Vitamin D and Depression: A Systematic Review and Meta-Analysis Comparing Studies with and without Biological Flaws
Abstract
:1. Introduction
2. Methods
- the primary evidence for Vitamin D supplementation and depression from RCTs;
- the types of subjects, the dose of Vitamin D supplementation, the control interventions and the measures of outcome used;
- methodological quality of the studies;
- biological flaws in the study design, and
- estimates of the size of the effect.
2.1. Search Approach
2.2. Independence
2.3. Eligible Studies
2.4. Decision-Making
2.5. Critical Appraisal
2.6. Data Extraction
2.7. Biological Flaws
- inappropriate interventions (interventions that did not include Vitamin D), or
- interventions producing the opposite effect of that intended (interventions that included Vitamin D, but reduced the 25OHD level in the intervention group), or
- ineffective interventions that did not improving Vitamin D status (did not significantly change the 25OHD level), or
- where the baseline 25OHD level was not measured in the majority of participants, or
- where the baseline 25OHD level indicated sufficiency (not deficiency) at baseline.
2.8. Meta-Analysis
3. Results
3.1. Systematic Review
| Author | Year | Reference Citation # | Population | Sample Size | Quality Score |
|---|---|---|---|---|---|
| Arvold et al. | 2009 | [36] | Individuals with Vit D deficiency (10–25 ng/mL) seen for medical care at a primary healthcare clinic | 100 (I 50, C 50) | 10 |
| Belcaro et al. | 2010 | [42] | Menopausal women with signs of depression and mood disorder | 65 (I 33, C 32) | 8 |
| Bertone-Johnson et al. | 2012 | [38] | PostmenopausalWomen with depressive symptoms | 36,282 (I 18176, C 18106) | 11 |
| Dean et al. | 2011 | [45] | Young healthy adults (University students) | 128 (I 63, C 65) | 11 |
| Dumville et al. | 2006 | [43] | Older women with seasonal affective disorder | 2117 (I 912,C 1205) | 11 |
| Gloth et al. | 1999 | [44] | Adults with Season Affective Disorder | 15 (I 8,C 7) | 6.5 |
| Harris & Dawson-Hughes | 1993 | [30] | Women with seasonal affective disorder | 250 (I 125, C 125) | 5 |
| Jorde et al. | 2008 | [37] | Overweight and obese adults | 441 (IH 150, ILl 142, C 149) | 8 |
| Khajehei et al. | 2009 | [46] | University female students with premenstrual syndrome | 180 (IOes 60, I 60, C 60) | 9 |
| Khoraminya et al. | 2013 | [49] | Adults with major depressive disorder based on DSM-IV criteria, without psychosis | 40 (I 20, C 20) | 10 |
| Landsdowne & Provost | 1998 | [39] | Adults with seasonal affective disorder | 44 (I 22, C 22) | 8 |
| Sanders et al. | 2011 | [47] | Community dwelling older women with seasonal mood disorders | 2012 (I 1001, C 1011) | 11 |
| Veith et al. | 2004 | [40] | Adults with serum 25(OH)D <61 nmol/L in summer, expected to develop 25(OH)D concentrations <40 nmol/L by winter | 64 ( I 32, C 32) | 10 |
| Yalamanchilli & Gallagher | 2012 | [48] | Older post-menopausal women with depression | 488 (Ioes+Calcitrol 122, Ioes 122, Calcitrol 123, placebo 123 ) | 11 |
| Zhang et al. | 2011 | [41] | Hospitalized patients | 32 (I 17, C 15) | 9 |
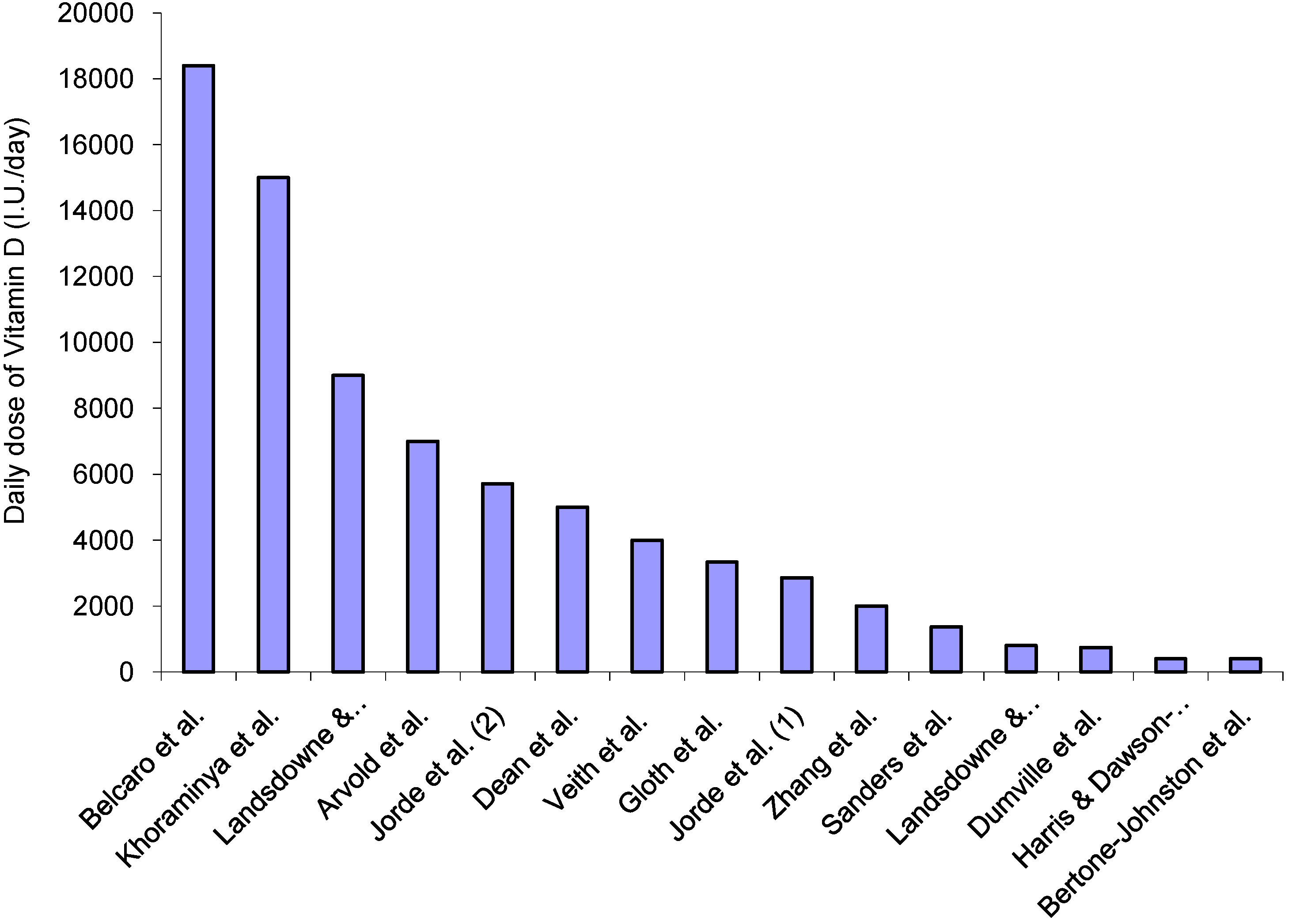
| Author | Year | Outcome Measures | Follow-up Time Period | Within Group Findings | Between Group Findings | |
|---|---|---|---|---|---|---|
| Arvold et al. | 2009 | Fibromyalgia Impact Questionnaire | 8 weeks | FIQ score Mean pre-post difference total (95%CI) intervention −3.71 (−7.5 to 0.1) (p < 0.03), control 1.91 (−2.9 to 6.7) (p > 0.05) | p < 0.05 favoring intervention | |
| Belcaro et al. | 2010 | Menopause Symptoms Questionnaire | 8 weeks | Total average symptom score reduced by 48% for intervention group (p < 0.05), control group increased by 10% (p > 0.05). | p < 0.05 favoring intervention | |
| Bertone-Johnson et al. | 2012 | Burnam Depression Scale | At 2 weeks, then twice yearly for 2 years | Mean overall change (SD) 0.004 (0.143) intervention, −0.002 (0.113) (control) | p > 0.05 | |
| Dean et al. | 2011 | Beck Depression Index | 6 weeks | Baseline: follow up mean (95%CI): Intervention 7.24 (5.58–8.90); 6.40 (4.73–8.07) (p > 0.05); control 5.72 (4.09–7.36); 5.38 (3.74–7.02) (p > 0.05) | p > 0.05 | |
| Dumville et al. | 2006 | SF12 mental component | 6 months | Mean difference (95%CI) between intervention and control at baseline −0.6 (−1.5 to 0.3) (p > 0.05); at follow up 1.8 (−0.8 to 1.2) (p > 0.05) | Mean adjusted (age- and baseline score) between group difference (95%CI) −0.49 (−1.34 to 0.81) p > 0.05 | |
| Gloth et al. | 1999 | SAD-8 | 1 month | Significant improvement in SAD-8 scores for intervention group, not control (explanatory data not provided) | Significant association between improvement in Vit D levels and SAD-8 scores in overall cohort (r2 = 0.26) | |
| Harris & Dawson-Hughes | 1993 | Profile of Mood States | 3 monthly for 12 months | No difference in pre-post scores for any domain of PoMS for either intervention or control (p > 0.05) | No difference between intervention or control change over time in any domain (p > 0.05) | |
| Jorde et al. | 2008 | Beck Depression Index (total score) | 12 months | Baseline: DD group 4.5 (0.0–24.0); DP group 5.0 (0.0–28.0); PP group 4.0 (0.0–24.0). Follow-up: DD group 3.0 (0.0–23.0) (p < 0.05); DP group 4.0 (0.0–26.0) (p < 0.05); PP group 3.8 (0.0–18.0) | DD and DP groups change was similar (p > 0.05) but significantly greater from PP (p < 0.05) | |
| Khajehei et al. | 2009 | PMS symptom rating form which captured psychological and physical symptoms including depression | Pre-mens for 2 cycles | Mean % total symptoms Pre: Dydrogesteron group 52.1%, Calcium plus Vitamin D group 50.7%, Placebo 53.7%. Post (respectively): 47.9%, 46.1%, 53.7%Both active treatment groups had significant decreases | The dydrogesterone and calcium plus Vitamin D treatments were significantly more effective than placebo in lessening the severity of PMS symptoms (p < 0.05) | |
| Khora-minya et al. | 2013 | 24-item Hamilton Depression Rating Scale (HDRS) (1°), 21-item Beck Depression Inventory (BDI) (2°) | Every 2 weeks for 8 weeks | BDI Intervention Wk0 32.45 ± 7.35; Wk2 27.73 ± 7.50; Wk4 20.44 ± 6.56; Wk6 16.73 ± 8.11; Wk8 13.2 ± 8.64 (p < 0.05) Control. Wk0 31.65 ± 7.33; Wk2 29.17 ± 6.78; Wk4 25.18 ± 6.93; Wk6 21.00 ± 6.81; Wk8 17.95 ± 6.31 (p < 0.05) | p < 0.05 for both outcomes, favoring intervention | |
| Lands-downe & Provost | 1998 | PANAS | 5 days | Sig within-group improvements for both active interventions (p < 0.05) | Sig improvements for both active interventions cf control for positive and negative affects (p < 0.05) | |
| Sanders et al. | 2011 | General Health Questionnaire SF12 (PCS, MCS), WHO Wellbeing Index | 3–5 years | Intervention: no intervention SF12 PCS effect size (95%CI) 0.27 (−2.40 to 2.94) 0.23 (−0.88 to 1.34) | Treatment effects SF12 effect size (95%CI) PCS 0.22 (−70.75 to 1.19); MCS 70.14 (−71.00 to 0.72) | |
| Veith et al. | 2004 | Self-developed Wellbeing Scale | 2–6 months | Pre-post mean (SD): 600 I.U. 2.2 (2.0); 2.3 (2.3) (p > 0.05) 4000 I.U. 2.0 (2.3); 1.1 (1.8) (p < 0.05) | Significant improvement in wellbeing, favoring higher Vit D dose | |
| Yalamanchilli & Gallagher | 2012 | Geriatric Depression Scale | 1. HT alone 2. calcitriol alone 3. HT & calcitrol 4. placebo | % with depression (pre/post) 13.8%; 8.9%; 9.7%; 7.3%; 8.2%; 6.6% 13.8%; 8.9% All groups p > 0.05 | No effect on depression in any treatment group compared with placebo (p > 0.05) | |
| Zhang et al. | 2011 | Profile of Mood States questionnaire | Average 8 days | Vit D group pre-post 23.1 ± 27.2; 22.4 ± 22.4 p > 0.05 Vit C group pre-post 28.6 ± 21.8; 18.8 ± 19.4) p < 0.05 | p < 0.05 favouring Vit D | |
3.2. Biological Flaws
| Study | Biological Flaws NOT Present | Biological Flaw(s) Present | Type of Flaw | Quality Score (Max 11) | Date of Publication | ||
|---|---|---|---|---|---|---|---|
| 25OHD not Assessed | Dose not Appropriate | ||||||
| Belcaro et al. | X | X | 8 | 2010 | |||
| Bertone-Johnson et al. | X | X | X (L) | 11 | 2012 | ||
| Dumville et al. | X | X | 11 | 2006 | |||
| Harris & Dawson-Hughes | X | X | X (L) | 5 | 1993 | ||
| Dean et al. | X | X | X (H) | 11 | 2011 | ||
| Khajehei et al. | X | X (I) | 9 | 2009 | |||
| Sanders et al. | X | X (SE) | 11 | 2011 | |||
| Yalamanchilli & Gallagher | X | X (I) | 11 | 2012 | |||
| Total-8 Studies with Biological Flaws | 0 | 8 | 5 | 6 | 3 | 5 | |
| Arvold et al. | X | 10 | 2009 | ||||
| Gloth et al. | X | 6.5 | 1999 | ||||
| Jorde et al. | X | 8 | 2008 | ||||
| Khoraminya et al. | X | 10 | 2013 | ||||
| Landsdowne & Provost | X | 8 | 1998 | ||||
| Veith et al. | X | 10 | 2004 | ||||
| Zhang et al. | X | 9 | 2011 | ||||
| Total—7 studies without flaws | 7 | 0 | 0 | 0 | 5 | 2 | |
3.3. Meta-Analysis
3.3.1. Meta-Analysis of Studies without Biological Flaws (Right Panel of Figure 2)
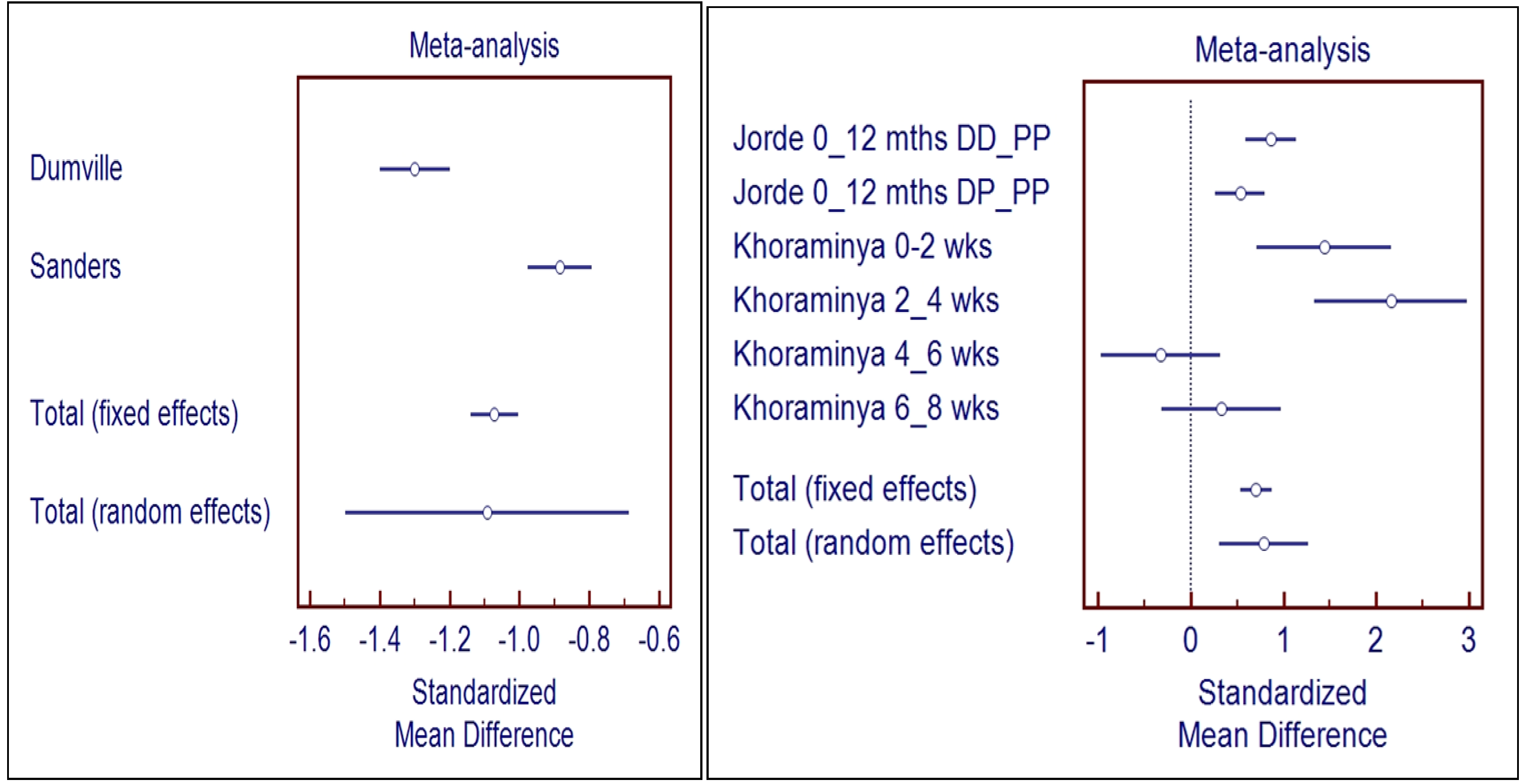
3.3.2. Meta-Analysis of Studies with Biological Flaws (Left Panel of Figure 2)
4. Discussion
5. Conclusion
Acknowledgments
References
- Hyman, S.; Chisholm, D.; Kessler, R.; Patel, V.; Whiteford, H. Mental disorders. In Disease Control Priorities in Developing Countries, 2nd ed.; Jamison, D.T., Breman, J.G., Measham, A.R., Alleyne, G., Claeson, M., Evans, D.B., Jha, P., Mills, A., Musgrove, P., Eds.; Oxford University Press: New York, NY, USA, 2006; pp. 605–626. [Google Scholar]
- Bracken, P.; Thomas, P.; Timimi, S. Psychiatry beyond the current paradigm. Br. J. Psychiat. 2012, 201, 430–434. [Google Scholar] [CrossRef]
- Jordanes. The Origin and Deeds of the Goths; Mierow, C.C., Ed.; Princeton University Press: Princeton, NJ, USA, 2012; pp. 19–21. Available online: http://people.ucalgary.ca/~vandersp/Courses/texts/jordgeti.html (accessed on 8 April 2014).
- Holick, M.F. The Vitamin D deficiency pandemic: A forgotten hormone important for health. Public Health Rev. 2010, 32, 267–283. [Google Scholar]
- Hollick, M.F. Vitamin D deficiency. N. Engl. J. Med. 2007, 357, 266–281. [Google Scholar] [CrossRef]
- Daly, R.M.; Gagnon, C.; Lu, Z.X.; Magliano, D.J.; Dunstan, D.W.; Sikaris, K.A.; Zimmet, P.Z.; Ebeling, P.R.; Shaw, J.E. Prevalence of Vitamin D deficiency and its determinants in Australian adults aged 25 years and older: A national, population-based study. Clin. Endocrinol. 2011, 77, 26–35. [Google Scholar]
- Munns, C.F.; Simm, P.J.; Rodda, C.P. Incidence of Vitamin D deficiency rickets among Australian children: An Australian Paediatric Surveillance Unit study. Med. J. Aust. 2012, 196, 466–468. [Google Scholar] [CrossRef]
- Thacher, T.D.; Fischer, P.R.; Strand, M.A.; Pettifor, J.M. Nutritional rickets around the world: Causes and future directions. Ann. Trop. Paediatr. 2006, 26, 1–16. [Google Scholar] [CrossRef]
- Ginde, A.A.; Liu, M.C.; Camargo, C.A. Vitamin D deficiency seems to be increasing in the US population. Arch. Intern. Med. 2009, 169, 616–32. [Google Scholar] [CrossRef]
- Maxmen, A. Nutrition advice: The Vitamin D-lemma. A vociferous debate about vitamin-D supplementation reveals the difficulty of distilling strong advice from weak evidence. Nature 2011, 475, 23–25. [Google Scholar] [CrossRef]
- Scragg, R. Vitamin D and public health: An overview of recent research on common diseases and mortality in adulthood. Public Health Nutr. 2011, 14, 1515–1532. [Google Scholar] [CrossRef]
- Heaney, R.P. Does inconclusive evidence for Vitamin D supplementation to reduce risk for cardiovascular disease warrant pessimism? [Letter to the editor]. Ann. Intern. Med. 2010, 153, 208–209. [Google Scholar] [CrossRef]
- Spedding, S. Vitamins are more adaptable than Casimir Funk thought. Australasian J. Med. 2013, in press. [Google Scholar]
- Hendrix, I.; Anderson, P.; May, B.; Morris, H. Regulation of gene expression by the CYP27B1 promoter—study of a transgenic model. J. Steroid Biochem. Mol. Biol. 2004, 89–90, 139–142. [Google Scholar] [CrossRef]
- Ramagopalan, S.V.; Heger, A.; Berlanga, A.J.; Maugeri, N.J.; Lincoln, M.R.; Burrell, A. A ChIP-seq defined genome-wide map of Vitamin D receptor binding: Associations with disease and evolution. Genome Res. 2010, 20, 1352–1360. [Google Scholar] [CrossRef]
- Obradovic, D.; Gronemeyer, H.; Lutz, B.; Rein, T. Cross-talk of Vitamin D and glucocorticoids in hippocampal cells. J. Neurochem. 2006, 96, 500–509. [Google Scholar] [CrossRef]
- Puchacz, E.; Stumpf, W.; Stachowiak, E.K.; Stachowiak, M.K. Vitamin D increases expression of the tyrosine hydroxylase gene in adrenal medullary cells. Mol. Brain Res. 1996, 36, 193–196. [Google Scholar] [CrossRef]
- Cass, W.A.; Smith, M.P.; Peters, L.E. Calcitriol protects against the dopamine- and serotonin-depleting effects of neurotoxic doses of methamphetamine. Ann. N. Y. Acad. Sci. 2006, 1074, 261–271. [Google Scholar]
- Ganji, V.; Milone, C.; Cody, M.; McCarty, F.; Wang, Y.T. Serum Vitamin D concentrations are related to depression in young adult US population: The Third National Health and Nutrition Examination Survey. Int. Arch. Med. 2010, 3, 29. [Google Scholar] [CrossRef]
- May, H.T.; Bair, T.L.; Lappé, D.L.; Anderson, J.L.; Horne, B.D.; Carlquist, J.F.; Muhlestein, J.B. Association of Vitamin D levels with incident depression among a general cardiovascular population. Am. Heart J. 2010, 159, 1037–1043. [Google Scholar] [CrossRef]
- Hoang, M.T.; DeFina, L.F.; Willis, B.L. Association between low serum 25-hydroxyVitamin D and depression in a large sample of healthy adults: The Cooper Center Longitudinal Study. Mayo Clin. Proc. 2011, 86, 1050–1055. [Google Scholar] [CrossRef]
- Kjærgaard, M.; Joakimsen, R.; Jorde, R. Low serum 25-hydroxyVitamin D levels are associated with depression in an adult Norwegian population. Psychiatry Res. 2011, 190, 221–225. [Google Scholar] [CrossRef]
- Umhau, J.C.; George, D.T.; Heaney, R.P.; Lewis, M.D.; Ursano, R.J. Low Vitamin D status and suicide: A case-control study of active duty military service members. PLoS One 2013, 8, e51543. [Google Scholar]
- Li, G.; Mbuagbaw, L.; Samaan, Z.; Zhang, S.; Adachi, J.D.; Papaioannou, A. ThabaneL Efficacy of vitamin D supplementation in depression in adults: a systematic review protocol. Syst. Rev. 2013, 2, 64. [Google Scholar] [CrossRef]
- Institute of Medicine. Dietary Reference Intakes for Calcium and Vitamin D; The National Academics Press: Washington DC, WA, USA, 2011. [Google Scholar]
- Morgan, A.; Jorm, A. Self-help interventions for depressive disorders and depressive symptoms: A systematic review. Ann. Gen. Psychiatry 2008, 7, 13. [Google Scholar] [CrossRef]
- Murphy, P.K.; Wagner, C.L. Vitamin D and mood disorders among women: An integrative review. J. Midwifery Womens Health 2008, 53, 440–446. [Google Scholar] [CrossRef]
- Barnard, K.; Colon-Emeric, C. Extraskeletal effects of Vitamin D in older adults: Cardiovascular disease, mortality, mood, and cognition. Am. J. Geriatr. Pharmacother. 2010, 8, 4–33. [Google Scholar] [CrossRef]
- Thys-Jacobs, S.; Starkey, P.; Bernstein, D.; Tian, J. Calcium carbonate and the premenstrual syndrome: Effects on premenstrual and menstrual symptoms. Premenstrual Syndrome Study Group. Am. J. Obstet. Gynecol. 1998, 179, 444–452. [Google Scholar] [CrossRef]
- Harris, S.; Dawson-Hughes, B. Seasonal mood changes in 250 normal women. Psychiatry Res. 1993, 49, 77–87. [Google Scholar] [CrossRef]
- Scragg, R. Do we need to take calcium with Vitamin D supplements to prevent falls, fractures, and death? Cur. Opin. Clin. Nutr. Metab. Care 2012, 15, 614–624. [Google Scholar] [CrossRef]
- Biesalski, H.K.; Aggett, P.J.; Anton, R.; Bernstein, P.S.; Blumberg, J.; Heaney, R.P. Scientific substantiation of health claims: Evidence-based nutrition. 26th Hohenheim Consensus Conference. Nutrition 2011, 27, S1–S20. [Google Scholar] [CrossRef]
- Lappe, J.M.; Heaney, R.P. Why randomized controlled trials of calcium and Vitamin D sometimes fail. Dermato Endocrinol. 2012, 4, 95–100. [Google Scholar] [CrossRef]
- Heaney, R.P. Vitamin D—Baseline status and effective dose. [Letters to the editor]. N. Engl. J. Med. 2012, 367, 1. [Google Scholar] [CrossRef]
- Sherrington, C.; Herbert, R.D.; Maher, C.G.; Moseley, A.M. PEDro. A database of randomized trials and systematic reviews in physiotherapy. Man. Ther. 2000, 5, 223–226. [Google Scholar] [CrossRef]
- Arvold, D.S.; Odean, M.J.; Dornfeld, M.P.; Regal, R.R.; Arvold, J.G.; Karwoski, G.C.; Mast, D.J.; Sanford, P.B.; Sjoberg, R.J. Correlation of symptoms with Vitamin D deficiency and symptom response to cholecalciferol treatment: A randomized controlled trial. Endocr. Pract. 2009, 15, 203–212. [Google Scholar] [CrossRef]
- Jorde, R.; Sneve, M.; Figenschau, Y.; Svartberg, J.; Waterloo, K. Effects of Vitamin D supplementation on symptoms of depression in overweight and obese subjects: Randomized double blind trial. J. Intern. Med. 2008, 264, 599–609. [Google Scholar] [CrossRef]
- Bertone-Johnson, E.R.; Powers, S.I.; Spangler, L.; Larson, J.; Michael, Y.L.; Millen, A.E.; Bueche, M.N.; Salmoirago-Blotcher, E.; Wassertheil-Smoller, S.; Brunner, R.L. Vitamin D supplementation and depression in the women’s health initiative calcium and Vitamin D trial. Am. J. Epidemiol. 2012, 176, 1–13. [Google Scholar] [CrossRef]
- Lansdowne, A.T.; Provost, S.C. Vitamin D3 enhances mood in healthy subjects during winter. Psychopharmacol. 1998, 135, 319–323. [Google Scholar] [CrossRef]
- Veith, R.; Kimball, S.; Hu, A.; Walfish, P.G. Randomized comparison of the effects of the Vitamin D3 adequate intake versus 100 mcg (4000 IU) per day on biochemical responses and the wellbeing of patients. Nutr. J. 2004, 3, 8. [Google Scholar] [CrossRef]
- Zhang, M.; Robitaille, L.; Eintracht, S.; Hoffer, L.J. Vitamin C provision improves mood in acutely hospitalized patients. Nutrition 2011, 27, 530–533. [Google Scholar] [CrossRef]
- Belcaro, G.; Cesarone, M.R.; Cornelli, U.; Dugall, M. MF Afragil® in the treatment of 34 menopause symptoms: A pilot study. Panminerva Med. 2010, 52, 49–54. [Google Scholar]
- Dumville, J.C.; Miles, J.N.; Porthouse, J.; Cockayne, S.; Saxon, L.; King, C. Can Vitamin D supplementation prevent winter-time blues? A randomised trial among older women. J. Nutr. Health Aging 2006, 10, 151–153. [Google Scholar]
- Gloth, F.M., III; Alam, W.; Hollis, B. Vitamin D vs. broad spectrum phototherapy in the treatment of seasonal affective disorder. J. Nutr. Health Aging 1999, 3, 5–7. [Google Scholar]
- Dean, A.J.; Bellgrove, M.A.; Hall, T.; Phan, W.M.; Eyles, D.W.; Kvaskoff, D.; McGrath, J.J. Effects of Vitamin D supplementation on cognitive and emotional functioning in young adults—A randomised controlled trial. PLoS One 2011, 6, e25966. [Google Scholar]
- Khajehei, M.; Abdali, K.; Parsanezhad, M.E.; Tabatabaee, H.R. Effect of treatment with dydrogesterone or calcium plus Vitamin D on the severity of premenstrual syndrome. Int. J. Gynecol. Obstet. 2009, 105, 158–161. [Google Scholar] [CrossRef]
- Sanders, K.M.; Stuart, A.L.; Williamson, E.J.; Jacka, F.N.; Dodd, S.; Nicholson, G.; Berk, M. Annual high-dose Vitamin D3 and mental well-being: randomised controlled trial. Br. J. Psychiatry 2011, 198, 357–364. [Google Scholar] [CrossRef]
- Yalamanchili, V.V.; Gallagher, J.C. Treatment with hormone therapy and calcitriol did not affect depression in older postmenopausal women: No interaction with estrogen and Vitamin D receptor genotype polymorphisms. Menopause 2012, 19, 697–703. [Google Scholar] [CrossRef]
- Khoraminya, N.; Tehrani-Doost, M.; Jazayeri, S.; Hosseini, A.; Djazayery, A. Therapeutic effects of Vitamin D as adjunctive therapy to fluoxetine in patients with major depressive disorder. Aust. N.Z. J. Psychiatry 2013, 47, 271–275. [Google Scholar] [CrossRef]
- Dawson-Hughes, B.; Dallal, G.E.; Krall, E.A.; Harris, S.; Sokoll, L.J.; Falconer, G. Effect of Vitamin D supplementation on wintertime and overall bone loss in healthy postmenopausal women. Ann. Intern. Med. 1991, 115, 505–512. [Google Scholar] [CrossRef]
- Gallagher, J.C.; Sai, A.; Templin, T., II; Smith, L. Dose response to Vitamin D supplementation in postmenopausal women: A randomized trial. Ann. Intern. Med. 2012, 156, 425–437. [Google Scholar] [CrossRef]
- Luppino, F.S.; Wit, L.M.; Bouvy, P.F. Overweight, obesity and depression. A systematic review and meta-analysis of longitudinal studies. Arch. Gen. Psychiatry 2010, 67, 220–229. [Google Scholar] [CrossRef]
- Kendrick, T.; Chatwin, J.; Dowrick, C.; Tylee, A.; Morriss, R.; Peveler, R. Randomised controlled trial to determine the clinical effectiveness and cost-effectiveness of selective serotonin reuptake inhibitors plus supportive care, versus supportive care alone, for mild to moderate depression with somatic symptoms in primary care: The THREAD (THREshold for AntiDepressant response) study. Health Technol. Assess. 2009, 13, 1–159. [Google Scholar]
- Turner, E.H.; Matthews, A.M.; Linardatos, E.; Tell, R.; Rosenthal, R. Selective publication of antidepressant trials and its influence on apparent efficacy. N. Engl. J. Med. 2008, 358, 252–260. [Google Scholar] [CrossRef]
- Maddock, J.; Berry, D.J.; Geoffroy, M.-C.; Power, C.; Hyppönen, E. Vitamin D and common mental disorders in mid-life: Cross-sectional and prospective findings. Clin. Nutr. 2013, 32, 758–764. [Google Scholar] [CrossRef]
- Bolland, M.J.; Grey, A.; Reid, I.R. Time for a moratorium on meta-analyses of Vitamin D? Letter to the editor. Br. Med. J. 2009, 339, 987. [Google Scholar]
- Moser, U. Vitamins—Wrong approaches. Int. J. Vitamin Nutr. Res. 2012, 82, 327–332. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Spedding, S. Vitamin D and Depression: A Systematic Review and Meta-Analysis Comparing Studies with and without Biological Flaws. Nutrients 2014, 6, 1501-1518. https://doi.org/10.3390/nu6041501
Spedding S. Vitamin D and Depression: A Systematic Review and Meta-Analysis Comparing Studies with and without Biological Flaws. Nutrients. 2014; 6(4):1501-1518. https://doi.org/10.3390/nu6041501
Chicago/Turabian StyleSpedding, Simon. 2014. "Vitamin D and Depression: A Systematic Review and Meta-Analysis Comparing Studies with and without Biological Flaws" Nutrients 6, no. 4: 1501-1518. https://doi.org/10.3390/nu6041501
APA StyleSpedding, S. (2014). Vitamin D and Depression: A Systematic Review and Meta-Analysis Comparing Studies with and without Biological Flaws. Nutrients, 6(4), 1501-1518. https://doi.org/10.3390/nu6041501




