Low-FODMAP Diet Improves Irritable Bowel Syndrome Symptoms: A Meta-Analysis
Abstract
1. Introduction
2. Materials and Methods
Statistical Analysis
3. Results
3.1. Low-FODMAP Diet Versus Traditional IBS Diet
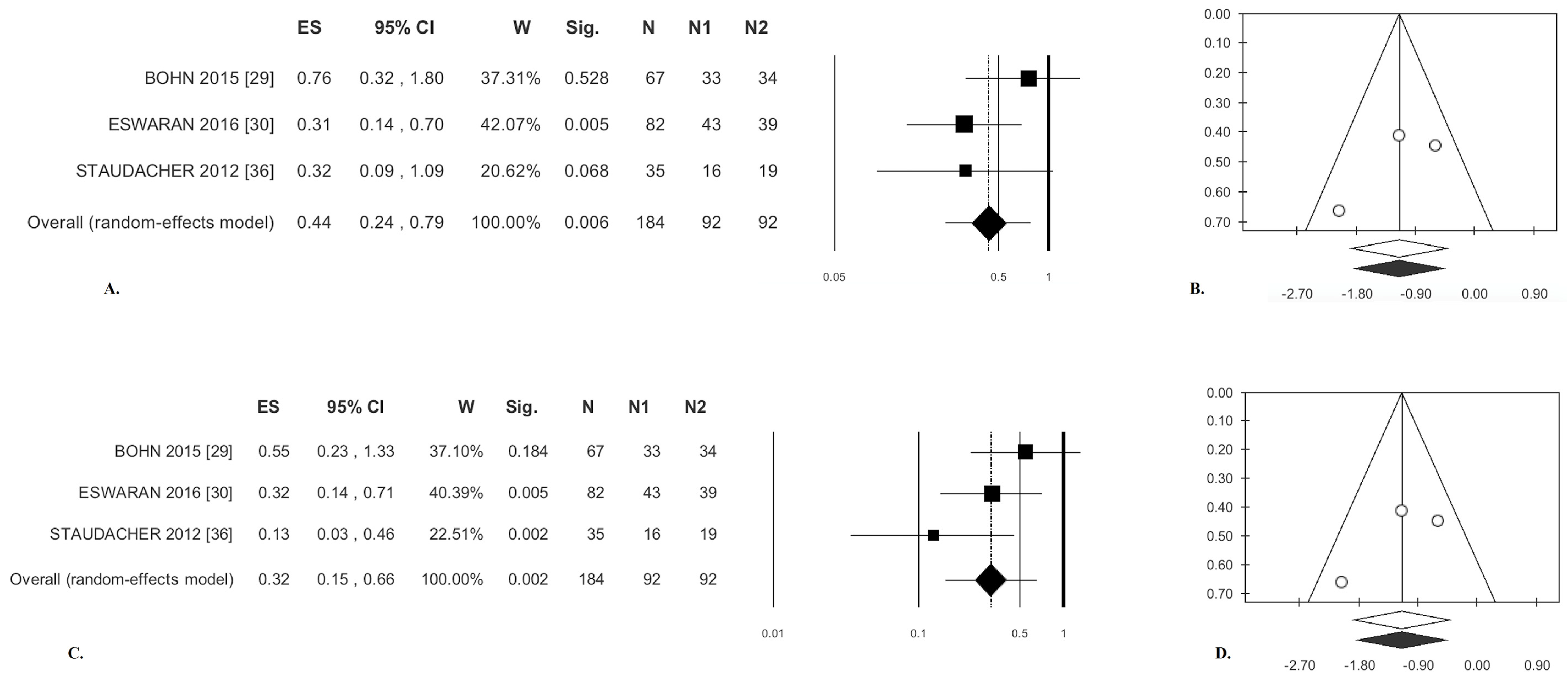
3.1.1. Abdominal Pain
3.1.2. Bloating
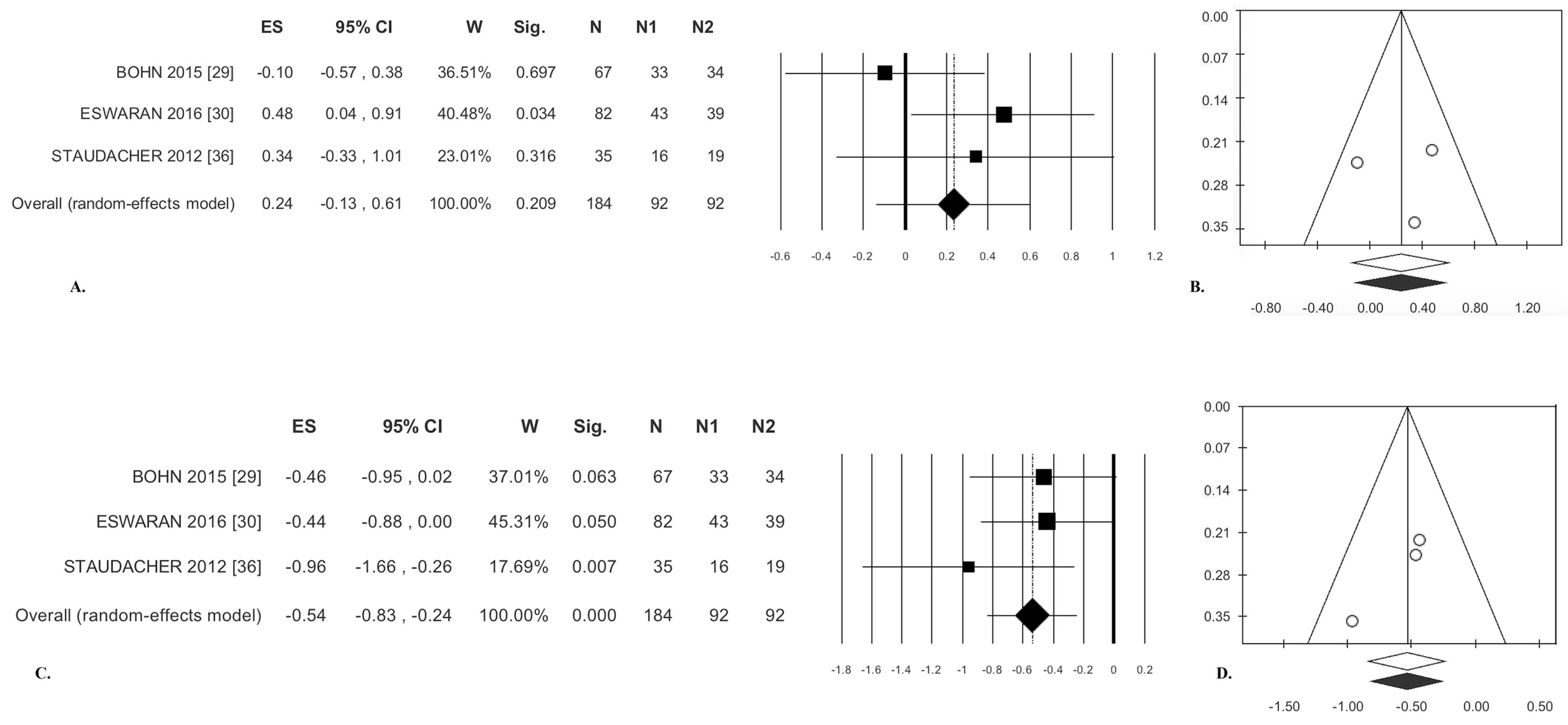
3.1.3. Stool Consistency
3.1.4. Stool Frequency
3.2. Low-FODMAP Diet vs. Medium/High-FODMAP Diet
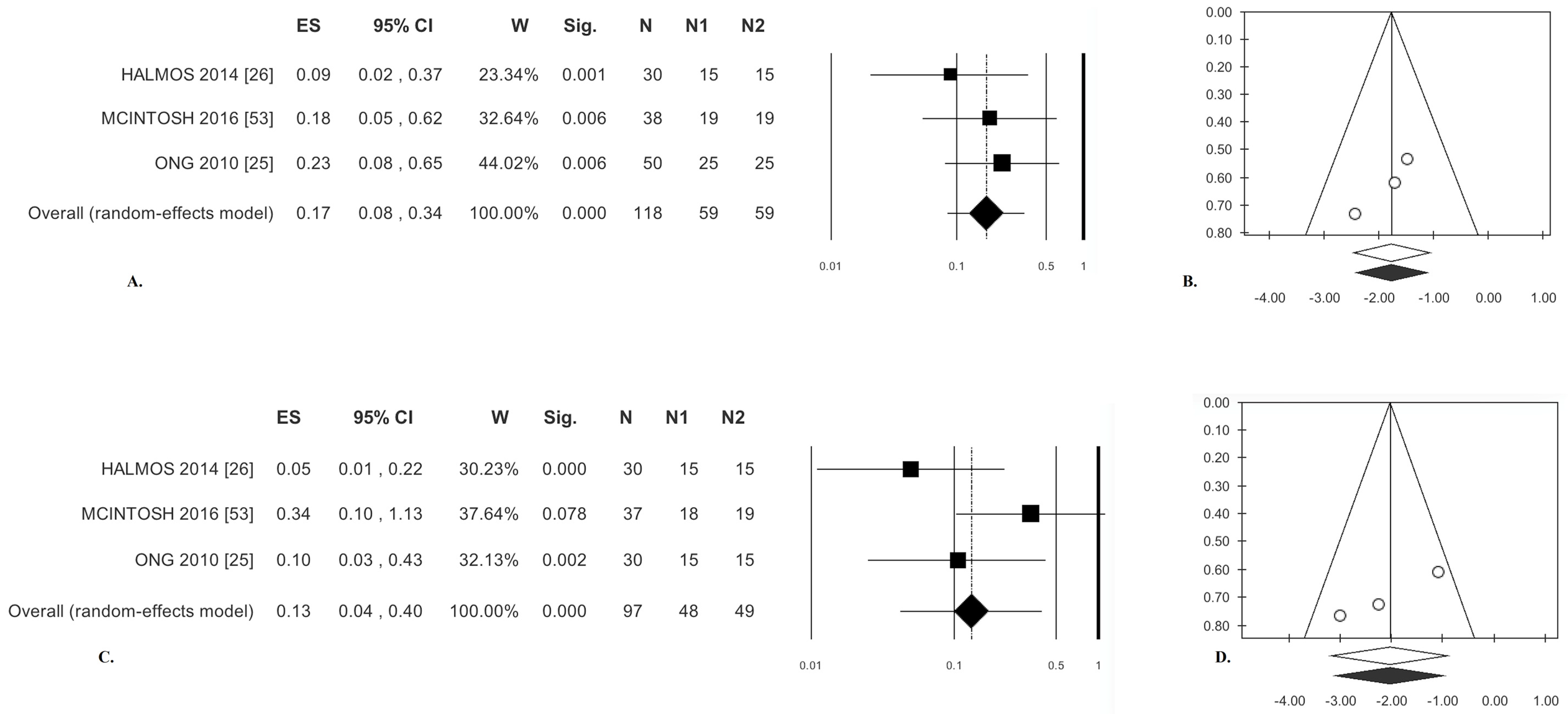
3.2.1. Abdominal Pain
3.2.2. Bloating
3.3. Cohort Studies
3.3.1. Abdominal Pain
3.3.2. Bloating
4. Discussion
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Altobelli, E.; Lattanzi, A.; Paduano, R.; Varrassi, G.; di Orio, F. Colorectal cancer prevention in Europe: Burden of disease and status of screening programs. Prev. Med. 2014, 62, 132–141. [Google Scholar] [CrossRef] [PubMed]
- Altobelli, E.; D’Aloisio, F.; Angeletti, P.M. Colorectal cancer screening in countries of European Council outside of the EU-28. World J. Gastroenterol. 2016, 22, 4946–4957. [Google Scholar] [CrossRef]
- Burisch, J.; Jess, T.; Martinato, M.; Lakatos, P.L. ECCO-EpiCom. The burden of inflammatory bowel disease in Europe. J. Crohn’s Colitis 2013, 7, 322–337. [Google Scholar] [CrossRef] [PubMed]
- American College of Gastroenterology Task Force on Irritable Bowel Syndrome; Brandt, L.J.; Chey, W.D.; Foxx-Orenstein, A.E.; Schiller, L.R.; Schoenfeld, P.S.; Spiegel, B.M.; Talley, N.J.; Quigley, E.M. An evidence-based position statement on the management of irritable bowel syndrome. Am. J. Gastroenterol. 2009, 104, S1–S35. [Google Scholar] [CrossRef] [PubMed]
- Andrews, E.B.; Eaton, S.C.; Hollis, K.A.; Hopkins, J.S.; Ameen, V.; Hamm, L.R.; Cook, S.F.; Tennis, P.; Mangel, A.W. Prevalence and demographics of irritable bowel syndrome: Results from a large web-based survey. Aliment. Pharmacol. Ther. 2005, 22, 935–942. [Google Scholar] [CrossRef] [PubMed]
- Müller-Lissner, S.A.; Bollani, S.; Brummer, R.J.; Coremans, G.; Dapoigny, M.; Marshall, J.K.; Muris, J.W.; Oberndorff-Klein, W.A.; Pace, F.; Rodrigo, L.; et al. Epidemiological aspects of irritable bowel syndrome in Europe and North America. Digestion 2001, 64, 200–204. [Google Scholar] [CrossRef] [PubMed]
- El-Salhy, M.; Gundersen, D.; Gilja, O.H.; Hatlebakk, J.G.; Hausken, T. Is irritable bowel syndrome an organic disorder? World J. Gastroenterol. 2014, 20, 384–400. [Google Scholar] [CrossRef] [PubMed]
- Horwitz, B.J.; Fisher, R.S. The irritable bowel syndrome. N. Engl. J. Med. 2001, 344, 1846–1850. [Google Scholar] [CrossRef] [PubMed]
- Chang, L. Review article: Epidemiology and quality of life in functional gastrointestinal disorders. Aliment. Pharmacol. Ther. 2004, 20, 31–39. [Google Scholar] [CrossRef] [PubMed]
- Maxion-Bergemann, S.; Thielecke, F.; Abel, F.; Bergemann, R. Costs of irritable bowel syndrome in the UK and US. Pharmacoeconomics 2006, 24, 21–37. [Google Scholar] [CrossRef] [PubMed]
- Chey, W.D.; Kurlander, J.; Eswaran, S. Irritable bowel syndrome: A clinical review. JAMA 2015, 313, 949–958. [Google Scholar] [CrossRef] [PubMed]
- Anastasi, J.K.; Capili, B.; Chang, M. Managing irritable bowel syndrome. Am. J. Nurs. 2013, 113, 42–52. [Google Scholar] [CrossRef] [PubMed]
- Mayer, E.A. Clinical practice. Irritable bowel syndrome. N. Engl. J. Med. 2008, 358, 1692–1699. [Google Scholar] [CrossRef] [PubMed]
- Mayer, E.A.; Labus, J.S.; Tillisch, K.; Cole, S.W.; Baldi, P. Towards a system view of IBS. Nat. Rev. Gastroenterol. Hepatol. 2015, 12, 592–605. [Google Scholar] [CrossRef] [PubMed]
- Rajilić-Stojanović, M.; Jonkers, D.M.; Salonen, A.; Hanevik, K.; Raes, J.; Jalanka, J.; de Vos, W.M.; Manichanh, C.; Golic, N.; Enck, P.; et al. Intestinal microbiota and diet in IBS: Causes, consequences, or epiphenomena? Am. J. Gastroenterol. 2015, 110, 278–287. [Google Scholar] [CrossRef] [PubMed]
- Böhn, L.; Störsrud, S.; Simrén, M. Nutrient intake in patients with irritable bowel syndrome compared with the general population. Neurogastroenterol. Motil. 2013, 25, 23–30. [Google Scholar] [CrossRef] [PubMed]
- Hayes, P.A.; Fraher, M.H.; Quigley, E.M. Irritable bowel syndrome: The role of food in pathogenesis and management. Gastroenterol. Hepatol. 2014, 10, 164–174. [Google Scholar]
- Eswaran, S.; Tack, J.; Chey, W.D. Food: The forgotten factor in the irritable bowel syndrome. Gastroenterol. Clin. N. Am. 2011, 40, 141–162. [Google Scholar] [CrossRef] [PubMed]
- Farré, R.; Tack, J. Food and symptom generation in functional gastrointestinal disorders: Physiological aspects. Am. J. Gastroenterol. 2013, 108, 698–706. [Google Scholar] [CrossRef] [PubMed]
- Moayyedi, P.; Ford, A.C.; Quigley, E.M.; Lembo, A.J.; Saito, Y.A.; Schiller, L.R.; Soffer, E.E.; Spiegel, B.M.; Quigley, E.M. Task Force on the Management of Functional Bowel Disorders. The American College of Gastroenterology irritable bowel syndrome monograph: Translating systematic review data to clinical practice. Gastroenterology 2010, 138, 789–791. [Google Scholar] [CrossRef] [PubMed]
- Dalrymple, J.; Bullock, I. Diagnosis and management of irritable bowel syndrome in adults in primary care: Summary of NICE guidance. BMJ 2008, 336, 556–568. [Google Scholar] [CrossRef] [PubMed]
- Tuck, C.J.; Muir, J.G.; Barrett, J.S.; Gibson, P.R. Fermentable oligosaccharides, disaccharides, monosaccharides and polyols: Role in irritable bowel syndrome. Expert Rev. Gastroenterol. Hepatol. 2014, 8, 819–834. [Google Scholar] [CrossRef] [PubMed]
- Gibson, P.R.; Varney, J.; Malakar, S.; Muir, J.G. Food components and irritable bowel syndrome. Gastroenterology 2015, 148, 1158–1574. [Google Scholar] [CrossRef] [PubMed]
- Barrett, J.S.; Gearry, R.B.; Muir, J.G.; Irving, P.M.; Rose, R.; Rosella, O.; Haines, M.L.; Shepherd, S.J.; Gibson, P.R. Dietary poorly absorbed, short-chain carbohydrates increase delivery of water and fermentable substrates to the proximal colon. Aliment. Pharmacol. Ther. 2010, 31, 874–882. [Google Scholar] [CrossRef] [PubMed]
- Ong, D.K.; Mitchell, S.B.; Barrett, J.S.; Shepherd, S.J.; Irving, P.M.; Biesiekierski, J.R.; Smith, S.; Gibson, P.R.; Muir, J.G. Manipulation of dietary short chain carbohydrates alters the pattern of gas production and genesis of symptoms in irritable bowel syndrome. J. Gastroenterol. Hepatol. 2010, 25, 1366–1373. [Google Scholar] [CrossRef] [PubMed]
- Halmos, E.P.; Power, V.A.; Shepherd, S.J.; Gibson, P.R.; Muir, J.G. A diet low in FODMAPs reduces symptoms of irritable bowel syndrome. Gastroenterology 2014, 146, 67–75. [Google Scholar] [CrossRef] [PubMed]
- Shepherd, S.J.; Lomer, M.C.; Gibson, P.R. Short-chain carbohydrates and functional gastrointestinal disorders. Am. J. Gastroenterol. 2013, 108, 707–717. [Google Scholar] [CrossRef] [PubMed]
- Camilleri, M.; Boeckxstaens, G. Dietary and pharmacological treatment of abdominal pain in IBS. Gut 2017, 66, 966–974. [Google Scholar] [CrossRef] [PubMed]
- Böhn, L.; Störsrud, S.; Liljebo, T.; Collin, L.; Lindfors, P.; Törnblom, H.; Simrén, M. Diet low in FODMAPs reduces symptoms of irritable bowel syndrome as well as traditional dietary advice: A randomized controlled trial. Gastroenterology 2015, 149, 1399–1407. [Google Scholar] [CrossRef] [PubMed]
- Eswaran, S.L.; Chey, W.D.; Han-Markey, T.; Ball, S.; Jackson, K. A Randomized Controlled Trial Comparing the Low FODMAP Diet vs. Modified NICE Guidelines in US Adults with IBS-D. Am J Gastroenterol. 2016, 111, 1824–1832. [Google Scholar] [CrossRef] [PubMed]
- Laatikainen, R.; Koskenpato, J.; Hongisto, S.M.; Loponen, J.; Poussa, T.; Hillilä, M.; Korpela, R. Randomised clinical trial: Low-FODMAP rye bread vs. regular rye bread to relieve the symptoms of irritable bowel syndrome. Aliment. Pharmacol. Ther. 2016, 44, 460–470. [Google Scholar] [CrossRef] [PubMed]
- Gibson, P.R. The evidence base for efficacy of the low FODMAP diet in irritable bowel syndrome: Is it ready for prime time as a first-line therapy? J. Gastroenterol. Hepatol. 2017, 32, 32–35. [Google Scholar] [CrossRef] [PubMed]
- Gibson, P.R.; Shepherd, S.J. Evidence-based dietary management of functional gastrointestinal symptoms: The FODMAP approach. J. Gastroenterol. Hepatol. 2010, 25, 252–258. [Google Scholar] [CrossRef] [PubMed]
- Valeur, J.; Røseth, A.G.; Knudsen, T.; Malmstrøm, G.H.; Fiennes, J.T.; Midtvedt, T.; Berstad, A. Fecal Fermentation in Irritable Bowel Syndrome: Influence of Dietary Restriction of Fermentable Oligosaccharides, Disaccharides, Monosaccharides and Polyols. Digestion 2016, 94, 50–56. [Google Scholar] [CrossRef] [PubMed]
- Hustoft, T.N.; Hausken, T.; Ystad, S.O.; Valeur, J.; Brokstad, K.; Hatlebakk, J.G.; Lied, G.A. Effects of varying dietary content of fermentable short-chain carbohydrates on symptoms, fecal microenvironment, and cytokine profiles in patients with irritable bowel syndrome. Neurogastroenterol. Motil. 2017. [Google Scholar] [CrossRef] [PubMed]
- Staudacher, H.M.; Lomer, M.C.; Anderson, J.L.; Barrett, J.S.; Muir, J.G.; Irving, P.M.; Whelan, K. Fermentable carbohydrate restriction reduces luminal bifidobacteria and gastrointestinal symptoms in patients with irritable bowel syndrome. J. Nutr. 2012, 142, 1510–1518. [Google Scholar] [CrossRef] [PubMed]
- Staudacher, H.M.; Irving, P.M.; Lomer, M.C.; Whelan, K. Mechanisms and efficacy of dietary FODMAP restriction in IBS. Nat. Rev. Gastroenterol. Hepatol. 2014, 11, 256–266. [Google Scholar] [CrossRef] [PubMed]
- Staudacher, H.M.; Whelan, K. Altered gastrointestinal microbiota in irritable bowel syndrome and its modification by diet: Probiotics, prebiotics and the low FODMAP diet. Proc. Nutr. Soc. 2016, 75, 306–318. [Google Scholar] [CrossRef] [PubMed]
- Chumpitazi, B.P.; Cope, J.L.; Hollister, E.B.; Tsai, C.M.; McMeans, A.R.; Luna, R.A.; Versalovic, J.; Shulman, R.J. Randomised clinical trial: Gut microbiome biomarkers are associated with clinical response to a low FODMAP diet in children with the irritable bowel syndrome. Aliment. Pharmacol. Ther. 2015, 42, 418–427. [Google Scholar] [CrossRef] [PubMed]
- Marsh, A.; Eslick, E.M.; Eslick, G.D. Does a diet low in FODMAPs reduce symptoms associated with functional gastrointestinal disorders? A comprehensive systematic review and meta-analysis. Eur. J. Nutr. 2016, 55, 897–906. [Google Scholar] [CrossRef] [PubMed]
- Moher, D.; Altman, D.G.; Liberati, A.; Tetzlaff, J. PRISMA statement. Epidemiology 2011, 22, 128. [Google Scholar] [CrossRef] [PubMed]
- Higgins, J.P.T.; Green, S.; Altman, D.G. Assessing Risk of Bias in Included Studies Published Online. In Cochrane Handbook for Systematic Reviews of Interventions: Cochrane Book Series Chapter 8; WILEY: Hoboken, NJ, USA, 2008. [Google Scholar]
- Wells, G.A.; Shea, B.; O’Connell, D.; Peterson, J.; Welch, V.; Losos, M.; Tugwell, P. The Newcastle-Ottawa Scale (NOS) for Assessing the Quality of Nonrandomised Studies in Meta-Analyses. 2001. Available online: http://www.ohri.ca/programs/clinical_epidemiology/oxford.asp (accessed on 31 March 2017).
- Rothstein, H.R.; Sutton, A.J.; Borenstein, M. Publication Bias in Meta-Analysis; Wiley: Chichester, UK, 2005. [Google Scholar]
- Fragkos, K.C.; Tsagris, M.; Frangos, C.C. Publication bias in meta-analysis: Confidence intervals for Rosenthal’s fail-safe number. Int. Sch. Res. Not. 2014, 2014, 825383. [Google Scholar] [CrossRef] [PubMed]
- Pedersen, N.; Andersen, N.N.; Végh, Z.; Jensen, L.; Ankersen, D.V.; Felding, M.; Simonsen, M.H.; Burisch, J.; Munkholm, P. Ehealth: Low FODMAP diet vs. Lactobacillus rhamnosus GG in irritable bowel syndrome. World J. Gastroenterol. 2014, 20, 16215–16226. [Google Scholar] [CrossRef] [PubMed]
- Peters, S.L.; Yao, C.K.; Philpott, H.; Yelland, G.W.; Muir, J.G.; Gibson, P.R. Randomized clinical trial: The efficacy of gut-directed hypnotherapy is similar to that of the low FODMAP diet for the treatment of irritable bowel syndrome. Aliment. Pharmacol. Ther. 2016, 44, 447–459. [Google Scholar] [CrossRef] [PubMed]
- Major, G.; Pritchard, S.; Murray, K.; Yelland, G.W.; Muir, J.G.; Gibson, P.R. Colon Hypersensitivity to Distension, Rather than Excessive Gas Production, Produces Carbohydrate-Related Symptoms in Individuals with Irritable Bowel Syndrome. Gastroenterology 2017, 152, 124–133. [Google Scholar] [CrossRef] [PubMed]
- Biesiekierski, J.R.; Peters, S.L.; Newnham, E.D.; Tsai, C.M.; McMeans, A.R.; Luna, R.A.; Versalovic, J.; Shulman, R.J. No effects of gluten in patients with self-reported non-celiac gluten sensitivity after dietary reduction of fermentable, poorly absorbed, short-chain carbohydrates. Gastroenterology 2013, 145, 320–328. [Google Scholar] [CrossRef] [PubMed]
- Whigham, L.; Joyce, T.; Harper, G.; Irving, P.M.; Staudacher, H.M.; Whelan, K.; Lomer, M.C. Clinical effectiveness and economic costs of group versus one-to-one education for short-chain fermentable carbohydrate restriction (low FODMAP diet) in the management of irritable bowel syndrome. J. Hum. Nutr. Diet. 2015, 28, 687–696. [Google Scholar] [CrossRef] [PubMed]
- Maagaard, L.; Ankersen, D.V.; Végh, Z.; Burisch, J.; Jensen, L.; Pedersen, N.; Munkholm, P. Follow-up of patients with functional bowel symptoms treated with a low FODMAP diet. World J. Gastroenterol. 2016, 22, 4009–4019. [Google Scholar] [CrossRef] [PubMed]
- Pedersen, N. EHealth: Self-management in inflammatory bowel disease and in irritable bowel syndrome using novel constant-care web applications. EHealth by constant-care in IBD and IBS. Dan. Med. J. 2015, 62, B5168. [Google Scholar] [CrossRef] [PubMed]
- McIntosh, K.; Reed, D.E.; Schneider, T.; Dang, F.; Keshteli, A.H.; De Palma, G.; Madsen, K.; Bercik, P.; Vanner, S. FODMAPs alter symptoms and the metabolome of patients with IBS: A randomised controlled trial. Gut 2016, 66, 1241–1251. [Google Scholar] [CrossRef] [PubMed]
- De Roest, R.H.; Dobbs, B.R.; Chapman, B.A.; Batman, B.; O’Brien, L.A.; Leeper, J.A.; Hebblethwaite, C.R.; Gearry, R.B. The low FODMAP diet improves gastrointestinal symptoms in patients with irritable bowel syndrome: A prospective study. Int. J. Clin. Pract. 2013, 67, 895–903. [Google Scholar] [CrossRef] [PubMed]
- Huamán, J.W.; Felip, A.; Guedea, E.; Jansana, M.; Videla, S.; Saperas, E. The diet low in fermentable carbohydrates short chain and polyols improves symptoms in patients with functional gastrointestinal disorders in Spain. Gastroenterol. Hepatol. 2015, 38, 113–122. [Google Scholar] [CrossRef] [PubMed]
- López, N.P.Y.; Torres-López, E.; Zamarripa-Dorsey, F. Clinical response in Mexican patients with irritable bowel syndrome treated with a low diet low in fermentable carbohydrates (FODMAP). Rev. Gastroenterol. Mex. 2015, 80, 180–185. [Google Scholar]
- Mazzawi, T.; Hausken, T.; Gundersen, D.; El-Salhy, M. Dietary guidance normalizes large intestinal endocrine cell densities in patients with irritable bowel syndrome. Eur. J. Clin. Nutr. 2016, 70, 175–181. [Google Scholar] [CrossRef] [PubMed]
- Staudacher, H.M.; Whelan, K.; Irving, P.M.; Lomer, M.C. Comparison of symptom response following advice for a diet low in fermentable carbohydrates (FODMAPs) versus standard dietary advice in patients with irritable bowel syndrome. J. Hum. Nutr. Diet. 2011, 24, 487–495. [Google Scholar] [CrossRef] [PubMed]
- Gibson, P.R. Use of the low-FODMAP diet in inflammatory bowel disease. J. Gastroenterol. Hepatol. 2017, 32, 40–42. [Google Scholar] [CrossRef] [PubMed]
- Nanayakkara, W.S.; Skidmore, P.M.; O’Brien, L.; Wilkinson, T.J.; Gearry, R.B. Efficacy of the low FODMAP diet for treating irritable bowel syndrome: The evidence to date. Clin. Exp. Gastroenterol. 2016, 9, 131–142. [Google Scholar] [PubMed]
- Liberati, A.; Altman, D.G.; Tetzlaff, J.; Mulrow, C.; Gøtzsche, P.C.; Ioannidis, J.P.; Clarke, M.; Devereaux, P.J.; Kleijnen, J.; Moher, D. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: Explanation and elaboration. J. Clin. Epidemiol. 2009, 62, e1–e34. [Google Scholar] [CrossRef] [PubMed]
- O’Keeffe, M.; Jansen, C.; Martin, L.; Williams, M.; Seamark, L.; Staudacher, H.M.; Irving, P.M.; Whelan, K.; Lomer, M.C. Long-term impact of the low-FODMAP diet on gastrointestinal symptoms, dietary intake, patient acceptability, and healthcare utilization in irritable bowel syndrome. Neurogastroenterol. Motil. 2017. [Google Scholar] [CrossRef] [PubMed]
- Irritable Bowel Syndrome in Adults: Diagnosis and Management. Available online: https://www.nice.org.uk/guidance/cg61 (accessed on 1 July 2017).
- Staudacher, H.M.; Lomer, M.C.E.; Farquharson, F.M.; Louis, P.; Fava, F.; Franciosi, E.; Scholz, M.; Tuohy, K.M.; Lindsay, J.O.; Irving, P.M.; et al. Diet low in FODMAPs reduces symptoms in patients with irritable bowel syndrome and probiotic restores bifidobacterium species: A randomized controlled trial. Gastroenterology 2017. [Google Scholar] [CrossRef] [PubMed]
- Catassi, G.; Lionetti, E.; Gatti, S.; Catassi, C. The low FODMAP diet: Many question marks for a catchy acronym. Nutrients 2017, 9, E292. [Google Scholar] [CrossRef] [PubMed]
- Vincenzi, M.; Del Ciondolo, I.; Pasquini, E.; Gennai, K.; Paolini, B. Effects of a low FODMAP diet and specific carbohydrate diet on symptoms and nutritional adequacy of patients with irritable bowel syndrome: Preliminary results of a single-blinded randomized trial. J. Transl. Int. Med. 2017, 30, 120–126. [Google Scholar] [CrossRef] [PubMed]
| Patient Population | Year (Mean ± SD or Median) | % Female | Symptoms and Stool Characteristics | |||||||||||||
| Study, Year, Country | Duration of Follow-up | Assessed for Eligibility | Randomised | Intervention/Control | Drop Outs | Low- FODMAP group | Traditional IBS group | p | Low- FODMAP group | Traditional IBS group | p | Abdom Pain * | Bloating | Stool Consistency | Stool Frequency | |
| Study group | Control group | |||||||||||||||
| RCT Low-FODMAP vs. Traditional IBS Diets | ||||||||||||||||
| Eswaran, 2016, USA [30] | 4 weeks | 171 | 92 | 50/42 | Study group n = 5 | Control group n = 3 | 41.6 ± 41.7 Low- FODMAP group | 43.8 ± 15.2 Traditional IBS group | (p = 0.49) | 66.0 Low- FODMAP group | 76.2 Traditional IBS group | (p = 0.35) | X | X | X | X |
| Böhn, 2015, Sweden [29] | 4 weeks | 84 | 75 | 38/37 | Study group n = 5 | Control group n = 3 | 44.0 Low- FODMAP group | 41.0 Traditional IBS group | (p = 0.35) | 79.0 Low- FODMAP group | 84.0 Traditional IBS group | (p = 0.59) | X | X | X | X |
| Staudacher, 2012, UK [36] | 4 weeks | 99 | 41 | 19/22 | Study group n = 3 | Control group n = 3 | 35.2 Low- FODMAP group | 35.0 Traditional IBS group | (p = 0.94) | 63.0 Low- FODMAP group | 68.0 Traditional IBS group | (p = 0.74) | X | X | X | X |
| RCT low-FODMAP vs. Medium/High FODMAP Diets | ||||||||||||||||
| McIntosh, 2016, Canada [53] | 3 weeks | 37 | 40 | 20/20 | Study group n = 2 | Control group n = 1 | 50.2 Low- FODMAP group | 51.4 High- FODMAP | (p = NS) ** | 83.3 Low- FODMAP group | 89.4 High- FODMAP | (p = NS) * | X | X | - | - |
| Halmos, 2014, Australia [26] | 3 weeks | 45 | 30 | 15/15 | IBS group n = 7 | Healthy subject group n = 8 | 41.0 IBS group | 31.0 Healthy subject group | (p = NS) ** | 70.0 IBS group | 75.0 Healthy subject group | (p = NS) ** | X | X | - | - |
| Ong, 2010, Australia [25] | 11 days | 15 | 15 | 15/15 | Not Reported | 50.2 Low- FODMAP group | 51.4 High- FODMAP | (p = NS) ** | 83.3 Low- FODMAP group | 89.4 High- FODMAP | (p = NS) ** | X | X | - | - | |
| Study, Year, Country | Duration of Follow-Up | Assessed for Eligibility | Completed Study (No. of Patients) | Lost at Follow-Up | Years (Mean) | % Female | Symptoms and Stool Characteristics | |||
|---|---|---|---|---|---|---|---|---|---|---|
| Abdominal Pain | Bloating | Stool Consistency | Stool Frequency | |||||||
| Valeur, 2016, Norway [34] | 4 weeks | 97 | 63 | 34 | 38.4 | 88.9 | X | X | - | - |
| De Roest, 2013, New Zeland [54] | 15 months | 192 | 90 | 102 | 47.0 | 84.4 | X | X | - | - |
| Huaman, 2015, Spain [55] | 2 months | 30 | 24 | 6 | 40.0 | 79.0 | X | X | - | - |
| Pérez y López, 2015, Mexico [56] | 3 weeks | Not reported | 31 | 0 | 46.4 | 87.0 | X | X | - | - |
| Mazzawi, 2013, Norway [57] | 3–9 months | Not reported | 46 | 0 | 35.0 | 76.0 | X | - | - | - |
| Staudacher, 2011, UK [58] | 9 months | Not reported | 43 | 0 | 37.8 | 65.0 | X | X | - | - |
| Pooled Analysis | Heterogeneity | Publication Bias | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Outcome | Effect Size | CI | p Value | Q | I2 | p Value | T2 | T | Egger’s | Begg and Mazdumdar’s | Fail-Safe | Rosenthal | ||
| T | p Value | Z | p Value | No. | No. | |||||||||
| RCTs Low-FODMAP vs. Traditional IBS Diet (k = 3) [29,30,36] | ||||||||||||||
| Abdominal Pain | 0.44 (OR) | (0.26; 0.79) | 0.006 | 2.43 | 17.81 | 0.296 | 0.05 | 0.23 | −0.19 | 0.877 | 0.52 | 0.602 | 4 | 25 |
| Bloating | 0.32 (OR) | (0.15; 0.66) | <0.0001 | 1.97 | 0.00 | 0.374 | 0.00 | 0.00 | −1.21 | 0.439 | −0.52 | 0.602 | 11 | 25 |
| Stool Consistency | 0.24 * | (−0.13; 0.61) | 0.209 | 3.07 | 34.84 | 0.216 | 0.04 | 0.19 | −0.02 | 0.989 | −0.52 | 0.602 | 0 | 25 |
| Stool Frequency | −0.54 * | (−0.83; −0.24) | <0.0001 | 1.67 | 0.00 | 0.434 | 0.00 | 0.00 | −5.74 | 0.110 | −1.57 | 0.117 | 8 | 25 |
| RCTs Low-FODMAP vs. Medium/High FODMAP (k = 3) [25,26,53] | ||||||||||||||
| Abdominal Pain | 0.17 (OR) | (0.08; 0.34) | <0.0001 | 1.14 | 0.00 | 0.567 | 0.00 | 0.00 | −4.69 | 0.150 | −1.54 | 0.018 | 17 | 25 |
| Bloating | 0.13 (OR) | (0.04; 0.40) | <0.0001 | 4.11 | 51.37 | 0.128 | 0.51 | 0.72 | −8.89 | 0.071 | −0.57 | 0.017 | 66 | 40 |
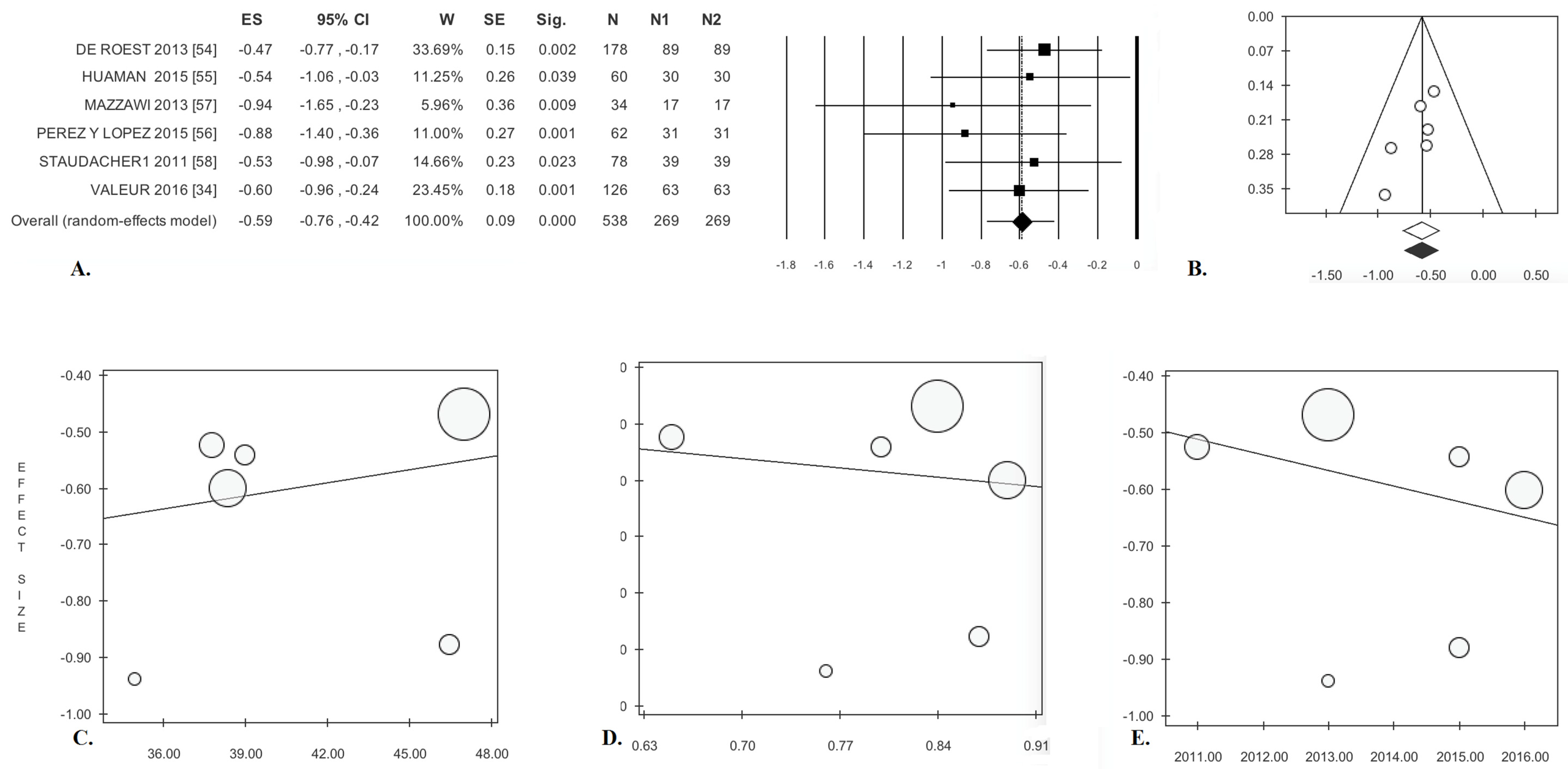
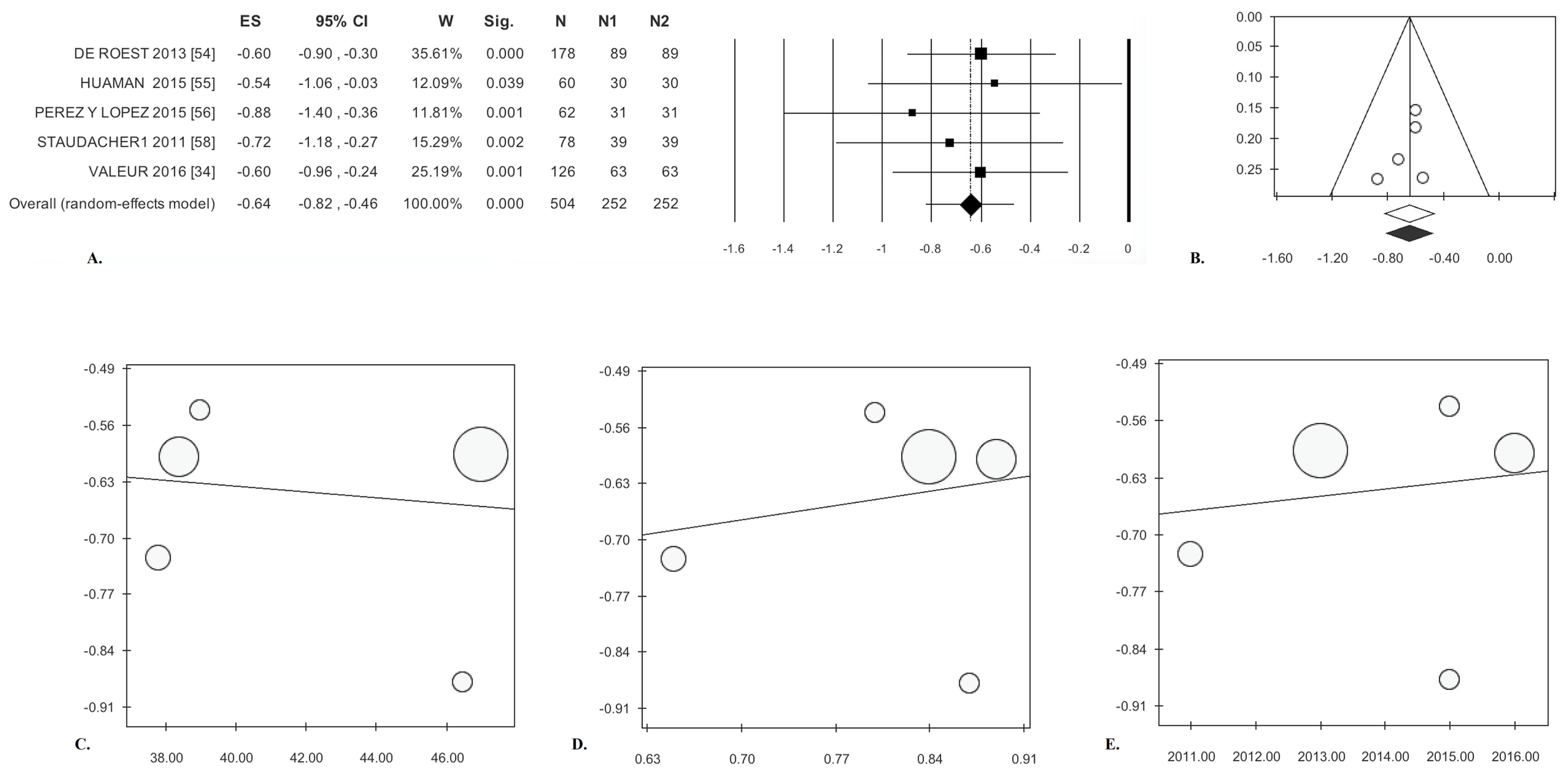
| Cohort Studies (k = 6) [34,54,55,56,57,58] | ||||||||||||||
| Abdominal Pain | −0.59 * | (−0.76; −0.42) | <0.0001 | 2.85 | 0.00 | 0.723 | 0.00 | 0.00 | −2.45 | 0.070 | −1.69 | 0.091 | 66 | 40 |
| Bloating | −0.64 * | (0.82; −0.46) | <0.0001 | 1.20 | 0.00 | 0.878 | 0.00 | 0.00 | −1.13 | 0.342 | −0.98 | 0.327 | 59 | 40 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Altobelli, E.; Del Negro, V.; Angeletti, P.M.; Latella, G. Low-FODMAP Diet Improves Irritable Bowel Syndrome Symptoms: A Meta-Analysis. Nutrients 2017, 9, 940. https://doi.org/10.3390/nu9090940
Altobelli E, Del Negro V, Angeletti PM, Latella G. Low-FODMAP Diet Improves Irritable Bowel Syndrome Symptoms: A Meta-Analysis. Nutrients. 2017; 9(9):940. https://doi.org/10.3390/nu9090940
Chicago/Turabian StyleAltobelli, Emma, Valerio Del Negro, Paolo Matteo Angeletti, and Giovanni Latella. 2017. "Low-FODMAP Diet Improves Irritable Bowel Syndrome Symptoms: A Meta-Analysis" Nutrients 9, no. 9: 940. https://doi.org/10.3390/nu9090940
APA StyleAltobelli, E., Del Negro, V., Angeletti, P. M., & Latella, G. (2017). Low-FODMAP Diet Improves Irritable Bowel Syndrome Symptoms: A Meta-Analysis. Nutrients, 9(9), 940. https://doi.org/10.3390/nu9090940






