Moderate Amounts of Vitamin D3 in Supplements are Effective in Raising Serum 25-Hydroxyvitamin D from Low Baseline Levels in Adults: A Systematic Review
Abstract
:1. Introduction
2. Methods
2.1. Search Strategy
2.2. Calculation of Rate Constant and Statistical Analysis
3. Results
| 25(OH)D Measurement (nmol/L) | |||||
|---|---|---|---|---|---|
| Study | Baseline Level ± SD (or CI range) by Dose (μg) | Net rise in 25(OH)D by Dose (μg) | Rate Constant: nmol/L per μg by Dose Level (μg) and Time (month) | ||
| Aloia et al. 2005 [13] | 43.0 ± 16.6 (0) | 23.9 (20) | 1.20 (20) at 3 months | ||
| 48.3 ± 20.9 (20) | |||||
| Andersen et al. 2008 [14] | M 20.0 (15.0, 25.2) (0) | M 16.0 (10) M 31.8 (20) F 39.4 (10) F 38.4 (20) | M 1.60 (10) M 1.59 (20) F 3.94 (10) F 1.29 (20) at 6 months | ||
| M 22.9 (12.6, 28.2) (10) | |||||
| M 18.9 (13.6, 29.2) (20) | |||||
| F 11.7 (7.5, 19.4) (0) | |||||
| F 10.0 (6.9, 14.3) (10) | |||||
| F 14.0 (8.3, 17.5) (20) | |||||
| Bischoff-Ferrari et al.2006 [15] | F 63.0 ± 30.3 (0) | F 29 (17.5) | F 1.66 (17.5) | ||
| F 70.0 ± 33.0 (17.5) | At 3 years | ||||
| Bolton-Smith et al. 2007 [16] | 57.0 ± 15.2 (0) | 22.0(10) | 2.2 (10) | ||
| 62.5 ± 15.5 (10) | at 12 months | ||||
| Brazier et al. 2005 [17] | 17.5 (0) | 44.2 (10) | 4.42 (10) | ||
| 18.25 (10) | at 12 months | ||||
| Bunout et al. 2006 [18] | 32.8 ± 6.8 (0) | 30.0 (10) | 3.0 (10) | ||
| 31.0 ± 5.5 (10) | at 9 months | ||||
| Cashman et al. 2009 [19] | 58.8 (44,78) (0) | 18.6 (5) 32.4(10) 39.0 (15) | 3.72 (5) | ||
| 51.8 (41, 71) (5) | 3.24 (10) | ||||
| 54.3 (43, 72) (10) | 2.6 (15) | ||||
| 55.1 (40, 70) (15) | at 5 months | ||||
| Chel et al. 2008 [20] | 25.2 ± 12.1 (0) | 35.9 (15) | 2.39 (15) | ||
| 23.0 ± 8.3 (15) | at 2 months | ||||
| Gallagher et al. 2012 [21] | 37.7 ± 9.1 (0) | 32.5 (10) * | 3.25 (10) | ||
| 37.8 ± 10.8 (10) | at 12 months | ||||
| Islam et al. 2010 [22] | 35.0 ± 9.4) (0) | 31.6 (10) | 3.16 (10) | ||
| 37.1 ±12.1 (10) | at 12 months | ||||
| Karkkaine et al. 2010 [23] | 49.2 ± 17.7 (0) | 23.7 (20) | 1.27 (20 | ||
| 50.1 ± 18.8 (20) | at 36 months | ||||
| Nelson et al. 2009 [24] | 61.9 ± 22.6 (0) | 25.5 (20) | 1.28 (20) | ||
| 62.1 ± 24.0 (20) | at 12 months | ||||
| Pfeifer et al. 2009 [25] | 54 ± 18 (0) | 26 (20) | 1.3 (20) | ||
| 55 ± 18 (20) | at 12 months | ||||
| Pignotti et al. 2010 [26] | 52.9 ± 21.4 (0) | 6.9 (10) | 0.69 (10) | ||
| 46.7 ± 14.0 (10) | At 3 months | ||||
| Smith et al. 2009 [27] | 36 ± 17 (0) | 15 (10) | 1.5 (10) | ||
| 44 ± 18 (10) | at 6 months | ||||
| Talwar et al. 2007 [28] | 43.2 ± 16.8 (0) | 29.6 (20) | 1.48 (20) | ||
| 46.9 ± 20.6 (20) | at 3 months | ||||
| Viljakainen et al. 2006 [29] | 52.2 ± 19.9 (0) | 10.9 (5) 21.4 (10) 35.1 (20) | 2.18 (5) | ||
| 46.0 ± 14.3 (5) | 2.14 (10) | ||||
| 46.5 ± 10.2 (10) | 1.76 (20) | ||||
| 44.1 ± 13.5 (20) | at 12 weeks | ||||
| Bonjour et al. 2013 [30] | 16.2 ± 0.6 (0) | 20.2 (10) | 2.02 (10) | ||
| 19.2 ± 1.2 (10) | at 8 weeks | ||||
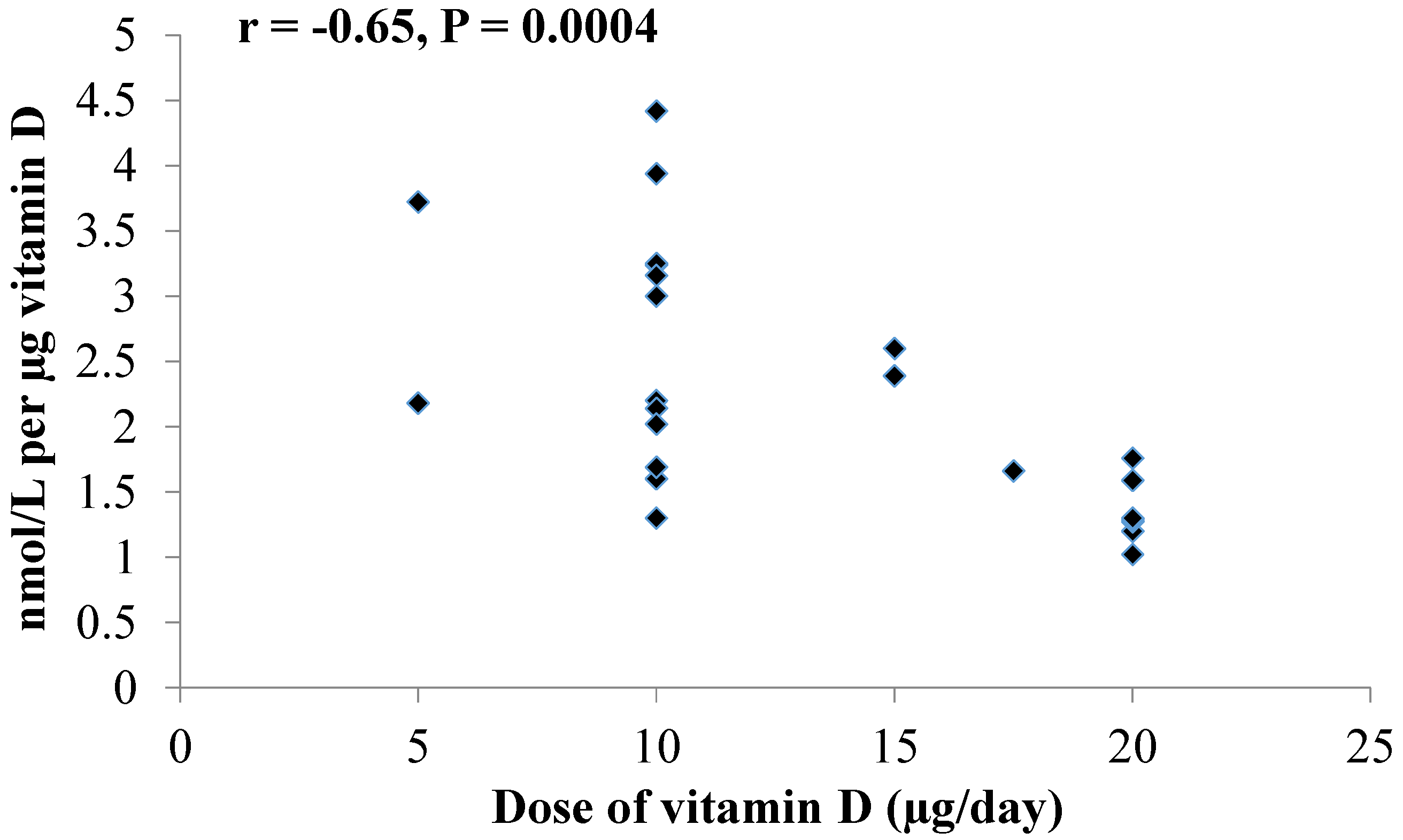
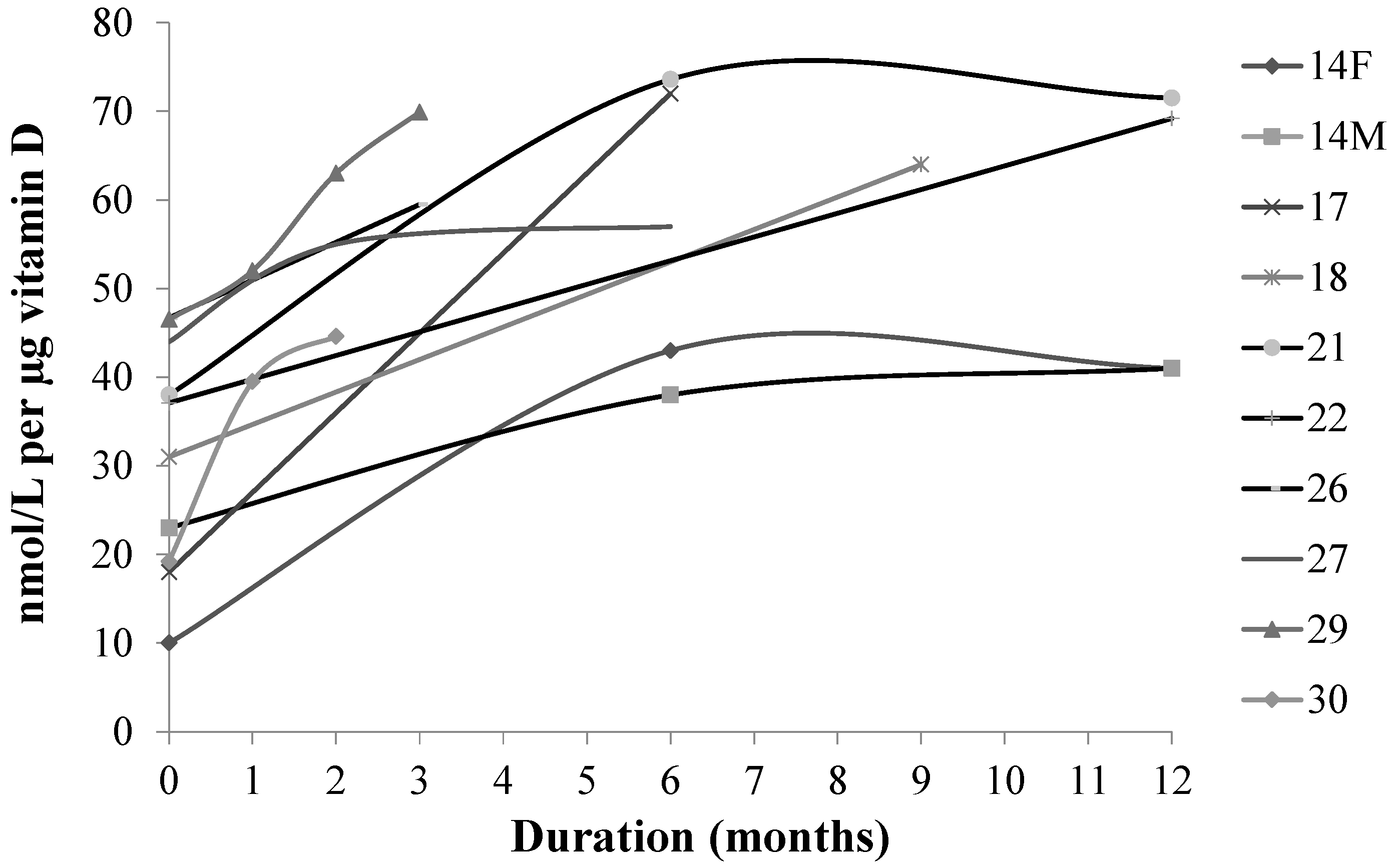
4. Discussion
Supplementary Files
Supplementary File 1Author Contributions
Appendix
| Design and Population at Baseline | |||||||
|---|---|---|---|---|---|---|---|
| Study | Dose μg (IU) | Form | Duration (Sampling) | Subjects | Background Intake and Sun Exposure | Assay | N per Group |
| Supplement Studies | |||||||
| Aloia et al. 2005 [13] | 20 (800) | Capsule D3 + Ca | 3 months | F only, 65–104 years African American BMI | Mean intake 4.6 μg/day | RIA | 280-total 104-placebo |
| Andersen et al. 2008 [14] | 10 (400) 20 (800) | Tablet D3 | 1 year (6, 12 months) | F & M 18–64 years; healthy; Pakistani; Denmark BMI ~ 27 | Dietary intake averaged 1.7 μg/day | HPLC | 199-total 37(F), 27(M)-placebo |
| Bischoff-Ferrari et al. 2006 [15] | 17.5 (700) | Tablet D3 + Ca | 3 y | ≥65 years; healthy; M & F; Boston USA (mixed ethnicity) BMI ~ 26.5 | No supplement since at least 2 month ago | CPBA | 445-total 125(F), placebo |
| Bolton-Smith et al. 2007 [16] | 10 (400) | Tablet D3 + 1000 mg Ca | 2 year (12, 24 months) | F only ≥60 years; Caucasian, Scotland BMI ~ 26 | No supplement that provided in excess of 10 μg vit D; Vit D intake ~5 μg/day | RIA | 244-total 58-placebo |
| Brazier et al. 2005 [17] | 10 (400) | Tablet D3 + 500 mg Ca | 1 year | F only, >65 years; France | Recruited having 25(OH)D <30 nmol/L; Vit D intake ~2 μg/day | CPBA | 191 total 96-placebo |
| Bunout et al. 2006 [18] | 10 (400) | Tablets with 800 mg Ca | 9 months | F & M, ≥70 years; Chile | Recruited having 25(OH)D <40 nmol/L | Not stated | 96-total 46-placebo |
| Cashman et al. 2009 [19] | 5 (200) 10 (400) 15 (600) | Capsule D3 | 22 weeks | F & M; ≥64 years; Caucasian, Ireland | Intervention during winter months | ELISA | 225-total 61-placebo |
| Chel et al. 2008 [20] | 15 (600) | Tablet D3 | 4.5 months | F & M 84 ± 6.2 years; nursing home residents; Caucasian, Netherlands | Outside ≤ once/week; no use of vitamin D supplementation; vitamin D fortified food or drink ≤1/day | RIA | 338-total 172-placebo |
| Gallagher et al. 2012 [21] | 10 (400) | Capsule D3 | 1 year | F only; 57–90 years; Caucasian Omaha USA | Screened in late winter; Chose low (<50 nmol/L levels) | RIA | 41-total 21-placebo |
| Islam et al. 2010 [22] | 10 (400) | Tablet D (form assumed to be D3) | 1 year | F 18–36 years; Bangladeshi, Bangladesh | Only hands and face uncovered | Enzyme immune- assay | 200-total 50-placebo |
| Karkkaine et al. 2010 [23] | 20 (800) | Tablet D3 | 3 years | F only 65–71 years; Finland BMI ~ 27.7 | Open-label trial of D + Ca asked all participants to follow usual diet (not specified) | RIA | 3139-total 1573-placebo |
| Nelson et al. 2009 [24] | 20 (800) | Capsule D3 | 21 weeks | 19–35 years; healthy; F; USA BMI = 25.5 | Baseline intake 3.5 μg/day; study run in winter | RIA | 112-total 31-placebo |
| Pfeifer et al. 2009 [25] | 20 (800) | Tablet D3 + Ca | 1 year | F & M; 70–94 years; community-dwelling seniors. Germany and Austria | Vitamin D supplementation was exclusion criterion | RIA | 242-total 121-placebo |
| Pignotti et al. 2010 [26] | 10 (400) | Tablet D3 | 3 months | F only 62 ± 8 years; Caucasian Brazil BMI: 26.7 | Dietary intake at baseline 3.5 μg/day Vit D supplement use excluded | RIA | 64-total 29-placebo |
| Smith et al. 2009 [27] | 10 (400) | Tablet D3 | 5 months | M & F 42 ± 12 years BMI: 19 ± 6 | Conducted during winter in Antarctica | RIA | 55-total 4(F), 3(M)-placebo |
| Talwar et al. 2007 [28] | 20(800) | capsule D3 | 2 years | F only 59.9 ± 6.2 years; African American, New York USA BMI = 29 | Dietary intake : 4.6 μg/day Exclusion of >10 μg/day vitamin D 6 mo before entry | RIA | 208-total 104-placebo |
| Viljakainen et al. 2006 [29] | 5 (200) 10 (400) 20 (800) | Tablet D3 | 12 weeks | F only 65–85 years; Helsinki Finland | Dietary intake at baseline ~10 μg/day | HPLC | 49-total 12-placebo |
| Bonjour et al. 2013 [30] | 10 (400) | Yogurt + 800 mg Ca | 8 weeks | F only >60 years; Institutionalized France BMI = 26 | Limited sun exposure, winter time, no supplementation | Immuno-Diagnostics System | 89-total 27-placebo |
Conflicts of Interest
References
- Hilger, J.; Friedel, A.; Herr, R.; Rausch, T.; Roos, F.; Wahl, D.A.; Pierroz, D.D.; Weber, P.; Hoffmann, K. A systematic review of vitamin D status in populations worldwide. Br. J. Nutr. 2014, 111, 23–45. [Google Scholar] [CrossRef] [PubMed]
- Calvo, M.S.; Whiting, S.J.; Barton, C.N. Vitamin D intake: A Global Perspective of Current Status. J. Nutr. 2005, 135, 310–316. [Google Scholar] [PubMed]
- Institute of Medicine (IOM). Dietary Reference Intakes for Calcium and Vitamin D; The National Academy Press: Washington, DC, USA, 2012. [Google Scholar]
- Holick, M.F.; Binkley, N.C.; Bischoff-Ferrari, H.A.; Gordon, C.M.; Hanley, D.A.; Heaney, R.P.; Murad, M.H.; Weaver, C.M.; Endocrine, S. Evaluation, treatment, and prevention of vitamin D deficiency: An Endocrine Society clinical practice guideline. J. Clin. Endocrinol. Metab. 2011, 96, 1911–1930. [Google Scholar] [CrossRef] [PubMed]
- Whiting, S.J.; Green, T.J.; Calvo, M.S. Vitamin D intakes in North America and Asia-Pacific countries are not sufficient to prevent vitamin D insufficiency. J. Steroid Biochem. Mol. Biol. 2007, 103, 626–630. [Google Scholar] [CrossRef] [PubMed]
- Whiting, S.J.; Langlois, K.A.; Vatanparast, H.; Greene-Finestone, L.S. The vitamin D status of Canadians relative to the 2011 Dietary Reference Intakes: An examination in children and adults with and without supplement use. Am. J. Clin. Nutr. 2011, 94, 128–135. [Google Scholar] [CrossRef] [PubMed]
- Position Paper on Sun Exposure for Australia. Available online: http://www.cancer.org.au/content/pdf/CancerControlPolicy/PositionStatements/PSRisksBenefitsSunExposure03May07.pdf (accessed on 12 December 2014).
- Sacco, J.E.; Dodd, K.W.; Kirkpatrick, S.I.; Tarasuk, V. Voluntary food fortification in the United States: Potential for excessive intakes. Eur. J. Clin. Nutr. 2013, 67, 592–597. [Google Scholar] [CrossRef] [PubMed]
- Bouillon, R.; van Schoor, N.M.; Gielen, E.; Boonen, S.; Mathieu, C.; Vanderschueren, D.; Lips, P. Optimal vitamin D status: A critical analysis on the basis of evidence-based medicine. J. Clin. Endocrinol. Metab. 2013, 98, E1283–E1304. [Google Scholar] [CrossRef] [PubMed]
- Heaney, R.P.; Davies, K.M.; Chen, T.C.; Holick, M.F.; Barger-Lux, M.J. Human serum 25-hydroxycholecalciferol response to extended oral dosing with cholecalciferol. Am. J. Clin. Nutr. 2003, 77, 204–210. [Google Scholar] [PubMed]
- Black, L.J.; Seamans, K.M.; Cashman, K.D.; Kiely, M. An updated systematic review and meta-analysis of the efficacy of vitamin D food fortification. J. Nutr. 2012, 142, 1102–1108. [Google Scholar] [CrossRef] [PubMed]
- McKenna, M.J.; Murray, B.F. Vitamin D dose response is underestimated by Endocrine Society’s Clinical Practice Guideline. Endocr. Connect. 2013, 2, 87–95. [Google Scholar] [CrossRef] [PubMed]
- Aloia, J.F.; Talwar, S.A.; Pollack, S.; Yeh, J. A randomized controlled trial of vitamin D3 supplementation in African American women. Arch. Intern. Med. 2005, 165, 1618–1623. [Google Scholar] [CrossRef] [PubMed]
- Andersen, R.; Molgaard, C.; Skovgaard, L.T.; Brot, C.; Cashman, K.D.; Jakobsen, J.; Lamberg-Allardt, C.; Ovesen, L. Effect of vitamin D supplementation on bone and vitamin D status among Pakistani immigrants in Denmark: A randomised double-blinded placebo-controlled intervention study. Br. J. Nutr. 2008, 100, 197–207. [Google Scholar] [CrossRef] [PubMed]
- Bischoff-Ferrari, H.A.; Orav, E.J.; Dawson-Hughes, B. Effect of cholecalciferol plus calcium on falling in ambulatory older men and women: A 3-year randomized controlled trial. Arch. Inter. Med. 2006, 166, 424–430. [Google Scholar]
- Bolton-Smith, C.; McMurdo, M.E.; Paterson, C.R.; Mole, P.A.; Harvey, J.M.; Fenton, S.T.; Prynne, C.J.; Mishra, G.D.; Shearer, M.J. Two-year randomized controlled trial of vitamin K1 (phylloquinone) and vitamin D3 plus calcium on the bone health of older women. J. Bone Min. Res. 2007, 22, 509–519. [Google Scholar] [CrossRef]
- Brazier, M.; Grados, F.; Kamel, S.; Mathieu, M.; Morel, A.; Maamer, M.; Sebert, J.L.; Fardellone, P. Clinical and laboratory safety of one year’s use of a combination calcium + vitamin D tablet in ambulatory elderly women with vitamin d insufficiency: Results of a multicenter, randomized, double-blind, placebo-controlled study. Clin. Ther. 2005, 27, 1885–1893. [Google Scholar] [CrossRef] [PubMed]
- Bunout, D.; Barrera, G.; Leiva, L.; Gattas, V.; de la Maza, M.; Avendan˜o, M.; Hirsch, S. Effects of vitamin D supplementation and exercise training on physical performance in Chilean vitamin D deficient elderly subjects. Exp. Gerontol. 2006, 41, 746–752. [Google Scholar] [CrossRef] [PubMed]
- Cashman, K.D.; Wallace, J.M.; Horigan, G.; Hill, T.R.; Barnes, M.S.; Lucey, A.J.; Bonham, M.P.; Taylor, N.; Duffy, E.M.; Seamans, K.; et al. Estimation of the dietary requirement for vitamin D in free-living adults ≥64 y of age. Am. J. Clin. Nutr. 2009, 89, 1366–1374. [Google Scholar] [CrossRef] [PubMed]
- Chel, V.; Wijnhoven, H.A.; Smit, J.H.; Ooms, M.; Lips, P. Efficacy of different doses and time intervals of oral vitamin D supplementation with or without calcium in elderly nursing home residents. Osteoporos. Int. 2008, 19, 663–671. [Google Scholar] [CrossRef] [PubMed]
- Gallagher, J.C.; Sai, A.; Templin, T.; Smith, L. Dose response to vitamin D supplementation in postmenopausal women: A randomized trial. Ann. Intern. Med. 2012, 156, 425–437. [Google Scholar] [CrossRef]
- Islam, M.Z.; Shamim, A.A.; Viljakainen, H.T.; Akhtaruzzaman, M.; Jehan, A.H.; Khan, H.U.; al-Arif, F.A.; Lamberg-Allardt, C. Effect of vitamin D, calcium and multiple micronutrient supplementation on vitamin D and bone status in Bangladeshi premenopausal garment factory workers with hypovitaminosis D: A double-blinded, randomised, placebo-controlled 1-year intervention. Br. J. Nutr. 2010, 104, 241–247. [Google Scholar] [CrossRef] [PubMed]
- Karkkainen, M.K.; Tuppurainen, M.; Salovaara, K.; Sandini, L.; Rikkonen, T.; Sirola, J.; Honkanen, R.; Arokoski, J.; Alhava, E.; Kroger, H. Does daily vitamin D 800 IU and calcium 1000 mg supplementation decrease the risk of falling in ambulatory women aged 65–71 years? A 3-year randomized population-based trial (OSTPRE-FPS). Maturitas 2010, 65, 359–365. [Google Scholar] [CrossRef] [PubMed]
- Nelson, M.L.; Blum, J.M.; Hollis, B.W.; Rosen, C.; Sullivan, S.S. Supplements of 20 μg/d cholecalciferol optimized serum 25-hydroxyvitamin D concentrations in 80% of premenopausal women in winter. J. Nutr. 2009, 139, 540–546. [Google Scholar] [CrossRef] [PubMed]
- Pfeifer, M.; Begerow, B.; Minne, H.W.; Suppan, K.; Fahrleitner-Pammer, A.; Dobnig, H. Effects of a long-term vitamin D and calcium supplementation on falls and parameters of muscle function in community-dwelling older individuals. Osteoporos. Int. 2009, 20, 315–322. [Google Scholar] [CrossRef] [PubMed]
- Pignotti, G.A.P.; Genaro, P.S.; Pinheiro, M.M.; Szejnfeld, V.L.; Martini, L.A. Is a lower dose of vitamin D supplementation enough to increase 25(OH)D status in a sunny country? Eur. J. Nutr. 2010, 49, 277–283. [Google Scholar] [CrossRef] [PubMed]
- Smith, S.A.; Gardner, K.K.; Locke, J.; Zwart, S.R. Vitamin D supplementation during Antarctic winter. Am. J. Clin. Nutr. 2009, 89, 1092–1098. [Google Scholar] [CrossRef] [PubMed]
- Talwar, S.A.; Aloia, J.F.; Pollack, S.; Yeh, J.K. Dose response to vitamin D supplementation among postmenopausal African American women. Am. J. Clin. Nutr. 2007, 86, 1657–1662. [Google Scholar] [PubMed]
- Viljakainen, H.T.; Palssa, A.; Karkkainen, M.; Jakobsen, J.; Lamberg-Allardt, C. How much vitamin D3 do the elderly need? J. Am. Coll. Nutr. 2006, 25, 429–435. [Google Scholar] [CrossRef] [PubMed]
- Bonjour, J.P.; Benoit, V.; Payen, F.; Kraenzlin, M. Consumption of yogurts fortified in vitamin D and calcium reduces serum parathyroid hormone and markers of bone resorption: A double-blind randomized controlled trial in institutionalized elderly women. J. Clin. Endocrinol. Metab. 2013, 98, 2915–2921. [Google Scholar] [CrossRef] [PubMed]
- Social Science Statistics. Available online: http://www.socscistatistics.com (accessed on 31 January 2015).
- Calvo, M.S.; Barton, C.N.; Whiting, S.J. Vitamin D fortification in the US and Canada: Current status and data needs. Am J. Clin. Nutr. 2004, 80, 1710S–1716S. [Google Scholar] [PubMed]
- Fulgoni, V.L.; Keast, D.R.; Bailey, R.L.; Dwyer, J. Foods, fortificants, and supplements: Where do Americans get their nutrients? J. Nutr. 2011, 141, 1847–1854. [Google Scholar] [CrossRef] [PubMed]
- Blum, M.; Dallal, G.E.; Dawson-Hughes, B. Body size and serum 25 hydroxyvitamin D response to oral supplements in healthy older adults. J. Am. Coll. Nutr. 2008, 27, 274–279. [Google Scholar] [CrossRef] [PubMed]
- Waterhouse, M.; Tran, B.; Armstrong, B.K.; Baxter, C.; Ebeling, P.R.; English, D.R.; Gebski, V.; Hill, C.; Kimlin, M.G.; Lucas, R.M.; et al. Environmental, personal, and genetic determinants of response to vitamin D supplementation in older adults. J. Clin. Endocrinol. Metab. 2014, 99, E1332–E1340. [Google Scholar] [CrossRef] [PubMed]
- Heaney, R.P.; Armas, L.A.; French, C. All-Source basal vitamin D inputs are greater than previously thought and cutaneous inputs are smaller. J. Nutr. 2013, 143, 571–575. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Whiting, S.J.; Bonjour, J.-P.; Payen, F.D.; Rousseau, B. Moderate Amounts of Vitamin D3 in Supplements are Effective in Raising Serum 25-Hydroxyvitamin D from Low Baseline Levels in Adults: A Systematic Review. Nutrients 2015, 7, 2311-2323. https://doi.org/10.3390/nu7042311
Whiting SJ, Bonjour J-P, Payen FD, Rousseau B. Moderate Amounts of Vitamin D3 in Supplements are Effective in Raising Serum 25-Hydroxyvitamin D from Low Baseline Levels in Adults: A Systematic Review. Nutrients. 2015; 7(4):2311-2323. https://doi.org/10.3390/nu7042311
Chicago/Turabian StyleWhiting, Susan J, Jean-Philippe Bonjour, Flore Dontot Payen, and Brigitte Rousseau. 2015. "Moderate Amounts of Vitamin D3 in Supplements are Effective in Raising Serum 25-Hydroxyvitamin D from Low Baseline Levels in Adults: A Systematic Review" Nutrients 7, no. 4: 2311-2323. https://doi.org/10.3390/nu7042311
APA StyleWhiting, S. J., Bonjour, J.-P., Payen, F. D., & Rousseau, B. (2015). Moderate Amounts of Vitamin D3 in Supplements are Effective in Raising Serum 25-Hydroxyvitamin D from Low Baseline Levels in Adults: A Systematic Review. Nutrients, 7(4), 2311-2323. https://doi.org/10.3390/nu7042311






