Antioxidant and Anti-Inflammatory Activities of Hydrolysates and Peptide Fractions Obtained by Enzymatic Hydrolysis of Selected Heat-Treated Edible Insects
Abstract
1. Introduction
2. Materials and Methods
2.1. Samples
2.2. Chemicals
2.3. Methods of Edible Insect Preparation
2.4. The Method of Obtaining Protein Preparation
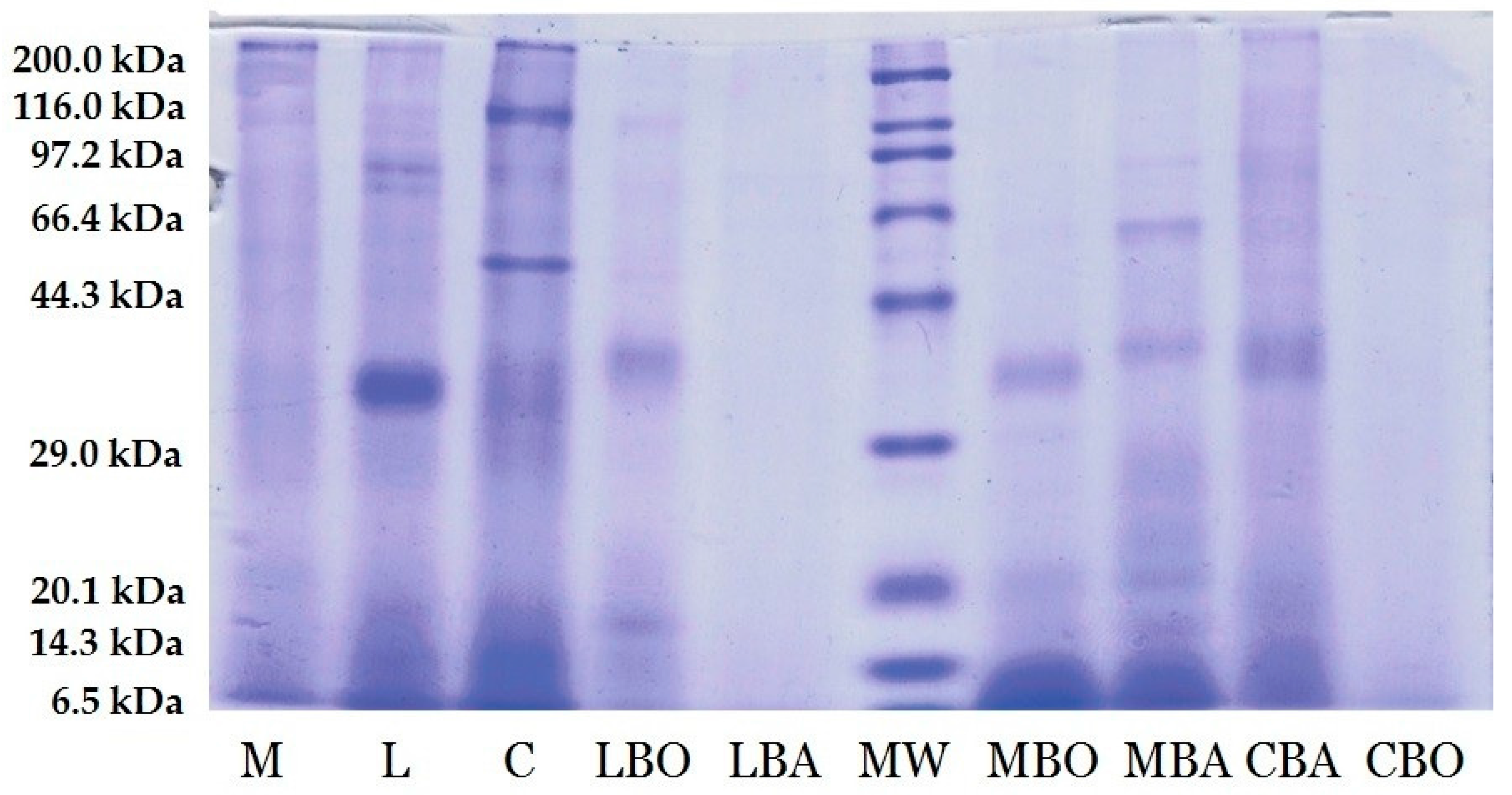
2.5. SDS-Polyacrylamide Gel Electrophoresis Protein Profile
2.6. Enzymatic Hydrolysis and Absorption Process
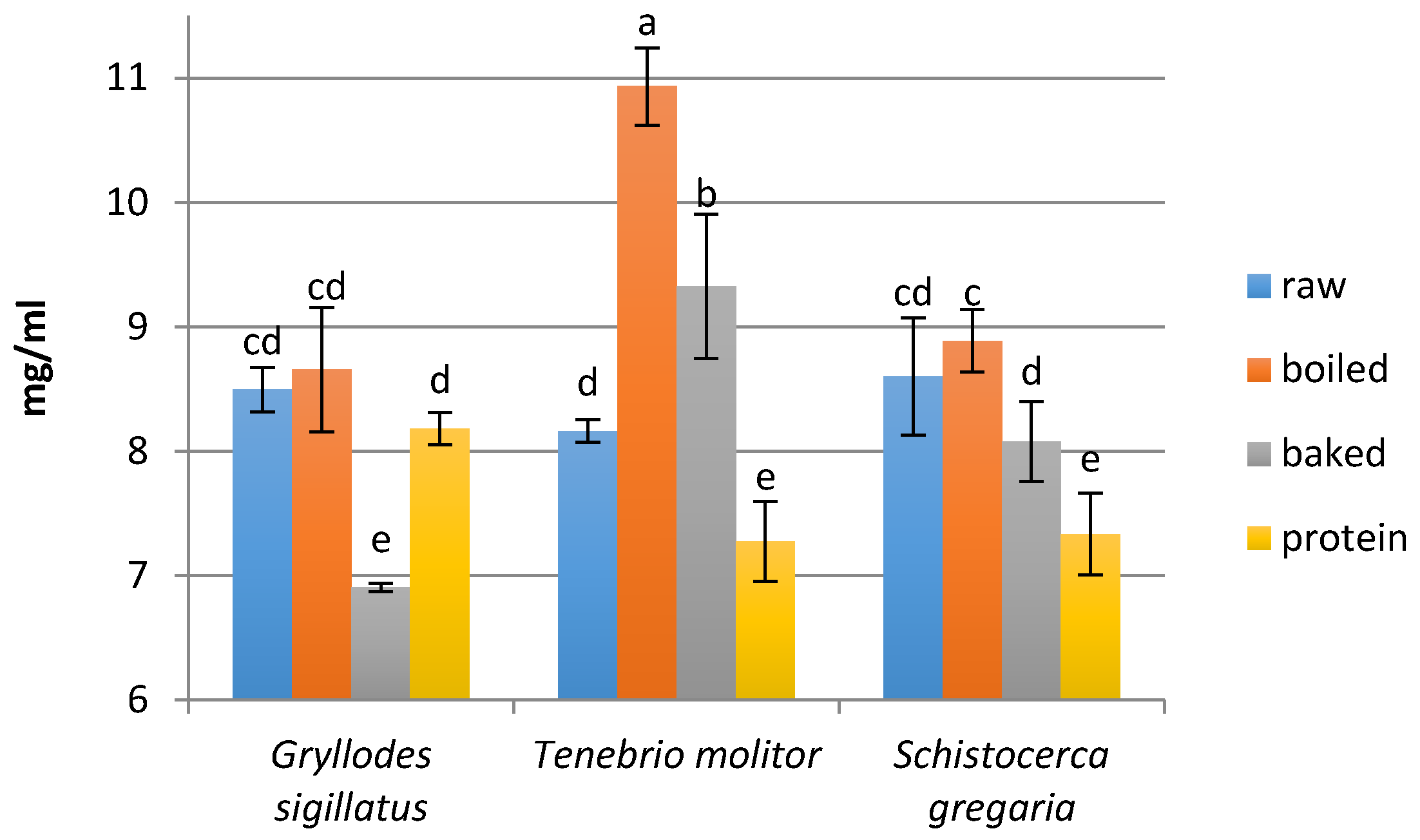
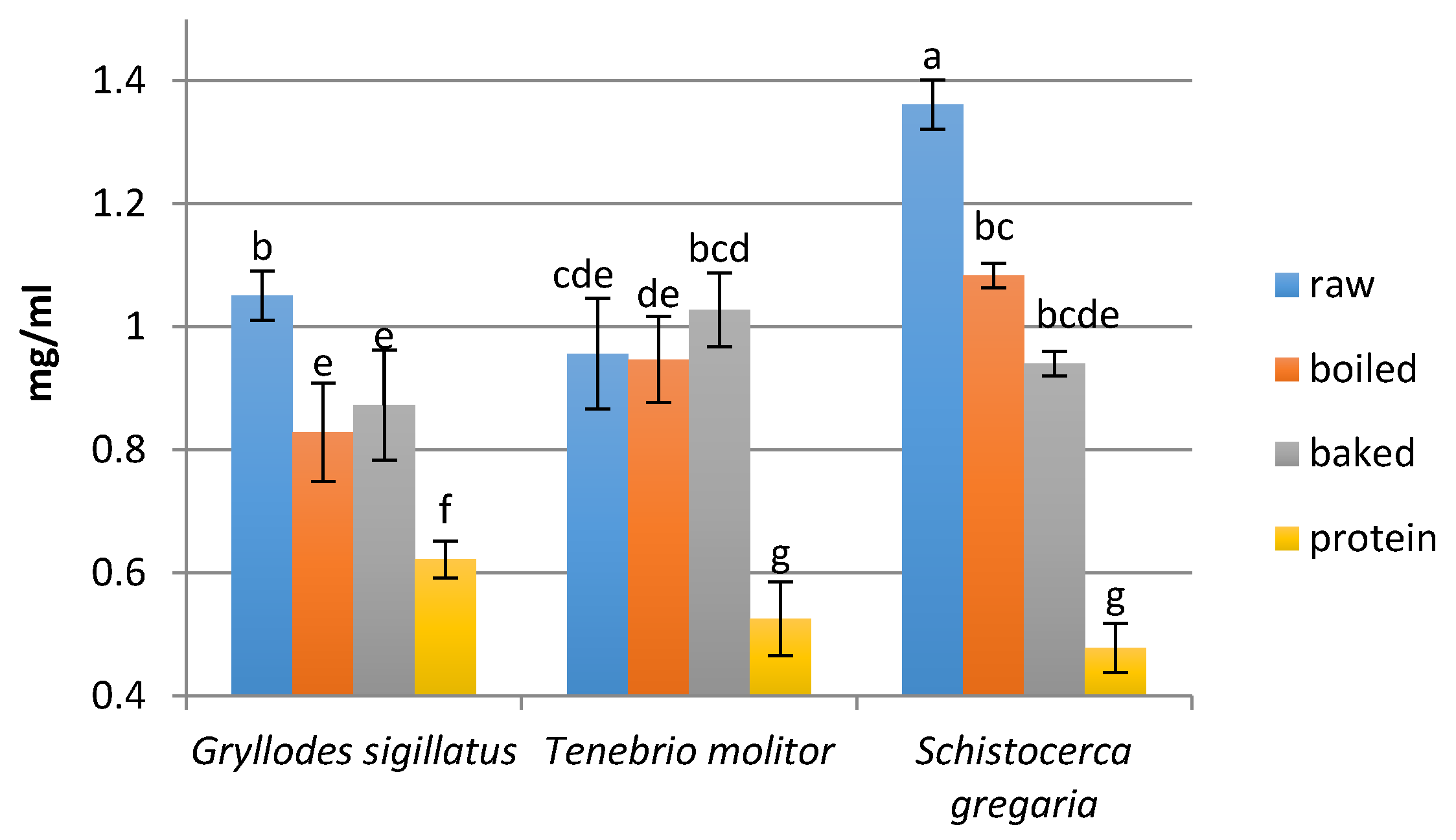
2.7. Determination of the Peptide Concentration
2.8. Free Radical Scavenging Assay
2.8.1. DPPH Radical Scavenging Activity Assay
2.8.2. ABTS Radical Scavenging Activity Assay
2.9. Determination of Fe2+ Chelating Activity
2.10. Ferric-Reducing Power
2.11. Anti-Inflammatory Activity
2.11.1. Lipoxygenase Inhibitory Activity Assay
2.11.2. Cyclooxygenase Inhibitory Activity Assay
2.12. Statistical Analysis
3. Results and Discussion
3.1. Electrophoresis Profile
3.2. Peptide Concentration
3.3. Free Radical Scavenging Assay
3.4. Ion Chelating Activity
3.5. Ferric-Reducing Power
3.6. Lipoxygenase and Cyclooxygenase-2 Inhibitory Activity
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Rumpold, B.A.; Schlüter, O.K. Potential and challenges of insects as an innovative source for food and feed production. Innov. Food Sci. Emerg. Technol. 2013, 17, 1–11. [Google Scholar] [CrossRef]
- Van der Spiegel, M.; Noordam, M.Y.; Van der Fels-Klerx, H.J. Safety of novel protein sources (insects, microalgae, seaweed, duckweed, and rapeseed) and legislative aspects for their application in food and feed production. Compr. Rev. Food Sci. Food Saf. 2013, 12, 662–678. [Google Scholar] [CrossRef]
- Van Huis, A.; Itterbeeck, J.V.; Klunder, H.; Mertens, E.; Halloran, A.; Muir, G.; Vantomme, P. Edible Insects: Future Prospects for Food and Feed Security; Commonwealth Forestry Association: Rome, Italy, 2013; Volume 171, pp. 1–79. [Google Scholar]
- Megido, R.C.; Gierts, C.; Blecker, C.; Brostaux, Y.; Haubruge, É.; Alabi, T.; Francis, F. Consumer acceptance of insect-based alternative meat products in Western countries. Food Qual. Prefer. 2016, 52, 237–243. [Google Scholar] [CrossRef]
- Verkerk, M.C.; Tramper, J.; Van Trijp, J.C.M.; Martens, D.E. Insect cells for human food. Biotechnol. Adv. 2007, 25, 198–202. [Google Scholar] [CrossRef] [PubMed]
- Zielińska, E.; Karaś, M.; Jakubczyk, A. Antioxidant activity of predigested protein obtained from a range of farmed edible insects. Int. J. Food Sci. Technol. 2017, 52, 306–312. [Google Scholar] [CrossRef]
- Gobbetti, M.; Stepaniak, L.; De Angelis, M.; Corsetti, A.; Di Cagno, R. Latent bioactive peptides in milk proteins: Proteolytic activation and significance in dairy processing. Crit. Rev. Food Sci. Nutr. 2002, 42, 223–239. [Google Scholar] [CrossRef] [PubMed]
- Saiga, A.; Tanabe, S.; Nishimura, T. Antioxidant activity of peptides obtained from porcine myofibrillar proteins by protease treatment. J. Agric. Food Chem. 2003, 51, 3661–3667. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Mu, T.H.; Wang, Y.B.; Sun, M.J. Evaluation of free radical-scavenging activities of sweet potato protein and its hydrolysates as affected by single and combination of enzyme systems. Int. J. Food Sci. Technol. 2012, 47, 696–702. [Google Scholar] [CrossRef]
- Karaś, M.; Jakubczyk, A.; Szymanowska, U.; Złotek, U.; Zielińska, E. Digestion and bioavailability of bioactive phytochemicals. Int. J. Food Sci. Technol. 2017, 52, 291–305. [Google Scholar] [CrossRef]
- Torres-Fuentes, C.; Alaiz, M.; Vioque, J. Affinity purification and characterisation of chelating peptides from chickpea protein hydrolysates. Food Chem. 2011, 129, 485–490. [Google Scholar] [CrossRef]
- Karaś, M.; Baraniak, B.; Rybczyńska, K.; Gmiński, J.; Gaweł-Bęben, K.; Jakubczyk, A. The influence of heat treatment of chickpea seeds on antioxidant and fibroblast growth-stimulating activity of peptide fractions obtained from proteins digested under simulated gastrointestinal conditions. Int. J. Food Sci. Technol. 2015, 50, 2097–2103. [Google Scholar] [CrossRef]
- Megías, C.; Pedroche, J.; Yust, M.M.; Girón-Calle, J.; Alaiz, M.; Millán, F.; Vioque, J. Affinity purification of copper chelating peptides from chickpea protein hydrolysates. J. Agric. Food Chem. 2007, 55, 3949–3954. [Google Scholar] [CrossRef] [PubMed]
- Stadtman, E.R. Protein oxidation and aging. Free Radic. Res. 2006, 40, 1250–1258. [Google Scholar] [CrossRef] [PubMed]
- Ali, S.S.; Kasoju, N.; Luthra, A.; Singh, A.; Sharanabasava, H.; Sahu, A.; Bora, U. Indian medicinal herbs as sources of antioxidants. Food Res. Int. 2008, 41, 1–15. [Google Scholar] [CrossRef]
- Carrasco-Castilla, J.; Hernández-Álvarez, A.J.; Jiménez-Martínez, C.; Jacinto-Hernández, C.; Alaiz, M.; Girón-Calle, J.; Vioque, J.; Dávila-Ortiz, G. Antioxidant and metal chelating activities of Phaseolus vulgaris L. var. Jamapa protein isolates, phaseolin and lectin hydrolysates. Food Chem. 2012, 131, 1157–1164. [Google Scholar] [CrossRef]
- Martel-Pelletier, J.; Lajeunesse, D.; Reboul, P.; Pellietier, J.P. Therapeutic role of dual inhibitors of 5-LOX and COX, selective and non-selective non-steroidal anti-inflammatory drugs. Ann. Rheum. Dis. 2003, 62, 501–509. [Google Scholar] [CrossRef] [PubMed]
- Shen, F.Q.; Wang, Z.C.; Wu, S.Y.; Ren, S.Z.; Man, R.; Wang, B.Z.; Zhu, H.L. Synthesis of novel hybrids of pyrazole and coumarin as dual inhibitors of COX-2 and 5-LOX. Bioorg. Med. Chem. Lett. 2017, 27, 3653–3660. [Google Scholar] [CrossRef] [PubMed]
- Zielińska, E.; Baraniak, B.; Karaś, M.; Rybczyńska, K.; Jakubczyk, A. Selected species of edible insects as a source of nutrient composition. Food Res. Int. 2015, 77, 460–466. [Google Scholar] [CrossRef]
- Finke, M.D. Complete nutrient composition of commercially raised invertebrates used as food for insectivores. Zoo Biol. 2012, 21, 269–285. [Google Scholar] [CrossRef]
- Ramos-Elorduy, J. Energy supplied by edible insects from Mexico and their nutritional and ecological importance. Ecol. Food Nutr. 2008, 47, 280–297. [Google Scholar] [CrossRef]
- Ramos-Elorduy, J.; Moreno, J.M.P.; Camacho, V.H.M. Could grasshoppers be a nutritive meal? Food Nutr. Sci. 2012, 3, 164–175. [Google Scholar]
- Rumpold, B.A.; Schlüter, O.K. Nutritional composition and safety aspects of edible insects. Mol. Nutr. Food Res. 2013, 57, 802–823. [Google Scholar] [CrossRef] [PubMed]
- Mlcek, J.; Rop, O.; Borkovcova, M.; Bednarova, M. A comprehensive look at the possibilities of edible insects as food in Europe—A Review. Pol. J. Food Nutr. Sci. 2014, 64, 147–157. [Google Scholar] [CrossRef]
- Adámková, A.; Adámek, M.; Mlček, J.; Borkovcová, M.; Bednářová, M.; Kouřimská, L.; Skácel, J.; Vítová, E. Welfare of the mealworm (Tenebrio molitor) breeding with regard to nutrition value and food safety. Potr. Slovak J. Food Sci. 2017, 11, 460–465. [Google Scholar] [CrossRef]
- EFSA Scientific Committee (EFSA). Risk profile related to production and consumption of insects as food and feed. EFSA J. 2015, 13, 4257. [Google Scholar]
- Girón-Calle, J.; Alaiz, M.; Vioque, J. Effect of chickpea protein hydrolysates on cell proliferation and in vitro bioavailability. Food Res. Int. 2010, 43, 1365–1370. [Google Scholar] [CrossRef]
- Jakubczyk, A.; Karaś, M.; Baraniak, B.; Pietrzak, M. The impact of fermentation and in vitro digestion on formation angiotensin converting enzyme (ACE) inhibitory peptides from pea proteins. Food Chem. 2013, 141, 3774–3780. [Google Scholar] [CrossRef] [PubMed]
- Adler-Nissen, J. Determination of the degree of hydrolysis of food protein hydrolysates with trinitrobenzenesulfonic acid. J. Agric. Food Chem. 1979, 27, 1256–1262. [Google Scholar] [CrossRef]
- Brand-Williams, W.; Cuvelier, M.; Berset, C. Use of a free radical method to evaluate antioxidant activity. LWT Food Sci. Technol. 1995, 28, 25–30. [Google Scholar] [CrossRef]
- Re, R.; Pellegrini, A.; Proteggente, A.; Pannala, M.; Yang, M.; Rice-Evans, C. Antioxidant activity applying an improved ABTS radical cation decolorization assay. Free Radic. Biol. Med. 1999, 26, 1231–1237. [Google Scholar] [CrossRef]
- Decker, E.A.; Welch, B. Role of ferritin as a lipid oxidation catalyst in muscle food. J. Agric. Food Chem. 1990, 38, 674–677. [Google Scholar] [CrossRef]
- Hu, L.; Song, R.; Gu, Z. An antioxidant peptide produced by autolysis reactions from wheat germ. Afr. J. Biotechnol. 2012, 11, 3640–3648. [Google Scholar]
- Axelroad, B.; Cheesborough, T.M.; Laakso, S. Lipoxygenases in soybeans. Methods Enzymol. 1981, 71, 441–451. [Google Scholar]
- You, L.; Zheng, L.; Regenstein, J.M.; Zhao, M.; Liu, D. Effect of thermal treatment on the characteristic properties of loach peptide. Int. J. Food Sci. Technol. 2012, 47, 2574–2581. [Google Scholar] [CrossRef]
- Fu, Y.; Young, J.F.; Therkildsen, M. Bioactive peptides in beef: Endogenous generation through postmortem aging. Meat Sci. 2017, 123, 134–142. [Google Scholar] [CrossRef] [PubMed]
- Serpen, A.; Gökmen, V.; Fogliano, V. Total antioxidant capacities of raw and cooked meats. Meat Sci. 2012, 90, 60–65. [Google Scholar] [CrossRef] [PubMed]
- Wu, Q.Y.; Jia, J.Q.; Tan, G.X.; Xu, J.L.; Gui, Z.Z. Physicochemical properties of silkworm larvae protein isolate and gastrointestinal hydrolysate bioactivities. Afr. J. Biotechnol. 2011, 10, 6145–6153. [Google Scholar] [CrossRef]
- Bräunlich, M.; Slimestad, R.; Wangensteen, H.; Brede, C.; Malterud, K.E.; Barsett, H. Extracts, anthocyanins and procyanidins from Aronia melanocarpa as radical scavengers and enzyme inhibitors. Nutrients 2013, 5, 663–678. [Google Scholar] [CrossRef] [PubMed]
- Nađpal, J.D.; Lesjak, M.M.; Šibul, F.S.; Anačkov, G.T.; Četojević-Simin, D.D.; Mimica-Dukić, N.M.; Beara, I.N. Comparative study of biological activities and phytochemical composition of two rose hips and their preserves: Rosa canina L. and Rosa arvensis Huds. Food Chem. 2016, 192, 907–914. [Google Scholar] [CrossRef] [PubMed]
- Zhu, L.J.; Chen, J.; Tang, X.Y.; Xiong, Y.L. Reducing, radical scavenging, and chelation properties of in vitro digests of alcalase-treated zein hydrolysate. J. Agric. Food Chem. 2008, 56, 2714–2721. [Google Scholar] [CrossRef] [PubMed]
- Lin, S.; Jin, Y.; Liu, M.; Yang, Y.; Zhang, M.; Guo, Y.; Jones, G.; Liu, J.; Yin, Y. Research on the preparation of antioxidant peptides derived from egg white with assisting of high-intensity pulsed electric field. Food Chem. 2013, 139, 300–306. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Luo, Y.; Shen, H.; You, J. Antioxidant activities and functional properties of grass carp (Ctenopharyngodon idellus) protein hydrolysates. J. Sci. Food Agric. 2012, 92, 292–298. [Google Scholar] [CrossRef] [PubMed]
- Raghavendra, M.; Reddy, A.M.; Yadav, P.R.; Raju, A.S.; Kumar, L.S. Comparative studies on the in vitro antioxidant properties of methanolic leafy extracts from six edible leafy vegetables of India. Asian J. Pharm. Clin. Res. 2013, 6, 96–99. [Google Scholar]
- Mukherjee, S.; Pawar, N.; Kulkarni, O.; Nagarkar, B.; Thopte, S.; Bhujbal, A.; Pawar, P. Evaluation of free-radical quenching properties of standard Ayurvedic formulation Vayasthapana Rasayana. BMC Complement. Altern. Med. 2011, 11, 38. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Jiang, B.; Zhang, T.; Mu, W.; Liu, J. Antioxidant and free radical-scavenging activities of chickpea protein hydrolysate (CPH). Food Chem. 2008, 106, 444–450. [Google Scholar] [CrossRef]
- Dong, X.P.; Zhu, B.W.; Zhao, H.X.; Zhou, D.Y.; Wu, H.T.; Yang, J.F.; Li, D.M.; Murata, Y. Preparation and in vitro antioxidant activity of enzymatic hydrolysates from oyster (Crassostrea talienwhannensis) meat. Int. J. Food Sci. Technol. 2010, 45, 978–984. [Google Scholar] [CrossRef]
- Szymanowska, U.; Złotek, U.; Karaś, M.; Baraniak, B. Anti-inflammatory and antioxidative activity of anthocyanins from purple basil leaves induced by selected abiotic elicitors. Food Chem. 2015, 172, 71–77. [Google Scholar] [CrossRef] [PubMed]
- Jäger, A.K.; Eldeen, I.M.S.; van Staden, J. COX-1 and -2 Activity of Rose Hip. Phytother. Res. 2007, 21, 1251–1252. [Google Scholar] [CrossRef] [PubMed]
| Studied Species | Type of Heat Treatment | 2,2′-Azino-bis(3-ethylbenzothiazoline-6-sulfonic Acid) Radical Cation (ABTS•+) Scavenge (IC50 µg/mL) | 2,2-Diphenyl-1-picrylhydrazyl Radical (DPPH•) Scavenge (IC50 µg/mL) | Fe2+ Chelating Activity (IC50 µg/mL) | Reducing Power (Abs700) | Lipoxygenase (LOX) Inhibitory Activity (IC50 mg/mL) | Cyclooxygenase (COX) Inhibitory Activity (IC50 µg/mL) |
|---|---|---|---|---|---|---|---|
| Gryllodes sigillatus | raw | 21.9 ± 1.13 d | 29.45 ± 1.63 h | 85.64 ± 1.02 a | 0.771 ± 0.02 a | 3.14 ± 0.06 a | 69.91 ± 0.4 h |
| boiled | 21.8 ± 0.71 d | 40.3 ± 0.57 g | 53.97 ± 0.88 f | 0.611 ± 0.02 b | 2.04 ± 0.05 c | 146.8 ± 1.6 c | |
| baked | 17.5 ± 0.57 f | 28.5 ± 0.57 i | 38.6 ± 0.6 j | 0.588 ± 0.02 bc | 2.24 ± 0.02 bc | 38.51 ± 0.84 k | |
| protein | 21.65 ± 0.47 de | 29.5 ± 1.6 h | 16.18 ± 0.1 k | 0.551 ± 0.01 bcd | 0.79 ± 0.02 f | 208.24 ± 1.6 a | |
| Tenebrio molitor | raw | 5.3 ± 0.42 j | 109.4 ± 1.13 a | 67.52 ± 0.8 c | 0.477 ± 0.04 defg | 1.57 ± 0.04 d | 58.09 ± 1.24 j |
| boiled | 28.9 ± 1.41 a | 97.45 ± 1.62 c | 69.71 ± 0.69 b | 0.566 ± 0.04 bcde | 1.59 ± 0.04 d | 108.51 ± 2.22 d | |
| baked | 24.5 ± 1.27 c | 85.85 ± 2.33 e | 52.14 ± 0.74 g | 0.504 ± 0.03 def | 2.29 ± 0.06 b | 82.59 ± 2.11 g | |
| protein | 21.4 ± 0.63 e | 40.3 ± 0.6 g | 60.78 ± 0.97 d | 0.373 ± 0.06 h | 0.89 ± 0.02 f | 92.06 ± 1.2 f | |
| Schistocerca gregaria | raw | 27.5 ± 0.99 b | 79.5 ± 1.27 f | 58.21 ± 1.02 e | 0.533 ± 0.02 bcde | 1.3 ± 0.02 e | 10.91 ± 0.4 l |
| boiled | 8.4 ± 0.85 h | 104.5 ± 3.39 b | 60.31 ± 0.87 d | 0.403 ± 0.02 gh | 1.28 ± 0.02 e | 61.6 ± 2.16 i | |
| baked | 6.9 ± 1.13 i | 91.1 ± 1.98 cd | 51.19 ± 0.65 h | 0.452 ± 0.02 efgh | 0.83 ± 0.02 f | 198.51 ± 3.16 b | |
| protein | 16.6 ± 0.86 g | 28.5 ± 0.6 i | 43.13 ± 0.99 i | 0.364 ± 0.02 fgh | 0.65 ± 0.02 f | 98.13 ± 2.14 e |
| Studied Species | Type of Heat Treatment | ABTS•+ Scavenge (IC50 µg/mL) | DPPH• Scavenge (IC50 µg/mL) | Fe2+ Chelating Activity (IC50 µg/mL) | Reducing Power (Abs700) | LOX Inhibitory Activity (IC50 µg/mL) | COX Inhibitory Activity (IC50 µg/mL) |
|---|---|---|---|---|---|---|---|
| Gryllodes sigillatus | raw | 15.2 ± 1.83 g | 12.35 ± 0.07 j | 54.78 ± 1.12 c | 0.255 ± 0.07 bc | 141.81 ± 2.43 b | 16.34 ± 0.21 e |
| boiled | 12.6 ± 3.04 i | 11.85 ± 0.21 k | 27.23 ± 0.88 d | 0.298 ± 0.04 a | 187.44 ± 2.11 a | 18.71 ± 0.2 d | |
| baked | 10.7 ± 0.71 k | 10.9 ± 0.14 l | 17.8 ± 0.65 f | 0.286 ± 0.09 a | 103.15 ± 1.65 d | 12.64 ± 0.45 f | |
| protein | 15.24 ± 1.7 g | 17.97 ± 0.05 i | 63.42 ± 2.01 a | 0.228 ± 0.03 cde | 6.95 ± 0.1 j | 9.89 ± 0.12 h | |
| Tenebrio molitor | raw | 18.1 ± 1.13 e | 59.5 ± 1.98 e | 19.5 ± 0.89 e | 0.238 ± 0.01 cd | 94.68 ± 0.42 e | 9.88 ± 0.32 h |
| boiled | 19.7 ± 1.55 d | 18.4 ± 1.83 h | 11.8 ± 0.54 g | 0.202 ± 0.02 ef | 62.17 ± 0.54 g | 11.33 ± 0.45 g | |
| baked | 13.1 ± 1.41 h | 25.1 ± 1.55 g | 9.93 ± 0.1 h | 0.280 ± 0.02 ab | 38.4 ± 0.2 i | 11.46 ± 0.3 g | |
| protein | 24.31 ± 1.88 a | 85.55 ± 1.83 b | 12.32 ± 0.21 g | 0.203 ± 0.01 ef | 3.82 ± 0.12 k | 9.53 ± 0.15 i | |
| Schistocerca gregaria | raw | 21.5 ± 1.4 b | 81.4 ± 1.4 d | 57.97 ± 0.64 b | 0.204 ± 0.02 ef | 133.4 ± 0.96 c | 26.37 ± 0.45 a |
| boiled | 20.2 ± 1.6 c | 81.8 ± 1.7 c | 2.57 ± 0.05 i | 0.197 ± 0.07 f | 80.62 ± 1.67 f | 20.92 ± 0.2 c | |
| baked | 17.8 ± 1.1 f | 29.1 ± 1.4 f | 11.93 ± 0.11 g | 0.211 ± 0.04 def | 49.57 ± 1.12 h | 23.82 ± 0.45 b | |
| protein | 12.1 ± 0.01 j | 88.81 ± 3.71 a | 10.30 ± 0.2 h | 0.207 ± 0.01 ef | 3.13 ± 0.23 l | 5.05 ± 0.32 j |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zielińska, E.; Baraniak, B.; Karaś, M. Antioxidant and Anti-Inflammatory Activities of Hydrolysates and Peptide Fractions Obtained by Enzymatic Hydrolysis of Selected Heat-Treated Edible Insects. Nutrients 2017, 9, 970. https://doi.org/10.3390/nu9090970
Zielińska E, Baraniak B, Karaś M. Antioxidant and Anti-Inflammatory Activities of Hydrolysates and Peptide Fractions Obtained by Enzymatic Hydrolysis of Selected Heat-Treated Edible Insects. Nutrients. 2017; 9(9):970. https://doi.org/10.3390/nu9090970
Chicago/Turabian StyleZielińska, Ewelina, Barbara Baraniak, and Monika Karaś. 2017. "Antioxidant and Anti-Inflammatory Activities of Hydrolysates and Peptide Fractions Obtained by Enzymatic Hydrolysis of Selected Heat-Treated Edible Insects" Nutrients 9, no. 9: 970. https://doi.org/10.3390/nu9090970
APA StyleZielińska, E., Baraniak, B., & Karaś, M. (2017). Antioxidant and Anti-Inflammatory Activities of Hydrolysates and Peptide Fractions Obtained by Enzymatic Hydrolysis of Selected Heat-Treated Edible Insects. Nutrients, 9(9), 970. https://doi.org/10.3390/nu9090970






