Inadequacy of Body Weight-Based Recommendations for Individual Protein Intake—Lessons from Body Composition Analysis
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Populations
2.1.1. Healthy Subjects
2.1.2. Patients with Liver Cirrhosis
2.1.3. Cancer Patients
2.2. Body Composition Analysis in Study Populations
2.2.1. Healthy Subjects
Magnetic Resonance Imaging (MRI)
Dual-Energy X-ray Absorptiometry (DXA)
Quantitative Magnetic Resonance Imaging (QMR)
2.2.2. Patients with Liver Cirrhosis
Bioelectrical Impedance (BIA)
Total Body Potassium (TBK)
2.2.3. Cancer Patients
Computer Tomography (CT)
Standardization of Lean Mass
Definition of Sarcopenia
2.3. Assessment of Resting Energy Expenditure (REE) in Study Populations
2.3.1. Healthy Subjects
2.3.2. Patients with Liver Cirrhosis
2.3.3. Cancer Patients
2.4. Calculation of Total Energy Expenditure (TEE)
Statistical Analysis
2.5. Calculations of Theoretical Protein Requirements
3. Results
3.1. Healthy and Clinical Populations Characteristics
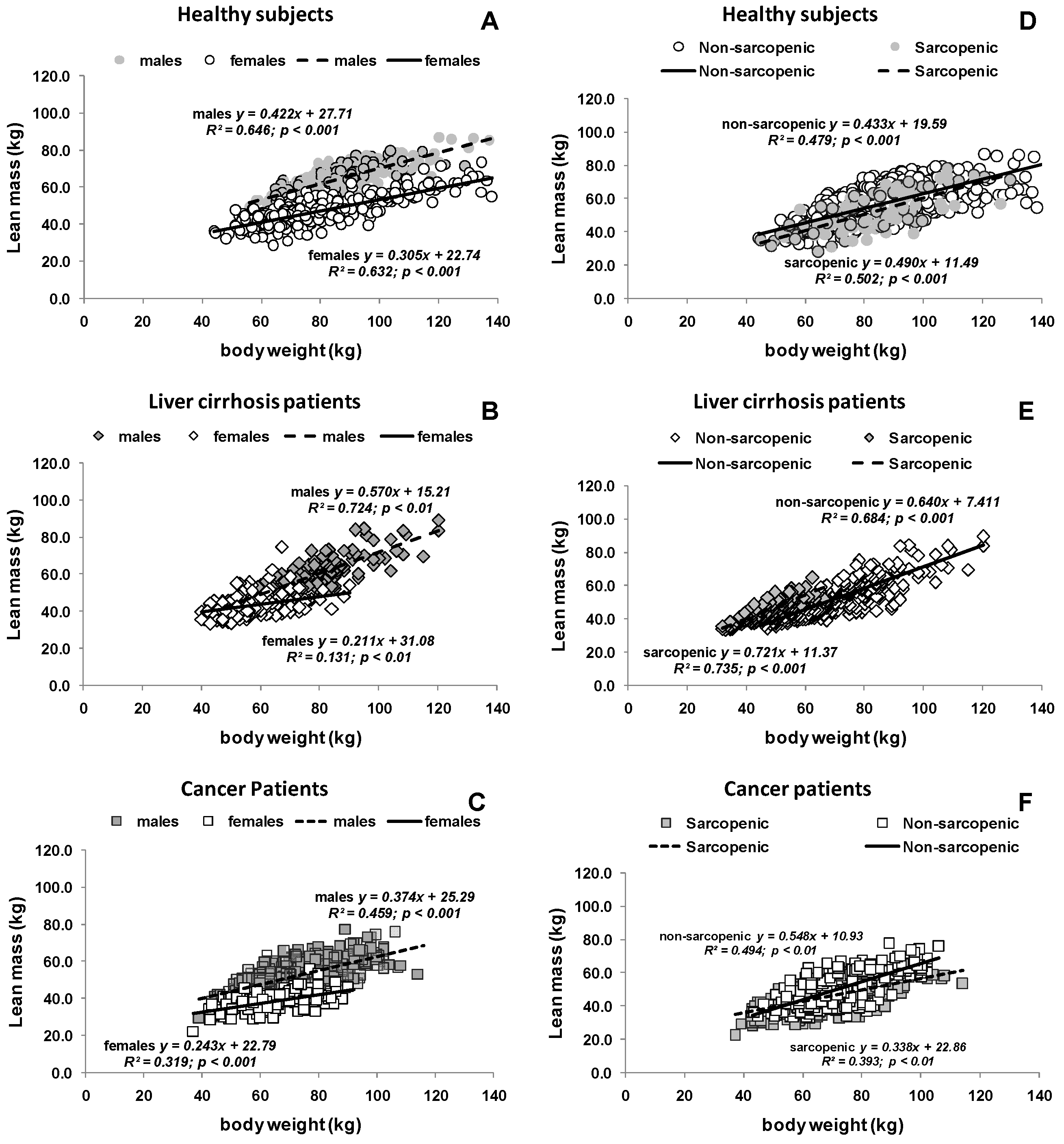
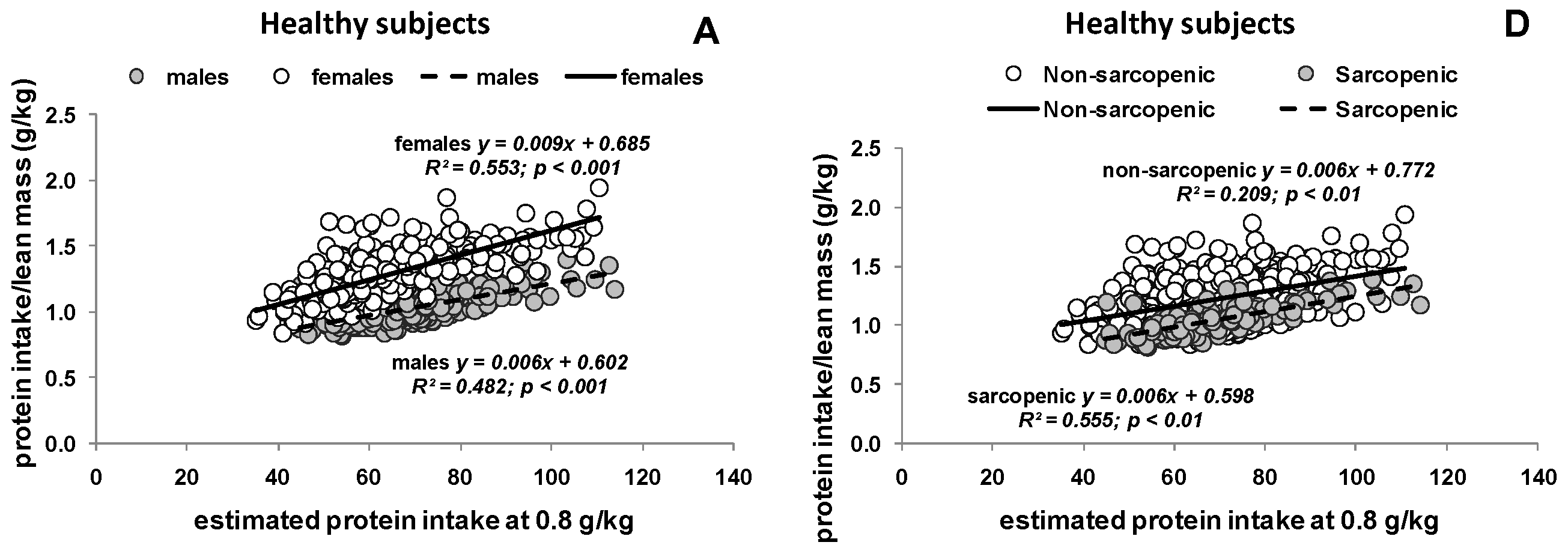
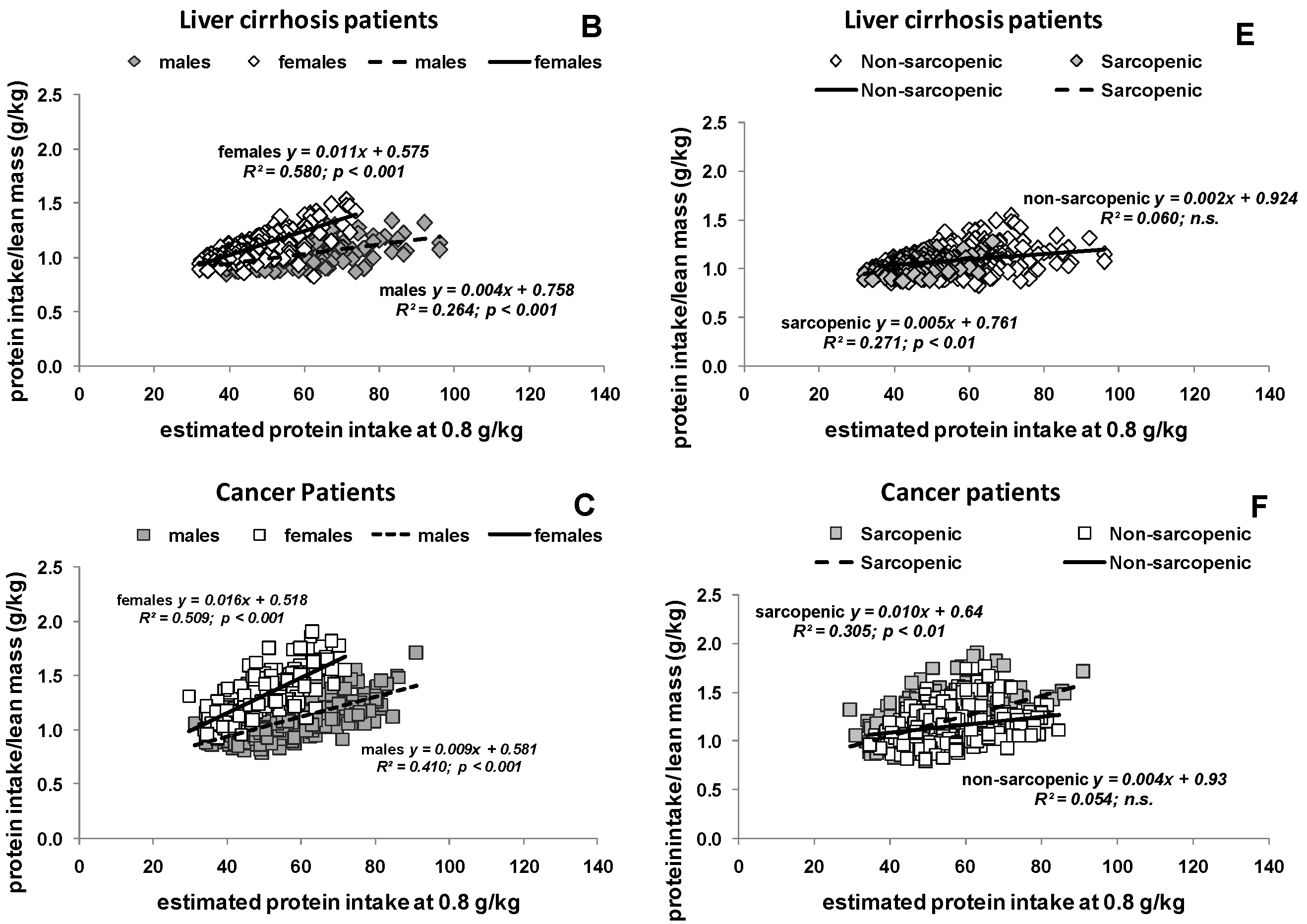
3.2. Estimated Protein Intake Based on either Body Weight or Body Composition
3.3. Potential Consequences of Variable Protein and Energy Recommendations
4. Discussion
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of interest
Abbreviations
| ASMI | Appendicular Skeletal Muscle Mass Index |
| BIA | Bioelectrical impedance |
| BMI | Body mass index |
| CR | Caloric restriction |
| CT | Computer Tomography |
| DXA | Dual energy X-ray absorptiometry |
| FFM | Fat free mass |
| FFMI | Fat free mass index |
| FM | Fat mass |
| FMI | Fat mass index |
| LM | Lean mass |
| LST | Lean soft tissue |
| MM | Muscle mass |
| MRI | Magnetic resonance imaging |
| OF | Overfeeding |
| REE | Resting energy expenditure |
| RF | Refeeding |
| TBK | Total body potassium |
| TBProTBK | Total body protein |
| TEE | Total energy expenditure |
References
- Medicine, I.O. Dietary Reference Intakes for Energy, Carbohydrate, Fiber, Fat, Fatty Acids, Cholesterol, Protein, and Amino Acids (Macronutrients); The National Academies Press: Washington, DC, USA, 2005; p. 1357. [Google Scholar]
- Houston, D.K.; Nicklas, B.J.; Ding, J.; Harris, T.B.; Tylavsky, F.A.; Newman, A.B.; Lee, J.S.; Sahyoun, N.R.; Visser, M.; Kritchevsky, S.B.; et al. Dietary protein intake is associated with lean mass change in older, community-dwelling adults: The health, aging, and body composition (health abc) study. Am. J. Clin. Nutr. 2008, 87, 150–155. [Google Scholar] [PubMed]
- Campbell, W.W.; Trappe, T.A.; Wolfe, R.R.; Evans, W.J. The recommended dietary allowance for protein may not be adequate for older people to maintain skeletal muscle. J. Gerontol. A Biol. Sci. Med. Sci. 2001, 56, M373–M380. [Google Scholar] [CrossRef] [PubMed]
- Argiles, J.M.; Busquets, S.; Stemmler, B.; Lopez-Soriano, F.J. Cachexia and sarcopenia: Mechanisms and potential targets for intervention. Curr. Opin. Pharmacol. 2015, 22, 100–106. [Google Scholar] [CrossRef] [PubMed]
- Kerstetter, J.E.; O’Brien, K.O.; Insogna, K.L. Low protein intake: The impact on calcium and bone homeostasis in humans. J. Nutr. 2003, 133, 855S–861S. [Google Scholar] [PubMed]
- Evans, W.J. Protein nutrition, exercise and aging. J. Am. Coll. Nutr. 2004, 23, 601S–609S. [Google Scholar] [CrossRef] [PubMed]
- Paddon-Jones, D.; Short, K.R.; Campbell, W.W.; Volpi, E.; Wolfe, R.R. Role of dietary protein in the sarcopenia of aging. Am. J. Clin. Nutr. 2008, 87, 1562S–1566S. [Google Scholar] [PubMed]
- Prado, C.M.; Heymsfield, S.B. Lean tissue imaging: A new era for nutritional assessment and intervention. J. Parenter. Enter. Nutr. 2014, 38, 940–953. [Google Scholar] [CrossRef] [PubMed]
- Prado, C.M.; Cushen, S.J.; Orsso, C.E.; Ryan, A.M. Sarcopenia and cachexia in the era of obesity: Clinical and nutritional impact. Proc. Nutr. Soc. 2016, 75, 188–198. [Google Scholar] [CrossRef] [PubMed]
- Prado, C.M.; Siervo, M.; Mire, E.; Heymsfield, S.B.; Stephan, B.C.; Broyles, S.; Smith, S.R.; Wells, J.C.; Katzmarzyk, P.T. A population-based approach to define body-composition phenotypes. Am. J. Clin. Nutr. 2014, 99, 1369–1377. [Google Scholar] [CrossRef] [PubMed]
- Anandavadivelan, P.; Brismar, T.B.; Nilsson, M.; Johar, A.M.; Martin, L. Sarcopenic obesity: A probable risk factor for dose limiting toxicity during neo-adjuvant chemotherapy in oesophageal cancer patients. Clin. Nutr. 2016, 35, 724–730. [Google Scholar] [CrossRef] [PubMed]
- Bosy-Westphal, A.; Schautz, B.; Lagerpusch, M.; Pourhassan, M.; Braun, W.; Goele, K.; Heller, M.; Gluer, C.C.; Muller, M.J. Effect of weight loss and regain on adipose tissue distribution, composition of lean mass and resting energy expenditure in young overweight and obese adults. Int. J. Obes. 2013, 37, 1371–1377. [Google Scholar] [CrossRef]
- Bosy-Westphal, A.; Schautz, B.; Later, W.; Kehayias, J.J.; Gallagher, D.; Muller, M.J. What makes a bia equation unique? Validity of eight-electrode multifrequency bia to estimate body composition in a healthy adult population. Eur. J. Clin. Nutr. 2013, 67, S14–S21. [Google Scholar] [CrossRef] [PubMed]
- Pourhassan, M.; Bosy-Westphal, A.; Schautz, B.; Braun, W.; Gluer, C.C.; Muller, M.J. Impact of body composition during weight change on resting energy expenditure and homeostasis model assessment index in overweight nonsmoking adults. Am. J. Clin. Nutr. 2014, 99, 779–791. [Google Scholar] [CrossRef] [PubMed]
- Lagerpusch, M.; Enderle, J.; Eggeling, B.; Braun, W.; Johannsen, M.; Pape, D.; Muller, M.J.; Bosy-Westphal, A. Carbohydrate quality and quantity affect glucose and lipid metabolism during weight regain in healthy men. J. Nutr. 2013, 143, 1593–1601. [Google Scholar] [CrossRef] [PubMed]
- Muller, M.J.; Enderle, J.; Pourhassan, M.; Braun, W.; Eggeling, B.; Lagerpusch, M.; Gluer, C.C.; Kehayias, J.J.; Kiosz, D.; Bosy-Westphal, A. Metabolic adaptation to caloric restriction and subsequent refeeding: The minnesota starvation experiment revisited. Am. J. Clin. Nutr. 2015, 102, 807–819. [Google Scholar] [CrossRef] [PubMed]
- Muller, M.J.; Bottcher, J.; Selberg, O.; Weselmann, S.; Boker, K.H.; Schwarze, M.; von zur Muhlen, A.; Manns, M.P. Hypermetabolism in clinically stable patients with liver cirrhosis. Am. J. Clin. Nutr. 1999, 69, 1194–1201. [Google Scholar] [PubMed]
- Crawford, J.; Prado, C.M.; Johnston, M.A.; Gralla, R.J.; Taylor, R.P.; Hancock, M.L.; Dalton, J.T. Study design and rationale for the phase 3 clinical development program of enobosarm, a selective androgen receptor modulator, for the prevention and treatment of muscle wasting in cancer patients (power trials). Curr. Oncol. Rep. 2016, 18, 37. [Google Scholar] [CrossRef] [PubMed]
- Snyder, W.S.; Cook, M.J.; Nasset, E.S.; Karhausen, L.R.; Howells, G.P.; Tipton, I.H. Report of the Task Group on Reference Man; Pergamon Press Oxford: Oxford, UK, 1975. [Google Scholar]
- Heymsfield, S.B.; Smith, R.; Aulet, M.; Bensen, B.; Lichtman, S.; Wang, J.; Pierson, R.N. Appendicular skeletal muscle mass: Measurement by dual-photon absorptiometry. Am. J. Clin. Nutr. 1990, 52, 214–218. [Google Scholar] [PubMed]
- Bosy-Westphal, A.; Muller, M.J. Assessment of fat and lean mass by quantitative magnetic resonance: A future technology of body composition research? Curr. Opin. Clin. Nutr. Metab. Care 2015, 18, 446–451. [Google Scholar] [CrossRef] [PubMed]
- Gallagher, D.; Thornton, J.C.; He, Q.; Wang, J.; Yu, W.; Bradstreet, T.E.; Burke, J.; Heymsfield, S.B.; Rivas, V.M.; Kaufman, R. Quantitative magnetic resonance fat measurements in humans correlate with established methods but are biased. Obesity (Silver Spring) 2010, 18, 2047–2054. [Google Scholar] [CrossRef] [PubMed]
- Sun, S.S.; Chumlea, W.C.; Heymsfield, S.B.; Lukaski, H.C.; Schoeller, D.; Friedl, K.; Kuczmarski, R.J.; Flegal, K.M.; Johnson, C.L.; Hubbard, V.S. Development of bioelectrical impedance analysis prediction equations for body composition with the use of a multicomponent model for use in epidemiologic surveys. Am. J. Clin. Nutr. 2003, 77, 331–340. [Google Scholar] [PubMed]
- Wang, Z.; Shen, W.; Kotler, D.P.; Heshka, S.; Wielopolski, L.; Aloia, J.F.; Nelson, M.E.; Pierson, R.N., Jr.; Heymsfield, S.B. Total body protein: A new cellular level mass and distribution prediction model. Am. J. Clin. Nutr. 2003, 78, 979–984. [Google Scholar]
- Mourtzakis, M.; Prado, C.M.; Lieffers, J.R.; Reiman, T.; McCargar, L.J.; Baracos, V.E. A practical and precise approach to quantification of body composition in cancer patients using computed tomography images acquired during routine care. Appl. Physiol. Nutr. Metab. 2008, 33, 997–1006. [Google Scholar] [CrossRef] [PubMed]
- Siervo, M.; Jebb, S.A. Body composition assessment: Theory into practice: Introduction of multicompartment models. IEEE Eng. Med. Biol. Mag. 2010, 29, 48–59. [Google Scholar] [CrossRef] [PubMed]
- Cederholm, T.; Bosaeus, I.; Barazzoni, R.; Bauer, J.; van Gossum, A.; Klek, S.; Muscaritoli, M.; Nyulasi, I.; Ockenga, J.; Schneider, S.M.; et al. Diagnostic criteria for malnutrition—An Espen consensus statement. Clin. Nutr. 2015, 34, 335–340. [Google Scholar] [CrossRef] [PubMed]
- Prado, C.M.M.; Lieffers, J.R.; McCargar, L.J.; Reiman, T.; Sawyer, M.B.; Martin, L.; Baracos, V.E. Prevalence and clinical implications of sarcopenic obesity in patients with solid tumours of the respiratory and gastrointestinal tracts: A population-based study. Lancet Oncol. 2008, 9, 629–635. [Google Scholar] [CrossRef]
- Cruz-Jentoft, A.J.; Baeyens, J.P.; Bauer, J.M.; Boirie, Y.; Cederholm, T.; Landi, F.; Martin, F.C.; Michel, J.P.; Rolland, Y.; Schneider, S.M.; et al. Sarcopenia: European consensus on definition and diagnosis: Report of the european working group on sarcopenia in older people. Age Ageing 2010, 39, 412–423. [Google Scholar] [CrossRef] [PubMed]
- Harris, J.A.; Benedict, F.G. A Biometric Study of Basal Metabolism in Man; Carnegie Institution of Washington: Washington, DC, USA, 1919. [Google Scholar]
- Rafii, M.; Chapman, K.; Owens, J.; Elango, R.; Campbell, W.W.; Ball, R.O.; Pencharz, P.B.; Courtney-Martin, G. Dietary protein requirement of female adults >65 years determined by the indicator amino acid oxidation technique is higher than current recommendations. J. Nutr. 2015, 145, 18–24. [Google Scholar] [CrossRef] [PubMed]
- Campbell, W.W.; Johnson, C.A.; McCabe, G.P.; Carnell, N.S. Dietary protein requirements of younger and older adults. Am. J. Clin. Nutr. 2008, 88, 1322–1329. [Google Scholar] [PubMed]
- Gorissen, S.H.; Burd, N.A.; Hamer, H.M.; Gijsen, A.P.; Groen, B.B.; van Loon, L.J. Carbohydrate coingestion delays dietary protein digestion and absorption but does not modulate postprandial muscle protein accretion. J. Clin. Endocrinol. Metab. 2014, 99, 2250–2258. [Google Scholar] [CrossRef] [PubMed]
- Symons, T.B.; Sheffield-Moore, M.; Wolfe, R.R.; Paddon-Jones, D. A moderate serving of high-quality protein maximally stimulates skeletal muscle protein synthesis in young and elderly subjects. J. Am. Diet. Assoc. 2009, 109, 1582–1586. [Google Scholar] [CrossRef] [PubMed]
- Campbell, W.W.; Trappe, T.A.; Jozsi, A.C.; Kruskall, L.J.; Wolfe, R.R.; Evans, W.J. Dietary protein adequacy and lower body versus whole body resistive training in older humans. J. Physiol. 2002, 542, 631–642. [Google Scholar] [CrossRef] [PubMed]
- Farnsworth, E.; Luscombe, N.D.; Noakes, M.; Wittert, G.; Argyiou, E.; Clifton, P.M. Effect of a high-protein, energy-restricted diet on body composition, glycemic control, and lipid concentrations in overweight and obese hyperinsulinemic men and women. Am. J. Clin. Nutr. 2003, 78, 31–39. [Google Scholar]
- Weigle, D.S.; Breen, P.A.; Matthys, C.C.; Callahan, H.S.; Meeuws, K.E.; Burden, V.R.; Purnell, J.Q. A high-protein diet induces sustained reductions in appetite, ad libitum caloric intake, and body weight despite compensatory changes in diurnal plasma leptin and ghrelin concentrations. Am. J. Clin. Nutr. 2005, 82, 41–48. [Google Scholar] [PubMed]
- Contaldo, F.; Di Biase, G.; Scalfi, L.; Presta, E.; Mancini, M. Protein-sparing modified fast in the treatment of severe obesity: Weight loss and nitrogen balance data. Int. J. Obes. 1980, 4, 189–196. [Google Scholar] [PubMed]
- Morais, J.A.; Chevalier, S.; Gougeon, R. Protein turnover and requirements in the healthy and frail elderly. J. Nutr. Health Aging 2006, 10, 272–283. [Google Scholar] [PubMed]
- Bunker, V.W.; Lawson, M.S.; Stansfield, M.F.; Clayton, B.E. Nitrogen balance studies in apparently healthy elderly people and those who are housebound. Br. J. Nutr. 1987, 57, 211–221. [Google Scholar] [CrossRef] [PubMed]
- Hutton, J.L.; Martin, L.; Field, C.J.; Wismer, W.V.; Bruera, E.D.; Watanabe, S.M.; Baracos, V.E. Dietary patterns in patients with advanced cancer: Implications for anorexia-cachexia therapy. Am. J. Clin. Nutr. 2006, 84, 1163–1170. [Google Scholar] [PubMed]
- Prado, C.M.; Lieffers, J.R.; Bergsten, G.; Mourtzakis, M.; Baracos, V.E.; Reiman, T.; Sawyer, M.B.; McCargar, L.J. Dietary patterns of patients with advanced lung or colorectal cancer. Can. J. Diet. Pract. Res. 2012, 73, e298–e303. [Google Scholar] [CrossRef] [PubMed]
| Healthy Subjects | |||
| All (n = 574) | Females (n = 319) | Males (n = 255) | |
| Age | 41.4 ± 15.2 | 39.9 ± 15.2 * | 43.2 ± 15.2 |
| Height (cm) | 172.8 ± 8.5 | 167.8 ± 6.5 * | 179.0 ± 6.3 |
| Weight (kg) | 82.2 ± 18.7 | 78.1 ± 19.8 * | 87.3 ± 15.8 |
| BMI (kg/m2) | 27.4 ± 5.6 | 27.6 ± 6.2 | 27.2 ± 4.5 |
| Lean massDXA (kg) 1 | 54.6 ± 11.9 | 46.6 ± 7.6 * | 64.6 ± 8.2 |
| Muscle massMRI (kg) | 26.1 ± 6.8 | 21.4 ± 3.8 * | 31.7 ± 5.1 |
| ASMIDXA (kg/m2) | 8.2 ± 1.6 | 7.4 ± 1.2 * | 9.3 ± 1.3 |
| FMIDXA (kg/m2) | 8.0 ± 15.2 | 9.8 ± 4.2 * | 9.3 ± 1.3 |
| Non Sarcopenic | 81.0% | 80.9% | 81.2% |
| Sarcopenic | 19.0% | 19.1% | 18.8% |
| Cirrhotic Patients | |||
| All (n = 403) | Females (n = 193) | Males (n = 210) | |
| Age | 44.7 ± 12.3 | 44.5 ± 12.9 | 44.9 ± 11.8 |
| Height (cm) | 170.5 ± 9.1 | 164.6 ± 6.7 * | 175.9 ± 7.5 |
| Body Weight (kg) | 67.5 ± 14.3 | 59.9 ± 11.0 * | 74.4 ± 13.5 |
| BMI (kg/m2) | 23.1 ± 3.9 | 22.1 ± 3.6 * | 24.0 ± 3.9 |
| Lean massbia (kg) 1 | 50.7 ± 10.5 | 43.1 ± 5.6 * | 57.7 ± 9.1 |
| Total body proteinTBK (kg) | 7.5 ± 2.3 | 6.5 ± 1.5 * | 8.8 ± 2.4 |
| FFMIBIA (kg/m2) | 17.3 ± 2.4 | 15.9 ± 1.6 * | 18.6 ± 2.3 |
| FMIBIA (kg/m2) | 5.8 ± 2.7 | 6.2 ± 2.8 * | 5.4 ± 2.5 |
| Non Sarcopenic | 70.7% | 69.4% | 71.9% |
| Sarcopenic | 29.3% | 30.6% | 28.1% |
| Cancer Patients | |||
| All (n = 547) | Females (n = 157) | Males (n = 390) | |
| Age | 61.3 ± 8.2 | 62.8 ± 7.8 * | 60.8 ± 8.4 |
| Height (cm) | 168.7 ± 9.6 | 158.4 ± 6.8 * | 172.8 ± 7.1 |
| Weight (kg) | 70.8 ± 14.0 | 63.6 ± 11.4 * | 73.7 ± 13.9 |
| BMI (kg/m2) | 24.8 ± 3.9 | 25.3 ± 4.1 * | 24.6 ± 3.8 |
| Lean massCT (kg) 1 | 48.6 ± 9.6 | 38.2 ± 4.9 | 52.8 ± 7.7 |
| ASMICT (kg/m2) | 6.6 ± 1.0 | 5.9 ± 0.8 * | 6.9 ± 0.9 |
| FMICT (kg/m2) 2 | 7.9 ± 2.3 | 9.2 ± 2.4 * | 7.3 ± 2.1 |
| Non Sarcopenic | 53.4% | 77.1% | 43.8% |
| Sarcopenic | 46.6% | 22.9% | 56.2% |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Geisler, C.; Prado, C.M.; Müller, M.J. Inadequacy of Body Weight-Based Recommendations for Individual Protein Intake—Lessons from Body Composition Analysis. Nutrients 2017, 9, 23. https://doi.org/10.3390/nu9010023
Geisler C, Prado CM, Müller MJ. Inadequacy of Body Weight-Based Recommendations for Individual Protein Intake—Lessons from Body Composition Analysis. Nutrients. 2017; 9(1):23. https://doi.org/10.3390/nu9010023
Chicago/Turabian StyleGeisler, Corinna, Carla M. Prado, and Manfred J. Müller. 2017. "Inadequacy of Body Weight-Based Recommendations for Individual Protein Intake—Lessons from Body Composition Analysis" Nutrients 9, no. 1: 23. https://doi.org/10.3390/nu9010023
APA StyleGeisler, C., Prado, C. M., & Müller, M. J. (2017). Inadequacy of Body Weight-Based Recommendations for Individual Protein Intake—Lessons from Body Composition Analysis. Nutrients, 9(1), 23. https://doi.org/10.3390/nu9010023




