Effect of Diets Differing in Glycemic Index and Glycemic Load on Cardiovascular Risk Factors: Review of Randomized Controlled-Feeding Trials
Abstract
:1. Introduction
2. Methods
2.1. Search Strategy
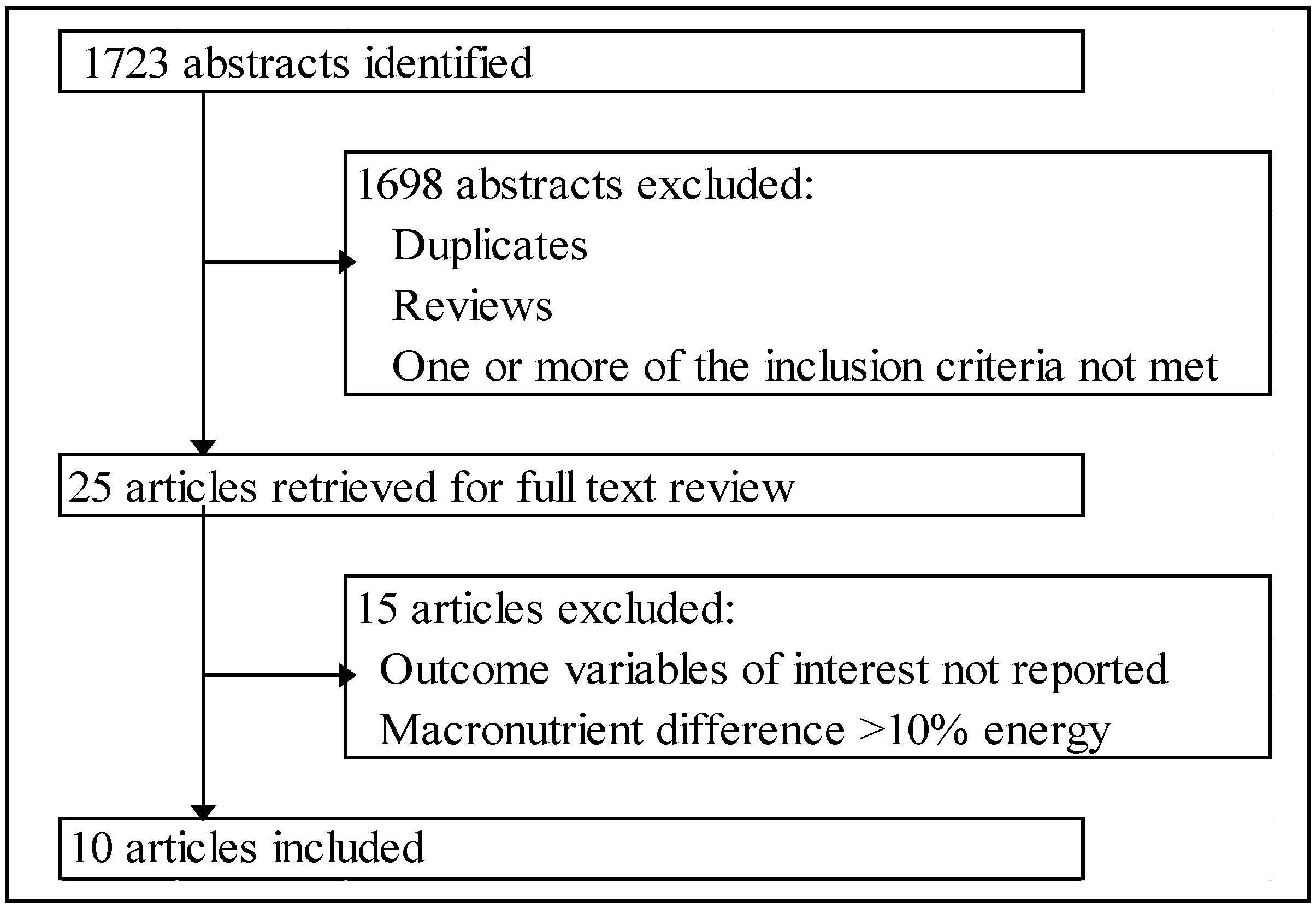
2.2. Inclusion Criteria
3. Results
3.1. Included Studies
| Trial | Reference | Design | Intervention | Duration, Weeks (Washout) | Sex ( n) | Participants | Age, Years, Mean (SD) or Range | BMI (SD) |
|---|---|---|---|---|---|---|---|---|
| 1 | [12] | R-X | LGI/GL-HGI/GL | 4 (4) | M (24) | Ob | 34.5 (8.1) | 29.5 (4.3) |
| 2 | [13] | R-X | LGI-HGI | 4 (~2) | M (64) | IS + IR | 54.5 (7.8) | 28.7 (3.5) |
| 2 | [14] | R-X | LGI-HGI | 4 (~2) | M (64) | IS + IR | 53.5 (7.6), IS | 27.4 (3.2), IS |
| 55.5 (8.0), IR | 30.3 (3.2), IR | |||||||
| 3 | [15] | R-X | LGL-HGL | 4 (4) | M (40), F (40) | NW + OW/Ob | 29.6 (8.2) | 27.4 (5.9) |
| 3 | [16] | R-X | LGL-HGL | 4 (4) | M (9), F (7) | NW + OW/Ob | 19–44 | 18.5–25, NW |
| 28–40, OW/Ob | ||||||||
| 3 | [17] | R-X | LGL-HGL | 4 (4) | M (40), F (40) | NW + OW/Ob | 18–45 | 27.5 (5.9) |
| 4 | [18] | R-P | LGI-HGI | 12 | M (5), F (14) | Ob | 18–70 | 30–40 |
| 5 | [19] | R-P | (LGI + E)-(HGI + E) | 12 | M (8), F (14) | Ob + prediabetic | 66 (1) | 34.4 (2.8) |
| 5 | [20] | R-P | (LGI + E)-(HGI + E) | 12 | M (13), F (15) | Ob + IR | 66 (1) | 34.2 (0.7) |
| 5 | [21] | R-P | (LGI + E)-(HGI + E) | 12 | M (11), F (10) | MetS | 66.2 (1.1) | 35.3 (0.9) |
| Trial | Reference | GI | GL | Carbohydrate | Fat | Protein | Fiber | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| %Energy | |||||||||||||
| low | high | low | high | low | high | low | high | low | high | low | high | ||
| 1 | [12] | 50 | 75 | 158 | 246 | 55 | 56 | 29 | 30 | 18 | 16 | 23 a | 21 a |
| 2 | [13,14] | 38 | 69 | 84 | 152 | 50 | 50 | 34 | 34 | 18 | 18 | 21 b | 9 b |
| 3 | [15,16,17] | 34 | 78 | ≤125 | ≥250 | 55 | 55 | 30 | 30 | 15 | 15 | 49 a | 24 a |
| 4 | [18] | 33 | 63 | 178 | 272 | 60 | 60 | 25 | 25 | 15 | 15 | 17 b | 9 b |
| 5 | [19,20,21] | 40 | 80 | 102 | 218 | 56 | 58 | 32 | 32 | 17 | 17 | 28 a | 28 a |
3.2. Outcome Variables
3.2.1. Glucose Homeostasis
| Trial | Reference | Participants | Glucose Homeostasis Markers | Lipid Profile | Inflammatory Markers | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Glc | Ins | %HbA1c | Glc OGTT | Ins OGTT | TC | LDL | HDL | TG | CRP | IL-6 | TNFα | |||
| Cross-Over Studies | ||||||||||||||
| 1 | [12] | Ob | ↔ | ↔ | – | – | – | ↑ | ↑ | ↑ | ↔ | ↔ | ↔ | ↔ |
| 2 | [13] | IS/IR | ↑ | ↔ | – | – | – | – | – | – | – | ↔ | – | – |
| IS | ↔ | ↔ | ↔ | |||||||||||
| IR | ↑ | ↔ | ↔ | |||||||||||
| 2 | [14] | IS/IR | – | – | – | – | – | ↓ | ↓ | ↓ | ↔ | – | – | – |
| IS | ↓ | ↓ | ↔ | ↔ | ||||||||||
| IR | ↔ | ↓ | ↓ | ↔ | ||||||||||
| 3 | [15] | LBF/HBF | ↑ | ↔ | – | – | – | – | – | – | – | – | – | – |
| LBF | ↔ | ↔ | ||||||||||||
| HBF | ↑ | ↔ | ||||||||||||
| 3 | [16] | LBF/HBF | ↔ | ↔ | – | – | – | – | – | – | – | – | – | – |
| LBF | ↔ | ↔ | ||||||||||||
| HBF | ↔ | ↔ | ||||||||||||
| 3 | [17] | LBF/HBF | – | – | – | – | – | – | – | – | – | ↔ | ↔ | – |
| LBF | ↔ | ↑ | ||||||||||||
| HBF | ↓ | ↔ | ||||||||||||
| Parallel Studies | ||||||||||||||
| 4 | [18] | Ob | ↔ | ↔ | – | – | – | – | – | – | ↔ | – | – | – |
| 5 | [19] | Ob | ↔ | ↔ | ↔ | ↓ | ↓ | ↔ | ↔ | ↔ | ↔ | – | – | – |
| 5 | [20] | Ob | ↔ | ↔ | ↔ | ↔ | ↔ | – | – | – | – | – | ↓ | ↓ |
| 5 | [21] | Ob | ↔ | ↔ | ↔ | – | – | ↔ | ↔ | ↔ | ↔ | – | – | – |
3.2.2. Lipid Profile
3.2.3. Inflammatory Markers
3.2.4. Additional Variables
4. Discussion
5. Conclusions
Conflict of Interest
References
- Crapo, P.A.; Reaven, G.; Olefsky, J. Plasma glucose and insulin responses to orally administered simple and complex carbohydrates. Diabetes 1976, 25, 741–747. [Google Scholar]
- Crapo, P.A.; Reaven, G.; Olefsky, J. Postprandial plasma-glucose and -insulin responses to different complex carbohydrates. Diabetes 1977, 26, 1178–1183. [Google Scholar]
- Jenkins, D.J.; Wolever, T.M.; Taylor, R.H.; Barker, H.; Fielden, H.; Baldwin, J.M.; Bowling, A.C.; Newman, H.C.; Jenkins, A.L.; Goff, D.V. Glycemic index of foods: A physiological basis for carbohydrate exchange. Am. J. Clin. Nutr. 1981, 34, 362–366. [Google Scholar]
- Salmeron, J.; Ascherio, A.; Rimm, E.B.; Colditz, G.A.; Spiegelman, D.; Jenkins, D.J.; Stampfer, M.J.; Wing, A.L.; Willett, W.C. Dietary fiber, glycemic load, and risk of NIDDM in men. Diabetes Care 1997, 20, 545–550. [Google Scholar]
- Pi-Sunyer, X. Do glycemic index, glycemic load, and fiber play a role in insulin sensitivity, disposition index, and type 2 diabetes? Diabetes Care 2005, 28, 2978–2979. [Google Scholar] [CrossRef]
- Franz, M.J. Is there a role for the glycemic index in coronary heart disease prevention or treatment? Curr. Atheroscler. Rep. 2008, 10, 497–502. [Google Scholar] [CrossRef]
- Dong, J.Y.; Zhang, Y.H.; Wang, P.; Qin, L.Q. Meta-analysis of dietary glycemic load and glycemic index in relation to risk of coronary heart disease. Am. J. Cardiol. 2012, 109, 1608–1613. [Google Scholar] [CrossRef]
- Brand-Miller, J.; Buyken, A.E. The glycemic index issue. Curr. Opin. Lipidol. 2012, 23, 62–67. [Google Scholar] [CrossRef]
- Opperman, A.M.; Venter, C.S.; Oosthuizen, W.; Thompson, R.L.; Vorster, H.H. Meta-analysis of the health effects of using the glycaemic index in meal-planning. Br. J. Nutr. 2004, 92, 367–381. [Google Scholar] [CrossRef]
- Kelly, S.; Frost, G.; Whittaker, V.; Summerbell, C. Low glycaemic index diets for coronary heart disease. Cochrane Database of Syst. Rev. 2004, 4, CD004467. [Google Scholar]
- Goff, L.M.; Cowland, D.E.; Hooper, L.; Frost, G.S. Low glycaemic index diets and blood lipids: A systematic review and meta-analysis of randomised controlled tria. Nutr. Metab. Cardiovasc. Dis. 2013, 23, 1–10. [Google Scholar] [CrossRef] [Green Version]
- Shikany, J.M.; Phadke, R.P.; Redden, D.T.; Gower, B.A. Effects of low- and high-glycemic index/glycemic load diets on coronary heart disease risk factors in overweight/obese men. Metabolism 2009, 58, 1793–1801. [Google Scholar] [CrossRef]
- Hartman, T.J.; Albert, P.S.; Zhang, Z.; Bagshaw, D.D.; Kris-Etherton, P.M.; Ulbrecht, J.; Miller, C.K.; Bobe, G.; Colburn, N.H.; Lanza, E. Consumption of a legume-enriched, low-glycemic index diet is associated with biomarkers of insulin resistance and inflammation among men at risk for colorectal cancer. J. Nutr. 2010, 140, 60–67. [Google Scholar] [CrossRef]
- Zhang, Z.; Lanza, E.; Kris-Etherton, P.M.; Colburn, N.H.; Bagshaw, D.; Rovine, M.J.; Ulbrecht, J.S.; Bobe, G.; Chapkin, R.S.; Hartman, T.J. A high legume low glycemic index diet improves serum lipid profiles in men. Lipids 2010, 45, 767–775. [Google Scholar]
- Runchey, S.S.; Pollak, M.N.; Valsta, L.M.; Coronado, G.D.; Schwarz, Y.; Breymeyer, K.L.; Wang, C.; Wang, C.Y.; Lampe, J.W.; Neuhouser, M.L. Glycemic load effect on fasting and post-prandial serum glucose, insulin, IGF-1 and IGFBP-3 in a randomized, controlled feeding study. Eur. J. Clin. Nutr. 2012, 66, 1146–1152. [Google Scholar] [CrossRef]
- Runchey, S.S.; Valsta, L.M.; Schwarz, Y.; Wang, C.; Song, X.; Lampe, J.W.; Neuhouser, M.L. Effect of low- and high-glycemic load on circulating incretins in a randomized clinical trial. Metabolism 2013, 62, 188–195. [Google Scholar] [CrossRef]
- Neuhouser, M.L.; Schwarz, Y.; Wang, C.; Breymeyer, K.; Coronado, G.; Wang, C.Y.; Noar, K.; Song, X.; Lampe, J.W. A low-glycemic load diet reduces serum C-reactive protein and modestly increases adiponectin in overweight and obese adults. J. Nutr. 2012, 142, 369–374. [Google Scholar] [CrossRef]
- Raatz, S.K.; Torkelson, C.J.; Redmon, J.B.; Reck, K.P.; Kwong, C.A.; Swanson, J.E.; Liu, C.; Thomas, W.; Bantle, J.P. Reduced glycemic index and glycemic load diets do not increase the effects of energy restriction on weight loss and insulin sensitivity in obese men and women. J. Nutr. 2005, 135, 2387–2391. [Google Scholar]
- Solomon, T.P.; Haus, J.M.; Kelly, K.R.; Cook, M.D.; Filion, J.; Rocco, M.; Kashyap, S.R.; Watanabe, R.M.; Barkoukis, H.; Kirwan, J.P. A low-glycemic index diet combined with exercise reduces insulin resistance, postprandial hyperinsulinemia, and glucose-dependent insulinotropic polypeptide responses in obese, prediabetic humans. Am. J. Clin. Nutr. 2010, 92, 1359–1368. [Google Scholar] [CrossRef]
- Kelly, K.R.; Haus, J.M.; Solomon, T.P.; Patrick-Melin, A.J.; Cook, M.; Rocco, M.; Barkoukis, H.; Kirwan, J.P. A low-glycemic index diet and exercise intervention reduces TNF(alpha) in isolated mononuclear cells of older, obese adults. J. Nutr. 2011, 141, 1089–1094. [Google Scholar] [CrossRef]
- Malin, S.K.; Niemi, N.; Solomon, T.P.; Haus, J.M.; Kelly, K.R.; Filion, J.; Rocco, M.; Kashyap, S.R.; Barkoukis, H.; Kirwan, J.P. Exercise training with weight loss and either a high- or low-glycemic index diet reduces metabolic syndrome severity in older adults. Ann. Nutr. Metab. 2012, 61, 135–141. [Google Scholar] [CrossRef]
- Fan, J.; Song, Y.; Wang, Y.; Hui, R.; Zhang, W. Dietary glycemic index, glycemic load, and risk of coronary heart disease, stroke, and stroke mortality: A systematic review with meta-analysis. PLoS One 2012, 7, e52182. [Google Scholar]
- Bohan Brown, M.M.; Brown, A.W.; Allison, D.B. Nutritional epidemiology in practice: Learning from data or promulgating beliefs? Am. J. Clin. Nutr. 2013, 97, 5–6. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Kristo, A.S.; Matthan, N.R.; Lichtenstein, A.H. Effect of Diets Differing in Glycemic Index and Glycemic Load on Cardiovascular Risk Factors: Review of Randomized Controlled-Feeding Trials. Nutrients 2013, 5, 1071-1080. https://doi.org/10.3390/nu5041071
Kristo AS, Matthan NR, Lichtenstein AH. Effect of Diets Differing in Glycemic Index and Glycemic Load on Cardiovascular Risk Factors: Review of Randomized Controlled-Feeding Trials. Nutrients. 2013; 5(4):1071-1080. https://doi.org/10.3390/nu5041071
Chicago/Turabian StyleKristo, Aleksandra S., Nirupa R. Matthan, and Alice H. Lichtenstein. 2013. "Effect of Diets Differing in Glycemic Index and Glycemic Load on Cardiovascular Risk Factors: Review of Randomized Controlled-Feeding Trials" Nutrients 5, no. 4: 1071-1080. https://doi.org/10.3390/nu5041071





