Loop Replacement Enhances the Ancestral Antibacterial Function of a Bifunctional Scorpion Toxin
Abstract
1. Introduction
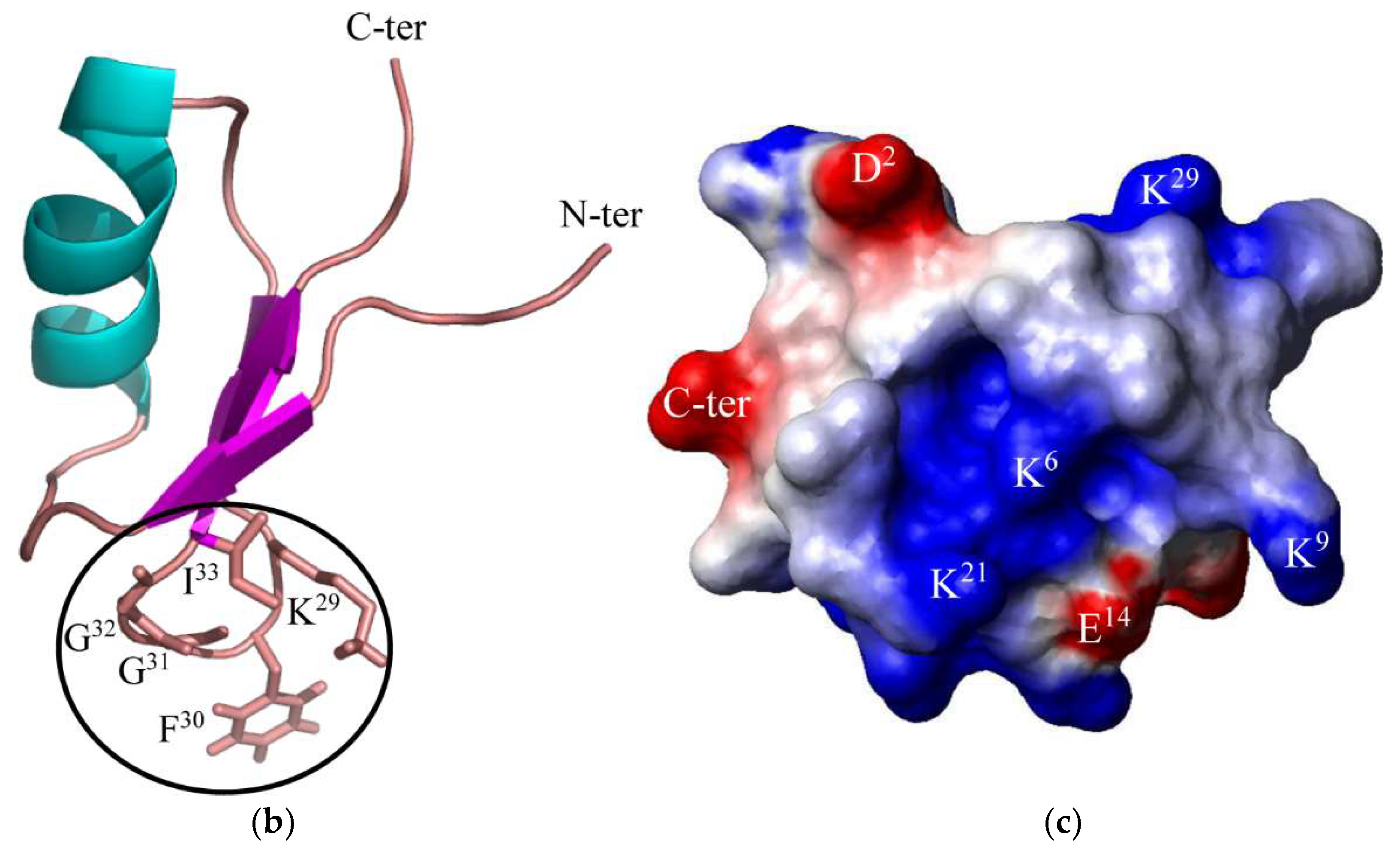
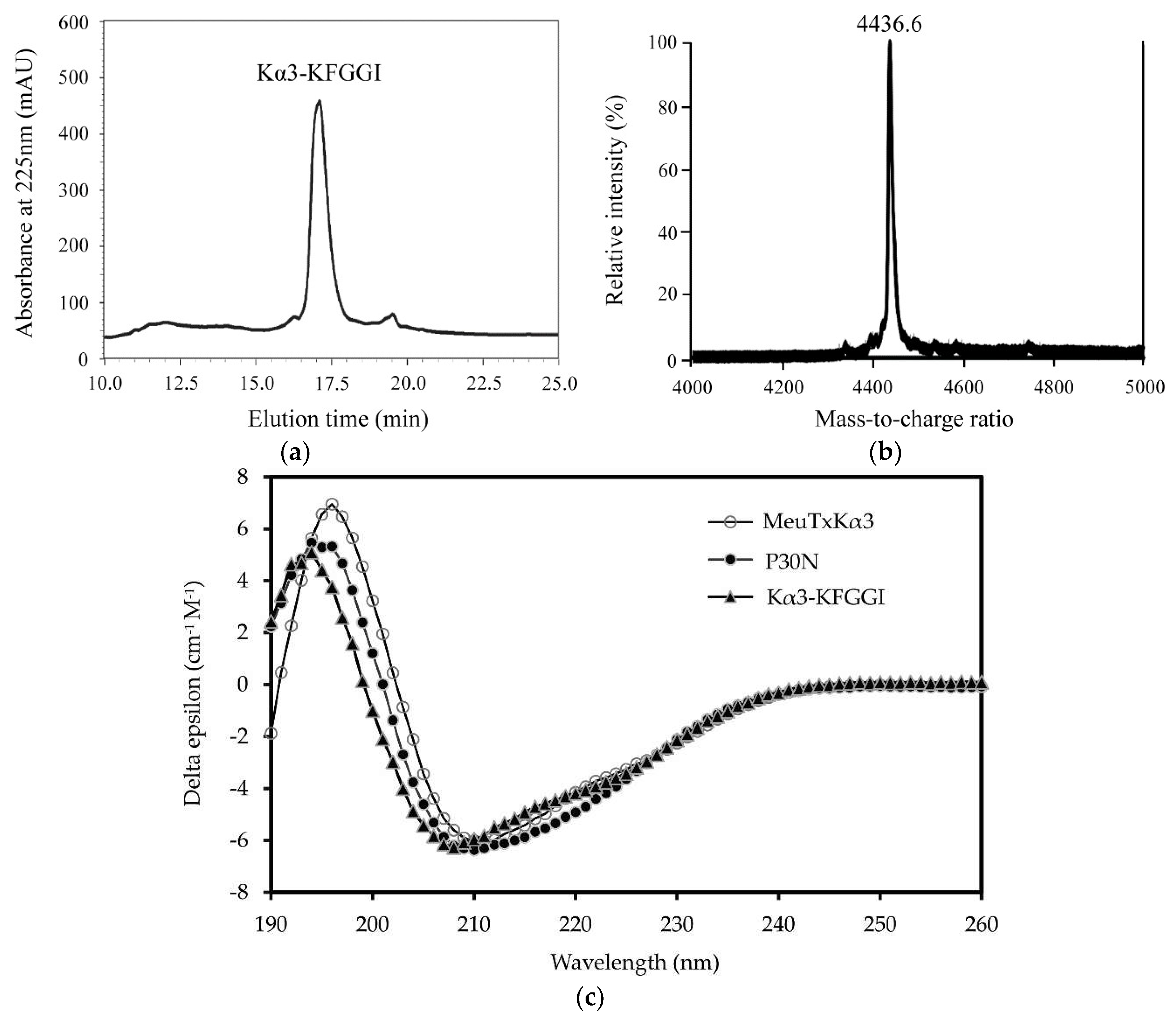
2. Results
3. Discussion
4. Conclusions
5. Materials and Methods
5.1. Site-Directed Mutagenesis
5.2. Expression, Purification, and Characterization of Recombinant Peptide
5.3. Circular Dichroism Spectroscopy
5.4. Inhibition Zone Assay
5.5. Structure Modeling
Author Contributions
Funding
Conflicts of Interest
References
- Rochat, H.; Martin-Eauclaire, M.F. Animal Toxins: Facts and Protocols; Birkhauser Verlag: Berlin, Germany, 2000. [Google Scholar]
- Quintero-Hernández, V.; Jiménez-Vargas, J.M.; Gurrola, G.B.; Valdivia, H.H.; Possani, L.D. Scorpion venom components that affect ion-channels function. Toxicon 2013, 76, 328–342. [Google Scholar] [CrossRef] [PubMed]
- Zhu, S.; Gao, B.; Tytgat, J. Phylogenetic distribution, functional epitopes and evolution of the CSαβ superfamily. Cell. Mol. Life Sci. 2005, 62, 2257–2269. [Google Scholar] [CrossRef] [PubMed]
- Santibáñez-López, C.E.; Possani, L.D. Overview of the Knottin scorpion toxin-like peptides in scorpion venoms: Insights on their classification and evolution. Toxicon 2015, 107, 317–326. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez de la Vega, R.C.; Merino, E.; Becerril, B.; Possani, L.D. Novel interactions between K+ channels and scorpion toxins. Trends Pharmacol. Sci. 2003, 24, 222–227. [Google Scholar] [CrossRef]
- Tytgat, J.; Chandy, K.G.; Garcia, M.L.; Gutman, G.A.; Martin-Eauclaire, M.F.; van der Walt, J.J.; Possani, L.D. A unified nomenclature for short-chain peptides isolated from scorpion venoms: A-KTx molecular subfamilies. Trends Pharmacol. Sci. 1999, 20, 444–447. [Google Scholar] [CrossRef]
- Kuzmenkov, A.I.; Krylov, N.A.; Chugunov, A.O.; Grishin, E.V.; Vassilevski, A.A. Kalium: A database of potassium channel toxins from scorpion venom. Database 2016, baw056. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez de la Vega, R.C.; Possani, L.D. Current views on scorpion toxins specific for K+ channels. Toxicon 2004, 43, 865–875. [Google Scholar] [CrossRef] [PubMed]
- Ashby, M.; Petkova, A.; Hilpert, K. Cationic antimicrobial peptides as potential new therapeutic agents in neonates and children: A review. Curr. Opin. Infect. Dis. 2014, 27, 258–267. [Google Scholar] [CrossRef] [PubMed]
- Malmsten, M. Antimicrobial peptides. Ups. J. Med. Sci. 2014, 119, 199–204. [Google Scholar] [CrossRef] [PubMed]
- Pasupuleti, M.; Schmidtchen, A.; Malmsten, M. Antimicrobial peptides: Key components of the innate immune system. Crit. Rev. Biotechnol. 2012, 32, 143–171. [Google Scholar] [CrossRef] [PubMed]
- Landon, C.; Barbault, F.; Legrain, M.; Guenneugues, M.; Vovelle, F. Rational design of peptides active against the gram positive bacteria Staphylococcus aureus. Proteins 2008, 72, 229–239. [Google Scholar] [CrossRef] [PubMed]
- Zhu, S.; Peigneur, S.; Gao, B.; Umetsu, Y.; Ohki, S.; Tytgat, J. Experimental conversion of a defensin into a neurotoxin: Implications for origin of toxic function. Mol. Biol. Evol. 2014, 31, 546–559. [Google Scholar] [CrossRef] [PubMed]
- Banerjee, A.; Lee, A.; Campbell, E.; Mackinnon, R. Structure of a pore-blocking toxin in complex with a eukaryotic voltage-dependent K+ channel. eLife 2013, 2, e00594. [Google Scholar] [CrossRef] [PubMed]
- Lange, A.; Giller, K.; Hornig, S.; Martin-Eauclaire, M.F.; Pongs, O.; Becker, S.; Baldus, M. Toxin-induced conformational changes in a potassium channel revealed by solid-state NMR. Nature 2006, 440, 959–962. [Google Scholar] [CrossRef] [PubMed]
- Thennarasu, S.; Nagaraj, R. Synthetic peptides corresponding to the β-hairpin loop of rabbit defensin NP-2 show antimicrobial activity. Biochem. Biophys. Res. Commun. 1999, 254, 281–283. [Google Scholar] [CrossRef] [PubMed]
- Yount, N.Y.; Yeaman, M.R. Multidimensional signatures in antimicrobial peptides. Proc. Natl. Acad. Sci. USA 2004, 101, 7363–7368. [Google Scholar] [CrossRef] [PubMed]
- Yount, N.Y.; Yeaman, M.R. Structural congruence among membrane-active host defense polypeptides of diverse phylogeny. Biochim. Biophys. Acta 2006, 1758, 1373–1386. [Google Scholar] [CrossRef] [PubMed]
- Gao, B.; Zhu, S. Identification and characterization of the parasitic wasp Nasonia defensins: Positive selection targeting the functional region? Dev. Comp. Immunol. 2010, 34, 659–668. [Google Scholar] [CrossRef] [PubMed]
- Zhu, S.; Peigneur, S.; Gao, B.; Luo, L.; Jin, D.; Zhao, Y.; Tytgat, J. Molecular diversity and functional evolution of scorpion potassium channel toxins. Mol. Cell. Proteom. 2011, 10, M110.002832. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Gao, B.; Zhu, S. A single-point mutation enhances dual functionality of a scorpion toxin. Comp. Biochem. Physiol. C Toxicol. Pharmacol. 2017, 179, 72–78. [Google Scholar] [CrossRef] [PubMed]
- Zhu, S.; Gao, B.; Harvey, P.J.; Craik, D.J. Dermatophytic defensin with antiinfective potential. Proc. Natl. Acad. Sci. USA 2012, 109, 8495–8500. [Google Scholar] [CrossRef] [PubMed]
- Mitta, G.; Vandenbulcke, F.; Hubert, F.; Roch, P. Mussel defensins are synthesised and processed in granulocytes then released into the plasma after bacterial challenge. J. Cell Sci. 1999, 112, 4233–4242. [Google Scholar] [PubMed]
- Bulet, P.; Cociancich, S.; Reuland, M.; Sauber, F.; Bischoff, R.; Hegy, G.; Van Dorsselaer, A.; Hetru, C.; Hoffmann, J.A. A novel insect defensin mediates the inducible antibacterial activity in larvae of the dragonfly Aeschna cyanea (Paleoptera, Odonata). Eur. J. Biochem. 1992, 209, 977–984. [Google Scholar] [CrossRef] [PubMed]
- Ehret-Sabatier, L.; Loew, D.; Goyffon, M.; Fehlbaum, P.; Hoffmann, J.A.; van Dorsselaer, A.; Bulet, P. Characterization of novel cysteine-rich antimicrobial peptides from scorpion blood. J. Biol. Chem. 1996, 271, 29537–29544. [Google Scholar] [CrossRef] [PubMed]
- Hidalgo, P.; MacKinnon, R. Revealing the architecture of a K+ channel pore through mutant cycles with a peptide inhibitor. Science 1995, 268, 307–310. [Google Scholar] [CrossRef] [PubMed]
- Romi, R.; Crest, M.; Gola, M.; Sampieri, F.; Jacquet, G.; Zerrouk, H.; Mansuelle, P.; Sorokine, O.; Van Dorsselaer, A.; Rochat, H.; et al. Synthesis and characterization of kaliotoxin. Is the 26-32 sequence essential for potassium channel recognition? J. Biol. Chem. 1993, 268, 26302–26309. [Google Scholar] [PubMed]
- Zhu, S.; Tytgat, J. The scorpine family of defensins: Gene structure, alternative polyadenylation and fold recognition. Cell. Mol. Life Sci. 2004, 61, 1751–1763. [Google Scholar] [CrossRef] [PubMed]
- Diego-García, E.; Abdel-Mottaleb, Y.; Schwartz, E.F.; de la Vega, R.C.; Tytgat, J.; Possani, L.D. Cytolytic and K+ channel blocking activities of beta-KTx and scorpine-like peptides purified from scorpion venoms. Cell. Mol. Life Sci. 2008, 65, 187–200. [Google Scholar] [CrossRef] [PubMed]
- Zhu, S.; Gao, B.; Aumelas, A.; del Carmen Rodriguez, M.; Lanz-Mendoza, H.; Peigneur, S.; Diego-Garcia, E.; Martin-Eauclaire, M.-F.; Tytgat, J.; Possani, L.D. MeuTXKβ1, a scorpion venom-derived two-domain potassium channel toxin-like peptide with cytolytic activity. Biochim. Biophys. Acta Proteins Proteom. 2010, 1804, 872–883. [Google Scholar] [CrossRef] [PubMed]
- Gao, B.; Zhu, S. Mesobuthus venom-derived antimicrobial peptides possess intrinsic multifunctionality and differential potential as drugs. Front. Microbiol. 2018, 9, 320. [Google Scholar] [CrossRef] [PubMed]
- Fry, B.G. From genome to “venome”: Molecular origin and evolution of the snake venom proteome inferred from phylogenetic analysis of toxin sequences and related body proteins. Genome Res. 2005, 15, 403–420. [Google Scholar] [CrossRef] [PubMed]
- Hargreaves, A.D.; Swain, M.T.; Hegarty, M.J.; Logan, D.W.; Mulley, J.F. Restriction and recruitment gene duplication and the origin and evolution of snake venom toxins. Genome Biol Evol. 2014, 6, 2088–2095. [Google Scholar] [CrossRef] [PubMed]
- Sartim, M.A.; Sampaio, S.V. Snake venom galactoside-binding lectins: A structural and functional overview. J. Venom. Anim. Toxins Incl. Trop. Dis. 2015, 21, 35. [Google Scholar] [CrossRef] [PubMed]
- Ostrowski, M.; Porowinska, D.; Prochnicki, T.; Prevost, M.; Raynal, B.; Baron, B.; Sauguet, L.; Corringer, P.J.; Faure, G. Neurotoxic phospholipase A2 from rattlesnake as a new ligand and new regulator of prokaryotic receptor GLIC (proton-gated ion channel from G. violaceus). Toxicon 2016, 116, 63–71. [Google Scholar] [CrossRef] [PubMed]
- Gao, B.; Sherman, P.; Luo, L.; Bowie, J.; Zhu, S. Structural and functional characterization of two genetically related meucin peptides highlights evolutionary divergence and convergence in antimicrobial peptides. FASEB J. 2009, 23, 1230–1245. [Google Scholar] [CrossRef] [PubMed]
- Danhier, F.; Le Breton, A.; Préat, V. RGD-based strategies to target α(v)β(3) integrin in cancer therapy and diagnosis. Mol. Pharm. 2012, 9, 2961–2973. [Google Scholar] [CrossRef] [PubMed]
- Matsubara, T.; Onishi, A.; Saito, T.; Shimada, A.; Inoue, H.; Taki, T.; Nagata, K.; Okahata, Y.; Sato, T. Sialic acid-mimic peptides as hemagglutinin inhibitors for anti-influenza therapy. J. Med. Chem. 2010, 53, 4441–4449. [Google Scholar] [CrossRef] [PubMed]
- Zhu, S.; Wei, L.; Yamasaki, K.; Gallo, R.L. Activation of cathepsin L by the cathelin-like domain of protegrin-3. Mol. Immunol. 2008, 45, 2531–2536. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Gao, B.; Zhu, S. Single-point mutation-mediated local amphipathic adjustment dramatically enhances antibacterial activity of a fungal defensin. FASEB J. 2016, 30, 2602–2614. [Google Scholar] [CrossRef] [PubMed]
- Hultmark, D. Quantification of antimicrobial activity, using the inhibition-zone assay. In Techniques in Insect Immunology; Wiesner, A., Dunphy, G.B., Marmaras, V.J., Morishima, I., Sugumaran, M., Yamakawa, M., Eds.; SOS Publications: Fair Haven, NJ, USA, 1998; pp. 103–107. [Google Scholar]
- Eisenberg, D.; Lüthy, R.; Bowie, J.U. VERIFY3D: Assessment of protein models with three-dimensional profiles. Methods Enzymol. 1997, 277, 396–404. [Google Scholar] [PubMed]
- Guex, N.; Peitsch, M.C. SWISS-MODEL and the Swiss-PdbViewer: An environment for comparative protein modeling. Electrophoresis 1997, 18, 2714–2723. [Google Scholar] [CrossRef] [PubMed]

| Species | MeuTxKα3 | P30N | Kα3-KFGGI |
|---|---|---|---|
| Methicillin-resistant Staphylococcus aureus (MRSA), P1374 | N.A. 1 | N.A. | 3.69 |
| Penicillin-resistant Staphylococcus aureus (PRSA), P1383 | 5.39 | 0.87 | 1.34 |
| Penicillin-resistant Staphylococcus epidermidis (PRSE), P1389 | N.A. | N.A. | 5.35 |
| Staphylococcus warneri, CGMCC 1.2824 | N.A. | N.A. | 5.39 |
| Streptococcus mutans, CGMCC 1.2499 | 33.80 | 24.06 | 8.84 |
| Streptococcus salivarius, CGMCC 1.2498 | N.A. | N.A. | 0.71 |
| Streptococcus sanguinis, CGMCC 1.2497 | 3.72 | N.A. | 2.14 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, S.; Gao, B.; Wang, X.; Zhu, S. Loop Replacement Enhances the Ancestral Antibacterial Function of a Bifunctional Scorpion Toxin. Toxins 2018, 10, 227. https://doi.org/10.3390/toxins10060227
Zhang S, Gao B, Wang X, Zhu S. Loop Replacement Enhances the Ancestral Antibacterial Function of a Bifunctional Scorpion Toxin. Toxins. 2018; 10(6):227. https://doi.org/10.3390/toxins10060227
Chicago/Turabian StyleZhang, Shangfei, Bin Gao, Xueli Wang, and Shunyi Zhu. 2018. "Loop Replacement Enhances the Ancestral Antibacterial Function of a Bifunctional Scorpion Toxin" Toxins 10, no. 6: 227. https://doi.org/10.3390/toxins10060227
APA StyleZhang, S., Gao, B., Wang, X., & Zhu, S. (2018). Loop Replacement Enhances the Ancestral Antibacterial Function of a Bifunctional Scorpion Toxin. Toxins, 10(6), 227. https://doi.org/10.3390/toxins10060227





