Zinc Fortification Decreases ZIP1 Gene Expression of Some Adolescent Females with Appropriate Plasma Zinc Levels
Abstract
:1. Introduction
2. Experimental Section
2.1. Subjects
2.2. Intervention
2.3. Blood Samples
2.3.1. Plasma Zinc and CRP Status
2.3.2. Primer Design and Gene Expression Analysis
| Primers | PCR length (bp) | Sequence 5′–3′ | Accession No. | |
|---|---|---|---|---|
| ZIP1 | Fw | 231 | CGTGCCTGTGTACTGGTGTT | NM_020342.2 |
| Rv | ATGACACCTCTAGGCATCGG | |||
| ZnT1 | Fw | 210 | TGGAGGTGGCTAAAACCATT | NM_021194.2 |
| Rv | TGCTAACTGCTGGGGTCTTT | |||
| Β-actin | Fw | 221 | GCAAGCAGGAGTATGACG | NM_001101.3 |
| Rv | GTCACCTTCACCGTTCCAGT | |||
2.4. Food Intake
2.5. Physical Activity Questionnaire
2.6. Socioeconomic Status
2.7. Statistics
3. Results
3.1. Baseline Characteristics of the Participants
| Characteristics | Mean ± SD | Range |
|---|---|---|
| Age (year) | 14.1 ± 1.1 | 12–16 |
| Weight (kg) | 56.3 ± 11.1 | 43–77.5 |
| Height (cm) | 159.3 ± 4.3 | 152.5–169.4 |
| BMI/age (kg/m2) | 22.2 ± 4.3 | 18.1–32.9 |
| Age at menarche (year) | 12.2 ± 1.2 | 10–14 |
| Physical activity (mMB) | Sedentary | <1.56 mBM |
3.2. Intake and Plasma Zinc Changes
| Variable | Basal | After intervention | p |
|---|---|---|---|
| Energy (kcal/day) | 1930.1 ± 709.5 | 2193.1 ± 693.8 | NS |
| Protein (g/day) | 65.15 ± 26.8 | 73.7 ± 21.0 | NS |
| Zinc (mg/day) | 10.5 ± 3.9 | 17.6 ± 4.4 | <0.001 |
| Phytate (mg/day) | 1105.5 ± 1110.5 | 1384.7 ± 1101.9 | NS |
| Phytate:zinc molar ratio | 11.6 ± 9.7 | 6.8 ± 5.4 | <0.05 |
| Estimated Zn absorption | 3.2 ± 1.2 | 5.3 ± 1.3 | <0.001 |
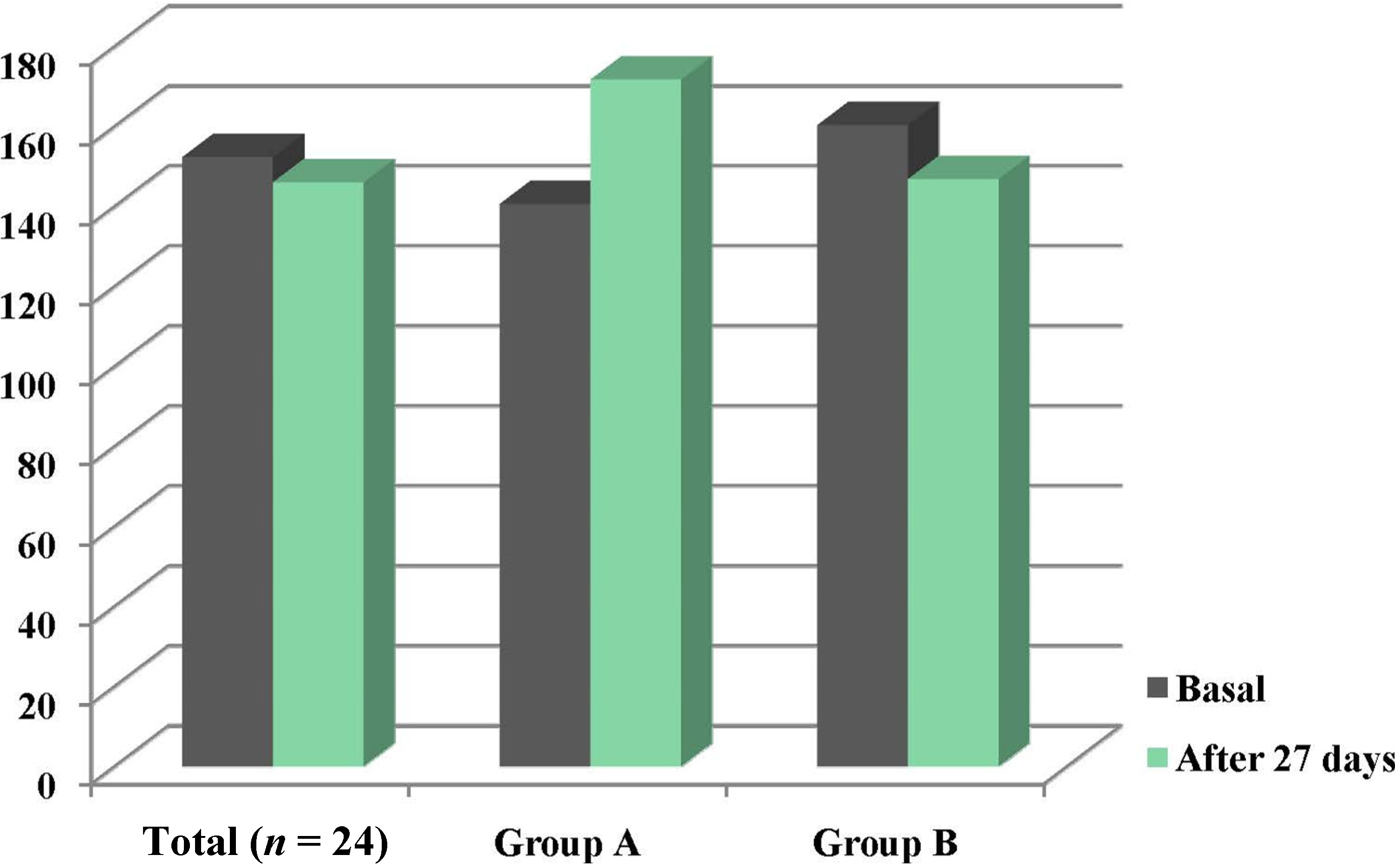
3.3. Changes in the ZnT1 and ZIP1 Expression Levels
| Study Group | Basal | After intervention | p * |
|---|---|---|---|
| Fold change of ZIP1 | |||
| A | 1 | −1.40 | 0.006 |
| B | 1 | −1.44 | 0.078 |
| Fold change of ZnT1 | |||
| A | 1 | 1.75 | 0.55 |
| B | 1 | 1.09 | 0.88 |
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- King, J. Zinc: An essential but elusive nutrient. Am. J. Clin. Nutr. 2011, 94, 679S–684S. [Google Scholar] [CrossRef]
- Prasad, A. Impact of the discovery of human zinc deficiency on health. J. Am. Coll. Nutr. 2009, 28, 257–265. [Google Scholar] [CrossRef]
- Brown, K.H.; Hambidge, K.M.; Ranum, P. Zinc fortification of cereal flours: Current recommendations and research needs. Food Nutr. Bull. 2010, 31, S62–S74. [Google Scholar]
- Serdula, M. Maximizing the impact of flour fortification to improve vitamin and mineral nutrition in populations. Food Nutr Bull. 2010, 31, S86–S93. [Google Scholar]
- Méndez, R.O.; Galdámez, K.; Grijalva, M.I.; Quihui, L.; García, H.S.; Calderon de la Barca, A.M. Effect of micronutrient-fortified milk on zinc intake and plasma concentration in adolescent girls. J. Am. Coll. Nutr. 2012, 31, 408–414. [Google Scholar] [CrossRef]
- Andree, K.B.; Kim, J.; Kirschke, C.P.; Gregg, J.P.; Paik, H.; Joung, H.; Woodhouse, L.; King, J.C.; Huang, L. Investigation of lymphocyte gene expression for use as biomarkers for zinc status in humans. J. Nutr. 2004, 134, 716–723. [Google Scholar]
- Ryu, M.S.; Guthrie, G.J.; Maki, A.B.; Aydemir, T.B.; Cousins, R.J. Proteomic analysis shows the upregulation of erythrocyte dematin in zinc-restricted human subjects. Am. J. Clin. Nutr. 2012, 95, 1096–1102. [Google Scholar] [CrossRef]
- Dufner-Beattie, J.; Kuo, Y.M.; Gitschier, J.; Andrews, G.K. The adaptive response to dietary zinc in mice involves the differential cellular localization and zinc regulation of the zinc transporters ZIP4 and ZIP5. J. Biol. Chem. 2004, 279, 49082–49090. [Google Scholar] [CrossRef]
- Liuzzi, J.; Cousin, R. Mammalian zinc transporters. Annu. Rev. Nutr. 2004, 24, 151–172. [Google Scholar]
- De Onis, M.; Onyango, A.W.; Borghi, E.; Siyam, A.; Nishida, C.; Siekmann, J. Development of a WHO growth reference for school-aged children and adolescents. Bull. World Health Organ. 2007, 85, 660–667. [Google Scholar] [CrossRef]
- Association of Official Analytical Chemists. Official Methods of Analysis of Association of Official Analytical Chemists, 16th ed.; Method 991.11. Zinc in Serum Flame Atomic Absorption Spectrophotometric Method; AOAC: Arlington, VA, USA, 1995. [Google Scholar]
- Livack, K.; Schmittgen, T. Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCt method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef]
- Ortega, M.I.; Quizán, T.; Morales, G.G.; Preciado, M. Cálculo de la ingestión dietaria y coeficientes de adecuación a partir de: Registro de 24 horas y frecuencia de consumo de alimentos [Food consumption and diet adequation analysis: 24 hours recall and food frequency questionnaires]. Ser. Eval. Consum. Aliment. 1999, 1, 1–48. [Google Scholar]
- World Health Organization. Energy and Protein Requirements; Report of a Joint FAO/WHO/UNU Expert Consultation; World Health Organization: Geneva, Switzerland, 1985. [Google Scholar]
- Asociación Mexicana de Agencias de Inteligencia de Mercado y Opinión A.C. Available online: http://www.amai.org/NSE/AMAINSE2004.ppt (accessed on 13 January 2013).
- Institute of Medicine (U.S.). Dietary Reference Intakes for Vitamin A, Vitamin K, Arsenic, Boron, Chromium, Copper, Iodine, Iron, Manganese, Molybdenum, Nickel, Silicon, Vanadium, and Zinc: A Report of the Panel on Micronutrients and the Standing Committee on the Scientific Evaluation of Dietary Reference Intakes; Food and Nutrition Board, Institute of Medicine, National Academy Press: Washington, DC, USA, 2001. [Google Scholar]
- Aydemir, T.; Blanchard, R.; Cousins, R. Zinc supplementation of young men alters metallothionein, zinc transporter, and cytokine gene expression in leukocyte populations. Proc. Natl. Acad. Sci. USA 2006, 103, 1699–1704. [Google Scholar] [CrossRef]
- Baqui, A.; Black, R.; Fischer, C.; Arifeen, S.; Zaman, K.; Yunus, M.; Wahed, M.; Caulfield, L. Zinc Supplementation and Serum Zinc During Diarrhea. Indian J. Pediatr. 2006, 73, 493–497. [Google Scholar] [CrossRef]
- Mocchegiani, E.; Giacconi, R.; Costarelli, L.; Muti, E.; Cipriano, C.; Tesei, S.; Pierpaoli, S.; Giuli, C.; Papa, R.; Marcellini, F.; et al. Zinc deficiency and IL-6 −174G/C polymorphism in old people from different European countries: Effect of zinc supplementation. ZINCAGE study. Exp. Gerontol. 2008, 43, 433–444. [Google Scholar] [CrossRef]
- Mariani, E.; Neri, S.; Cattini, L.; Mocchegiani, E.; Malavolta, M.; Dedoussis, G.V.; Kanoni, S.; Rink, L.; Jajte, J.; Facchini, A. Effect of zinc supplementation on plasma IL-6 and MCP-1 production and NK cell function in healthy elderly: Interactive influence of +647 MT1a and −174 IL-6 polymorphic alleles. Exp. Gerontol. 2008, 43, 462–471. [Google Scholar] [CrossRef]
- IZiNCG. Preventing Zinc Deficiency through Diet Diversification and Modification; Technical Brief. No. 05 2007; IZiNCG: Davis, CA, USA, 2007. [Google Scholar]
- Ortega, M.I.; Valencia, M.E. Measuring the intakes of foods and nutrients of marginal population in North-West Mexico. Public Health Nutr. 2012, 5, 907–910. [Google Scholar] [CrossRef]
- Rodríguez-Ramírez, S.; Mundo-Rosas, V.; Shamah, T.; Ponce-Martínez, X.; Jiménez, A.; González-de-Cossío, T. Energy and nutrient intake in Mexican adolescents: Analysis of the Mexican national health and nutrition survey 2006. Salud Pública Méx 2009, 51, S551–S561. [Google Scholar]
- Noh, H.; Paik, H.Y.; Kim, J.; Chung, J. The Alteration of Zinc Transporter Gene Expression Is Associated with Inflammatory Markers in Obese Women. Biol. Trace Elem. Res. 2014, 158, 1–8. [Google Scholar] [CrossRef]
- Encuesta Nacional de Salud y Nutrición, 2012. Estado de Nutrición, Anemia, Seguridad Alimentaria en la Población Mexicana. Available online: http://ensanut.insp.mx/doctos/ENSANUT2012_Nutricion.pdf (accessed on 12 January 2013).
- Encuesta Nacional de Salud y Nutrición, 2012. Resultados por Entidad Federativa. Available online: http://ensanut.insp.mx/informes/Sonora-OCT.pdf (accessed on 12 January 2013).
- Tungtrongchitr, R.; Pongpaew, P.; Phonrat, B.; Tungtrongchitr, A.; Viroonudomphol, D.; Vudhivai, N.; Schelp, F.P. Serum copper, zinc, ceruloplasmin and superoxide dismutase in Thai overweight and obese. J. Med. Assoc. Thail. 2003, 86, 543–551. [Google Scholar]
- Ennes, D.F.; de Sousa, V.B.; Mello, N.R.; Franciscato, S.M.; do Nascimento, D. Biomarkers of metabolic syndrome and its relationship with the zinc nutritional status in obese women. Nutr. Hosp. 2011, 26, 650–654. [Google Scholar]
- Foster, M.; Hancock, D.; Petocz, P.; Samman, S. Zinc transporter genes are coordinately expressed in men and women independently of dietary or plasma zinc. J. Nutr. 2011, 141, 1195–1201. [Google Scholar] [CrossRef]
- Sekler, I.; Sensi, S.L.; Hershfinkel, M.; Silverman, W.F. Mechanism and regulation of cellular zinc transport. Mol. Med. 2007, 13, 337–343. [Google Scholar]
- Liuzzi, J.P.; Bobo, J.A.; Lichten, L.A.; Samuelson, D.A.; Cousins, R.J. Responsive transporter genes within the murine intestinal-pancreatic axis form a basis of zinc homeostasis. Proc. Natl. Acad. Sci. USA 2004, 101, 14355–14360. [Google Scholar]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Méndez, R.O.; Santiago, A.; Yepiz-Plascencia, G.; Peregrino-Uriarte, A.B.; Barca, A.M.C.d.l.; García, H.S. Zinc Fortification Decreases ZIP1 Gene Expression of Some Adolescent Females with Appropriate Plasma Zinc Levels. Nutrients 2014, 6, 2229-2239. https://doi.org/10.3390/nu6062229
Méndez RO, Santiago A, Yepiz-Plascencia G, Peregrino-Uriarte AB, Barca AMCdl, García HS. Zinc Fortification Decreases ZIP1 Gene Expression of Some Adolescent Females with Appropriate Plasma Zinc Levels. Nutrients. 2014; 6(6):2229-2239. https://doi.org/10.3390/nu6062229
Chicago/Turabian StyleMéndez, Rosa O., Alejandra Santiago, Gloria Yepiz-Plascencia, Alma B. Peregrino-Uriarte, Ana M. Calderón de la Barca, and Hugo S. García. 2014. "Zinc Fortification Decreases ZIP1 Gene Expression of Some Adolescent Females with Appropriate Plasma Zinc Levels" Nutrients 6, no. 6: 2229-2239. https://doi.org/10.3390/nu6062229
APA StyleMéndez, R. O., Santiago, A., Yepiz-Plascencia, G., Peregrino-Uriarte, A. B., Barca, A. M. C. d. l., & García, H. S. (2014). Zinc Fortification Decreases ZIP1 Gene Expression of Some Adolescent Females with Appropriate Plasma Zinc Levels. Nutrients, 6(6), 2229-2239. https://doi.org/10.3390/nu6062229




