Effect of Whey Supplementation on Circulating C-Reactive Protein: A Meta-Analysis of Randomized Controlled Trials
Abstract
:1. Introduction
2. Methods
2.1. Literature Search
2.2. Study Selection
2.3. Data Extraction and Quality Assessment
2.4. Data Synthesis and Analysis
3. Results
3.1. Search Results
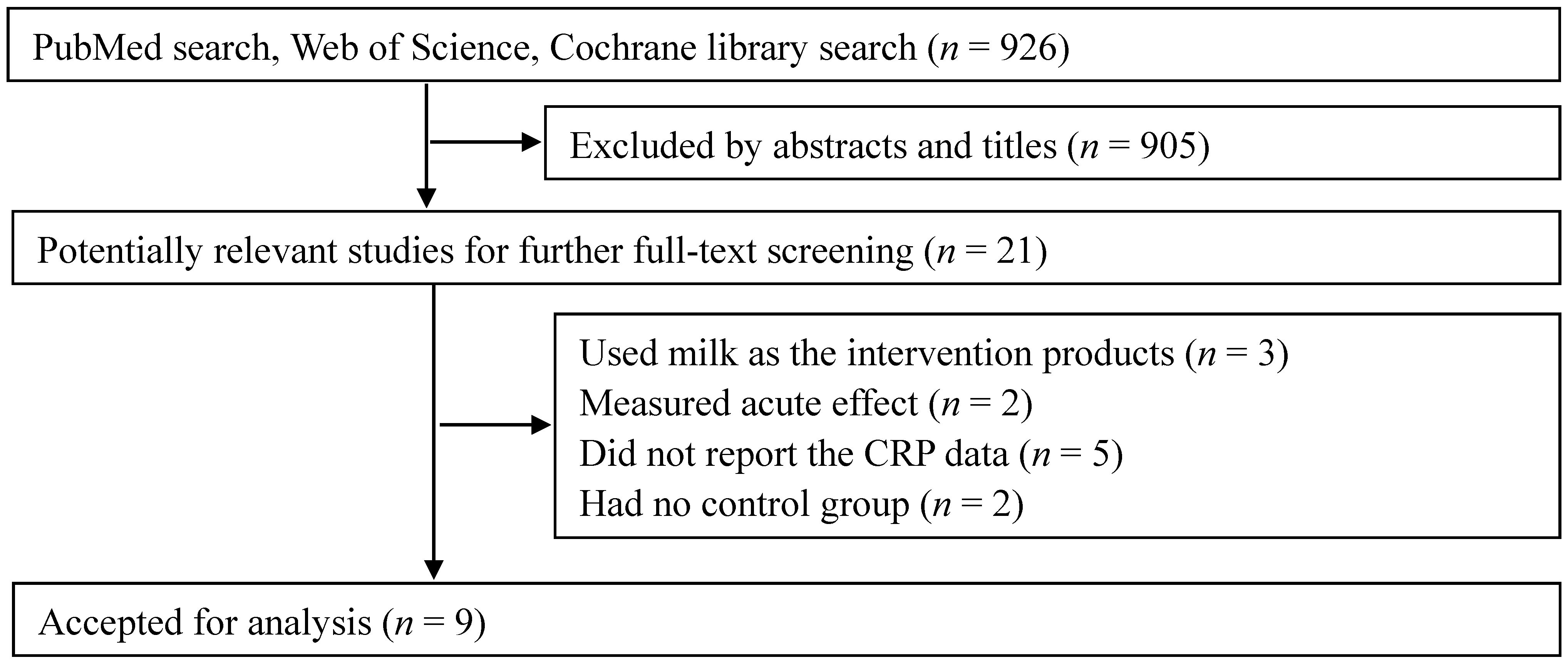
3.2. Study Characteristics
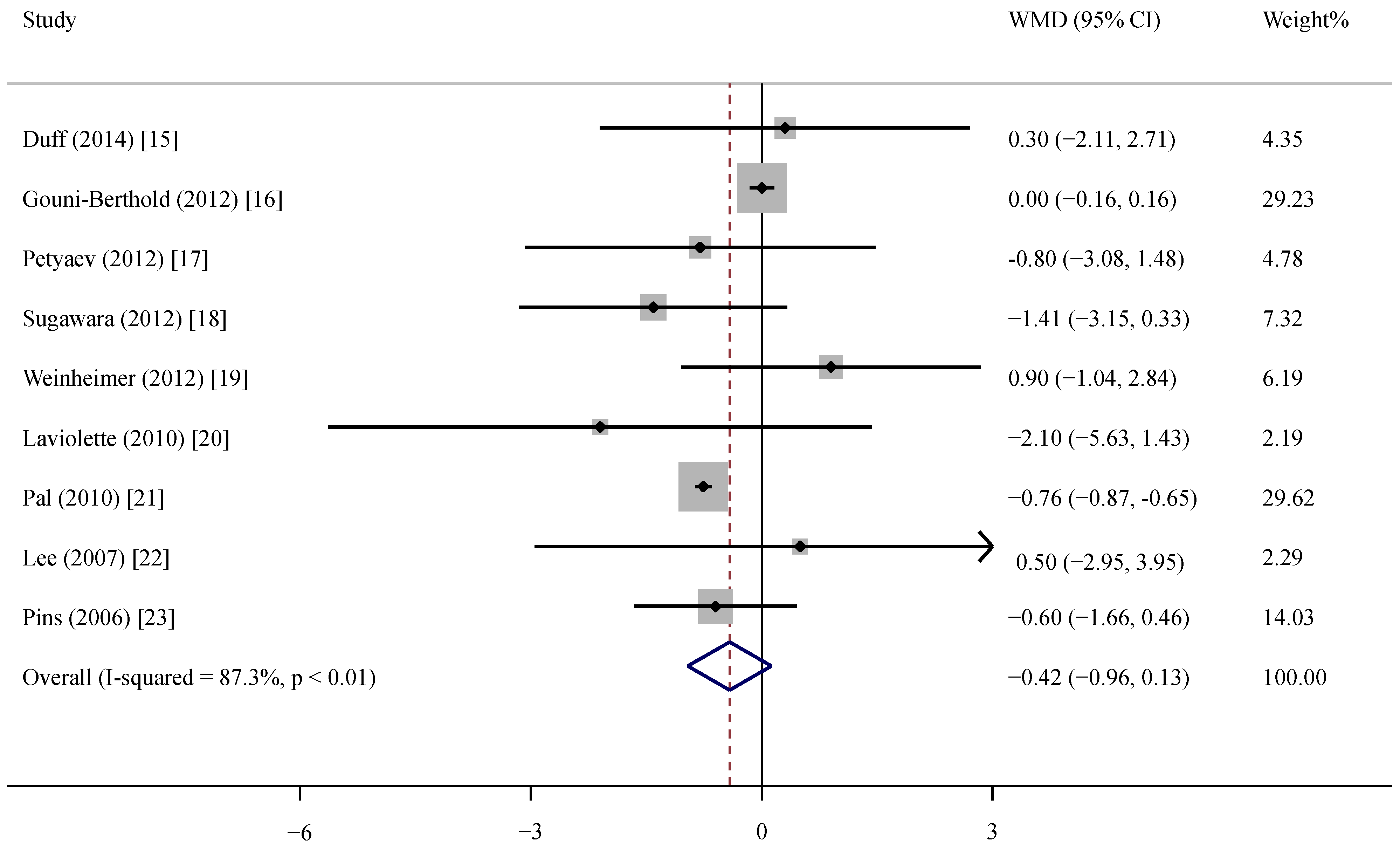
3.3. Effect of Whey Protein on CRP
| Author | Year | Country | Study Design | Sample Size a | Health Status | Male (%) | Age (year) | BMI (kg/m2) | Baseline CRP (mg/L) | CRP Assay | Duration (Weeks) | Intervention | Daily Dose (g) | Jadad Scores |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Duff | 2014 | Canada | P, DB | 21/19 | Adults b | 37.5 | 59.5 | NR | 2.2 | ELISA | 8 | Whey protein complex | 38 | 4 |
| Gouni-Berthold | 2012 | Germany | P, DB | 88/92 | Metabolic syndrome | 52.8 | 53.4 | 31.1 | 0.4 | Immunoturbidimetric latex highly-sensitive assay | 12 | Whey fermentation products | 15.3 | 5 |
| Petyaev | 2012 | Russia | P c | 10/10 | Prehypertension | 55.0 | 54.5 | 26.4 | 7.6 | Commercially available kits | 4 | Whey protein isolate | 0.7 | 3 |
| Sugawara | 2012 | Japan | P, DB | 17/14 | COPD b | 93.5 | 77.3 | NR | 2.0 | Latex turbidimetric immunoassay | 12 | Whey peptides | 10 | 4 |
| Weinheimer | 2012 | USA | P, DB | 30/84 | Overweight/obesity b | 40.4 | 48.0 | 30.0 | 3.2 | COBAS Integra 400 | 36 | Whey protein | 60 | 3 |
| Laviolette | 2010 | Canada | P, DB | 12/10 | COPD | 63.6 | 65.3 | 28.2 | 4.0 | Immunonephelometry | 8 | Pressurized whey | 20 | 4 |
| Pal | 2010 | Australia | P, SB | 25/25 | Overweight/obesity | 14.0 | 48.5 | 31.3 | 3.8 | Solid phase enzyme amplified sensitivity immunoassay | 12 | Whey protein isolate | 54 | 3 |
| Lee | 2007 | Germany | P, DB | 27/26 | Mild hypertension | 56.7 | 51.6 | 27.9 | 2.3 | Immunonephelometry | 12 | Whey peptides | 3.0 | 4 |
| Pins | 2006 | USA | P, DB | 15/15 | Prehypertension or stage 1 hypertension | 46.7 | 46.1 | 29.0 | 2.6 | Immunonephelometry | 6 | Hydrolyzed whey protein | 20 | 3 |
3.4. Subgroup and Sensitivity Analyses
| Group | No. | Net Change (95% CI) | p | Pheterogeneity | I2 (%) |
|---|---|---|---|---|---|
| Total | 9 | −0.42 (−0.96, 0.13) | 0.20 | <0.01 | 87.3 |
| Study duration, week | |||||
| <12 | 4 | −0.60 (−1.47, 0.26) | 0.17 | 0.74 | 0 |
| ≥12 | 5 | −0.30 (−1.02, 0.42) | 0.41 | <0.01 | 94.9 |
| Whey dose, g/day | |||||
| <20 | 4 | −0.10 (−0.69, 0.49) | 0.74 | 0.32 | 14.1 |
| ≥20 | 5 | −0.72 (−0.97, −0.47) | <0.01 | 0.39 | 3.8 |
| CRP/hsCRP in study | |||||
| CRP | 6 | −0.75 (−0.85, −0.64) | <0.01 | 0.47 | 0 |
| hsCRP | 3 | −0.32 (−0.99, 0.35) | 0.35 | 0.16 | 45.2 |
| Baseline CRP, mg/L | |||||
| <3 | 5 | −0.06 (−0.46, 0.35) | 0.79 | 0.34 | 11.8 |
| ≥3 | 4 | −0.67 (−1.21, −0.14) | 0.01 | 0.34 | 10.4 |
3.5. Meta-Regression Analysis
3.6. Publication Bias
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Sousa, G.T.; Lira, F.S.; Rosa, J.C.; de Oliveira, E.P.; Oyama, L.M.; Santos, R.V.; Pimentel, G.D. Dietary whey protein lessens several risk factors for metabolic diseases: A review. Lipids Health Dis. 2012, 11, 67. [Google Scholar] [CrossRef] [PubMed]
- Tong, X.; Li, W.; Xu, J.Y.; Han, S.; Qin, L.Q. Effects of whey protein and leucine supplementation on insulin sensitivity in non-obese insulin-resistant model rats. Nutrition 2014, 30, 1076–1080. [Google Scholar] [CrossRef] [PubMed]
- Qin, L.Q.; Xu, J.Y.; Dong, J.Y.; Zhao, Y.; van Bladeren, P.; Zhang, W. Lactotripeptides intake and blood pressure management: A meta-analysis of randomized controlled clinical trials. Nutr. Metab. Cardiovasc. Dis. 2013, 23, 395–402. [Google Scholar] [CrossRef] [PubMed]
- Broekhuizen, R.; Wouters, E.F.; Creutzberg, E.C.; Schols, A.M. Raised CRP levels mark metabolic and functional impairment in advanced COPD. Thorax 2006, 61, 17–22. [Google Scholar] [CrossRef] [PubMed]
- Ridker, P.M. Clinical application of C-reactive protein for cardiovascular disease detection and prevention. Circulation 2003, 107, 363–369. [Google Scholar] [CrossRef] [PubMed]
- Androulakis, E.; Tousoulis, D.; Papageorgiou, N.; Latsios, G.; Siasos, G.; Tsioufis, C.; Giolis, A.; Stefanadis, C. Inflammation in hypertension: Current therapeutic approaches. Curr. Pharm. Des. 2011, 17, 4121–4131. [Google Scholar] [CrossRef] [PubMed]
- Donath, M.Y. Targeting inflammation in the treatment of type 2 diabetes: Time to start. Nat. Rev. Drug Discov. 2014, 13, 465–476. [Google Scholar] [CrossRef] [PubMed]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G. PRISMA Group. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. Ann. Intern. Med. 2009, 151, 264–269. [Google Scholar] [CrossRef] [PubMed]
- Jadad, A.R.; Moore, R.A.; Carroll, D.; Jenkinson, C.; Reynolds, D.J.; Gavaghan, D.J.; McQuay, H.J. Assessing the quality of reports of randomized clinical trials: Is blinding necessary? Control Clin. Trials 1996, 17, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Higgins, J.; Green, S. Cochrane Handbook for Systematic Reviews of Interventions Version 5.1.0. The Cochrane Collaboration. Available online: http://www.cochrane-handbook.org (accessed on 20 June 2014).
- Follmann, D.; Elliott, P.; Suh, I.; Cutler, J. Variance imputation for overviews of clinical trials with continuous response. J. Clin. Epidemiol. 1992, 45, 769–773. [Google Scholar] [CrossRef] [PubMed]
- Higgins, J.P.; Thompson, S.G.; Deeks, J.J.; Altman, D.G. Measuring inconsistency in meta-analyses. BMJ 2003, 327, 557–560. [Google Scholar] [CrossRef] [PubMed]
- DerSimonian, R.; Laird, N.P. Meta-analysis in clinical trials. Control Clin. Trials 1986, 7, 177–188. [Google Scholar] [CrossRef] [PubMed]
- Egger, M.; Davey Smith, G.; Schneider, M.; Minder, C. Bias in meta-analysis detected by a simple, graphical test. BMJ 1997, 315, 629–634. [Google Scholar] [CrossRef] [PubMed]
- Duff, W.R.; Chilibeck, P.D.; Rooke, J.J.; Kaviani, M.; Krentz, J.R.; Haines, D.M. The effect of bovine colostrum supplementation in older adults during resistance training. Int. J. Sport Nutr. Exerc. Metab. 2014, 24, 276–285. [Google Scholar] [CrossRef] [PubMed]
- Gouni-Berthold, I.; Schulte, D.M.; Krone, W.; Lapointe, J.F.; Lemieux, P.; Predel, H.G.; Berthold, H.K. The whey fermentation product malleable protein matrix decreases TAG concentrations in patients with the metabolic syndrome: A randomized placebo-controlled trial. Br. J. Nutr. 2012, 107, 1694–1706. [Google Scholar] [CrossRef] [PubMed]
- Petyaev, I.M.; Dovgalevsky, P.Y.; Klochkov, V.A.; Chalyk, N.E.; Kyle, N. Whey protein lycosome formulation improves vascular functions and plasma lipids with reduction of markers of inflammation and oxidative stress in prehypertension. Sci. World J. 2012, 2012. [Google Scholar] [CrossRef]
- Sugawara, K.; Takahashi, H.; Kashiwagura, T.; Yamada, K.; Yanagida, S.; Homma, M.; Dairiki, K.; Sasaki, H.; Kawagoshi, A.; Satake, M.; et al. Effect of anti-inflammatory supplementation with whey peptide and exercise therapy in patients with COPD. Respir. Med. 2012, 106, 1526–1534. [Google Scholar] [CrossRef] [PubMed]
- Weinheimer, E.M.; Conley, T.B.; Kobza, V.M.; Sands, L.P.; Lim, E.; Janle, E.M.; Campbell, W.W. Whey protein supplementation does not affect exercise training-induced changes in body composition and indices of metabolic syndrome in middle-aged overweight and obese adults. J. Nutr. 2012, 142, 1532–1539. [Google Scholar] [CrossRef] [PubMed]
- Laviolette, L.; Lands, L.C.; Dauletbaev, N.; Saey, D.; Milot, J.; Provencher, S.; LeBlanc, P.; Maltais, F. Combined effect of dietary supplementation with pressurized whey and exercise training in chronic obstructive pulmonary disease: A randomized, controlled, double-blind pilot study. J. Med. Food 2010, 13, 589–598. [Google Scholar] [CrossRef] [PubMed]
- Pal, S.; Ellis, V. The chronic effects of whey proteins on blood pressure, vascular function, and inflammatory markers in overweight individuals. Obesity 2010, 18, 1354–1359. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.M.; Skurk, T.; Hennig, M.; Hauner, H. Effect of a milk drink supplemented with whey peptides on blood pressure in patients with mild hypertension. Eur. J. Nutr. 2007, 46, 21–27. [Google Scholar] [CrossRef] [PubMed]
- Pins, J.J.; Keenan, J.M. Effects of whey peptides on cardiovascular disease risk factors. J. Clin. Hypertens. 2006, 8, 775–782. [Google Scholar] [CrossRef]
- Wang, C.; Yatsuya, H.; Tamakoshi, K.; Uemura, M.; Li, Y.; Wada, K.; Yamashita, K.; Kawaguchi, L.; Toyoshima, H.; Aoyama, A. Positive association between high-sensitivity C-reactive protein and incidence of type 2 diabetes mellitus in Japanese workers: 6-year follow-up. Diabetes Metab. Res. Rev. 2013, 29, 398–405. [Google Scholar] [CrossRef] [PubMed]
- Choi, J.L.; Joseph, L.; Pilote, L. Obesity and C-reactive protein in various populations: A systematic review and meta-analysis. Obes. Rev. 2013, 14, 232–244. [Google Scholar] [CrossRef] [PubMed]
- Buyken, A.E.; Goletzke, J.; Joslowski, G.; Felbick, A.; Cheng, G.; Herder, C.; Brand-Miller, J.C. Association between carbohydrate quality and inflammatory markers: Systematic review of observational and interventional studies. Am. J. Clin. Nutr. 2014, 99, 813–833. [Google Scholar] [CrossRef] [PubMed]
- Zhou, H.; Li, Y.; Huang, G.; Gu, X.; Zeng, J.; Li, Y.; Luo, C.; Ou, B.; Zhang, Y.; Wu, Z.; et al. Interleukin 6 augments mechanical strain-induced C-reactive protein synthesis via the stretch-activated channel-nuclear factor κB signal pathway. Heart 2013, 99, 570–576. [Google Scholar] [CrossRef] [PubMed]
- Skurk, T.; van Harmelen, V.; Hauner, H. Angiotensin II stimulates the release of interleukin-6 and interleukin-8 from cultured human adipocytes by activation of NF-κB. Arterioscler Thromb. Vasc. Biol. 2004, 24, 1199–1203. [Google Scholar] [CrossRef] [PubMed]
- Xiao, Y.; Haynes, W.L.; Michalek, J.E.; Russell, I.J. Elevated serum high- sensitivity C-reactive protein levels in fibromyalgia syndrome patients correlate with body mass index, interleukin-6, interleukin-8, erythrocyte sedimentation rate. Rheumatol. Int. 2013, 33, 1259–1264. [Google Scholar] [CrossRef] [PubMed]
- Comassi, M.; Vitolo, E.; Pratali, L.; Del Turco, S.; Dellanoce, C.; Rossi, C.; Santini, E.; Solini, A. Acute effects of different degrees of ultra-endurance exercise on systemic inflammatory responses. Intern. Med. J. 2014. [Google Scholar] [CrossRef]
- Kerasioti, E.; Stagos, D.; Jamurtas, A.; Kiskini, A.; Koutedakis, Y.; Goutzourelas, N.; Pournaras, S.; Tsatsakis, A.M.; Kouretas, D. Anti-inflammatory effects of a special carbohydrate-whey protein cake after exhaustive cycling in humans. Food Chem. Toxicol. 2013, 61, 42–46. [Google Scholar] [CrossRef] [PubMed]
- Lands, L.C.; Iskandar, M.; Beaudoin, N.; Meehan, B.; Dauletbaev, N.; Berthiuame, Y. Dietary supplementation with pressurized whey in patients with cystic fibrosis. J. Med. Food 2010, 13, 77–82. [Google Scholar] [CrossRef] [PubMed]
- Bondia-Pons, I.; Ryan, L.; Martinez, J.A. Oxidative stress and inflammation interactions in human obesity. J. Physiol. Biochem. 2012, 68, 701–711. [Google Scholar] [CrossRef] [PubMed]
- Marshall, K. Therapeutic applications of whey protein. Altern. Med. Rev. 2004, 9, 136–156. [Google Scholar] [PubMed]
- Zavorsky, G.S.; Kubow, S.; Grey, V.; Riverin, V.; Lands, L.C. An open-label dose-response study of lymphocyte glutathione levels in healthy men and women receiving pressurized whey protein isolate supplements. Int. J. Food. Sci. Nutr. 2007, 58, 429–436. [Google Scholar] [CrossRef] [PubMed]
- Kerasioti, E.; Stagos, D.; Priftis, A.; Aivazidis, S.; Tsatsakis, A.M.; Kouretas, D. Antioxidant effects of whey protein on muscle C2C12 cells. Food Chem. 2014, 155, 271–278. [Google Scholar] [CrossRef] [PubMed]
- Zemel, M.B.; Bruckbauer, A. Effects of a leucine and pyridoxine-containing nutraceutical on fat oxidation, and oxidative and inflammatory stress in overweight and obese subjects. Nutrients 2012, 4, 529–541. [Google Scholar] [CrossRef] [PubMed]
- Ricci-Cabello, I.; Herrera, M.O.; Artacho, R. Possible role of milk-derived bioactive peptides in the treatment and prevention of metabolic syndrome. Nutr. Rev. 2012, 70, 241–255. [Google Scholar] [CrossRef] [PubMed]
- Aihara, K.; Osaka, M.; Yoshida, M. Oral administration of milk casein-derived tripeptides Val-Pro-Pro attenuates high-fat diet-induced adipose tissue inflammation in mice. Br. J. Nutr. 2014, 112, 513–519. [Google Scholar] [CrossRef] [PubMed]
- Hirota, T.; Ohki, K.; Kawagishi, R.; Kajimoto, Y.; Mizuno, S.; Nakamura, Y.; Kitakaze, M. Casein hydrolysate containing the antihypertensive tripeptides Val-Pro-Pro and Ile-Pro-Pro improves vascular endothelial function independent of blood pressure-lowering effects: Contribution of the inhibitory action of angiotensin-converting enzyme. Hypertens. Res. 2007, 30, 489–496. [Google Scholar] [CrossRef] [PubMed]
- Arnberg, K.; Larnkjar, A.; Michaelsen, K.F.; Jensen, S.M.; Hoppe, C.; Molgaard, C. Casein improves brachial and central aortic diastolic blood pressure in overweight adolescents: A randomized, controlled trial. J. Nutr. Sci. 2013, 2, 1–10. [Google Scholar] [CrossRef]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhou, L.-M.; Xu, J.-Y.; Rao, C.-P.; Han, S.; Wan, Z.; Qin, L.-Q. Effect of Whey Supplementation on Circulating C-Reactive Protein: A Meta-Analysis of Randomized Controlled Trials. Nutrients 2015, 7, 1131-1143. https://doi.org/10.3390/nu7021131
Zhou L-M, Xu J-Y, Rao C-P, Han S, Wan Z, Qin L-Q. Effect of Whey Supplementation on Circulating C-Reactive Protein: A Meta-Analysis of Randomized Controlled Trials. Nutrients. 2015; 7(2):1131-1143. https://doi.org/10.3390/nu7021131
Chicago/Turabian StyleZhou, Ling-Mei, Jia-Ying Xu, Chun-Ping Rao, Shufen Han, Zhongxiao Wan, and Li-Qiang Qin. 2015. "Effect of Whey Supplementation on Circulating C-Reactive Protein: A Meta-Analysis of Randomized Controlled Trials" Nutrients 7, no. 2: 1131-1143. https://doi.org/10.3390/nu7021131
APA StyleZhou, L.-M., Xu, J.-Y., Rao, C.-P., Han, S., Wan, Z., & Qin, L.-Q. (2015). Effect of Whey Supplementation on Circulating C-Reactive Protein: A Meta-Analysis of Randomized Controlled Trials. Nutrients, 7(2), 1131-1143. https://doi.org/10.3390/nu7021131




