Association between Dietary Patterns and Atopic Dermatitis in Relation to GSTM1 and GSTT1 Polymorphisms in Young Children
Abstract
1. Introduction
2. Methods
2.1. Participants and Study Design
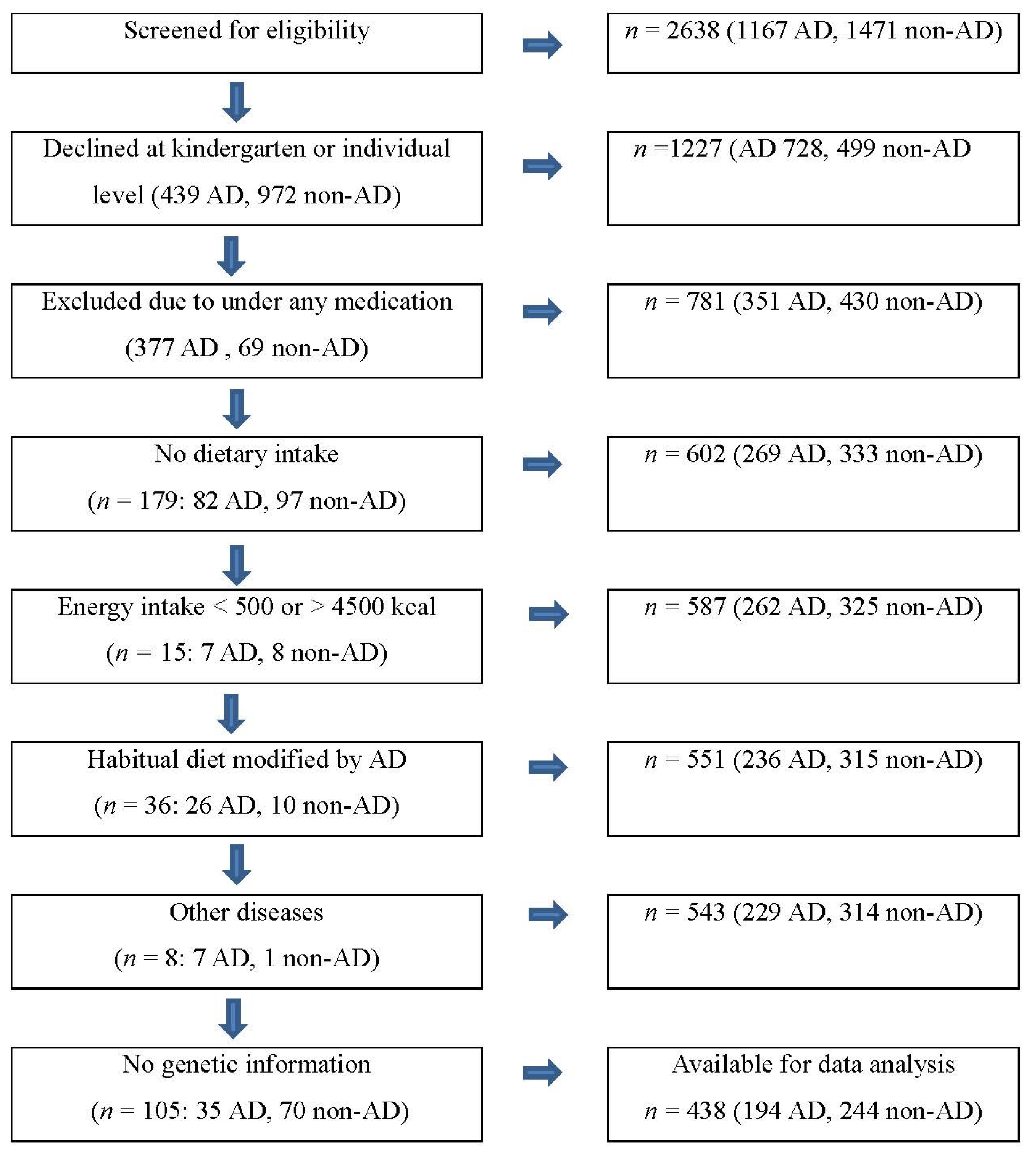
2.2. Dietary Assessment
| Food/Food Group | Food |
|---|---|
| Beans | Soybean curd (tofu)/curd residue, soybean (boiled with soy sauce), soymilk |
| Beef | Sliced beef with sauces (Galbi, Bulgogi), beef (loin, tender loin), beef soup/beef broiled down in soy |
| Bread | White and dark breads |
| Cereals | Breakfast cereals |
| Cheese | Cheese |
| Chicken | Chicken (fried), chicken (boiled, braised) |
| Chocolate | Chocolate |
| Eggs | Eggs |
| Fast food | Hamburger, pizza, French fries |
| Fats | Butter/margarine, mayonnaise |
| Fresh fish | White fish (pan fried, fried), white fish (grilled, broiled down in soy) blue fish (pan fried, fried), blue fish (grilled, broiled down in soy), squid/octopus, shrimps, clams/oysters |
| Fruit juice | Orange juice, tomato juice, other fruit juices |
| Fruits | Strawberries, apple, pear, mandarin/orange, tomato, banana, melon/muskmelon, watermelon, peaches/plum, grapes |
| Ice cream | Ice cream |
| Kimchi | Korean cabbage kimchi/seasoned cubed radish roots/young radish kimchi, other kinds of kimchi |
| Milk | Whole milk, flavored milk, low fat milk |
| Mulchi | Anchovy (stir-fried) |
| Noodles & Dumplings | Korean style noodles, spaghetti/bean sauce noodles, dumplings |
| Nuts | Nuts |
| Pork | Pork (loin, tender loin, shoulder), pork (belly) |
| Potatoes | Potatoes, sweet potatoes (not fried) |
| Processed fish | Canned tuna, fish paste |
| Processed meat | Ham/sausage |
| Ramyeon | Ramyeon |
| Rice | White rice, other grains |
| Rice cake | Rice cakes |
| Seaweeds | Dried laver, sea mustard |
| Snacks | Chips, crackers |
| Sweet bread | Sweet bread |
| Sweet drinks | Cocoa, soft drinks, sport drinks, traditional sweet drinks |
| Sweets | Candies, jam |
| Vegetables | Lettuce/cabbage (raw), lettuce/cabbage (cooked), radish, bean sprout/mungbean sprout, cucumber, spinach, perilla leaves, unripe hot pepper, onion, carrots, squash, mushrooms, roots of balloon flower/fernbrake |
| Yogurt | Yogurt, yogurt drinks |
2.3. Genotyping
2.4. Other Factors
2.5. Statistical Analysis
| Food/Food Groups | Traditional Healthy | Animal Foods | Sweets |
|---|---|---|---|
| Vegetables | 0.62 | 0.13 | −0.13 |
| Fruit | 0.58 | −0.01 | 0.18 |
| Seaweeds | 0.48 | 0.06 | −0.02 |
| Beans | 0.44 | 0.01 | 0.10 |
| Mulchi | 0.46 | −0.03 | −0.06 |
| Potatoes | 0.46 | 0.21 | −0.04 |
| Kimchi | 0.38 | −0.08 | −0.22 |
| Fresh fish | 0.37 | 0.21 | −0.10 |
| Ramyeon | −0.35 | 0.29 | −0.17 |
| Noodles and dumplings | −0.21 | 0.52 | 0.03 |
| Bread | 0.04 | 0.49 | 0.12 |
| Rice cake | 0.15 | 0.48 | −0.04 |
| Chicken | −0.07 | 0.47 | −0.02 |
| Fast food | −0.19 | 0.43 | 0.19 |
| Sweet bread | −0.03 | 0.40 | 0.06 |
| Beef | 0.15 | 0.37 | 0.04 |
| Sweets | −0.01 | 0.36 | 0.35 |
| Fats | 0.11 | 0.33 | −0.01 |
| Pork | 0.05 | 0.30 | 0.05 |
| Processed meat | −0.21 | 0.28 | 0.09 |
| Processed fish | 0.09 | 0.25 | 0.01 |
| Milk | 0.01 | −0.34 | 0.19 |
| Fruit juice | 0.24 | 0.01 | 0.50 |
| Sweet drinks | −0.23 | 0.17 | 0.46 |
| Chocolate | −0.14 | 0.14 | 0.46 |
| Snacks | −0.28 | 0.14 | 0.41 |
| Ice cream | 0.02 | 0.06 | 0.38 |
| Cheese | 0.32 | −0.13 | 0.33 |
| Rice | −0.23 | −0.01 | −0.60 |
| Yogurt | 0.20 | −0.23 | 0.29 |
| Eggs | 0.16 | 0.15 | −0.27 |
| Nuts | 0.27 | −0.05 | 0.27 |
| Cereals | −0.04 | 0.02 | 0.16 |
2.6. Ethics Statement
3. Results
| Nutrient | Traditional Healthy | Animal Foods | Sweets | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Low (Q1–Q3) | High (Q4) | p | Low (Q1–Q3) | High (Q4) | p | Low (Q1–Q3) | High (Q4) | p | |||||||
| Mean | SE | Mean | SE | Mean | SE | Mean | SE | Mean | SE | Mean | SE | ||||
| Energy (kJ) | 1556.3 | 37.1 | 1511.9 | 64.5 | 0.551 | 1583.0 | 37.0 | 1431.4 | 64.2 | 0.041 | 1497.5 | 36.8 | 1689.3 | 64.0 | 0.010 |
| Animal protein (g) | 32.0 | 0.6 | 39.3 | 1.1 | <.0.001 | 32.8 | 0.6 | 36.8 | 1.1 | 0.003 | 33.9 | 0.7 | 33.4 | 1.1 | 0.706 |
| Plant protein (g) | 22.6 | 0.3 | 23.8 | 0.5 | 0.048 | 23.1 | 0.3 | 22.5 | 0.5 | 0.363 | 23.7 | 0.3 | 20.5 | 0.5 | <0.001 |
| Animal fat (g) | 28.0 | 0.6 | 32.6 | 1.1 | <0.001 | 28.4 | 0.6 | 31.5 | 1.1 | 0.011 | 28.7 | 0.6 | 30.5 | 1.1 | 0.140 |
| Plant fat (g) | 18.5 | 0.4 | 20.8 | 0.7 | 0.004 | 18.0 | 0.4 | 22.4 | 0.7 | <0.001 | 17.9 | 0.4 | 22.8 | 0.7 | <0.001 |
| Vitamin A (μg, RE) | 413.6 | 10.3 | 652.4 | 17.8 | <0.001 | 481.1 | 11.7 | 448.7 | 20.4 | 0.170 | 459.9 | 11.7 | 512.8 | 20.4 | 0.025 |
| Retinol (μg) | 229.5 | 5.7 | 258.5 | 9.9 | 0.012 | 240.8 | 5.7 | 224.4 | 10.0 | 0.157 | 216.3 | 5.4 | 298.2 | 9.5 | <0.001 |
| β-carotene (μg) | 1247.3 | 55.0 | 2561.9 | 95.6 | <0.001 | 1598.4 | 63.4 | 1502.3 | 110.4 | 0.452 | 1560.3 | 63.5 | 1617.3 | 110.7 | 0.656 |
| Vitamin C (mg) | 65.3 | 2.3 | 97.3 | 4.0 | <0.001 | 73.8 | 2.4 | 71.6 | 4.2 | 0.659 | 65.6 | 2.3 | 96.4 | 4.0 | <0.001 |
| Folate (μg) | 172.1 | 2.8 | 231.4 | 4.9 | <0.001 | 188.7 | 3.1 | 181.3 | 5.5 | 0.242 | 186.6 | 3.2 | 187.8 | 5.5 | 0.855 |
| Vitamin E (mg, α-TE) | 8.5 | 0.2 | 12.3 | 0.4 | <0.001 | 8.7 | 0.2 | 11.6 | 0.4 | <0.001 | 9.5 | 0.2 | 9.2 | 0.4 | 0.513 |
| Saturated fatty acids (g) | 12.1 | 0.3 | 13.1 | 0.6 | 0.131 | 12.5 | 0.3 | 11.9 | 0.6 | 0.326 | 11.9 | 0.3 | 13.8 | 0.6 | 0.004 |
| Monounsaturated fatty acids (g) | 8.0 | 0.2 | 9.7 | 0.4 | <0.001 | 8.1 | 0.2 | 9.4 | 0.4 | 0.003 | 8.3 | 0.2 | 8.8 | 0.4 | 0.224 |
| Polyunsaturated fatty acids (g) | 3.9 | 0.1 | 5.5 | 0.2 | <0.001 | 4.0 | 0.1 | 5.2 | 0.2 | <0.001 | 4.3 | 0.1 | 4.2 | 0.2 | 0.749 |
| AD (n = 194) | Non-AD (n = 244) | p * | |||
|---|---|---|---|---|---|
| Mean | SD | Mean | SD | ||
| Age (year) | 5.4 | 1.2 | 5.3 | 1.3 | 0.270 |
| BMI (kg/m2) | 18.3 | 3.5 | 17.9 | 3.4 | 0.260 |
| Birth weight (kg) | 3.3 | 0.7 | 3.3 | 0.8 | 0.876 |
| Daily nutrient intake | |||||
| Energy (kJ) | 6305.2 | 2622.3 | 6592.8 | 2957.5 | 0.289 |
| Animal protein (g) | 31.8 | 18.6 | 35.4 | 23.3 | 0.073 |
| Plant protein (g) | 22.4 | 9.4 | 23.3 | 10.2 | 0.324 |
| Animal fat (g) | 27.5 | 16.8 | 30.5 | 21.0 | 0.095 |
| Plant fat (g) | 18.2 | 10.5 | 19.8 | 11.9 | 0.130 |
| Vitamin A (μg RE) | 442.5 | 274.3 | 497.3 | 321.0 | 0.055 |
| Retinol (μg) | 231 | 158.8 | 241.2 | 161.8 | 0.509 |
| β-carotene (μg) | 1442.2 | 1244.3 | 1679.6 | 1397.8 | 0.065 |
| Vitamin C (mg) | 69.3 | 57.1 | 76.4 | 61.8 | 0.217 |
| Folate (μg) | 173.8 | 88.0 | 197.3 | 106.5 | 0.012 |
| Vitamin E (mg α-TE) | 8.5 | 5.3 | 10.2 | 7.4 | 0.005 |
| Saturated fatty acids (g) | 11.9 | 7.8 | 12.7 | 9.1 | 0.357 |
| Monounsaturated fatty acids (g) | 7.8 | 4.6 | 8.8 | 6.8 | 0.072 |
| Polyunsaturated fatty acids (g) | 3.9 | 2.4 | 4.6 | 3.5 | 0.009 |
| Total IgE (U/mL) † | 325.8 | 617.2 | 187.7 | 307.9 | 0.006 |
| n | (%) | n | (%) | ||
| Gender (boys) | 103 | (53.4) | 123 | (50.4) | 0.539 |
| AD status ‡ | |||||
| Symptoms (n = 192) | 88 | 45.8 | |||
| Itchy rash in the last year (n = 180) | 89 | 49.5 | |||
| Diagnosis by physician (n = 194) | 156 | 80.4 | |||
| Treatment (n = 190) | 43 | 22.6 | |||
| Current exposure to smoking at home | 31 | (17.0) | 35 | (15.4) | 0.645 |
| Supplementary multivitamin use | 80 | (46.0) | 110 | (48.9) | 0.564 |
| Household income (104 Won/mo) § | |||||
| <200 | 28 | (15.0) | 30 | (12.7) | 0.733 |
| 200–399 | 90 | (48.1) | 113 | (47.7) | |
| ≥400 | 69 | (36.9) | 94 | (39.7) | |
| Maternal educational level (≥16 year) | 86 | (44.8) | 99 | (40.7) | 0.396 |
| Allergic history of mother | |||||
| Asthma | 9 | (4.6) | 13 | (5.3) | 0.743 |
| Rhinitis | 46 | (23.7) | 57 | (23.4) | 0.932 |
| AD | 26 | (13.4) | 14 | (5.7) | 0.006 |
| Allergic history of father | |||||
| Asthma | 9 | (4.6) | 6 | (2.5) | 0.213 |
| Rhinitis | 47 | (24.2) | 35 | (14.3) | 0.008 |
| AD | 22 | (11.3) | 17 | (7.0) | 0.110 |
| AD | Non-AD | p * | |||
|---|---|---|---|---|---|
| n | % | n | % | ||
| Genotypes | |||||
| GSTM1 | 0.466 | ||||
| Null | 118 | 60.8 | 140 | 57.4 | |
| Present | 76 | 39.2 | 104 | 42.6 | |
| GSTT1 | 0.686 | ||||
| Null | 98 | 50.5 | 128 | 52.5 | |
| Present | 96 | 49.5 | 116 | 47.5 | |
| GSTM1/GSTT1 | 0.638 | ||||
| Double null | 57 | 29.4 | 75 | 30.7 | |
| Either null | 102 | 52.6 | 118 | 48.4 | |
| Double present | 35 | 18.0 | 51 | 20.9 | |
| Dietary patterns | |||||
| Traditional healthy | 0.466 | ||||
| Low (Q1-Q3) | 149 | 45.3 | 180 | 54.7 | |
| High (Q4) | 45 | 41.3 | 64 | 58.7 | |
| Animal foods | 0.873 | ||||
| Low (Q1-Q3) | 145 | 44.1 | 184 | 55.9 | |
| High (Q4) | 49 | 45.0 | 60 | 55.0 | |
| Sweets | 0.776 | ||||
| Low (Q1-Q3) | 147 | 44.7 | 182 | 55.3 | |
| High (Q4) | 47 | 43.1 | 62 | 56.9 | |
| AD vs. Non AD | ||||||
|---|---|---|---|---|---|---|
| aOR | 95% CI | p for Chi-Square † | aOR | 95% CI | p for Interaction ‡ | |
| Tradition healthy | ||||||
| GSTM1 | ||||||
| Null | 1.24 | (0.68, 2.26) | 0.495 | 1 | ||
| Present | 0.31 | (0.13, 0.75) | 0.009 | 0.71 | (0.54, 0.92) | 0.011 |
| GSTT1 | ||||||
| Null | 0.67 | (0.34, 1.30) | 0.239 | 1 | ||
| Present | 0.90 | (0.45, 1.81) | 0.788 | 1.08 | (0.85, 1.37) | 0.542 |
| GSTM1/GSTT1 | ||||||
| Double null | 0.93 | (0.41, 2.13) | 0.881 | 1 | ||
| Either null | 0.97 | (0.49, 1.90) | 0.931 | 1.27 | (0.90.1.37) | 0.176 |
| Double present | 0.24 | (0.06, 0.93) | 0.039 | 0.63 | (0.39, 1.03) | 0.065 |
| Animal foods | ||||||
| GSTM1 | ||||||
| Null | 1.07 | (0.56, 2.08) | 0.843 | 1 | ||
| Present | 1.25 | (0.62, 2.52) | 0.550 | 1.04 | (0.82, 1.32) | 0.760 |
| GSTT1 | ||||||
| Null | 1.07 | (0.55, 2.07) | 0.861 | 1 | ||
| Present | 1.18 | (0.60, 2.32) | 0.649 | 1.03 | (0.81, 1.30) | 0.836 |
| GSTM1/GSTT1 | ||||||
| Double null | 1.18 | (0.47, 2.98) | 0.741 | 1 | ||
| Either null | 0.95 | (0.49, 1.84) | 0.891 | 0.88 | (0.64, 1.21) | 0.423 |
| Double present | 1.68 | (0.60, 4.68) | 0.329 | 1.17 | (0.79. 1.72) | 0.441 |
| Sweets | ||||||
| GSTM1 | ||||||
| Null | 0.68 | (0.36, 1.26) | 0.220 | 1 | ||
| Present | 1.53 | (0.73, 3.20) | 0.266 | 1.23 | (0.96, 1.56) | 0.098 |
| GSTT1 | ||||||
| Null | 0.85 | (0.42, 1.73) | 0.665 | |||
| Present | 1.02 | (0.54, 1.93) | 0.963 | 1.05 | (0.82,1.33) | 0.712 |
| GSTM1/GSTT1 | ||||||
| Double null | 0.59 | (0.24, 1.44) | 0.247 | 1 | ||
| Either null | 1.04 | (0.52, 2.10) | 0.920 | 1.02 | (0.74, 1.41) | 0.897 |
| Double present | 1.62 | (0.61, 4.33) | 0.343 | 1.27 | (0.87, 1.86) | 0.212 |
4. Discussion
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Eichenfield, L.F.; Tom, W.L.; Berger, T.G.; Krol, A.; Paller, A.S.; Schwarzenberger, K.; Bergman, J.N.; Chamlin, S.L.; Cohen, D.E.; Cooper, K.D. Guidelines of care for the management of atopic dermatitis: Section 2. management and treatment of atopic dermatitis with topical therapies. J. Am. Acad. Dermatol. 2014, 71, 116–132. [Google Scholar] [PubMed]
- Lee, J.Y.; Seo, J.H.; Kwon, J.W.; Yu, J.; Kim, B.J.; Lee, S.Y.; Kim, H.B.; Kim, W.K.; Kim, K.W.; Shin, Y.J.; et al. Exposure to gene-environment interactions before 1 year of age may favor the development of atopic dermatitis. Int. Arch. Allergy Immunol. 2012, 157, 363–371. [Google Scholar] [CrossRef] [PubMed]
- Williams, H.C. Atopic dermatitis. N. Engl. J. Med. 2005, 352, 2314–2324. [Google Scholar] [CrossRef] [PubMed]
- Bieber, T. Atopic dermatitis 2.0: From the clinical phenotype to the molecular taxonomy and stratified medicine. Allergy 2012, 67, 1475–1482. [Google Scholar] [CrossRef] [PubMed]
- Larsen, F.S. Atopic dermatitis: A genetic-epidemiologic study in a population-based twin sample. J. Am. Acad. Dermatol. 1993, 28, 719–723. [Google Scholar] [CrossRef]
- Omata, N.; Tsukahara, H.; Ito, S.; Ohshima, Y.; Yasutomi, M.; Yamada, A.; Jiang, M.; Hiraoka, M.; Nambu, M.; Deguchi, Y. Increased oxidative stress in childhood atopic dermatitis. Life Sci. 2001, 69, 223–228. [Google Scholar] [CrossRef]
- Tsukahara, H.; Shibata, R.; Ohshima, Y.; Todoroki, Y.; Sato, S.; Ohta, N.; Hiraoka, M.; Yoshida, A.; Nishima, S.; Mayumi, M. Oxidative stress and altered antioxidant defenses in children with acute exacerbation of atopic dermatitis. Life Sci. 2003, 72, 2509–2516. [Google Scholar] [CrossRef]
- Kapp, A.; Zeck-Kapp, G.; Czech, W.; Schöpf, E. The chemokine rantes is more than a chemoattractant: characterization of its effect on human eosinophil oxidative metabolism and morphology in comparison with IL-5 and GM-CSF. J. Investig. Dermatol. 1994, 102, 906–914. [Google Scholar] [CrossRef] [PubMed]
- Portugal, M.; Barak, V.; Ginsburg, I.; Kohen, R. Interplay among oxidants, antioxidants, and cytokines in skin disorders: Present status and future considerations. Biomed. Pharmacother. 2007, 61, 412–422. [Google Scholar] [CrossRef] [PubMed]
- Oh, S.; Chung, J.; Kim, M.; Kwon, S.; Cho, B. Antioxidant nutrient intakes and corresponding biomarkers associated with the risk of atopic dermatitis in young children. Eur. J. Clin. Nutr. 2010, 64, 245–252. [Google Scholar] [CrossRef] [PubMed]
- Saadeh, D.; Salameh, P.; Baldi, I.; Raherison, C. Diet and allergic diseases among population aged 0 to 18 years: myth or reality? Nutrients 2013, 5, 3399–3423. [Google Scholar] [CrossRef] [PubMed]
- Nurmatov, U.; Devereux, G.; Sheikh, A. Nutrients and foods for the primary prevention of asthma and allergy: Systematic review and meta-analysis. J. Allergy Clin. Immunol. 2011, 127, 724–733. [Google Scholar] [CrossRef] [PubMed]
- Howard, A.L.; Robinson, M.; Smith, G.J.; Ambrosini, G.L.; Piek, J.P.; Oddy, W.H. ADHD is associated with a “western” dietary pattern in adolescents. J. Atten. Disord. 2011, 15, 403–411. [Google Scholar] [CrossRef] [PubMed]
- Millichap, J.G.; Yee, M.M. The diet factor in attention-deficit/hyperactivity disorder. Pediatrics 2012, 129, 330–337. [Google Scholar] [CrossRef] [PubMed]
- Northstone, K.; Joinson, C.; Emmett, P.; Ness, A.; Paus, T. Are dietary patterns in childhood associated with IQ at 8 years of age? A population-based cohort study. J. Epidemiol. Community Health 2012, 66, 624–628. [Google Scholar] [CrossRef] [PubMed]
- Smithers, L.G.; Golley, R.K.; Mittinty, M.N.; Brazionis, L.; Northstone, K.; Emmett, P.; Lynch, J.W. Dietary patterns at 6, 15 and 24 months of age are associated with IQ at 8 years of age. Eur. J. Epidemiol. 2012, 27, 525–535. [Google Scholar] [CrossRef] [PubMed]
- Castaldi, P.J.; Cho, M.H.; Cohn, M.; Langerman, F.; Moran, S.; Tarragona, N.; Moukhachen, H.; Venugopal, R.; Hasimja, D.; Kao, E.; et al. The COPD genetic association compendium: A comprehensive online database of COPD genetic associations. Hum. Mol. Genet. 2010, 19, 526–534. [Google Scholar] [CrossRef] [PubMed]
- Karban, A.; Krivoy, N.; Elkin, H.; Adler, L.; Chowers, Y.; Eliakim, R.; Efrati, E. Non-Jewish Israeli IBD patients have significantly higher glutathione S-transferase GSTT1-Null frequency. Dig. Dis. Sci. 2011, 56, 2081–2087. [Google Scholar] [CrossRef] [PubMed]
- Thakur, H.; Gupta, L.; Sobti, R.C.; Janmeja, A.K.; Seth, A.; Singh, S.K. Association of GSTM1T1 GENES with COPD and prostate cancer in North Indian Population. Mol. Biol. Rep. 2011, 38, 1733–1739. [Google Scholar] [CrossRef] [PubMed]
- Oniki, K.; Hori, M.; Saruwatari, J.; Morita, K.; Kajiwara, A.; Sakata, M.; Mihara, S.; Ogata, Y.; Nakagawa, K. Interactive effects of smoking and glutathione S-transferase polymorphisms on the development of non-alcoholic fatty liver Disease. Toxicol. Lett. 2013, 220, 143–149. [Google Scholar] [CrossRef] [PubMed]
- Vavilin, V.; Safronova, O.; Lyapunova, A.; Lyakhovich, V.; Kaznacheeva, L.; Manankin, N.; Molokova, A. Interaction of GSTM1, GSTT1, and GSTP1 genotypes in determination of predisposition to atopic dermatitis. Bull. Exp. Biol. Med. 2003, 136, 388–391. [Google Scholar] [CrossRef] [PubMed]
- Cho, H.; Uhm, Y.; Kim, H.; Ban, J.; Chung, J.; Yim, S.; Choi, B.; Lee, M. Glutathione S-transferase M1 (GSTM1) polymorphism is associated with atopic dermatitis susceptibility in a Korean population. Int. J. Immunogenet. 2011, 38, 145–150. [Google Scholar] [CrossRef] [PubMed]
- Chung, J.; Oh, S.; Shin, Y. Association of glutathione-S-transferase polymorphisms with atopic dermatitis risk in preschool age children. Clin. Chem. Lab. Med. 2009, 47, 1475–1481. [Google Scholar] [CrossRef] [PubMed]
- Shin, K.O.; Oh, S.; Park, H.S. Empirically derived major dietary patterns and their associations with overweight in Korean Preschool Children. Br. J. Nutr. 2007, 98, 416–421. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Physical Status: The Use of and Interpretation of Anthropometry; Report of a WHO Expert Committee; World Health Organization: Geneva, Switzerland, 1995. [Google Scholar]
- Cody, R.; Smith, J. Applied Statistics and the SAS Programming Language, 5th ed.; Prentice-Hall: Upper Saddle River, NJ, USA, 2005. [Google Scholar]
- Bhuvaneswari, V.; Velmurugan, B.; Nagini, S. Induction of glutathione-dependent hepatic biotransformation enzymes by lycopene in the hamster cheek pouch carcinogenesis model. J. Biochem. Mol. Biol. Biophys. 2002, 6, 257–260. [Google Scholar] [PubMed]
- Munday, R.; Munday, J.S.; Munday, C.M. Comparative effects of mono-, di-, tri-, and tetrasulfides derived from plants of the Allium Family: Redox cycling in vitro and hemolytic activity and phase 2 enzyme induction in vivo. Free Radic. Biol. Med. 2003, 34, 1200–1211. [Google Scholar] [CrossRef]
- Kuo, W.; Chou, F.; Young, S.; Chang, Y.; Wang, C. Geniposide activates GSH S-transferase by the induction of GST M1 and GST M2 Subunits involving the transcription and phosphorylation of MEK-1 Signaling in rat hepatocytes. Toxicol. Appl. Pharmacol. 2005, 208, 155–162. [Google Scholar] [CrossRef] [PubMed]
- Yuan, L.; Zhang, L.; Ma, W.; Zhou, X.; Ji, J.; Li, N.; Xiao, R. Glutathione S-transferase M1 and T1 gene polymorphisms with consumption of high fruit-juice and vegetable diet affect antioxidant capacity in healthy adults. Nutrition 2013, 29, 965–971. [Google Scholar] [CrossRef] [PubMed]
- Joseph, M.A.; Moysich, K.B.; Freudenheim, J.L.; Shields, P.G.; Bowman, E.D.; Zhang, Y.; Marshall, J.R.; Ambrosone, C.B. Cruciferous vegetables, genetic polymorphisms in glutathione S-transferases M1 and T1, and prostate cancer risk. Nutr. Cancer 2004, 50, 206–213. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.I.; Giovannucci, E.L.; Hunter, D.; Neuberg, D.; Su, L.; Christiani, D.C. Dietary intake of cruciferous vegetables, glutathione S-transferase (GST) polymorphisms and lung cancer risk in a Caucasian population. Cancer Causes Control 2004, 15, 977–985. [Google Scholar] [CrossRef] [PubMed]
- Safronova, O.; Vavilin, V.; Lyapunova, A.; Makarova, S.; Lyakhovich, V.; Kaznacheeva, L.; Manankin, N.; Batychko, O.; Gavalov, S. Relationship between glutathione S-transferase P1 polymorphism and bronchial asthma and atopic dermatitis. Bull. Exp. Biol. Med. 2003, 136, 73–75. [Google Scholar] [CrossRef] [PubMed]
- Wang, I.; Guo, Y.L.; Lin, T.; Chen, P.; Wu, Y. GSTM1, GSTP1, prenatal smoke exposure, and atopic dermatitis. Ann. Allergy Asthma Immunol. 2010, 105, 124–129. [Google Scholar] [CrossRef] [PubMed]
- Miyake, Y.; Sasaki, S.; Tanaka, K.; Hirota, Y. Consumption of vegetables, fruit, and antioxidants during pregnancy and wheeze and eczema in infants. Allergy 2010, 65, 758–765. [Google Scholar] [CrossRef] [PubMed]
- Okuda, M.; Bando, N.; Terao, J.; Sasaki, S.; Sugiyama, S.; Kunitsugu, I.; Hobara, T. Association of serum carotenoids and tocopherols with atopic diseases in japanese children and adolescents. Pediatr. Allergy Immunol. 2010, 21, e705–e710. [Google Scholar] [CrossRef] [PubMed]
- Nwaru, B.I.; Erkkola, M.; Lumia, M.; Kronberg-Kippilä, C.; Ahonen, S.; Kaila, M.; Ilonen, J.; Simell, O.; Knip, M.; Veijola, R. Maternal intake of fatty acids during pregnancy and allergies in the offspring. Br. J. Nutr. 2012, 108, 720–732. [Google Scholar] [CrossRef] [PubMed]
- Rosenlund, H.; Magnusson, J.; Kull, I.; Håkansson, N.; Wolk, A.; Pershagen, G.; Wickman, M.; Bergström, A. Antioxidant intake and allergic disease in children. Clin. Exp. Allergy 2012, 42, 1491–1500. [Google Scholar] [CrossRef] [PubMed]
- Fung, T.T.; Hu, F.B.; Holmes, M.D.; Rosner, B.A.; Hunter, D.J.; Colditz, G.A.; Willett, W.C. Dietary patterns and the risk of postmenopausal breast cancer. Int. J. Cancer 2005, 116, 116–121. [Google Scholar] [CrossRef] [PubMed]
- Chatzi, L.; Kogevinas, M. Prenatal and childhood mediterranean diet and the development of asthma and allergies in children. Public Health Nutr. 2009, 12, 1629–1634. [Google Scholar] [CrossRef] [PubMed]
- Chatzi, L.; Torrent, M.; Romieu, I.; Garcia-Esteban, R.; Ferrer, C.; Vioque, J.; Kogevinas, M.; Sunyer, J. Mediterranean diet in pregnancy is protective for wheeze and atopy in childhood. Thorax 2008, 63, 507–513. [Google Scholar] [CrossRef] [PubMed]
- Suarez-Varela, M.M.; Alvarez, L.G.; Kogan, M.D.; Ferreira, J.C.; Martinez Gimeno, A.; Aguinaga Ontoso, I.; Gonzalez Diaz, C.; Arnedo Pena, A.; Dominguez Aurrecoechea, B.; Busquets Monge, R.M.; et al. Diet and prevalence of atopic eczema in 6 to 7-year-old schoolchildren in Spain: ISAAC Phase III. J. Investig. Allergol. Clin. Immunol. 2010, 20, 469–475. [Google Scholar] [PubMed]
- Lee, S. Prevalence of childhood asthma in Korea: International study of asthma and allergies in childhood. Allergy Asthma Immunol. Res. 2010, 2, 61–64. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.H.; Jeong, K.S.; Ha, E.; Park, H.; Ha, M.; Hong, Y.; Lee, S.; Lee, K.Y.; Jeong, J.; Kim, Y. Association between prenatal exposure to cadmium and atopic dermatitis in infancy. J. Korean Med. Sci. 2013, 28, 516–521. [Google Scholar] [CrossRef] [PubMed]
- Alvarez, A.E.; de Lima Marson, F.A.; Bertuzzo, C.S.; Arns, C.W.; Ribeiro, J.D. Epidemiological and genetic characteristics associated with the severity of acute viral bronchiolitis by respiratory syncytial virus. J. Pediatr. 2013, 89, 531–543. [Google Scholar] [CrossRef] [PubMed]
- Jung, Y.; Seo, J.; Kim, H.Y.; Kwon, J.; Kim, B.; Kim, H.; Lee, S.; Jang, G.C.; Song, D.J.; Kim, W.K. The relationship between asthma and bronchiolitis is modified by TLR4, CD14, and IL-13 polymorphisms. Pediatr. Pulmonol. 2015, 50, 8–16. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chung, J.; Kwon, S.-O.; Ahn, H.; Hwang, H.; Hong, S.-J.; Oh, S.-Y. Association between Dietary Patterns and Atopic Dermatitis in Relation to GSTM1 and GSTT1 Polymorphisms in Young Children. Nutrients 2015, 7, 9440-9452. https://doi.org/10.3390/nu7115473
Chung J, Kwon S-O, Ahn H, Hwang H, Hong S-J, Oh S-Y. Association between Dietary Patterns and Atopic Dermatitis in Relation to GSTM1 and GSTT1 Polymorphisms in Young Children. Nutrients. 2015; 7(11):9440-9452. https://doi.org/10.3390/nu7115473
Chicago/Turabian StyleChung, Jayong, Sung-Ok Kwon, Hyogin Ahn, Hyojung Hwang, Soo-Jong Hong, and Se-Young Oh. 2015. "Association between Dietary Patterns and Atopic Dermatitis in Relation to GSTM1 and GSTT1 Polymorphisms in Young Children" Nutrients 7, no. 11: 9440-9452. https://doi.org/10.3390/nu7115473
APA StyleChung, J., Kwon, S.-O., Ahn, H., Hwang, H., Hong, S.-J., & Oh, S.-Y. (2015). Association between Dietary Patterns and Atopic Dermatitis in Relation to GSTM1 and GSTT1 Polymorphisms in Young Children. Nutrients, 7(11), 9440-9452. https://doi.org/10.3390/nu7115473




