Evolutionary Developments in Interpreting the Gluten‐Induced Mucosal Celiac Lesion: An Archimedian Heuristic
Abstract
:1. Introduction
2. Early (Mis-) Interpretations of Intestinal Biopsies
3. The Immunological Functions of Intestinal Mucosa
4. Re-Evaluating Intraepithelial Lymphocyte (IEL) Counts Derived from the Existing Literature
5. Objective (Computerised) Measurements of Intestinal Mucosa
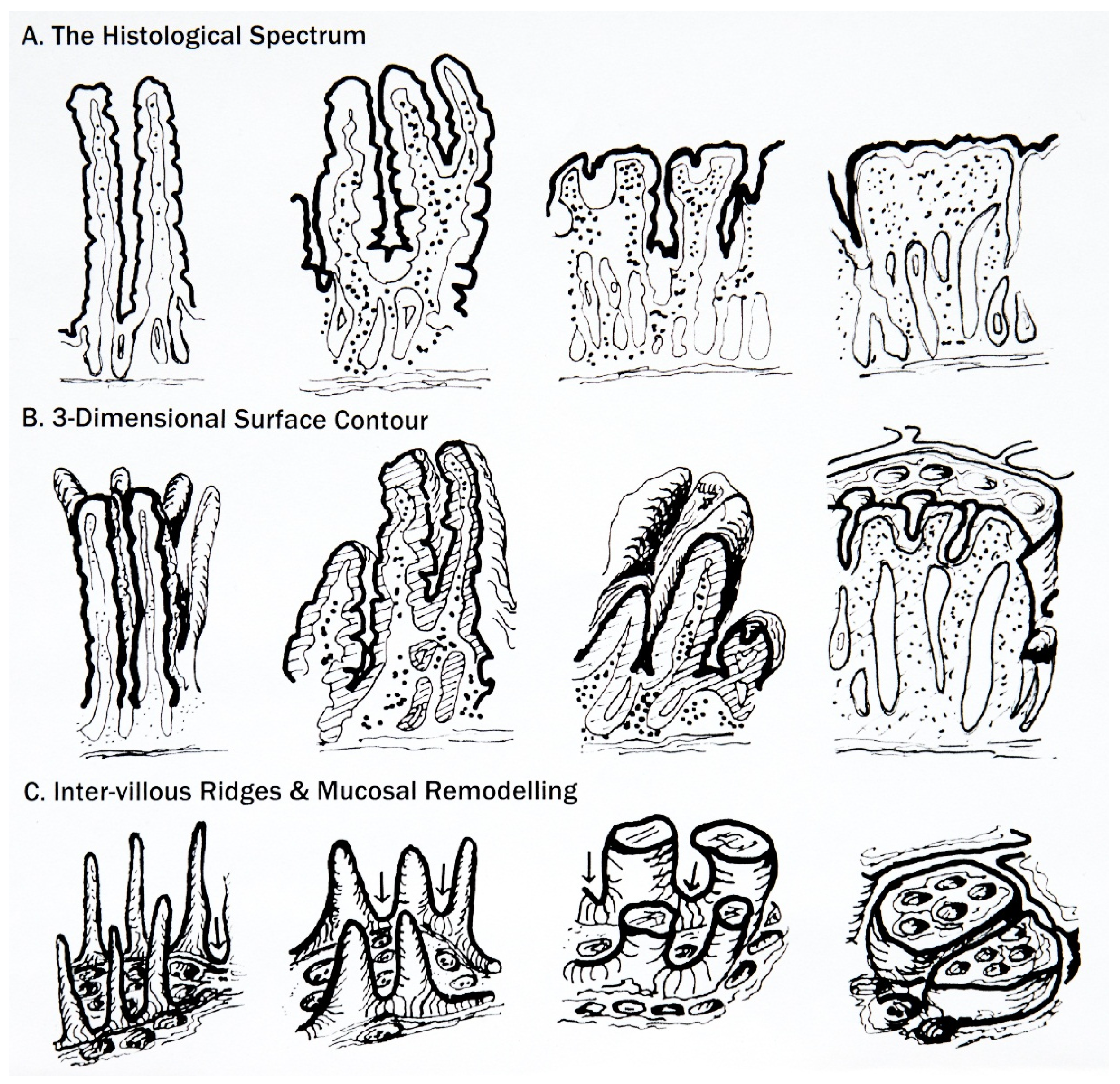
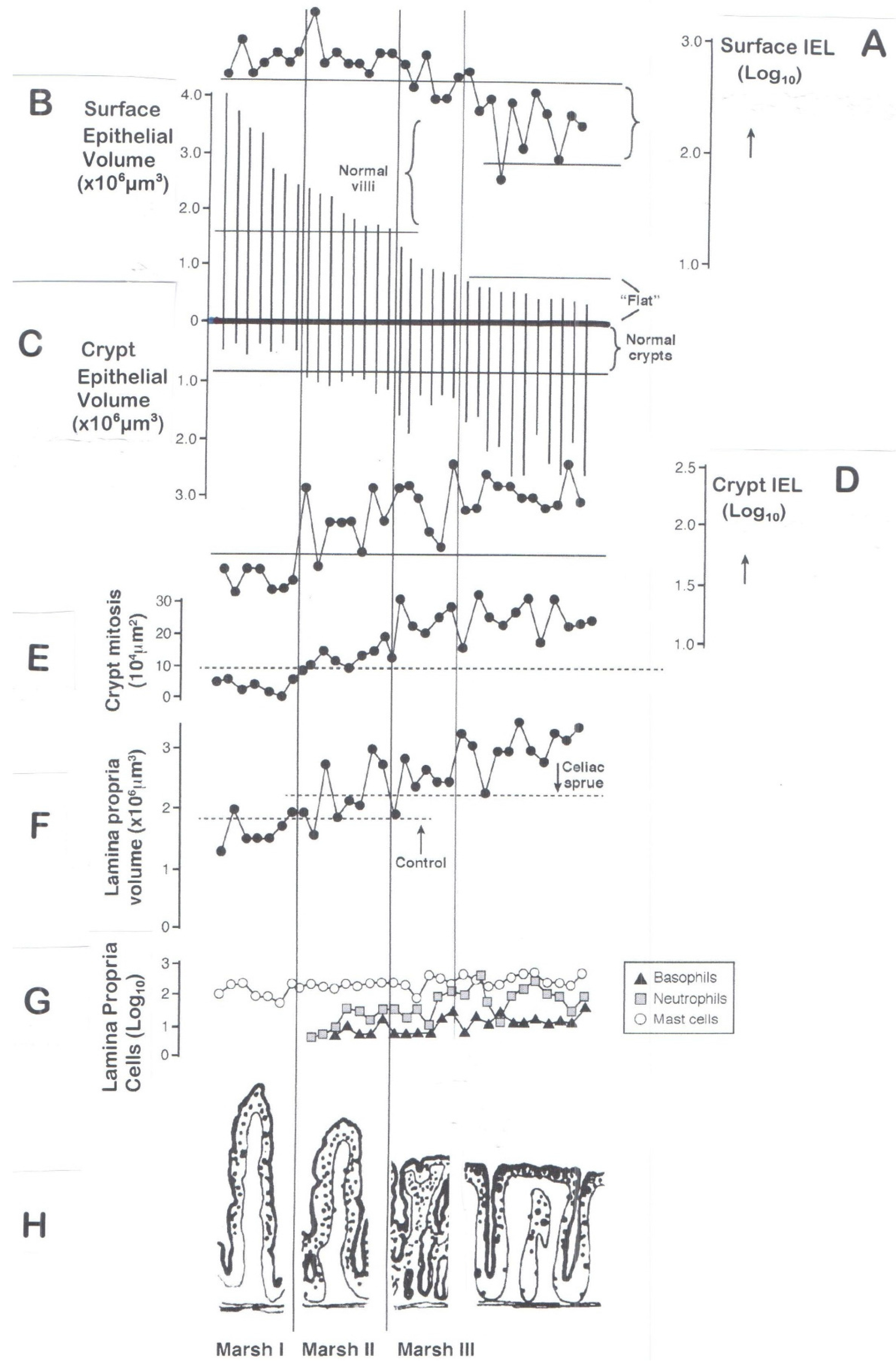
6. Classification of Mucosal Remodelling: A Major Hypertrophic Process
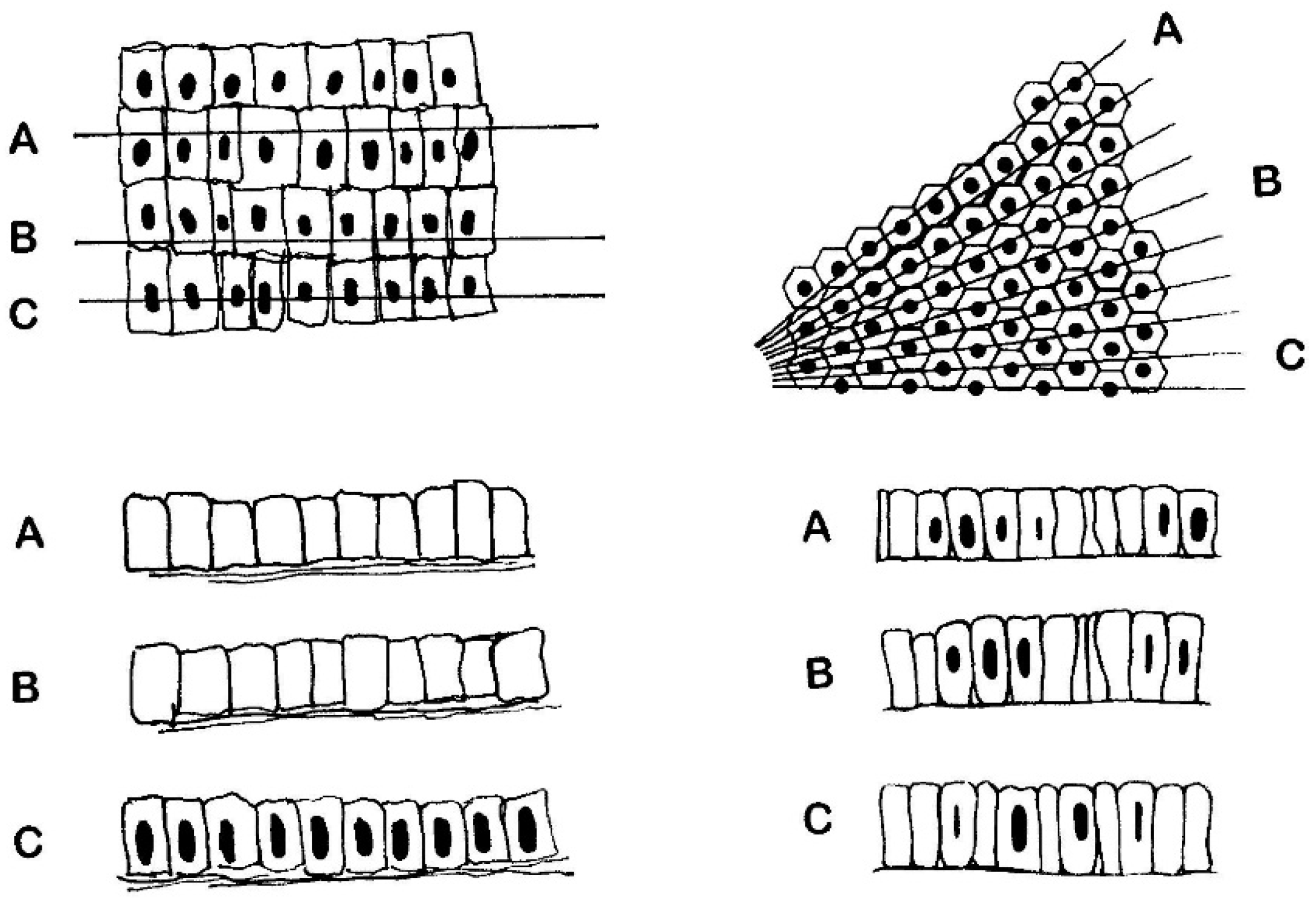
6.1. The Surface Epithelium
6.2. The Crypts
6.3. The Lamina Propria
7. Interpreting the Marsh Classification
7.1. So-Called “Non-Specificity” of the Marsh I and II Lesions
7.2. Irrelevance of the Marsh III Sub-Classification
- (a)
- Absence of appropriate criteria: these subdivisions were never precisely defined morphologically as verification of the proposed subdivisions. It is interesting to envisage how (and why) so many histologists thought they were identifying real structures. Even the micrographs illustrated in a later publication [74] written by histopathologists, for the help of other histologists, failed to correspond to the originals, again demonstrative of the subjective nature of the whole scheme.Oberhuber’s approach has now been further degraded by additional studies:
- (b)
- morphological—which highlight the misinterpretations of sectioned mosaic plateaus as supposedly representing ‘blunted’, ‘degenerate’ ‘villi’ [75];
- (c)
- immunohistochemical—demonstrating that varied sub-immunophenotype IEL are equally represented in each subdivision, when their density should have increased with the worsening histological picture alleged to represent each successive stage: a, b, c [76];
- (d)
- mathematical—the regression equations employed by Charlesworth and colleagues failed to identify the a,b,c subgrades as valid entities for improved pathological recognition [77];
- (e)
- clinical—there appear to be no published accounts in which a gastroenterologist necessarily had to rely, ultimately and crucially, on the pathologist’s sub-classification of the relevant mucosal biopsy in order to facilitate diagnosis, treatment, or offer a prognosis for the patients concerned;
- (f)
- generalised usage—finally, given the failure of this attempted reclassification, it seems to follow that more recently revised classifications of Marsh were based, however, on these sub-divisions, offering no further decisive clarity. In fact, they could be said to increase complexity and interpretational difficulties. For example, from a review of relevant papers published over the last decade, it is abundantly clear that these recent contenders for the job have not surfaced either as being more useful, more acceptable, or more easily employed. The original classification is as simple as could be.
7.3. The “Normal” Mucosa
7.4. Failures in Understanding the Marked Hypertrophic Remodelling Response
8. Afterword
- there are no (immuno)histologically unique diagnostic features for celiac disease that “absolutely” distinguish it from other mucosal enteropathies or more importantly, disease-control biopsies;
- the spectre of the “normal” mucosa, but which is consistent with true gluten sensitivity, remains a difficult problem to deal with, including its redefinition;
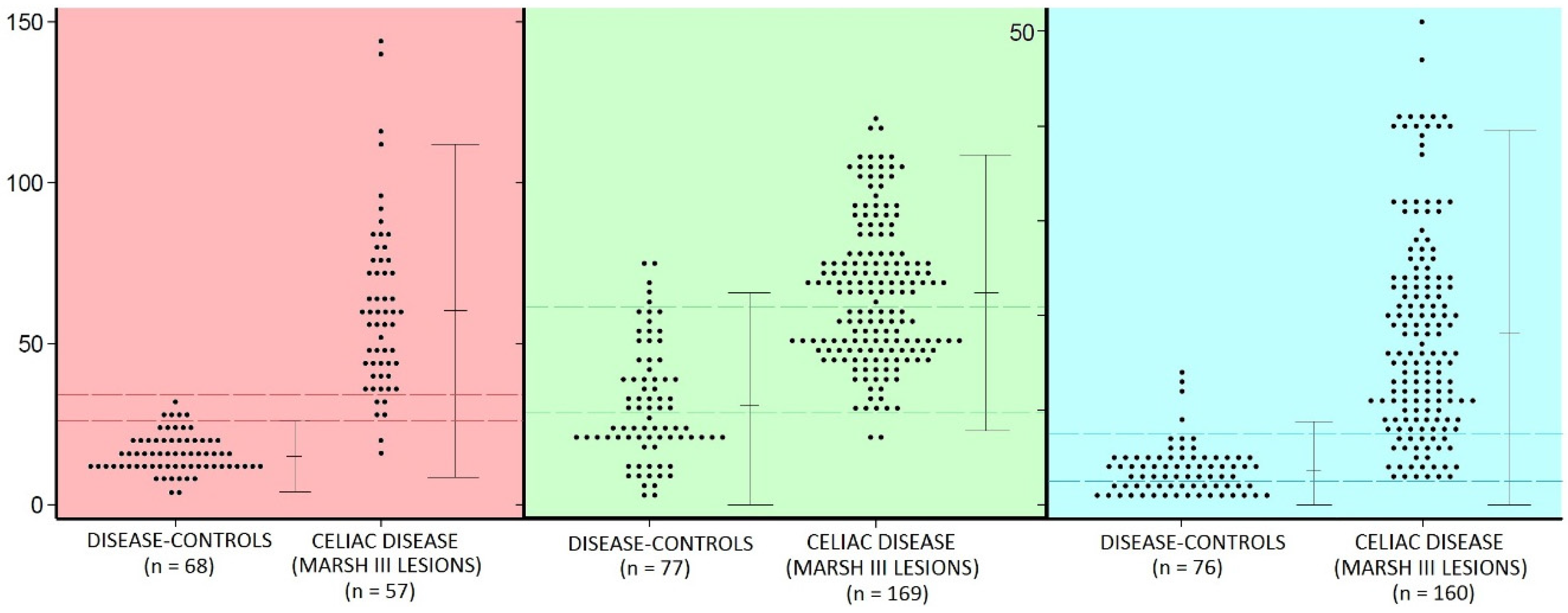
- there is considerable overlap between the populations of celiac intraepithelial lymphocyte (IEL) and controls (Figure 2)—regardless of the identifying technique used;
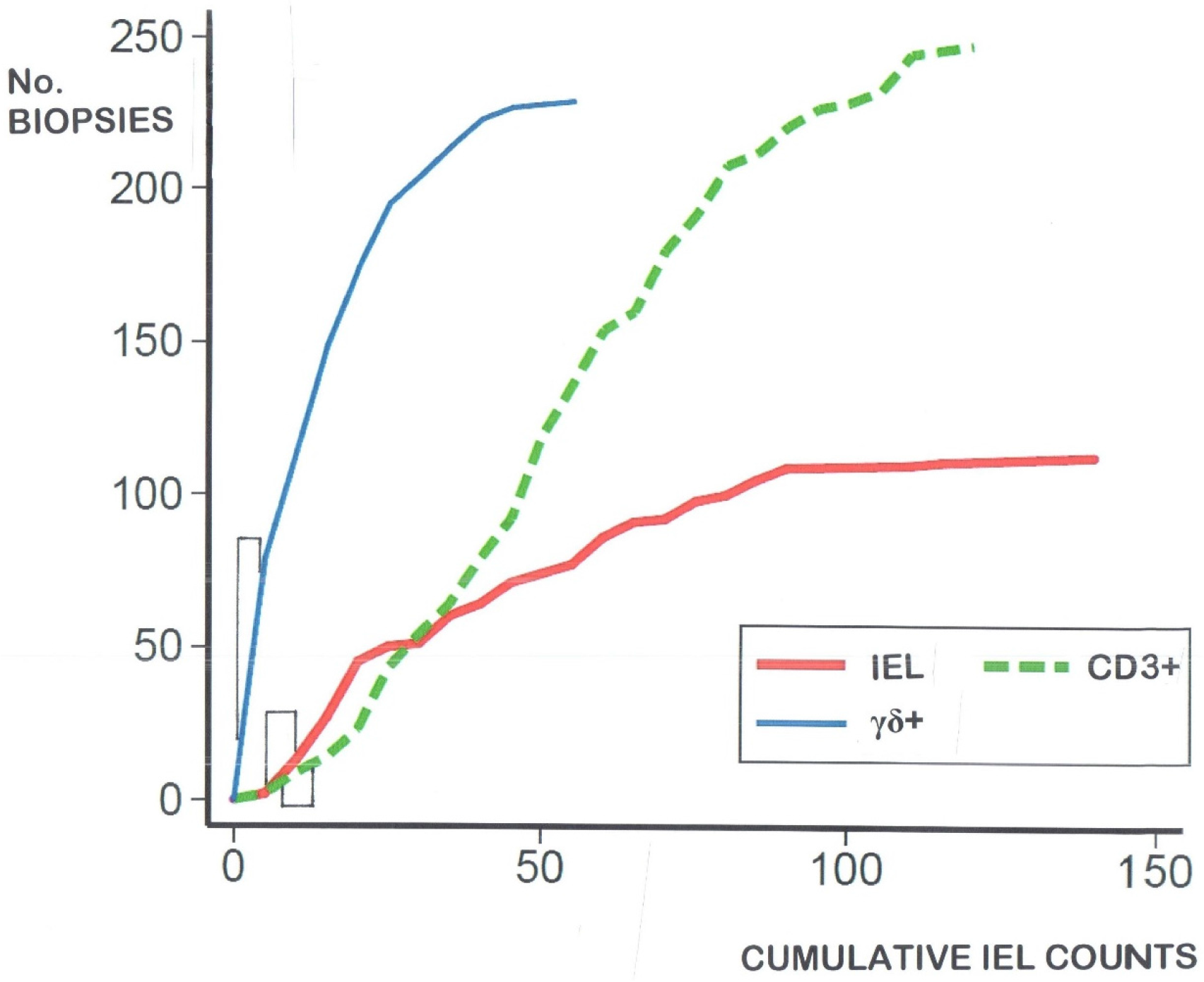
- IEL populations do not comprise two separate populations (bimodal), but represent graded biological outcomes (to luminal antigens), analogous to height, weight, blood pressure or acid secretion (Figure 3);
- additionally detailed studies of the dose-response characteristics of the CD3− innate pool of IEL, and their CD127+ and CD127− components may bring new insights to diagnosis and mucosal interpretation;
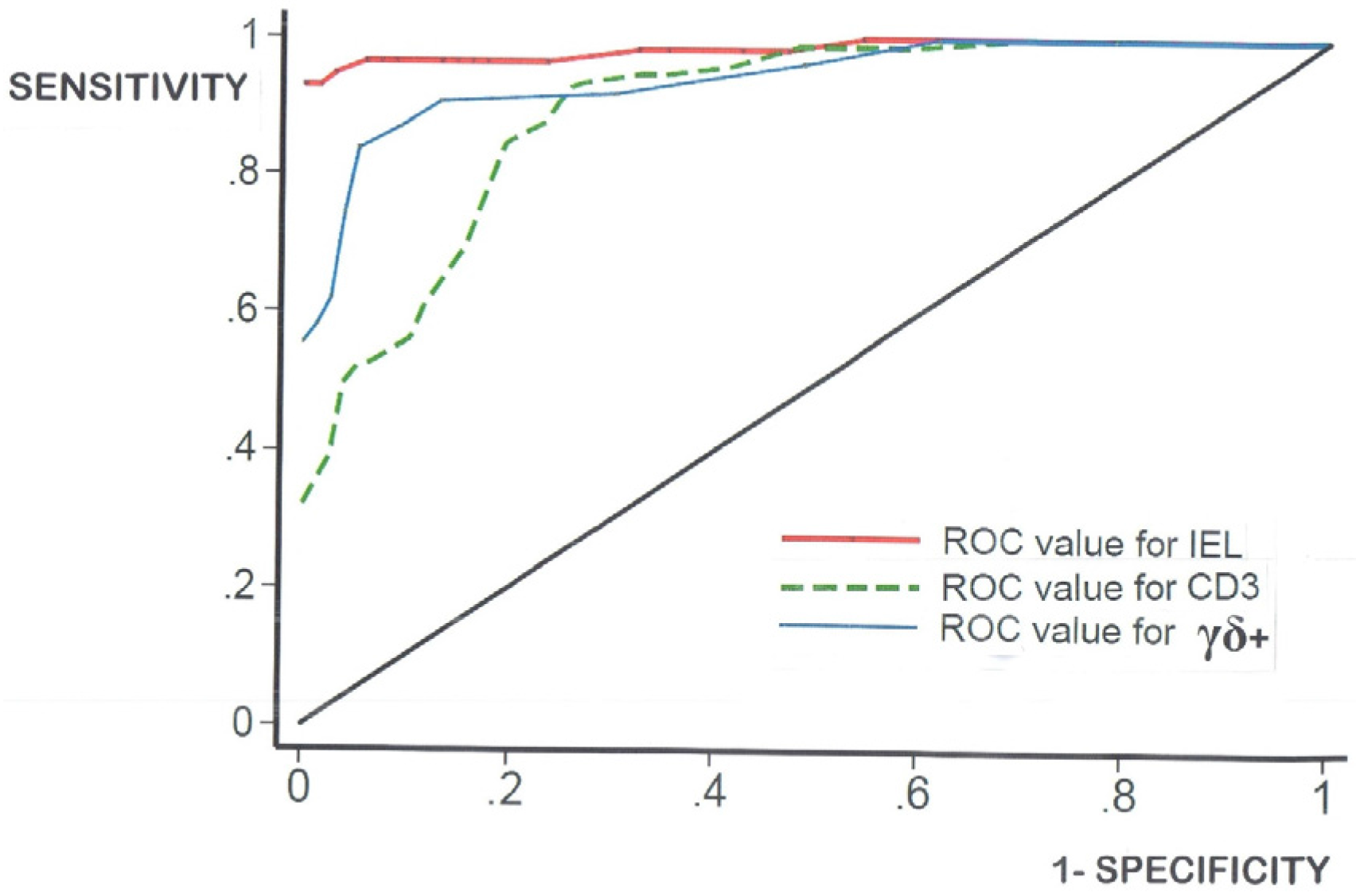
- log-transformation of the skewed celiac data does not produce means which materially differ from the numerical means (data not shown). Together, these results confirm that histopathologists do not need to log-transform their numerical counts, and that IEL counts in routine hematoxylin and eosin (H&E)sections can now be seen as a very easy and resourceful way of defining one’s cut-off, provided receiver-operating characteristic (ROC) curve analysis is additionally carried out;
- there is a vast cavern between high-level research still needed in continued interrogations of the mucosal response to gluten ingestion, and the somewhat more unsophisticated approaches deployable at histopathological level during routine diagnostic service work.
Conflicts of Interest
References
- Gee, S. On the Coeliac Affection. St. Bartholomew’s Hosp. Rep. 1888, 24, 17–20. [Google Scholar]
- Shiner, M. Jejunal biopsy tube. Lancet 1956, 1, 85. [Google Scholar] [CrossRef]
- Crosby, W.; Kugler, H. Intraluminal biopsy of the small intestine. The intestinal biopsy capsule. Am. J. Dig. Dis. 1958, 5, 217–222. [Google Scholar]
- Wood, I.; Doig, R.; Motteram, R.; Hughes, A. Gastric Biopsy: Report on fifty-five biopsies using a new flexible gastric biopsy tube. Lancet 1949, 1, 18–21. [Google Scholar] [CrossRef]
- Paulley, J. Observations on the aetiology of idiopathic steatorrhoea: Jejunal and lymph node biopsies. Br. Med. J. 1954, 2, 1318–1321. [Google Scholar] [CrossRef] [PubMed]
- Dicke, W.; Weijers, H.; van der Kamer, J. Coeliac disease. II—The presence in wheat ofa factor having a deleterious effect in cases of celiac disease. Acta Paediatr. 1953, 42, 34–42. [Google Scholar] [CrossRef]
- Haas, S. The value of the banana in the treatment of celiac disease. Am. J. Dis. Child. 1924, 28, 421–437. [Google Scholar] [CrossRef]
- Anderson, C. Histological changes in the duodenal mucosa in celiac disease: Reversibility during treatment with a gluten-free diet. Arch. Dis. Child. 1960, 35, 419–427. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Booth, C.; Stewart, J.; Holmes, R.; Brackenbury, W. Dissecting Microscope Appearances of Intestinal Mucosa; Intestinal Biopsy (Ciba Foundation Study Group No. 14); Wolstenholme, G., Cameron, M., Eds.; Churchill: London, UK, 1962; pp. 2–23. [Google Scholar]
- Cocco, A.; Dorhmann, M.; Hendrix, T. Reconstruction of normal jejunal biopsies: Three-dimensional histology. Gastroenterology 1966, 51, 24–31. [Google Scholar] [PubMed]
- Loehry, C.; Creamer, B. Three-dimensional structure of the human small intestinal mucosa in health and disease. Gut 1969, 10, 6–12. [Google Scholar] [CrossRef] [PubMed]
- Toner, P.; Carr, K.; Ferguson, A.; Mackay, C. Scanning and transmission electron microscopic studies of human intestinal mucosa. Gut 1970, 11, 471–481. [Google Scholar] [CrossRef] [PubMed]
- Creamer, B.; Leppard, P. Post-mortem examination of a small intestine in the coeliac syndrome. Gut 1965, 6, 466–471. [Google Scholar] [CrossRef] [PubMed]
- Padykula, H.; Strauss, E.; Ladman, A.; Gardner, F. A morphological and histochemical analysis of the human jejunal epithelium in nontropicalsprue. Gastroenterology 1961, 40, 735–765. [Google Scholar] [PubMed]
- Marsh, M.N.; Rostami, K. What is a normal intestinal mucosa? Gastroenterology 2016, 151, 784–788. [Google Scholar] [CrossRef] [PubMed]
- Gowans, J.L.; Knight, E.J. The route of re-circulation of lymphocytes in the rat. Proc. R. Soc. B 1964, 159, 257–282. [Google Scholar] [CrossRef]
- Crabbé, P.; Carbonara, A.; Heremans, J. The normal human intestinal mucosa as a major source of plasma cells containing γA-immunoglobulin. Lab. Investig. 1965, 14, 235–248. [Google Scholar] [PubMed]
- Tomasi, T.; Tan, E.; Solomon, A.; Prendergast, R. Characteristics of an immune system common to certain external secretions. J. Exp. Med. 1965, 121, 101–125. [Google Scholar] [CrossRef] [PubMed]
- Crabbé, P.; Nash, D.; Bazin, H.; Eyssen, H.; Heremans, J. Antibodies of the IgA type in intestinal plasma cells of germfree mice after oral or parenteral immunization with ferritin. J. Exp. Med. 1969, 130, 723–738. [Google Scholar] [CrossRef] [PubMed]
- Loft, D.; Marsh, M.N.; Sandle, G.; Crowe, P.; Garner, V.; Gordon, D.; Baker, R. Studies of intestinal lymphoid tissue. XII-Epithelial lymphocyte and mucosal responses to rectal gluten challenge in celiac sprue. Gastroenterology 1989, 97, 29–37. [Google Scholar] [CrossRef]
- Ensari, A.; Marsh, M.N.; Morgan, S.; Lobley, R.; Unsworth, D.; Kounali, D.; Crowe, P.; Paisley, J.; Moriarty, K.; Lowry, J. Diagnosing coeliac disease by rectal gluten challenge: A prospective study based on immunopathology, computerized image analysis and logistic regression analysis. Clin. Sci. 2001, 101, 199–207. [Google Scholar] [CrossRef] [PubMed]
- Quiding-Järbrink, M.; Lakew, M.; Nordström, I.; Banchereau, J.; Butcher, E.; Holmgren, J.; Czerkinsky, C. Human circulating specific antibody-forming cells after systemic and mucosal immunizations: Differential homing commitments and cell surface differentiation markers. Eur. J. Immunol. 1995, 25, 322–327. [Google Scholar] [CrossRef] [PubMed]
- Barratt, M.; Powell, J.; Allen, W.; Porter, P. Immunopathology of intestinal disorders in farm animals. In Immunopathology of the Small Intestine; Marsh, M.N., Ed.; Wiley: Chichester, UK, 1987; pp. 253–281. [Google Scholar]
- Kantele, J.; Arvilommi, H.; Kontiainen, S.; Salmi, M.; Jalkanen, S.; Savilahti, E.; Westerholm, M.; Kantele, A. Mucosally activated circulating human B cells in diarrhea express homing receptors directing them back to the gut. Gastroenterology 1996, 110, 1061–1067. [Google Scholar] [CrossRef] [PubMed]
- Ráki, M.; Fallang, L.; Brottveit, M.; Bergseng, E.; Quarsten, H.; Lundin, K.; Sollid, L. Tetramer visualisation of gut-homing gluten-specific T cells in the peripheral blood of celiac disease patients. Proc. Natl. Acad. Sci. USA 2007, 104, 2831–2836. [Google Scholar] [CrossRef] [PubMed]
- Brottveit, M.; Ráki, M.; Bergsen, E.; Fallang, L.; Simonsen, B.; Løvik, A.; Larsen, S.; Løberg, E.M.; Jahnsen, F.L.; Sollid, L.M.; et al. Assessing possible celiac disease by an HLA-DQ2-gliadin tetramer test. Am. J. Gastroenterol. 2011, 106, 1318–1324. [Google Scholar] [CrossRef] [PubMed]
- Douglas, A.; Crabbé, P.; Hobbs, J. Immunochemical studies of the serum, intestinal secretions and intestinal mucosa in patients with adult celiac disease and other forms of the celiac syndrome. Gastroenterology 1970, 59, 414–425. [Google Scholar] [PubMed]
- Ferguson, A.; Murray, D. Quantitation of intraepithelial lymphocytes in human jejunum. Gut 1971, 12, 988–994. [Google Scholar] [CrossRef] [PubMed]
- Batman, P.; Miller, A.; Harris, J.; Pinching, A.; Griffin, G. Jejunal enteropathy associated with human immunodeficiency virus infection: Quantitative histology. J. Clin. Pathol. 1989, 42, 275–281. [Google Scholar] [CrossRef] [PubMed]
- Hayat, M.; Cairns, A.; Dixon, M. Quantitation of intraepithelial lymphocytes in human duodenum: What is normal? J. Clin. Pathol. 2002, 55, 393–395. [Google Scholar] [CrossRef] [PubMed]
- Mahadeva, S.; Wyatt, J.; Howdle, P. Is a raised intraepithelial lymphocyte count with normal duodenal villous architecture clinically relevant? J. Clin. Pathol. 2002, 55, 424–428. [Google Scholar] [CrossRef] [PubMed]
- Kakar, S.; Nehra, V.; Murray, J.; Dayharsh, G.; Burgart, L. Significance of intraepithelial lymphocytosis in small bowel biopsy samples with normal mucosal architecture. Am. J. Gastroenterol. 2003, 98, 2027–2033. [Google Scholar] [CrossRef] [PubMed]
- Veress, B.; Franzén, L.; Bodin, L.; Borch, K. Duodenal intraepithelial lymphocyte-count revisited. Scand. J. Gastroenterol. 2004, 9, 138–144. [Google Scholar] [CrossRef]
- Biagi, F.; Luinette, O.; Campanella, J.; Klersy, C.; Zambelli, C.; Villanacci, V. Intraepithelial lymphocytes in the villous tip: Do they indicate potential celiac disease? J. Clin. Pathol. 2004, 57, 835–839. [Google Scholar] [CrossRef] [PubMed]
- Nasseri-Moghaddam, S.; Mofid, A.; Nouraie, M.; Abedi, B.; Pourshams, A.; Malekzadeh, R.; Sotoudeh, M. The normal range of duodenal intraepithelial lymphocytes. Arch. Iran. Med. 2008, 11, 136–142. [Google Scholar] [PubMed]
- Siriweera, E.; Qi, Z.; Yong, J. Validity of intraepithelial lymphocyte counts in the diagnosis of celiac disease: A histopathological study. Int. J. Celiac Dis. 2015, 3, 136–142. [Google Scholar]
- Guix, M.; Skinner, J.; Whitehead, R. Measuring intraepithelial lymphocytes, surface area, and volume of lamina propria in the jejunal mucosa of celiac patients. Gut 1979, 20, 275–278. [Google Scholar] [CrossRef] [PubMed]
- Weibel, E.R. Stereological Methods; Academic Press: New York, NY, USA, 1979; Volume I. [Google Scholar]
- Marsh, M.N.; Crowe, P.; Moriarty, K.; Ensari, A. Morphometric Analysis of Intestinal Mucosa: The measurement of volume compartments and cell volumes in human intestinal mucosa. Methods Mol. Med. 2000, 41, 125–145. [Google Scholar] [PubMed]
- Marsh, M.N. Gluten, major histocompatibility complex, and the small intestine: A molecular and mmunobiologic approach to the spectrum of gluten sensitivity (‘celiac sprue’). Gastroenterology 1992, 102, 330–354. [Google Scholar] [CrossRef]
- Marsh, M.N. Grains of truth: Evolutionary changes in small intestinal mucosa in response to environmental antigenic challenge. Gut 1990, 31, 111–114. [Google Scholar] [CrossRef] [PubMed]
- Doherty, M.; Barry, R. Gluten-induced mucosal changes in subjects without overt small-bowel disease. Lancet 1981, 317, 517–520. [Google Scholar] [CrossRef]
- Egan-Mitchell, B.; Fottrell, P.; McNichol, B. Early or pre-coeliac mucosa: Development of gluten enteropathy. Gut 1981, 22, 65–69. [Google Scholar] [CrossRef] [PubMed]
- Marsh, M.N. Studies of intestinal lymphoid tissue. XIII—Immunopathololgy of the evolving celiac sprue lesion. Pathol. Res. Pract. 1989, 185, 774–777. [Google Scholar] [CrossRef]
- Maki, M.; Holm, K.; Koskimies, S.; Hallstgrom, O.; Visakorpi, J. Normal small bowel biopsy followed by celiac disease. Arch. Dis. Child. 1990, 65, 1137–1141. [Google Scholar] [CrossRef] [PubMed]
- O’Mahony, S.; Vestey, J.; Ferguson, A. Similarities in intestinal humoral immunity in Dermatitis herpetiformis without enteropathy and in coeliac disease. Lancet 1990, 35, 1487–1490. [Google Scholar] [CrossRef]
- Collin, P.; Helin, H.; Mäki, M.; Hallström, O.; Karvonen, A.-L. Follow-up of patients positive in reticulin and gliadin antibody tests with normal small-bowel biopsy findings. Scand. J. Gastroenterol. 1993, 28, 595–598. [Google Scholar] [CrossRef] [PubMed]
- Niveloni, S.; Pedreira, S.; Sugai, E.; Vazquez, H.; Smecuol, E.; Fiorini, A. The natural history of gluten sensitivity: Report of two patients from a long-term follow-up of non-atrophic, first-degree relatives. Am. J. Gastroenterol. 2000, 95, 463–468. [Google Scholar] [CrossRef] [PubMed]
- Marsh, M.N. Mucosal pathology in gluten sensitivity. In Coeliac Disease; Marsh, M.N., Ed.; Blackwell Scientific Publications: Oxford, UK, 1992; pp. 136–191. [Google Scholar]
- Marsh, M.N.; Loft, D.; Garner, V.; Gordon, D. Time/dose responses of celiac mucosae to graded oral challenges with Frazer’s fraction III of gliadin. Eur. J. Gastroenterol. Hepatol. 1992, 4, 667–674. [Google Scholar]
- Mowat, A.; Ferguson, A. Intraepithelial lymphocyte counts and crypt hyperplasia measure the mucosal component of the graft-versus-host reaction in mouse small intestine. Gastroenterology 1982, 83, 417–423. [Google Scholar] [PubMed]
- Bjarnason, I.; Peters, T.; Veall, N. A persistent defect in intestinal permeability in celiac disease demonstrated by a 51Cr-labelled EDTA absorption test. Lancet 1983, 12, 323–325. [Google Scholar] [CrossRef]
- Marsh, M.N.; Bjarnason, I.; Shaw, J.; Ellis, A.; Baker, R. Studies of intestinal lymphoid tissue: XIV—HLA status, mucosal morphology, permeability and epithelial lymphocyte populations in first degree relatives of patients with coeliac disease. Gut 1990, 31, 32–36. [Google Scholar] [CrossRef] [PubMed]
- Crowe, P.; Marsh, M.N. Morphometric analysis of small intestinal mucosa IV. Determining cell volumes. Virchows Arch. A 1993, 422, 459–466. [Google Scholar] [CrossRef]
- Crowe, P.; Marsh, M.N. Morphometric analysis of small intestinal mucosa. VI—Principles in enumerating intra-epithelial lymphocytes. Virchows Arch. 1994, 424, 301–306. [Google Scholar] [CrossRef] [PubMed]
- Marsh, M.N. Studies of intestinal lymphoid tissue. III—Quantitative studies of epithelial lymphocytes in small intestinal mucosa of control human subjects and of patients with celiac sprue. Gastroenterology 1980, 79, 481–492. [Google Scholar] [PubMed]
- Marsh, M.N. Studies of intestinal lymphoid tissue. IV – The predictive value of raised mitotic indices among jejunal epithelial lymphocytes in the diagnosis of gluten-sensitive enteropathy. J. Clin. Pathol. 1982, 35, 517–525. [Google Scholar] [CrossRef] [PubMed]
- Marsh, M.N.; Haeney, M. Studies of intestinal lymphoid tissue. VI—Proliferative response of small intestinal lymphocytes distinguishes gluten- from non-gluten-induced enteropathy. J. Clin. Pathol. 1983, 76, 149–160. [Google Scholar] [CrossRef]
- Trier, J.; Browning, T. Epithelial-cell renewal in cultured duodenal biopsies in celiac sprue. N. Eng. J. Med. 1970, 283, 1245–1250. [Google Scholar] [CrossRef] [PubMed]
- Watson, A.; Wright, N. Morphology and cell kinetics of the jejunal mucosa in untreated patients. Clin. Gastroenterol. 1974, 3, 11–31. [Google Scholar] [PubMed]
- Mowat, A.; Ferguson, A. Hypersensitivity reactions in the small intestine: 6—Pathogenesis of the graft-versus-host reaction in the small intestinal mucosa of the mouse. Transplantation 1981, 32, 238–243. [Google Scholar] [CrossRef] [PubMed]
- Elson, C.; Reilly, R.; Rosenberg, I. Small intestinal injury in the GvHR: An innocent bystander phenomenon. Gastroenterology 1977, 72, 886–889. [Google Scholar] [PubMed]
- Marsh, M.N.; Hinde, J. Morphometric analysis of small intestinal mucosa. III—The quantitation of crypt epithelial volumes and lymphoid cell infiltrates, with reference to celiac sprue mucosae. Virchows Arch. 1986, 409, 11–22. [Google Scholar] [CrossRef]
- Marsh, M.N.; Hinde, J. Inflammatory component of celiac sprue mucosa. I—Mast cells, basophils, and eosinophils. Gastroenterology 1985, 89, 92–101. [Google Scholar] [CrossRef]
- Dhesi, I.; Marsh, M.N.; Kelly, C.; Crowe, P. Morphometric analysis of small intestinal mucosa. II—Determination of lamina propria volumes, plasma cell and neutrophil populations within control and coeliac disease mucosae. Virchows Arch. 1984, 403, 173–180. [Google Scholar] [CrossRef]
- Dvorak, H.; Dvorak, A. Basophils, mast cells and cellular immunity in animals and man. Hum. Pathol. 1972, 3, 454–456. [Google Scholar] [CrossRef]
- Ruitenberg, E.; Elgersma, A. Absence of intestinal mast cell response in congenitally athymic mice during Trichinella spiralis infection. Nature 1976, 264, 258–260. [Google Scholar] [CrossRef] [PubMed]
- Befus, D.; Pearce, F.; Gauldie, J.; Horsewood, P.; Bienenstock, J. Mucosal mast cells: I—Isolation and functional characteristics of rat intestinal mast cells. J. Immunol. 1982, 128, 2475–2480. [Google Scholar] [CrossRef]
- Schmidt, E.; Smyrk, T.; Faubion, W.; Oxentenko, A. Duodenal intraepithelial lymphocytosis with normal villous architecture in pediatric patients: Mayo Clinic experiences, 2000–2009. J. Pediatr. Gastroenterol. Nutr. 2013, 56, 51–55. [Google Scholar] [CrossRef] [PubMed]
- Zanini, B.; Lanzarotto, F.; Villanacci, V.; Carabellese, N.; Ricci, C.; Lanzini, A. Clinical expression of lymphocytic duodenosis in “mild enteropathy” celiac disease and in functional gastrointestinal syndromes. Scand. J. Gastroenterol. 2014, 49, 794–800. [Google Scholar] [CrossRef] [PubMed]
- Rostami, K.; Aldulaimi, D.; Holmes, G.; Johnson, M.; Robert, M.; Srivastava, A.; Flejou, J.F.; Sanders, D.S.; Volta, U.; Derakhshan, M.H.; et al. Microscopic Enteritis: The Bucharest Consensus. World J. Gastroenterol. 2015, 21, 2593–2604. [Google Scholar] [CrossRef] [PubMed]
- Not, T.; Zivberna, F.; Vatta, S.; Quaglia, S.; Martelossi, S.; Villanacci, V.; Marzari, R.; Florian, F.; Vecchiet, M.; Sulic, A.M.; et al. Cryptic genetic gluten intolerance revealed by intestinal anti-transaminase antibodies and response to gluten-free diet. Gut 2011, 60, 1487–1493. [Google Scholar] [CrossRef] [PubMed]
- Oberhuber, G.; Granditsch, G.; Vogelsang, H. The histopathology of celiac disease: Time for a standardized report scheme for pathologists. Eur. J. Gastroenterol. Hepatol. 1999, 11, 1185–1194. [Google Scholar] [CrossRef] [PubMed]
- Dickson, B.; Streutket, C.; Chetty, R. Coeliac disease: An update for pathologists. J. Clin. Pathol. 2006, 59, 1008–1016. [Google Scholar] [CrossRef] [PubMed]
- Marsh, M.N.; Johnson, M.; Rostami, K. Mucosal histopathology in celiac disease: A rebuttal of Oberhüber’s sub-division of Marsh III. Gastroenterol. Hepatol. Bed Bench 2015, 8, 99–109. [Google Scholar]
- De Andrés, A.; Camarero, C. Distal duodenum versus bulb: Intraepithelial lymphocytes have something to say about celiac disease diagnosis. Dig. Dis. Sci. 2015, 60, 1004–1009. [Google Scholar] [CrossRef] [PubMed]
- Charlesworth, R.; Andronicus, N.; Scott, D.; McFarlane, J.; Agnew, L. Can the sensitivity of the histopathological diagnosis of celiac disease be increased and can treatment progression be monitored using mathematical modelling of histological section?—A pilot study. J. Adv. Med. Sci. 2017, in press. [Google Scholar]
- Kaukinen, K.; Peraaho, M.; Collin, P. Small-bowel mucosal transaminase 2-specific IgA deposits in celiac disease without villous atrophy: A prospective and randomized clinical study. Scand. J. Gastroenterol. 2005, 40, 564–572. [Google Scholar] [CrossRef] [PubMed]
- Sbarbati, A.; Valletta, E.; Bertini, M.; Cipolli, M.; Morroni, M.; Pinelli, L.; Tatò, L. Gluten sensitivity and “normal” histology: Is the intestinal mucosa really normal? Dig. Liver Dis. 2003, 35, 768–773. [Google Scholar] [CrossRef]
- Tosca, A.; Maglio, M.; Paparo, F.; Rapacciuolo, L.; Sannino, A.; Miele, E. Immunoglobulin A anti-tissue transglutaminase antibody deposits in the small intestinal mucosa of children with no villous atrophy. J. Pediatr. Gastroenterol. Nutr. 2008, 47, 293–398. [Google Scholar] [CrossRef] [PubMed]
- Rubio-Tapia, A.; Hill, I.; Kelly, C.; Claderwood, A.; Murray, J. ACG guidelines: Diagnosis and management of celiac disease. Am. J. Gastroenterol. 2013, 108, 656–676. [Google Scholar] [CrossRef] [PubMed]
- Ludvigsson, J.; Bai, J.; Biagi, F.; Card, T.R.; Ciacci, C.; Ciclitira, P.J.; Green, P.H.; Hadjivassiliou, M.; Holdoway, A.; van Heel, D.A.; et al. Diagnosis and management of adult celiac disease: Guidelines from the British Society of Gastroenterology. Gut 2014, 63, 1210–1228. [Google Scholar] [CrossRef] [PubMed]
- Tursi, A.; Brandimarte, G. The symptomatic and histologic response to a gluten-free diet in patients with borderline enteropathy. J. Clin. Gastroenterol. 2003, 36, 13–17. [Google Scholar] [CrossRef] [PubMed]
| Paper | Methods | Number of Biopsies | Upper Range | Comments |
|---|---|---|---|---|
| Ferguson and Murray, 1971 [28] | H&E staining IEL/100 enterocytes | 40 | 40 | Used controls, celiac and autoimmune conditions. Incorrect about normally distributed IEL. Highest IEL count recorded, of 155 |
| 7 μm sections | ||||
| Batman et al., 1989 [29] | H&E staining 5 μm sections | 8 | 33 | Study of HIV enteropathy |
| Hayat et al., 2002 [30] | H&E staining | 20 | 25 | Counts made on uninterrupted length of epithelium >500 epithelial cells: Controls defined only by a “normal” sugar permeability |
| 4 μm sections | ||||
| Mahadeva et al., 2002 [31] | H&E staining | ?? | 22 | Major interest in normal villi with IEL infiltrate |
| 3 μm sections | Really difficult to infer group numbers here | |||
| Kakar et al., 2003 [32] | H&E staining | 12 | 39 | Interest in normal villi with IEL infiltrates |
| Veress et al., 2004 [33] | H&E staining | 64 | 20 | 3 μm H&E sections: |
| CD3+ counts | 5–9 | If IEL to EC ratio >5:1, do CD3 count | ||
| Biagi et al., 2004 [34] | H&E staining | 17 | 45 | Major interest in villous tip counts |
| Nasseri-Moghaddam et al., 2008 [35] | H&E staining | 46 | 46 | Establishing normal criteria by histology and immuno-cytology |
| CD45+ counts | 47 | |||
| Siriweera et al., 2015 [36] | H&E staining | 75 | 8 | Retrospective study on 38 control specimens and 37 celiacs. Inexplicably small upper ranges for both groups |
| Lymphocyte Subtype | H&E Stained | CD3+ | γδ+ |
|---|---|---|---|
| AUC | 0.985 | 0.891 | 0.943 |
| OPTIMALCUT-OFF | 27 | 40 | 6 |
| FALSE-POSITIVE | 5 | 21 | 10 |
| FALSE-NEGATIVE | 3 | 11 | 15 |
| Disease Controls | Celiac Disease | |
|---|---|---|
| Surface Epithelium | ||
| Volume (×106 μm3) | 2.3 (1.5–3.6) | 0.4 (0.2–0.6) |
| Cell Height (μm) | 37 (30–43) | 33 (27–33) |
| Cell Width (μm) | 5.1 (4.1–6.2) | 4.7 (3.8–5.8) |
| Cell Volume (μm3) | 800 (500–1250) | 600 (390–920) |
| No. Enterocytes/Volume | 3000 (1935–4435) | 600 (320–1100) |
| No. IEL/Volume | 350 (275–450) | 190 (150–240) |
| IEL/100 enterocytes | ||
| (‘Absolute’ by Image Analysis) | 12 (10–16) | 32 (27–37) |
| (Ferguson, per 100 cells) | 24 (11–53) | 61 (31–122) |
| Enterocytes per Lymphocyte | 8 (7–11) | 3 (2–4) |
| CRYPTS | ||
| Volume (×106 μm3) | 0.5–0.6 | 1.7 |
| IEL (‘Absolute’/volume) | 30 (12–48) | 173 (121–225) |
| LAMINA PROPRIA | ||
| Volume (106 μm3) | 1.4 (1.12–1.6) | 3.1 (2.8–3.5) |
| Cells/Volume | ||
| (‘Absolute’) | ||
| Mast Cells | 14 (10–20) | 38 (22–54) |
| Eosinophils | 18 (16–20) | 62 (50–74) |
| Basophils | 0.7 (0.48–1.12) | |
| Neutrophils | 45 (25–65) |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Marsh, M.N.; Heal, C.J. Evolutionary Developments in Interpreting the Gluten‐Induced Mucosal Celiac Lesion: An Archimedian Heuristic. Nutrients 2017, 9, 213. https://doi.org/10.3390/nu9030213
Marsh MN, Heal CJ. Evolutionary Developments in Interpreting the Gluten‐Induced Mucosal Celiac Lesion: An Archimedian Heuristic. Nutrients. 2017; 9(3):213. https://doi.org/10.3390/nu9030213
Chicago/Turabian StyleMarsh, Michael N., and Calvin J. Heal. 2017. "Evolutionary Developments in Interpreting the Gluten‐Induced Mucosal Celiac Lesion: An Archimedian Heuristic" Nutrients 9, no. 3: 213. https://doi.org/10.3390/nu9030213
APA StyleMarsh, M. N., & Heal, C. J. (2017). Evolutionary Developments in Interpreting the Gluten‐Induced Mucosal Celiac Lesion: An Archimedian Heuristic. Nutrients, 9(3), 213. https://doi.org/10.3390/nu9030213




