Suboptimal Plasma Long Chain n-3 Concentrations are Common among Adults in the United States, NHANES 2003–2004
Abstract
:1. Introduction
2. Materials and Methods
2.1. Comparison with Other Studies
2.2. Statistical Analysis
3. Results
| All | Hispanic | Non-Hispanic White | Non-Hispanic Black | |
|---|---|---|---|---|
| Women, N (%) | 693 (48.5) | 158 (43.4) | 374 (52.2) | 130 (55.2) |
| Age, N (%) | ||||
| ≥20 years | 1386 | 334 (11.3) | 739 (72.1) | 253 (10.9) |
| 20–39 years | 412 (37.0) | 101 (55.1) | 196 (33.0) | 99 (48.3) |
| 40–59 years | 423 (40.1) | 96 (32.2) | 223 (41.6) | 78 (33.0) |
| ≥60 years | 551 (23.0) | 137 (12.7) | 320 (25.5) | 76 (18.7) |
| Education, N (%) | ||||
| <High school | 411 (18.4) | 202 (51.2) | 116 (11.4) | 80 (30.0) |
| High school | 339 (25.6) | 65 (23.3) | 203 (26.7) | 59 (24.2) |
| >High school | 634 (55.9) | 67 (25.5) | 420 (61.9) | 112 (45.6) |
| BMI, kg/m2, Mean ± SE | 28.3 ± 0.20 | 29.0 ± 0.38 | 28.0 ± 0.25 | 30.2 ± 0.52 |
| Smoking status, N (%) | ||||
| Never | 687 (49.9) | 177 (52.7) | 332 (47.7) | 145 (60.0) |
| Current | 307 (25.0) | 57 (21.4) | 168 (25.3) | 69 (27.7) |
| Former | 392 (25.1) | 100 (25.9) | 69 (27.7) | 39 (12.3) |
| Prevalent CVD, N (%) | 159 (8.08) | 27 (3.70) | 99 (8.41) | 22 (6.84) |
| All | Hispanic | Non-Hispanic White | Non-Hispanic Black | |
|---|---|---|---|---|
| ≥20 years | ||||
| Mean (95% CI) | 2.07 (1.95–2.19) | 1.82 (1.65–2.00) a | 2.04 (1.92–2.17) a | 2.30 (2.08–2.52) b |
| 20–39 years | ||||
| Mean (95% CI) | 1.98 (1.83–2.13) a | 1.76 (1.44–2.07) | 1.95 (1.80–2.10) | 2.19 (1.93–2.44) |
| 40–59 years | ||||
| Mean (95% CI) | 2.04 (1.87–2.21) a,b | 1.89 (1.68–2.10) | 1.99 (1.81–2.17) | 2.28 (2.01–2.55) |
| ≥60 years | ||||
| Mean (95% CI) | 2.28 (2.15–2.41) b | 1.96 (1.67–2.24) | 2.25 (2.11–2.40) | 2.60 (2.25–2.95) |
| N (%) | All | Hispanic | Non-Hispanic White | Non-Hispanic Black |
|---|---|---|---|---|
| Below dietary recommendations; LCn-3 plasma PUFA < 2.49% | ||||
| ≥20 years | 1109 (80.6) | 311 (91.2) a | 588 (82.0) a,b | 178 (73.2) b |
| 20–39 years | 352 (84.2) | 98 (94.7) | 165 (83.6) | 78 (78.7) |
| 40–59 years | 351 (81.8) | 90 (87.5) | 190 (84.6) | 58 (76.1) |
| ≥60 years | 406 (72.9) | 123 (85.2) | 233 (75.5) | 42 (53.6) |
| Below concentrations associated with cardio-protection b; LCn-3 plasma PUFA < 3.86% | ||||
| ≥20 years | 1322 (95.7) | 328 (98.2) | 707 (96.0) | 235 (93.8) |
| 20–39 years | 399 (96.9) | 100 (98.2) | 191 (97.5) | 94 (94.5) |
| 40–59 years | 401 (95.0) | 94 (98.2) | 214 (95.8) | 71 (92.4) |
| ≥60 years | 522 (94.8) | 134 (97.9) | 302 (94.5) | 70 (94.3) |
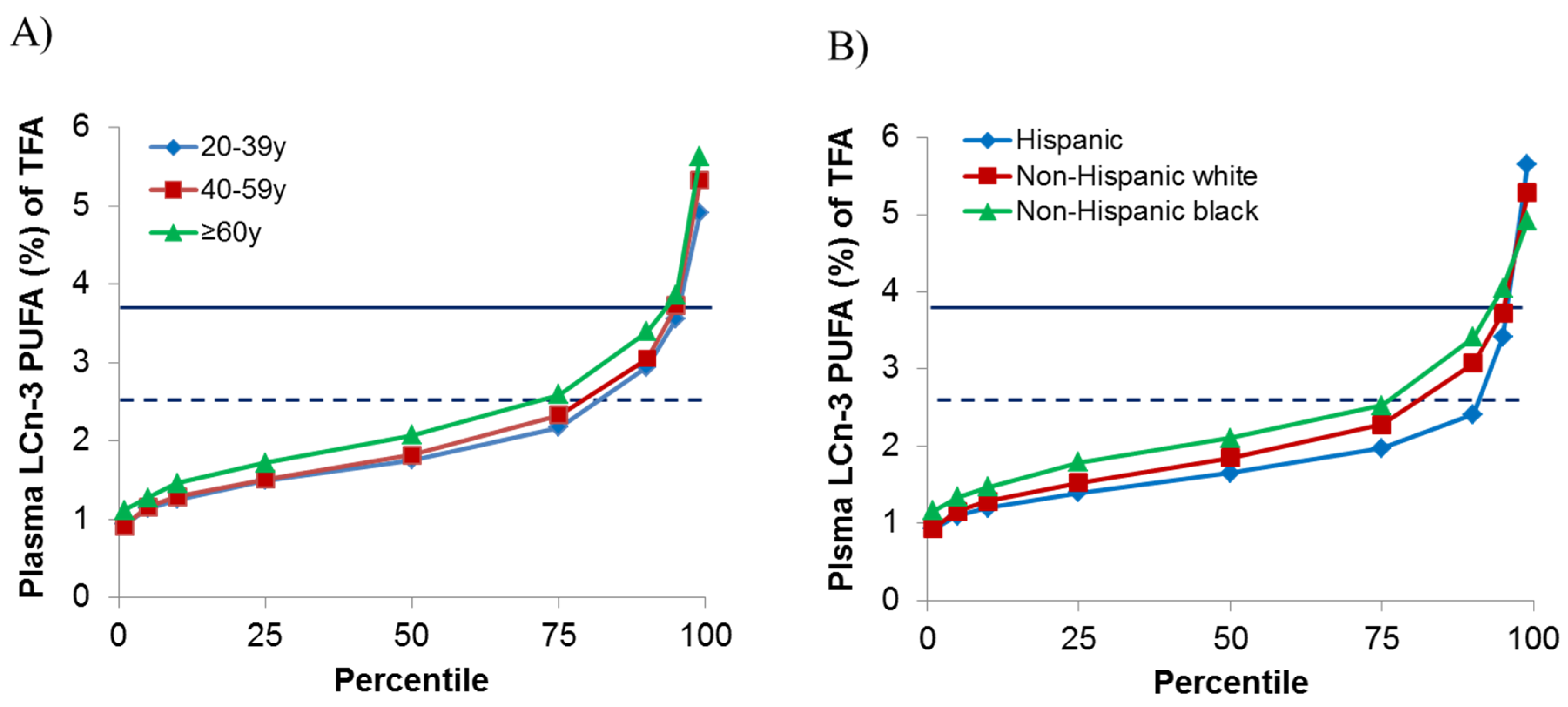
4. Discussion
Strengths and Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Conflicts of Interest
References
- USA Department of Agriculture and USA Department of Health and Human Services. Dietary Guidelines for Americans, 2010, 7th ed.USA Government Printing Office: Washington, DC, USA, 2010.
- Burr, M.L.; Fehily, A.M.; Gilbert, J.F.; Rogers, S.; Holliday, R.M.; Sweetnam, P.M.; Elwood, P.C.; Deadman, N.M. Effects of changes in fat, fish, and fibre intakes on death and myocardial reinfarction: Diet and reinfarction trial (DART). Lancet 1989, 2, 757–761. [Google Scholar] [CrossRef]
- Hu, F.B.; Bronner, L.; Willett, W.C.; Stampfer, M.J.; Rexrode, K.M.; Albert, C.M.; Hunter, D.; Manson, J.E. Fish and omega-3 fatty acid intake and risk of coronary heart disease in women. JAMA 2002, 287, 1815–1821. [Google Scholar] [CrossRef] [PubMed]
- Siscovick, D.S.; Raghunathan, T.E.; King, I.; Weinmann, S.; Wicklund, K.G.; Albright, J.; Bovbjerg, V.; Arbogast, P.; Smith, H.; Kushi, L.H.; et al. Dietary intake and cell membrane levels of long-chain n-3 polyunsaturated fatty acids and the risk of primary cardiac arrest. JAMA 1995, 274, 1363–1367. [Google Scholar] [CrossRef] [PubMed]
- American Heart Association. Fish and Omega-3 Fatty Acids. Available online: http://www.heart.org/HEARTORG/GettingHealthy/NutritionCenter/HealthyEating/Fish-and-Omega-3-Fatty-Acids_UCM_303248_Article.jsp (accessed on 7 April 2015).
- Papanikolaou, Y.; Brooks, J.; Reider, C.; Fulgoni, V.L., 3rd. U.S. Adults are not meeting recommended levels for fish and omega-3 fatty acid intake: Results of an analysis using observational data from NHANES 2003–2008. Nutr. J. 2014, 13, 31. [Google Scholar] [CrossRef] [PubMed]
- Dietary Guidelines Advisory Committee. Scientific Report of the 2015 Dietary Guidelines Advisory Committee USDA; Department of Health and Human Services: Washington, DC, USA, 2015. [Google Scholar]
- Hedrick, V.E.; Dietrich, A.M.; Estabrooks, P.A.; Savla, J.; Serrano, E.; Davy, B.M. Dietary biomarkers: Advances, limitations and future directions. Nutr. J. 2012, 11, 109. [Google Scholar] [CrossRef] [PubMed]
- Sun, Q.; Ma, J.; Campos, H.; Hankinson, S.E.; Hu, F.B. Comparison between plasma and erythrocyte fatty acid content as biomarkers of fatty acid intake in US women. Am. J. Clin. Nutr. 2007, 86, 74–81. [Google Scholar] [PubMed]
- Age-Related Eye Disease Study 2 Research Group. Lutein + zeaxanthin and omega-3 fatty acids for age-related macular degeneration: The age-related eye disease study 2 (AREDS2) randomized clinical trial. JAMA 2013, 309, 2005–2015. [Google Scholar]
- USA Centers for Disease Control and Prevetion. Second National Report on Biochemical Indicators of Diet and Nutrition in the U.S. Population 2012; National Center for Environmental Health: Atlanta, GA, USA, 2012. Available online: http://www.cdc.gov/nutritionreport (accessed on 10 October 2014).
- Crowe, F.L.; Appleby, P.N.; Travis, R.C.; Barnett, M.; Brasky, T.M.; Bueno-de-Mesquita, H.B.; Chajes, V.; Chavarro, J.E.; Chirlaque, M.D.; English, D.R.; et al. Circulating fatty acids and prostate cancer risk: Individual participant meta-analysis of prospective studies. J. Natl. Cancer Inst. 2014, 106. [Google Scholar] [CrossRef] [PubMed]
- National Center for Health Statistics. About the National Health and Nutrition Examination Survey. Available online: http://www.cdc.gov/nchs/nhanes/about_nhanes.htm (accessed on 5 January 2015).
- Lagerstedt, S.A.; Hinrichs, D.R.; Batt, S.M.; Magera, M.J.; Rinaldo, P.; McConnell, J.P. Quantitative determination of plasma c8–c26 total fatty acids for the biochemical diagnosis of nutritional and metabolic disorders. Mol. Genet. Metab. 2001, 73, 38–45. [Google Scholar] [CrossRef] [PubMed]
- Harris, T.B.; Song, X.; Reinders, I.; Lang, T.F.; Garcia, M.E.; Siggeirsdottir, K.; Sigurdsson, S.; Gudnason, V.; Eiriksdottir, G.; Sigurdsson, G.; et al. Plasma phospholipid fatty acids and fish-oil consumption in relation to osteoporotic fracture risk in older adults: The age, gene/environment susceptibility study. Am. J. Clin. Nutr. 2015, 101, 947–955. [Google Scholar] [CrossRef] [PubMed]
- Reinders, I.; Murphy, R.A.; Song, X.; Visser, M.; Cotch, M.F.; Lang, T.F.; Garcia, M.E.; Launer, L.J.; Siggeirsdottir, K.; Eiriksdottir, G.; et al. Polyunsaturated fatty acids in relation to incident mobility disability and decline in gait speed; the age, gene/environment susceptibility-reykjavik study. Eur. J. Clin. Nutr. 2015, 69, 489–493. [Google Scholar] [CrossRef] [PubMed]
- Virtanen, J.K.; Laukkanen, J.A.; Mursu, J.; Voutilainen, S.; Tuomainen, T. Serum long-chain n-3 polyunsaturated fatty acids, mercury, and risk of sudden cardiac death in men: A prospective population-based study. PLoS ONE 2012, 7, e41046. [Google Scholar] [CrossRef] [PubMed]
- Harris, W.S.; von Schacky, C. The omega-3 index: A new risk factor for death from coronary heart disease? Prev. Med. 2004, 39, 212–220. [Google Scholar] [CrossRef] [PubMed]
- Flock, M.R.; Harris, W.S.; Kris-Etherton, P.M. Long-chain omega-3 fatty acids: Time to establish a dietary reference intake. Nutr. Rev. 2013, 71, 692–707. [Google Scholar] [CrossRef] [PubMed]
- Harris, W.S.; Dayspring, T.D.; Moran, T.J. Omega-3 fatty acids and cardiovascular disease: New developments and applications. Postgrad. Med. 2013, 125, 100–113. [Google Scholar] [CrossRef] [PubMed]
- Heidenreich, P.A.; Trogdon, J.G.; Khavjou, O.A.; Butler, J.; Dracup, K.; Ezekowitz, M.D.; Finkelstein, E.A.; Hong, Y.; Johnston, S.C.; Khera, A.; et al. Forecasting the future of cardiovascular disease in the United States: A policy statement from the American Heart Association. Circulation 2011, 123, 933–944. [Google Scholar] [CrossRef] [PubMed]
- Shanahan, C.; de Lorimier, R. Smart Prevention-Health Care Cost Savings Resulting from the Targeted Use of Dietary Supplements; Frost & Sullivan: Mountain View, CA, USA, 2014. [Google Scholar]
- Krogstad, J.; Lopez, M. Hispanic Nativity Sift; Pew Research Center: Washington, DC, USA, 2014. [Google Scholar]
- Richardson, A.J.; Burton, J.R.; Sewell, R.P.; Spreckelsen, T.F.; Montgomery, P. Docosahexaenoic acid for reading, cognition and behavior in children aged 7–9 years: A randomized, controlled trial (the DOLAB study). PLoS ONE 2012, 7, e43909. [Google Scholar] [CrossRef] [PubMed]
- Richardson, A.J.; Montgomery, P. The oxford-durham study: A randomized, controlled trial of dietary supplementation with fatty acids in children with developmental coordination disorder. Pediatrics 2005, 115, 1360–1366. [Google Scholar] [CrossRef] [PubMed]
- Burgess, J.R.; Stevens, L.; Zhang, W.; Peck, L. Long-chain polyunsaturated fatty acids in children with attention-deficit hyperactivity disorder. Am. J. Clin. Nutr. 2000, 71, 327S–330S. [Google Scholar] [PubMed]
- Birch, R.J.; Bigler, J.; Rogers, J.W.; Zhuang, Y.; Clickner, R.P. Trends in blood mercury concentrations and fish consumption among U.S. Women of reproductive age, NHANES, 1999–2010. Environ. Res. 2014, 133, 431–438. [Google Scholar] [CrossRef] [PubMed]
- Clarke, T.C.; Black, L.I.; Stussman, B.J.; Barnes, P.M.; Nahin, R.L. Trends in the use of complementary health approaches among adults: United States, 2002–2012. Natl. Health Stat. Rep. 2015, 79, 1–16. [Google Scholar]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Murphy, R.A.; Yu, E.A.; Ciappio, E.D.; Mehta, S.; McBurney, M.I. Suboptimal Plasma Long Chain n-3 Concentrations are Common among Adults in the United States, NHANES 2003–2004. Nutrients 2015, 7, 10282-10289. https://doi.org/10.3390/nu7125534
Murphy RA, Yu EA, Ciappio ED, Mehta S, McBurney MI. Suboptimal Plasma Long Chain n-3 Concentrations are Common among Adults in the United States, NHANES 2003–2004. Nutrients. 2015; 7(12):10282-10289. https://doi.org/10.3390/nu7125534
Chicago/Turabian StyleMurphy, Rachel A., Elaine A. Yu, Eric D. Ciappio, Saurabh Mehta, and Michael I. McBurney. 2015. "Suboptimal Plasma Long Chain n-3 Concentrations are Common among Adults in the United States, NHANES 2003–2004" Nutrients 7, no. 12: 10282-10289. https://doi.org/10.3390/nu7125534
APA StyleMurphy, R. A., Yu, E. A., Ciappio, E. D., Mehta, S., & McBurney, M. I. (2015). Suboptimal Plasma Long Chain n-3 Concentrations are Common among Adults in the United States, NHANES 2003–2004. Nutrients, 7(12), 10282-10289. https://doi.org/10.3390/nu7125534







