Diurnal Triglyceridemia in Relation to Alcohol Intake in Men
Abstract
:1. Introduction
2. Experimental Section
2.1. Subjects
2.2. Analytical Determinations
2.3. TG Self-Measurements
2.4. Dietary Intake
2.5. Statistics
3. Results
| Characteristic | (n = 139) |
|---|---|
| Age (years) | 39.9 ± 14.2 |
| BMI (kg/m2) | 24.6 ± 3.4 |
| SBP (mmHg) | 125.0 ± 13.5 |
| DBP (mmHg) | 81.5 ± 10.2 |
| Smoking (n, %) | 35 (25.2%) |
| History of CAD (n, %) | 27 (19.4%) |
| History of T2DM (n, %) | 3 (2.2%) |
| Total cholesterol (mmol/L) | 5.30 ± 1.04 |
| LDL-C (mmol/L) | 3.45 ± 1.00 |
| HDL-C (mmol/L) | 1.23 ± 0.31 |
| Apolipoprotein B (g/L) | 0.97 ± 0.24 |
| Apolipoprotein A–I (g/L) | 1.32 ± 0.22 |
| Plasma triglycerides (mmol/L) | 1.35 ± 0.77 |
| Alcohol Intake | |
| None (n, %) | 34 (24.5%) |
| Low, <10 g/day (n, %) | 27 (19.4%) |
| Moderate, 20–30 g/day (n, %) | 54 (38.8%) |
| High, >30 g/day (n, %) | 24 (17.3%) |
| Characteristics | Alcohol Intake | |||
|---|---|---|---|---|
| No | Low | Moderate | High | |
| (n = 34) | (n = 27) | (n = 54) | (n = 24) | |
| Alcohol intake (g/day) | 0.0 ± 0.0 | 5.4 ± 2.7 | 19.9 ± 6.2 | 61.5 ± 43.0 |
| Age (years) | 34.9 ± 14.5 | 41.7 ± 13.4 | 39.9 ± 15.0 | 45.2 ± 10.9 a |
| BMI (kg/m2) | 24.6 ± 3.4 | 23.8 ± 2.9 | 24.6 ± 3.6 | 25.4 ± 3.8 |
| SBP (mmHg) | 127.4 ± 14.2 | 122.1 ± 11.1 | 124.4 ± 13.5 | 126.4 ± 15.1 |
| DBP (mmHg) | 83.0 ± 8.9 | 81.9 ± 9.2 | 80.5 ± 10.2 | 81.1 ± 12.6 |
| Smoking (n, %) | 6 (17.6%) | 5 (18.5%) | 12 (22.2%) | 12 (50.0%) b |
| History of CAD (n, %) | 6 (17.6%) | 3 (11.1%) | 10 (18.5%) | 8 (33.3%) |
| History of T2DM (n, %) | 2 (5.9%) | 1 (3.7%) | 2 (3.7%) | 1 (4.2%) |
| Total cholesterol (mmol/L) | 5.08 ± 1.13 | 5.17 ± 1.11 | 5.33 ± 1.04 | 5.34 ± 0.83 |
| LDL-C (mmol/L) | 3.27 ± 1.01 | 3.35 ± 1.09 | 3.47 ± 0.98 | 3.43 ± 0.84 |
| HDL-C (mmol/L) | 1.20 ± 0.29 | 1.20 ± 0.37 | 1.24 ± 0.32 | 1.27 ± 0.27 |
| Apolipoprotein B (g/L) | 0.89 ± 0.22 | 0.99 ± 0.29 | 0.99 ± 0.22 | 1.02 ± 0.26 |
| Apolipoprotein A–I (g/L) | 1.34 ± 0.24 | 1.34 ± 0.23 | 1.27 ± 0.19 | 1.38 ± 0.22 |
| Plasma triglycerides (mmol/L) | 1.32 ± 0.78 | 1.32 ± 0.75 | 1.38 ± 0.66 | 1.38 ± 1.04 |
| Fasting plasma glucose (mmol/L) | 5.17 ± 0.60 | 5.29 ± 093 | 5.43 ± 1.95 | 5.32 ± 0.69 |
| Fasting plasma insulin (UI/L) | 8.47 ± 3.89 | 9.20 ± 6.07 | 8.25 ± 418 | 10.65 ± 11.59 |
| Dietary carbohydrates intake (g/day) | 278.48 ± 81.68 | 282.26 ± 58.33 | 273.60 ± 78.79 | 265.15 ± 68.05 |
| Dietary fat intake (g/day) | 86.95 ± 28.37 | 93.00 ± 24.93 | 87.48 ± 26.67 | 89.98 ± 20.95 |
| Dietary MUFA intake (g/day) | 32.96 ± 13.22 | 36.80 ± 10.77 | 33.90 ± 11.39 | 35.07 ± 8.93 |
| Dietary PUFA intake (g/day) | 13.61 ± 5.66 | 15.72 ± 4.67 | 13.68 ± 5.21 | 15.47 ± 4.48 |
| Dietary saturated fat intake (g/day) | 32.59 ± 10.72 | 34.02 ± 9.13 | 32.48 ± 10.36 | 32.66 ± 8.02 |
| Energyintake (kcal/day) | 2301.30 ± 586.35 | 2402.38 ± 481.40 | 2405.81 ± 608.94 | 2651.58 ± 599.32 |
| ∆cTG-AUC (mmol·h/L) | 6.8 ± 7.0 | 2.8 ± 7.3 | 6.5 ± 5.6 | 7.4 ± 6.3 |
| ∆cTG-AUC (mmol·h/L) adjusted * | 7.1 ± 1.8 | 3.1 ± 1.9 c | 6.5 ± 1.8 | 7.3 ± 2.2 |
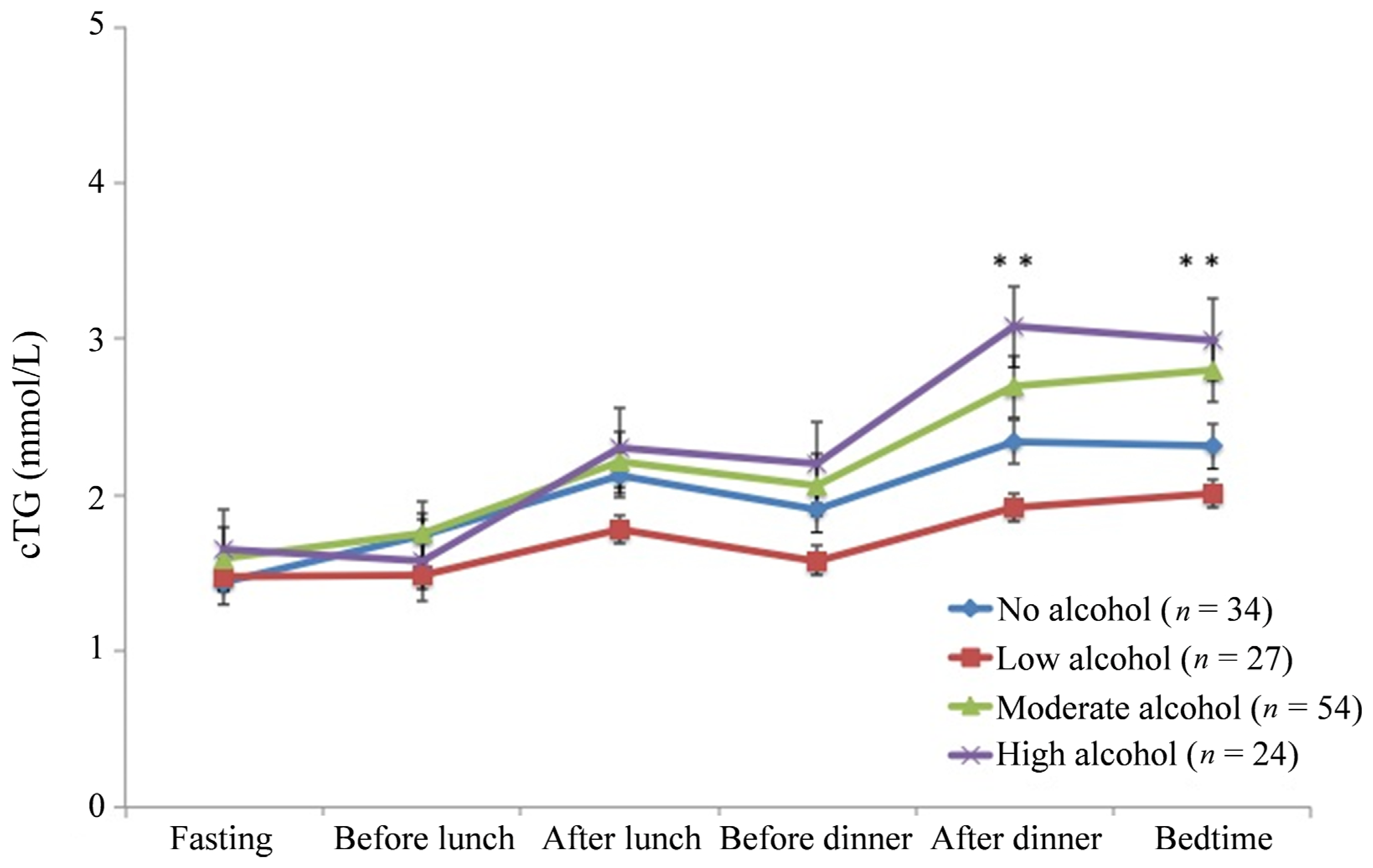
4. Discussion
5. Conclusions
Acknowledgments
Conflicts of Interest
References
- Schieffer, B.; Selle, T.; Hilfiker, A.; Hilfiker-Kleiner, D.; Grote, K.; Tietge, U.J.; Trautwein, C.; Luchtefeld, M.; Schmittkamp, C.; Heeneman, S.; et al. Impact of interleukin-6 on plaque development and morphology in experimental atherosclerosis. Circulation 2004, 110, 3493–3500. [Google Scholar] [CrossRef]
- Castro Cabezas, M.; Halkes, C.J.; Meijssen, S.; van Oostrom, A.J.; Erkelens, D.W. Diurnal triglyceride profiles: A novel approach to study triglyceride changes. Atherosclerosis 2001, 155, 219–228. [Google Scholar] [CrossRef]
- Halkes, C.J.; van Wijk, J.P.; Ribalta, J.; Masana, L.; Castro Cabezas, M. Diurnal triglyceridaemia and insulin resistance in mildly obese subjects with normal fasting plasma lipids. J. Intern. Med. 2004, 255, 74–81. [Google Scholar] [CrossRef]
- Masana, L.; Ribalta, J.; Salazar, J.; Fernandez-Ballart, J.; Joven, J.; Cabezas, M.C. The apolipoprotein AV gene and diurnal triglyceridaemia in normolipidaemic subjects. Clin. Chem. Lab. Med. 2003, 41, 517–521. [Google Scholar]
- Ribalta, J.; Halkes, C.J.; Salazar, J.; Masana, L.; Cabezas, M.C. Additive effects of the PPARγ, APOE, and FABP-2 genes in increasing daylong triglycerides of normolipidemic women to concentrations comparable to those in men. Clin. Chem. 2005, 51, 864–871. [Google Scholar] [CrossRef]
- Klop, B.; Rego, A.T.; Cabezas, M.C. Alcohol and plasma triglycerides. Curr. Opin. Lipidol. 2013, 24, 321–326. [Google Scholar] [CrossRef]
- Foerster, M.; Marques-Vidal, P.; Gmel, G.; Daeppen, J.B.; Cornuz, J.; Hayoz, D.; Pecoud, A.; Mooser, V.; Waeber, G.; Vollenweider, P.; et al. Alcohol drinking and cardiovascular risk in a population with high mean alcohol consumption. Am. J. Cardiol. 2009, 103, 361–368. [Google Scholar] [CrossRef]
- Whitfield, J.B.; Heath, A.C.; Madden, P.A.; Pergadia, M.L.; Montgomery, G.W.; Martin, N.G. Metabolic and biochemical effects of low-to-moderate alcohol consumption. Alcohol. Clin. Exp. Res. 2013, 37, 575–586. [Google Scholar] [CrossRef]
- Lehto, S.; Ronnemaa, T.; Haffner, S.M.; Pyorala, K.; Kallio, V.; Laakso, M. Dyslipidemia and hyperglycemia predict coronary heart disease events in middle-aged patients with niddm. Diabetes 1997, 46, 1354–1359. [Google Scholar] [CrossRef]
- Nordestgaard, B.G.; Benn, M.; Schnohr, P.; Tybjaerg-Hansen, A. Nonfasting triglycerides and risk of myocardial infarction, ischemic heart disease, and death in men and women. JAMA 2007, 298, 299–308. [Google Scholar] [CrossRef]
- O’Keefe, J.H.; Bybee, K.A.; Lavie, C.J. Alcohol and cardiovascular health: The razor-sharp double-edged sword. J. Am. Coll. Cardiol. 2007, 50, 1009–1014. [Google Scholar] [CrossRef]
- Ruidavets, J.B.; Ducimetiere, P.; Evans, A.; Montaye, M.; Haas, B.; Bingham, A.; Yarnell, J.; Amouyel, P.; Arveiler, D.; Kee, F.; et al. Patterns of alcohol consumption and ischaemic heart disease in culturally divergent countries: The prospective epidemiological study of myocardial infarction (prime). BMJ 2010, 341, c6077. [Google Scholar] [CrossRef]
- Sarwar, N.; Danesh, J.; Eiriksdottir, G.; Sigurdsson, G.; Wareham, N.; Bingham, S.; Boekholdt, S.M.; Khaw, K.T.; Gudnason, V. Triglycerides and the risk of coronary heart disease: 10,158 Incident cases among 262,525 participants in 29 western prospective studies. Circulation 2007, 115, 450–458. [Google Scholar] [CrossRef]
- Brien, S.E.; Ronksley, P.E.; Turner, B.J.; Mukamal, K.J.; Ghali, W.A. Effect of alcohol consumption on biological markers associated with risk of coronary heart disease: Systematic review and meta-analysis of interventional studies. BMJ 2011, 342, d636. [Google Scholar]
- Gaziano, J.M.; Buring, J.E.; Breslow, J.L.; Goldhaber, S.Z.; Rosner, B.; VanDenburgh, M.; Willett, W.; Hennekens, C.H. Moderate alcohol intake, increased levels of high-density lipoprotein and its subfractions, and decreased risk of myocardial infarction. N. Engl. J. Med. 1993, 329, 1829–1834. [Google Scholar] [CrossRef]
- Marques-Vidal, P.; Cambou, J.P.; Nicaud, V.; Luc, G.; Evans, A.; Arveiler, D.; Bingham, A.; Cambien, F. Cardiovascular risk factors and alcohol consumption in france and northern ireland. Atherosclerosis 1995, 115, 225–232. [Google Scholar] [CrossRef]
- Savolainen, M.J.; Hannuksela, M.; Seppanen, S.; Kervinen, K.; Kesaniemi, Y.A. Increased high-density lipoprotein cholesterol concentration in alcoholics is related to low cholesteryl ester transfer protein activity. Eur. J. Clin. Investig. 1990, 20, 593–599. [Google Scholar]
- Rimm, E.B.; Williams, P.; Fosher, K.; Criqui, M.; Stampfer, M.J. Moderate alcohol intake and lower risk of coronary heart disease: Meta-analysis of effects on lipids and haemostatic factors. BMJ 1999, 319, 1523–1528. [Google Scholar] [CrossRef]
- Sharrett, A.R.; Heiss, G.; Chambless, L.E.; Boerwinkle, E.; Coady, S.A.; Folsom, A.R.; Patsch, W. Metabolic and lifestyle determinants of postprandial lipemia differ from those of fasting triglycerides: The atherosclerosis risk in communities (aric) study. Arterioscler. Thromb. Vasc. Biol. 2001, 21, 275–281. [Google Scholar] [CrossRef]
- Baraona, E.; Lieber, C.S. Alcohol and lipids. Recent Dev. Alcohol. 1998, 14, 97–134. [Google Scholar]
- Delawi, D.; Meijssen, S.; Castro Cabezas, M. Intra-individual variations of fasting plasma lipids, apolipoproteins and postprandial lipemia in familial combined hyperlipidemia compared to controls. Clin. Chim. Acta 2003, 328, 139–145. [Google Scholar] [CrossRef]
- Halkes, C.J.; Castro Cabezas, M.; van Wijk, J.P.; Erkelens, D.W. Gender differences in diurnal triglyceridemia in lean and overweight subjects. Int. J. Obes. Relat. Metab. Disord. 2001, 25, 1767–1774. [Google Scholar] [CrossRef]
- Van Oostrom, A.J.; Castro Cabezas, M.; Ribalta, J.; Masana, L.; Twickler, T.B.; Remijnse, T.A.; Erkelens, D.W. Diurnal triglyceride profiles in healthy normolipidemic male subjects are associated to insulin sensitivity, body composition and diet. Eur. J. Clin. Investig. 2000, 30, 964–971. [Google Scholar] [CrossRef]
- Van Wijk, J.P.; Cabezas, M.C.; Halkes, C.J.; Erkelens, D.W. Effects of different nutrient intakes on daytime triacylglycerolemia in healthy, normolipemic, free-living men. Am. J. Clin. Nutr. 2001, 74, 171–178. [Google Scholar]
- Van Wijk, J.P.; van Oostrom, A.J.; Castro Cabezas, M. Normal ranges of non-fasting triglycerides in healthy dutch males and females. Clin. Chim. Acta 2003, 337, 49–57. [Google Scholar] [CrossRef]
- Moses, R.G.; Calvert, D.; Storlien, L.H. Evaluation of the Accutrend GCT with respect to triglyceride monitoring. Diabetes Care 1996, 19, 1305–1306. [Google Scholar]
- Dutch Food Composition Datebase. Available online: http://www.rivm.nl/en/Topics/D/Dutch_Food_Composition_Database (accessed on 9 December 2013).
- Hansel, B.; Thomas, F.; Pannier, B.; Bean, K.; Kontush, A.; Chapman, M.J.; Guize, L.; Bruckert, E. Relationship between alcohol intake, health and social status and cardiovascular risk factors in the urban paris-ile-de-france cohort: Is the cardioprotective action of alcohol a myth? Eur. J. Clin. Nutr. 2010, 64, 561–568. [Google Scholar] [CrossRef]
- Wakabayashi, I. Associations of alcohol drinking and cigarette smoking with serum lipid levels in healthy middle-aged men. Alcohol Alcohol. 2008, 43, 274–280. [Google Scholar] [CrossRef]
- Tolstrup, J.S.; Gronbaek, M.; Nordestgaard, B.G. Alcohol intake, myocardial infarction, biochemical risk factors, and alcohol dehydrogenase genotypes. Circ. Cardiovasc. Genet. 2009, 2, 507–514. [Google Scholar] [CrossRef]
- Daher, C.F.; Slaiby, R.; Haddad, N.; Boustany, K.; Baroody, G.M. Effect of acute and chronic moderate red or white wine consumption on fasted and postprandial lipemia in the rat. J. Toxicol. Environ. Health A 2006, 69, 1117–1131. [Google Scholar] [CrossRef]
- Mudrakova, E.; Poledne, R.; Kovar, J. Postprandial triglyceridemia after single dose of alcohol in healthy young men. Nutr. Metab. Cardiovasc. Dis. 2013, 23, 183–188. [Google Scholar] [CrossRef]
- Schneider, J.; Liesenfeld, A.; Mordasini, R.; Schubotz, R.; Zofel, P.; Kubel, F.; Vandre-Plozzitzka, C.; Kaffarnik, H. Lipoprotein fractions, lipoprotein lipase and hepatic triglyceride lipase during short-term and long-term uptake of ethanol in healthy subjects. Atherosclerosis 1985, 57, 281–291. [Google Scholar] [CrossRef]
- Zhong, W.; Zhao, Y.; Tang, Y.; Wei, X.; Shi, X.; Sun, W.; Sun, X.; Yin, X.; Sun, X.; Kim, S.; et al. Chronic alcohol exposure stimulates adipose tissue lipolysis in mice: Role of reverse triglyceride transport in the pathogenesis of alcoholic steatosis. Am. J. Pathol. 2012, 180, 998–1007. [Google Scholar]
- Park, H.; Kim, K. Relationship between alcohol consumption and serum lipid levels in elderly korean men. Arch. Gerontol. Geriatr. 2012, 55, 226–230. [Google Scholar] [CrossRef]
- Van Tol, A.; van der Gaag, M.S.; Scheek, L.M.; van Gent, T.; Hendriks, H.F. Changes in postprandial lipoproteins of low and high density caused by moderate alcohol consumption with dinner. Atherosclerosis 1998, 141, S101–S103. [Google Scholar] [CrossRef]
- Malarcher, A.M.; Giles, W.H.; Croft, J.B.; Wozniak, M.A.; Wityk, R.J.; Stolley, P.D.; Stern, B.J.; Sloan, M.A.; Sherwin, R.; Price, T.R.; et al. Alcohol intake, type of beverage, and the risk of cerebral infarction in young women. Stroke 2001, 32, 77–83. [Google Scholar] [CrossRef]
- Blanco-Colio, L.M.; Munoz-Garcia, B.; Martin-Ventura, J.L.; Alvarez-Sala, L.A.; Castilla, M.; Bustamante, A.; Lamuela-Raventos, R.M.; Gomez-Gerique, J.; Fernandez-Cruz, A.; Millan, J.; et al. Ethanol beverages containing polyphenols decrease nuclear factor kappa-B activation in mononuclear cells and circulating MCP-1 concentrations in healthy volunteers during a fat-enriched diet. Atherosclerosis 2007, 192, 335–341. [Google Scholar] [CrossRef]
- Covas, M.I.; Konstantinidou, V.; Mysytaki, E.; Fito, M.; Weinbrenner, T.; de la Torre, R.; Farre-Albadalejo, M.; Lamuela-Raventos, R. Postprandial effects of wine consumption on lipids and oxidative stress biomarkers. Drugs Exp. Clin. Res. 2003, 29, 217–223. [Google Scholar]
- Naissides, M.; Mamo, J.C.; James, A.P.; Pal, S. The effect of chronic consumption of red wine on cardiovascular disease risk factors in postmenopausal women. Atherosclerosis 2006, 185, 438–445. [Google Scholar] [CrossRef]
- Barefoot, J.C.; Gronbaek, M.; Feaganes, J.R.; McPherson, R.S.; Williams, R.B.; Siegler, I.C. Alcoholic beverage preference, diet, and health habits in the unc alumni heart study. Am. J. Clin. Nutr. 2002, 76, 466–472. [Google Scholar]
- Timmers, S.; Konings, E.; Bilet, L.; Houtkooper, R.H.; van de Weijer, T.; Goossens, G.H.; Hoeks, J.; van der Krieken, S.; Ryu, D.; Kersten, S.; et al. Calorie restriction-like effects of 30 days of resveratrol supplementation on energy metabolism and metabolic profile in obese humans. Cell Metab. 2011, 14, 612–622. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Torres do Rego, A.; Klop, B.; Birnie, E.; Elte, J.W.F.; Ramos, V.C.; Walther, L.A.A.-S.; Cabezas, M.C. Diurnal Triglyceridemia in Relation to Alcohol Intake in Men. Nutrients 2013, 5, 5114-5126. https://doi.org/10.3390/nu5125114
Torres do Rego A, Klop B, Birnie E, Elte JWF, Ramos VC, Walther LAA-S, Cabezas MC. Diurnal Triglyceridemia in Relation to Alcohol Intake in Men. Nutrients. 2013; 5(12):5114-5126. https://doi.org/10.3390/nu5125114
Chicago/Turabian StyleTorres do Rego, Ana, Boudewijn Klop, Erwin Birnie, Jan Willem F. Elte, Victoria Cachofeiro Ramos, Luis A. Alvarez-Sala Walther, and Manuel Castro Cabezas. 2013. "Diurnal Triglyceridemia in Relation to Alcohol Intake in Men" Nutrients 5, no. 12: 5114-5126. https://doi.org/10.3390/nu5125114
APA StyleTorres do Rego, A., Klop, B., Birnie, E., Elte, J. W. F., Ramos, V. C., Walther, L. A. A.-S., & Cabezas, M. C. (2013). Diurnal Triglyceridemia in Relation to Alcohol Intake in Men. Nutrients, 5(12), 5114-5126. https://doi.org/10.3390/nu5125114




