Dietary Patterns and Maternal Anthropometry in HIV-Infected, Pregnant Malawian Women
Abstract
:1. Introduction
2. Experimental Section
Statistical Analysis
3. Results
| Characteristic | N = 577 |
|---|---|
| Age (year) [mean ± SD] 1 | 25.9 ± 4.9 |
| Education | |
| No school (%) | 11.3 |
| Primary (%) | 52.8 |
| Secondary or higher (%) | 35.9 |
| Occupation Status | |
| Unemployed (%) | 81.3 |
| Employed (%) | 18.7 |
| Experienced famine season | |
| None (%) | 39.7 |
| Some (%) | 14.6 |
| All (%) | 45.7 |
| Parity (live births) [mean ± SD] | 1.7 ± 1.3 |
| Gestational age (weeks) [mean ± SD] | 25.2 ± 5.4 |
| CD4 count (cells/uL) [range (IQR) 2] | 442 (325–601) |
| Hemoglobin (g/dL) [mean ± SD] | 10.8 ± 1.2 |
| Daily energy total intake (kcal) [mean ± SD] | 1378 ± 821 |
| Mid-upper arm circumference (cm) [mean ± SD] | 26.4 ± 2.6 |
| Arm muscle area (cm2) [mean ± SD] | 36.6 ± 6.4 |
| Arm fat area (cm2) [mean ± SD] | 19.5 ± 7.8 |
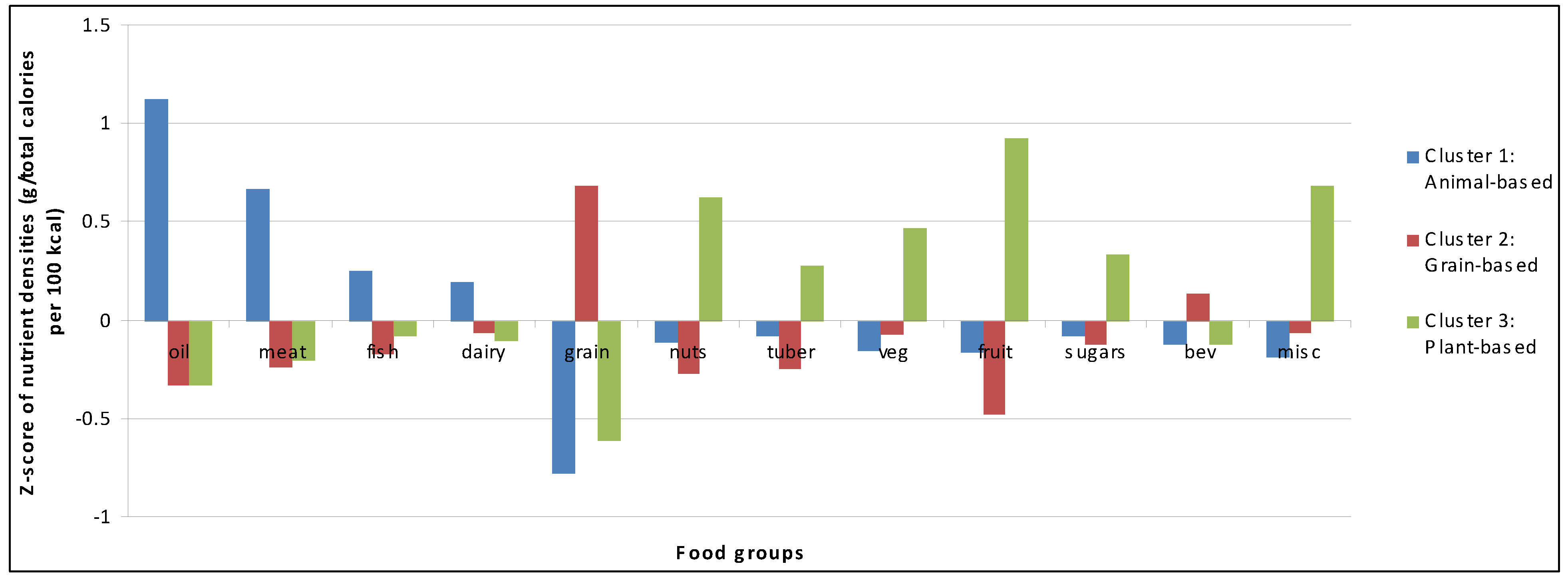
| Indicators | Animal-Based 1 (n = 160) | Grain-based 2,3 (n = 254) | Plant-based 4 (n = 163) | P value for Pairwise Cluster Comparisons 5 | ||
|---|---|---|---|---|---|---|
| Animal vs. Grain | Animal vs. Plant | Grain vs. Plant | ||||
| Characteristics 6 | ||||||
| Age (year) (mean ± SD) | 25.0 ± 4.4 | 26.2 ± 5.1 | 26.2 ± 4.9 | - | - | - |
| No school (n = 65) [n(%)] | 11 (6.9) | 37 (14.5) | 17 (10.4) | - | - | - |
| Lowest wealth index (n = 108) [n(%)] | 17 (15.7) | 63 (58.3) | 28 (25.9) | 0.001 | - | - |
| Parity > 2 (n = 133) [n(%)] | 23(14.3) | 67 (26.4) | 43 (26.3) | 0.014 | - | - |
| Any famine season exposure (n = 348) [n(%)] | 83 (51.9) | 180 (70.9) | 85 (51.2) | <0.001 | - | <0.001 |
| Nutrition status | ||||||
| Daily energy total intake (kcal) | 1776.8 ± 859.5 | 1083.5 ± 672.2 | 1445.4 ± 818.5 | <0.001 | <0.001 | <0.001 |
| Carbohydrates (g/day) | 195.8 ± 116.3 | 201.1 ± 128.9 | 237.4 ± 135.3 | - | 0.010 | 0.014 |
| Protein (g/day) | 69.3 ± 57.1 | 32.6 ± 28.3 | 47.1 ± 35.9 | <0.001 | <0.001 | 0.001 |
| Total fat (g/day) | 82.9 ± 51.2 | 19.9 ± 18.5 | 41.2 ± 43.8 | <0.001 | <0.001 | <0.001 |
| Iron (g/day) | 10.1 ± 9.9 | 6.9 ± 5.9 | 11.3 ± 8.4 | <0.001 | - | <0.001 |
| Hemoglobin (g/dL) | 11.0 ± 1.1 | 10.7 ± 1.2 | 10.7 ± 1.1 | 0.017 | - | - |
| MUAC (cm) | 26.3 ± 2.7 | 26.3 ± 2.6 | 26.8 ± 2.6 | - | - | - |
| AMA (cm2) | 35.3 ± 6.2 | 37.3 ± 6.5 | 37.0 ± 6.3 | 0.007 | - | - |
| AFA (cm2) | 20.2 ± 8.0 | 18.7 ± 7.0 | 20.7 ± 8.5 | - | - | 0.003 |
| Infant weight at delivery (g) | 3058.6 ± 431.4 | 2976.9 ± 434.4 | 3053.9 ± 395.2 | - | - | - |
| Outcome | Mean Difference | 95% CI | p value | |
|---|---|---|---|---|
| AMA 1 (cm2) | ||||
| Grain (compared to animal) | 1.86 | 0.53 | 3.19 | 0.01 |
| Plant (compared to animal) | 1.23 | −0.14 | 2.60 | 0.08 |
| Plant (compared to grain) | −0.63 | −1.89 | 0.63 | 0.33 |
| AFA 1 (cm2) | ||||
| Grain (compared to animal) | −2.09 | −3.75 | −0.44 | 0.01 |
| Plant (compared to animal) | 0.38 | −1.33 | 2.08 | 0.67 |
| Plant (compared to grain) | 2.47 | 0.90 | 4.03 | <0.01 |
| Hb 1 (g/dL) | ||||
| Grain (compared to animal) | −0.27 | −0.52 | −0.01 | 0.04 |
| Plant (compared to animal) | −0.32 | −0.59 | −0.07 | 0.01 |
| Plant (compared to grain) | −0.06 | −0.30 | 0.18 | 0.62 |
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Scrimshaw, N.; SanGiovanni, J. Synergism of nutrition, infection and immunity: An overview. Am. J. Clin. Nutr. 1997, 66, 464S–477S. [Google Scholar] [PubMed]
- HIV/AIDS JUNPo. Global report: UNAIDS report on the global AIDS epidemic: 2012; UNAIDS: Geneva, Switzerland, 2012. [Google Scholar]
- Villamor, E.; Msamanga, G.; Spiegelman, D.; Coley, J.; Hunter, D.J.; Peterson, K.E.; Fawzi, W.W. HIV status and sociodemographic correlates of maternal body size and wasting during pregnancy. Eur. J. Clin. Nutr. 2002, 56, 415–424. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Fawzi, W.W.; Hunter, D.J. Vitamins in HIV disease progression and vertical transmission. Epidemiology 1998, 9, 457–466. [Google Scholar] [CrossRef] [PubMed]
- Hartikainen, H.M.K.; Kulmala, T.; Ashorn, P. Seasonality of gestational weight gain and fetal growth in rural Malawi. East Afr. Med. J. 2005, 82, 294–299. [Google Scholar] [CrossRef] [PubMed]
- Larmarange, J. [HIV prevalence in sub-Saharan Africa: background of an estimation]. Med. Sci. (Paris) 2009, 25, 87–92. [Google Scholar] [CrossRef]
- Moses, A.; Zimba, C.; Kamanga, E.; Nkhoma, J.; Maida, A.; Martinson, F.; Mofolo, I.; Joaki, G.; Muita, J.; Spensley, A.; et al. Prevention of mother-to-child transmission: Program changes and the effect on uptake of the HIVNET 012 regimen in Malawi. Aids 2008, 22, 83–87. [Google Scholar] [CrossRef] [PubMed]
- Van der Horst, C.; Chasela, C.; Ahmed, Y.; Hoffman, I.; Hosseinipour, M.; Knight, R.; Fiscus, S.; Hudgens, M.; Kazembe, P.; Bentley, M.; et al. Modifications of a large HIV prevention clinical trial to fit changing realities: A case study of the Breastfeeding, Antiretroviral, and Nutrition (BAN) protocol in Lilongwe, Malawi. Contemp. Clin. Trials 2009, 30, 24–33. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Chasela, C.S.; Hudgens, M.G.; Jamieson, D.J.; Kayira, D.; Hosseinipour, M.C.; Kourtis, A.P.; Martinson, F.; Tegha, G.; Knight, R.J.; Ahmed, Y.I.; et al. Maternal or infant antiretroviral drugs to reduce HIV-1 transmission. N. Engl. J. Med. 2010, 362, 2271–2281. [Google Scholar] [CrossRef] [PubMed]
- Ferguson, E.L.; Gadowsky, S.L.; Huddle, J.M.; Cullinan, T.R.; Lehrfeld, J.; Gibson, R.S. An interactive 24-h recall technique for assessing the adequacy of trace mineral intakes of rural Malawian women; its advantages and limitations. Eur. J. Clin. Nutr. 1995, 49, 565–578. [Google Scholar] [PubMed]
- Lukmanji, Z.H.E.; Mlingi, N.; Assey, V.; Ndossi, G.; Fawzi, W. Tanzania Food Composition Tables; MUHAS-TFNC, HSPH: Dar es Salaam, Tanzania, 2008. [Google Scholar]
- USDA National Nutrient Database for Standard Reference, Release 22; United States Department of Agriculture, Agricultural Research Service: Washington, DC, USA, 2010.
- National Statistical Office (NSO) [Malawi] aOM. Malawi Demographic and Health Survey 2004; NSO: Lilongwe, Malawi, 2005. [Google Scholar]
- Chen, H.; Ward, M.H.; Graubard, B.I.; Heineman, E.F.; Markin, R.M.; Potischman, N.A.; Russell, R.M.; Weisenburger, D.D.; Tucker, K.L. Dietary patterns and adenocarcinoma of the esophagus and distal stomach. Am. J. Clin. Nutr. 2002, 75, 137–144. [Google Scholar] [PubMed]
- Newby, P.K.; Tucker, K.L. Empirically derived eating patterns using factor or cluster analysis: A review. Nutr. Rev. 2004, 62, 177–203. [Google Scholar] [CrossRef] [PubMed]
- StataCorp. Stata Statistical Software: Release 9; StataCorp LP: College Station, TX, USA, 2005. [Google Scholar]
- Kolenikov, S.A.G. The Use of Discrete Data in PCA: Theory, Simulations, and Applications to Socioeconomic Indices; University of North Carolina at Chapel Hill and Carolina Population Center: Chapel Hill, NC, USA, 2004. [Google Scholar]
- Harrell, F.E. Regression Modeling Strategies: With Application to Linear Models, Logistic Regression and Survival Analysis; Springer Science+Business Media, Inc: New York, NY, USA, 2001. [Google Scholar]
- Huddle, J.M.; Gibson, R.S.; Cullinan, T.R. Is zinc a limiting nutrient in the diets of rural pregnant Malawian women? Br. J. Nutr. 1998, 79, 257–265. [Google Scholar] [CrossRef] [PubMed]
- Friis, H.; Gomo, E.; Nyazema, N.; Ndhlovu, P.; Kaestel, P.; Krarup, H.; Michaelsen, K.F. HIV-1 viral load and elevated serum alpha(1)-antichymotrypsin are independent predictors of body composition in pregnant Zimbabwean women. J. Nutr. 2002, 132, 3747–3753. [Google Scholar] [PubMed]
- Friis, H.; Gomo, E.; Nyazema, N.; Ndhlovu, P.; Krarup, H.; Kaestel, P.; Michaelsen, K.F. Maternal body composition, HIV infection and other predictors of gestation length and birth size in Zimbabwe. Br. J. Nutr. 2004, 92, 833–840. [Google Scholar] [CrossRef] [PubMed]
- Antelman, G.; Msamanga, G.I.; Spiegelman, D.; Urassa, E.J.; Narh, R.; Hunter, D.J.; Fawzi, W.W. Nutritional factors and infectious disease contribute to anemia among pregnant women with human immunodeficiency virus in Tanzania. J. Nutr. 2000, 130, 1950–1957. [Google Scholar] [PubMed]
- Brocklehurst, P.; French, R. The association between maternal HIV infection and perinatal outcome: A systematic review of the literature and meta-analysis. Br. J. Obstet. Gynaecol. 1998, 105, 836–848. [Google Scholar] [CrossRef] [PubMed]
- Ramlal, R.T.; Tembo, M.; Soko, A.; Chigwenembe, M.; Tohill, B.C.; Kayira, D.; King, C.C.; Chasela, C.; Jamieson, D.; van der Horst, C. Patterns of Body Composition Among HIV-Infected, Pregnant Malawians and the Effects of Famine Season. Matern. Child Health J. 2013, 17, 265–273. [Google Scholar] [CrossRef] [PubMed]
- Nyambose, J.; Koski, K.G.; Tucker, K.L. High intra/interindividual variance ratios for energy and nutrient intakes of pregnant women in rural Malawi show that many days are required to estimate usual intake. J. Nutr. 2002, 132, 1313–1318. [Google Scholar] [PubMed]
- Newby, P.K.; Muller, D.; Tucker, K.L. Associations of empirically derived eating patterns with plasma lipid biomarkers: A comparison of factor and cluster analysis methods. Am. J. Clin. Nutr. 2004, 80, 759–767. [Google Scholar] [PubMed]
- Hu, F.B. Dietary pattern analysis: A new direction in nutritional epidemiology. Curr. Opin. Lipidol. 2002, 13, 3–9. [Google Scholar] [CrossRef] [PubMed]
- Kant, A.K. Dietary patterns and health outcomes. J. Am. Diet. Assoc. 2004, 104, 615–635. [Google Scholar] [CrossRef] [PubMed]
- Panagiotakos, D.B.; Pitsavos, C.; Skoumas, Y.; Stefanadis, C. The association between food patterns and the metabolic syndrome using principal components analysis: The ATTICA Study. J. Am. Diet. Assoc. 2007, 107, 979–987. [Google Scholar] [CrossRef] [PubMed]
- Newby, P.K.; Muller, D.; Hallfrisch, J.; Andres, R.; Tucker, K.L. Food patterns measured by factor analysis and anthropometric changes in adults. Am. J. Clin. Nutr. 2004, 80, 504–513. [Google Scholar] [PubMed]
- Newby, P.K.; Muller, D.; Hallfrisch, J.; Qiao, N.; Andres, R.; Tucker, K.L. Dietary patterns and changes in body mass index and waist circumference in adults. Am. J. Clin. Nutr. 2003, 77, 1417–1425. [Google Scholar] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ramlal, R.T.; Tembo, M.; King, C.C.; Ellington, S.; Soko, A.; Chigwenembe, M.; Chasela, C.; Jamieson, D.J.; Van der Horst, C.; Bentley, M.; et al. Dietary Patterns and Maternal Anthropometry in HIV-Infected, Pregnant Malawian Women. Nutrients 2015, 7, 584-594. https://doi.org/10.3390/nu7010584
Ramlal RT, Tembo M, King CC, Ellington S, Soko A, Chigwenembe M, Chasela C, Jamieson DJ, Van der Horst C, Bentley M, et al. Dietary Patterns and Maternal Anthropometry in HIV-Infected, Pregnant Malawian Women. Nutrients. 2015; 7(1):584-594. https://doi.org/10.3390/nu7010584
Chicago/Turabian StyleRamlal, Roshan T., Martin Tembo, Caroline C. King, Sascha Ellington, Alice Soko, Maggie Chigwenembe, Charles Chasela, Denise J. Jamieson, Charles Van der Horst, Margaret Bentley, and et al. 2015. "Dietary Patterns and Maternal Anthropometry in HIV-Infected, Pregnant Malawian Women" Nutrients 7, no. 1: 584-594. https://doi.org/10.3390/nu7010584
APA StyleRamlal, R. T., Tembo, M., King, C. C., Ellington, S., Soko, A., Chigwenembe, M., Chasela, C., Jamieson, D. J., Van der Horst, C., Bentley, M., Adair, L., & The BAN Study Team. (2015). Dietary Patterns and Maternal Anthropometry in HIV-Infected, Pregnant Malawian Women. Nutrients, 7(1), 584-594. https://doi.org/10.3390/nu7010584




