Resveratrol as a Bioenhancer to Improve Anti-Inflammatory Activities of Apigenin
Abstract
:1. Introduction
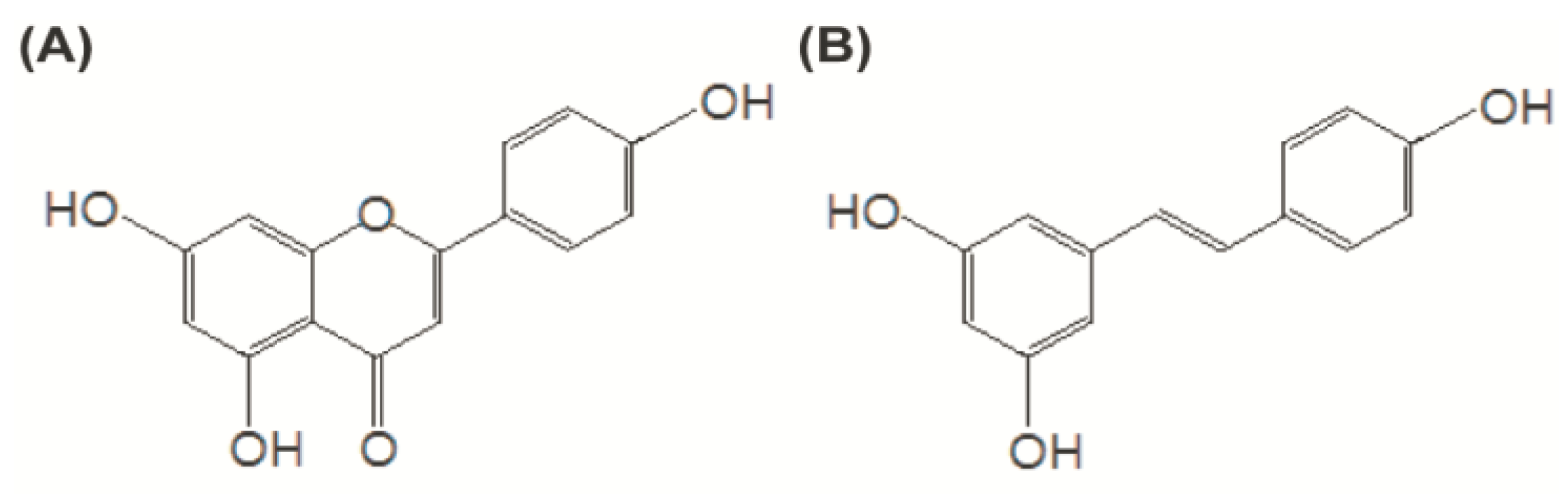
2. Materials and Methods
2.1. Cell Culture
2.2. Production of Hepatic Metabolites of Apigenin and/or Resveratrol in HepG2 Cells
2.3. Determination of NO Production
2.4. Measurement of PGE2 and Pro-Inflammatory Cytokine Production
2.5. Preparation of Whole Cell Extracts
2.6. Western Blot Analysis
2.7. Experimental Animal
2.8. Carrageenan-Induced Paw Edema in Mice
2.9. Apigenin and Apigenin Metabolite Analysis
2.10. Kinetics of Glucuronidation of Apigenin by UGT1A9
2.11. HPLC Analysis
2.12. Data Analysis
2.13. Statistical Analysis
3. Results
3.1. Inhibition of NO and PGE2 Production, and iNOS and COX-2 Expressions in Vitro
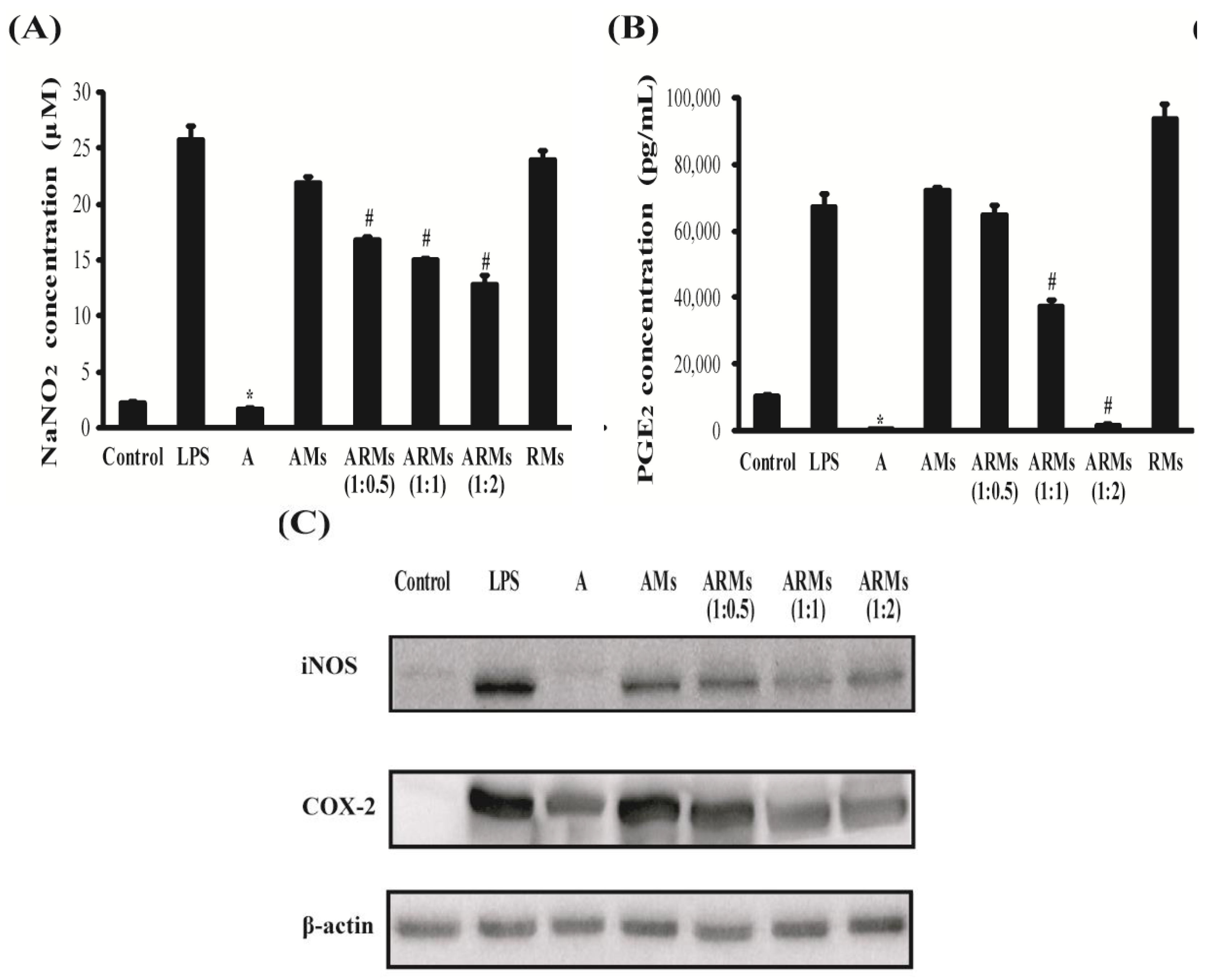
3.2. Inhibition of Secretion of Pro-Inflammatory Cytokines in RAW 264.7 Cells

3.3. Carrageenan-Induced Paw Edema in Mice
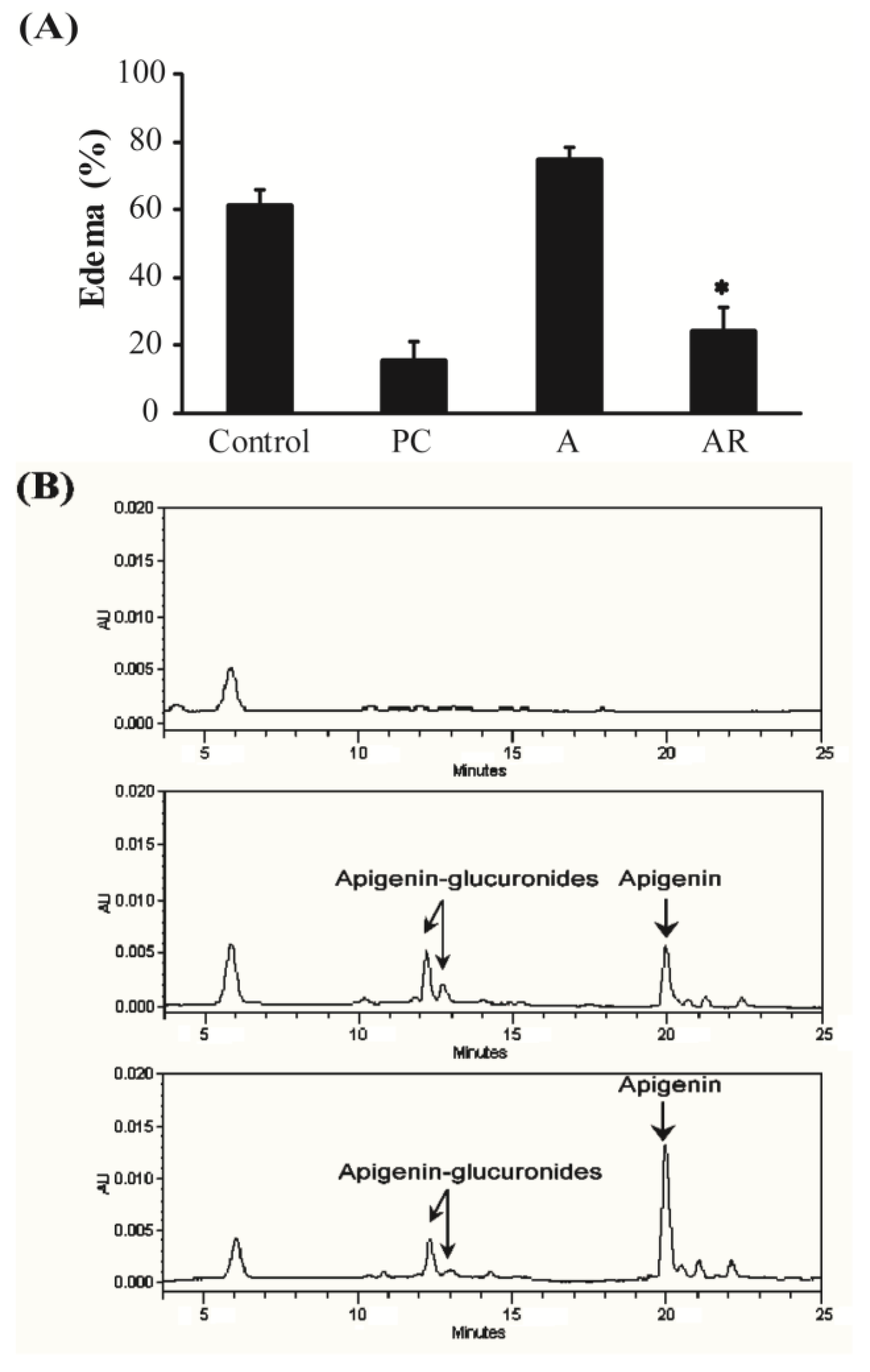
| Group | Concentration of Apigenin (ng/mL) |
|---|---|
| Apigenin | 452.996 ± 155.857 |
| Apigenin + Resveratrol | 1084.600 ± 243.508 * |
3.4. Kinetic Pattern of Action of Resveratrol on Formation of Glucuronide Conjugates of Apigenin by Recombinant UGT1A9
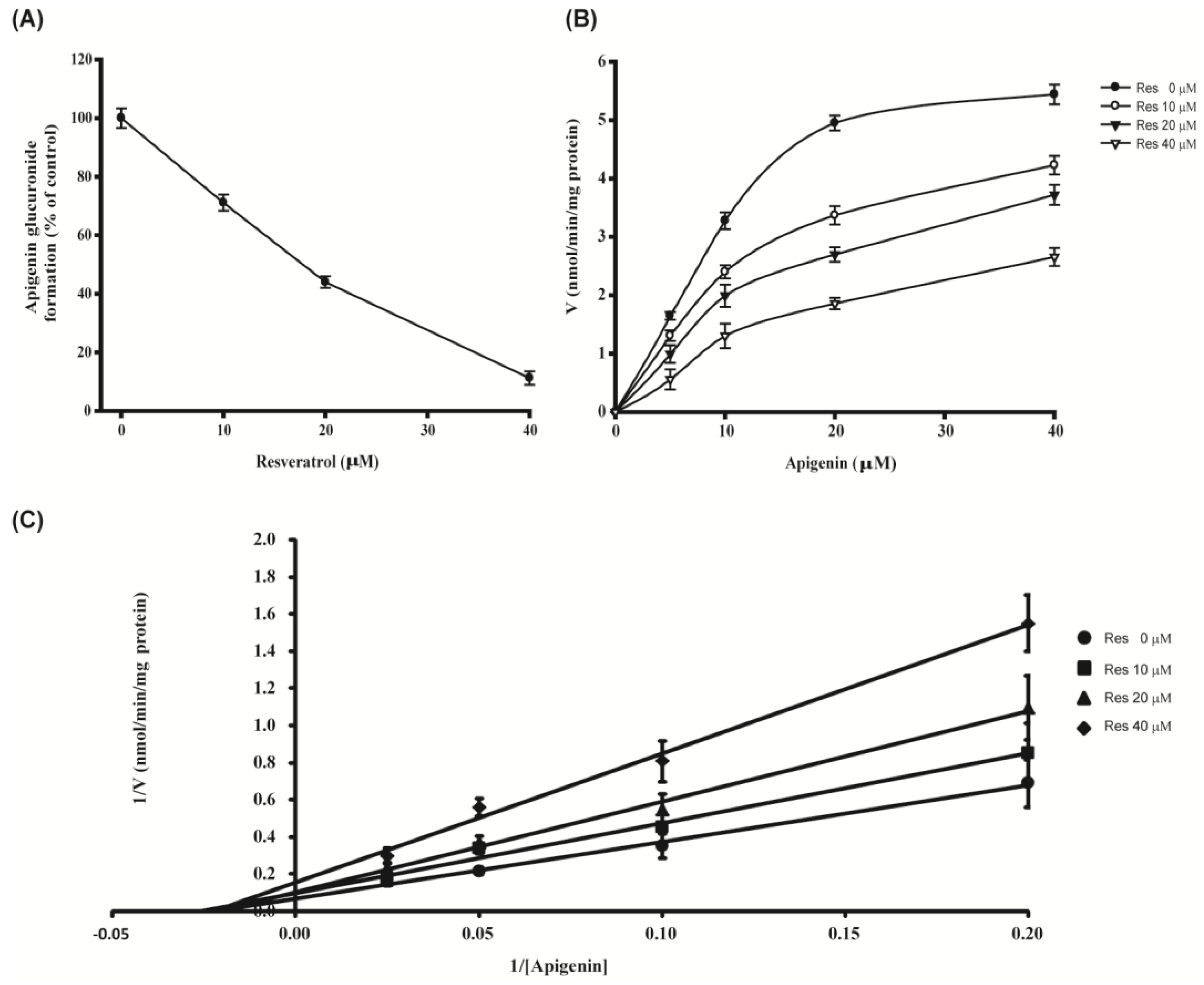
| UGT Source | Vmax (nmol/min/mg Protein) | Km (μM) | Ki (μM) |
|---|---|---|---|
| UGT1A9 a | 3.935 ± 0.65 | 0.478 ± 0.02 | 7.782 ± 0.84 |
4. Discussion
5. Conclusions
Author Contributions
Conflicts of Interest
References
- Nicholas, C.; Batra, S.; Vargo, M.A.; Voss, O.H.; Gavrilin, M.A.; Wewers, M.D.; Guttridge, D.C.; Grotewold, E.; Doseff, A.I. Apigenin blocks lipopolysaccharide-induced lethality in vivo and proinflammatory cytokines expression by inactivating NF-kappaB through the suppression of p65 phosphorylation. J. Immunol. 2007, 179, 7121–7127. [Google Scholar] [CrossRef] [PubMed]
- Nielsen, S.E.; Dragsted, L.O. Column-switching high-performance liquid chromatographic assay for determination of apigenin and acacetin in human urine with ultraviolet absorbance detection. J. Chromatogr. B Biomed. Sci. Appl. 1998, 713, 379–386. [Google Scholar] [CrossRef]
- Havsteen, B.H. The biochemistry and medical significance of the flavonoids. Pharmacol. Ther. 2002, 96, 67–202. [Google Scholar] [CrossRef]
- Hu, M. Commentary: Bioavailability of flavonoids and polyphenols: Call to arms. Mol. Pharm. 2007, 4, 803–806. [Google Scholar] [CrossRef] [PubMed]
- Bose, K.C. Pharmacopoeia India: Being a Collection of Vegetable, Mineral (and) Animal Drugs in Common Use in India; The Book Co.: Calcutta, India, 1932. [Google Scholar]
- Shoba, G.; Joy, D.; Joseph, T.; Maieed, M.; Raiendran, R.; Srinivas, P.S. Influence of piperine on the pharmacokinetics of curcumin in animals and human volunteers. Planta Med. 1998, 64, 353–356. [Google Scholar] [CrossRef] [PubMed]
- Lambert, J.D.; Hong, J.; Kim, D.H.; Mishin, V.M.; Yang, C.S. Piperine enhances the bioavailability of the tea polyphenol (-)-epigallocatechin-3-gallate in mice. J. Nutr. 2004, 134, 1948–1952. [Google Scholar] [PubMed]
- Williams, J.A.; Ring, B.J.; Cantrell, V.E.; Campanale, K.; Jones, D.R.; Hall, S.D.; Wrighton, S.A. Differential modulation of UGT1A1-catalyzed estradiol-3-glucuronidation by the addition of UGT1A1 substrates and other compounds to human liver microsomes. Drug Metab. Dispos. 2002, 30, 1266–1273. [Google Scholar] [CrossRef] [PubMed]
- Kale, A.; Gawande, S.; Kotwal, S.; Netke, S.; Roomi, W.; Ivanov, V.; Niedzwiecki, A.; Rath, M. Studies on the effects of oral administration of nutrient mixture, quercetin and red onions on the bioavailability of epigallocatechin gallate from green tea extract. Phytother. Res. 2010, 24, S48–S55. [Google Scholar] [CrossRef] [PubMed]
- Singh, R.; Wu, B.; Tang, L.; Hu, M. Uridine diphosphate glucuronosyltransferase isoform-dependent regiospecificity of glucuronidation of flavonoids. J. Agric. Food Chem. 2011, 59, 7452–7464. [Google Scholar] [CrossRef] [PubMed]
- Kowalski, J.; Samojedny, A.; Paul, M.; Pietsz, G.; Wilczok, T. Effect of apigenin, kaempferol and resveratrol on the expression of interleukin-1 beta and tumor necrosis factor-alpha genes in J774.2 macrophages. Phamacol. Rep. 2005, 57, 390–394. [Google Scholar]
- Pollard, S.E.; Kuhnle, G.G.; Vauzour, D.; Vafeiadou, K.; Tzounis, X.; Whiteman, M.; Rice-Evans, C.; Spencer, J.P. The reaction of flavonoid metabolites with peroxynitrite. Biochem. Biophys. Res. Commun. 2006, 350, 960–968. [Google Scholar] [CrossRef] [PubMed]
- Spencer, J.P.; Kuhnle, G.G.; Williams, R.J.; Rice-Evans, C. Intracellular metabolism and bioactivity of quercetin and its in vivo metabolites. Biochem. J. 2003, 372, 173–181. [Google Scholar] [CrossRef] [PubMed]
- Williamson, G.; Barron, D.; Shimoi, K.; Terao, J. In vitro biological properties of flavonoid conjugates found in vivo. Free Radical. Res. 2005, 39, 457–469. [Google Scholar] [CrossRef] [PubMed]
- Atal, C.K.; Dubey, R.K.; Singh, J. Biochemical basis of enhanced drug bioabailability by piperine: Evidence that piperine is a potent inhibitor of drug metabolism. J. Pharmacol. Exp. Ther. 1985, 232, 258–262. [Google Scholar] [PubMed]
- Singh, J.; Dubey, R.K.; Atal, C.K. Piperine-mediated inhibition of glucuronidation activity in isolated epithelial cells of the guinea-pig small intestine: Evidence that piperine lowers the endogeneous UDP-glucuronic acid content. J. Pharmacol. Exp. Ther. 1986, 236, 488–493. [Google Scholar] [PubMed]
- Fremont, L.; Belguendouz, L.; Delpal, S. Antioxidant activity of resveratrol and alcohol-free wine polyphenols related to LDL oxidation and polyunsaturated fatty acids. Life Sci. 1999, 64, 2511–2521. [Google Scholar] [CrossRef]
- Zou, J.G.; Huang, Y.Z.; Chen, Q.; Wei, E.H.; Hsieh, T.C.; Wu, J.M. Resveratrol inhibits copper ion-induced and azo compound-initiated oxidative modification of human low density lipoprotein. Biochem. Mol. Biol. Int. 1999, 47, 1089–1096. [Google Scholar] [CrossRef] [PubMed]
- Revel, A.; Raanani, H.; Younglai, E.; Xu, J.; Rogers, I.; Han, R.; Savouret, J.F.; Casper, R.F. Resveratrol, a natural aryl hydrocarbon receptor antagonist, protects lung from DNA damage and apoptosis caused by benz (a) pyrene. J. Appl. Toxicol. 2003, 23, 255–261. [Google Scholar] [CrossRef] [PubMed]
- Piver, B.; Berthou, F.; Dreano, Y.; Lucas, D. Inhibition of CYP3A, CYP1A and CYP2E1 activities by resveratrol and other non-volatile red wine components. Toxicol. Lett. 2001, 125, 83–91. [Google Scholar] [CrossRef]
- Brill, S.S.; Furimsky, A.M.; Ho, M.N.; Furniss, M.J.; Li, Y.; Green, A.G.; Bradford, W.W.; Green, C.E.; Kapetanovic, I.M.; Iyer, L.V. Glucuronidation of trans-resveratrol by human liver and intestinal microsomes and UGT isoforms. J. Pharm. Pharmacol. 2006, 58, 469–479. [Google Scholar] [CrossRef] [PubMed]
- Otake, Y.; Nolan, A.L.; Walle, U.K.; Walle, T. Quercetin and resveratrol potently reduce estrogen sulfotransferase activity in normal human mammary epithelial cells. J. Steroid Biochem. Mol. Biol. 2000, 73, 265–270. [Google Scholar] [CrossRef]
- Lawrence, T.; Willloughby, D.A.; Gilroy, D.W. Anti-inflammatory lipid mediators and insights into the resolution of inflammation. Nat. Rev. Immunol. 2002, 2, 787–795. [Google Scholar] [CrossRef] [PubMed]
- Rimal, B.; Greenberg, A.K.; Rom, W.N. Basic pathogenetic mechanisms in silicosis: Current understanding. Curr. Opin. Pulm. Med. 2005, 11, 169–173. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.H.; Soyoola, E.; Chanmugam, P.; Hart, S.; Sun, W.; Zhong, H.; Liou, S.; Simmons, D.; Hwang, D. Selective expression of mitogen-inducible cyclooxygenase in macrophages stimulated with lipopolysaccharide. J. Biol. Chem. 1992, 267, 25934–25938. [Google Scholar] [PubMed]
- Hua, Y.Q.; Su, S.L.; Duan, J.A.; Wang, Q.J.; Chen, L. Danggui-Shaoyao-San, a tranditional Chinese prescription, suppresses PGE2 alpha production in endometrial epithelial cells by inhibition COX-2 expression and activity. Phytomedicine 2008, 15, 1046–1052. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Wang, G.; Gurley, E.C.; Zhou, H. Flavonoid apigenin inhibits lipopolysaccharide-induced inflammatory response through multiple mechanisms in macrophages. PLoS ONE 2014, 9, e107072. [Google Scholar] [CrossRef] [PubMed]
- Gambero, A.; Becker, T.L.; Gurgueira, S.A.; Benvengo, Y.H.; Ribeiro, M.L.; de Mendonca, S.; Pedrazzoli, J., Jr. Acute inflammatory response induced by Helicobacter pylori in the rat air pouch. FEMS. Immunol. Med. Microbiol. 2003, 38, 193–198. [Google Scholar] [CrossRef]
- Mackenzie, P.I.; Gregory, P.A.; Lewinsky, R.H.; Yasmin, S.N.; Height, T.; Mckinnon, R.A.; Gardner-Stephen, D.A. Polymorphic variations in the expression of the chemical detoxifying UDP glucuronosyltransferases. Toxicol. Appl. Pharmacol. 2005, 207, 77–83. [Google Scholar] [CrossRef] [PubMed]
- Li, X.M.; Li, X.; Chen, S.Q.; Zeng, S. Construction of pcDNA3.1-UDPGT1A9 expression plasmid and establishment of its transgenic cell line. Chin. J. Pharmacol. Toxicol. 2004, 18, 115–120. [Google Scholar]
- Barbier, O.; Girard, H.; Inoue, Y.; Duez, H.; Villeneuve, L.; Kamiya, A.; Fruchart, J.C.; Guillemette, C.; Gonzalez, F.J.; Staels, B. Hepatic expression of the UGT1A9 gene is governed by hepatocyte nuclear factor 4α. Mol. Pharmacol. 2005, 67, 241–249. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lee, J.-A.; Ha, S.K.; Cho, E.; Choi, I. Resveratrol as a Bioenhancer to Improve Anti-Inflammatory Activities of Apigenin. Nutrients 2015, 7, 9650-9661. https://doi.org/10.3390/nu7115485
Lee J-A, Ha SK, Cho E, Choi I. Resveratrol as a Bioenhancer to Improve Anti-Inflammatory Activities of Apigenin. Nutrients. 2015; 7(11):9650-9661. https://doi.org/10.3390/nu7115485
Chicago/Turabian StyleLee, Jin-Ah, Sang Keun Ha, EunJung Cho, and Inwook Choi. 2015. "Resveratrol as a Bioenhancer to Improve Anti-Inflammatory Activities of Apigenin" Nutrients 7, no. 11: 9650-9661. https://doi.org/10.3390/nu7115485
APA StyleLee, J.-A., Ha, S. K., Cho, E., & Choi, I. (2015). Resveratrol as a Bioenhancer to Improve Anti-Inflammatory Activities of Apigenin. Nutrients, 7(11), 9650-9661. https://doi.org/10.3390/nu7115485





