Changes in Bioavailability of Omega-3 (DHA) through Alpha-Tocopheryl Phosphate Mixture (TPM) after Oral Administration in Rats
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Animals
3. Methods
3.1. Study Design
3.2. Administration of Formulations and Sample Collection
3.3. Calculation of Pharmacokinetics Parameters
4. Results
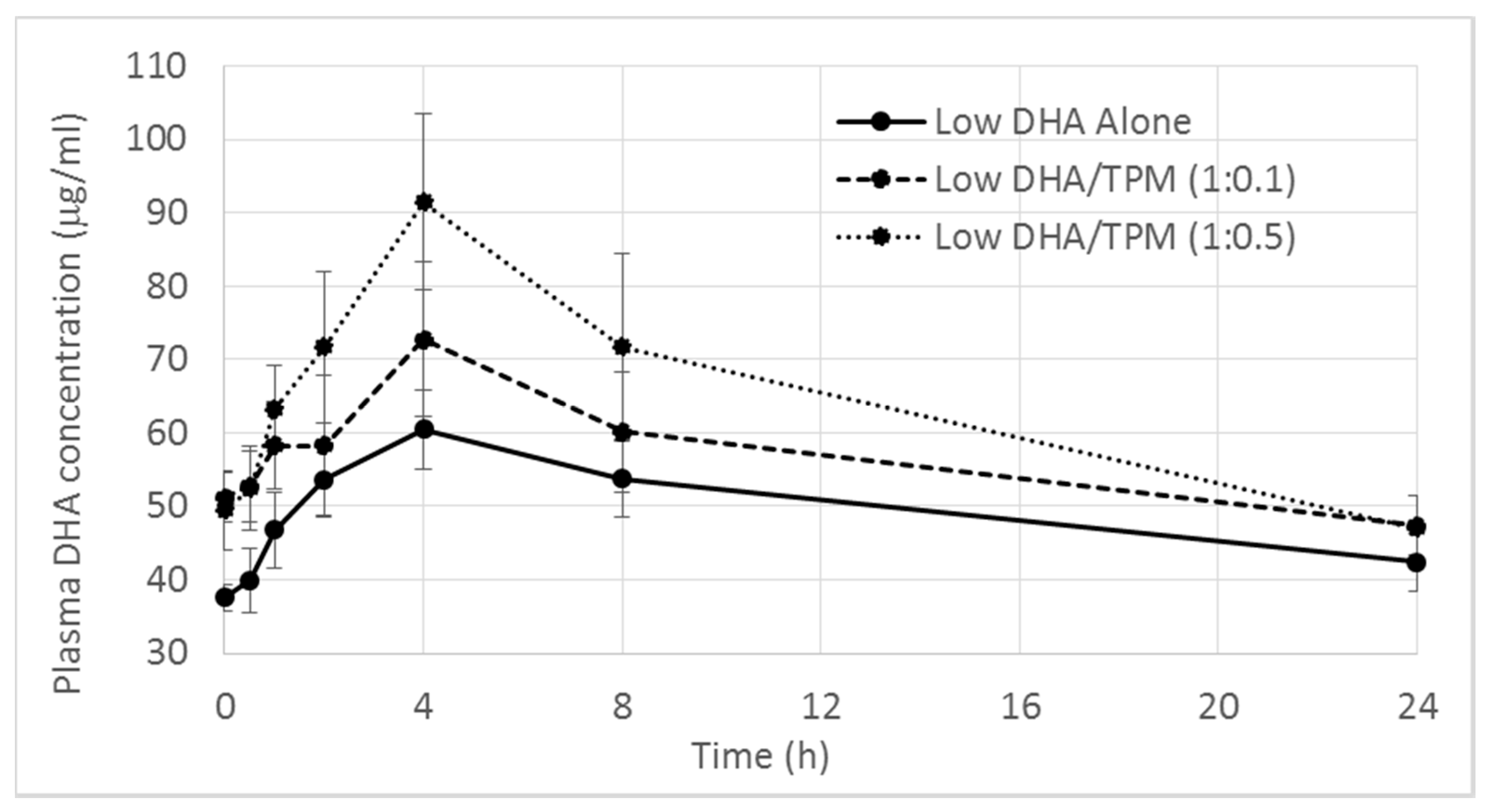
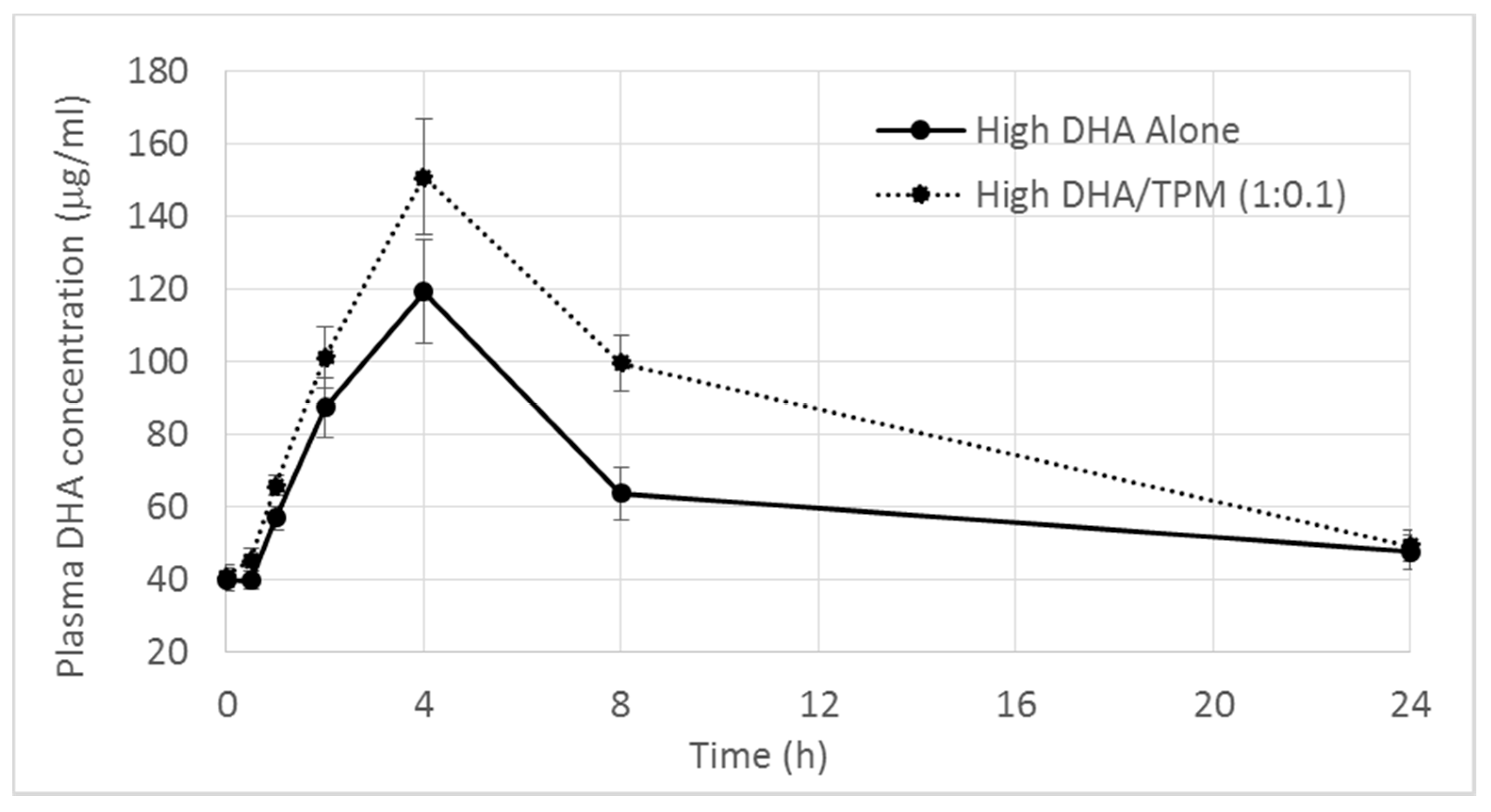
DHA Levels
5. Discussion
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Authority Nutrition. Available online: https://authoritynutrition.com/omega-3-6-9-overview (accessed on 2 May 2017).
- Greenberg, J.A.; Bell, S.J.; Ausdal, W.V. Omega-3 Fatty Acid supplementation during pregnancy. Rev. Obstet. Gynecol. 2008, 1, 162–169. [Google Scholar] [PubMed]
- National Institutes of Health (NIH). Available online: https://ods.od.nih.gov/factsheets/Omega3FattyAcids-HealthProfessional (accessed on 7 August 2017).
- Naini, A.E.; Keyvandarian, N.; Mortazavi, M.; Taheri, S.; Hosseini, S.M. Effect of Omega-3 fatty acids on blood pressure and serum lipids in continuous ambulatory peritoneal dialysis patients. J. Res. Pharm. Pract. 2015, 4, 135–141. [Google Scholar] [PubMed]
- Denman, M. In patients with early rheumatoid arthritis, fish oil reduced failure of treatment with DMARDs. Ann. Intern. Med. 2014, 160, JC11. [Google Scholar] [CrossRef] [PubMed]
- Swanson, D.; Block, R.; Mousa, S.A. Omega-3 fatty acids EPA and DHA: Health benefits throughout life. Adv. Nutr. 2012, 3, 1–7. [Google Scholar] [CrossRef] [PubMed]
- McNamara, R.K.; Strawn, J.R. Role of Long-Chain Omega-3 Fatty Acids in Psychiatric Practice. PharmaNutrition 2013, 1, 41–49. [Google Scholar] [CrossRef] [PubMed]
- Walker, R.; Decker, E.A.; McClements, D.J. Development of food-grade nanoemulsions and emulsions for delivery of omega-3 fatty acids: Opportunities and obstacles in the food industry. Food Funct. 2015, 6, 42–55. [Google Scholar] [CrossRef] [PubMed]
- Raper, N.R.; Cronin, F.J.; Exler, J. Omega-3 fatty acid content of the US food supply. J. Am. Coll. Nutr. 1992, 11, 304. [Google Scholar] [CrossRef] [PubMed]
- Van Vliet, T.; Katan, M.B. Lower ratio of n-3 to n-6 fatty acids in cultured than in wild fish. Am. J. Clin. Nutr. 1990, 51, 1–2. [Google Scholar] [PubMed]
- Simopoulos, A.P.; Salem, N., Jr. Egg yolk as a source of long-chain polyunsaturated fatty acids in infant feeding. Am. J. Clin. Nutr. 1992, 55, 411–414. [Google Scholar] [PubMed]
- Daley, C.A.; Abbott, A.; Doyle, P.S.; Nader, G.A.; Larson, S. A review of fatty acid profiles and antioxidant content in grass-fed and grain-fed beef. Nutr. J. 2010, 9, 10. [Google Scholar] [CrossRef] [PubMed]
- Simopoulos, A.P. An Increase in the Omega-6/Omega-3 Fatty Acid Ratio Increases the Risk for Obesity. Nutrients 2016, 8, 128. [Google Scholar] [CrossRef] [PubMed]
- Porter, C.J.; Trevaskis, N.L.; Charman, W.N. Lipids and lipid-based formulations: Optimizing the oral delivery of lipophilic drugs. Nat. Rev. Drug Discov. 2007, 6, 231–248. [Google Scholar] [CrossRef] [PubMed]
- Pouton, C.M. Formulation of poorly water-soluble drugs for oral administration: Physicochemical and physiological issues and the lipid formulation classification system. Eur. J. Pharm. Sci. 2006, 29, 278–287. [Google Scholar] [CrossRef] [PubMed]
- Humberstone, A.J.; Charman, W.N. Lipid-based vehicles for the oral delivery of poorly water soluble drugs. Adv. Drug Deliv. Rev. 1997, 25, 103–128. [Google Scholar] [CrossRef]
- Kossena, G.A.; Boyd, B.J.; Porter, C.J.; Charman, W.N. Separation and characterization of the colloidal phases produced on digestion of common formulation lipids and assessment of their impact on the apparent solubility of selected poorly water-soluble drugs. J. Pharm. Sci. 2003, 92, 634–648. [Google Scholar] [CrossRef] [PubMed]
- Porter, C.J.; Kaukonen, A.M.; Taillardat-Bertschinger, A.; Boyd, B.J.; O’Connor, J.M.; Edwards, G.A.; Charman, W.N. Use of in vitro lipid digestion data to explain the in vivo performance of triglyceride-based oral lipid formulations of poorly water-soluble drugs: Studies with halofantrine. J. Pharm. Sci. 2004, 93, 1110–1121. [Google Scholar] [CrossRef] [PubMed]
- Holub, B. Function Follows Form. Nutr. Outlook 2011, 14, 34–40. [Google Scholar]
- Garaiova, I.; Guschina, I.A.; Plummer, S.F.; Tang, J.; Wang, D.; Plummer, N.T. A randomized cross-over trial in healthy adults indicating improved absorption of omega-3 fatty acids by pre-emulsification. Nutr. J. 2007, 6, 4. [Google Scholar] [CrossRef] [PubMed]
- Raatz, S.K.; Redmon, J.B.; Wimmergren, N.; Donadio, J.V.; Bibus, D.M. Enhanced absorption of n-3 fatty acids from emulsified compared with encapsulated fish oil. J. Am. Diet. Assoc. 2009, 109, 1076–1081. [Google Scholar] [CrossRef] [PubMed]
- Horn, A.F.; Nielsen, N.K.; Andersen, U.; Sogaard, L.H.; Horsewell, A.; Jacobsen, C. Oxidative stability of 70% fish oil-in-water emulsions: Impact of emulsifiers and pH. Eur. J. Lipid Sci. Technol. 2011, 113, 1243–1257. [Google Scholar] [CrossRef]
- Gianello, R.; Hall, W.C.; Kennepohl, E.; Libinaki, R.; Ogru, E. Subchronic oral toxicity study of mixed tocopheryl phosphates in rats. Int. J. Toxicol. 2007, 26, 475–490. [Google Scholar] [CrossRef] [PubMed]
- Gavin, P.D.; El-Tamimy, M.; Keah, H.H.; Boyd, B.J. Tocopheryl phosphate mixture (TPM) as a novel lipid-based transdermal drug delivery carrier: Formulation and evaluation. Drug Deliv. Transl. Res. 2017, 7, 53–65. [Google Scholar] [CrossRef] [PubMed]
- Mukherjee, S.; Lekli, I.; Das, M.; Azzi, A.; Das, D.K. Cardioprotection with α-tocopheryl phosphate: Amelioration of myocardial ischemia reperfusion injury is linked with its ability to generate a survival signal through Akt activation. Biochim. Biophys. Acta 2008, 1782, 498–503. [Google Scholar] [CrossRef] [PubMed]
- Ogru, E.; Libinaki, R.; Gianello, R.; West, S.; Munteanu, A.; Zingg, J.M.; Azzi, A. Modulation of Cell Proliferation and Gene Expression by α-Tocopheryl Phosphates: Relevance to Atherosclerosis and Inflammation. Ann. N. Y. Acad. Sci. 2004, 1031, 405–411. [Google Scholar] [CrossRef] [PubMed]
- Negis, Y.; Aytan, N.; Özer, N.; Ogru, E.; Libinaki, R.; Gianello, R.; Azzi, A.; Zingg, J.M. The effect of tocopheryl phosphates on atherosclerosis progression in rabbits fed with a high cholesterol diet. Arch. Biochem. Biophys. 2006, 450, 63–66. [Google Scholar] [CrossRef] [PubMed]
- Libinaki, R.; Tesanovic, S.; Heal Nikolovski, B.; Vinh, A.; Widdop, R.E.; Gaspari, T.A.; Devaraj, S.; Ogru, E. Effect of tocopheryl phosphate on key biomarkers of inflammation: Implication in the reduction of atherosclerosis progression in a hypercholesterolaemic rabbit model. Clin. Exp. Pharmacol. Physiol. 2010, 37, 587–592. [Google Scholar] [CrossRef] [PubMed]
- Libinaki, R.; Vinh, A.; Tesanovic-Klajiac, S.; Woddop, R.; Gaspari, T. The effect of tocopheryl phosphates (TPM) on the development of atherosclerosis in apolipoprotein-E deficient mice. Clin. Exp. Pharmacol. Physiol. 2017, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Pham, A.; Gavin, P.; Libinaki, R.; Ramirez, G.; Boyd, B.J. A new lipid excipient, phosphorylated tocopherol mixture, TPM® enhances the solubilisation and oral bioavailability of poorly water soluble CoQ10 in a lipid formulation. J. Control. Release 2017, in press. [Google Scholar]
- Pham, A.; Gavin, P.; Libinaki, R.; Ramirez, G.; Khan, J.T.; Montagnat, O.; Boyd, B.J. Differential effects of TPM, a phosphorylated tocopherol mixture, and other tocopherol derivatives as excipients for enhancing the solubilization of lipophilic drugs during digestion of lipid-based formulations. AAPS PhamSciTech 2017. submitted. [Google Scholar]
- US Food and Drug Administration (USFDA). Guidance for Industry: Estimating the Maximum Safe Starting Dose in Adult Healthy Volunteer; USFDA: Rockville, MD, USA, 2005.
- Ghasemifard, S.; Sinclair, A.J.; Kaur, G.; Lewandowski, P.; Turchini, G.M. What Is the Most Effective Way of Increasing the Bioavailability of Dietary Long Chain Omega-3 Fatty Acids—Daily vs. Weekly Administration of Fish Oil? Nutrients 2015, 7, 5628–5645. [Google Scholar] [CrossRef] [PubMed]
- Singh, M. Essential Fatty Acids, DHA and the Human Brain. Indian J. Pediatr. 2005, 72, 239–242. [Google Scholar] [CrossRef] [PubMed]
- Youdim, K.A.; Martin, A.; Joseph, J.A. Essential fatty acids and the brain: Possible health implications. Int. J. Dev. Neurosci. 2000, 18, 383–3999. [Google Scholar] [CrossRef]
- Spector, A.A. Essential fatty acids. Lipids 1999, 34, S1–S3. [Google Scholar] [CrossRef] [PubMed]
- Kuratko, C.N.; Barrett, E.C.; Nelson, E.B.; Salem, N., Jr. The relationship of docosahexaenoic acid (DHA) with learning and behavior in healthy children: A review. Nutrients 2013, 5, 2777–2810. [Google Scholar] [CrossRef] [PubMed]
- Davidson, M.H.; Johnson, J.; Rooney, M.W.; Kyle, M.L.; Kling, D.F. A novel omega-3 free fatty acid formulation has dramatically improved bioavailability during a low-fat diet compared with omega-3-acid ethyl esters: The ECLIPSE (Epanova(®) compared to 22. Lovaza(®) in a pharmacokinetic single-dose evaluation) study. J. Clin. Lipidol. 2012, 6, 573–584. [Google Scholar] [CrossRef] [PubMed]
- Offman, E.; Marenco, T.; Ferber, S.; Johnson, J.; Kling, D.; Curcio, D.; Davidson, M. Steady-state bioavailability of prescription omega-3 on a low-fat diet is significantly improved with a free fatty acid formulation compared with an ethyl ester formulation: The ECLIPSE II study. Vasc. Health Risk Manag. 2013, 9, 563–573. [Google Scholar] [CrossRef] [PubMed]
- Maki, K.C.; Orloff, D.G.; Nicholls, S.J.; Dunbar, R.L.; Roth, E.M.; Curcio, D.; Johnson, J.; Kling, D.; Davidson, M.H. A highly bioavailable omega-3 free fatty acid formulation improves the cardiovascular risk profile in high-risk, statin-treated patients with residual hypertriglyceridemia (the ESPRIT trial). Clin. Ther. 2013, 35, 1400-11.e1-3. [Google Scholar] [CrossRef] [PubMed]
- Lopez-Toledano, M.A.; Thorsteinsson, T.; Daak, A.; Maki, K.C.; Johns, C.; Rabinowicz, A.L.; Sancilio, F.D. A Novel ω-3 Acid Ethyl Ester Formulation Incorporating Advanced Lipid TechnologiesTM (ALT®) Improves Docosahexaenoic Acid and Eicosapentaenoic Acid Bioavailability Compared with Lovaza®. Clin. Ther. 2017, 39, 581–591. [Google Scholar] [CrossRef] [PubMed]
- Hussey, E.K.; Portelli, S.; Fossler, M.J.; Gao, F.; Harris, W.S.; Blum, R.A.; Lates, C.D.; Gould, E.; Abu-Baker, O.; Johnson, S.; et al. Relative bioavailability of an emulsion formulation for omega-3-acid ethyl esters compared to the commercially available formulation: A randomized, parallel-group, single-dose study followed by repeat dosing in healthy volunteers. Clin. Pharmacol. Drug Dev. 2012, 1, 14–23. [Google Scholar] [CrossRef] [PubMed]
- Hoogevest, P.; Wendel, A. The use of natural and synthetic phospholipids as pharmaceutical excipients. Eur. J. Lipid Sci. Technol. 2014, 116, 1088–1107. [Google Scholar] [CrossRef] [PubMed]
| Dose | Low Dose DHA Treatments | High Dose DHA Treatments | |||
|---|---|---|---|---|---|
| Low DHA Alone | Low DHA/TPM (1:0.1) | Low DHA/TPM (1:0.5) | High DHA Alone | High DHA/TPM (1:0.1) | |
| DHA (mg/kg) | 88.6 | 88.6 | 88.6 | 265.7 | 265.7 |
| TPM (mg/kg) | - | 8.86 | 44.3 | - | 26.57 |
| Parameter | Low Dose DHA | High Dose DHA | |||
|---|---|---|---|---|---|
| Low DHA Alone (n = 9) | Low DHA/TPM (1:0.1) (n = 7) | Low DHA/TPM (1:0.5) (n = 9) | High DHA Alone (n = 10) | High DHA/TPM (1:0.1) (n = 8) | |
| Tmax (h) | 4.56 ± 2.79 | 3.33 ± 0.94 | 4.44 ± 1.33 | 3.2 ± 1.03 | 4.25 ± 1.67 |
| Cmax (µg/mL) | 67.20 ± 18.11 | 78.39 ± 25.21 | 91.95 ± 37.04 | 124.80 ± 37.19 | 160.50 ± 35.93 |
| p-value vs. control | - | p = 0.780 | p = 0.186 | - | p = 0.057 |
| AUC0–24 h (µg/mL·h) | 1208.00 ± 281.40 | 1396.60 ± 411.07 | 1560.60 ± 638.23 | 1581.18 ± 372.94 | 2078.30 ± 319.02 |
| p-value vs. control | - | p = 0.730 | p = 0.290 | - | p = 0.007 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Libinaki, R.; Gavin, P.D. Changes in Bioavailability of Omega-3 (DHA) through Alpha-Tocopheryl Phosphate Mixture (TPM) after Oral Administration in Rats. Nutrients 2017, 9, 1042. https://doi.org/10.3390/nu9091042
Libinaki R, Gavin PD. Changes in Bioavailability of Omega-3 (DHA) through Alpha-Tocopheryl Phosphate Mixture (TPM) after Oral Administration in Rats. Nutrients. 2017; 9(9):1042. https://doi.org/10.3390/nu9091042
Chicago/Turabian StyleLibinaki, Roksan, and Paul D. Gavin. 2017. "Changes in Bioavailability of Omega-3 (DHA) through Alpha-Tocopheryl Phosphate Mixture (TPM) after Oral Administration in Rats" Nutrients 9, no. 9: 1042. https://doi.org/10.3390/nu9091042
APA StyleLibinaki, R., & Gavin, P. D. (2017). Changes in Bioavailability of Omega-3 (DHA) through Alpha-Tocopheryl Phosphate Mixture (TPM) after Oral Administration in Rats. Nutrients, 9(9), 1042. https://doi.org/10.3390/nu9091042





