Probiotic Supplements Beneficially Affect Tryptophan–Kynurenine Metabolism and Reduce the Incidence of Upper Respiratory Tract Infections in Trained Athletes: A Randomized, Double-Blinded, Placebo-Controlled Trial
Abstract
:1. Introduction
2. Materials and Methods
2.1. Subjects
2.2. Study Intervention
2.3. Study Protocol
2.4. Exercise Tests
2.5. Blood Measurements
2.6. Statistical Analysis
3. Results
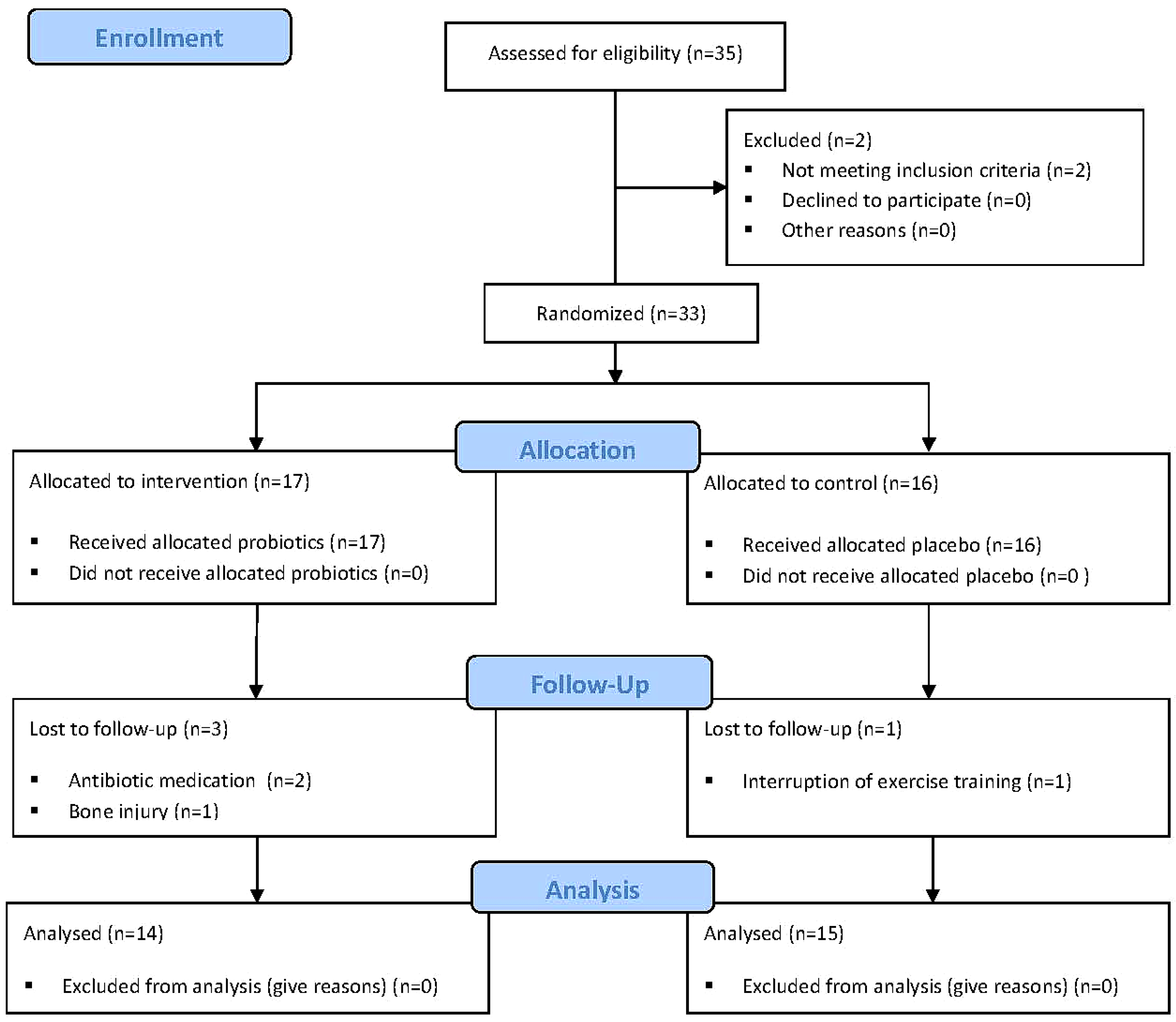
3.1. Study Population
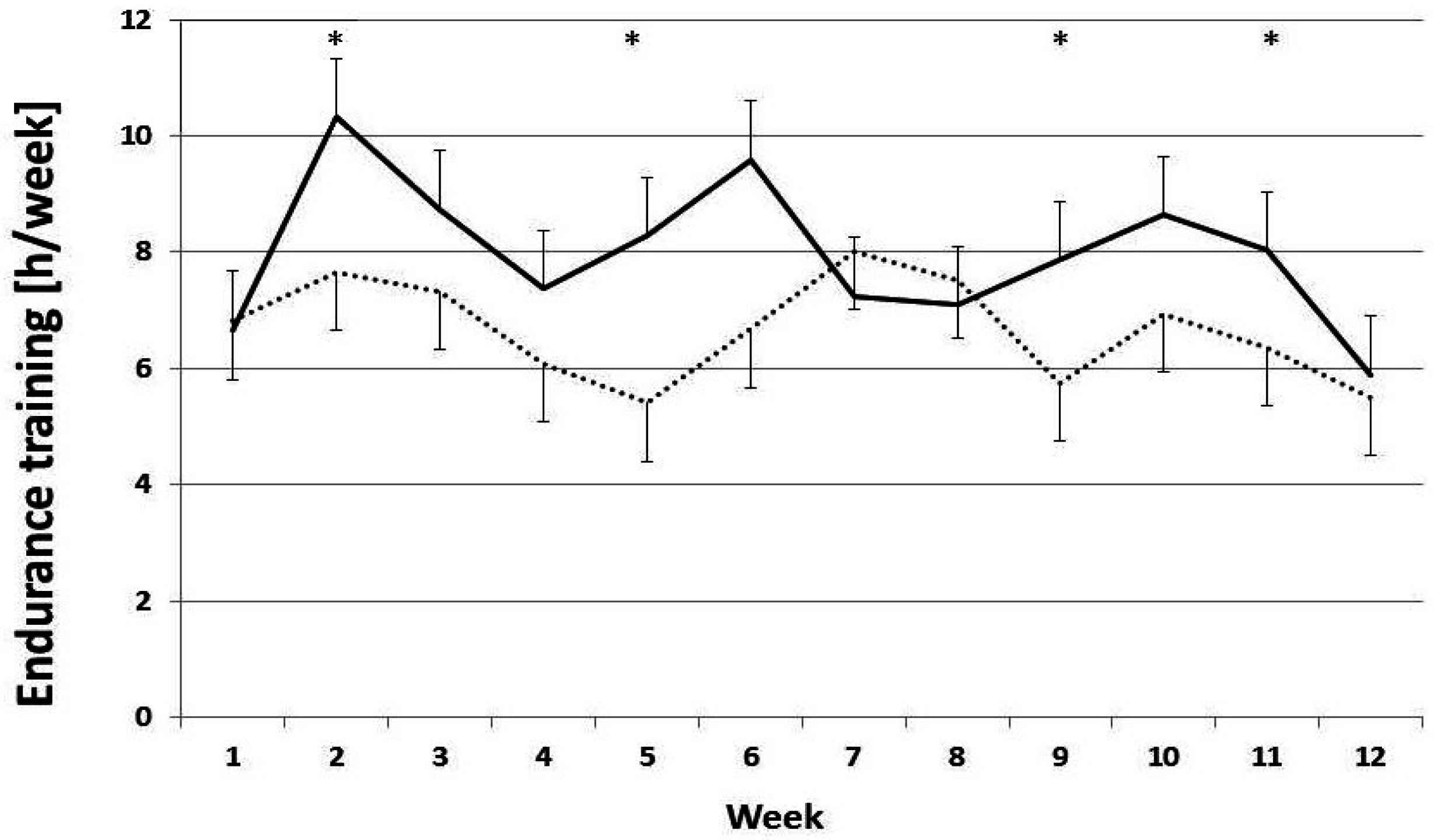
3.2. Training Loads
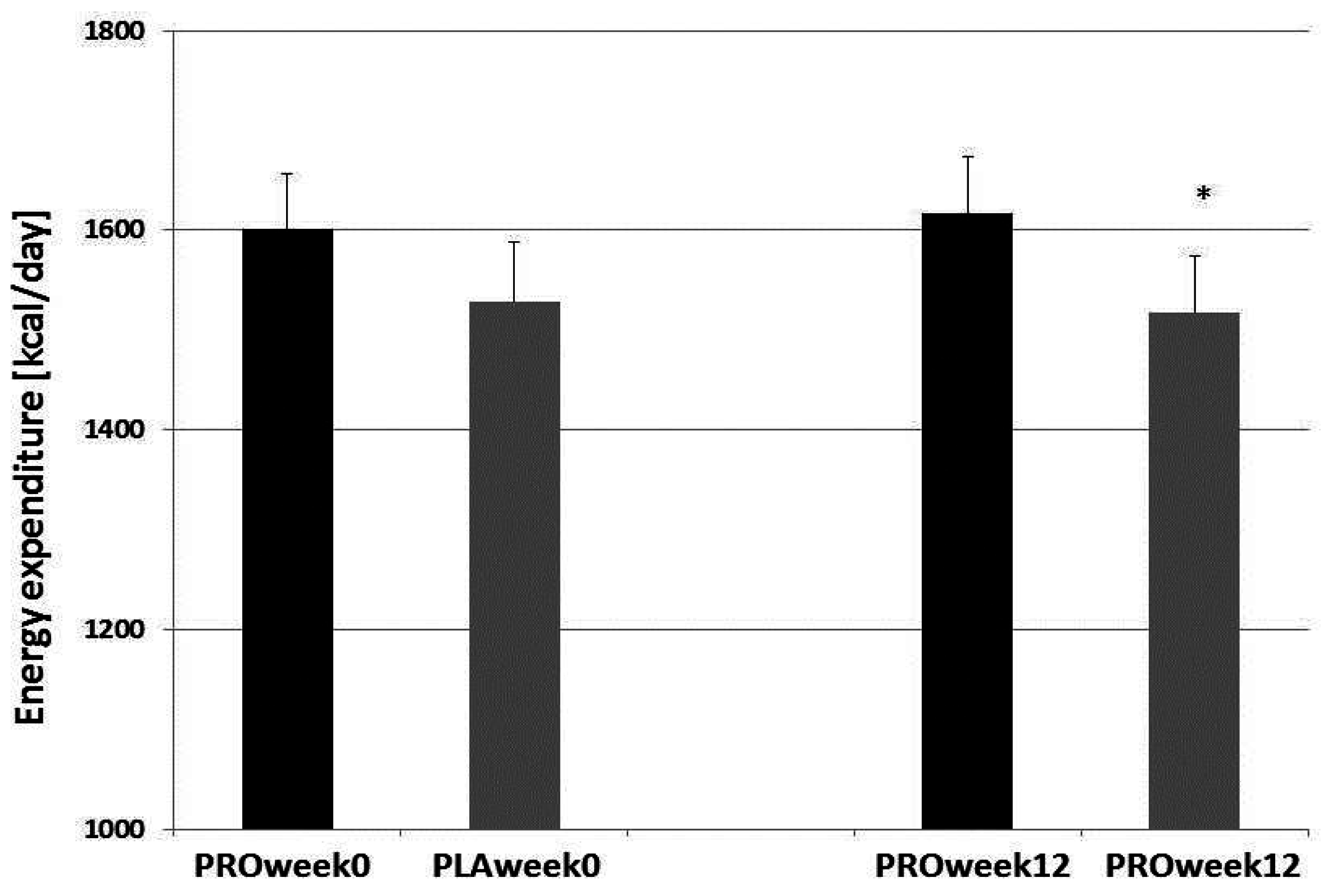
3.3. Body Composition, Nutrition, and Performance
3.4. Amino Acids
3.5. Immune System Biomarkers
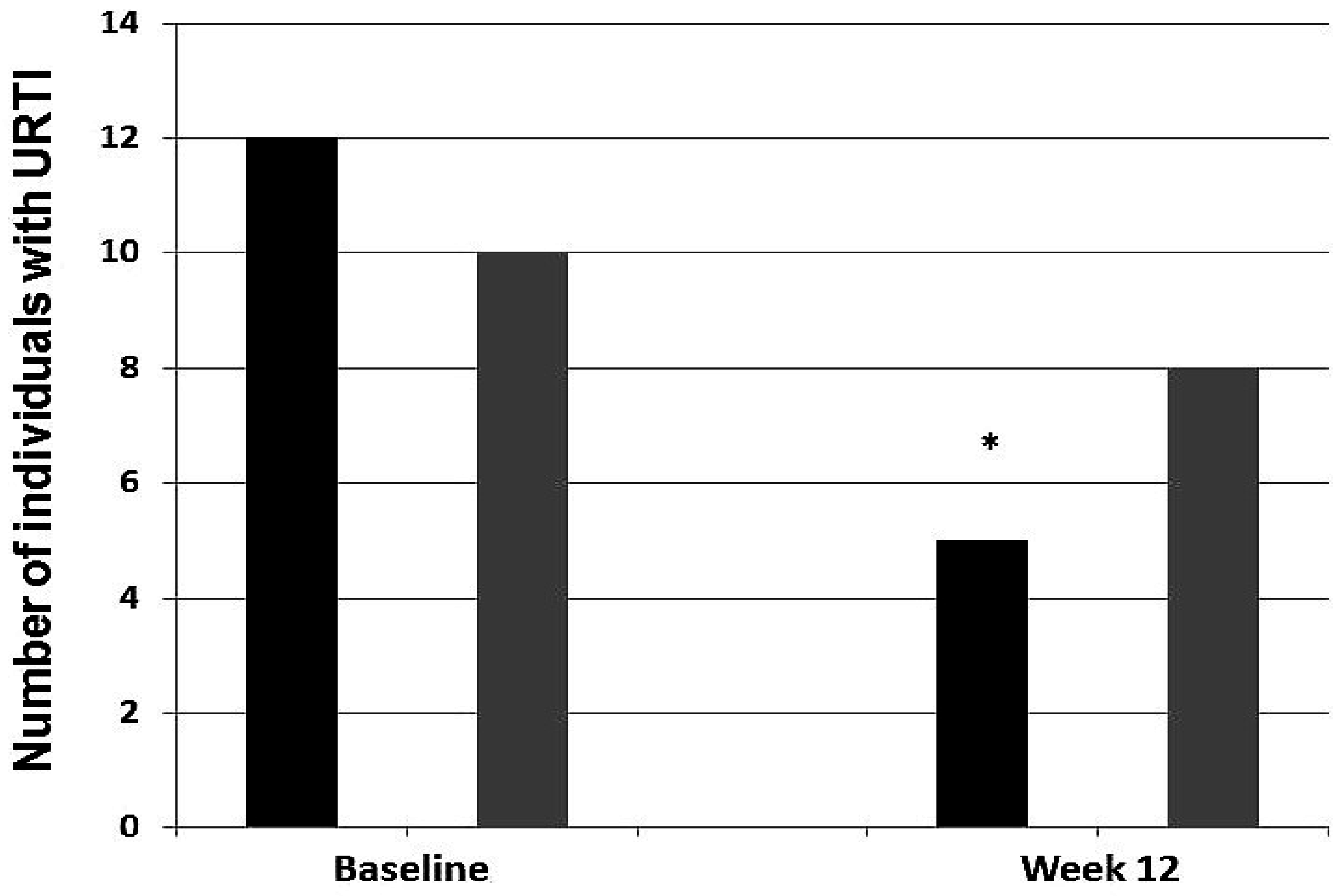
3.6. Infection Incidence
4. Discussion
4.1. Training Adherence
4.2. Tryptophan and the Gut Microbiome
4.3. Probiotics to Prevent URTIs
4.4. Study Strengths and Limitations
5. Conclusions and Future Research
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Nieman, D.C.; Henson, D.A.; Austin, M.D.; Sha, W. Upper respiratory tract infection is reduced in physically fit and active adults. Br. J. Sports Med. 2011, 45, 987–992. [Google Scholar] [CrossRef] [PubMed]
- Gleeson, M.; Bishop, N.C. URI in athletes: Are mucosal immunity and cytokine responses key risk factors? Exerc. Sport Sci. Rev. 2013, 41, 148–153. [Google Scholar] [CrossRef] [PubMed]
- Walsh, N.P.; Gleeson, M.; Shephard, R.J.; Gleeson, M.; Woods, J.A.; Bishop, N.C.; Fleshner, M.; Green, C.; Pedersen, B.K.; Hoffman-Goetz, L.; et al. Position statement. Part one: Immune function and exercise. Exerc. Immunol. Rev. 2011, 17, 6–63. [Google Scholar] [PubMed]
- Nieman, D.C. Immune response to heavy exertion. J. Appl. Physiol. 1997, 82, 1385–1394. [Google Scholar] [PubMed]
- He, C.S.; Bishop, N.C.; Handzlik, M.K.; Muhamad, A.S.; Gleeson, M. Sex differences in upper respiratory symptoms prevalence and oral-respiratory mucosal immunity in endurance athletes. Exerc. Immunol. Rev. 2014, 20, 8–22. [Google Scholar] [PubMed]
- Fahlman, M.M.; Engels, H.J. Mucosal IgA and URTI in American college football players: A year longitudinal study. Med. Sci. Sports Exerc. 2005, 37, 374–380. [Google Scholar] [CrossRef] [PubMed]
- Gleeson, M.; McDonald, W.A.; Pyne, D.B.; Cripps, A.W.; Francis, J.L.; Fricker, P.A.; Clancy, R.L. Salivary IgA levels and infection risk in elite swimmers. Med. Sci. Sports Exerc. 1999, 31, 67–73. [Google Scholar] [CrossRef] [PubMed]
- Neville, V.; Gleeson, M.; Folland, J.P. Salivary IgA as a risk factor for upper respiratory infections in elite professional athletes. Med. Sci. Sports Exerc. 2008, 40, 1228–1236. [Google Scholar] [CrossRef] [PubMed]
- Lancaster, G.I.; Halson, S.L.; Khan, Q.; Drysdale, P.; Wallace, F.; Jeukendrup, A.E.; Drayson, M.T.; Gleeson, M. Effects of acute exhaustive exercise and chronic exercise training on type 1 and type 2 T lymphocytes. Exerc. Immunol. Rev. 2004, 10, 91–106. [Google Scholar] [PubMed]
- Sprenger, H.; Jacobs, C.; Nain, M.; Gressner, A.M.; Prinz, H.; Wesemann, W.; Gemsa, D. Enhanced release of cytokines, interleukin-2 receptors, and neopterin after long-distance running. Clin. Immunol. Immunopathol. 1992, 63, 188–195. [Google Scholar] [CrossRef]
- Tilz, G.P.; Domej, W.; Diez-Ruiz, A.; Weiss, G.; Brezinschek, R.; Brezinschek, H.P.; Hüttl, E.; Pristautz, H.; Wachter, H.; Fuchs, D. Increased immune activation during and after physical exercise. Immunobiology 1993, 188, 194–202. [Google Scholar] [CrossRef]
- Strasser, B.; Gostner, J.M.; Fuchs, D. Mood, food, and cognition: Role of tryptophan and serotonin. Curr. Opin. Clin. Nutr. Metab. Care 2016, 19, 55–61. [Google Scholar] [CrossRef] [PubMed]
- Stone, T.W.; Perkins, M.N. Quinolinic acid: A potent endogenous excitant at amino acid receptors in CNS. Eur. J. Pharmacol. 1981, 72, 411–412. [Google Scholar] [CrossRef]
- Chen, Y.; Guillemin, G.J. Kynurenine pathway metabolites in humans: Disease and healthy states. Int. J. Tryptophan Res. 2009, 2, 1–19. [Google Scholar] [PubMed]
- Strasser, B.; Geiger, D.; Schauer, M.; Gatterer, H.; Burtscher, M.; Fuchs, D. Effects of exhaustive aerobic exercise on tryptophan-kynurenine metabolism in trained athletes. PLoS ONE 2016, 11, e0153617. [Google Scholar] [CrossRef] [PubMed]
- Strasser, B.; Sperner-Unterweger, B.; Fuchs, D.; Gostner, J.M. Mechanisms of Inflammation-Associated Depression: Immune Influences on Tryptophan and Phenylalanine Metabolisms. Curr. Top. Behav. Neurosci. 2016, in press. [Google Scholar]
- Jenkins, T.A.; Nguyen, J.C.; Polglaze, K.E.; Bertrand, P.P. Influence of tryptophan and serotonin on mood and cognition with a possible role of the gut-brain axis. Nutrients 2016, 8. [Google Scholar] [CrossRef] [PubMed]
- Evans, J.M.; Morris, L.S.; Marchesi, J.R. The gut microbiome: The role of a virtual organ in the endocrinology of the host. J. Endocrinol. 2013, 218, R37–R47. [Google Scholar] [CrossRef] [PubMed]
- Leblhuber, F.; Geisler, S.; Steiner, K.; Fuchs, D.; Schütz, B. Elevated fecal calprotectin in patients with Alzheimer’s dementia indicates leaky gut. J. Neural Transm. 2015, 122, 1319–1322. [Google Scholar] [CrossRef] [PubMed]
- Bermon, S.; Petriz, B.; Kajėnienė, A.; Prestes, J.; Castell, L.; Franco, O.L. The microbiota: An exercise immunology perspective. Exerc. Immunol. Rev. 2015, 21, 70–79. [Google Scholar] [PubMed]
- Cerdá, B.; Pérez, M.; Pérez-Santiago, J.D.; Tornero-Aguilera, J.F.; González-Soltero, R.; Larrosa, M. Gut Microbiota Modification: Another Piece in the Puzzle of the Benefits of Physical Exercise in Health? Front. Physiol. 2016, 7, 51. [Google Scholar] [CrossRef] [PubMed]
- Gleeson, M.; McFarlin, B.; Flynn, M. Exercise and Toll-like receptors. Exerc. Immunol. Rev. 2006, 12, 34–53. [Google Scholar] [PubMed]
- Matteoli, G.; Mazzini, E.; Iliev, I.D.; Mileti, E.; Fallarino, F.; Puccetti, P.; Chieppa, M.; Rescigno, M. Gut CD103+ dendritic cells express indoleamine 2,3-dioxygenase which influences T regulatory/T effector cell balance and oral tolerance induction. Gut 2010, 59, 595–604. [Google Scholar] [CrossRef] [PubMed]
- Nagata, S.; Asahara, T.; Wang, C.; Suyama, Y.; Chonan, O.; Takano, K.; Daibou, M.; Takahashi, T.; Nomoto, K.; Yamashiro, Y. The Effectiveness of Lactobacillus Beverages in Controlling Infections among the Residents of an Aged Care Facility: A Randomized Placebo-Controlled Double-Blind Trial. Ann. Nutr. Metab. 2016, 68, 51–59. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Li, X.; Ge, T.; Xiao, Y.; Liao, Y.; Cui, Y.; Zhang, Y.; Ho, W.; Yu, G.; Zhang, T. Probiotics for prevention and treatment of respiratory tract infections in children: A systematic review and meta-analysis of randomized controlled trials. Medicine 2016, 95, e4509. [Google Scholar] [CrossRef] [PubMed]
- O’Sullivan, O.; Cronin, O.; Clarke, S.F.; Murphy, E.F.; Molloy, M.G.; Shanahan, F.; Cotter, P.D. Exercise and the microbiota. Gut Microbes 2015, 6, 131–136. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Clarke, S.F.; Murphy, E.F.; O’Sullivan, O.; Lucey, A.J.; Humphreys, M.; Hogan, A.; Hayes, P.; O’Reilly, M.; Jeffery, I.B.; Wood-Martin, R.; et al. Exercise and associated dietary extremes impact on gut microbial diversity. Gut 2014, 63, 1913–1920. [Google Scholar] [CrossRef] [PubMed]
- Jäger, R.; Purpura, M.; Stone, J.D.; Turner, S.M.; Anzalone, A.J.; Eimerbrink, M.J.; Pane, M.; Amoruso, A.; Rowlands, D.S.; Oliver, J.M. Probiotic Streptococcus thermophilus FP4 and Bifidobacterium breve BR03 Supplementation Attenuates Performance and Range-of-Motion Decrements Following Muscle Damaging Exercise. Nutrients 2016, 8, 642. [Google Scholar] [CrossRef] [PubMed]
- Lamprecht, M.; Bogner, S.; Schippinger, G.; Steinbauer, K.; Fankhauser, F.; Hallstroem, S.; Schuetz, B.; Greilberger, J.F. Probiotic supplementation affects markers of intestinal barrier, oxidation, and inflammation in trained men; a randomized, double-blinded, placebo-controlled trial. J. Int. Soc. Sports Nutr. 2012, 9, 45. [Google Scholar] [CrossRef] [PubMed]
- Cox, A.J.; Pyne, D.B.; Saunders, P.U.; Fricker, P.A. Oral administration of the probiotic Lactobacillus fermentum VRI-003 and mucosal immunity in endurance athletes. Br. J. Sports Med. 2010, 44, 222–226. [Google Scholar] [CrossRef] [PubMed]
- Gleeson, M.; Bishop, N.C.; Oliveira, M.; Tauler, P. Daily probiotic’s (Lactobacillus casei Shirota) reduction of infection incidence in athletes. Int. J. Sport Nutr. Exerc. Metab. 2011, 21, 55–64. [Google Scholar] [CrossRef] [PubMed]
- Haywood, B.A.; Black, K.E.; Baker, D.; McGarvey, J.; Healey, P.; Brown, R.C. Probiotic supplementation reduces the duration and incidence of infections but not severity in elite rugby union players. J. Sci. Med. Sport 2014, 17, 356–360. [Google Scholar] [CrossRef] [PubMed]
- West, N.P.; Pyne, D.B.; Peake, J.M.; Cripps, A.W. Probiotics, immunity and exercise: A review. Exerc. Immunol. Rev. 2009, 15, 107–126. [Google Scholar] [PubMed]
- Edvardsen, E.; Scient, C.; Hansen, B.H.; Holme, I.M.; Dyrstad, S.M.; Anderssen, S.A. Reference values for cardiorespiratory response and fitness on the treadmill in a 20- to 85-year-old population. Chest 2013, 144, 241–248. [Google Scholar] [CrossRef] [PubMed]
- Faulhaber, M.; Gatterer, H.; Haider, T.; Patterson, C.; Burtscher, M. Intermittent hypoxia does not affect endurance performance at moderate altitude in well-trained athletes. J. Sports Sci. 2010, 28, 513–519. [Google Scholar] [CrossRef] [PubMed]
- Widner, B.; Werner, E.R.; Schennach, H.; Wachter, H.; Fuchs, D. Simultaneous measurement of serum tryptophan and kynurenine by HPLC. Clin. Chem. 1997, 43, 2424–2426. [Google Scholar] [PubMed]
- Neurauter, G.; Scholl-Bürgi, S.; Haara, A.; Geisler, S.; Mayersbach, P.; Schennach, H.; Fuchs, D. Simultaneous measurement of phenylalanine and tyrosine by high performance liquid chromatography (HPLC) with fluorescence detection. Clin. Biochem. 2013, 46, 1848–1851. [Google Scholar] [CrossRef] [PubMed]
- Geisler, S.; Mayersbach, P.; Becker, K.; Schennach, H.; Fuchs, D.; Gostner, J.M. Serum tryptophan, kynurenine, phenylalanine, tyrosine and neopterin concentrations in 100 healthy blood donors. Pteridines 2015, 26, 31–36. [Google Scholar] [CrossRef]
- Cohen, J.W. Statistical Power Analysis for the Behavioral Sciences, 2nd ed.; Lawrence Erlbaum Associates Publishers: Mahwah, NJ, USA, 1988; pp. 284–287. [Google Scholar]
- Areces, F.; González-Millán, C.; Salinero, J.J.; Abian-Vicen, J.; Lara, B.; Gallo-Salazar, C.; Ruiz-Vicente, D.; Del Coso, J. Changes in serum free amino acids and muscle fatigue experienced during a half-ironman triathlon. PLoS ONE 2015, 10, e0138376. [Google Scholar] [CrossRef] [PubMed]
- Schlittler, M.; Goiny, M.; Agudelo, L.Z.; Venckunas, T.; Brazaitis, M.; Skurvydas, A.; Kamandulis, S.; Ruas, J.L.; Erhardt, S.; Westerblad, H.; et al. Endurance exercise increases skeletal muscle kynurenine aminotransferases and plasma kynurenic acid in humans. Am. J. Physiol. Cell Physiol. 2016, 310, C836–C840. [Google Scholar] [CrossRef] [PubMed]
- Meeusen, R. Exercise, nutrition and the brain. Sports Med. 2014, 44 (Suppl. 1), S47–S56. [Google Scholar] [CrossRef] [PubMed]
- Agudelo, L.Z.; Femenía, T.; Orhan, F.; Porsmyr-Palmertz, M.; Goiny, M.; Martinez-Redondo, V.; Correia, J.C.; Izadi, M.; Bhat, M.; Schuppe-Koistinen, I.; et al. Skeletal muscle PGC-1α1 modulates kynurenine metabolism and mediates resilience to stress-induced depression. Cell 2014, 159, 33–45. [Google Scholar] [CrossRef] [PubMed]
- Desbonnet, L.; Garrett, L.; Clarke, G.; Bienenstock, J.; Dinan, T.G. The probiotic Bifidobacteria infantis: An assessment of potential antidepressant properties in the rat. J. Psychiatr. Res. 2008, 43, 164–174. [Google Scholar] [CrossRef] [PubMed]
- Hao, Q.; Dong, B.R.; Wu, T. Probiotics for preventing acute upper respiratory tract infections. Cochrane Database Syst. Rev. 2015, 2, CD006895. [Google Scholar]
- Pyne, D.B.; West, N.P.; Cox, A.J.; Cripps, A.W. Probiotics supplementation for athletes—Clinical and physiological effects. Eur. J. Sport Sci. 2015, 15, 63–72. [Google Scholar] [CrossRef] [PubMed]
- West, N.P.; Pyne, D.B.; Cripps, A.W.; Hopkins, W.G.; Eskesen, D.C.; Jairath, A.; Christophersen, C.T.; Conlon, M.A.; Fricker, P.A. Lactobacillus fermentum (PCC®) supplementation and gastrointestinal and respiratory-tract illness symptoms: A randomised control trial in athletes. Nutr. J. 2011, 10, 30. [Google Scholar] [CrossRef] [PubMed]
- Gleeson, M. Nutritional support to maintain proper immune status during intense training. Nestle Nutr. Inst. Workshop Ser. 2013, 75, 85–97. [Google Scholar] [PubMed]
- Ho, P.M.; Bryson, C.L.; Rumsfeld, J.S. Medication adherence: Its importance in cardiovascular outcomes. Circulation 2009, 119, 3028–3035. [Google Scholar] [CrossRef] [PubMed]
- Carretti, N.; Florio, P.; Bertolin, A.; Costa, C.V.; Allegri, G.; Zilli, G. Serum fluctuations of total and free tryptophan levels during the menstrual cycle are related to gonadotrophins and reflect brain serotonin utilization. Hum. Reprod. 2005, 20, 1548–1553. [Google Scholar] [CrossRef] [PubMed]

| Variable | Unit | Probiotics (n = 14) Mean ± SD | Placebo (n = 15) Mean ± SD |
|---|---|---|---|
| Gender | male/female | 8/6 | 5/10 |
| Age | year | 25.7 ± 3.5 | 26.6 ± 3.5 |
| BMI | kg/m2 | 22.2 ± 1.5 | 21.2 ± 2.7 |
| Weight | kg | 67.4 ± 9.6 | 62.9 ± 11.1 |
| Body cell mass | kg | 31.2 ± 6.6 | 28.7 ± 7.4 |
| Total body fat | % | 20.1 ± 5.7 | 19.5 ± 4.4 |
| VO2max | mL/kg/min | 55.1 ± 6.4 | 47.5 ± 7.1 ** |
| Pmax | watt | 325 ± 54.2 | 274 ± 51.6 * |
| Prel | watt/kg | 4.8 ± 0.3 | 4.3 ± 0.4 ** |
| PTT | watt | 222 ± 41.9 | 181 ± 38.3 * |
| Energy intake | kcal/day | 2821 ± 1374 | 2840 ± 1161 |
| REE | kcal/day | 1602 ± 206 | 1519 ± 2031 |
| Protein | % | 14.9 ± 3.3 | 15.0 ± 3.5 |
| Carbohydrates | % | 49.5 ± 12.4 | 49.3 ± 12.7 |
| Fat | % | 32.5 ± 10.8 | 33.0 ± 12.1 |
| Fibers | g | 33.0 ± 10.1 | 32.0 ± 14.2 |
| Alcohol | g | 11.1 ± 10.7 | 9.4 ± 9.5 |
| Water | L | 3.38 ± 0.58 | 3.37 ± 0.84 |
| Food | Energy (kcal) | Protein (g) | Carbohydrates (g) | Fat (g) |
|---|---|---|---|---|
| 2 wheat rolls 100 g | 260 | 8.70 | 52.7 | 0.90 |
| Marmalade/jam 50 g | 114 | 0.30 | 28.0 | 0.00 |
| 250 mL tea | 5 | 0.75 | 0.25 | 0.25 |
| 250 mL water | - | - | - | - |
| Total | 379 | 9.75 | 80.95 | 1.15 |
| Meal energy (%) | 11 | 88 | 1 |
| Probiotics (n = 14) | Baseline PRE | Baseline POST | Week 12 PRE | Week 12 POST |
| Tryptophan (µmol/L) | 70.07 ± 3.20 a,e | 57.99 ± 2.47 b | 68.64 ± 2.12 c,k | 58.76 ± 2.11 d |
| Kynurenine (µmol/L) | 1.98 ± 0.11 | 1.97 ± 0.07 | 1.83 ± 0.10 | 1.92 ± 0.11 |
| Kyn/Trp (µmol/mmol) | 28.35 ± 1.16 f | 34.50 ± 1.46 | 26.94 ± 1.51 l | 33.32 ± 2.19 |
| Neopterin (nmol/L) | 5.19 ± 0.23 g | 8.43 ± 1.00 | 4.92 ± 0.31 m | 7.74 ± 0.86 |
| Tyrosine (µmol/L) | 138.58 ± 29.96 h | 145.06 ± 6.23 | 147.25 ± 24.01 n | 149.22 ± 5.60 |
| Phenylalanine (µmol/L) | 69.59 ± 8.27 i | 68.72 ± 2.06 | 72.16 ± 1.99 o | 71.76 ± 1.90 |
| Phe/Tyr (mol/mol) | 0.52 ± 0.08 j | 0.48 ± 0.01 | 0.50 ± 0.03 p | 0.49 ± 0.02 |
| Placebo (n = 15) | Baseline PRE | Baseline POST | Week 12 PRE | Week 12 POST |
| Tryptophan (µmol/L) | 62.27 ± 1.72 | 58.27 ± 2.37 | 61.50 ± 1.84 | 52.26 ± 1.86 |
| Kynurenine (µmol/L) | 1.77 ± 0.13 | 2.02 ± 0.07 | 1.75 ± 0.08 | 1.77 ± 0.09 |
| Kyn/Trp (µmol/mmol) | 28.38 ± 1.81 | 34.49 ± 2.17 | 28.49 ± 1.03 | 34.03 ± 1.51 |
| Neopterin (nmol/L) | 6.63 ± 0.95 | 10.48 ± 1.56 | 5.65 ± 0.70 | 9.55 ± 2.06 |
| Tyrosine (µmol/L) | 131.15 ± 5.28 | 137.40 ± 5.42 | 126.41 ± 6.29 | 129.28 ± 5.76 |
| Phenylalanine (µmol/L) | 69.23 ± 2.45 | 68.55 ± 1.87 | 72.53 ± 1.67 | 70.09 ± 2.71 |
| Phe/Tyr (mol/mol) | 0.53 ± 0.02 | 0.50 ± 0.02 | 0.59 ± 0.03 | 0.55 ± 0.02 |
| URTI | Baseline PRE | Baseline POST | Week 12 PRE | Week 12 POST |
|---|---|---|---|---|
| yes | 28.9 ± 1.7 | 34.9 ± 6.0 | 31.1 ± 5.1 | 38.9 ± 7.3 |
| no | 28.2 ± 6.5 | 34.4 ± 7.2 | 26.7 ± 4.3 | 32.0 ± 6.0 |
| U/p-value | 0.535/0.592 | 0.102/0.919 | 2.039/0.041 | 2.090/0.037 |
| Illness Symptoms | Baseline U | p | Week 12 U | p | |
|---|---|---|---|---|---|
| URTI | PRO | 0.212 | 0.832 | 0.155 | 0.877 |
| PLA | 0.748 | 0.454 | 0.399 | 0.690 | |
| Runny nose | PRO | 0.212 | 0.832 | 0.329 | 0.742 |
| PLA | 0 | 1 | 1.080 | 0.280 | |
| Cough | PRO | 0.931 | 0.352 | 0.362 | 0.717 |
| PLA | 1.497 | 0.134 | 1.497 | 0.134 | |
| Sore throat | PRO | 1.041 | 0.298 | 0.866 | 0.386 |
| PLA | 0.374 | 0.708 | 1.497 | 0.134 | |
| Fever | PRO | 0.823 | 0.411 | 0 | 1 |
| PLA | 0 | 1 | 0 | 1 | |
| Weakness | PRO | 0.866 | 0.386 | 0 | 1 |
| PLA | 0.519 | 0.604 | 0.519 | 0.604 |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Strasser, B.; Geiger, D.; Schauer, M.; Gostner, J.M.; Gatterer, H.; Burtscher, M.; Fuchs, D. Probiotic Supplements Beneficially Affect Tryptophan–Kynurenine Metabolism and Reduce the Incidence of Upper Respiratory Tract Infections in Trained Athletes: A Randomized, Double-Blinded, Placebo-Controlled Trial. Nutrients 2016, 8, 752. https://doi.org/10.3390/nu8110752
Strasser B, Geiger D, Schauer M, Gostner JM, Gatterer H, Burtscher M, Fuchs D. Probiotic Supplements Beneficially Affect Tryptophan–Kynurenine Metabolism and Reduce the Incidence of Upper Respiratory Tract Infections in Trained Athletes: A Randomized, Double-Blinded, Placebo-Controlled Trial. Nutrients. 2016; 8(11):752. https://doi.org/10.3390/nu8110752
Chicago/Turabian StyleStrasser, Barbara, Daniela Geiger, Markus Schauer, Johanna M. Gostner, Hannes Gatterer, Martin Burtscher, and Dietmar Fuchs. 2016. "Probiotic Supplements Beneficially Affect Tryptophan–Kynurenine Metabolism and Reduce the Incidence of Upper Respiratory Tract Infections in Trained Athletes: A Randomized, Double-Blinded, Placebo-Controlled Trial" Nutrients 8, no. 11: 752. https://doi.org/10.3390/nu8110752
APA StyleStrasser, B., Geiger, D., Schauer, M., Gostner, J. M., Gatterer, H., Burtscher, M., & Fuchs, D. (2016). Probiotic Supplements Beneficially Affect Tryptophan–Kynurenine Metabolism and Reduce the Incidence of Upper Respiratory Tract Infections in Trained Athletes: A Randomized, Double-Blinded, Placebo-Controlled Trial. Nutrients, 8(11), 752. https://doi.org/10.3390/nu8110752






