Glycaemic Index and Load Values Tested in Normoglycemic Adults for Five Staple Foodstuffs: Pounded Yam, Pounded Cassava-Plantain, Placali, Attieke and Maize Meal Stiff Porridge
Abstract
:1. Introduction
2. Experimental Section
2.1. Setting
2.2. Subjects
2.3. Study Protocol
2.4. Test Foods Procedure—Collection of Samples and Description/Preparation of Experimental Diets
| Food (Raw Material Used) Agricultural Form | Test Food Description/Preparation |
|---|---|
| White yam tuber based (Dioscorea cayenensis-rotundata; variety Kponan) | Group 1: Pounded yam or yam foutou. Yam has taken the first place in rank with its volume (6,932,950 tons per year), among the consumer crops produced in Côte d’Ivoire [3]. Generally, Kponan variety is mainly used for the preparation of foutou [10]. For pounded yam production, the tubers were peeled, cut into pieces and boiled until soft. The water is then drained off and the pieces pounded in a wooden mortar and pestle until stiff glutinous dough is formed, usually taking 15–30 min [11,12]. |
| Cassava root based food items (Manihot esculenta Crantz; Improved African Cassava (IAC) variety) | Group 2: Attieke (a fermented cassava couscous). The most popular food derived from fermented cassava is attieke [13]. This product is widely consumed in Côte d’Ivoire and in neighbouring countries [14]. It is a traditional foodstuff made by fermentation and steam-cooking cassava root [15]. In Côte d’Ivoire, attieke is mainly prepared from the local variety called Improved African Cassava (IAC), which is bitter, pest-resistant and produces high yields [16,17]. Attieke processing technology comprises peeling the roots, reducing them into mash, inoculating obtained dough with a cooked and fermented cassava pulp and adding palm oil. The fresh mash is fermented for two or three days, mechanically squeezed in order to remove as much water as possible, granulated, sun-dried before sieving and finally steamed to get the final product attieke [14,15]. |
| Group 3: Placali (a fermented cassava paste). Bitter cassava roots are usually used to prepare placali. In Côte d’Ivoire, placali is commonly consumed and it is the second well-known cassava product after attieke [18]. The placali meal was prepared as follows: cassava are peeled, crushed and mixed with a small amount of fermented cassava. The paste obtained is fermented for one to two days and then sieved to remove fibers. The fermented dough is transformed into a gel called “placali” after simmering [19]. | |
| Plantain based (Musa paradisiaca L.; variety ameletia) Musa spp., AAB group, cv. False Horn in ripeness stage 5. | Group 4: Pounded plantain or pounded cassava-plantain (pounded cassava mixed with pounded plantain). The plantain and cassava are washed, peeled and plantain is sliced lengthways into two with a stainless steel knife to cut out the black spots (atrophied seeds). Peeled cassava (750 g) and ripe plantain pieces (2 kg) are cooked in water for 1 h. After cooling, the water was drained and the boiled plantain/cassava was transferred into a traditional wooden mortar and pounded to obtain a smooth consistent paste. Pounded plantain sample was moulded into spherical balls [20]. |
| Maize based (Zea mays L.) | Group 5: Maize meal stiff porridge or cabatôh. This is a traditional Ivorian recipe from a classic starchy staple made from corn dough boiled in water until it forms a stiff porridge-like paste. It is also known as cabatôh or tôh de mais and is made from cornmeal. The maize meal was prepared as follows: flour of maize (1.2 kg) obtained by pounding whole grains in a mortar, is poured into boiling water (3 L) and stirred until a solid paste is formed. |
2.5. Physico-Chemical Analyses
2.6. Blood Glucose Measurements
2.7. Incremental Area under the Curve, Glycaemic Index and Glycaemic Load
2.8. Ethical Considerations
2.9. Data Management
2.10. Statistical Analysis
3. Results
3.1. Descriptive Characteristics of Subjects
3.2. Chemical Composition
3.3. Within-Subject Variation of Reference Food
3.4. Blood Glucose Response to Test Meals
3.5. Glycaemic Index/Load Testing Portions
3.6. Factors that Affect the GI of Tested Foods
| Food Samples | Moisture Content (g/100 g) | Dry Matter Content (g/100 g) | Ashes Content (g/100 g) | Total Dietary Fiber Content (g/100 g) | Proteins Content (g/100 g) | Lipids Content (g/100 g) | Available CHO Content (g/100 g) * | Total Content CHO (g/100 g) * | Energetic Value (kcal/100 g) ** | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Mean (range) | SD | Mean (range) | SD | Mean (range) | SD | Mean (range) | SD | Mean (range) | SD | Mean (range) | SD | Mean (range) | SD | Mean (range) | SD | Mean (range) | SD | |
| Pounded yam | 71.0 c (70.9–71.0) | 0.0 | 29.0 d (29.0–29.1) | 0.0 | 0.8 b (0.8–0.8) | 0.0 | 0.6 b (0.6–0.7) | 0.2 | 1.5 b (1.5–1.6) | 0.0 | 0.8 b (0.8–0.8) | 0.0 | 25.3 c (25.3–25.4) | 0.1 | 25.9 d (25.9–26.0) | 0.0 | 114.3 c (114.0–114.8) | 0.4 |
| Placali | 81.0 a (81.0–81.0) | 0.0 | 19.0 f (19.0–19.0) | 0.0 | 0.8 b (0.8–0.8) | 0.0 | 1.6 a (1.5–1.7) | 0.1 | 0.8 c (0.8–0.9) | 0.0 | 0.0 e (0.0–0.0) | 0.0 | 15.8 d (15.7–15.8) | 0.1 | 17.4 c (17.3–17.4) | 0.0 | 66.5 d (66.7–66.9) | 0.5 |
| Pounded cassava-plantain | 68.3 d (68.2–68.6) | 0.2 | 31.7 c (31.4–31.8) | 0.2 | 0.2 c (0.2–0.3) | 0.0 | 0.6 b (0.6–0.6) | 0.0 | 1.7 b (1.6–1.7) | 0.0 | 0.4 c (0.4–0.4) | 0.0 | 28.8 b (28.5–28.9) | 0.2 | 29.3 b (29.1–29.5) | 0.2 | 125.4 b(124.5–126.1) | 0.3 |
| Maize meal stiff porridge | 73.4 b (73.0–73.7) | 0.4 | 26.6 e (26.3–27.0) | 0.4 | 1.1 a (1.0–1.3) | 0.1 | 1.5 a (1.4–1.6) | 0.1 | 2.2 a (2.1–2.3) | 0.1 | 0.2 d (0.2–0.2) | 0.0 | 21.6 e (21.3–22.0) | 0.4 | 23.1 e (22.7–23.6) | 0.5 | 97.0 e (96.2–98.4) | 1.2 |
| Attieke | 51.2 e (49.3–52.3) | 1.7 | 48.8 b (47.8–50.7) | 1.7 | 0.7 b (0.7–0.7) | 0.0 | 0.2 c (0.2–0.2) | 0.0 | 0.4 d (0.4–0.4) | 0.0 | 1.3 a (1.3–1.3) | 0.0 | 46.2 a (45.1–48.1) | 1.7 | 46.4 a (45.3–48.3) | 1.7 | 198.1 a (193.9–205.7) | 6.6 |
| Food | Available CHO † (g/100 g of Food) | Experimental Portion (g) | GI 1 (Glucose = 100) 3 | GL 2 (per Experimental Portion Size) | ||||
|---|---|---|---|---|---|---|---|---|
| Mean | SE | Category | Mean | SE | Category | |||
| Pounded yam | 25.3 | 197.5 | 85 b | 4 | high | 22 cd | 1 | high |
| Placali | 15.8 | 317.1 | 106 a | 5 | high | 17 de | 1 | medium |
| Pounded plantain | 28.8 | 173.9 | 91 ab | 4 | high | 26 bc | 1 | high |
| Maize meal stiff porridge | 21.6 | 231.5 | 74 bc | 5 | high | 16 e | 1 | medium |
| Attieke | 46.2 | 108.2 | 63 c | 2 | medium | 29 b | 1 | high |
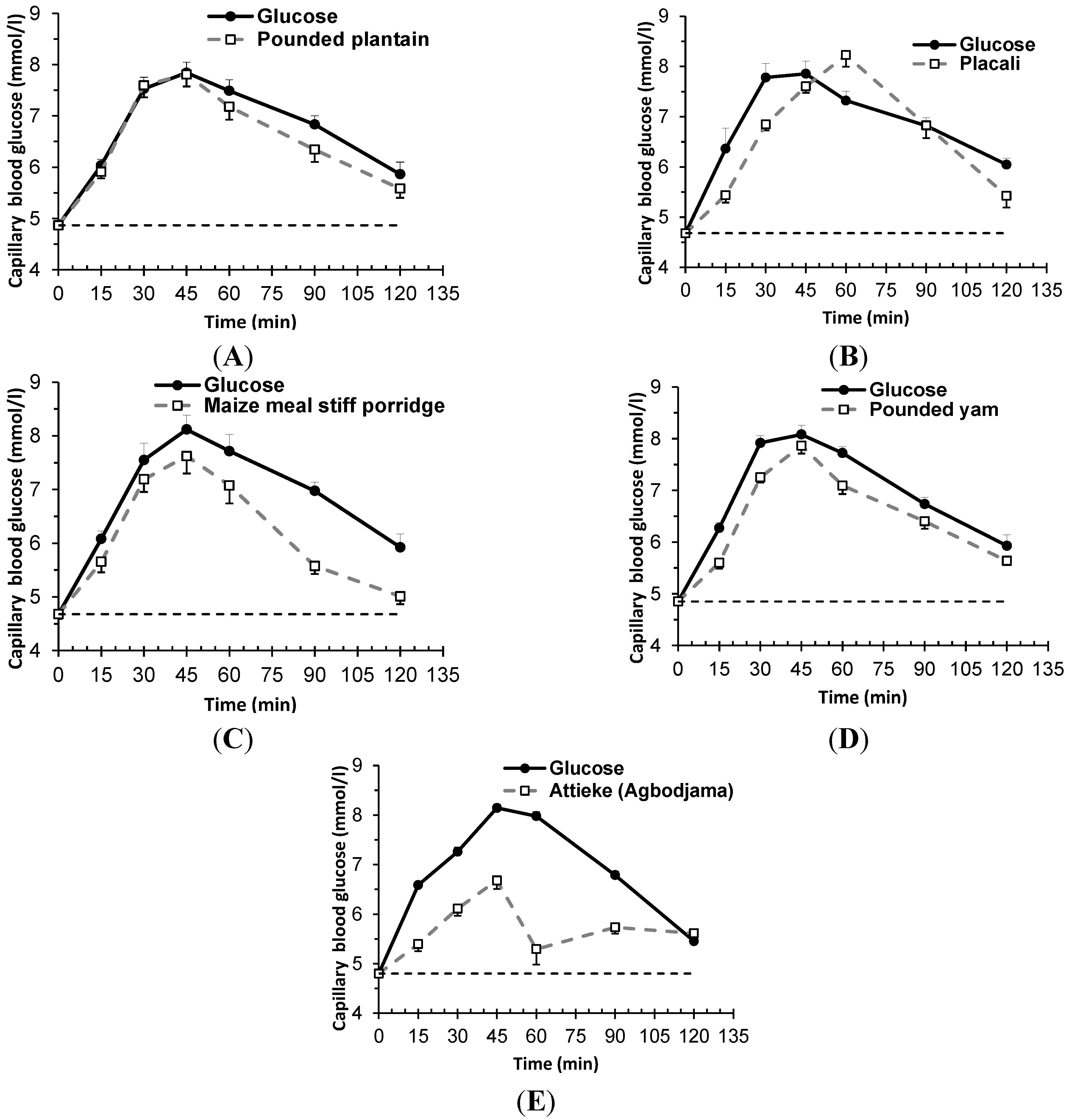
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- National Geographic. Staple Food Crops of the World: Where Are Some of the World’s Staple Crops Grown? Interactive Map Media Spotlight. Available online: http://education.nationalgeographic.com/education/maps/staple-food-crops-world/?ar_a=1 (accessed on 12 December 2014).
- Mourey, A. Manuel de Nutrition Pour l’Intervention Humanitaire. Available online: https://www.icrc.org/fre/assets/files/other/icrc_001_0820.pdf (accessed on 5 January 2015).
- Amani, N.G.; Kamenan, A. Potentialités nutritionnelles et technologie traditionnelle de transformation des denrées amylacées en Côte d’Ivoire. In Proceedings of the 2nd Ateliers Internationaux sur les Voies Alimentaires d’Améliorations des Situations Nutritionnelles en Afrique de l’Ouest, les Rôles des Technologies Alimentaires et Nutritionnistes, Ouagadougou, Burkina Faso, 23–28 November 2003.
- Pineau, H. Etat Nutritionnel et Qualité de l’Alimentation des Enfants de Moins de Deux ans Dans le Village de Damé (Côte d’Ivoire) Identification et Estimation de la Valeur et de l’Apport Nutritionnels des Aliments de Complément. DESS Nutrition et Alimentation dans les Pays en Développement. Available online: http://www.museum.agropolis.fr/pages/savoirs/agroalim_ci/Pineau_DESS_2004.pdf (accessed on 5 January 2015).
- Avallone, S.; Tiemtore, T.W.E.; Mouquet-rivier, V.; Trèche, S. Nutritional value of six multi-ingredient sauces from Burkina Faso. J. Food Comp. Anal. 2008, 21, 553–558. [Google Scholar] [CrossRef]
- Agriculture and Consumer Protection Department, Food and Agriculture Organization of the United Nations. Carbohydrates in human nutrition (FAO Food and Nutrition Paper No. 66). In Proceedings of the Joint FAO/WHO Expert Consultation, Rome, Italy, 14–18 April 1997.
- International Standards Organisation. ISO 26642–2010. Food Products—Determination of the Glycaemic Index (GI) and Recommendation for Food Classification; International Standards Organisation: Geneva, Switzerland, 2010. [Google Scholar]
- Finocchiaro, F.; Ferrari, B.; Gianinetti, A.; Scazzina, F.; Pellegrini, N.; Caramanico, R.; Salati, C.; Shirvanian, V.; Stanca, A.M. Effects of barley β-glucan-enriched flour fractions on the glycaemic index of bread. Int. J. Food Sci. Nutr. 2012, 63, 23–29. [Google Scholar] [CrossRef] [PubMed]
- Stessens, J.; Dao, D. Côte d’Ivoire: Profil des Marchés Pour les Evaluations d’Urgence de la Sécurité Alimentaire; Programme alimentaire mondiales Nations Unies, Service de l’évaluation des besoins d’urgence (ODAN): Rome, Italy, 2006; p. 78. [Google Scholar]
- Digbeu, D.Y.; Due, A.E.; Brou, K.; Kouakou, A.M.; Nemlin, G.J.; DAGO, G. Characterization of yam land races in Côte d’Ivoire with respect to food quality and end uses. J. Appl. Biosci. 2009, 20, 1203–1214. [Google Scholar]
- Coursey, D.G. Yams; Longmans: London, UK, 1967. [Google Scholar]
- Kouamé, A.C.; Kouassi, K.N.; Coulibaly, A.; N’dri, Y.D.; Tiahou, G.G.; Lokrou, A.; Amani, N.G. Glycemic index and glycemic load of selected staples based on rice, yam and cassava commonly consumed in Côte d’Ivoire. FNS 2014, 5, 308–315. [Google Scholar] [CrossRef]
- Toka, D.M.; Djéni, N.T.; Dje, K.M. Improved process of cassava processing into “Attiéké”, a traditional food product of Côte D’Ivoire. Int. J. Food. Eng. 2008, 4, 1–13. [Google Scholar] [CrossRef]
- Aboua, F. Optimization of traditional fermentation of cassava. Trop. Sci. 1995, 35, 68–75. [Google Scholar]
- Nimaga, D.; Tetchi, F.A.; Kakou, C.A.; Nindjin, C.; Amani, N.G. Influence of traditional inoculum and fermentation time on the organoleptic quality of “Attiéké”. FNS 2012, 3, 1335–1339. [Google Scholar] [CrossRef]
- Kakou, A.C. Optimisation des conditions d’application d’une méthode conservation longue durée de la pâte de manioc (Manihot esculenta, Crantz) en vue d’améliorer la qualité alimentaire de l’attiéké et du placali. Master’s Thesis, Université de Cocody, Abidjan, Côte D’Ivoire, 2000. [Google Scholar]
- Assanvo, J.B.; Agbo, G.N.; Behi, Y.E.N.; Coulin, P.; Farah, Z. La microflore du ferment de manioc pour la production de l’attiéké adjoukrou à Dabou (Côte d’Ivoire). Available online: http://www.csrs.ch/fichiers/bioterre/assanvo-justine-2002.pdf (accessed on 5 January 2015).
- Soro-Yao, A.A.; Kouakou, B.; Koffi-Nevry, R.; Djè, K.M. Microbiology of Ivorian fermented products: A review. Asian J. Agric. Food Sci. 2013, 1, 37–47. [Google Scholar]
- Zoumenou, V.; Aboua, F.; Gnakri, D.; Kamenan, A. Etude des caractéristiques physicochimique de certains plats traditionnels dérivés du manioc (foutou, placali et kokondé). Tropicultura 1998–1999, 16–17, 120–126. [Google Scholar]
- Aboua, F. Effect of home processing methods on the nutritional value of plantains in Ivory Coast. Trop. Sci. 1994, 34, 274–281. [Google Scholar]
- Williams, S. Official Methods of Analysis of the Association of Official Analytical Chemists, 16th ed.; Association of Official Analytical Chemists (AOAC): Arlington, VA, USA, 2000. [Google Scholar]
- Prosky, L.; Asp, N.G.; Schweitzer, T.F.; DeVries, J.W.; Furda, I. Determination of insoluble and soluble and total dietary fiber in foods and food products: Interlaboratory study. J. AOAC Int. 1992, 75, 360–367. [Google Scholar]
- Food and Agriculture Organization of the United Nations (FAO) and World Health Organization (WHO). Energy and Protein Requirements. (FAO Nutrition Meetings Report Series No. 52). Available online: http://whqlibdoc.who.int/trs/WHO_TRS_522.pdf (accessed on 8 January 2015).
- Salmeron, J.; Manson, J.E.; Stampfer, M.J.; Colditz, G.A.; Wing, A.L.; Willett, W.C. Dietary fiber, glycaemic load, and risk of non-insulin dependent diabetes mellitus in women. JAMA 1997, 277, 472–477. [Google Scholar] [CrossRef] [PubMed]
- Venn, B.J.; Green, T.J. Glycemic index and glycemic load: Measurement issues and their effect on diet disease relationships. Eur. J. Clin. Nutr. 2007, 61, S122–S131. [Google Scholar] [CrossRef] [PubMed]
- Wolever, T.M.S.; Katzman-Relle, L.; Jenkins, A.L.; Vuksan, V.; Josse, R.B.; Jenkins, D.J. Glycaemic index of 102 complex carbohydrate foods in patients with diabetes. Nutr. Res. 1994, 14, 651–669. [Google Scholar] [CrossRef]
- Henry, C.J.K.; Lightowler, H.J.; Strik, C.M.; Renton, R.; Hails, S. Glycaemic index and glycaemic load values of commercially available products in the UK. Br. J. Nutr. 2005, 94, 922–930. [Google Scholar] [CrossRef] [PubMed]
- Omoregie, E.S.; Osagie, A.U. Glycemic indices and glycemic load of some Nigerian foods. Pak. J. Nutr. 2008, 7, 710–716. [Google Scholar] [CrossRef]
- Mahgoub, S.O.; Sabone, M.; Jackson, J. Glycaemic index of selected staple carbohydrate-rich foods commonly consumed in Botswana. South Afr. J. Clin. Nutr. 2013, 26, 182–187. [Google Scholar]
- Bhupathiraju, N.S.; Tobias, K.D.; Malik, S.V.; Pan, A.; Adela, H.; JoAnn, E.; Manson, E.A.J.; Willett, C.W.; Hu, B.F. Glycemic index, glycemic load, and risk of type 2 diabetes: Results from 3 large US cohorts and an updated meta-analysis. AJCN 2014, 100, 218–232. [Google Scholar] [CrossRef]
- Leeman, M.; Östman, E.; Björck, I. Glycaemic and satiating properties of potato products. Eur. J. Clin. Nutr. 2008, 62, 87–95. [Google Scholar] [CrossRef] [PubMed]
- Che, L.M.; Wang, L.J.; Li, D.; Bhandari, B.; Özkan, N.; Chen, D.X.; Mao, Z.H. Starch pastes thinning during high-pressure homogenization. Carbohydr. Polym. 2009, 75, 32–38. [Google Scholar] [CrossRef]
- Cândido, F.G.; Pereira, E.V.; Alfenas, R.C.G. Use of the glycemic index in nutrition education. Rev. Nutr. 2013, 26, 89–96. [Google Scholar] [CrossRef]
- Tako, M.; Tamaki, Y.; Teruya, T.; Takeda, Y. The principles of starch gelatinization and retrogradation. FNS 2014, 5, 280–291. [Google Scholar] [CrossRef]
- Vosloo, M.C. Some factors affecting the digestion of glycaemic carbohydrates and the blood glucose response. J. Fam. Ecol. Consum. Sci. 2005, 33, 1–9. [Google Scholar]
- Giménez, A.M.; González, J.R.; Wagner, J.; Torres, R.; Lobo, O.M.; Samman, C.N. Effect of extrusion conditions on physicochemical and sensorial properties of corn-broad beans (Vicia faba) spaghetti type pasta. Food Chem. 2013, 136, 538–545. [Google Scholar] [CrossRef] [PubMed]
- Thompson, L.U.; Yoon, J.H.; Jenkins, D.J.; Wolever, T.M.; Jenkins, A.L. Relationship between polyphenol intake and blood glucose response of normal and diabetic individuals. Am. J. Clin. Nutr. 1984, 39, 745–751. [Google Scholar] [PubMed]
- Nuttall, F.D.; Mooradian, A.D.; Gannon, M.C.; Billington, C.; Krezowski, P. Effect of protein ingestion on the glucose and insulin response to a standardized oral glucose load. Diabetes Care 1984, 7, 465–470. [Google Scholar] [CrossRef] [PubMed]
- Manthou, E.; Kanaki, M.; Georgakouli, K.; Deli, C.K.; Kouretas, D.; Koutedakis, Y.; Jamurtas, A.Z. Glycemic response of a carbohydrate-protein bar with ewe-goat whey. Nutrients 2014, 6, 2240–2250. [Google Scholar] [CrossRef] [PubMed]
- Ihediohanma, C.N. Determination of the glycemic indices of three different cassava granules (garri) and the effect of fermentation period on their glycemic responses. Pak. J. Nutr. 2011, 10, 6–9. [Google Scholar] [CrossRef]
- Mendosa, D. Revised International Table of Glycemic Index (GI) and Glycemic Load (GL) Values—2008. Available online: http://www.mendosa.com/gilists.htm (accessed on 12 February 2012).
- Atkinson, F.S.; Foster-Powell, K.; Brand-Miller, J.C. International tables of glycemic index and glycemic load values: 2008. Diabetes Care 2008, 31, 2281–2283. [Google Scholar] [CrossRef] [PubMed]
- Salmeron, J.; Ascherio, A.; Rimm, E.B.; Colditz, G.A.; Spiegelman, D.; Jenkins, D.J.; Stampfer, M.J.; Wing, A.L.; Willett, W.C. Dietary fiber, glycaemic load, and risk of NIDDM in men. Diabetes Care 1997, 20, 545–550. [Google Scholar] [CrossRef] [PubMed]
- Willett, W.; Manson, J.; Liu, S. Glycemic index, glycemic load, and risk of type 2 diabetes. Am. J. Clin. Nutr. 2002, 76, S274–S280. [Google Scholar]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kouamé, A.C.; Kouassi, K.N.; N'dri, Y.D.; Amani, N.G. Glycaemic Index and Load Values Tested in Normoglycemic Adults for Five Staple Foodstuffs: Pounded Yam, Pounded Cassava-Plantain, Placali, Attieke and Maize Meal Stiff Porridge. Nutrients 2015, 7, 1267-1281. https://doi.org/10.3390/nu7021267
Kouamé AC, Kouassi KN, N'dri YD, Amani NG. Glycaemic Index and Load Values Tested in Normoglycemic Adults for Five Staple Foodstuffs: Pounded Yam, Pounded Cassava-Plantain, Placali, Attieke and Maize Meal Stiff Porridge. Nutrients. 2015; 7(2):1267-1281. https://doi.org/10.3390/nu7021267
Chicago/Turabian StyleKouamé, Adam C., Kouakou N. Kouassi, Yao D. N'dri, and N'guessan G. Amani. 2015. "Glycaemic Index and Load Values Tested in Normoglycemic Adults for Five Staple Foodstuffs: Pounded Yam, Pounded Cassava-Plantain, Placali, Attieke and Maize Meal Stiff Porridge" Nutrients 7, no. 2: 1267-1281. https://doi.org/10.3390/nu7021267
APA StyleKouamé, A. C., Kouassi, K. N., N'dri, Y. D., & Amani, N. G. (2015). Glycaemic Index and Load Values Tested in Normoglycemic Adults for Five Staple Foodstuffs: Pounded Yam, Pounded Cassava-Plantain, Placali, Attieke and Maize Meal Stiff Porridge. Nutrients, 7(2), 1267-1281. https://doi.org/10.3390/nu7021267





