Assessment of Fusarium Infection and Mycotoxin Contamination of Wheat Kernels and Flour Using Hyperspectral Imaging
Abstract
:1. Introduction
2. Results
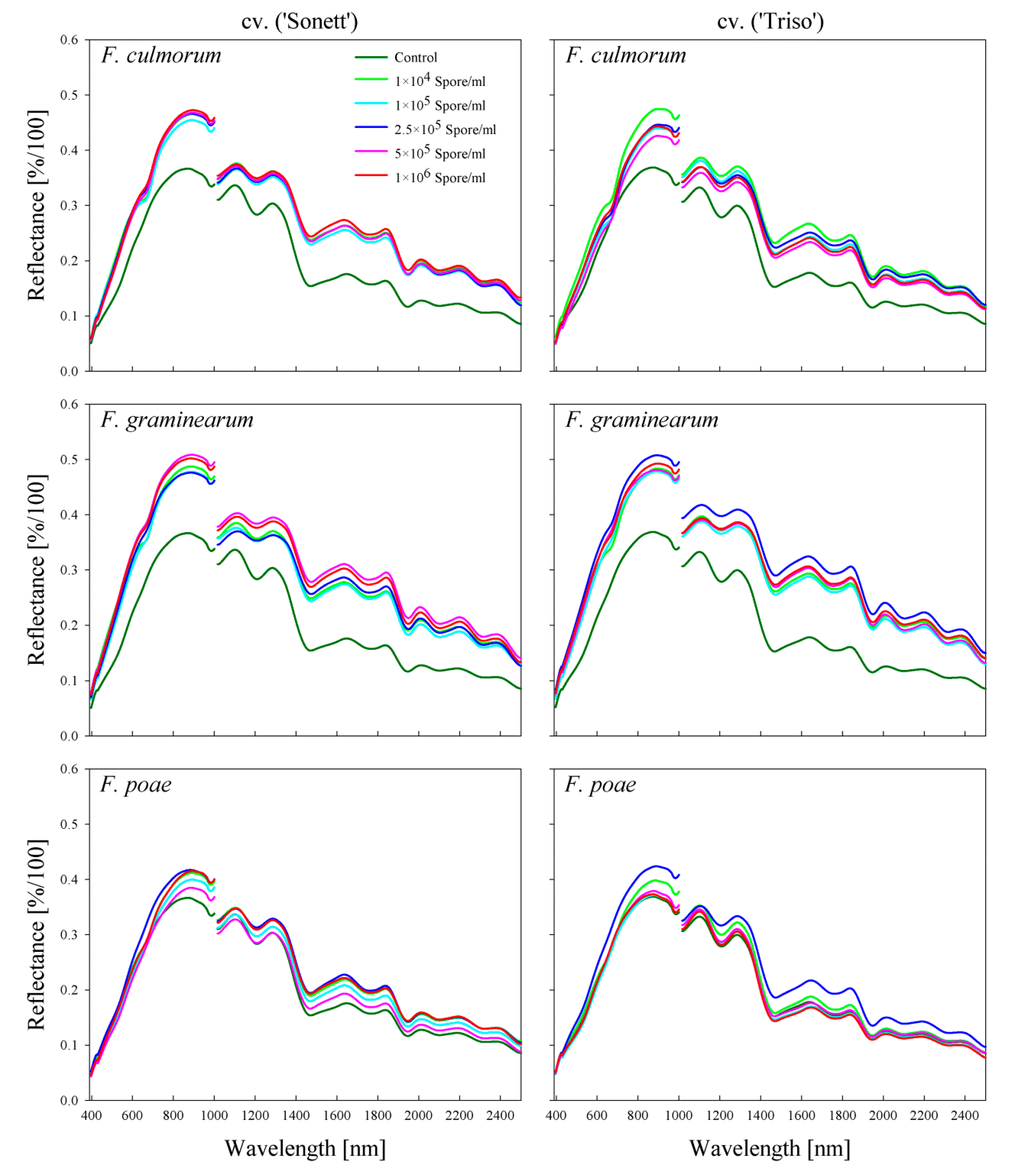
2.1. Effect of Fusarium Infection on the Spectral Signature of the Kernels
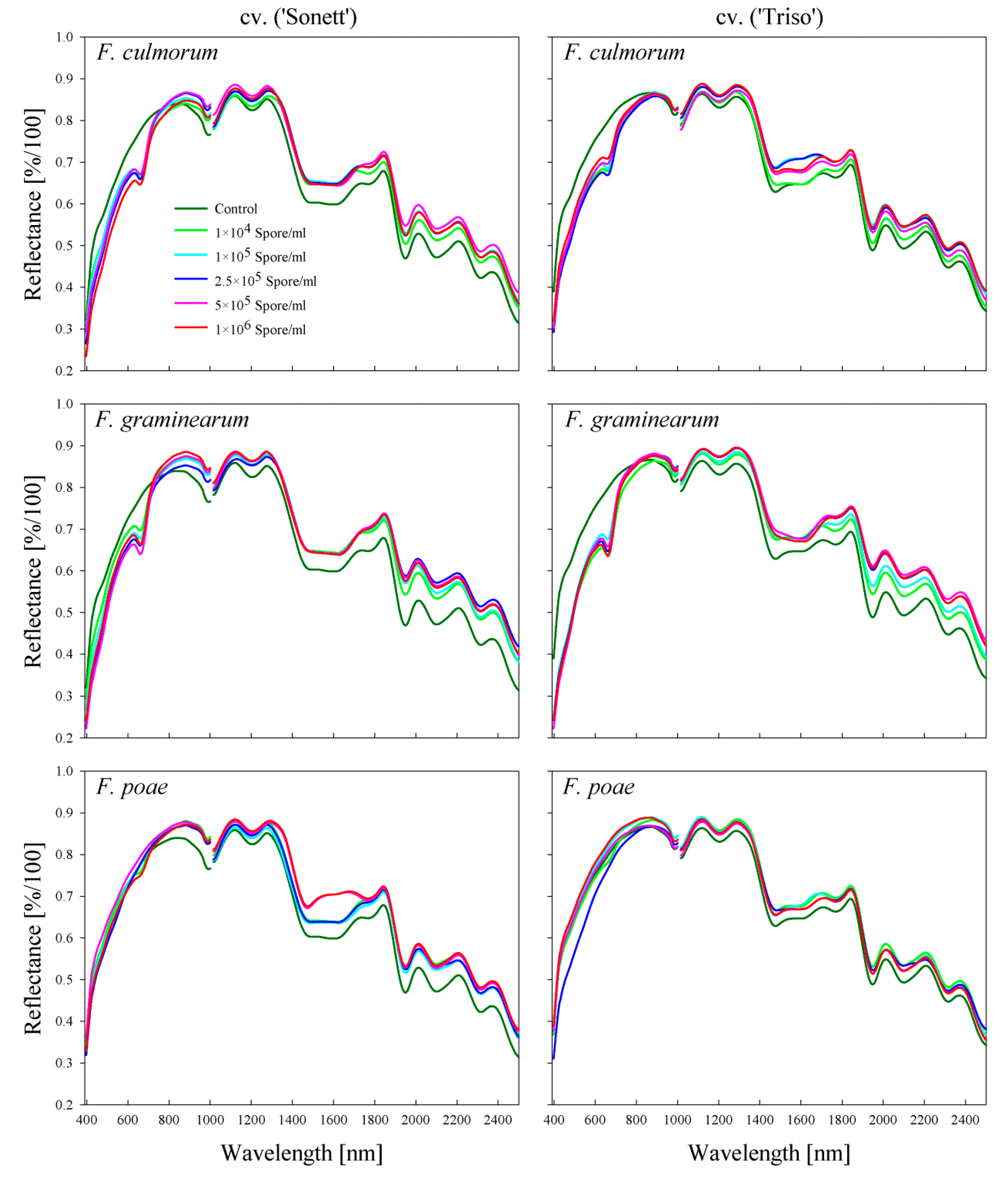
2.2. Comparison between the Spectral Signatures of the Kernels and the Produced Flour
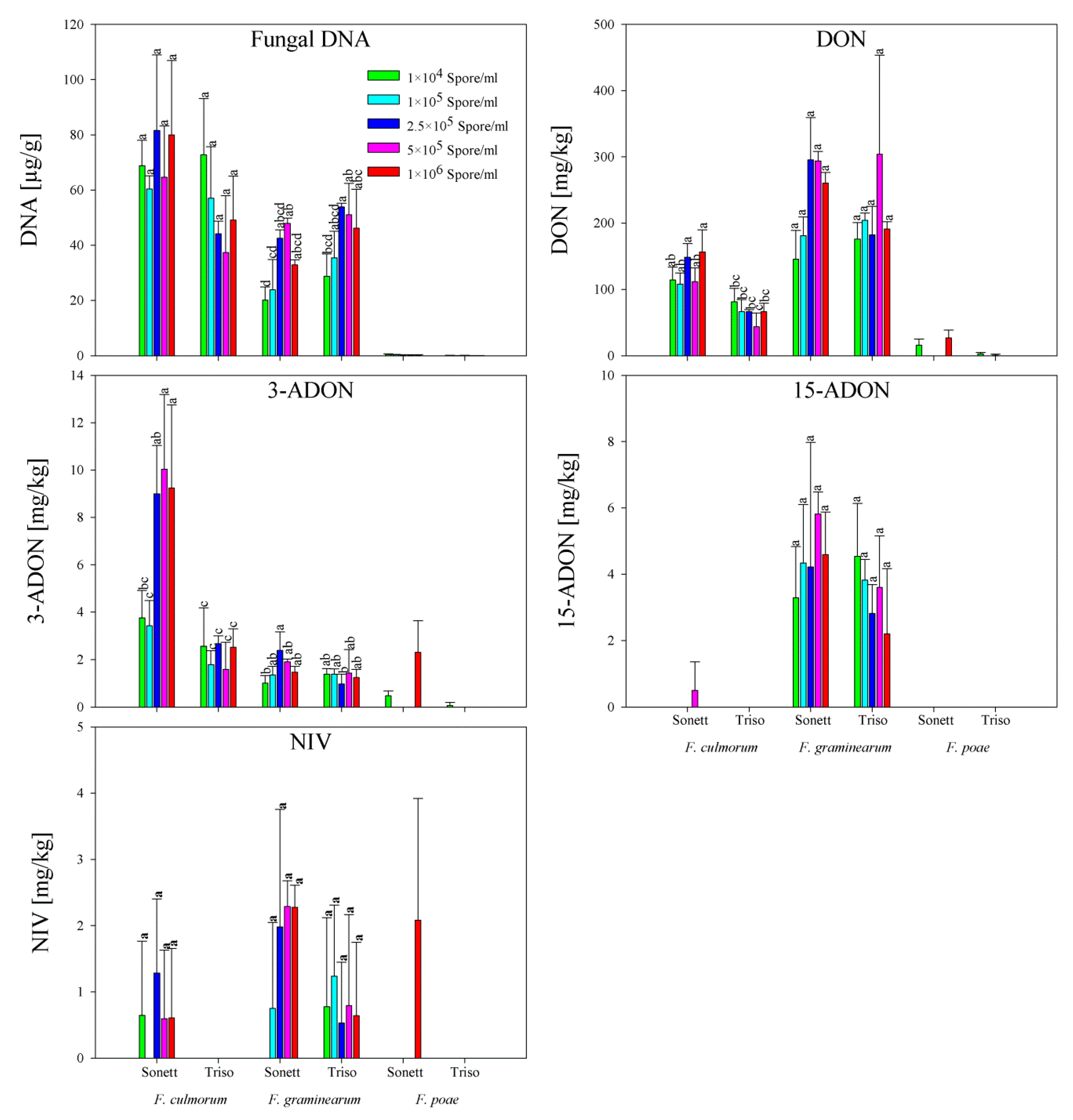
2.3. Fungal DNA and Mycotoxin Content in Wheat Flour
2.4. Correlation between Fungal DNA and Mycotoxin Content
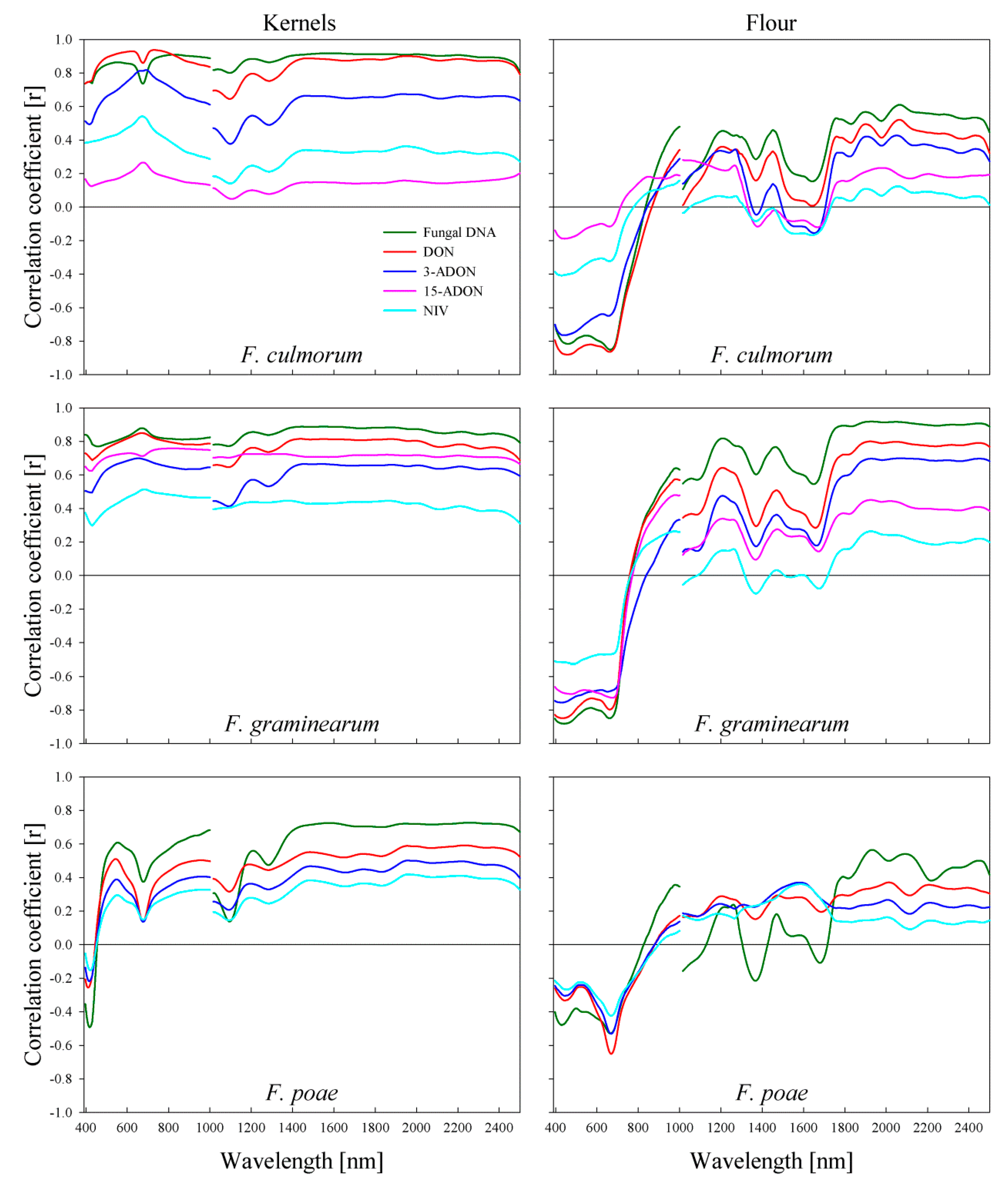
2.5. Correlation of Spectral Signature of Wheat Kernels to Fungal DNA and Mycotoxin Content
2.6. Correlation of Spectral Signature of Wheat Flour to Fungal DNA and Mycotoxin Content
3. Discussion
4. Conclusions
5. Materials and Methods
5.1. Plant Material
5.2. Plant Pathogens and Inoculation Techniques
5.3. Hyperspectral Measurements
5.4. DNA Extraction and Fungal DNA Quantification
5.5. Mycotoxin Extraction and Quantification
5.6. Statistical Analysis
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- McMullen, M.; Bergstrom, G.; De Wolf, E.; Dill-Macky, R.; Hershman, D.; Shaner, G.; Van Sanford, D. A unified effort to fight an enemy of wheat and barley: Fusarium head blight. Plant Dis. 2012, 96, 1712–1728. [Google Scholar] [CrossRef] [PubMed]
- Talas, F.; Parzies, H.K.; Miedaner, T. Diversity in genetic structure and chemotype composition of Fusarium graminearum sensu stricto populations causing wheat head blight in individual fields in Germany. Eur. J. Plant Pathol. 2011, 131, 39–48. [Google Scholar] [CrossRef]
- Alkadri, D.; Rubert, J.; Prodi, A.; Pisi, A.; Manes, J.; Soler, C. Natural co-occurrence of mycotoxins in wheat grains from Italy and Syria. Food Chem. 2014, 157, 111–118. [Google Scholar] [CrossRef] [PubMed]
- van der Lee, T.; Zhang, H.; van Diepeningen, A.; Waalwijk, C. Biogeography of Fusarium graminearum species complex and chemotypes: A review. Food Addit. Contam. Part A Chem. Anal. Control Expo. Risk Assess. 2015, 32, 453–460. [Google Scholar] [CrossRef] [PubMed]
- Pasquali, M.; Beyer, M.; Logrieco, A.; Audenaert, K.; Balmas, V.; Basler, R.; Boutigny, A.-L.; Chrpová, J.; Czembor, E.; Gagkaeva, T.; et al. A European database of Fusarium graminearum and F. culmorum trichothecene genotypes. Front. Microbiol. 2016, 7, 406. [Google Scholar] [CrossRef] [PubMed]
- Alvarez, C.L.; Azcarate, M.P.; Pinto, V.F. Toxigenic potential of Fusarium graminearum sensu stricto isolates from wheat in Argentina. Int. J. Food Microbiol. 2009, 135, 131–135. [Google Scholar] [CrossRef] [PubMed]
- Umpiérrez-Failache, M.; Garmendia, G.; Pereyra, S.; Rodríguez-Haralambides, A.; Ward, T.J.; Vero, S. Regional differences in species composition and toxigenic potential among Fusarium head blight isolates from Uruguay indicate a risk of nivalenol contamination in new wheat production areas. Int. J. Food Microbiol. 2013, 166, 135–140. [Google Scholar] [CrossRef] [PubMed]
- Ward, T.J.; Clear, R.M.; Rooney, A.P.; O’Donnell, K.; Gaba, D.; Patrick, S.; Starkey, D.E.; Gilbert, J.; Geiser, D.M.; Nowicki, T.W. An adaptive evolutionary shift in Fusarium head blight pathogen populations is driving the rapid spread of more toxigenic Fusarium graminearum in North America. Fungal Genet. Biol. 2008, 45, 473–484. [Google Scholar] [CrossRef] [PubMed]
- Kelly, A.C.; Clear, R.M.; O’Donnell, K.; McCormick, S.; Turkington, T.K.; Tekauz, A.; Gilbert, J.; Kistler, H.C.; Busman, M.; Ward, T.J. Diversity of Fusarium head blight populations and trichothecene toxin types reveals regional differences in pathogen composition and temporal dynamics. Fungal Genet. Biol. 2015, 82, 22–31. [Google Scholar] [CrossRef]
- Varga, E.; Wiesenberger, G.; Hametner, C.; Ward, T.J.; Dong, Y.; Schöfbeck, D.; McCormick, S.; Broz, K.; Stückler, R.; Schuhmacher, R.; et al. New tricks of an old enemy: Isolates of Fusarium graminearum produce a type a trichothecene mycotoxin. Environ. Microbiol. 2015, 17, 2588–2600. [Google Scholar] [CrossRef]
- Schaad, N.W.; Frederick, R.D. Real-time PCR and its application for rapid plant disease diagnostics. Can. J. Plant Pathol. 2002, 24, 250–258. [Google Scholar] [CrossRef]
- Krska, R.; Schubert-Ullrich, P.; Molinelli, A.; Sulyok, M.; Macdonal, S.; Crews, C. Mycotoxin analysis: An update. Food Addit. Contam. 2008, 25, 152–163. [Google Scholar] [CrossRef] [PubMed]
- Meneely, J.P.; Ricci, F.; van Egmond, H.P.; Elliott, C.T. Current methods of analysis for the determination of trichothecene mycotoxins in food. Trends Anal. Chem. 2011, 30, 192–203. [Google Scholar] [CrossRef]
- Del Fiore, A.; Reverberi, M.; Ricelli, A.; Pinzari, F.; Serranti, S.; Fabbri, A.A.; Bonifazi, G.; Fanelli, C. Early detection of toxigenic fungi on maize by hyperspectral imaging analysis. Int. J. Food Microbiol. 2010, 144, 64–71. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Heitschmidt, G.W.; Windham, W.R.; Feldner, P.; Ni, X.; Chu, X. Feasibility of detecting aflatoxin B1 on inoculated maize kernels surface using VIS/NIR hyperspectral imaging. J. Food Sci. 2015, 80, 116–122. [Google Scholar] [CrossRef] [PubMed]
- Kimuli, D.; Lawrence, K.C.; Yoon, S.-C.; Wang, W.; Heitschmidt, G.W.; Zhao, X. A SWIR hyperspectral imaging method for classifying Aflatoxin B1 contaminated maize kernels. In Proceedings of the 2017 ASABE Annual International Meeting, Spokane, WA, USA, 16–19 July 2017. Paper No. 1700764. [Google Scholar]
- Chu, X.; Wang, W.; Yoon, S.-C.; Ni, X.; Heitschmidt, G.W. Detection of aflatoxin B1 (AFB1) in individual maize kernels using short wave infrared (SWIR) hyperspectral imaging. Biosyst. Eng. 2017, 157, 13–23. [Google Scholar] [CrossRef] [Green Version]
- Singh, C.B.; Jayas, D.S.; Paliwal, J.; White, N.D.G. Detection of sprouted and midge-damaged wheat kernels using near-infrared hyperspectral imaging. In Proceedings of the CSBE/SCGAB 2008 Annual Conference, North Vancouver, BC, Canada, 13–16 July 2008. Paper No. CSBE08-198. [Google Scholar]
- Singh, C.B.; Jayas, D.S.; Paliwal, J.; White, N.D.G. Detection of insect-damaged wheat kernels using near-infrared hyperspectral imaging. J. Stored Prod. Res. 2009, 45, 151–158. [Google Scholar] [CrossRef]
- Peiris, K.H.S.; Pumphrey, M.O.; Dowell, F.E. NIR absorbance characteristics of deoxynivalenol and of sound and Fusarium- damaged wheat kernels. J. Near Infrared Spectrosc. 2009, 17, 213–221. [Google Scholar] [CrossRef]
- Peiris, K.H.S.; Pumphrey, M.O.; Dong, Y.; Maghirang, E.B.; Berzonsky, W.; Dowell, F.E. Near-infrared spectroscopic method for identification of Fusarium head blight damage and prediction of deoxynivalenol in single wheat kernels. Cereal Chem. 2010, 87, 511–517. [Google Scholar] [CrossRef]
- Kautzman, M.E.; Wickstrom, M.L.; Scott, T.A. The use of near infrared transmittance kernel sorting technology to salvage high quality grain from grain downgraded due to Fusarium damage. Anim. Nutr. 2015, 1, 41–46. [Google Scholar] [CrossRef]
- Beyer, M.; Pogoda, F.; Ronellenfitsch, F.K.; Hoffmann, L.; Udelhoven, T. Estimating deoxynivalenol contents of wheat samples containing different levels of Fusarium-damaged kernels by diffuse reflectance spectrometry and partial least square regression. Int. J. Food Microbiol. 2010, 142, 370–374. [Google Scholar] [CrossRef] [PubMed]
- Delwiche, S.R.; Hareland, G.A. Detection of scab-damaged hard red spring wheat kernels by near-infrared reflectance. Cereal Chem. 2004, 81, 643–649. [Google Scholar] [CrossRef]
- Delwiche, S.D.; Kim, M.S.; Dong, Y. Fusarium damage assessment in wheat kernels by VIS/NIR hyperspectral imaging. Sens. Instrum. Food Qual. Saf. 2011, 5, 63–71. [Google Scholar] [CrossRef]
- Dowell, F.E.; Ram, M.S.; Seitz, L.M. Predicting scab, vomitoxin, and ergosterol in single wheat kernels using near-infrared spectroscopy. Cereal Chem. 1999, 76, 573–576. [Google Scholar] [CrossRef]
- Shahin, M.A.; Symons, S.J. Detection of Fusarium damage in Canadian wheat using visible/near-infrared hyperspectral imaging. J. Food Meas. Charact. 2012, 6, 3–11. [Google Scholar] [CrossRef]
- Yang, I.C.; Delwiche, S.R.; Chen, S.; Lo, Y.M. Enhancement of Fusarium head blight detection in free-falling wheat kernels using a bichromatic pulsed LED design. Opt. Eng. 2009, 48, 023602. [Google Scholar]
- Jirsa, O.; Polišenská, I. Identification of Fusarium damaged wheat kernels using image analysis. Acta Univ. Agric. Silvic. Mendel. Brun. 2011, 59, 125–130. [Google Scholar] [CrossRef]
- Barbedo, J.G.A.; Tibola, C.S.; Fernandes, J.M.C. Detecting Fusarium head blight in wheat kernels using hyperspectral imaging. Biosyst. Eng. 2015, 131, 65–76. [Google Scholar] [CrossRef]
- McMullen, M.; Jones, R.; Gallenberg, D. Scab of wheat and barley: A re-emerging disease of devastating impact. Plant Dis. 1997, 81, 1340–1348. [Google Scholar] [CrossRef]
- Alisaac, E.; Behmann, J.; Kuska, M.T.; Dehne, H.W.; Mahlein, A.K. Hyperspectral quantification of wheat resistance to Fusarium head blight: Comparison of two Fusarium species. Eur. J. Plant Pathol. 2018, 152, 869–884. [Google Scholar] [CrossRef]
- Mahlein, A.-K.; Alisaac, E.; Al Masri, A.; Behmann, J.; Dehne, H.-W.; Oerke, E.-C. Comparison and combination of thermal, fluorescence and hyperspectral imaging for monitoring Fusarium head blight of wheat on spikelet scale. Sensors 2019, 19, 2281. [Google Scholar] [CrossRef] [PubMed]
- Mahlein, A.-K.; Kuska, M.T.; Behmann, J.; Polder, G.; Walter, A. Hyperspectral sensors and imaging technologies in phytopathology: State of the art. Annu. Rev. Phytopathol. 2018, 56, 535–558. [Google Scholar] [CrossRef] [PubMed]
- Boyacioglu, D.; Hettiarachchy, N.S. Changes in some biochemical components of wheat grain that was infected with Fusarium graminearum. J. Cereal Sci. 1995, 21, 57–62. [Google Scholar] [CrossRef]
- Siuda, R.; Grabowski, A.; Lenc, L.; Ralcewicz, M.; Spychaj-Fabisiak, E. Influence of the degree of fusariosis on technological traits of wheat grain. Int. J. Food Sci. Technol. 2010, 45, 2596–2604. [Google Scholar] [CrossRef]
- Kreuzberger, M.; Limsuwan, S.; Eggert, K.; Karlovsky, P.; Pawelzik, E. Impact of Fusarium spp. infection of bread wheat (Triticum aestivum L.) on composition and quality of flour in association with EU maximum level for deoxynivalenol. J. Appl. Bot. Food Qual. 2015, 88, 177–185. [Google Scholar]
- Wieser, H. Simple determination of gluten protein types in wheat flour by turbidimetry. Cereal Chem. 2000, 77, 48–52. [Google Scholar] [CrossRef]
- Wang, J.; Wieser, H.; Pawelzik, E.; Weinert, J.; Keutgen, A.J.; Wolf, G.A. Impact of the fungal protease produced by Fusarium culmorum on the protein quality and breadmaking properties of winter wheat. Eur. Food Res. Technol. 2005, 220, 552–559. [Google Scholar] [CrossRef]
- Nightingale, M.J.; Marchylo, B.A.; Clear, R.M.; Dexter, J.E.; Preston, K.R. Fusarium head blight: Effect of fungal proteases on wheat storage proteins. Cereal Chem. 1999, 76, 150–158. [Google Scholar] [CrossRef]
- Gärtner, B.H.; Munich, M.; Kleijer, G.; Mascher, F. Characterisation of kernel resistance against Fusarium infection in spring wheat by baking quality and mycotoxin assessments. Eur. J. Plant Pathol. 2008, 120, 61–68. [Google Scholar] [CrossRef]
- Hellin, P.; Duvivier, M.; Dedeurwaerder, G.; Bataille, C.; De Proft, M.; Legrève, A. Evaluation of the temporal distribution of Fusarium graminearum airborne inoculum above the wheat canopy and its relationship with Fusarium head blight and DON concentration. Eur. J. Plant Pathol. 2018, 151, 1049–1064. [Google Scholar] [CrossRef]
- Castañares, E.; Albuquerque, D.R.; Dinolfo, M.I.; Pinto, V.F.; Patriarca, A.; Stenglein, S.A. Trichothecene genotypes and production profiles of Fusarium graminearum isolates obtained from barley cultivated in Argentina. Int. J. Food Microbiol. 2014, 179, 57–63. [Google Scholar] [CrossRef] [PubMed]
- Nielsen, L.K.; Cook, D.J.; Edwards, S.G.; Ray, R.V. The prevalence and impact of Fusarium head blight pathogens and mycotoxins on malting barley quality in UK. Int. J. Food Microbiol. 2014, 179, 38–49. [Google Scholar] [CrossRef] [PubMed]
- Siou, D.; Gelisse, S.; Laval, V.; Repincay, C.; Canales, R.; Suffert, F.; Lannou, C. Effect of wheat spike infection timing on Fusarium head blight development and mycotoxin accumulation. Plant Pathol. 2014, 63, 390–399. [Google Scholar] [CrossRef]
- Beccari, G.; Arellano, C.; Covarelli, L.; Tini, F.; Sulyok, M.; Cowger, C. Effect of wheat infection timing on Fusarium head blight causal agents and secondary metabolites in grain. Int. J. Food Microbiol. 2019, 290, 214–225. [Google Scholar] [CrossRef] [PubMed]
- Delwiche, S.R. Classification of scab-and other mold-damaged wheat kernels by near-infrared reflectance spectroscopy. Trans. ASAE 2003, 46, 731–738. [Google Scholar] [CrossRef]
- Lancashire, P.D.; Bleiholder, H.; van den Boom, T.; Langeluddecke, P.; Stauss, R.; Weber, E.; Witzenberger, A. A uniform decimal code for growth stages of crops and weeds. Ann. Appl. Biol. 1991, 119, 561–601. [Google Scholar] [CrossRef]
- Schilling, A.G.; Möller, E.M.; Geiger, H.G. Polymerase chain reaction-based assays for species-specific detection of Fusarium culmorum, F. graminearum and F. avenaceum. Phytopathology 1996, 86, 515–522. [Google Scholar] [CrossRef]
- Nicholson, P.; Simpson, D.R.; Weston, G.; Rezanoor, H.N.; Lees, A.K.; Parry, D.W.; Joyce, D. Detection and quantification of Fusarium culmorum and Fusarium graminearum in cereals using PCR assays. Physiol. Mol. Plant Pathol. 1998, 53, 17–37. [Google Scholar] [CrossRef]
- Parry, D.W.; Nicholson, P. Development of a PCR assay to detect Fusarium poae in wheat. Plant Pathol. 1996, 45, 383–391. [Google Scholar] [CrossRef]

| Fungal DNA-Mycotoxin | F. culmorum | F. graminearum | F. poae |
|---|---|---|---|
| Fungal DNA-DON | 0.90 | 0.80 | n.s. |
| Fungal DNA-3-DON | 0.70 | n.s. | n.s. |
| Fungal DNA-15-DON | n.s. * | 0.51 | n.s. |
| DON-3-ADON | 0.85 | n.s | n.s. |
| DON-15-ADON | n.s. | 0.79 | n.s. |
| Pathogen | Denaturation | Annealing | Elongation | Melt Curve | Primers | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| °C | min | °C | min | °C | min | °C | °C | Primer name | Primer sequence (5′-3′) | Amplified fragment | Reference | |
| F. culmorum | 94.0 | 0:20 | 62.0 | 0:40 | 68.0 | 0:45 | 65.0 | 95.0 | OPT18 F OPT18 R | GATGCCAGACCAAGACGAAG GATGCCAGACGCACTAAGAT | 472 bp | Schilling et al. [49] |
| F. graminearum | 94.0 | 0:30 | 61.0 | 0:30 | 68.0 | 0:30 | 55.0 | 95.0 | Fg16N F Fg16N R | ACAGATGACAAGATTCAGGCACA TTCTTTGACATCTGTTCAACCCA | 280 bp | Nicholson et al. [50] |
| F. poae | 94.0 | 0:35 | 62.5 | 0:30 | 68.0 | 0:35 | 55.0 | 95.0 | Fp82 F Fp82 R | CAAGCAAACAGGCTCTTCACC TGTTCCACCTCAGTGACAGGTT | 220 bp | Parry and Nicholson [51] |
| Toxin | Obtained From | Molecular Ion | Parent Ion | Collision Energy [V] | Product Ions | LOD * | LOQ * | Recovery *** |
|---|---|---|---|---|---|---|---|---|
| [mg/kg] | [mg/kg] | % | ||||||
| NIV | Merck | [M-H]− | 357.1 | 10 | 311.1 | 0.007 | 0.025 | 89 |
| (Darmstadt, Germany) | 10 | 281.1 ** | ||||||
| DON | Merck | [M+H]+ | 297.1 | 4 | 249.1 ** | 0.006 | 0.018 | 113 |
| (Darmstadt, Germany) | 64 | 91.2 | ||||||
| 3-ADON | Merck | [M+H]+ | 339.2 | 8 | 231.1 ** | 0.022 | 0.072 | 110 |
| (Darmstadt, Germany) | 8 | 203 | ||||||
| 15-ADON | Merck | [M+H]+ | 339.2 | 10 | 261.0 ** | 0.07 | 0.23 | 111 |
| (Darmstadt, Germany) | 10 | 203 | ||||||
| HT-2 | Enzo Life Sciences | [M+Na]+ | 447 | 17 | 345.1 ** | 0.029 | 0.097 | 96 |
| (Lörrach, Germany) | 17 | 285 | ||||||
| T-2 | Enzo Life Sciences | [M+Na]+ | 489.2 | 98 | 128.1 | 0.029 | 0.092 | 119 |
| (Lörrach, Germany) | 142 | 115.1 ** | ||||||
| ZEA | Romer Labs | [M+H]+ | 319.2 | 12 | 301.1 | 0.098 | 0.322 | 76 |
| (Tulln, Austria) | 12 | 283.0 ** | ||||||
| Enniatin A1 | Merck | [M+H]+ | 668.4 | 20 | 228.2 | 0.029 | 0.095 | 68 |
| (Darmstadt, Germany) | 20 | 210.2 ** | ||||||
| Enniatin B1 | Merck | [M+H]+ | 654.4 | 23 | 228.2 | 0.046 | 0.151 | 80 |
| (Darmstadt, Germany) | 23 | 210.2 ** |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Alisaac, E.; Behmann, J.; Rathgeb, A.; Karlovsky, P.; Dehne, H.-W.; Mahlein, A.-K. Assessment of Fusarium Infection and Mycotoxin Contamination of Wheat Kernels and Flour Using Hyperspectral Imaging. Toxins 2019, 11, 556. https://doi.org/10.3390/toxins11100556
Alisaac E, Behmann J, Rathgeb A, Karlovsky P, Dehne H-W, Mahlein A-K. Assessment of Fusarium Infection and Mycotoxin Contamination of Wheat Kernels and Flour Using Hyperspectral Imaging. Toxins. 2019; 11(10):556. https://doi.org/10.3390/toxins11100556
Chicago/Turabian StyleAlisaac, Elias, Jan Behmann, Anna Rathgeb, Petr Karlovsky, Heinz-Wilhelm Dehne, and Anne-Katrin Mahlein. 2019. "Assessment of Fusarium Infection and Mycotoxin Contamination of Wheat Kernels and Flour Using Hyperspectral Imaging" Toxins 11, no. 10: 556. https://doi.org/10.3390/toxins11100556
APA StyleAlisaac, E., Behmann, J., Rathgeb, A., Karlovsky, P., Dehne, H.-W., & Mahlein, A.-K. (2019). Assessment of Fusarium Infection and Mycotoxin Contamination of Wheat Kernels and Flour Using Hyperspectral Imaging. Toxins, 11(10), 556. https://doi.org/10.3390/toxins11100556







