Sasa borealis Stem Extract Attenuates Hepatic Steatosis in High-Fat Diet-induced Obese Rats
Abstract
:1. Introduction
2. Experimental Section
2.1. Preparation of Sasa borealis Stem (SBS) Extracts
2.2. Measurement of Total Phenolic Content Using the Folin-Ciocalteu Assay
2.3. Measurement of Total Flavonoids
2.4. Measurement of Free Radical Scavenging Activity Using the 2,2-Diphenyl-1-picrylhydrazyl (DPPH) Assay
2.5. Measurement of Hydroxyl (OH−) Radical Scavenging Activity
2.6. Measurement of ABTS Radical Scavenging Activity
2.7. High-Performance Liquid Chromatography (HPLC) Analysis of SBS Compounds
2.8. Animals and Diets
| Ingredient (milligram) | HFD | HFD + SBS (mg/kg/day) |
|---|---|---|
| Casein | 200 | 200 |
| l-Cystine | 3 | 3 |
| Maltodextrin 10 | 125 | 125 |
| Sucrose | 68.8 | 68.8 |
| Cellulose | 50 | 50 |
| Soybean Oil | 25 | 25 |
| Lard | 245 | 245 |
| Mineral Mix S10026 | 10 | 10 |
| DiCalcium Phosphate | 13 | 13 |
| Calcium Carbonate | 5.5 | 5.5 |
| Potassuium Citrate | 16.5 | 16.5 |
| Vitamin Mix V10001 | 10 | 10 |
| Choline Bitartrate | 2 | 2 |
| Protein (milligram%) | 26.2 | 26.2 |
| Carbohydrate (milligram%) | 26.3 | 26.3 |
| Fat (milligram%) | 34.9 | 34.9 |
| SBS extracts | - | 150 |
2.9. Biochemical Analysis
2.10. Histopathological Examinations
2.11. Gene Expression Analysis
2.12. Statistical Analysis
3. Results
3.1. Total Phenol Content (TPC) and Total Flavonoid Content (TFC) of SBS Extracts
| TPC (mg Gallic acid/g extract) | TFC (mg Quercetin/g extract) | IC50 (µg/mL) | ABTS assay TEAC | ||
|---|---|---|---|---|---|
| DPPH scavenging assay | HRSA scavenging assay | ||||
| SBS | 430.0 ± 40.2 | 127.5 ± 12.0 | 43.7 ± 2.3 | 450.5 ± 28.5 | 0.6 ± 0.05 |
3.2. Antioxidant Activity of SBS Extracts
| Standards | RT 1 | λ 2 | Calibration curve | LOQ 3 | Compounds |
|---|---|---|---|---|---|
| Phenolic acids | |||||
| Phloroglucinol | 7.27 | 280 | Y = 397.949X + 0.655 | 50.00 | 0.60 ± 0.00 |
| 4-Hydroxy benzhydrazide derivative | 7.57 | 280 | Y = 8119.555X − 59.083 | 50.00 | 0.26 ± 0.00 |
| Gallic acid | 8.74 | 280 | Y = 18,200.182X − 28.003 | 50.00 | 0.53 ± 0.00 |
| Vanillic acid | 21.91 | 280 | Y = 11,026.185X + 14.026 | 50.00 | 1.44 ± 0.01 |
| Caffeic acid | 22.21 | 280 | Y = 19,697.774X − 13.018 | 50.00 | 0.74 ± 0.00 |
| Syringic acid | 24.10 | 280 | Y = 17,500.224X − 1.523 | 5.00 | 1.41 ± 0.01 |
| Chlorogenic acid | 24.92 | 280 | Y = 6240.064X − 10.524 | 50.00 | 1.61 ± 0.01 |
| p-Coumaric acid | 32.87 | 280 | Y = 23,926.358X + 0.631 | 5.00 | 10.41 ± 0.05 |
| trans-Ferulic acid | 34.48 | 280 | Y = 16,058.167X − 17.063 | 50.00 | 3.44 ± 0.02 |
| Sinapic acid | 34.91 | 280 | Y = 7025.930X + 0.785 | 50.00 | 2.98 ± 0.01 |
| 2-Amino-3,4-dimethyl-benzoicacid | 35.30 | 280 | Y = 1209.000X + 0.000 | 50.00 | 6.95 ± 0.04 |
| p-Anisic acid | 35.40 | 280 | Y = 9558.576X + 5.493 | 50.00 | - |
| Protocatechuic acid ethyl ester | 36.89 | 280 | Y = 8796.340X − 2.765 | 50.00 | 13.05 ± 0.06 |
| Coumarin | 38.27 | 280 | Y = 24,055.754X + 48.641 | 4.00 | 14.91 ± 0.04 |
| DPBA 4 | 39.85 | 280 | Y = 2971.415X − 7.872 | 50.00 | - |
| Alizarin | 43.86 | 280 | Y = 15,428.805X + 19.936 | 1.00 | - |
| Total phenolic acids | 62.98 ± 0.08 | ||||
| Flavonoids | |||||
| Gallocatechin | 17.68 | 280 | Y = 1331.637X + 0.000 | 50.00 | 1.59 ± 0.00 |
| Epigallocatechin | 18.58 | 280 | Y = 96.137X − 0.550 | 50.00 | 0.73 ± 0.00 |
| Catechin hydrate | 23.66 | 280 | Y = 3982.083X − 6.943 | 5.00 | 2.65 ± 0.01 |
| Epicatechin | 28.00 | 280 | Y = 7641.670X − 14.487 | 50.00 | 2.47 ± 0.00 |
| Epigallocatechin gallate | 29.53 | 280 | Y = 6425.894X − 6.592 | 50.00 | 0.98 ± 0.00 |
| Rutin hydrate | 32.93 | 370 | Y = 4763.242X − 4.752 | 50.00 | 0.29 ± 0.01 |
| Catechin gallate | 33.77 | 280 | Y = 1462.905X − 1.970 | 50.00 | - |
| Naringin | 34.14 | 280 | Y = 8230.457X − 42.997 | 50.00 | 2.60 ± 0.02 |
| Quercetin hydrate | 37.53 | 370 | Y = 7476.858X − 6.972 | 50.00 | 2.48 ± 0.02 |
| Myricetin | 37.41 | 370 | Y = 9908.955X − 0.383 | 5.00 | 0.43 ± 0.00 |
| Morin hydrate | 38.48 | 320 | Y = 4100.693X − 4.129 | 50.00 | - |
| Quercetin dehydrate | 40.19 | 370 | Y = 5623.574X − 0.729 | 50.00 | 1.93 ± 0.00 |
| Luteolin | 40.28 | 370 | Y = 12,303.249X − 8.820 | 50.00 | 6.32 ± 0.03 |
| Kaempferol | 42.89 | 370 | Y = 12,894.258X + 38.962 | 20.00 | 0.95 ± 0.03 |
| 3-Hydroxyflavone | 45.75 | 320 | Y = 4687.303X + 0.191 | 50.00 | - |
| Total flavonoids | 23.42 ± 0.09 |
3.3. Effect of SBS on Body Weight and Liver Weight in HFD-Fed Rats
| ND | HFD | HFD + SBS | |
|---|---|---|---|
| Food intake (g/day) | 12.03 ± 1.1 | 11.04 ± 1.6 | 11.44 ± 1.5 |
| Body weight | |||
| Initial weight (g) | 136.2 ± 4.3 | 135.5 ± 2.7 | 134.2 ± 3.7 |
| Final weight (g) | 338.7 ± 16.8 | 447.3 ± 20.5 * | 388.5 ± 15.5 # |
| Weight gain (g/5 weeks) | 202.5 ± 9.3 | 311.8 ± 16.3 * | 254.3 ± 10.7 # |
| Liver weight (g/5 weeks) | 2.5 ± 0.2 | 3.8 ± 0.3 * | 3.2 ± 0.3 # |
3.4. Effect of SBS on Serum Total Cholesterols and Triglyceride Levels
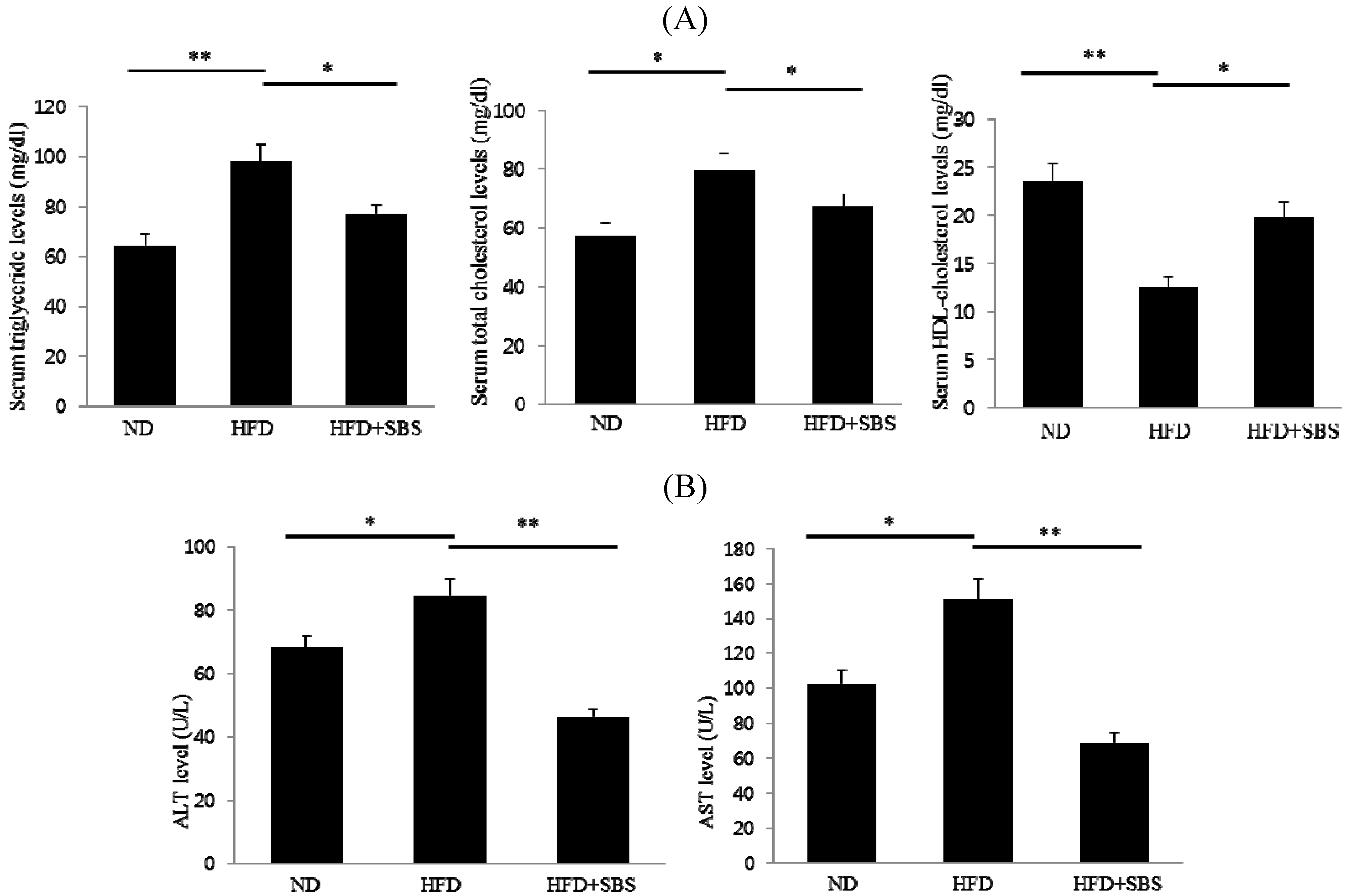
3.5. Effects of SBS on Hepatic Total Cholesterols Levels and Triglyceride Levels
3.6. Effects of SBS on Hapatic Steatosis
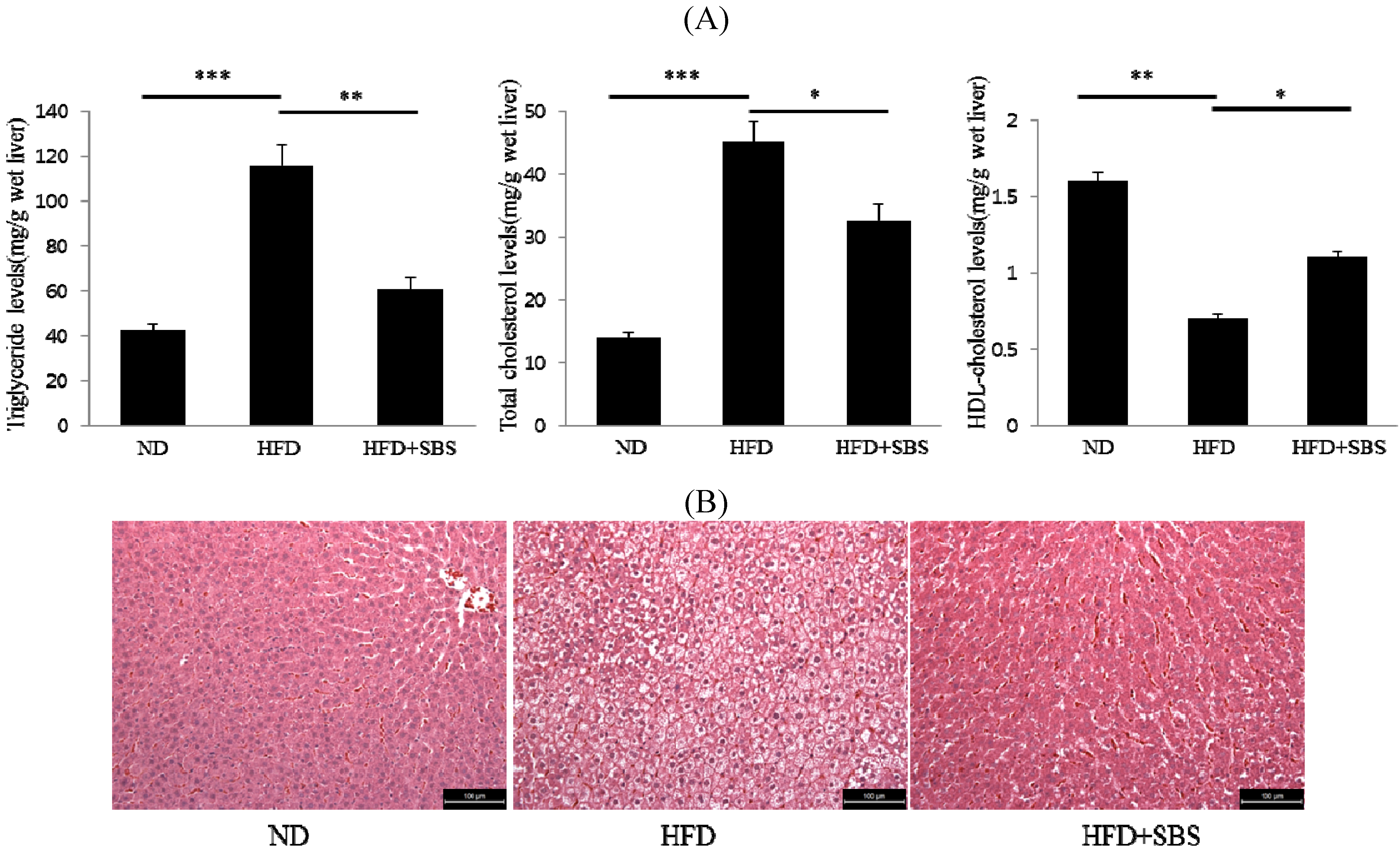
3.7. Effects of SBS on Hepatic mRNA Levels of Lipid-Related Gene Expression
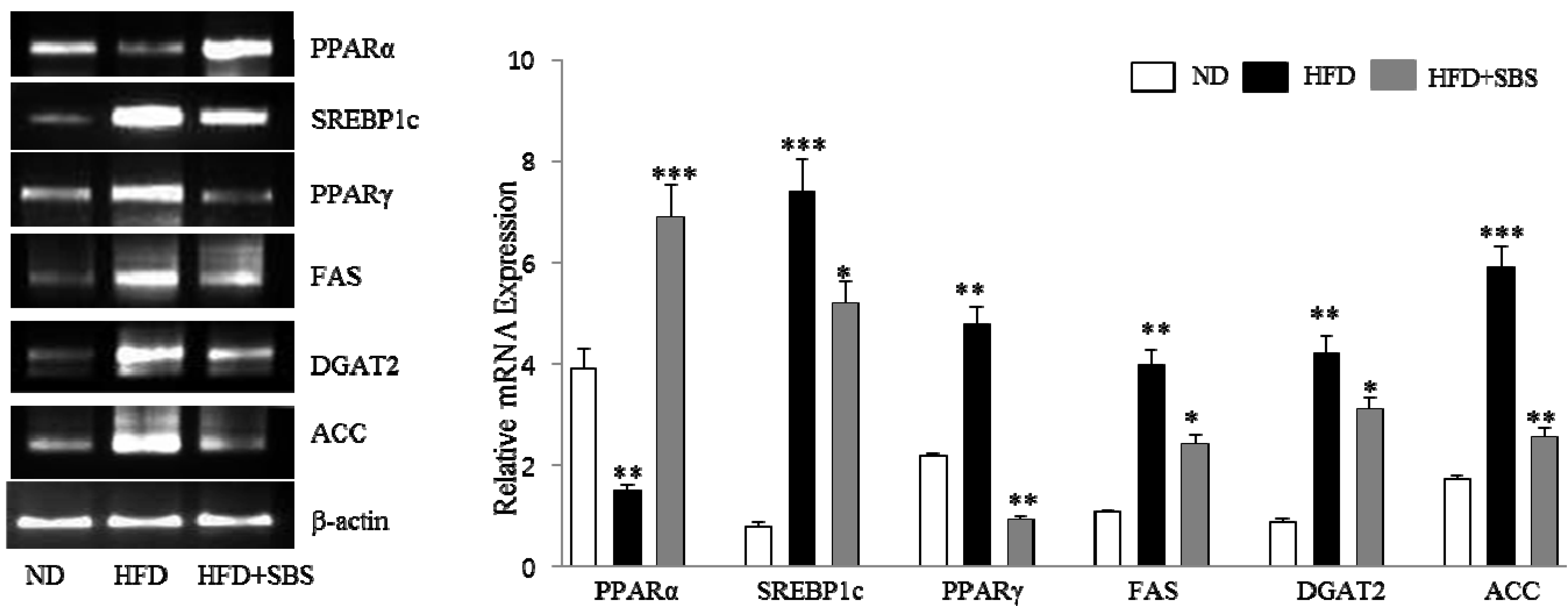
4. Discussion
5. Conclusions
Acknowledgements
Conflicts of Interest
References
- Ludwig, J.; Viggiano, T.R.; McGill, D.B.; Oh, B.J. Nonalcoholic steatohepatitis: Mayo clinic experience with a hitherto unnamed disease. Mayo Clin. Proc. 1980, 55, 434–438. [Google Scholar]
- Marchesini, G.; Bugianesi, E.; Forlani, G.; Cerrelli, F.; Lenzi, M.; Manini, R.; Natale, S.; Vanni, E.; Villanova, N.; Melchionda, N.; Rizzetto, M. Nonalcoholic fatty liver, steatohepatitis, and the metabolic syndrome. Hepatology 2003, 37, 917–923. [Google Scholar] [CrossRef]
- Powell, E.E.; Cooksley, W.G.; Hanson, R.; Searle, J.; Halliday, J.W.; Powell, L.W. The natural history of nonalcoholic steatohepatitis: A follow-up study of forty-two patients for up to 21 years. Hepatology 1990, 11, 74–80. [Google Scholar] [CrossRef]
- Marchesini, G.; Brizi, M.; Bianchi, G.; Tomassetti, S.; Bugianesi, E.; Lenzi, M.; McCullough, A.J.; Natale, S.; Forlani, G.; Melchionda, N. Nonalcoholic fatty liver disease: A feature of the metabolic syndrome. Diabetes 2001, 50, 1844–1850. [Google Scholar] [CrossRef]
- Fon Tacer, K.; Rozman, D. Nonalcoholic fatty liver disease: Focus on lipoprotein and lipid deregulation. J. Lipids 2011, 2011, 783976. [Google Scholar]
- Browning, J.D.; Horton, J.D. Molecular mediators of hepatic steatosis and liver injury. J. Clin. Investig. 2004, 114, 147–152. [Google Scholar] [CrossRef]
- Nagle, C.A.; Klett, E.L.; Coleman, R.A. Hepatic triacylglycerol accumulation and insulin resistance. J. Lipid Res. 2009, 50, S74–S79. [Google Scholar] [CrossRef]
- Prior, R.L.; Cao, G. Antioxidant capacity and polyphenolic components of teas: Implications for altering in vivo antioxidant status. Proc. Soc. Exp. Biol. Med. 1999, 220, 255–261. [Google Scholar] [CrossRef]
- Lu, B.; Wu, X.; Tie, X.; Zhang, Y.; Zhang, Y. Toxicology and safety of anti-oxidant of bamboo leaves. Part 1: Acute and subchronic toxicity studies on anti-oxidant of bamboo leaves. Food Chem. Toxicol. 2005, 43, 783–792. [Google Scholar] [CrossRef]
- Lu, B.; Wu, X.; Shi, J.; Dong, Y.; Zhang, Y. Toxicology and safety of antioxidant of bamboo leaves. Part 2: Developmental toxicity test in rats with antioxidant of bamboo leaves. Food Chem. Toxicol. 2006, 44, 1739–1743. [Google Scholar] [CrossRef]
- Choi, Y.J.; Lim, H.S.; Choi, J.S.; Shin, S.Y.; Bae, J.Y.; Kang, S.W.; Kang, I.J.; Kang, Y.H. Blockade of chronic high glucose-induced endothelial apoptosis by Sasa borealis bamboo extract. Exp. Biol. Med. (Maywood) 2008, 233, 580–591. [Google Scholar] [CrossRef]
- Lee, M.J.; Park, W.H.; Song, Y.S.; Lee, Y.W.; Song, Y.O.; Moon, G.S. Effect of bamboo culm extract on oxidative stress and genetic expression: Bamboo culm extract ameliorates cell adhesion molecule expression and NFkappaB activity through the suppression of the oxidative stress. Clin. Nutr. 2008, 27, 755–763. [Google Scholar] [CrossRef]
- Park, E.J.; Jhon, D.Y. Effects of bamboo shoot consumption on lipid profiles and bowel function in healthy young women. Nutrition 2009, 25, 723–728. [Google Scholar] [CrossRef]
- Zhang, Y.; Yao, X.; Bao, B.; Zhang, Y. Anti-fatigue activity of a triterpenoid-rich extract from Chinese bamboo shavings (Caulis bamfusae in taeniam). Phytother. Res. 2006, 20, 872–876. [Google Scholar] [CrossRef]
- Ko, B.S.; Jun, D.W.; Jang, J.S.; Kim, J.H.; Park, S. Effect of Sasa borealis and white lotus root and leaves on insulin action and secretion in vitro. Korea J. Food Sci. Technol. 2006, 38, 114–120. [Google Scholar]
- Kim, E.Y.; Jung, E.Y.; Lim, H.S.; Heo, Y.R. The effects of the Sasa borealis leaves extract on plasma adiponectin, restin, C-reactive protein and homocyteine levels in high fat diet-induced obese C57/BL6J mice. Korean J. Nutr. 2007, 40, 303–311. [Google Scholar]
- Park, H.S.; Lim, J.H.; Kim, H.J.; Choi, H.J.; Lee, I.S. Antioxidant flavone glycosides from the leaves of Sasa borealis. Arch. Pharm. Res. 2007, 30, 161–166. [Google Scholar] [CrossRef]
- Shibata, M.; Yamatake, M.; Sakamoto, M.; Kanamori, K.; Takagi, K.; Okabe, S. Pharmacological studies on bamboo grass. Nippon Yakurigaku Zassi 1975, 71, 481–485. [Google Scholar] [CrossRef]
- Singleton, V.L.; Rossi, J.R. Colorimetry of total phenolics with phosphomolybdic-phosphotungstic acid. Am. J. Enol. Vitic. 1965, 16, 144–158. [Google Scholar]
- Meda, A.; Lamien, C.E.; Romito, M.; Millogo, J.; Nacoulma, O.G. Determination of the total phenolic, flavonoid and proline contents in Burkina Fasan honey, as well as their radical scavenging activity. Food Chem. 2005, 91, 571–577. [Google Scholar] [CrossRef]
- Brand-Williams, W.; Cuvelier, M.E.; Berset, C. Use of a free radical method to evaluate antioxidant activity. LWT Food Sci. 1995, 28, 25–30. [Google Scholar] [CrossRef]
- Halliwell, B.; Gutteridge, J.M.C.; Arurma, O.I. The deoxyribose method: A simple “test-tube” assay for determination of rate constants for reactions of hydroxyl radicals. Anal. Biochem. 1987, 165, 215–219. [Google Scholar] [CrossRef]
- Re, R.; Pellegrini, N.; Proteggente, A.; Pannala, A.; Yang, M.; Rice-Evans, C. Antioxidant activity applying an improved ABTS radical cation decolorization assay. Free Radic. Biol. Med. 1999, 26, 1231–1237. [Google Scholar] [CrossRef]
- Beaton, M.D. Current treatment options for nonalcoholic fatty liver disease and nonalcoholic steatohepatitis. Can. J Gastroenterol. 2012, 26, 353–357. [Google Scholar]
- Hirako, S.; Kim, H.J.; Shimizu, S.; Chiba, H.; Matsumoto, A. Low-dose fish oil consumption prevents hepatic lipid accumulation in high cholesterol diet fed mice. J Agric. Food Chem. 2011, 59, 13353–13359. [Google Scholar] [CrossRef]
- Bradbury, M.W. Lipid metabolism and liver inflammation. I. Hepatic fatty acid uptake: Possible role in steatosis. Am. J. Physiol. Gastrointest. Liver Physiol. 2006, 290, G194–G198. [Google Scholar] [CrossRef]
- Cho, A.S.; Jeon, S.M.; Kim, M.J.; Yeo, J. Seo, K.I.; Choi, M.S.; Lee, M.K. Chlorogenic acid exhibits anti-obesity property and improves lipid metabolism in high-fat diet-induced-obese mice. Food Chem. Toxicol. 2010, 48, 937–943. [Google Scholar] [CrossRef]
- Kwon, H.J.; Kim, Y.Y.; Choung, S.Y. Amelioration effects of traditional Chinese medicine on alcohol-induced fatty liver. World J. Gastroenterol. 2005, 11, 5512–5516. [Google Scholar]
- Sekiya, M.; Yahagi, N.; Matsuzaka, T.; Najima, Y.; Nakakuki, M.; Nagai, R.; Ishibashi, S.; Osuga, J.; Yamada, N.; Shimano, H. Polyunsaturated fatty acids ameliorate hepatic steatosis in obese mice by SREBP-1 suppression. Hepatology 2003, 38, 1529–1539. [Google Scholar] [CrossRef]
- Shimano, H.; Horton, J.D.; Shimomura, I.; Hammer, R.E.; Brown, M.S.; Goldstein, J.L. Isoform 1c of sterol regulatory element binding protein is less active than isoform 1a in livers of transgenic mice and in cultured cells. J. Clin. Investig. 1997, 99, 846–854. [Google Scholar] [CrossRef]
- Yang, Z.X.; Shen, W.; Sun, H. Effects of nuclear receptor FXR on the regulation of liver lipid metabolism in patients with non-alcoholic fatty liver disease. Hepatol. Int. 2010, 4, 741–748. [Google Scholar] [CrossRef]
- Takahashi, N.; Senda, M.; Lin, S.; Goto, T.; Yano, M.; Sasaki, T.; Murakami, S.; Kawada, T. Auraptene regulates gene expression involved in lipid metabolism through PPARα activation in diabetic obese mice. Mol. Nutr. Food Res. 2011, 55, 1791–1797. [Google Scholar] [CrossRef]
- Nagasawa, T.; Inada, Y.; Nakano, S.; Tamura, T.; Takahashi, T.; Maruyama, K.; Yamazaki, Y.; Kuroda, J.; Shibata, N. Effects of bezafibrate, PPAR pan-agonist, and GW501516, PPARdelta agonist, on development of steatohepatitis in mice fed a methionine- and choline-deficient diet. Eur. J. Pharmacol. 2006, 536, 182–191. [Google Scholar] [CrossRef]
- Stone, S.J.; Myers, H.M.; Watkins, S.M.; Brown, B.E.; Feingold, K.R.; Elias, P.M.; Farese, R.V., Jr. Lipopenia and skin barrier abnormalities in DGAT2-deficient mice. J. Biol. Chem. 2004, 279, 11767–11776. [Google Scholar]
- Yamazaki, T.; Sasaki, E.; Kakinuma, C.; Yano, T.; Miura, S.; Ezaki, O. Increased very low density lipoprotein secretion and gonadal fat mass in mice overexpressing liver DGAT. J. Biol. Chem. 2005, 280, 21506–21514. [Google Scholar] [CrossRef]
- Sookoian, S.; Rosselli, M.S.; Gemma, C.; Burgueño, A.L.; Fernández Gianotti, T.; Castaño, G.O.; Pirola, C.J. Epigenetic regulation of insulin resistance in nonalcoholic fatty liver disease: Impact of liver methylation of the peroxisome proliferator-activated receptor γ coactivator 1α promoter. Hepatology 2010, 52, 1992–2000. [Google Scholar] [CrossRef]
- Fujiki, K.; Kano, F.; Shiota, K.; Murata, M. Expression of the peroxisome proliferator activated receptor gamma gene is repressed by DNA methylation in visceral adipose tissue of mouse models of diabetes. BMC Biol. 2009, 7, 38. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Song, Y.; Lee, S.-J.; Jang, S.-H.; Ha, J.H.; Song, Y.M.; Ko, Y.-G.; Kim, H.-D.; Min, W.; Kang, S.N.; Cho, J.-H. Sasa borealis Stem Extract Attenuates Hepatic Steatosis in High-Fat Diet-induced Obese Rats. Nutrients 2014, 6, 2179-2195. https://doi.org/10.3390/nu6062179
Song Y, Lee S-J, Jang S-H, Ha JH, Song YM, Ko Y-G, Kim H-D, Min W, Kang SN, Cho J-H. Sasa borealis Stem Extract Attenuates Hepatic Steatosis in High-Fat Diet-induced Obese Rats. Nutrients. 2014; 6(6):2179-2195. https://doi.org/10.3390/nu6062179
Chicago/Turabian StyleSong, Yuno, Soo-Jung Lee, Sun-Hee Jang, Ji Hee Ha, Young Min Song, Yeoung-Gyu Ko, Hong-Duck Kim, Wongi Min, Suk Nam Kang, and Jae-Hyeon Cho. 2014. "Sasa borealis Stem Extract Attenuates Hepatic Steatosis in High-Fat Diet-induced Obese Rats" Nutrients 6, no. 6: 2179-2195. https://doi.org/10.3390/nu6062179
APA StyleSong, Y., Lee, S.-J., Jang, S.-H., Ha, J. H., Song, Y. M., Ko, Y.-G., Kim, H.-D., Min, W., Kang, S. N., & Cho, J.-H. (2014). Sasa borealis Stem Extract Attenuates Hepatic Steatosis in High-Fat Diet-induced Obese Rats. Nutrients, 6(6), 2179-2195. https://doi.org/10.3390/nu6062179





