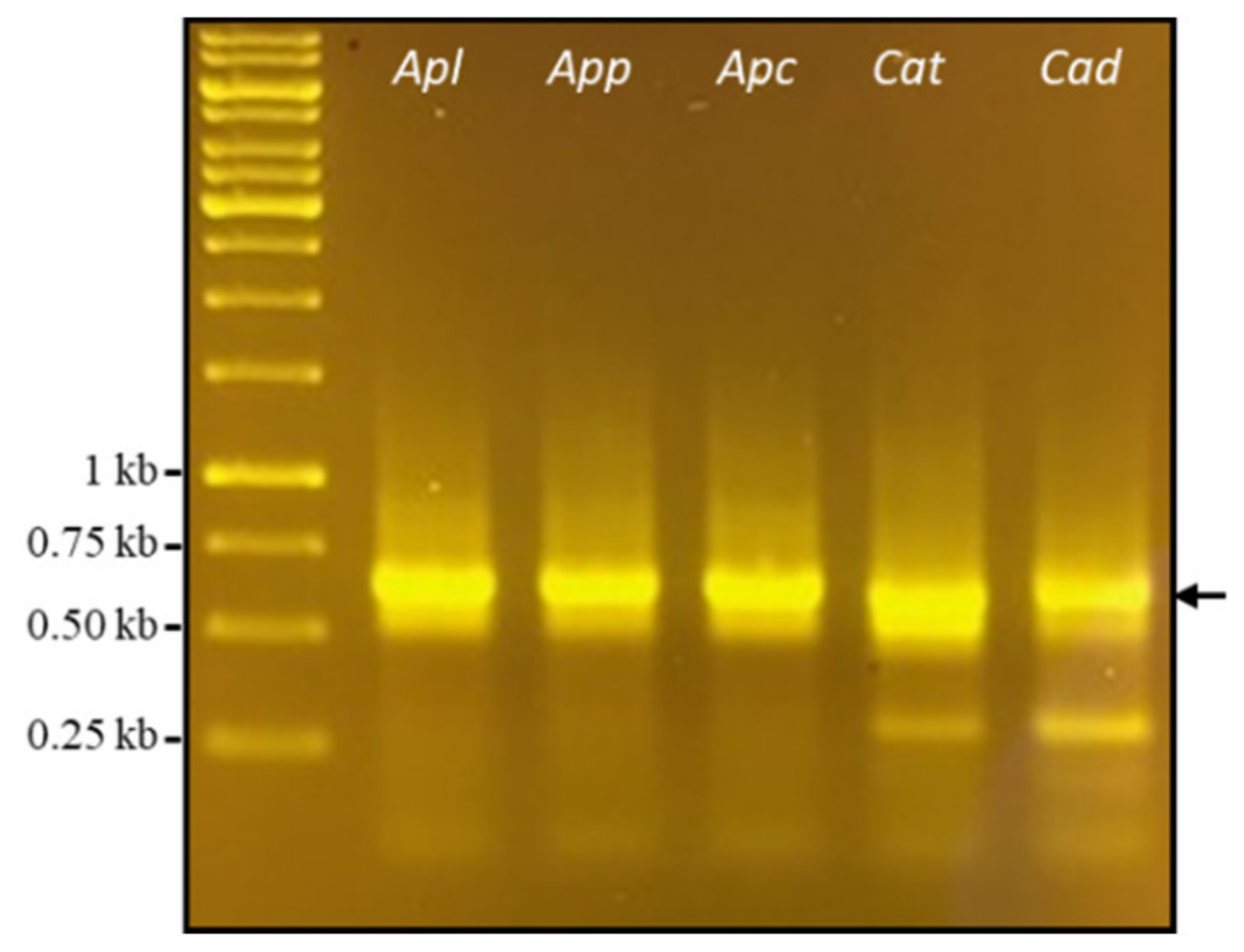
2.1. Cloning PLA2 cDNAs from Snake Venoms
- a)
- PLA2 transcript sequences are intraspecifically conserved;
- b)
- None or one AluI site is at the 5’- or 3’- end of cottonmouth PLA2 transcripts;
- c)
- One AluI site is near the middle of diamondback PLA2 transcripts.
2.2. Placement of Cloned PLA2 Transcripts in Snake Venom PLA2s
3. Materials and Methods
3.1. Cloning PLA2 cDNAs from Crude Venoms
3.2. Phylogenetic Analysis of Snake Venom PLA2s
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Cullis, P.R.; Hope, M.J. Physical properties and functional roles of lipids. In Biochemistry of Lipids and Membranes; Vance, D., Vance, J.E., Eds.; Benjamin-Cummings: Menlo Park, CA, 1985. [Google Scholar]
- Schaloske, R.H.; Dennis, E.A. The phospholipase A2 superfamily and its group numbering system. Biochim. Biophys. Acta 2006, 1761, 1246–1259. [Google Scholar] [CrossRef] [PubMed]
- Murakami, M.; Taketomi, Y.; Girad, C.; Yamamoto, K.; Lambeau, G. Emerging role of secreted phospholipase A2 enzymes: Lessons from transgenic and knockout mice. Biochimie 2010, 92, 561–582. [Google Scholar] [CrossRef] [PubMed]
- Heinrikson, R.L.; Krueger, E.T.; Keim, P.S. Amino acid sequence of phospholipase A2-α from the venom of Crotalus adamanteus. J. Biol. Chem. 1977, 252, 4913–4921. [Google Scholar] [PubMed]
- Dennis, E.A. Diversity of group types, regulation, and function of phospholipase A2. J. Biol. Chem. 1994, 269, 13057–13060. [Google Scholar] [PubMed]
- Maraganore, J.M.; Merutka, G.; Cho, W.; Welches, W.; Kezdy, F.J.; Heinrikson, R.L. A new class of phospholipases A2 with lysine in place of aspartate 49. Functional consequences for calcium and substrate binding. J. Biol. Chem. 1984, 259, 13839–13843. [Google Scholar] [PubMed]
- Chakraborti, S. Phospholipase A2 isoforms: A perspective. Cell. Signal. 2003, 15, 637–665. [Google Scholar] [CrossRef]
- Kini, R.M. Excitement ahead: Structure, functional and mechanism of snake venom phospholipase A2 enzymes. Toxicon 2003, 42, 827–480. [Google Scholar] [CrossRef]
- Angulo, Y.; Lomonte, B. Biochemistry and toxicology of toxins purified from the venom of the snake Bothrops asper. Toxicon 2009, 54, 949–957. [Google Scholar] [CrossRef]
- Fry, B.G.; Wüster, W. Assembling an arsenal: Origin and evolution of the snake venom proteome inferred from phylogenetic analysis of toxin sequences. Mol. Biol. Evol. 2004, 21, 870–883. [Google Scholar] [CrossRef]
- Casewell, N.R.; Wagstaff, S.C.; Wüster, W.; Cook, D.A.N.; Bolton, F.M.S.; King, S.I.; Pla, D.; Sanz, L.; Calvete, J.J.; Harrison, R.A. Medically important differences in snake venom composition are dictated by distinct postgenomic mechanisms. PNAS 2014, 111, 9205–9210. [Google Scholar] [CrossRef]
- Nakashima, K.; Ogawa, T.; Oda, N.; Hattori, M.; Sakaki, Y.; Kihara, H.; Ohno, M. Accelerated evolution of Trimeresurus flavoviridis venom gland phospholipase A2 isozyme. Proc. Natl. Acad. Sci. USA 1993, 90, 5964–5968. [Google Scholar] [CrossRef] [PubMed]
- Ikeda, N.; Chijiwa, T.; Matsubara, K.; Oda-Ueda, N.; Hattori, S.; Matsuda, Y.; Ohno, M. Unique structural characteristics and evolution of a cluster of venom phospholipase A2 isozymes. Proc. Natl. Acad. Sci. USA 2010, 90, 5964–5968. [Google Scholar]
- Shibata, H.; Chijiwa, T.; Oda-Uedaa, N.; Nakamura, H.; Yamajuchi, K.; Hattori, S.; Matsubara, K.; Matsuda, Y.; Yamashita, A.; Isomoto, A.; et al. The habu genome reveals accelerated evolution of venom protein genes. Sci. Rep. 2018, 8, 11300. [Google Scholar] [CrossRef] [PubMed]
- Junqueira-de-Azevedo Ide, L.; Ho, P.L. A survey of gene expression and diversity in the venom glands of the pitviper snake Bothrops insularis through the generation of expressed sequence tags (ESTs). Gene 2002, 299, 279–291. [Google Scholar] [CrossRef]
- Wagstaff, S.C.; Harrison, R.A. Venom gland EST analysis of the saw-scaled viper, Echis ocellatus, reveals novel alpha9beta1 integrin-binding motifs in venom metalloproteinases and a new group of putative toxins, renin-like aspartic proteases. Gene 2006, 377, 21–32. [Google Scholar] [CrossRef] [PubMed]
- Jia, Y.; Cantu, B.A.; Sánchez, E.; Pérez, J.C. Complementary DNA sequencing and identification of mRNAs from venomous gland of Agkistrodon piscivorus leucostoma. Toxicon 2008, 51, 1457–1466. [Google Scholar] [CrossRef] [PubMed]
- Jia, Y.; Ermolinsky, B.; Garza, A.; Provenzano, D. Phospholipase A2 in the venom of three cottonmouth snakes. Toxicon 2017, 135, 84–92. [Google Scholar] [CrossRef]
- Lomonte, B.; Tsai, W.C.; Ureña-Diaz, J.M.; Sanz, L.; Mora-Obando, D.; Sánchez, E.E.; Fry, B.G.; Gutiérrez, J.M.; Gibbs, H.L.; Sovic, M.G.; et al. Venomics of New World pit vipers: Genus-wide comparisons of venom proteomes across Agkistrodon. J. Proteomics 2014, 96, 103–116. [Google Scholar] [CrossRef]
- Ogawa, T.; Oda, N.; Nakashima, K.; Sasaki, H.; Hattori, M.; Sakaki, Y.; Sakaki, Y.; Kihara, H.; Ohno, M. Unusually high conservation of untranslated sequence in cDNA for Trimeresurus flavoviridis phospholipase A2 isozymes. PNAS 1992, 89, 8557–8561. [Google Scholar] [CrossRef]
- Chijiwa, T.; Deshimaru, M.; Nobuhisa, I.; Nakai, M.; Ogawa, T.; Oda, N.; Nakashima, K.; Fukumaki, Y.; Shimohigashi, Y.; Hattori, S.; et al. Regional evolution of venom-gland phospholipase A2 isoenzymes of Trimeresurus flavoviridis snake in the southwestern islands of Japan. Biochem. J. 2000, 347, 491–499. [Google Scholar] [CrossRef]
- Jia, Y.; Pérez, J. Molecular cloning and characterization of cDNAs encoding metalloproteinases from snake venom glands. Toxicon 2010, 55, 2–3. [Google Scholar] [CrossRef] [PubMed]
- Chen, T.B.; Bjourson, A.J.; Orr, D.F.; Kwok, H.F.; Rao, P.F.; Ivanyi, C.; Shaw, C. Unmasking venom gland transcriptomes in reptile venoms. Anal. Biochem. 2002, 311, 152–156. [Google Scholar] [CrossRef]
- Currier, R.B.; Calvete, J.J.; Sanz, L.; Harrison, R.A.; Rowley, P.D.; Wagstaff, S.C. Unusual stability of messenger RNA in snake venom reveals gene expression dynamics of venom replenishment. Plos ONE 2012, 7, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Chen, X.; Wang, L.; Chen, W.; Zhou, M.; Chen, T.; Shaw, C. Cloning and characterization of three novel disintegrin precursors from the venoms of three Atheris species: Atheris chlorechis, Atheris nitschei and Atheris squamigera. Toxicon 2013, 71, 31–40. [Google Scholar] [CrossRef] [PubMed]
- Modahl, C.M.; Mackessy, S.P. Full-length venom protein cDNA sequences from venom-derived mRNA: exploring compositional variation and adaptive multigene evolution. PloS Negl. Trop. Dis. 2016, 10, e0004587. [Google Scholar] [CrossRef] [PubMed]
- Yamaguchi, K.; Chijiwa, T.; Ikeda, N.; Shibata, H.; Fukumaki, Y.; Oda-Ueda, N.; Hattori, S.; Ohno, M. The finding of a group IIE phospholipase A2 gene in a specified segment of Protobothrops flavoviridis genome and its possible evolutionary relationship to group IIA phospholipase A2 genes. Toxins 2014, 6, 3471–3487. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.M.; Wang, J.H.; Tsai, I.H. Molecular cloning and deduced primary structures of acidic and basic phospholipases A2 from the venom of Deinagkistrodon acutus. Toxicon 1996, 34, 1191–1196. [Google Scholar] [CrossRef]
- Malhotra, A.; Creer, S.; Thorpe, R.S.; Harris, J.B.; Stocklin, R.; Favreau, P. Sequence submitted in Genbank. 2013; Unpublished. [Google Scholar]
- Zheng, Y.; Wiens, J.J. Combining phylogenomic and supermatrix approaches, and a time-calibrated phylogeny for squamate reptile (lizards and snakes) based on 52 genes and 4162 species. Mol. Phylogenet. Evol. 2016, 94, 537–547. [Google Scholar] [CrossRef]
- Pyron, R.A.; Burbrink, F.T.; Colli, G.R.; Montes de Oca, A.N.; Vitt, L.J.; Kuczynski, C.A.; Wiens, J.J. The phylogeny of advanced snakes (Colubroidea), with discovery of a new subfamily and comparison of support methods for likelihood trees. Mol. Phylogenet. Evol. 2011, 58, 329–342. [Google Scholar] [CrossRef]
- Pearson, W.R. An introduction to sequence similarity (“homology”) searching. Curr. Protoc. Bioinf. 2013. [Google Scholar] [CrossRef]
- Hall, B.G. Phylogenetic Trees Made Easy: A How-to Manual, 5th ed.; Sinauer Associates, Inc.: Sunderland, MA, USA, 2018; pp. 57–62. [Google Scholar]
- Drummond, A.J.; Suchard, M.A.; Xie, D.; Rambaut, A. Bayesian phylogenetics with BEAUTi and BEAST 1.7. Mol. Biol. Evol. 2012, 29, 1969–1973. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Stecher, G.; Tamura, K. MEGA7: Molecular Evolutionary Genetics Analysis version 7.0 for bigger datasets. Mol. Biol. Evol. 2016, 33, 1870–1874. [Google Scholar] [CrossRef] [PubMed]
- Gouy, M.; Guindon, S.; Gascuel, O. SeaView version 4: A multiplatform graphical user interface for sequence alignment and phylogenetic tree building. Mol. Biol. Evol. 2010, 27, 221–224. [Google Scholar] [CrossRef] [PubMed]
- Rambaut, A.; Drummond, A.J.; Xie, D.; Baele, G.; Suchard, M.A. Posterior summarisation in Bayesian phylogenetics using Tracer 1.7. Syst. Biol. 2018, 67, 901–904. [Google Scholar] [CrossRef] [PubMed]
- Drummond, A.J.; Rambaut, A. BEAST: Bayesian evolutionary analysis by sampling trees. BMC Evol Biol. 2007, 7, 214. [Google Scholar] [CrossRef] [PubMed]
- Fry, G.G.; Winter, K.; Norman, J.A.; Roelants, K.; Nabuurs, R.J.; van Osch, M.J.; Teeuwisse, W.M.; van der Weerd, L.; McNaughtan, J.E.; Kwok, H.F.; et al. Functional and structural diversification of the Anguimoupha lizard venom system. Mol. Cell. Proteomics 2010, 9, 2369–2390. [Google Scholar] [CrossRef] [PubMed]
- Figtree, version 1.4.3; graphical viewer of phylogenetic trees; Institute of Evolutionary Biology, University of Edinburgh: Edinburgh, UK, 2014.



© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jia, Y.; Olvera, P.; Rangel, F.; Mendez, B.; Reddy, S. Rapid Identification of Phospholipase A2 Transcripts from Snake Venoms. Toxins 2019, 11, 69. https://doi.org/10.3390/toxins11020069
Jia Y, Olvera P, Rangel F, Mendez B, Reddy S. Rapid Identification of Phospholipase A2 Transcripts from Snake Venoms. Toxins. 2019; 11(2):69. https://doi.org/10.3390/toxins11020069
Chicago/Turabian StyleJia, Ying, Pablo Olvera, Frida Rangel, Bianca Mendez, and Samir Reddy. 2019. "Rapid Identification of Phospholipase A2 Transcripts from Snake Venoms" Toxins 11, no. 2: 69. https://doi.org/10.3390/toxins11020069
APA StyleJia, Y., Olvera, P., Rangel, F., Mendez, B., & Reddy, S. (2019). Rapid Identification of Phospholipase A2 Transcripts from Snake Venoms. Toxins, 11(2), 69. https://doi.org/10.3390/toxins11020069



