Biomarkers to Monitor Gluten-Free Diet Compliance in Celiac Patients
Abstract
:1. Introduction
2. Monitoring of Gluten-Free Diet Compliance
2.1. Symptom Assessment
2.2. Validated Surveys and Dietary Interviews
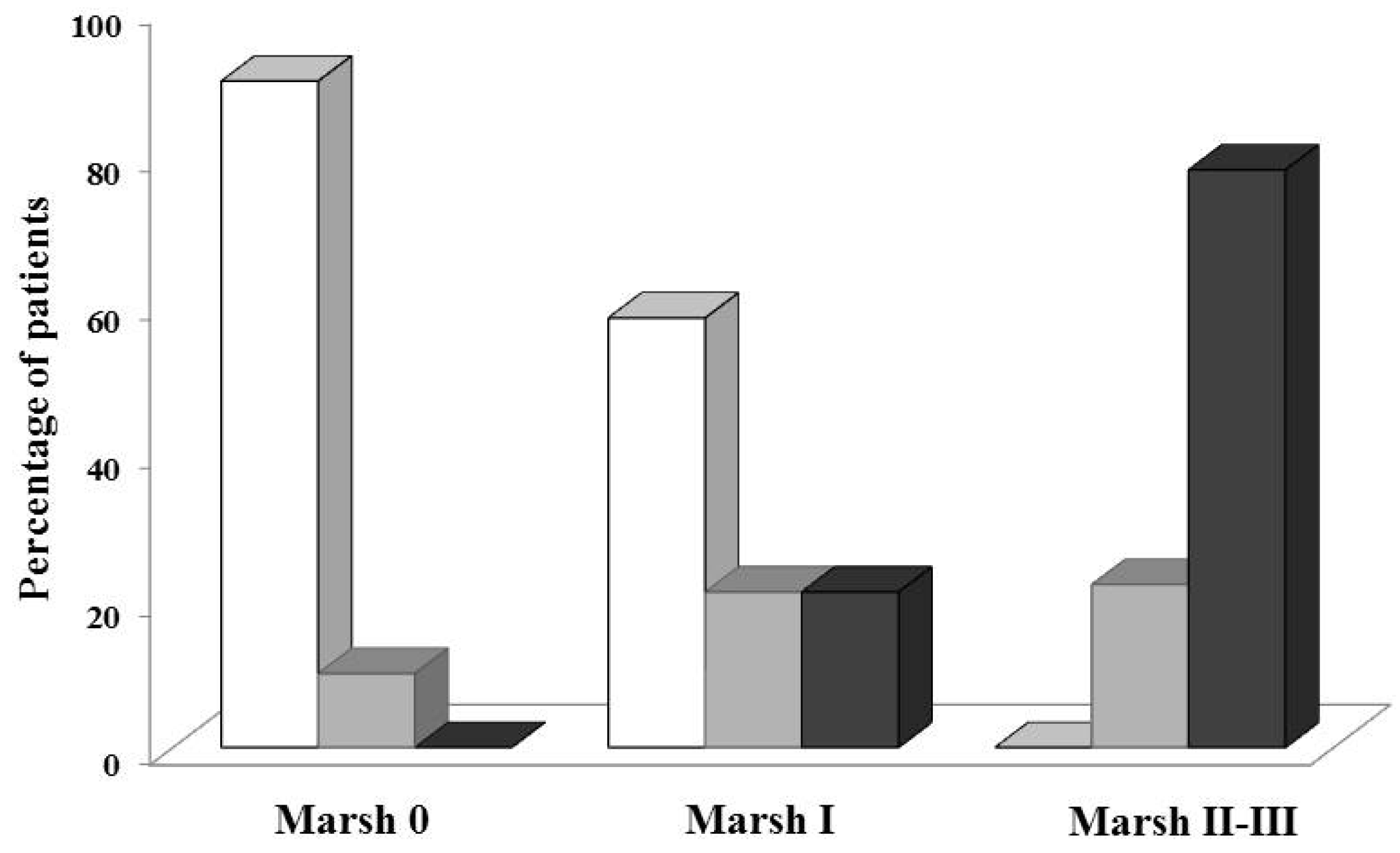
2.3. Biopsies
2.4. Serological Tests
2.5. Other Markers
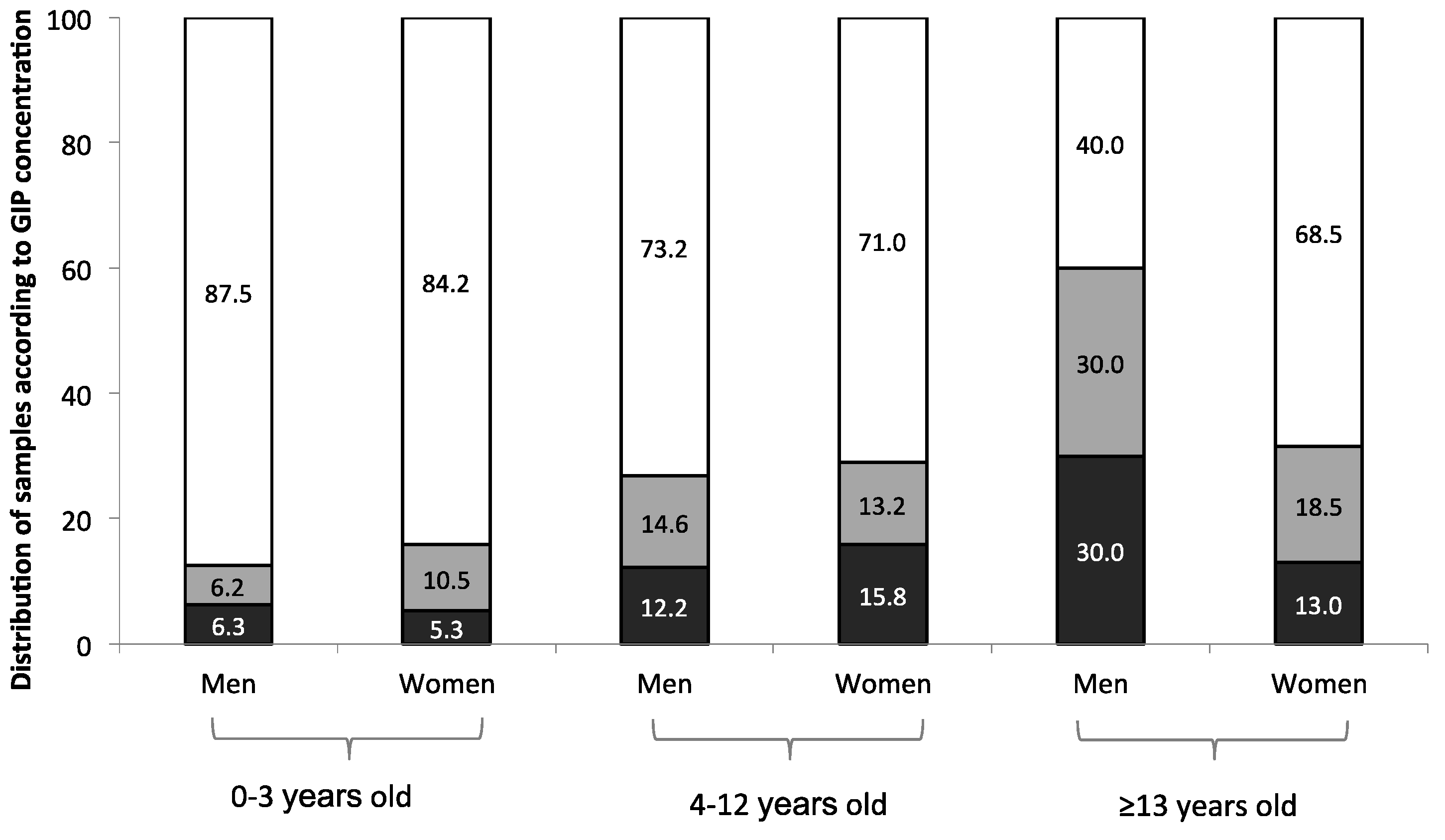
2.6. Detection of Gluten Immunogenic Peptides (GIP)
2.6.1. Feces
2.6.2. Urine
3. Conclusions and Future Directions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Jabri, B.; Sollid, L.M. Mechanisms of disease: Immunopathogenesis of celiac disease. Nat. Clin. Pract. Gastroenterol. Hepatol. 2006, 3, 516–525. [Google Scholar] [CrossRef] [PubMed]
- Makharia, G.K.; Catassi, C.; Goh, K.L.; Mulder, C.J. Celiac disease. Gastroenterol. Res. Pract. 2012, 2012, 758560. [Google Scholar] [CrossRef] [PubMed]
- Ludvigsson, J.F.; Leffler, D.A.; Bai, J.C.; Biagi, F.; Fasano, A.; Green, P.H.; Hadjivassiliou, M.; Kaukinen, K.; Kelly, C.P.; Leonard, J.N.; et al. The Oslo definitions for coeliac disease and related terms. Gut 2013, 62, 43–52. [Google Scholar] [CrossRef] [PubMed]
- Bernardo, D.; Peña, A.S. Developing strategies to improve the quality of life of patients with gluten intolerance in patients with and without coeliac disease. Eur. J. Intern. Med. 2012, 23, 6–8. [Google Scholar] [CrossRef] [PubMed]
- Hall, N.J.; Rubin, G.P.; Charnock, A. Intentional and inadvertent non-adherence in adult coeliac disease. A cross-sectional survey. Appetite 2013, 68, 56–62. [Google Scholar] [CrossRef] [PubMed]
- Ludvigsson, J.F.; Bai, J.C.; Biagi, F.; Card, T.R.; Ciacci, C.; Ciclitira, P.J.; Green, P.H.R.; Hadjivassiliou, M.; Holdoway, A.; van Heel, D.A.; et al. BSG Coeliac Disease Guidelines Development Group and British Society of Gastroenterology. Diagnosis and management of adult coeliac disease: Guidelines from the British Society of Gastroenterology. Gut 2014, 63, 1210–1228. [Google Scholar] [CrossRef] [PubMed]
- Bai, J.C.; Gonzalez, D.; Mautalen, C.; Mazure, R.; Pedreira, S.; Vazquez, H.; Smecuol, E.; Siccardi, A.; Cataldi, M.; Niveloni, S.; et al. Long-term eff ect of gluten restriction on bone mineral density of patients with coeliac disease. Aliment. Pharmacol. Ther. 1997, 11, 157–164. [Google Scholar] [CrossRef] [PubMed]
- Jafri, M.R.; Nordstrom, C.W.; Murray, J.A.; Van Dyke, C.T.; Dierkhising, R.A.; Zinsmeister, A.R.; Melton, L.J. Long-term fracture risk in patients with celiac disease: A population-based study in Olmsted County, Minnesota. Dig. Dis. Sci. 2008, 53, 964–971. [Google Scholar] [CrossRef] [PubMed]
- Sanchez, M.I.; Mohaidle, A.; Baistrocchi, A.; Matoso, D.; Vázquez, H.; González, A.; Mazure, R.; Maffei, E.; Ferrari, G.; Smecuol, E.; et al. Risk of fracture in celiac disease: Gender, dietary compliance, or both? World J. Gastroenterol. 2011, 17, 3035–3042. [Google Scholar] [CrossRef] [PubMed]
- Dickey, W. Low serum vitamin B12 is common in coeliac disease and is not due to autoimmune gastritis. Eur. J. Gastroenterol. Hepatol. 2002, 14, 425–427. [Google Scholar] [CrossRef] [PubMed]
- Halfdanarson, T.R.; Kumar, N.; Hogan, W.J.; Murray, J.A. Copper deficiency in celiac disease. J. Clin. Gastroenterol. 2009, 43, 162–164. [Google Scholar] [CrossRef] [PubMed]
- Codex Alimentarius, International Food Standars. Standard for Foods for Special Dietary Use for Persons Intolerant to Gluten CODEX STAN 118-1979. Available online: http://www.codexalimentarius.net (accessed on 2 January 2017).
- Peña, A.; Bernardo, D. Immunogenetic pathogenesis of celiac disease and non-celiac gluten sensitivity. Curr. Gastroenterol. Rep. 2016, 18, 36. [Google Scholar]
- See, J.A.; Kaukinen, K.; Makharia, G.K.; Gibson, P.R.; Murray, J.A. Practical insights into gluten-free diets. Nat. Rev. Gastroenterol. Hepatol. 2015, 12, 580–591. [Google Scholar] [CrossRef] [PubMed]
- Vriezinga, S.L.; Schweizer, J.J.; Koning, F.; Mearin, M.L. Coeliac disease and gluten-related disorders in childhood. Nat. Rev. Gastroenterol. Hepatol. 2015, 12, 527–536. [Google Scholar] [CrossRef] [PubMed]
- Ciacci, C.; Cirillo, M.; Cavallaro, R.; CiacMazzaccaci, G. Long-term follow-up of celiac adults on gluten-free diet: Prevalence and correlates of intestinal damage. Digestion 2002, 66, 178–185. [Google Scholar] [CrossRef] [PubMed]
- Högberg, L.; Grodzinsky, E.; Stenhammar, L. Better dietary compliance in patients with coeliac disease diagnosed in early childhood. Scand. J. Gastroenterol. 2003, 38, 751–754. [Google Scholar] [PubMed]
- Pietzak, M.M. Follow-up of patients with celiac disease: Achieving compliance with treatment. Gastroenterology 2005, 128, 135–141. [Google Scholar] [CrossRef]
- Herman, M.L.; Rubio-Tapia, A.; Lahr, B.D.; Larson, J.J.; Dyke, V.C.T.; Murray, J.A. Patients with celiac disease are not followed up adequately. Clin. Gastroenterol. Hepatol. 2012, 10, 893–899. [Google Scholar] [CrossRef] [PubMed]
- Ventura, A.; Magazzù, G.; Greco, L. Duration of exposure to gluten and risk for autoimmune disorders in patients with celiac disease. SIGEP study group for autoimmune disorders in celiac disease. Gastroenterology 1999, 117, 297–303. [Google Scholar] [CrossRef] [PubMed]
- Corrao, G.; Corazza, G.R.; Bagnardi, V.; Brusco, G.; Ciacci, C.; Cottone, M.; Guidetti, C.S.; Usai, P.; Cesari, P.; Pelli, M.A.; et al. Mortality in patients with coeliac disease and their relatives: A cohort study. Lancet 2001, 358, 356–361. [Google Scholar] [CrossRef]
- Rampertab, S.D.; Fleischauer, A.; Neugut, A.I.; Green, P.H.R. Risk of duodenal adenoma in celiac disease. Scand. J. Gastroenterol. 2003, 38, 831–833. [Google Scholar] [PubMed]
- Ludvigsson, J.F.; Montgomery, S.M.; Ekbom, A. Celiac disease and risk of adverse fetal outcome: A population-based cohort study. Gastroenterology 2005, 129, 454–463. [Google Scholar] [CrossRef] [PubMed]
- Khashan, A.S.; Henriksen, T.B.; McNamee, R.; Mortensen, P.B.; McCarthy, F.P.; Kenny, L.C. Parental celiac disease and offspring sex ratio. Epidemiology 2010, 21, 913–914. [Google Scholar] [CrossRef] [PubMed]
- Lebwohl, B.; Granath, F.; Ekbom, A.; Montgomery, S.M.; Murray, J.A.; Rubio-Tapia, A.; Green, P.H.; Ludvigsson, J.F. Mucosal healing and mortality in coeliac disease. Aliment. Pharmacol. Ther. 2013, 37, 332–339. [Google Scholar] [CrossRef] [PubMed]
- Silano, M.; Volta, U.; De Vincenzi, A.; Dessì, M.; De Vincenzi, M. Collaborating Centers of the Italian Registry of the Complications of Coeliac Disease. Effect of a gluten-free diet on the risk of enteropathy-associated T-cell lymphoma in celiac disease. Digest Dis. Sci. 2008, 53, 972–976. [Google Scholar] [CrossRef] [PubMed]
- Olén, O.; Askling, J.; Ludvigsson, J.F.; Hildebrand, H.; Ekbom, A.; Smedby, K.E. Coeliac disease characteristics, compliance to a gluten free diet and risk of lymphoma by subtype. Digest Liver Dis. 2011, 43, 862–868. [Google Scholar] [CrossRef] [PubMed]
- Leffler, D.A.; Dennis, M.; Hyett, B.; Kelly, E.; Schuppan, D.; Kelly, C.P. Etiologies and predictors of diagnosis in nonresponsive celiac disease. Clin. Gastroenterol. Hepatol. 2007, 5, 445–450. [Google Scholar] [CrossRef] [PubMed]
- Zuidmeer, L.; Goldhahn, K.; Rona, R.J.; Gislason, D.; Madsen, C.; Summers, C.; Sodergren, E.; Dahlstrom, J.; Lindner, T.; Sigurdardottir, S.T.; et al. The prevalence of plant food allergies: A systematic review. J. Allergy Clin. Immunol. 2008, 121, 1210–1218. [Google Scholar] [CrossRef] [PubMed]
- Sapone, A.; Bai, J.C.; Ciacci, C.; Dolinsek, J.; Green, P.H.; Hadjivassiliou, M.; Kaukinen, K.; Rostami, K.; Sanders, D.S.; Schumann, M.; et al. Spectrum of gluten-related disorders: Consensus on new nomenclature and classification. BMC Med. 2012, 7, 10–13. [Google Scholar] [CrossRef] [PubMed]
- Volta, U.; Caio, G.; De Giorgio, R.; Henriksen, C.; Skodje, G.; Lundin, K.E. Non-celiac gluten sensitivity: A work-in-progress entity in the spectrum of wheat-related disorders. Best Pract. Res. Clin. Gastroenterol. 2015, 29, 477–491. [Google Scholar] [CrossRef] [PubMed]
- Bai, J.C.; Fried, M.; Corazza, G.R.; Schuppan, D.; Farthing, M.; Catassi, C.; Greco, L.; Cohen, H.; Ciacci, C.; Eliakim, R.; et al. World Gastroenterology Organisation Global Guidelines on Celiac Disease. J. Clin. Gastroenterol. 2013, 47, 121–126. [Google Scholar] [CrossRef] [PubMed]
- Sainsbury, A.; Sanders, D.S.; Ford, A.C. Prevalence of irritable bowel syndrome-type symptoms in patients with celiac disease: A meta-analysis. Clin. Gastroenterol. Hepatol. 2013, 11, 359–365. [Google Scholar] [CrossRef] [PubMed]
- Sharkey, L.M.; Corbett, G.; Currie E Lee, J.; Sweeney, N.; Woodward, J.M. Optimising delivery of care in coeliac disease -comparison of the benefits of repeat biopsy and serological follow-up. Aliment. Pharmacol. Ther. 2013, 38, 1278–1291. [Google Scholar] [CrossRef] [PubMed]
- Lähdeaho, M.L.; Mäki, M.; Laurila, K.; Huhtala, H.; Kaukinen, K. Small-bowel mucosal changes and antibody responses after low- and moderate-dose gluten challenge in celiac disease. BMC Gastroenterol. 2011, 11, 129. [Google Scholar] [CrossRef] [PubMed]
- Ciacci, C.; D’Agate, C.; De Rosa, A.; Franzese, C.; Errichiello, S.; Gasperi, V.; Pardi, A.; Quagliata, D.; Visentini, S.; Greco, L. Self-rated quality of life in celiac disease. Dig. Dis. Sci. 2003, 48, 2216–2220. [Google Scholar] [CrossRef] [PubMed]
- Edwards George, J.B.; Leffler, D.A.; Dennis, M.D.; Franko, D.L.; Blom-Hoffman, J.; Kelly, C.P. Psychological correlates of gluten-free diet adherence in adults with celiac disease. J. Clin. Gastroenterol. 2009, 43, 301–306. [Google Scholar] [CrossRef] [PubMed]
- Hopman, E.G.; Koopman, H.M.; Wit, J.M.; Mearin, M.L. Dietary compliance and health-related quality of life in patients with coeliac disease. Eur. J. Gastroenterol. Hepatol. 2009, 21, 1056–1061. [Google Scholar] [CrossRef] [PubMed]
- Leffler, D.A.; Dennis, M.; Edwards George, J.B.; Jamma, S.; Magge, S.; Cook, E.F.; Schuppan, D.; Kelly, C.P. A simple validated gluten-free diet adherence survey for adults with celiac disease. Clin. Gastroenterol. Hepatol. 2009, 7, 530–536. [Google Scholar] [CrossRef]
- Chauhan, J.C.; Kumar, P.; Dutta, A.K.; Basu, S.; Kumar, A. Assessment of dietary compliance to gluten free diet and psychosocial problems in Indian children with celiac disease. Indian J. Pediatr. 2010, 77, 649–654. [Google Scholar] [CrossRef] [PubMed]
- Sainsbury, K.; Mullan, B. Measuring beliefs about gluten free diet adherence in adult coeliac disease using the theory of planned behaviour. Appetite 2011, 56, 476–483. [Google Scholar] [CrossRef] [PubMed]
- Leffler, D.; Schuppan, D.; Pallav, K.; Najarian, R.; Goldsmith, J.D.; Hansen, J.; Kabbani, T.; Dennis, M.; Kelly, C.P. Kinetics of the histological, serological and symptomatic responses to gluten challenge in adults with coeliac disease. Gut 2013, 62, 996–1004. [Google Scholar] [CrossRef] [PubMed]
- Comino, I.; Fernández-Bañares, F.; Esteve, M.; Ortigosa, L.; Castillejo, G.; Fambuena, B.; Ribes-Koninckx, C.; Sierra, C.; Rodríguez-Herrera, A.; Salazar, J.C.; et al. Fecal gluten peptides reveal limitations of serological tests and food questionnaires for monitoring gluten-free diet in celiac disease patients. Am. J. Gastroenterol. 2016, 111, 1456–1465. [Google Scholar] [CrossRef] [PubMed]
- Husby, S.; Koletzko, S.; Korponay-Szabó, I.R.; Mearin, M.L.; Phillips, A.; Shamir, R.; Troncone, R.; Giersiepen, K.; Branski, D.; Catassi, C.; et al. ESPGHAN Working Group on Coeliac Disease Diagnosis; ESPGHAN Gastroenterology Committee; European Society for Pediatric Gastroenterology, Hepatology, and Nutrition. European society for pediatric gastroenterology, hepatology, and nutrition guidelines for the diagnosis of coeliac disease. J. Pediatr. Gastroenterol. Nutr. 2012, 54, 136–160. [Google Scholar] [PubMed]
- Marsh, M.N. Gluten, major histocompatibility complex, and the small intestine. A molecular and immunobiologic approach to the spectrum of gluten sensitivity (‘celiac sprue’). Gastroenterology 1992, 102, 330–354. [Google Scholar] [CrossRef]
- Oberhuber, G.; Granditsch, G.; Vogelsang, H. The histopathology of coeliac disease: Time for a standardised report scheme for pathologist. Eur. J. Gastroenterol. Hepatol. 1999, 11, 1185–1194. [Google Scholar] [CrossRef] [PubMed]
- Corazza, G.R.; Villanacci, V. Coeliac disease some considerations on the histological diagnosis. J. Clin. Pathol. 2005, 58, 573–574. [Google Scholar] [CrossRef] [PubMed]
- Ensari, A. Gluten-sensitive enteropathy (celiac disease): Controversies in diagnosis and classification. Arch. Pathol. Lab. Med. 2010, 134, 826–836. [Google Scholar] [PubMed]
- Villanacci, V. What is the best histopathological classification for celiac disease? Does it matter? A letter of comment to the review of Amado Salvador Pena; a new proposal. Gastroenterol. Hepatol. Bed Bench 2015, 8, 306–308. [Google Scholar] [PubMed]
- Ensari, A. Coeliac disease: To classify or not to classify-that is the question! Gastroenterol. Hepatol. Bed Bench 2016, 9, 73–74. [Google Scholar] [PubMed]
- Villanacci, V. The histological classification of biopsy in celiac disease: Time for a change. Dig. Liver Dis. 2015, 47, 2–3. [Google Scholar] [CrossRef] [PubMed]
- Peña, A.S. What is the best histopathological classification for celiac disease? Does it matter? Gastroenterol. Hepatol. Bed Bench 2015, 8, 239–243. [Google Scholar] [PubMed]
- Taavela, J.; Kurppa, K.; Collin, P.; Lähdeaho, M.L.; Salmi, T.; Saavalainen, P.; Haimila, K.; Huhtala, H.; Laurila, K.; Sievänen, H.; et al. Degree of damage to the small bowel and serum antibody titers correlate with clinical presentation of patients with celiac disease. Clin. Gastroenterol. Hepatol. 2013, 11, 166–171. [Google Scholar] [CrossRef] [PubMed]
- Peña, A.S. Counting Intraepithelial Lymphocytes. Immunohistochemistry and flow cytometer are necessary new steps in the diagnosis of celiac disease. Int. J. Celiac Dis. 2016, 4, 7–8. [Google Scholar] [CrossRef]
- De Andres, A.; Camarero, C.; Roy, G. Distal duodenum versus duodenal bulb: Intraepithelial lymphocytes have something to say in celiac disease diagnosis. Dig. Dis. Sci. 2015, 60, 1004–1009. [Google Scholar] [CrossRef] [PubMed]
- Sanchez-Munoz, L.B.; Santon, A.; Cano, A.; Lopez, A.; Almeida, J.; Orfao, A.; Escribano, L.; Roy, G. Flow cytometric analysis of intestinal intraepithelial lymphocytes in the diagnosis of refractory celiac sprue. Eur. J. Gastroenterol. Hepatol. 2008, 20, 478–487. [Google Scholar] [CrossRef] [PubMed]
- Antonioli, D. Coeliac disease: A progress report. Mod. Pathol. 2003, 6, 342–346. [Google Scholar] [CrossRef] [PubMed]
- Siriweera, E.H.; Qi, Z.; Yong, J.L.C. Validity of intraepithelial lymphocyte count in the diagnosis of celiac disease: A histopathological study. Int. J. Celiac Dis. 2015, 3, 156–158. [Google Scholar] [CrossRef]
- Buchanan, R.; Dennis, S.; Gendel, S.; Acheson, D.; Assimon, S.A.; Beru, N.; Bolger, P.; Carlson, D.; Carvajal, R.; Copp, C.; et al. Approaches to establish thresholds for major food allergens and for gluten in food. J. Food Prot. 2008, 71, 1043–1088. [Google Scholar] [PubMed]
- Tuire, I.; Marja-Leena, L.; Teea, S.; Katri, H.; Jukka, P.; Päivi, S.; Heini, H.; Markku, M.; Pekka, C.; Katri, K. Persistent duodenal intraepithelial lymphocytosis despite a long-term strict gluten-free diet in celiac disease. Am. J. Gastroenterol. 2012, 107, 1563–1569. [Google Scholar] [CrossRef] [PubMed]
- Rashid, M. Serologic testing in celiac disease. Can. Fam. Phys. 2016, 62, 38–43. [Google Scholar]
- Rubio-Tapia, A.; Hill, I.D.; Kelly, C.P.; Calderwood, A.H.; Murray, J.A. American College of Gastroenterology. ACG clinical guidelines: Diagnosis and management of celiac disease. Am. J. Gastroenterol. 2013, 108, 656–676. [Google Scholar] [CrossRef] [PubMed]
- Berger, E.; Buergin-Wolff, A.; Freudenberg, E. Diagnostic value of the demonstration of gliadin antibodies in celiac disease. Klin. Wochenschr. 1964, 42, 788–790. [Google Scholar] [CrossRef] [PubMed]
- Adriaanse, M.; Leffler, D.A. Serum markers in the clinical management of celiac disease. Dig. Dis. 2015, 33, 236–243. [Google Scholar] [CrossRef] [PubMed]
- Kaukinen, K.; Peräaho, M.; Lindfors, K.; Partanen, J.; Woolley, N.; Pikkarainen, P.; Karvonen, A.L.; Laasanen, T.; Sievänen, H.; Mäki, M.; et al. Persistent small bowel mucosal villous atrophy without symptoms in coeliac disease. Aliment. Pharmacol. Ther. 2007, 25, 1237–1245. [Google Scholar] [CrossRef] [PubMed]
- Kaukinen, K.; Sulkanen, S.; Mäki, M.; Collin, P. IgA-class transglutaminase antibodies in evaluating the efficacy of gluten-free diet in coeliac disease. Eur. J. Gastroenterol. Hepatol. 2002, 14, 311–315. [Google Scholar] [CrossRef] [PubMed]
- Tursi, A.; Brandimarte, G.; Giorgetti, G.M. Lack of usefulness of anti-transglutaminase antibodies in assessing histologic recovery after gluten-free diet in celiac disease. J. Clin. Gastroenterol. 2003, 37, 387–391. [Google Scholar] [CrossRef] [PubMed]
- Bardella, M.T.; Velio, P.M.; Cesana, B.M.; Prampolini, L.; Casella, G.; Di Bella, C.; Lanzini, A.; Gambarotti, M.; Bassotti, G.; Villanacci, V. Coeliac disease: A histological follow-up study. Histopathology 2007, 50, 465–471. [Google Scholar] [CrossRef] [PubMed]
- Lanzini, A.; Lanzarotto, F.; Villanacci, V.; Mora, A.; Bertolazzi, S.; Turini, D.; Carella, G.; Malagoli, A.; Ferrante, G.; Cesana, B.M.; et al. Complete recovery of intestinal mucosa occurs very rarely in adult coeliac patients despite adherence to gluten-free diet. Aliment. Pharmacol. Ther. 2009, 29, 1299–1308. [Google Scholar] [CrossRef] [PubMed]
- Rubio-Tapia, A.; Rahim, M.W.; See, J.A.; Lahr, B.D.; Wu, T.T.; Murray, J.A. Mucosal recovery and mortality in adults with celiac disease after treatment with a gluten-free diet. Am. J. Gastroenterol. 2010, 105, 1412–1420. [Google Scholar] [CrossRef] [PubMed]
- Duerksen, D.R.; Wilhelm-Boyles, C.; Parry, D.M. Intestinal permeability in long-term follow-up of patients with celiac disease on a gluten-free diet. Dig. Dis. Sci. 2005, 50, 785–790. [Google Scholar] [CrossRef] [PubMed]
- Ertekin, V.; Selimoğlu, M.A.; Turgut, A.; Bakan, N. Fecal calprotectin concentration in celiac disease. J. Clin. Gastroenterol. 2010, 44, 544–546. [Google Scholar] [CrossRef] [PubMed]
- Planas, R.; Pujol-Autonell, I.; Ruiz, E.; Montraveta, M.; Cabre, E.; Lucas-Martin, A.; Pujol-Borrell, R.; Martinez-Caceres, E.; Vives-Pi, M. Regenerating gene Iα is a biomarker for diagnosis and monitoring of celiac disease: A preliminary study. Transl. Res. 2011, 158, 140–145. [Google Scholar] [CrossRef] [PubMed]
- Lind, M.V.; Madsen, M.L.; Rumessen, J.J.; Vestergaard, H.; Gøbel, R.J.; Hansen, T.; Lauritzen, L.; Pedersen, O.B.; Kristensen, M.; Ross, A.B. Plasma alkylresorcinols reflect gluten intake and distinguish between gluten-rich and gluten-poor diets in a population at risk of metabolic syndrome. J. Nutr. 2016, 146, 1991–1998. [Google Scholar] [CrossRef] [PubMed]
- Balamtekın, N.; Baysoy, G.; Uslu, N.; Orhan, D.; Akçören, Z.; Özen, H.; Gürakan, F.; Saltik-Temızel, İ.N.; Yüce, A. Fecal calprotectin concentration is increased in children with celiac disease: Relation with histopathological findings. Turk. J. Gastroenterol. 2012, 23, 503–508. [Google Scholar] [CrossRef] [PubMed]
- Capone, P.; Rispo, A.; Imperatore, N.; Caporaso, N.; Tortora, N. Fecal calprotectin in coeliac disease. World J. Gastroenterol. 2014, 20, 611–612. [Google Scholar] [CrossRef] [PubMed]
- Duerksen, D.R.; Wilhelm-Boyles, C.; Veitch, R.; Kryszak, D.; Parry, D.M. A comparison of antibody testing, permeability testing, and zonulin levels with small-bowel biopsy in celiac disease patients on a gluten-free diet. Dig. Dis. Sci. 2010, 55, 1026–1031. [Google Scholar] [CrossRef] [PubMed]
- Laass, M.W.; Röber, N.; Range, U.; Noß, L.; Roggenbuck, D.; Conrad, K. Loss and gain of tolerance to pancreatic glycoprotein 2 in celiac disease. PLoS ONE 2015, 10, e0128104. [Google Scholar] [CrossRef] [PubMed]
- Roggenbuck, D.; Vermeire, S.; Hoffman, I.; Reinhold, D.; Schierack, P.; Goihl, A.; von Arnim, U.; De Hertogh, G.; Polymeros, D.; Bogdanos, D.P.; et al. Evidence of Crohn’s disease-related anti-glycoprotein 2 antibodies in patients with celiac disease. Clin. Chem. Lab. Med. 2015, 53, 1349–1357. [Google Scholar] [CrossRef] [PubMed]
- Ryan, D.; Newnham, E.D.; Prenzler, P.D.; Gibson, P.R. Metabolomics as a tool for diagnosis and monitoring in coeliac disease. Metabolomics 2015, 11, 980–990. [Google Scholar] [CrossRef]
- Bertini, I.; Calalbro, A.; De Carli, V.; Luchinat, C.; Nepi, S.; Porfirio, B.; Renzi, D.; Saccenti, E.; Tenori, L. The metabonomic signature of celiac disease. J. Proteome Res. 2009, 8, 170–177. [Google Scholar] [CrossRef] [PubMed]
- Bernini, P.; Bertini, I.; Calabro, A.; la Marca, G.; Lami, G.; Luchinat, C.; Renzi, D.; Tenori, L. Are patients with potential celiac disease really potential? The answer of metabonomics. J. Proteome Res. 2011, 10, 714–721. [Google Scholar] [CrossRef] [PubMed]
- Tursi, A.; Brandimarte, G.; Giorgetti, G.M.; Elisei, W.; Inchingolo, C.D.; Monardo, E.; Aiello, F. Endoscopic and histological findings in the duodenum of adults with celiac disease before and after changing to a gluten-free diet: A 2-year prospective study. Endoscopy 2006, 38, 702–707. [Google Scholar] [CrossRef] [PubMed]
- Biagi, F.; Campanella, J.; Martucci, S.; Pezzimenti, D.; Ciclitira, P.J.; Ellis, H.J.; Corazza, G.R. A milligram of gluten a day keeps the mucosal recovery away: A case report. Nutr. Rev. 2004, 62, 360–363. [Google Scholar] [CrossRef] [PubMed]
- Rashtak, S.; Ettore, M.W.; Homburger, H.A.; Murray, J.A. Comparative usefulness of deamidated gliadin antibodies in the diagnosis of celiac disease. Clin. Gastroenterol. Hepatol. 2008, 6, 426–432. [Google Scholar] [CrossRef] [PubMed]
- Dipper, C.R.; Maitra, S.; Thomas, R.; Lamb, C.A.; McLean-Tooke, A.P.; Ward, R.; Smith, D.; Spickett, G.; Mansfield, J.C. Anti-tissue transglutaminase antibodies in the follow-up of adult coeliac disease. Aliment. Pharmacol. Ther. 2009, 30, 236–244. [Google Scholar] [CrossRef] [PubMed]
- Vives-Pi, M.; Takasawa, S.; Pujol-Autonell, I.; Planas, R.; Cabre, E.; Ojanguren, I.; Montraveta, M.; Santos, A.L.; Ruiz-Ortiz, E. Biomarkers for diagnosis and monitoring of celiac disease. J. Clin. Gastroenterol. 2013, 47, 308–313. [Google Scholar] [CrossRef] [PubMed]
- Vallejo-Diez, S.; Bernardo, D.; Moreno, M.L.; Muñoz-Suano, A.; Fernández-Salazar, L.; Calvo, C.; Sousa, C.; Garrote, J.A.; Cebolla, A.; Arranz, E. Detection of specific IgA antibodies against a novel deamidated 8-Mer gliadin peptide in blood plasma samples from celiac patients. PLoS ONE 2013, 8, e80982. [Google Scholar] [CrossRef] [PubMed]
- Hischenhuber, C.; Crevel, R.; Jarry, B.; Mäki, M.; Moneret-Vautrin, D.A.; Romano, A.; Troncone, R.; Ward, R. Review article: Safe amounts of gluten for patients with wheat allergy or coeliac disease. Aliment. Pharmacol. Ther. 2006, 23, 559–575. [Google Scholar] [CrossRef] [PubMed]
- Akobeng, A.K.; Thomas, A.G. Systematic review: Tolerable amount of gluten for people with coeliac disease. Aliment. Pharmacol. Ther. 2008, 27, 1044–1052. [Google Scholar] [CrossRef] [PubMed]
- Shan, L.; Molberg, Ø.; Parrot, I.; Hausch, F.; Filiz, F.; Gray, G.M.; Sollid, L.M.; Khosla, C. Structural basis for gluten intolerance in celiac sprue. Science 2002, 297, 2275–2279. [Google Scholar] [CrossRef] [PubMed]
- Morón, B.; Bethune, M.T.; Comino, I.; Manyani, H.; Ferragud, M.; López, M.C.; Cebolla, A.; Khosla, C.; Sousa, C. Toward the assessment of food toxicity for celiac patients: Characterization of monoclonal antibodies to a main immunogenic gluten peptide. PLoS ONE 2008, 3, e2294. [Google Scholar] [CrossRef] [PubMed]
- Morón, B.; Cebolla, A.; Manyani, H.; Alvarez-Maqueda, M.; Megías, M.; del Thomas, M.C.; López, M.C.; Sousa, C. Sensitive detection of cereal fractions that are toxic to celiac disease patients by using monoclonal antibodies to a main immunogenic wheat peptide. Am. J. Clin. Nutr. 2008, 87, 405–414. [Google Scholar] [PubMed]
- Comino, I.; Real, A.; De Moreno, M.L.; Montes, R.; Cebolla, A.; Sousa, C. Immunological determination of gliadin 33-mer equivalent peptides in beers as a specific and practical analytical method to assess safety for celiac patients. J. Sci. Food Agric. 2013, 93, 933–943. [Google Scholar] [CrossRef] [PubMed]
- Halbmayr-Jech, E.; Hammer, E.; Fielder, R.; Coutts, J.; Rogers, A.; Cornish, M. Characterization of G12 sandwich ELISA, a next-generation immunoassay for gluten toxicity. J. AOAC Int. 2012, 95, 372–376. [Google Scholar] [CrossRef] [PubMed]
- Real, A.; Comino, I.; Moreno Mde, L.; López-Casado, M.Á.; Lorite, P.; Torres, M.I.; Cebolla, Á.; Sousa, C. Identification and in vitro reactivity of celiac immunoactive peptides in an apparent gluten-free beer. PLoS ONE 2014, 9, e100917. [Google Scholar] [CrossRef] [PubMed]
- De Moreno, M.L.; Muñoz-Suano, A.; López-Casado, M.Á.; Torres, M.I.; Sousa, C.; Cebolla, Á. Selective capture of most celiac immunogenic peptides from hydrolyzed gluten proteins. Food Chem. 2016, 205, 36–42. [Google Scholar] [CrossRef] [PubMed]
- Comino, I.; Real, A.; Vivas, S.; Síglez, M.Á.; Caminero, A.; Nistal, E.; Casqueiro, J.; Rodríguez-Herrera, A.; Cebolla, A.; Sousa, C. Monitoring of gluten-free diet compliance in celiac patients by assessment of gliadin 33-mer equivalent epitopes in feces. Am. J. Clin. Nutr. 2012, 95, 670–677. [Google Scholar] [CrossRef] [PubMed]
- Auricchio, S. An innovative approach to measure compliance to a gluten-free diet. Am. J. Clin. Nutr. 2012, 95, 537–538. [Google Scholar] [CrossRef] [PubMed]
- Caminero, A.; Nistal, E.; Arias, L.; Vivas, S.; Comino, I.; Real, A.; Sousa, C.; de Morales, J.M.; Ferrero, M.A.; Rodríguez-Aparicio, L.B.; et al. A gluten metabolism study in healthy individuals shows the presence of faecal glutenasic activity. Eur. J. Nutr. 2012, 51, 293–299. [Google Scholar] [CrossRef] [PubMed]
- Riestra, S. Enfermedades asociadas. In Libro Blanco de la Enfermedad Celiaca; Polanco, I., Ed.; España: Madrid, Spain, 2008; pp. 41–49. [Google Scholar]
- Ludvigsson, J.F.; Green, P.H. Clinical management of coeliac disease. J. Intern. Med. 2011, 269, 560–571. [Google Scholar] [CrossRef] [PubMed]
- Moreno, M.L.; Cebolla, Á.; Muñoz-Suano, A.; Carrillo-Carrion, C.; Comino, I.; Pizarro, Á.; León, F.; Rodríguez-Herrera, A.; Sousa, C. Detection of gluten immunogenic peptides in the urine of patients with coeliac disease reveals transgressions in the gluten-free diet and incomplete mucosal healing. Gut 2015. [Google Scholar] [CrossRef] [PubMed]
- Matoori, S.; Fuhrmann, G.; Leroux, J.C. Celiac disease: A challenging disease for pharmaceutical scientists. Pharm. Res. 2013, 30, 619–626. [Google Scholar] [CrossRef] [PubMed]
- Tio, M.; Cox, M.R.; Eslick, G.D. Meta-analysis: Coeliac disease and the risk of all-cause mortality, any malignancy and lymphoid malignancy. Aliment. Pharmacol. Ther. 2012, 35, 540–551. [Google Scholar] [CrossRef] [PubMed]
- Stoven, S.; Murray, J.A.; Marietta, E. Celiac disease: Advances in treatment via gluten modification. Clin. Gastroenterol. Hepatol. 2012, 10, 859–862. [Google Scholar] [CrossRef] [PubMed]
- Soler, M.; Estevez, M.C.; de Moreno, M.L.; Cebolla, A.; Lechuga, L.M. Label-free SPR detection of gluten peptides in urine for non-invasive celiac disease follow-up. Biosens. Bioelectron. 2016, 79, 158–164. [Google Scholar] [CrossRef] [PubMed]
© 2017 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Moreno, M.D.L.; Rodríguez-Herrera, A.; Sousa, C.; Comino, I. Biomarkers to Monitor Gluten-Free Diet Compliance in Celiac Patients. Nutrients 2017, 9, 46. https://doi.org/10.3390/nu9010046
Moreno MDL, Rodríguez-Herrera A, Sousa C, Comino I. Biomarkers to Monitor Gluten-Free Diet Compliance in Celiac Patients. Nutrients. 2017; 9(1):46. https://doi.org/10.3390/nu9010046
Chicago/Turabian StyleMoreno, María De Lourdes, Alfonso Rodríguez-Herrera, Carolina Sousa, and Isabel Comino. 2017. "Biomarkers to Monitor Gluten-Free Diet Compliance in Celiac Patients" Nutrients 9, no. 1: 46. https://doi.org/10.3390/nu9010046
APA StyleMoreno, M. D. L., Rodríguez-Herrera, A., Sousa, C., & Comino, I. (2017). Biomarkers to Monitor Gluten-Free Diet Compliance in Celiac Patients. Nutrients, 9(1), 46. https://doi.org/10.3390/nu9010046



