The Role of Omega-3 Polyunsaturated Fatty Acids in Heart Failure: A Meta-Analysis of Randomised Controlled Trials
Abstract
:1. Introduction
2. Materials and Methods
2.1. Literature Search Strategy
2.2. Study Selection
2.3. Data Extraction
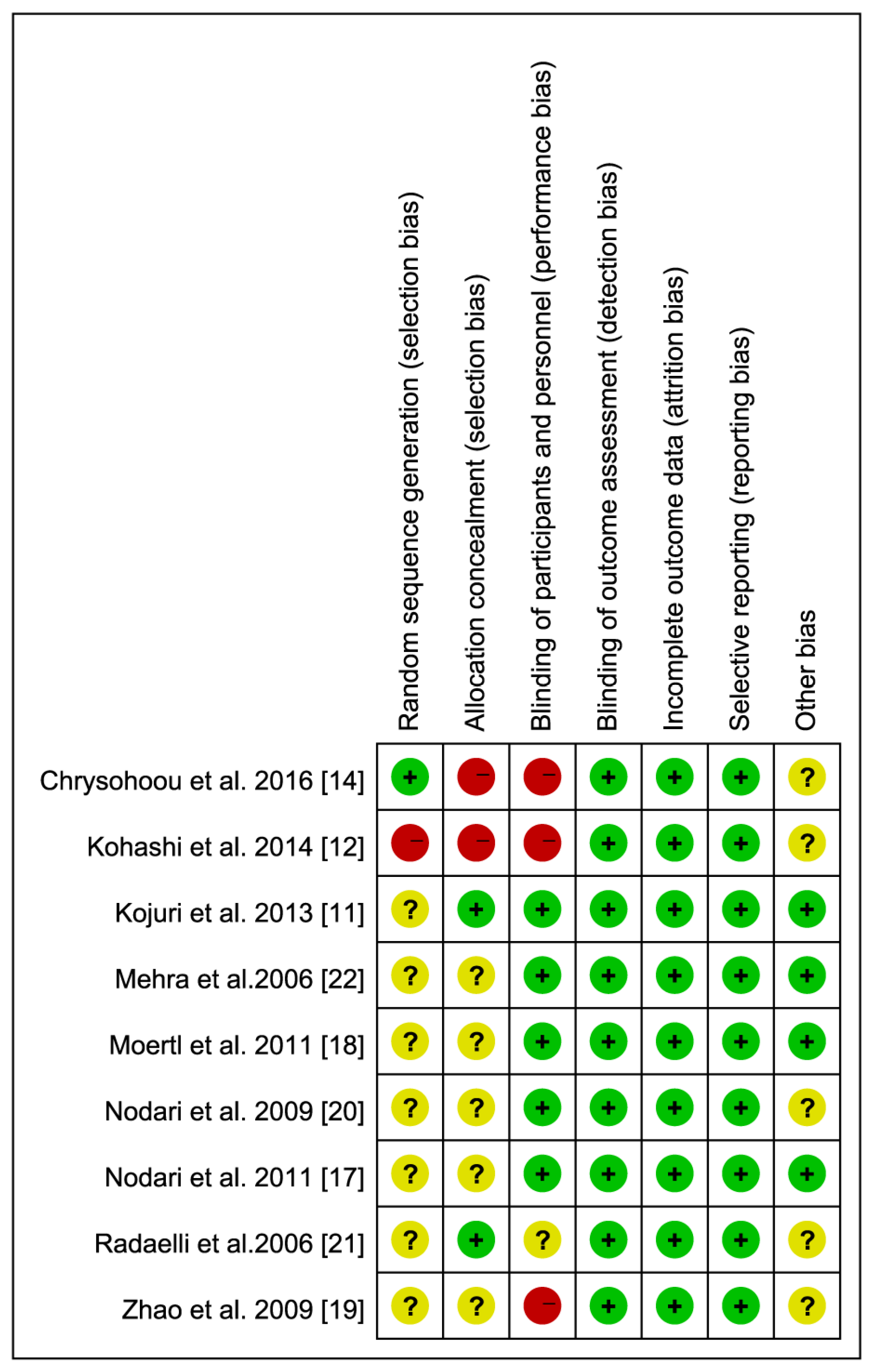
2.4. Assessment of Risk of Bias
2.5. Statistical Analysis
3. Results
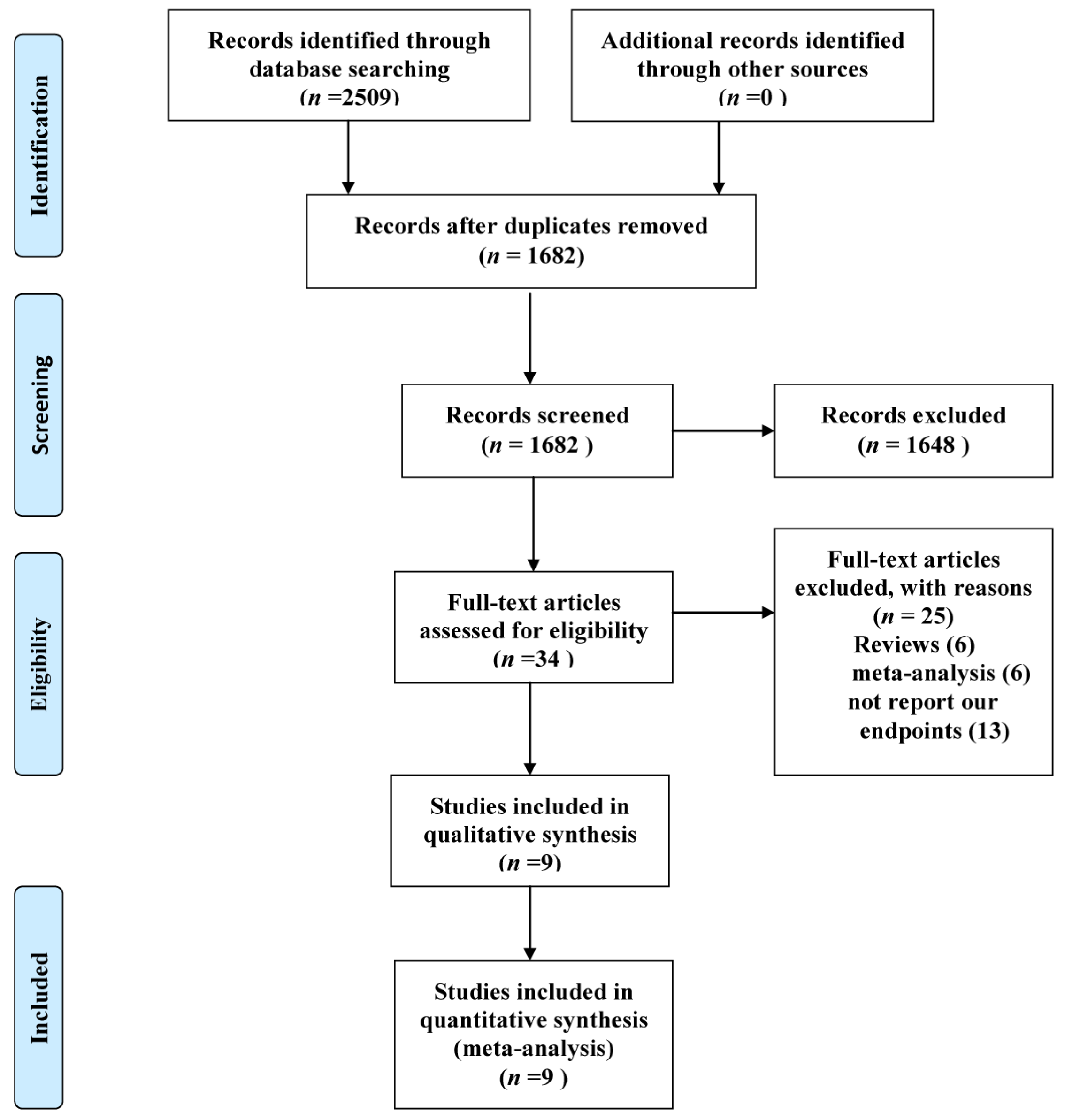
3.1. Eligible Studies
3.2. Study Characteristics
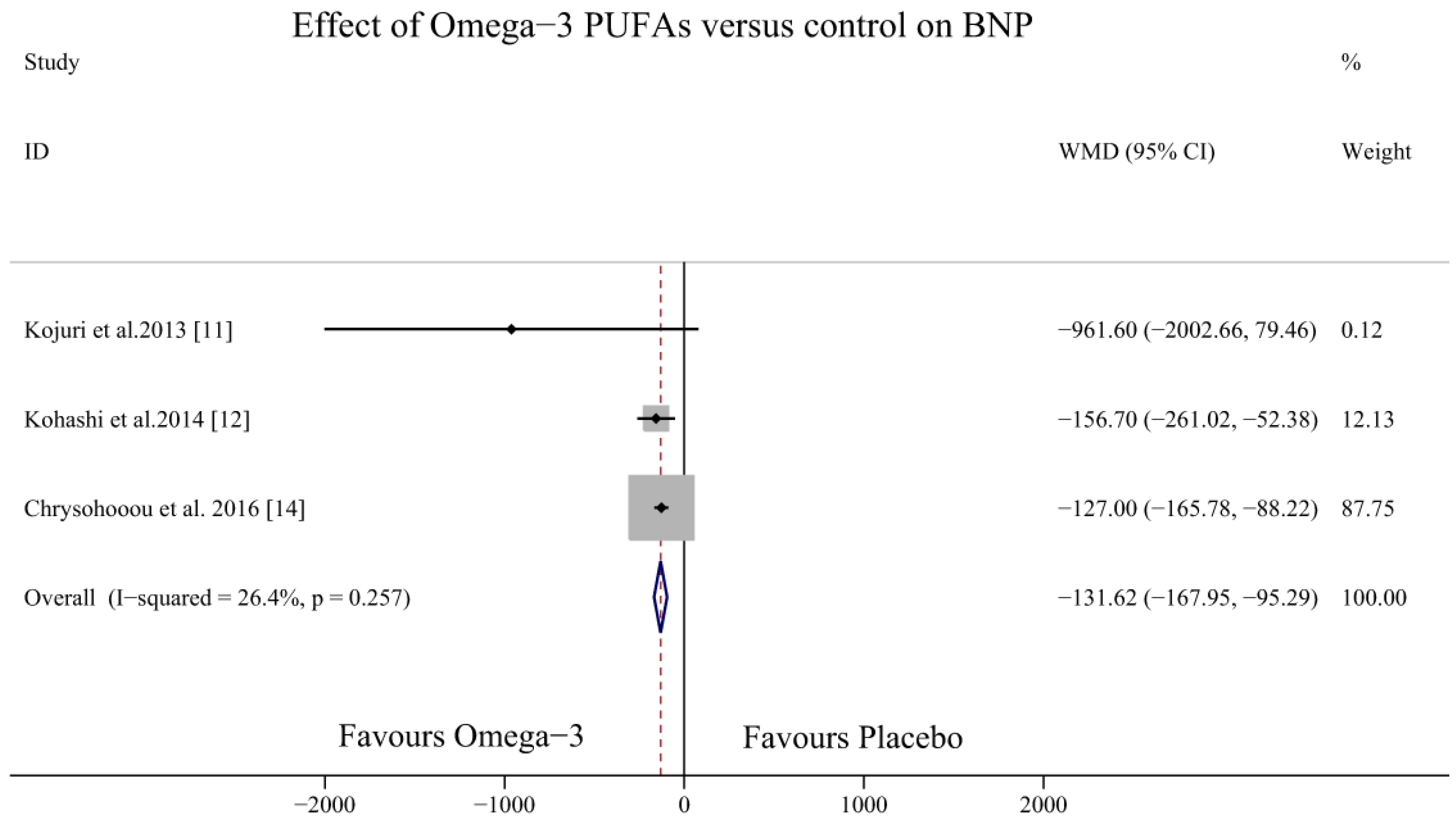
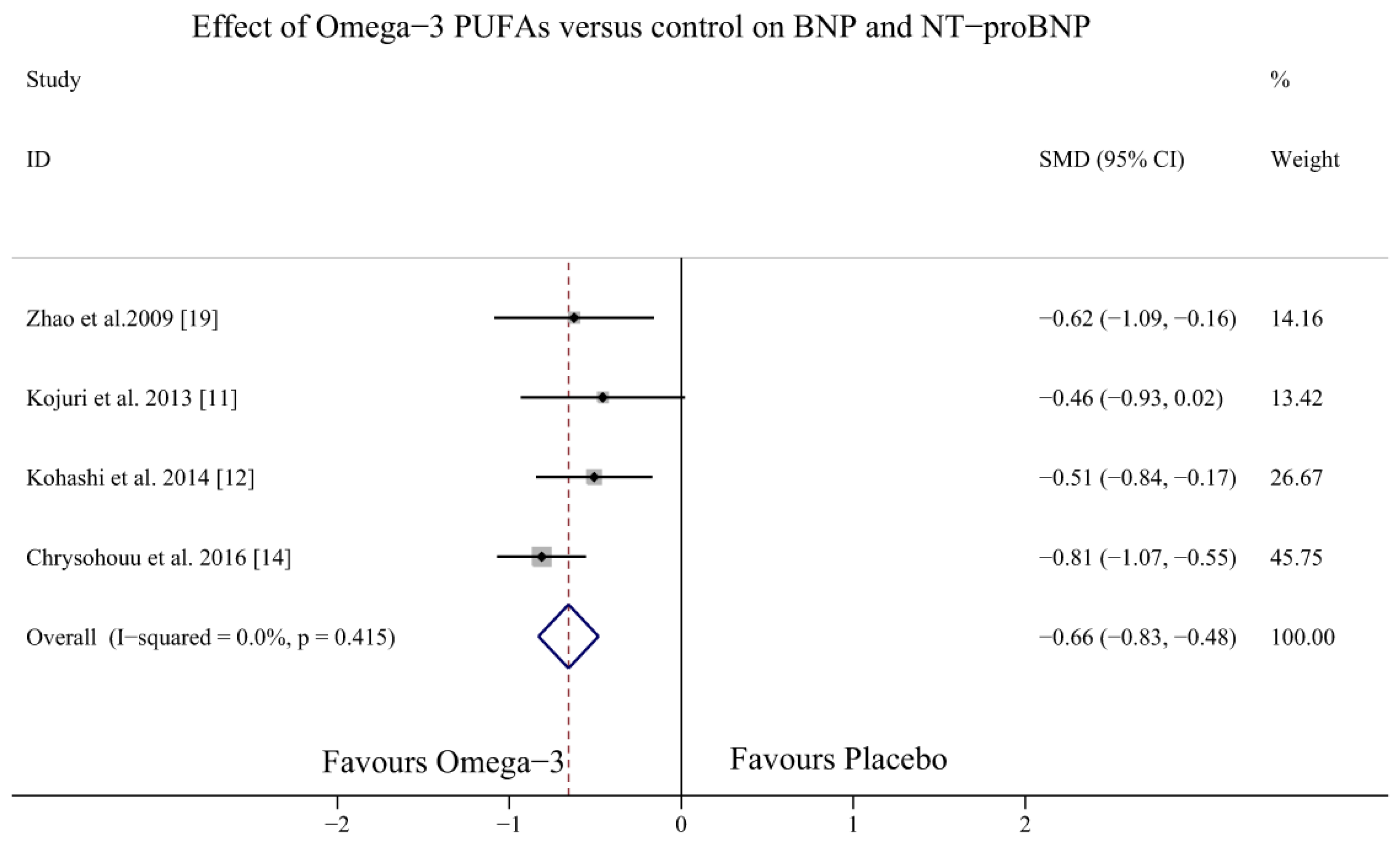
3.3. Effect of Omega-3 PUFAs on BNP Levels
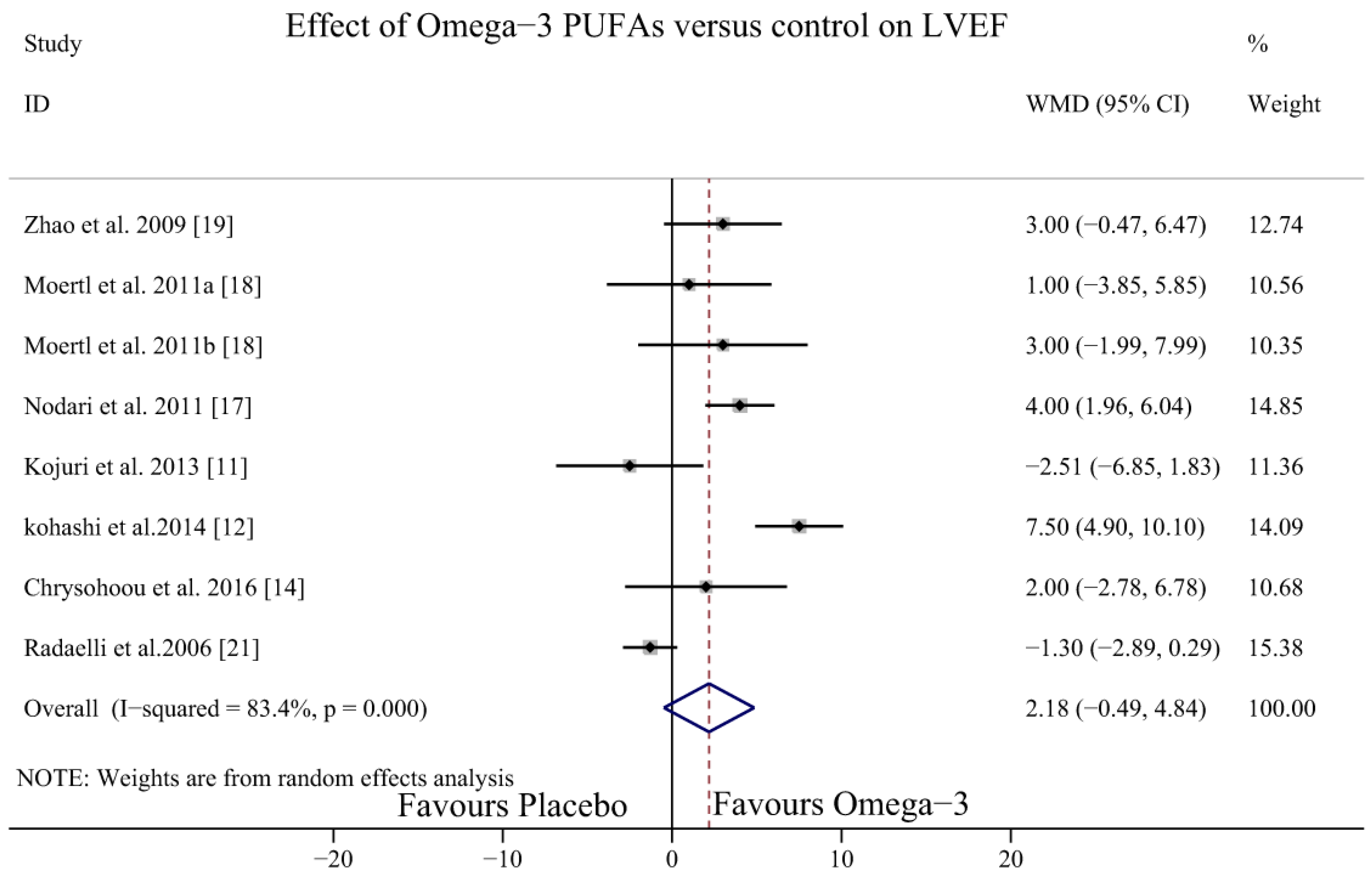
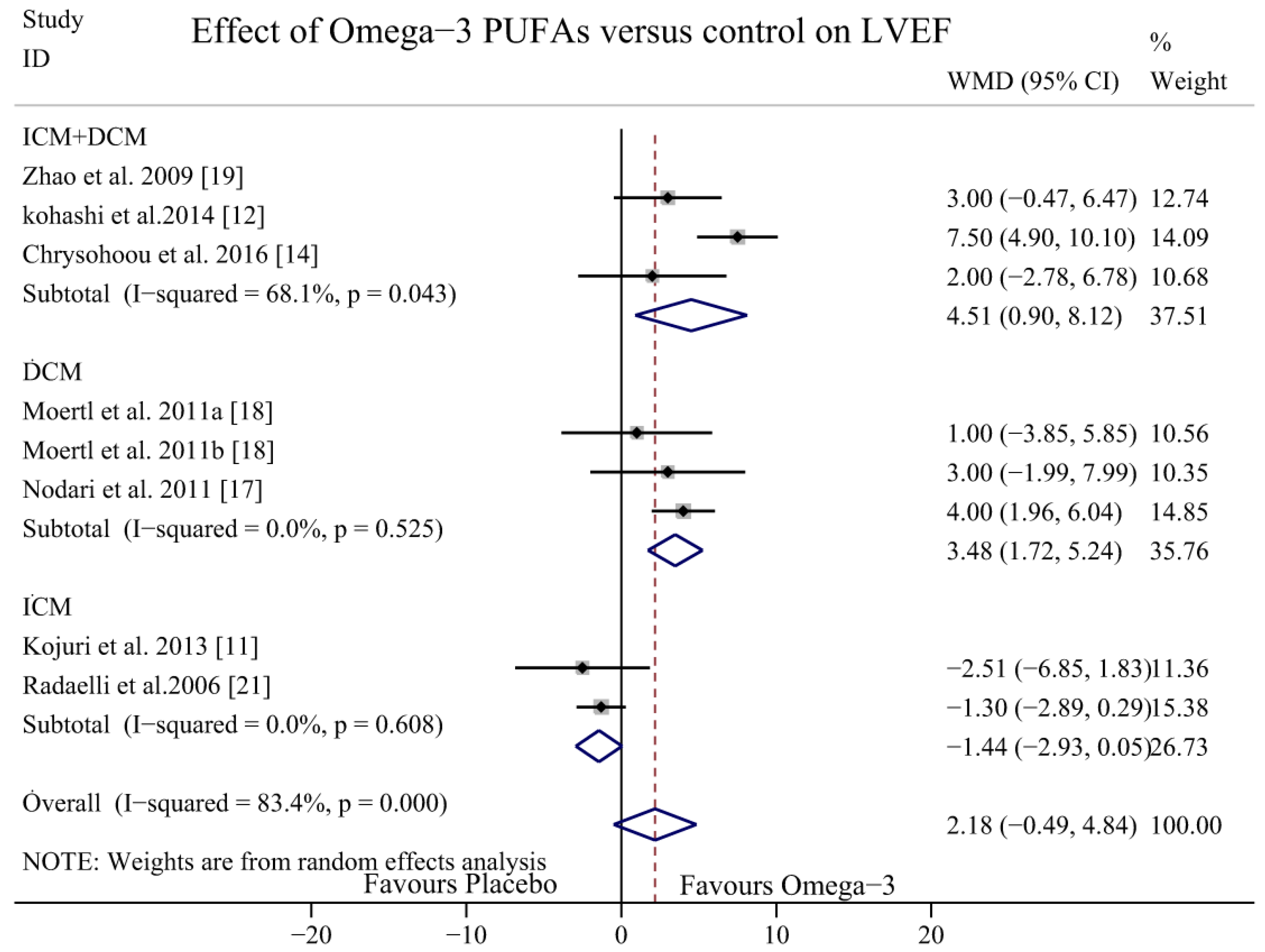
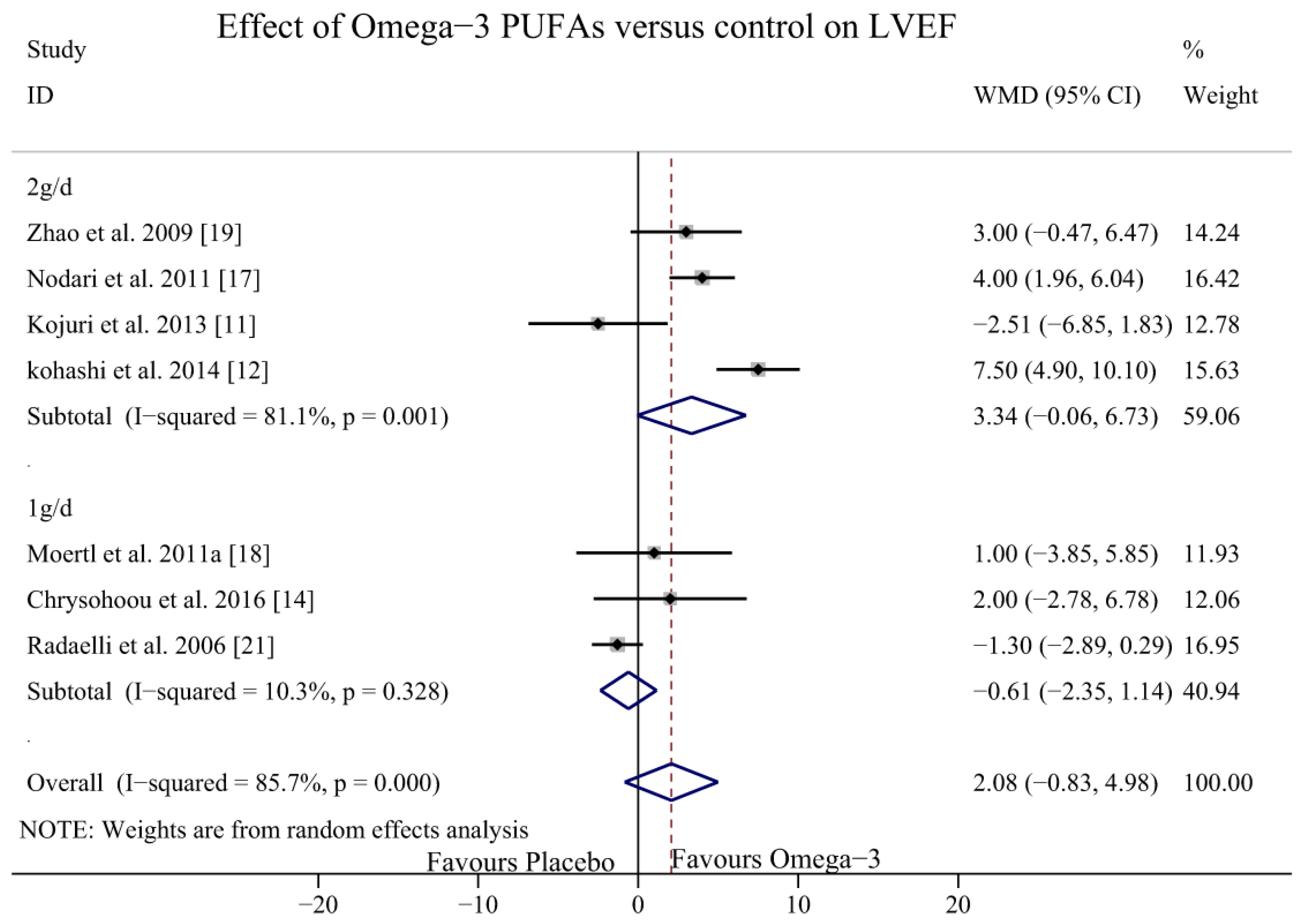
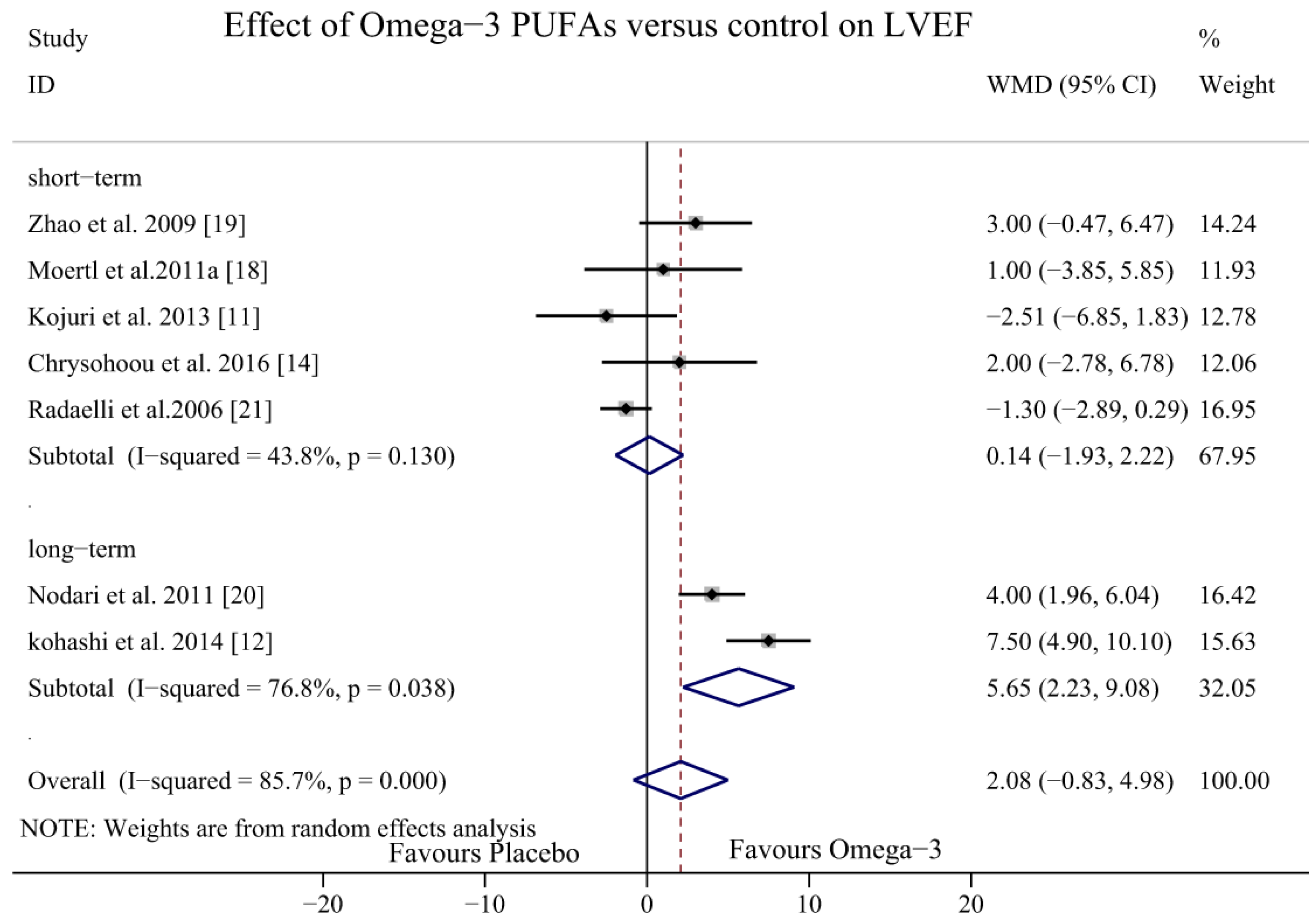
3.4. Effect of Omega-3 PUFAs on LVEF
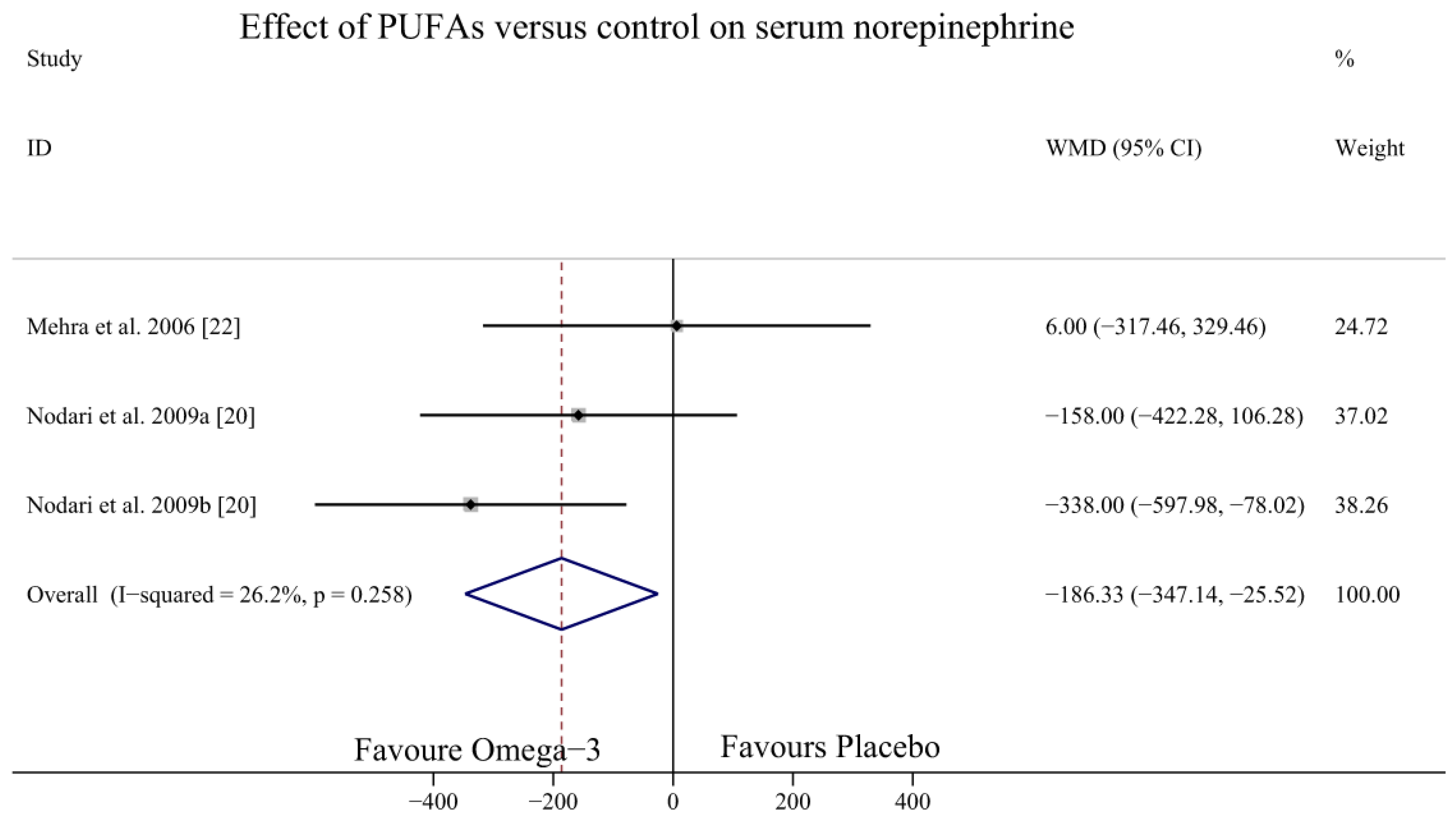
3.5. Effect of Omega-3 PUFAs on SNE Level
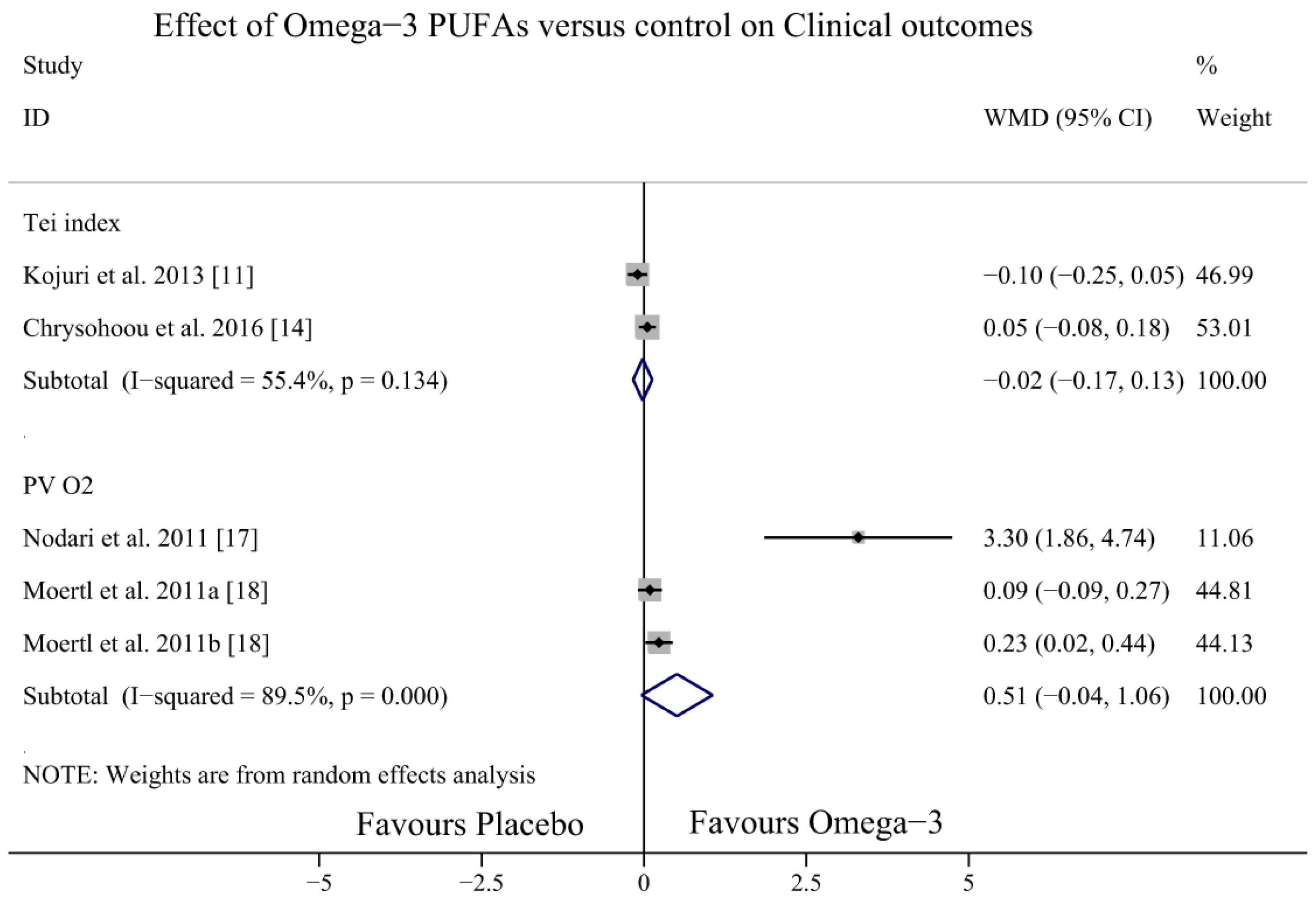
3.6. Effect of Omega-3 PUFAs on Otherclinical Outcomes
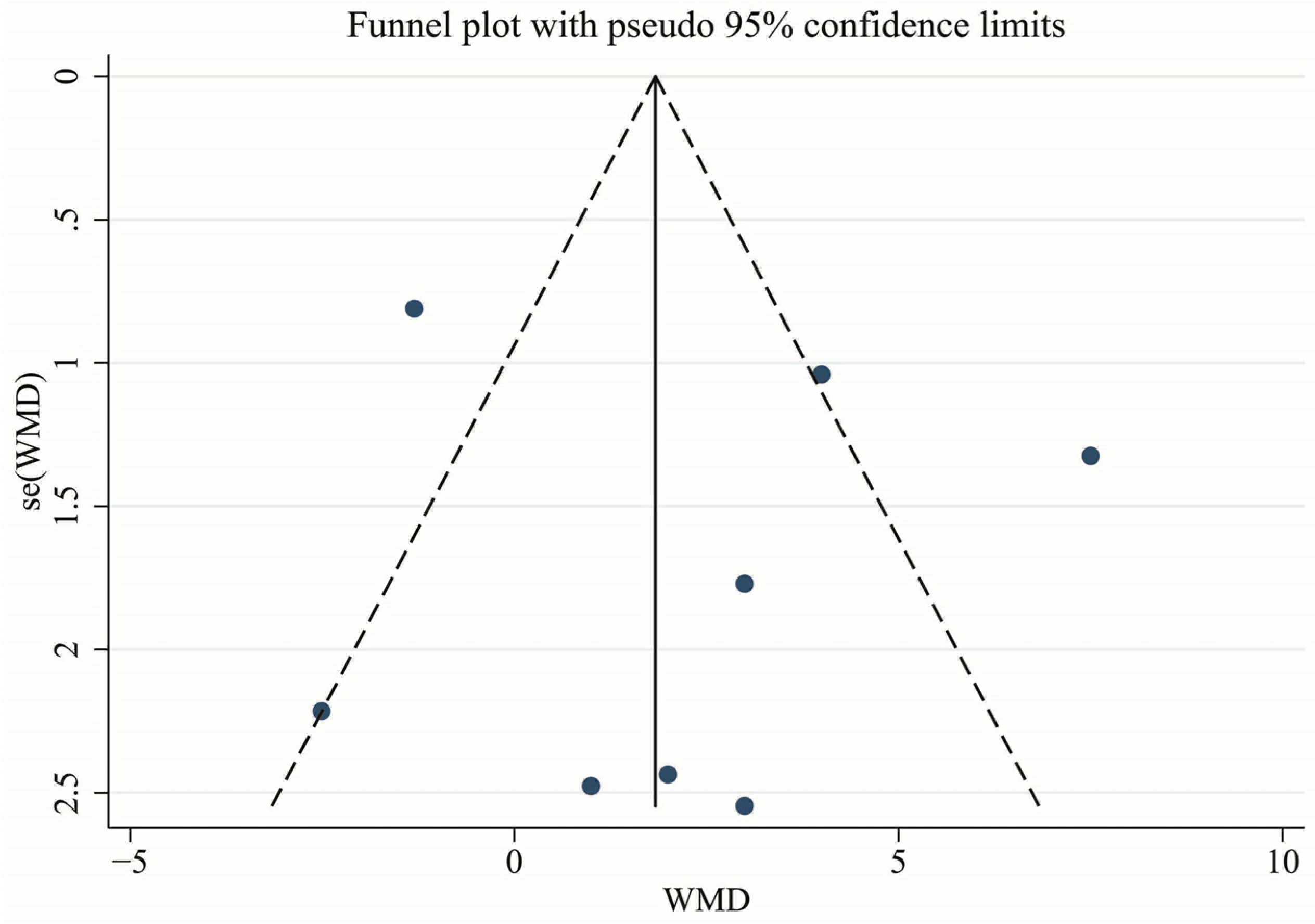
3.7. Publication Bias and Sensitivity Analysis
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Conflicts of Interest
Appendix A. Search Strategy in PubMed
References
- Piotr, P.; Anker, S.D.; Alhabib, K.F.; Cowie, M.R.; Force, T.L.; Hu, S.; Tiny, J.; Henry, K.; Vishal, R.; Rohde, L.E. Heart Failure. Preventing Disease and Death Worldwide. ESC Heart Fail. 2014, 1, 4–25. [Google Scholar]
- Roger, V.L.; Go, A.S.; Lloyd-Jones, D.M.; Adams, R.J.; Berry, J.D.; Brown, T.M.; Carnethon, M.R.; Dai, S.; De Simone, G.; Ford, E.S.; et al. Heart disease and stroke statistics—2011 update: A report from the American Heart Association. Circulation 2011, 123, e18–e209. [Google Scholar] [CrossRef] [PubMed]
- Jhund, P.S.; Macintyre, K.; Simpson, C.R.; Lewsey, J.D.; Stewart, S.; Redpath, A.; Chalmers, J.W.; Capewell, S.; McMurray, J.J. Long-term trends in first hospitalization for heart failure and subsequent survival between 1986 and 2003: A population study of 5.1 million people. Circulation 2009, 119, 515–523. [Google Scholar] [CrossRef] [PubMed]
- Wall, R.; Ross, R.P.; Fitzgerald, G.F.; Stanton, C. Fatty acids from fish: The anti-inflammatory potential of long-chain omega-3 fatty acids. Nutr. Rev. 2010, 68, 280–289. [Google Scholar] [CrossRef] [PubMed]
- Mozaffarian, D.; Bryson, C.L.; Lemaitre, R.N.; Burke, G.L.; Siscovick, D.S. Fish intake and risk of incident heart failure. J. Am. Coll. Cardiol. 2005, 45, 2015–2021. [Google Scholar] [CrossRef] [PubMed]
- Macchia, A.; Levantesi, G.; Franzosi, M.G.; Geraci, E.; Maggioni, A.P.; Marfisi, R.; Nicolosi, G.L.; Schweiger, C.; Tavazzi, L.; Tognoni, G.; et al. Left ventricular systolic dysfunction, total mortality, and sudden death in patients with myocardial infarction treated with n-3 polyunsaturated fatty acids. Eur. J. Heart Fail. 2005, 7, 904–909. [Google Scholar] [CrossRef] [PubMed]
- Yamagishi, K.; Iso, H.; Date, C.; Fukui, M.; Wakai, K.; Kikuchi, S.; Inaba, Y.; Tanabe, N.; Tamakoshi, A. Fish, omega-3 polyunsaturated fatty acids, and mortality from cardiovascular diseases in a nationwide community-based cohort of Japanese men and women the JACC (Japan Collaborative Cohort Study for Evaluation of Cancer Risk) study. J. Am. Coll. Cardiol. 2008, 52, 988–996. [Google Scholar] [CrossRef] [PubMed]
- Yamagishi, K.; Nettleton, J.A.; Folsom, A.R. Plasma fatty acid composition and incident heart failure in middle-aged adults: The atherosclerosis risk in Communities (ARIC) Study. Am. Heart J. 2008, 156, 965–974. [Google Scholar] [CrossRef] [PubMed]
- Tavazzi, L.; Maggioni, A.P.; Marchioli, R.; Barlera, S.; Franzosi, M.G.; Latini, R.; Tognoni, G. Effect of n-3 polyunsaturated fatty acids in patients with chronic heart failure (the GISSI-HF trial): A randomised, double-blind, placebo-controlled trial. Lancet 2008, 372, 1223–1230. [Google Scholar] [PubMed]
- Xin, W.; Wei, W.; Li, X. Effects of fish oil supplementation on cardiac function in chronic heart failure: A meta-analysis of randomised controlled trials. Heart (Br. Card. Soc.) 2012, 98, 1620–1625. [Google Scholar] [CrossRef] [PubMed]
- Kojuri, J.; Ostovan, M.A.; Rezaian, G.R.; Archin Dialameh, P.; Zamiri, N.; Sharifkazemi, M.B.; Jannati, M. Effect of omega-3 on brain natriuretic peptide and echocardiographic findings in heart failure: Double-blind placebo-controlled randomized trial. J. Cardiovasc. Dis. Res. 2013, 4, 20–24. [Google Scholar] [CrossRef] [PubMed]
- Kohashi, K.; Nakagomi, A.; Saiki, Y.; Morisawa, T.; Kosugi, M.; Kusama, Y.; Atarashi, H.; Shimizu, W. Effects of eicosapentaenoic acid on the levels of inflammatory markers, cardiac function and long-term prognosis in chronic heart failure patients with dyslipidemia. J. Atheroscleros. Thromb. 2014, 21, 712–729. [Google Scholar] [CrossRef]
- Hoogeveen, E.K.; Geleijnse, J.M.; Kromhout, D.; Van’T Sant, P.; Gemen, E.F.; Kusters, R.; Giltay, E.J. No effect of n-3 fatty acids supplementation on NT-proBNP after myocardial infarction: The alpha omega trial. Eur. J. Prev. Cardiol. 2015, 22, 648–655. [Google Scholar] [CrossRef] [PubMed]
- Chrysohoou, C.; Metallinos, G.; Georgiopoulos, G.; Mendrinos, D.; Papanikolaou, A.; Magkas, N.; Pitsavos, C.; Vyssoulis, G.; Stefanadis, C.; Tousoulis, D. Short term omega-3 polyunsaturated fatty acid supplementation induces favorable changes in right ventricle function and diastolic filling pressure in patients with chronic heart failure; a randomized clinical trial. Vasc. Pharmacol. 2016, 79, 43–50. [Google Scholar] [CrossRef] [PubMed]
- Ghio, S.; Scelsi, L.; Latini, R.; Masson, S.; Eleuteri, E.; Palvarini, M.; Vriz, O.; Pasotti, M.; Gorini, M.; Marchioli, R.; et al. Effects of n-3 polyunsaturated fatty acids and of rosuvastatin on left ventricular function in chronic heart failure: A substudy of GISSI-HF trial. Eur. J. Heart Fail. 2010, 12, 1345–1353. [Google Scholar] [CrossRef] [PubMed]
- Liberati, A.; Altman, D.G.; Tetzlaff, J.; Mulrow, C.; Gotzsche, P.C.; Ioannidis, J.P.; Clarke, M.; Devereaux, P.J.; Kleijnen, J.; Moher, D. The prisma statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: Explanation and elaboration. Ann. Intern. Med. 2009, 151, W65–W94. [Google Scholar] [CrossRef] [PubMed]
- Nodari, S.; Triggiani, M.; Campia, U.; Manerba, A.; Milesi, G.; Cesana, B.M.; Gheorghiade, M.; Dei Cas, L. Effects of n-3 polyunsaturated fatty acids on left ventricular function and functional capacity in patients with dilated cardiomyopathy. J. Am. Coll. Cardiol. 2011, 57, 870–879. [Google Scholar] [CrossRef] [PubMed]
- Moertl, D.; Hammer, A.; Steiner, S.; Hutuleac, R.; Vonbank, K.; Berger, R. Dose-dependent effects of omega-3-polyunsaturated fatty acids on systolic left ventricular function, endothelial function, and markers of inflammation in chronic heart failure of nonischemic origin: A double-blind, placebo-controlled, 3-arm study. Am. Heart J. 2011, 161, 915.e1–915.e9. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.T.; Shao, L.; Teng, L.L.; Hu, B.; Luo, Y.; Yu, X.; Zhang, D.F.; Zhang, H. Effects of n-3 polyunsaturated fatty acid therapy on plasma inflammatory markers and N-terminal pro-brain natriuretic peptide in elderly patients with chronic heart failure. J. Int. Med. Res. 2009, 37, 1831–1841. [Google Scholar] [CrossRef] [PubMed]
- Nodari, S.; Metra, M.; Milesi, G.; Manerba, A.; Cesana, B.M.; Gheorghiade, M.; Dei Cas, L. The role of n-3 PUFAs in preventing the arrhythmic risk in patients with idiopathic dilated cardiomyopathy. Cardiovasc. Drugs Ther. 2009, 23, 5–15. [Google Scholar] [CrossRef] [PubMed]
- Radaelli, A.; Cazzaniga, M.; Viola, A.; Balestri, G.; Janetti, M.B.; Signorini, M.G.; Castiglioni, P.; Azzellino, A.; Mancia, G.; Ferrari, A.U. Enhanced baroreceptor control of the cardiovascular system by polyunsaturated Fatty acids in heart failure patients. J. Am. Coll. Cardiol. 2006, 48, 1600–1606. [Google Scholar] [CrossRef] [PubMed]
- Mehra, M.R.; Lavie, C.J.; Ventura, H.O.; Milani, R.V. Fish oils produce anti-inflammatory effects and improve body weight in severe heart failure. J. Heart Lung Transplant. 2006, 25, 834–838. [Google Scholar] [CrossRef] [PubMed]
- Koglin, J.; Pehlivanli, S.; Schwaiblmair, M.; Vogeser, M.; Cremer, P.; Von Scheidt, W. Role of brain natriuretic peptide in risk stratification of patients with congestive heart failure. J. Am. Coll. Cardiol. 2001, 38, 1934–1941. [Google Scholar] [CrossRef]
- Stanek, B.; Frey, B.; Hulsmann, M.; Berger, R.; Sturm, B.; Strametz-Juranek, J.; Bergler-Klein, J.; Moser, P.; Bojic, A.; Hartter, E.; et al. Prognostic evaluation of neurohumoral plasma levels before and during beta-blocker therapy in advanced left ventricular dysfunction. J. Am. Coll. Cardiol. 2001, 38, 436–442. [Google Scholar] [CrossRef]
- Berger, R.; Huelsman, M.; Strecker, K.; Bojic, A.; Moser, P.; Stanek, B.; Pacher, R. B-type natriuretic peptide predicts sudden death in patients with chronic heart failure. Circulation 2002, 105, 2392–2397. [Google Scholar] [CrossRef] [PubMed]
- De Groote, P.; Dagorn, J.; Soudan, B.; Lamblin, N.; McFadden, E.; Bauters, C. B-type natriuretic peptide and peak exercise oxygen consumption provide independent information for risk stratification in patients with stable congestive heart failure. J. Am. Coll. Cardiol. 2004, 43, 1584–1589. [Google Scholar] [CrossRef] [PubMed]
- Linssen, G.C.; Bakker, S.J.; Voors, A.A.; Gansevoort, R.T.; Hillege, H.L.; De Jong, P.E.; Van Veldhuisen, D.J.; Gans, R.O.; De Zeeuw, D. N-terminal pro-B-type natriuretic peptide is an independent predictor of cardiovascular morbidity and mortality in the general population. Eur. Heart J. 2010, 31, 120–127. [Google Scholar] [CrossRef] [PubMed]
- Oreopoulos, A.; Padwal, R.; Kalantar-Zadeh, K.; Fonarow, G.C.; Norris, C.M.; McAlister, F.A. Body mass index and mortality in heart failure: A meta-analysis. Am. Heart J. 2008, 156, 13–22. [Google Scholar] [CrossRef] [PubMed]
- Gluck, T.; Alter, P. Marine omega-3 highly unsaturated fatty acids: From mechanisms to clinical implications in heart failure and arrhythmias. Vasc. Pharmacol. 2016, 82, 11–19. [Google Scholar] [CrossRef] [PubMed]
- Molfino, A.; Gioia, G.; Rossi Fanelli, F.; Muscaritoli, M. The role for dietary omega-3 fatty acids supplementation in older adults. Nutrients 2014, 6, 4058–4073. [Google Scholar] [CrossRef] [PubMed]
- Kromhout, D.; Giltay, E.J.; Geleijnse, J.M. n-3 fatty acids and cardiovascular events after myocardial infarction. N. Engl. J. Med. 2010, 363, 2015–2026. [Google Scholar] [CrossRef] [PubMed]
- Halade, G.V.; Kain, V.; Black, L.M.; Prabhu, S.D.; Ingle, K.A. Aging dysregulates D- and E-series resolvins to modulate cardiosplenic and cardiorenal network following myocardial infarction. Aging 2016, 8, 2611–2634. [Google Scholar] [CrossRef] [PubMed]
- Kain, V.; Prabhu, S.D.; Halade, G.V. Inflammation revisited: Inflammation versus resolution of inflammation following myocardial infarction. Basic Res. Cardiol. 2014, 109, 444. [Google Scholar] [CrossRef] [PubMed]
- Toma, M.; Ezekowitz, J.A.; Bakal, J.A.; O’Connor, C.M.; Hernandez, A.F.; Sardar, M.R.; Zolty, R.; Massie, B.M.; Swedberg, K.; Armstrong, P.W.; et al. The relationship between left ventricular ejection fraction and mortality in patients with acute heart failure: Insights from the ASCEND-HF Trial. Eur. J. Heart Fail. 2014, 16, 334–341. [Google Scholar] [CrossRef] [PubMed]
- Tang, W.H.; Samara, M.A. Polyunsaturated fatty acids in heart failure: Should we give more and give earlier? J. Am. Coll. Cardiol. 2011, 57, 880–883. [Google Scholar] [CrossRef] [PubMed]
- Duda, M.K.; O’Shea, K.M.; Stanley, W.C. Omega-3 polyunsaturated fatty acid supplementation for the treatment of heart failure: Mechanisms and clinical potential. Cardiovasc. Res. 2009, 84, 33–41. [Google Scholar] [CrossRef] [PubMed]
- Marchioli, R.; Silletta, M.G.; Levantesi, G.; Pioggiarella, R.; Tognoni, G. n-3 polyunsaturated fatty acids in heart failure: Mechanisms and recent clinical evidence. Cell. Mol. Biol. 2010, 56, 110–130. [Google Scholar] [PubMed]
- Pepe, S.; McLennan, P.L. (n-3) Long Chain PUFA dose-dependently increase oxygen utilization efficiency and inhibit arrhythmias after saturated fat feeding in rats. J. Nutr. 2007, 137, 2377–2383. [Google Scholar] [PubMed]
- Parati, G.; Esler, M. The human sympathetic nervous system: Its relevance in hypertension and heart failure. Eur. Heart J. 2012, 33, 1058–1066. [Google Scholar] [CrossRef] [PubMed]
- Cohn, J.N.; Levine, T.B.; Olivari, M.T.; Garberg, V.; Lura, D.; Francis, G.S.; Simon, A.B.; Rector, T. Plasma norepinephrine as a guide to prognosis in patients with chronic congestive heart failure. N. Engl. J. Med. 1984, 311, 819–823. [Google Scholar] [CrossRef] [PubMed]
- Triposkiadis, F.; Karayannis, G.; Giamouzis, G.; Skoularigis, J.; Louridas, G.; Butler, J. The sympathetic nervous system in heart failure physiology, pathophysiology, and clinical implications. J. Am. Coll. Cardiol. 2009, 54, 1747–1762. [Google Scholar] [CrossRef] [PubMed]
- La Rovere, M.T.; Christensen, J.H. The autonomic nervous system and cardiovascular disease: Role of n-3 PUFAs. Vasc. Pharmacol. 2015, 71, 1–10. [Google Scholar] [CrossRef] [PubMed]
| Author/Year | Country | Design | Sample Size Total (Control/Treatment) | Population of Heart Failure | Mean Dose of Omega 3 (g/Day) | Duration (Months) | Control | Main Indexes |
|---|---|---|---|---|---|---|---|---|
| Mehra et al., 2006 [22] | USA | RCT | 14 (7/7) | NYHA III-IV ICM and DCM | 1 (EPA/DHA) | 4.5 | Placebo | TNF-a, IL-1 Serum norepinphrine |
| Radaelli et al., 2006 [21] | Italy | RCT | 25 (15/10) | post-MI EF < 40% | 2 (EPA/DHA) | 4 | Placebo | R-R interval, NYHA class LVEF |
| Nodari et al., 2009 [20] | Italy | RCT | 44 (22/22) | DCM EF ≤ 45% | 1 (EPA/DHA) | 6 | Placebo | Serum norepinephrine TNF-α, IL-1, IL-6 |
| Zhao et al., 2009 [19] | China | RCT | 76 (37/38) | NYHA II-III DCM and ICM EF < 40% | 2 (EPA/DHA) | 3 | Placebo | hs-CRP LVEF, NT-proBNP |
| Moertl et al., 2011 [18] | Austria | RCT | 49 (16/14,13) | NYHA III-IV DCM EF < 35% | 1.4 (EPA/DHA) | 3 | Placebo | LVEF, hsIL-6 INF-α, Peak VO2 |
| Nodari et al., 2011 [17] | Italy, USA | RCT | 133 (66/67) | NYHA II-III DCM EF ≤ 45% | 2 (EPA/DHA) | 12 | Placebo | LVEF Peak VO2 IL-6, IL-1, TNF-α |
| Kojuri et al., 2013 [11] | Iran | RCT | 70 (32/38) | NYHA II-III ICM EF < 40% | 2 (EPA/DHA) | 6 | Placebo | BNP, Tei index 6-min walk test LVEF |
| Kohashi et al., 2014 [12] | Japan | RCT | 139 (68/71) | ICM and DCM EF 37.6% ± 8% | 1.8 (EPA) | 12 | no | LVEF NYHA class BNP hsCRP, TNF-α |
| Chrysohoou et al., 2016 [14] | Greece | RCT | 250 (125/125) | NYHA I-III ICM and DCM | 1 (EFA/DHA) | 6 | Placebo | LVEF BNP Tei index |
| Study | Age, Year (Treatment/Control) | Weight (kg) | Body Mass Index (kg/m2) | Treatment Initiation | Ratio of EPA to DHA |
|---|---|---|---|---|---|
| Mehra et al., 2006 [22] | 59 ± 7/61 ± 8 | 58 ± 11/65 ± 11 | NA | Progression of HF | 44% EPA, 24% DHA and 12% other |
| Radaelli et al., 2006 [21] | 59.4 ± 2.5/60.1 ± 2.7 | NA | NA | Chronic post-MI HF | EPA/DHA ratio of 0.9 to 1.5 |
| Nodari et al., 2009 [20] | 61.09 ± 11.22/64.82 ± 9.46 | NA | NA | Progression of HF | EPA/DHA of 0.9:1.5 |
| Zhao et al., 2009 [19] | 74 ± 6/71 ± 10 | NA | 24.7 ± 3.6/24.0 ± 2.9 | Progression of HF | EPA 180 mg, DHA 120 mg |
| Moertl et al., 2011 [18] | 58.6 ± 7.0, 61.9 ± 9.6/55.1 ± 12.7 | NA | 27.0 ± 3.3, 27.8 ± 5.0/28.0 ± 5.6 | Progression of HF | EPA 465 mg, DHA 375 mg |
| Nodari et al., 2011 [17] | 61 ± 11/64 ± 9 | 76.9 ± 10.1/76.0 ± 7.6 | 25.9 ± 2.3/25.7 ± 2.22 | Progression of HF | EPA/DHA ratio of 0.9 to 1.5 |
| Kojuri et al., 2013 [11] | 56/58 | NA | NA | Progression of HF | NA |
| Kohashi et al., 2014 [12] | 71.4 ± 7.7 | NA | 22.7 ± 2.4/23.1 ± 2.9 | Progression of HF | EPA |
| Chrysohoou et al., 2016 [14] | 63 ± 12.8/63.4 ± 14.6 | NA | 28.72 ± 3.88/27.6 ± 4.76 | Progression of HF | NA |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, C.; Xiong, B.; Huang, J. The Role of Omega-3 Polyunsaturated Fatty Acids in Heart Failure: A Meta-Analysis of Randomised Controlled Trials. Nutrients 2017, 9, 18. https://doi.org/10.3390/nu9010018
Wang C, Xiong B, Huang J. The Role of Omega-3 Polyunsaturated Fatty Acids in Heart Failure: A Meta-Analysis of Randomised Controlled Trials. Nutrients. 2017; 9(1):18. https://doi.org/10.3390/nu9010018
Chicago/Turabian StyleWang, Chunbin, Bo Xiong, and Jing Huang. 2017. "The Role of Omega-3 Polyunsaturated Fatty Acids in Heart Failure: A Meta-Analysis of Randomised Controlled Trials" Nutrients 9, no. 1: 18. https://doi.org/10.3390/nu9010018
APA StyleWang, C., Xiong, B., & Huang, J. (2017). The Role of Omega-3 Polyunsaturated Fatty Acids in Heart Failure: A Meta-Analysis of Randomised Controlled Trials. Nutrients, 9(1), 18. https://doi.org/10.3390/nu9010018




