Advanced Glycation End Products: Link between Diet and Ovulatory Dysfunction in PCOS?
Abstract
:1. Introduction
| Study | Subjects, Animals, or Cell Lines | Intervention | Outcome |
|---|---|---|---|
| Leuner B. et al., 2012 [13] | C57BL/6 mice | RAGE+ or RAGE− mice given high-fat diet to induce obesity | RAGE- mice had high insulin levels and accelerated weight gain |
| Cai W. et al., 2012 [14] | C57BL6 mice | Isocaloric diet with or without synthetic MG | Mice given MG developed premature insulin resistance, adiposity and the inflammatory changes |
| Hofmann S.M. et al., 2002 [15] | C57/BL/KsJ db/db female mice | High-AGE vs. low-AGE diet | Low-AGE diet: lower fasting insulin levels, reduction in body weight, improved glucose and insulin tolerance tests, increased plasma HDL and lower serum CML and MG levels |
| Sandu O. et al., 2005 [16] | C57/BL6 and db/db (++) female mice | High-AGE vs. low-AGE diet | High-AGE diet: elevated insulin levels, change in pancreatic islet structure |
| Chatzigeorgiou A. et al., 2013 [17] | female wistar rats | High-AGE vs. low-AGE diet | High-AGE diet: increased glucose, insulin, and testosterone levels |
| Cassese A. et al., 2008 [18] | C57/BL6 female mice | High-AGE vs. low-AGE diet | High-AGE diet: increase in insulin resistance and impairment in insulin sensitivity |
| Kandaraki E. et al., 2012 [19] | female Wistar rats | High-AGE vs. low-AGE diet | High-AGE diet: reduced ovarian GLO-I activity and high AGE expression in the granulosa cells |
| Diamanti-Kandarakis et al., 2007 [20] | Female rats | High-AGE vs. low-AGE diet | High-AGE diet: high fasting glucose, insulin, testosterone, and serum AGEs; higher AGE localization in the theca interna cells; elevated RAGE expression in granulosa cells |
| Gaens K.H. et al., 2014 [21] | Obese RAGE- vs. RAGE + mice | Measured CML in plasma and adipose tissue | RAGE+: reduced plasma CML level with entrapment in the adipose tissue, altered inflammatory profile and glucose homeostasis |
| Diamanti-Kandarakis E. et al., 2015 [22] | KGN: human granulosa cell line | Culture was done with HGA or insulin or both HGA + insulin | Altered insulin signaling and Glut-4 translocation after HGA exposure |
| Diamanti-Kandarakis E. et al., 2008 [23] | Young lean non-insulin resistant women with PCOS vs. healthy women and vs. women with isolated features of PCOS | Measurement of serum AGEs | Elevated levels of AGEs in young lean non-insulin resistant women with PCOS |
| Mark A.B. et al., 2014 [24] | Overweight women | High-AGE vs. low-AGE diet | Low-AGE diet: lower fasting insulin levels, urinary AGEs, and insulin resistance |
| Tantalaki E. et al., 2014 [12] | Women with PCOS | High-AGE vs. low-AGE diet | Low-AGE diet: reduction in insulin level and HOMA |
| Diamanti-Kandarakis et al., 2009 [25] | Women with or without PCOS | Measured serum AMH and AGEs | Higher AMH and AGEs in PCOS women with ovulatory dysfunction. Positive correlation with AMH/AGEs ratio to number of follicles |
| Diamanti-Kandarakis E. et al., 2007 [10] | Women with or without PCOS | AGE and RAGE immunoreactivity | PCOS: higher AGE and RAGE immunoexpression in granulosa cells |
| Diamanti-Kandarakis et al., 2005 [26] | Women with or without PCOS | Measured serum AGE levels and RAGE expression in circulating monocytes | PCOS: higher AGEs’ levels with increased RAGE expression, higher testosterone and free androgen index (FAI), waist-to-hip ratio and HOMA |
| Gaens K.H. et al., 2014 [21] | Human preadipocytes | Measured CML levels and RAGE expression | The activation of AGE-RAGE axis is involved in the dysregulation of adipokines in obesity, thereby contributing to the development of obesity-associated insulin resistance |
2. Dietary AGEs and Their Receptors
2.1. Dietary AGEs Activate the Pro-Inflammatory Receptor RAGE
2.2. Soluble Receptor for AGEs (sRAGE) Protects against Dietary AGEs
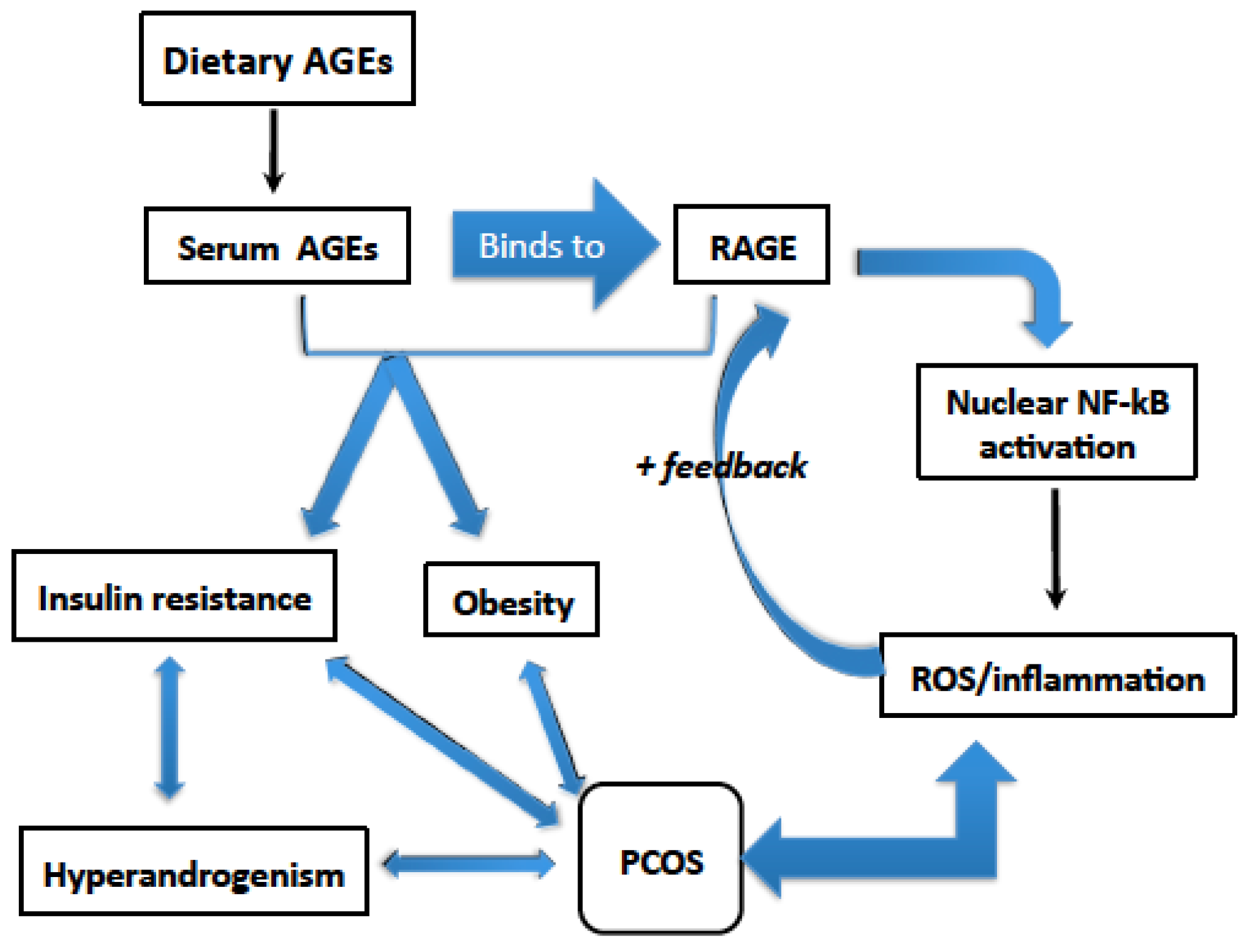
3. Dietary AGEs Induce Inflammation in PCOS
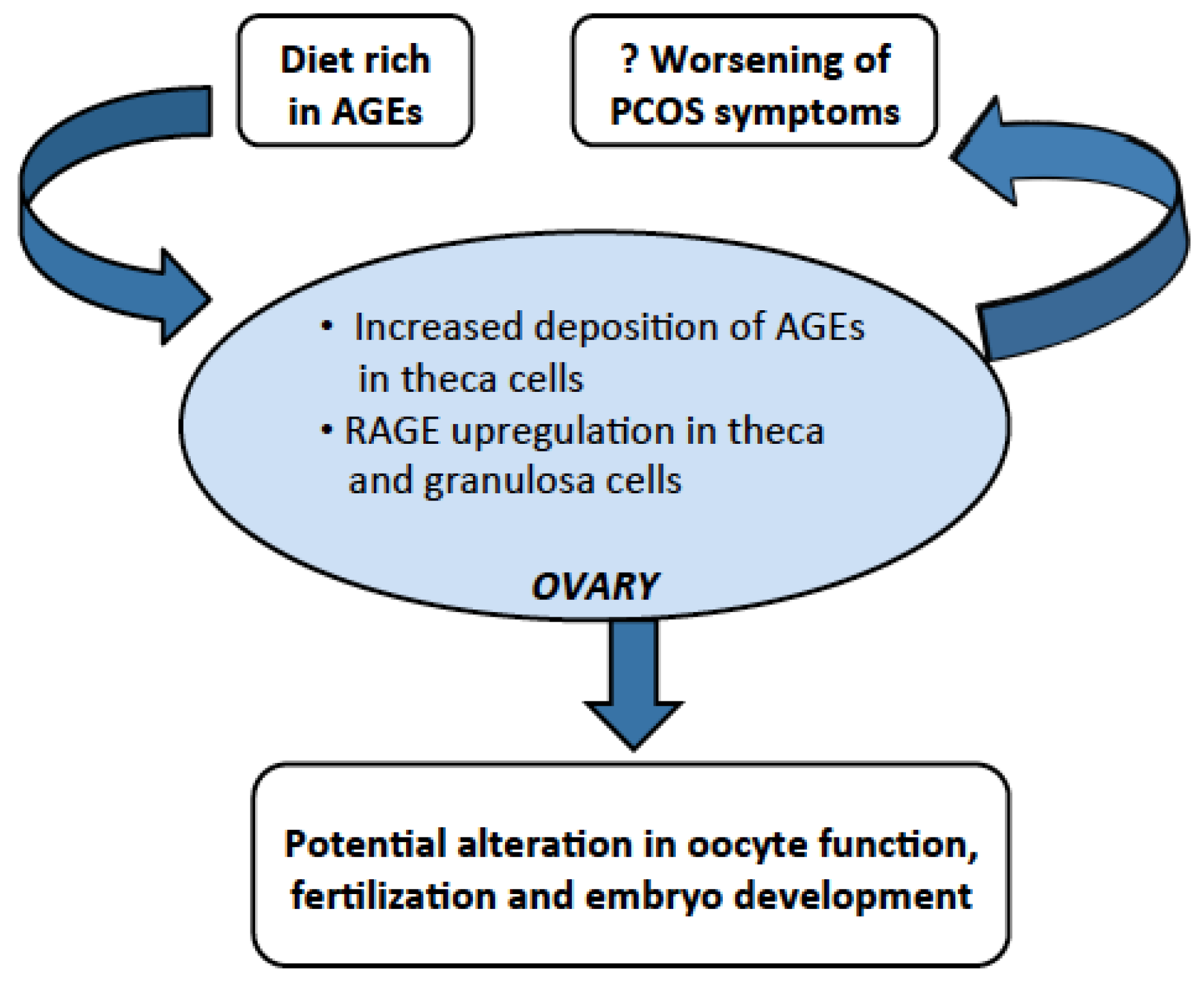
4. Dietary AGEs Cause Ovarian Dysfunction in PCOS
5. Dietary AGEs Are Associated with Insulin Resistance in PCOS
6. Dietary AGEs are Associated with Obesity in PCOS
7. Reduced Intake of Dietary AGEs and Inhibition of the AGE-RAGE System
| Methods | Effect on AGEs |
|---|---|
| 1. Change in the food preparation methods | |
| Reduce the dietary intake of AGEs |
| |
| 2. Vitamin D supplementation | Elevate serum sRAGE levels in women with PCOS |
| 3. Oral adsorption of dietary AGEs | |
|
|
| |
|
|
| 4. Insulin sensitizer | |
|
|
| |
| 5. Lipase inhibitor | Reduce post-meal serum AGEs’ levels |
| |
| 6. Alpha-lipoic acid (ALA) | Reduce formation of AGEs |
| 7. Pyridoxamine | Reduce formation of AGEs |
8. Conclusions
Author Contributions
Conflicts of Interest
References
- Azziz, R.; Woods, K.S.; Reyna, R.; Key, T.J.; Knochenhauer, E.S.; Yildiz, B.O. The prevalence and features of the polycystic ovary syndrome in an unselected population. J. Biol. Chem. 2004, 89, 2745–2749. [Google Scholar] [CrossRef] [PubMed]
- Diamanti-Kandarakis, E.; Christakou, C.; Marinakis, E. Phenotypes and enviromental factors: Their influence in PCOS. Curr. Pharm. Des. 2012, 18, 270–282. [Google Scholar] [CrossRef] [PubMed]
- Piperi, C.; Adamopoulos, C.; Dalagiorgou, G.; Diamanti-Kandarakis, E.; Papavassiliou, A.G. Crosstalk between advanced glycation and endoplasmic reticulum stress: Emerging therapeutic targeting for metabolic diseases. J. Biol. Chem. 2012, 97, 2231–2242. [Google Scholar] [CrossRef] [PubMed]
- O'Brien, J.; Morrissey, P.A. Nutritional and toxicological aspects of the maillard browning reaction in foods. J. Clin. Endocrinol. Metab. 1989, 28, 211–248. [Google Scholar] [CrossRef] [PubMed]
- Goldberg, T.; Cai, W.; Peppa, M.; Dardaine, V.; Baliga, B.S.; Uribarri, J.; Vlassara, H. Advanced glycoxidation end products in commonly consumed foods. J. Am. Diet. Assoc. 2004, 104, 1287–1291. [Google Scholar] [CrossRef] [PubMed]
- Tan, K.C.; Shiu, S.W.; Wong, Y.; Tam, X. Serum advanced glycation end products (AGEs) are associated with insulin resistance. Diet. Metab. Res. Rev. 2011, 27, 488–492. [Google Scholar] [CrossRef] [PubMed]
- Kume, S.; Takeya, M.; Mori, T.; Araki, N.; Suzuki, H.; Horiuchi, S.; Kodama, T.; Miyauchi, Y.; Takahashi, K. Immunohistochemical and ultrastructural detection of advanced glycation end products in atherosclerotic lesions of human aorta with a novel specific monoclonal antibody. Am. J. Pathol. 1995, 147, 654–667. [Google Scholar] [PubMed]
- Yan, S.D.; Yan, S.F.; Chen, X.; Fu, J.; Chen, M.; Kuppusamy, P.; Smith, M.A.; Perry, G.; Godman, G.C.; Nawroth, P.; et al. Non-enzymatically glycated tau in Alzheimer’s disease induces neuronal oxidant stress resulting in cytokine gene expression and release of amyloid beta-peptide. Nat. Med. 1995, 1, 693–699. [Google Scholar] [CrossRef] [PubMed]
- Ulrich, P.; Cerami, A. Protein glycation, diabetes, and aging. Recent Progress Horm. Res. 2001, 56, 1–21. [Google Scholar] [CrossRef]
- Diamanti-Kandarakis, E.; Piperi, C.; Patsouris, E.; Korkolopoulou, P.; Panidis, D.; Pawelczyk, L.; Papavassiliou, A.G.; Duleba, A.J. Immunohistochemical localization of advanced glycation end-products (AGEs) and their receptor (RAGE) in polycystic and normal ovaries. Histochem. Cell Biol. 2007, 127, 581–589. [Google Scholar] [CrossRef] [PubMed]
- Basta, G. Receptor for advanced glycation endproducts and atherosclerosis: From basic mechanisms to clinical implications. Atherosclerosis 2008, 196, 9–21. [Google Scholar] [CrossRef] [PubMed]
- Tantalaki, E.; Piperi, C.; Livadas, S.; Kollias, A.; Adamopoulos, C.; Koulouri, A.; Christakou, C.; Diamanti-Kandarakis, E. Impact of dietary modification of advanced glycation end products (AGEs) on the hormonal and metabolic profile of women with polycystic ovary syndrome (PCOS). Hormones 2014, 13, 65–73. [Google Scholar] [PubMed]
- Leuner, B.; Max, M.; Thamm, K.; Kausler, C.; Yakobus, Y.; Bierhaus, A.; Sel, S.; Hofmann, B.; Silber, R.E.; Simm, A.; et al. RAGE influences obesity in mice. Effects of the presence of RAGE on weight gain, AGE accumulation, and insulin levels in mice on a high fat diet. Z. Gerontol. Geriatr. 2012, 45, 102–108. [Google Scholar] [CrossRef] [PubMed]
- Cai, W.; Ramdas, M.; Zhu, L.; Chen, X.; Striker, G.E.; Vlassara, H. Oral advanced glycation endproducts (AGEs) promote insulin resistance and diabetes by depleting the antioxidant defenses AGE receptor-1 and sirtuin 1. Proc. Natl. Acad. Sci. USA 2012, 109, 15888–15893. [Google Scholar] [CrossRef] [PubMed]
- Hofmann, S.M.; Dong, H.J.; Li, Z.; Cai, W.; Altomonte, J.; Thung, S.N.; Zeng, F.; Fisher, E.A.; Vlassara, H. Improved insulin sensitivity is associated with restricted intake of dietary glycoxidation products in the db/db mouse. Diabetes 2002, 51, 2082–2089. [Google Scholar] [CrossRef] [PubMed]
- Sandu, O.; Song, K.; Cai, W.; Zheng, F.; Uribarri, J.; Vlassara, H. Insulin resistance and type 2 diabetes in high-fat-fed mice are linked to high glycotoxin intake. Diabetes 2005, 54, 2314–2319. [Google Scholar] [CrossRef] [PubMed]
- Chatzigeorgiou, A.; Kandaraki, E.; Piperi, C.; Livadas, S.; Papavassiliou, A.G.; Koutsilieris, M.; Papalois, A.; Diamanti-Kandarakis, E. Dietary glycotoxins affect scavenger receptor expression and the hormonal profile of female rats. J. Endocrinol. 2013, 218, 331–337. [Google Scholar] [CrossRef] [PubMed]
- Cassese, A.; Esposito, I.; Fiory, F.; Barbagallo, A.P.; Paturzo, F.; Mirra, P.; Ulianich, L.; Giacco, F.; Iadicicco, C.; Lombardi, A.; et al. In skeletal muscle advanced glycation end products (AGEs) inhibit insulin action and induce the formation of multimolecular complexes including the receptor for AGEs. J. Biol. Chem. 2008, 283, 36088–36099. [Google Scholar] [CrossRef] [PubMed]
- Kandaraki, E.; Chatzigeorgiou, A.; Piperi, C.; Palioura, E.; Palimeri, S.; Korkolopoulou, P.; Koutsilieris, M.; Papavassiliou, A.G. Reduced ovarian glyoxalase-I activity by dietary glycotoxins and androgen excess: A causative link to polycystic ovarian syndrome. Mol. Med. 2012, 18, 1183–1189. [Google Scholar] [CrossRef] [PubMed]
- Diamanti-Kandarakis, E.; Piperi, C.; Korkolopoulou, P.; Kandaraki, E.; Levidou, G.; Papalois, A.; Patsouris, E.; Papavassiliou, A.G. Accumulation of dietary glycotoxins in the reproductive system of normal female rats. J. Mol. Med. 2007, 85, 1413–1420. [Google Scholar] [CrossRef] [PubMed]
- Gaens, K.H.; Goossens, G.H.; Niessen, P.M.; van Greevenbroek, M.M.; van der Kallen, C.J.; Niessen, H.W.; Rensen, S.S.; Buurman, W.A.; Greve, J.W.; Blaak, E.E.; et al. Nepsilon-(carboxymethyl)lysine-receptor for advanced glycation end product axis is a key modulator of obesity-induced dysregulation of adipokine expression and insulin resistance. Arterioscler. Thromb. Vasc. Biol. 2014, 34, 1199–1208. [Google Scholar] [CrossRef] [PubMed]
- Diamanti-Kandarakis, E.; Chatzigeorgiou, A.; Papageorgiou, E.; Koundouras, D.; Koutsilieris, M. Advanced glycation end-products and insulin signaling in granulosa cells. Exp. Biol. Med. 2015. [Google Scholar] [CrossRef] [PubMed]
- Diamanti-Kandarakis, E.; Katsikis, I.; Piperi, C.; Kandaraki, E.; Piouka, A.; Papavassiliou, A.G.; Panidis, D. Increased serum advanced glycation end-products is a distinct finding in lean women with polycystic ovary syndrome (PCOs). Clin. Endocrinol. 2008, 69, 634–641. [Google Scholar] [CrossRef] [PubMed]
- Mark, A.B.; Poulsen, M.W.; Andersen, S.; Andersen, J.M.; Bak, M.J.; Ritz, C.; Holst, J.J.; Nielsen, J.; de Courten, B.; Dragsted, L.O.; et al. Consumption of a diet low in advanced glycation end products for 4 weeks improves insulin sensitivity in overweight women. Diabetes Care 2014, 37, 88–95. [Google Scholar] [CrossRef] [PubMed]
- Diamanti-Kandarakis, E.; Piouka, A.; Livadas, S.; Piperi, C.; Katsikis, I.; Papavassiliou, A.G.; Panidis, D. Anti-mullerian hormone is associated with advanced glycosylated end products in lean women with polycystic ovary syndrome. Eur. J. Endocrinol. 2009, 160, 847–853. [Google Scholar] [CrossRef] [PubMed]
- Diamanti-Kandarakis, E.; Piperi, C.; Kalofoutis, A.; Creatsas, G. Increased levels of serum advanced glycation end-products in women with polycystic ovary syndrome. Clin. Endocrinol. 2005, 62, 37–43. [Google Scholar] [CrossRef] [PubMed]
- Inagi, R. Inhibitors of advanced glycation and endoplasmic reticulum stress. Methods Enzymol. 2011, 491, 361–380. [Google Scholar] [PubMed]
- Singh, R.; Barden, A.; Mori, T.; Beilin, L. Advanced glycation end-products: A review. Diabetologia 2001, 44, 129–146. [Google Scholar] [CrossRef] [PubMed]
- Cerami, C.; Founds, H.; Nicholl, I.; Mitsuhashi, T.; Giordano, D.; Vanpatten, S.; Lee, A.; al-Abed, Y.; Vlassara, H.; Bucala, R.; et al. Tobacco smoke is a source of toxic reactive glycation products. Proc. Natl. Acad. Sci. USA 1997, 94, 13915–13920. [Google Scholar] [CrossRef] [PubMed]
- Vlassara, H.; Uribarri, J. Glycoxidation and diabetic complications: Modern lessons and a warning? Rev. Endocrine Metab. Disord. 2004, 5, 181–188. [Google Scholar] [CrossRef] [PubMed]
- Koschinsky, T.; He, C.J.; Mitsuhashi, T.; Bucala, R.; Liu, C.; Buenting, C.; Heitmann, K.; Vlassara, H. Orally absorbed reactive glycation products (glycotoxins): An environmental risk factor in diabetic nephropathy. Proc. Natl. Acad. Sci. USA 1997, 94, 6474–6479. [Google Scholar] [CrossRef] [PubMed]
- Uribarri, J.; Woodruff, S.; Goodman, S.; Cai, W.; Chen, X.; Pyzik, R.; Yong, A.; Striker, G.E.; Vlassara, H. Advanced glycation end products in foods and a practical guide to their reduction in the diet. J. Am. Diet. Assoc. 2010, 110, 911–916. [Google Scholar] [CrossRef] [PubMed]
- Story, M.; Hayes, M.; Kalina, B. Availability of foods in high schools: Is there cause for concern? J. Am. Diet. Assoc. 1996, 96, 123–126. [Google Scholar] [CrossRef]
- Bucala, R.; Makita, Z.; Koschinsky, T.; Cerami, A.; Vlassara, H. Lipid advanced glycosylation: Pathway for lipid oxidation in vivo. Proc. Natl. Acad. Sci. USA 1993, 90, 6434–6438. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, N.; Mirshekar-Syahkal, B.; Kennish, L.; Karachalias, N.; Babaei-Jadidi, R.; Thornalley, P.J. Assay of advanced glycation endproducts in selected beverages and food by liquid chromatography with tandem mass spectrometric detection. Mol. Nutr. Food Res. 2005, 49, 691–699. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, A.M.; Yan, S.D.; Yan, S.F.; Stern, D.M. The biology of the receptor for advanced glycation end products and its ligands. Biochim. Biophys. Acta 2000, 1498, 99–111. [Google Scholar] [CrossRef]
- Kalea, A.Z.; Schmidt, A.M.; Hudson, B.I. RAGE: A novel biological and genetic marker for vascular disease. Clin. Sci. 2009, 116, 621–637. [Google Scholar] [CrossRef] [PubMed]
- Wautier, M.P.; Chappey, O.; Corda, S.; Stern, D.M.; Schmidt, A.M.; Wautier, J.L. Activation of NADPH oxidase by AGE links oxidant stress to altered gene expression via RAGE. Am. J. Pathol. Endocrinol. Metab. 2001, 280, E685–E694. [Google Scholar]
- Raucci, A.; Cugusi, S.; Antonelli, A.; Barabino, S.M.; Monti, L.; Bierhaus, A.; Reiss, K.; Saftig, P.; Bianchi, M.E. A soluble form of the receptor for advanced glycation endproducts (RAGE) is produced by proteolytic cleavage of the membrane-bound form by the sheddase a disintegrin and metalloprotease 10 (ADAM10). FASEB J. 2008, 22, 3716–3727. [Google Scholar] [CrossRef] [PubMed]
- Koyama, H.; Shoji, T.; Yokoyama, H.; Motoyama, K.; Mori, K.; Fukumoto, S.; Emoto, M.; Shoji, T.; Tamei, H.; Matsuki, H.; et al. Plasma level of endogenous secretory RAGE is associated with components of the metabolic syndrome and atherosclerosis. Arterioscler. Thromb. Vasc. Biol. 2005, 25, 2587–2593. [Google Scholar] [CrossRef] [PubMed]
- Hanford, L.E.; Enghild, J.J.; Valnickova, Z.; Petersen, S.V.; Schaefer, L.M.; Schaefer, T.M.; Reinhart, T.A.; Oury, T.D. Purification and characterization of mouse soluble receptor for advanced glycation end products (sRAGE). J. Biol. Chem. 2004, 279, 50019–50024. [Google Scholar] [CrossRef] [PubMed]
- Park, L.; Raman, K.G.; Lee, K.J.; Lu, Y.; Ferran, L.J., Jr.; Chow, W.S.; Stern, D.; Schmidt, A.M. Suppression of accelerated diabetic atherosclerosis by the soluble receptor for advanced glycation endproducts. Nat. Med. 1998, 4, 1025–1031. [Google Scholar] [CrossRef] [PubMed]
- Goova, M.T.; Li, J.; Kislinger, T.; Qu, W.; Lu, Y.; Bucciarelli, L.G.; Nowygrod, S.; Wolf, B.M.; Caliste, X.; Yan, S.F.; et al. Blockade of receptor for advanced glycation end-products restores effective wound healing in diabetic mice. Am. J. Pathol. 2001, 159, 513–525. [Google Scholar] [CrossRef]
- Basta, G.; Sironi, A.M.; Lazzerini, G.; del Turco, S.; Buzzigoli, E.; Casolaro, A.; Natali, A.; Ferrannini, E.; Gastaldelli, A. Circulating soluble receptor for advanced glycation end products is inversely associated with glycemic control and S100A12 protein. J. Biol. Chem. 2006, 91, 4628–4634. [Google Scholar] [CrossRef] [PubMed]
- Bonetti, T.C.; Borges, E., Jr.; Braga, D.P.; Iaconelli, A., Jr.; Kleine, J.P.; Silva, I.D. Intrafollicular soluble receptor for advanced glycation end products (sRAGE) and embryo quality in assisted reproduction. Reprod. Biomed. Online 2013, 26, 62–67. [Google Scholar] [CrossRef] [PubMed]
- Vlassara, H.; Cai, W.; Crandall, J.; Goldberg, T.; Oberstein, R.; Dardaine, V.; Peppa, M.; Rayfield, E.J. Inflammatory mediators are induced by dietary glycotoxins, a major risk factor for diabetic angiopathy. Proc. Natl. Acad. Sci. USA 2002, 99, 15596–15601. [Google Scholar] [CrossRef] [PubMed]
- Uribarri, J.; Cai, W.; Peppa, M.; Goodman, S.; Ferrucci, L.; Striker, G.; Vlassara, H. Circulating glycotoxins and dietary advanced glycation endproducts: Two links to inflammatory response, oxidative stress, and aging. J. Gerontol. Ser. A Biol. Sci. Med. Sci. 2007, 62, 427–433. [Google Scholar] [CrossRef]
- Cai, W.; Gao, Q.D.; Zhu, L.; Peppa, M.; He, C.; Vlassara, H. Oxidative stress-inducing carbonyl compounds from common foods: Novel mediators of cellular dysfunction. Mol. Med. 2002, 8, 337–346. [Google Scholar] [PubMed]
- Gonzalez, F.; Rote, N.S.; Minium, J.; Kirwan, J.P. Increased activation of nuclear factor κB triggers inflammation and insulin resistance in polycystic ovary syndrome. J. Biol. Chem. 2006, 91, 1508–1512. [Google Scholar] [CrossRef] [PubMed]
- Thornalley, P.J. Glyoxalase I-structure, function and a critical role in the enzymatic defence against glycation. Biochem. Soc. Trans. 2003, 31, 1343–1348. [Google Scholar] [CrossRef] [PubMed]
- Brouwers, O.; Niessen, P.M.; Ferreira, I.; Miyata, T.; Scheffer, P.G.; Teerlink, T.; Schrauwen, P.; Brownlee, M.; Stehouwer, C.D.; Schalkwijk, C.G. Overexpression of glyoxalase-I reduces hyperglycemia-induced levels of advanced glycation end products and oxidative stress in diabetic rats. J. Biol. Chem. 2011, 286, 1374–1380. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.M.; Kim, Y.S.; Jung, D.H.; Lee, J.; Kim, J.S. Increased glyoxalase i levels inhibit accumulation of oxidative stress and an advanced glycation end product in mouse mesangial cells cultured in high glucose. Exp. Cell Res. 2012, 318, 152–159. [Google Scholar] [CrossRef] [PubMed]
- Tatone, C.; Eichenlaub-Ritter, U.; Amicarelli, F. Dicarbonyl stress and glyoxalases in ovarian function. Biochem. Soc. Trans. 2014, 42, 433–438. [Google Scholar] [CrossRef] [PubMed]
- Park, J.H.; Choi, T.S. Polycystic ovary syndrome (PCOS)-like phenotypes in the d-galactose-induced aging mouse model. Biochem. Biophys. Res. Commun. 2012, 427, 701–704. [Google Scholar] [CrossRef] [PubMed]
- Tatone, C.; Heizenrieder, T.; di Emidio, G.; Treffon, P.; Amicarelli, F.; Seidel, T.; Eichenlaub-Ritter, U. Evidence that carbonyl stress by methylglyoxal exposure induces DNA damage and spindle aberrations, affects mitochondrial integrity in mammalian oocytes and contributes to oocyte ageing. Hum. Reprod. 2011, 26, 1843–1859. [Google Scholar] [CrossRef] [PubMed]
- Chang, Y.J.; Chan, W.H. Methylglyoxal has injurious effects on maturation of mouse oocytes, fertilization, and fetal development, via apoptosis. Toxicol. Lett. 2010, 193, 217–223. [Google Scholar] [CrossRef] [PubMed]
- Sutton-McDowall, M.L.; Gilchrist, R.B.; Thompson, J.G. The pivotal role of glucose metabolism in determining oocyte developmental competence. Reproduction 2010, 139, 685–695. [Google Scholar] [CrossRef] [PubMed]
- Diamanti-Kandarakis, E.; Piperi, C.; Livadas, S.; Kandaraki, E.A.; Papageorgiou, E.; Koutsilieris, M. Interference of AGE-RAGE signaling with steroidogenic enzyme action in human ovarian cells. In Proceedings of the Endocrine Society’s 95th Annual Meeting and Expo, San Francisco, CA, USA, 15–18 June 2013.
- Su, Y.-Q.; Denegre, J.M.; Wigglesworth, K.; Pendola, F.L.; O’Brien, M.J.; Eppig, J.J. Oocyte-dependent activation of mitogen-activated protein kinase (ERK1/2) in cumulus cells is required for the maturation of the mouse oocyte–cumulus cell complex. Dev. Biol. 2003, 263, 126–138. [Google Scholar] [CrossRef]
- La Marca, A.; Orvieto, R.; Giulini, S.; Jasonni, V.M.; Volpe, A.; de Leo, V. Mullerian-inhibiting substance in women with polycystic ovary syndrome: Relationship with hormonal and metabolic characteristics. Fertil. Steril. 2004, 82, 970–972. [Google Scholar] [CrossRef] [PubMed]
- Weenen, C.; Laven, J.S.; von Bergh, A.R.; Cranfield, M.; Groome, N.P.; Visser, J.A.; Kramer, P.; Fauser, B.C.; Themmen, A.P. Anti-mullerian hormone expression pattern in the human ovary: Potential implications for initial and cyclic follicle recruitment. Mol. Hum. Reprod. 2004, 10, 77–83. [Google Scholar] [CrossRef] [PubMed]
- Pigny, P.; Merlen, E.; Robert, Y.; Cortet-Rudelli, C.; Decanter, C.; Jonard, S.; Dewailly, D. Elevated serum level of anti-mullerian hormone in patients with polycystic ovary syndrome: Relationship to the ovarian follicle excess and to the follicular arrest. J. Biol. Chem. 2003, 88, 5957–5962. [Google Scholar] [CrossRef] [PubMed]
- Legro, R.S.; Kunselman, A.R.; Dodson, W.C.; Dunaif, A. Prevalence and predictors of risk for type 2 diabetes mellitus and impaired glucose tolerance in polycystic ovary syndrome: A prospective, controlled study in 254 affected women. J. Biol. Chem. 1999, 84, 165–169. [Google Scholar] [CrossRef]
- Burghen, G.A.; Givens, J.R.; Kitabchi, A.E. Correlation of hyperandrogenism with hyperinsulinism in polycystic ovarian disease. J. Biol. Chem. 1980, 50, 113–116. [Google Scholar] [CrossRef] [PubMed]
- Diamanti-Kandarakis, E. Insulin resistance in PCOS. Endocrine 2006, 30, 13–17. [Google Scholar] [CrossRef]
- Peppa, M.; Brem, H.; Ehrlich, P.; Zhang, J.G.; Cai, W.; Li, Z.; Croitoru, A.; Thung, S.; Vlassara, H. Adverse effects of dietary glycotoxins on wound healing in genetically diabetic mice. Diabetes 2003, 52, 2805–2813. [Google Scholar] [CrossRef] [PubMed]
- Diamanti-Kandarakis, E.; Lambrinoudaki, I.; Economou, F.; Christou, M.; Piperi, C.; Papavassiliou, A.G.; Creatsas, G. Androgens associated with advanced glycation end-products in postmenopausal women. Menopause 2010, 17, 1182–1187. [Google Scholar] [CrossRef] [PubMed]
- Unoki, H.; Yamagishi, S. Advanced glycation end products and insulin resistance. Curr. Pharm. Des. 2008, 14, 987–989. [Google Scholar] [CrossRef] [PubMed]
- Merhi, Z. Advanced glycation end products and their relevance in female reproduction. Hum. Reprod. 2014, 29, 135–145. [Google Scholar] [CrossRef] [PubMed]
- Hernandez-Valencia, M.; Hernandez-Quijano, T.; Vargas-Giron, A.; Vargas-Lopez, C.; Arturo, Z. Decreased insulin resistance with amino acids, extracts and antioxidants in patients with polycystic ovary syndrome. Ginecol. Obstet. Mex. 2013, 81, 573–577. [Google Scholar] [PubMed]
- Barbieri, R.L.; Smith, S.; Ryan, K.J. The role of hyperinsulinemia in the pathogenesis of ovarian hyperandrogenism. Fertil. Steril. 1988, 50, 197–212. [Google Scholar] [PubMed]
- Gonzalez, D.; Rojas, A.; White, A.K.; Romero, J.; Davies, J.; Gracia, R.; Velez, C.; Joels, L.; White, J.O.; Conlan, R.S. Synergistic Effect of RAGE and AR Pathways in PCOS. In Proceedings of the Endocrine Society’s 94th Annual Meeting and Expo, Houston, TX, USA, 23–26 June 2012.
- Ehrmann, D.A. Polycystic ovary syndrome. N. Engl. J. Med. 2005, 352, 1223–1236. [Google Scholar] [CrossRef] [PubMed]
- Wright, C.E.; Zborowski, J.V.; Talbott, E.O.; McHugh-Pemu, K.; Youk, A. Dietary intake, physical activity, and obesity in women with polycystic ovary syndrome. Int. J. Obes. Relat. Metab. Disord. 2004, 28, 1026–1032. [Google Scholar] [CrossRef] [PubMed]
- Larsson, I.; Hulthen, L.; Landen, M.; Palsson, E.; Janson, P.; Stener-Victorin, E. Dietary intake, resting energy expenditure, and eating behavior in women with and without polycystic ovary syndrome. Clin. Nutr. 2015. [Google Scholar] [CrossRef] [PubMed]
- Georgopoulos, N.A.; Saltamavros, A.D.; Vervita, V.; Karkoulias, K.; Adonakis, G.; Decavalas, G.; Kourounis, G.; Markou, K.B.; Kyriazopoulou, V. Basal metabolic rate is decreased in women with polycystic ovary syndrome and biochemical hyperandrogenemia and is associated with insulin resistance. Fertil. Steril. 2009, 92, 250–255. [Google Scholar] [CrossRef] [PubMed]
- Picard, F.; Kurtev, M.; Chung, N.; Topark-Ngarm, A.; Senawong, T.; Machado De Oliveira, R.; Leid, M.; McBurney, M.W.; Guarente, L. Sirt1 promotes fat mobilization in white adipocytes by repressing PPAR-γ. Nature 2004, 429, 771–776. [Google Scholar] [CrossRef] [PubMed]
- Jia, X.; Chang, T.; Wilson, T.W.; Wu, L. Methylglyoxal mediates adipocyte proliferation by increasing phosphorylation of Akt1. PLoS ONE 2012, 7, e36610. [Google Scholar] [CrossRef] [PubMed]
- Yamamoto, Y.; Yamamoto, H. RAGE-mediated inflammation, type 2 diabetes, and diabetic vascular complication. Front. Endocrinol. 2013, 4. [Google Scholar] [CrossRef] [PubMed]
- Ueno, H.; Koyama, H.; Shoji, T.; Monden, M.; Fukumoto, S.; Tanaka, S.; Otsuka, Y.; Mima, Y.; Morioka, T.; Mori, K.; et al. Receptor for advanced glycation end-products (RAGE) regulation of adiposity and adiponectin is associated with atherogenesis in apoe-deficient mouse. Atherosclerosis 2010, 211, 431–436. [Google Scholar] [CrossRef] [PubMed]
- He, C.T.; Lee, C.H.; Hsieh, C.H.; Hsiao, F.C. Soluble form of receptor for advanced glycation end products is associated with obesity and metabolic syndrome in adolescents. 2014. [Google Scholar] [CrossRef] [PubMed]
- Davis, K.E.; Prasad, C.; Vijayagopal, P.; Juma, S.; Imrhan, V. Serum soluble receptor for advanced glycation end products correlates inversely with measures of adiposity in young adults. Nutr. Res. 2014, 34, 478–485. [Google Scholar] [CrossRef] [PubMed]
- Brix, J.M.; Hollerl, F.; Kopp, H.P.; Schernthaner, G.H.; Schernthaner, G. The soluble form of the receptor of advanced glycation endproducts increases after bariatric surgery in morbid obesity. Int. J. Obes. 2012, 36, 1412–1417. [Google Scholar] [CrossRef] [PubMed]
- Phelan, N.; O’Connor, A.; Kyaw Tun, T.; Correia, N.; Boran, G.; Roche, H.M.; Gibney, J. Hormonal and metabolic effects of polyunsaturated fatty acids in young women with polycystic ovary syndrome: Results from a cross-sectional analysis and a randomized, placebo-controlled, crossover trial. Am. J. Clin. Nutr. 2011, 93, 652–662. [Google Scholar] [CrossRef] [PubMed]
- Rafraf, M.; Mohammadi, E.; Asghari-Jafarabadi, M.; Farzadi, L. Omega-3 fatty acids improve glucose metabolism without effects on obesity values and serum visfatin levels in women with polycystic ovary syndrome. J. Am. Coll. Nutr. 2012, 31, 361–368. [Google Scholar] [CrossRef] [PubMed]
- Cai, W.; He, J.C.; Zhu, L.; Chen, X.; Zheng, F.; Striker, G.E.; Vlassara, H. Oral glycotoxins determine the effects of calorie restriction on oxidant stress, age-related diseases, and lifespan. Am. J. Pathol. 2008, 173, 327–336. [Google Scholar] [CrossRef] [PubMed]
- Teillet, L.; Verbeke, P.; Gouraud, S.; Bakala, H.; Borot-Laloi, C.; Heudes, D.; Bruneval, P.; Corman, B. Food restriction prevents advanced glycation end product accumulation and retards kidney aging in lean rats. J. Am. Soc. Nephrol. 2000, 11, 1488–1497. [Google Scholar] [PubMed]
- Gugliucci, A.; Kotani, K.; Taing, J.; Matsuoka, Y.; Sano, Y.; Yoshimura, M.; Egawa, K.; Horikawa, C.; Kitagawa, Y.; Kiso, Y.; et al. Short-term low calorie diet intervention reduces serum advanced glycation end products in healthy overweight or obese adults. Ann. Nutr. Metab. 2009, 54, 197–201. [Google Scholar] [CrossRef] [PubMed]
- Irani, M.; Minkoff, H.; Seifer, D.B.; Merhi, Z. Vitamin d increases serum levels of the soluble receptor for advanced glycation end products in women with pcos. J. Biol. Chem. 2014, 99, E886–E890. [Google Scholar] [CrossRef] [PubMed]
- Sung, J.Y.; Chung, W.; Kim, A.J.; Kim, H.S.; Ro, H.; Chang, J.H.; Lee, H.H.; Jung, J.Y. Calcitriol treatment increases serum levels of the soluble receptor of advanced glycation end products in hemodialysis patients with secondary hyperparathyroidism. Tohoku J. Exp. Med. 2013, 230, 59–66. [Google Scholar] [CrossRef] [PubMed]
- Sebekova, K.; Sturmer, M.; Fazeli, G.; Bahner, U.; Stab, F.; Heidland, A. Is vitamin D deficiency related to accumulation of advanced glycation end products, markers of inflammation, and oxidative stress in diabetic subjects? BioMed Res. Int. 2015. [Google Scholar] [CrossRef]
- Salum, E.; Kals, J.; Kampus, P.; Salum, T.; Zilmer, K.; Aunapuu, M.; Arend, A.; Eha, J.; Zilmer, M. Vitamin D reduces deposition of advanced glycation end-products in the aortic wall and systemic oxidative stress in diabetic rats. Diabetes Res. Clin. Pract. 2013, 100, 243–249. [Google Scholar] [CrossRef] [PubMed]
- Peng, X.; Ma, J.; Chen, F.; Wang, M. Naturally occurring inhibitors against the formation of advanced glycation end-products. Food Funct. 2011, 2, 289–301. [Google Scholar] [CrossRef] [PubMed]
- Yamagishi, S.; Nakamura, K.; Matsui, T.; Inoue, H.; Takeuchi, M. Oral administration of AST-120 (Kremezin) is a promising therapeutic strategy for advanced glycation end product (AGE)-related disorders. Med. Hypotheses 2007, 69, 666–668. [Google Scholar] [CrossRef] [PubMed]
- Diamanti-Kandarakis, E.; Alexandraki, K.; Piperi, C.; Aessopos, A.; Paterakis, T.; Katsikis, I.; Panidis, D. Effect of metformin administration on plasma advanced glycation end product levels in women with polycystic ovary syndrome. Metab. Clin. Exp. 2007, 56, 129–134. [Google Scholar] [CrossRef] [PubMed]
- Diamanti-Kandarakis, E.; Katsikis, I.; Piperi, C.; Alexandraki, K.; Panidis, D. Effect of long-term orlistat treatment on serum levels of advanced glycation end-products in women with polycystic ovary syndrome. Clin. Endocrinol. 2007, 66, 103–109. [Google Scholar] [CrossRef] [PubMed]
- Ueda, S.; Yamagishi, S.; Takeuchi, M.; Kohno, K.; Shibata, R.; Matsumoto, Y.; Kaneyuki, U.; Fujimura, T.; Hayashida, A.; Okuda, S. Oral adsorbent AST-120 decreases serum levels of AGEs in patients with chronic renal failure. Mol. Med. 2006, 12, 180–184. [Google Scholar] [CrossRef] [PubMed]
- Dewailly, D.; Pigny, P.; Soudan, B.; Catteau-Jonard, S.; Decanter, C.; Poncelet, E.; Duhamel, A. Reconciling the definitions of polycystic ovary syndrome: The ovarian follicle number and serum anti-mullerian hormone concentrations aggregate with the markers of hyperandrogenism. J. Biol. Chem. 2010, 95, 4399–4405. [Google Scholar] [CrossRef] [PubMed]
- He, C.; Sabol, J.; Mitsuhashi, T.; Vlassara, H. Dietary glycotoxins: Inhibition of reactive products by aminoguanidine facilitates renal clearance and reduces tissue sequestration. Diabetes 1999, 48, 1308–1315. [Google Scholar] [CrossRef] [PubMed]
- Marchetti, P.; Del Guerra, S.; Marselli, L.; Lupi, R.; Masini, M.; Pollera, M.; Bugliani, M.; Boggi, U.; Vistoli, F.; Mosca, F.; et al. Pancreatic islets from type 2 diabetic patients have functional defects and increased apoptosis that are ameliorated by metformin. J. Biol. Chem. 2004, 89, 5535–5541. [Google Scholar] [CrossRef] [PubMed]
- Schurman, L.; McCarthy, A.D.; Sedlinsky, C.; Gangoiti, M.V.; Arnol, V.; Bruzzone, L.; Cortizo, A.M. Metformin reverts deleterious effects of advanced glycation end-products (AGEs) on osteoblastic cells. Exp. Clin. Endocrinol. Diabetes 2008, 116, 333–340. [Google Scholar] [CrossRef] [PubMed]
- Ouslimani, N.; Mahrouf, M.; Peynet, J.; Bonnefont-Rousselot, D.; Cosson, C.; Legrand, A.; Beaudeux, J.L. Metformin reduces endothelial cell expression of both the receptor for advanced glycation end products and lectin-like oxidized receptor 1. Metab. Clin. Exp. 2007, 56, 308–313. [Google Scholar] [CrossRef] [PubMed]
- Diamanti-Kandarakis, E.; Piperi, C.; Alexandraki, K.I.; Papailiou, J.; Ekonomou, F.; Koulouri, E.; Kandarakis, H.; Creatsas, G. The acute effect of orlistat on dietary glycotoxins in diabetics and healthy women. Min. Endocrinol. 2009, 34, 97–104. [Google Scholar]
- Diamanti-Kandarakis, E.; Piperi, C.; Alexandraki, K.; Katsilambros, N.; Kouroupi, E.; Papailiou, J.; Lazaridis, S.; Koulouri, E.; Kandarakis, H.A.; Douzinas, E.E.; et al. Short-term effect of orlistat on dietary glycotoxins in healthy women and women with polycystic ovary syndrome. Metab. Clin. Exp. 2006, 55, 494–500. [Google Scholar] [CrossRef] [PubMed]
- Desai, K.; Wu, L. Methylglyoxal and advanced glycation endproducts: New therapeutic horizons? Recent Pat. Cardiovasc. Drug Discov. 2007, 2, 89–99. [Google Scholar] [CrossRef] [PubMed]
- Peyroux, J.; Sternberg, M. Advanced glycation endproducts (AGEs): Pharmacological inhibition in diabetes. Pathol. Biol. 2006, 54, 405–419. [Google Scholar] [CrossRef] [PubMed]
- Nangaku, M.; Miyata, T.; Sada, T.; Mizuno, M.; Inagi, R.; Ueda, Y.; Ishikawa, N.; Yuzawa, H.; Koike, H.; van Ypersele de Strihou, C.; et al. Anti-hypertensive agents inhibit in vivo the formation of advanced glycation end products and improve renal damage in a type 2 diabetic nephropathy rat model. J. Am. Soc. Nephrol. 2003, 14, 1212–1222. [Google Scholar] [CrossRef] [PubMed]
- Rahbar, S.; Natarajan, R.; Yerneni, K.; Scott, S.; Gonzales, N.; Nadler, J.L. Evidence that pioglitazone, metformin and pentoxifylline are inhibitors of glycation. Clin. Chim. Acta 2000, 301, 65–77. [Google Scholar] [CrossRef]
- Yamagishi, S.-I.; Nakamura, K.; Matsui, T.; Ueda, S.; Fukami, K.; Okuda, S. Agents that block advanced glycation end product (AGE)-RAGE (receptor for AGEs)-oxidative stress system: A novel therapeutic strategy for diabetic vascular complications. Exp. Opin. Investig. Drugs 2008, 17, 983–996. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Garg, D.; Merhi, Z. Advanced Glycation End Products: Link between Diet and Ovulatory Dysfunction in PCOS? Nutrients 2015, 7, 10129-10144. https://doi.org/10.3390/nu7125524
Garg D, Merhi Z. Advanced Glycation End Products: Link between Diet and Ovulatory Dysfunction in PCOS? Nutrients. 2015; 7(12):10129-10144. https://doi.org/10.3390/nu7125524
Chicago/Turabian StyleGarg, Deepika, and Zaher Merhi. 2015. "Advanced Glycation End Products: Link between Diet and Ovulatory Dysfunction in PCOS?" Nutrients 7, no. 12: 10129-10144. https://doi.org/10.3390/nu7125524
APA StyleGarg, D., & Merhi, Z. (2015). Advanced Glycation End Products: Link between Diet and Ovulatory Dysfunction in PCOS? Nutrients, 7(12), 10129-10144. https://doi.org/10.3390/nu7125524





