Effects of Supplementation with Milk Proteins on Body Composition and Anthropometric Parameters: A Systematic Review and Dose–Response Meta-Analysis
Abstract
1. Introduction
2. Methods
2.1. Search Strategy
2.2. Selection Criteria
2.3. Data Extraction
2.4. Risk of Bias Assessment
2.5. Certainty Assessment
2.6. Statistical Analysis
3. Results
3.1. Study Selection
3.2. Study Characteristics
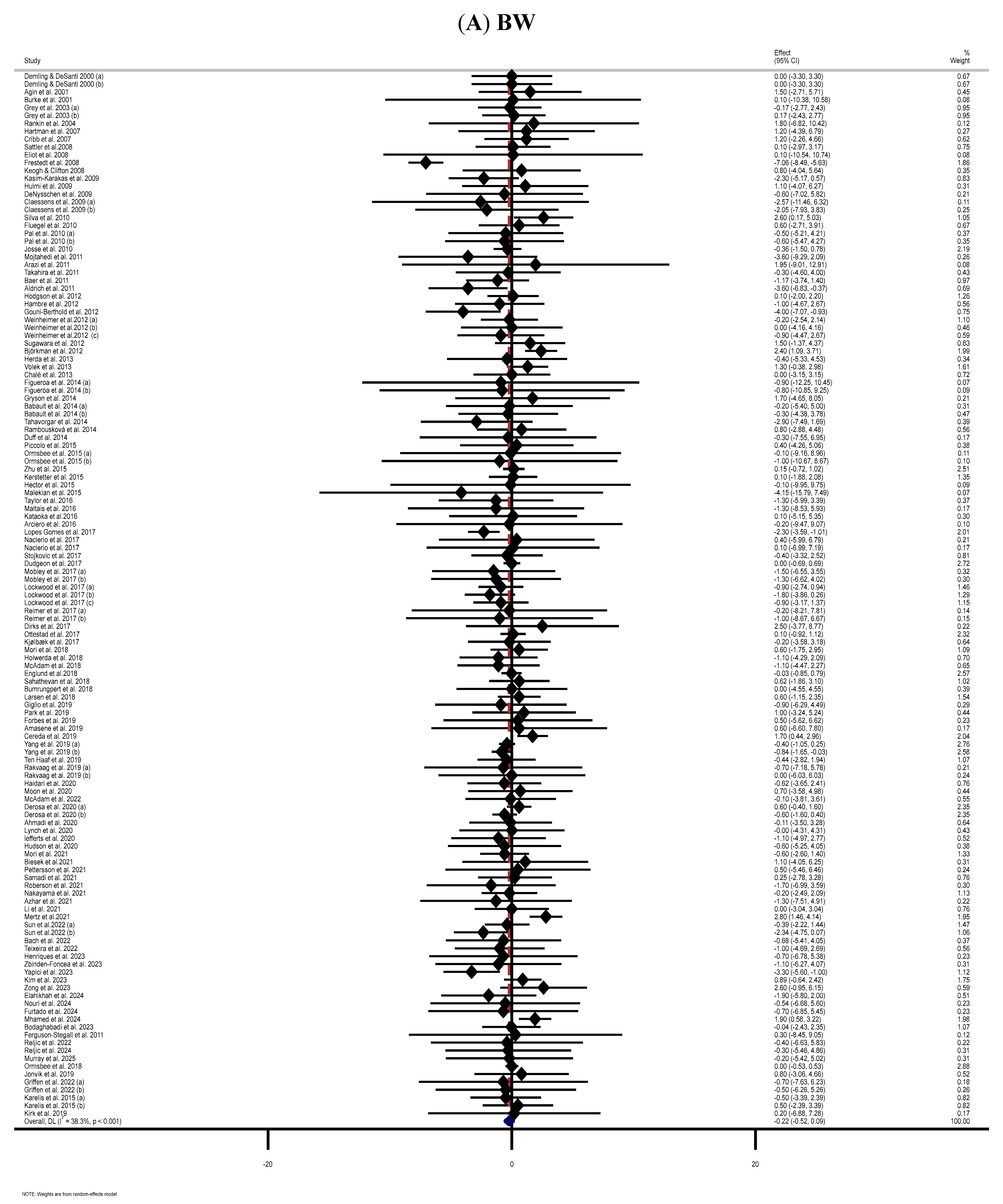
3.3. Effect of Supplementation with MP on BW
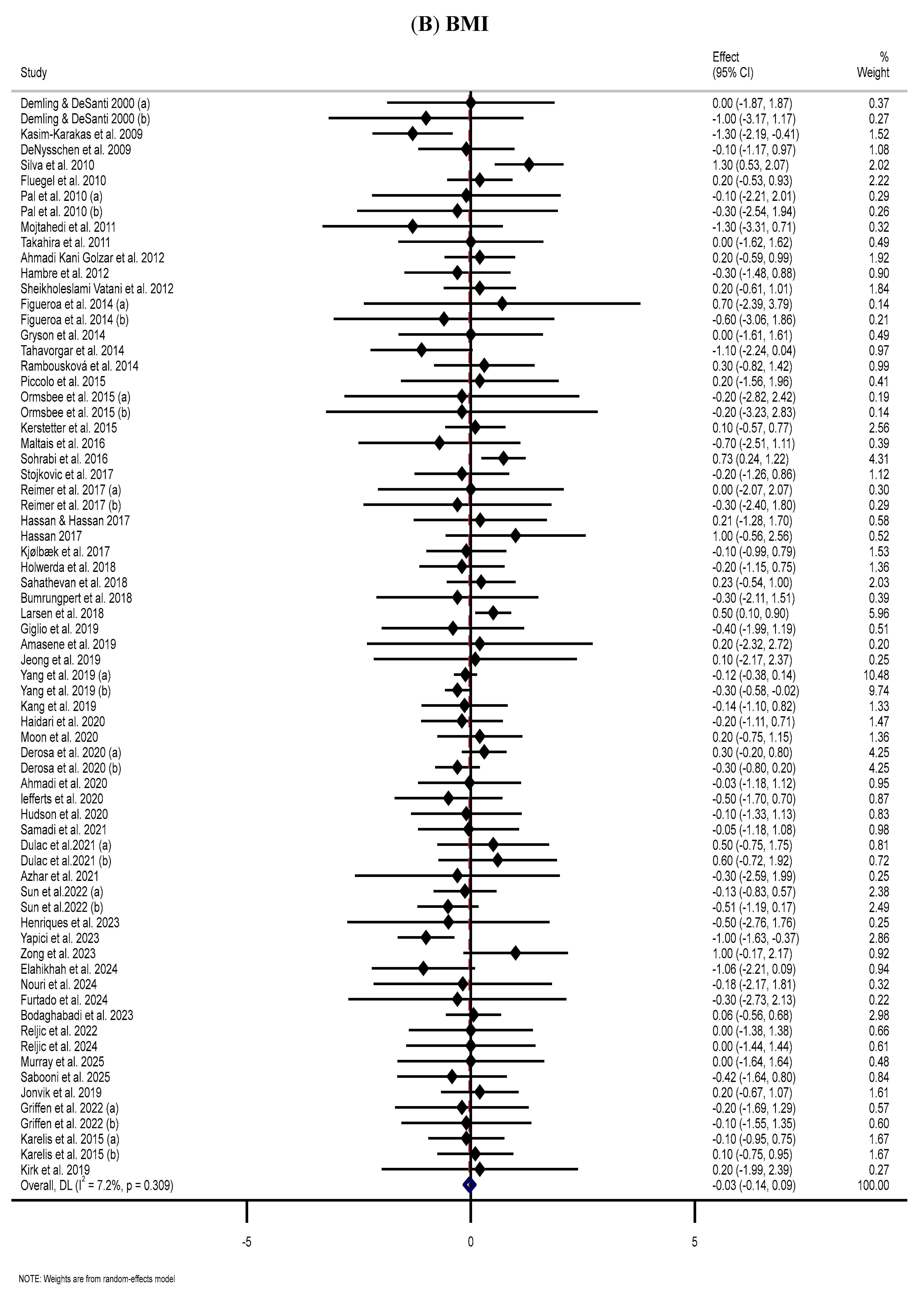
3.4. Effect of Supplementation with MP on BMI
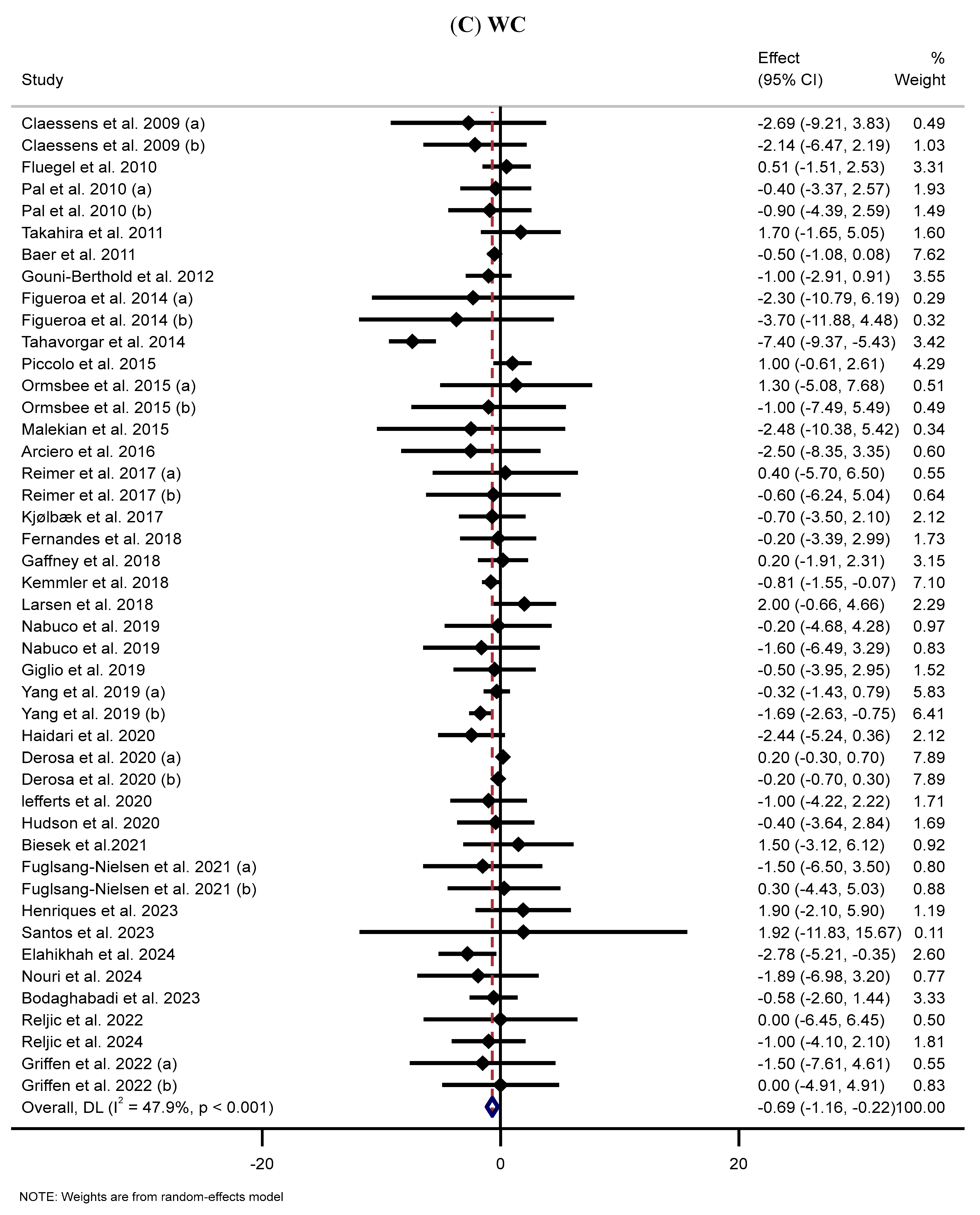
3.5. Effect of Supplementation with MP on WC
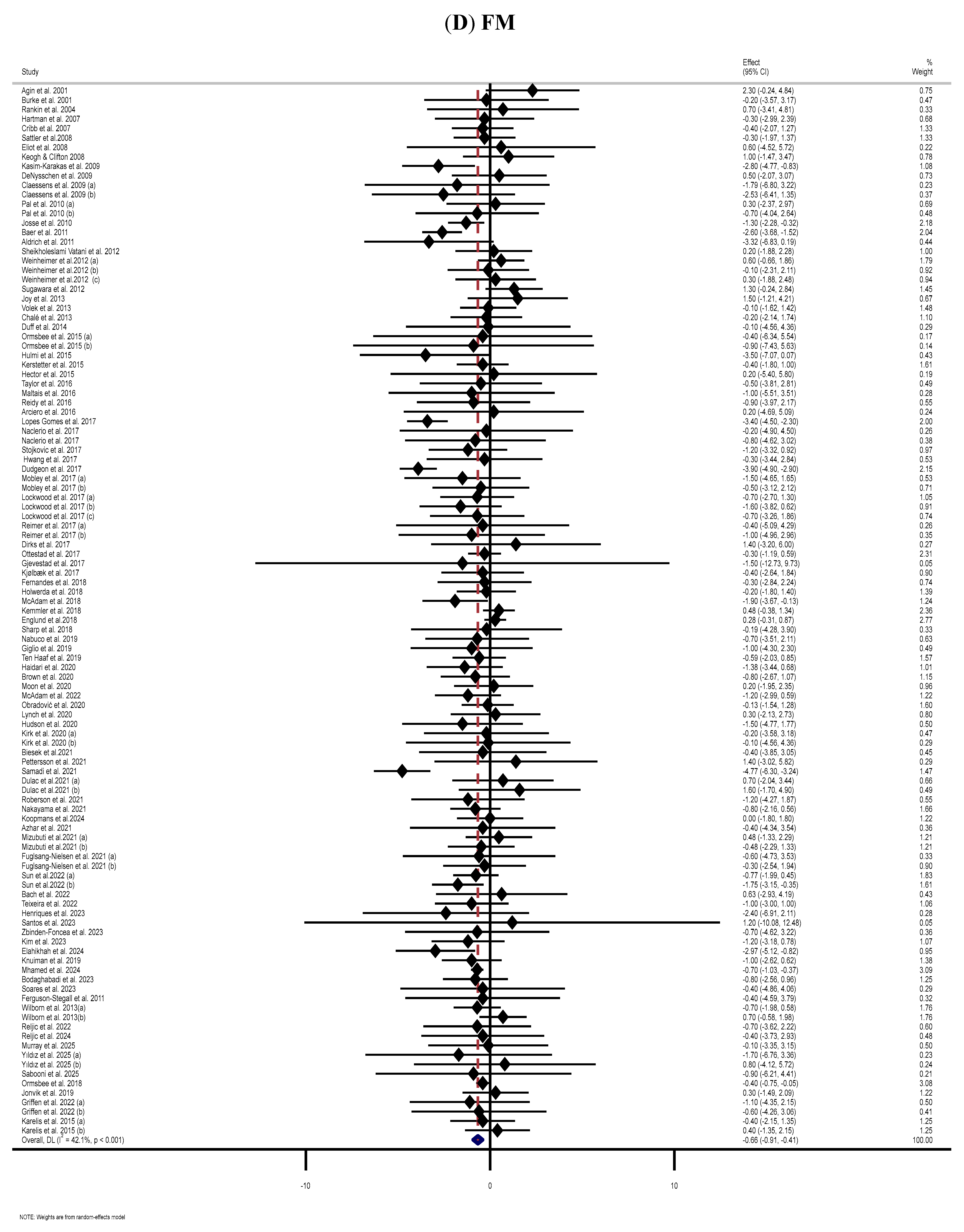
3.6. Effect of Supplementation with MP on FM
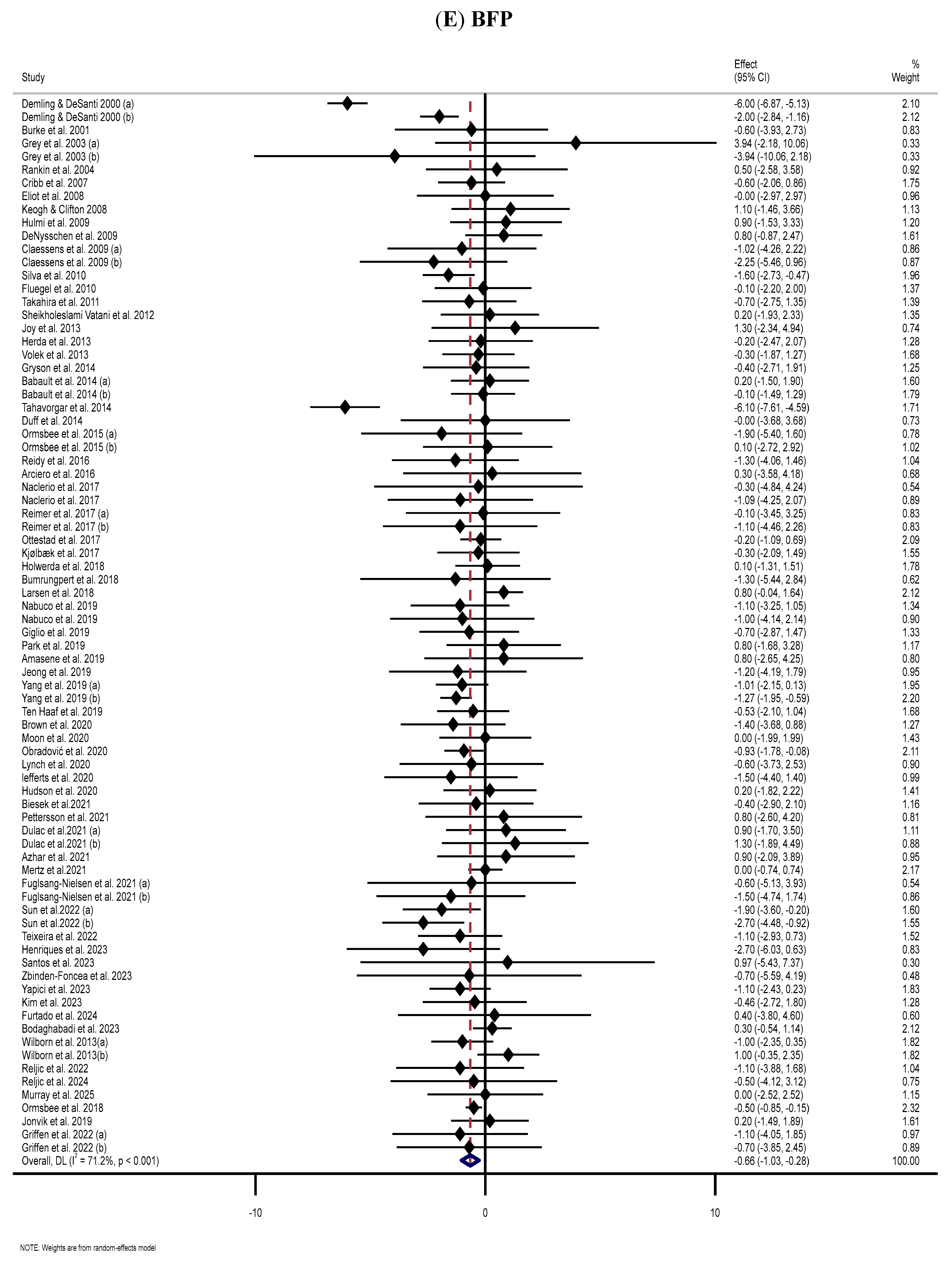
3.7. Effect of Supplementation with MP on BFP
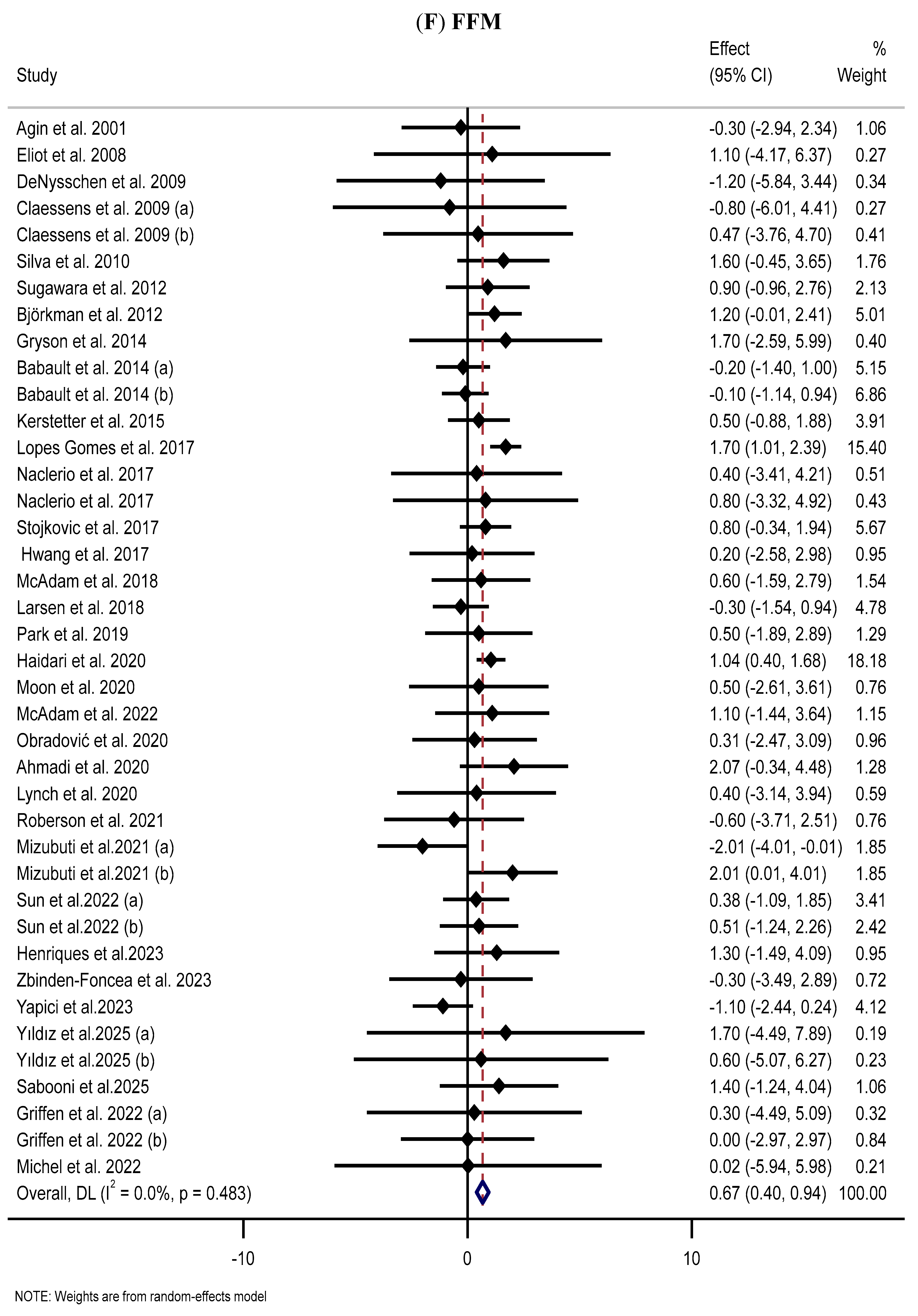
3.8. Effect of Supplementation with MP on FFM
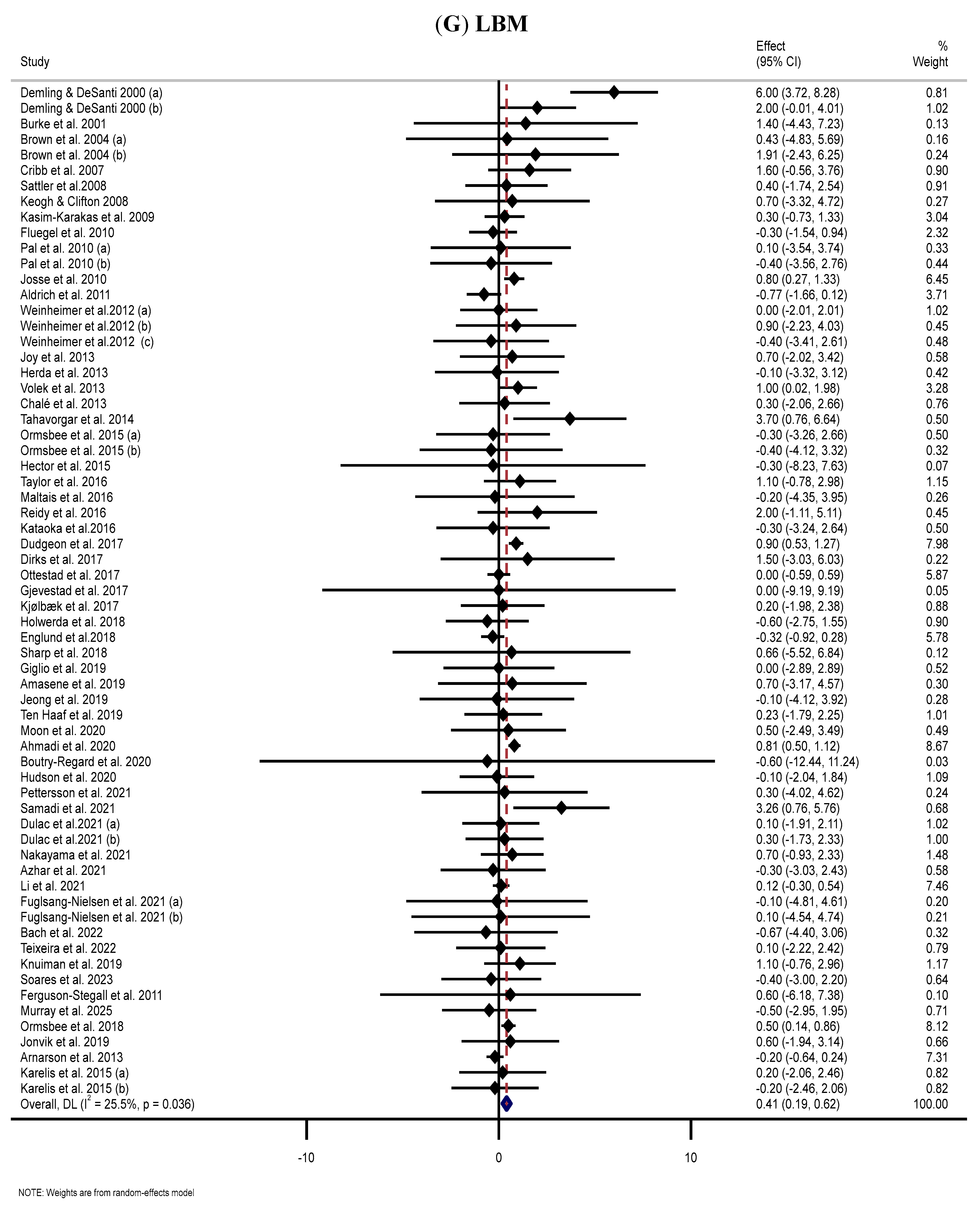
3.9. Effect of Supplementation with MP on LBM
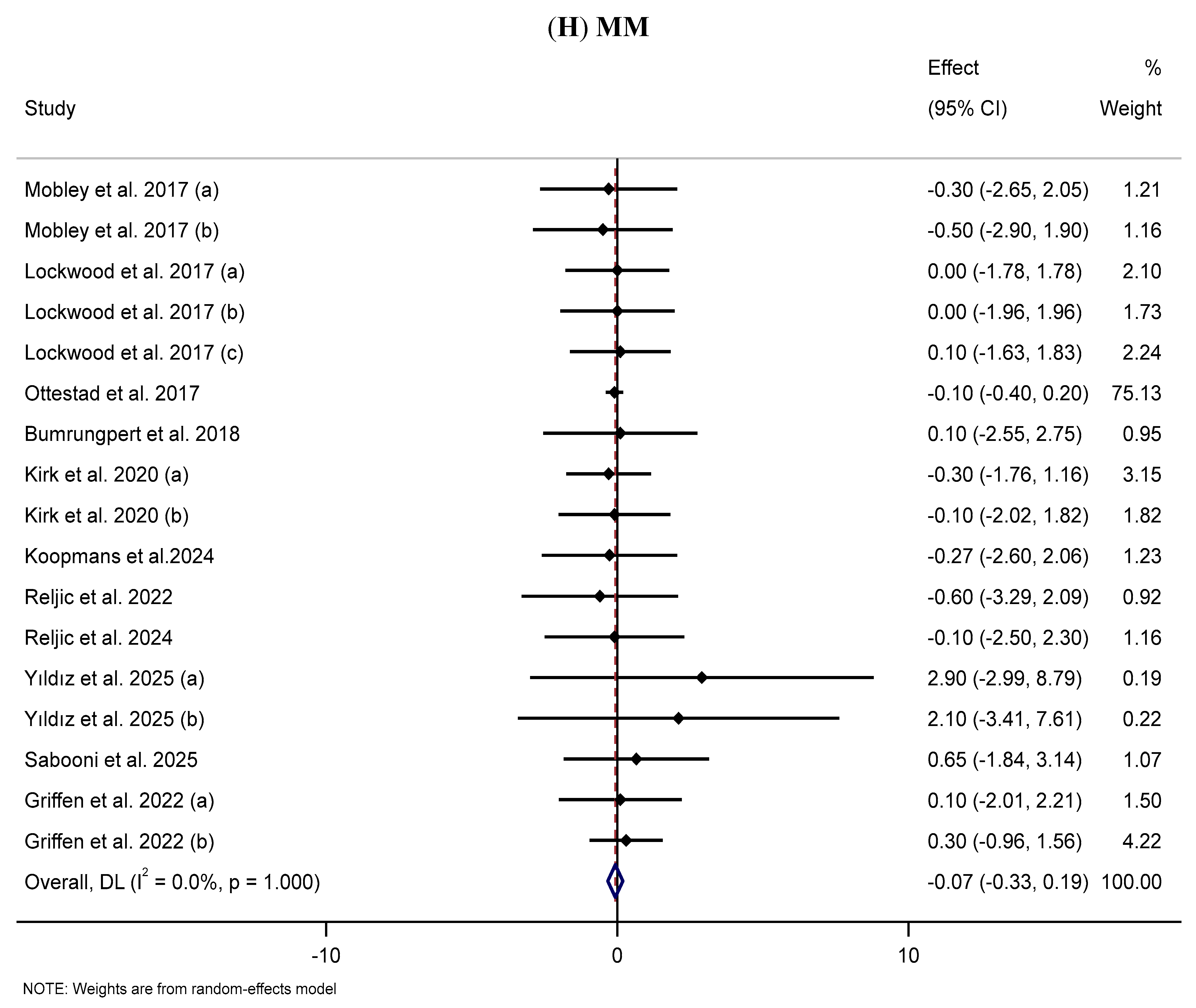
3.10. Effect of Supplementation with MP on MM
3.11. Publication Bias
3.12. Risk of Bias Evaluation
3.13. GRADE
3.14. Linear and Non-Linear Dose–Response Relations
3.15. Sensitivity Analysis
4. Discussion
4.1. Possible Underlying Mechanisms
4.2. Strengths and Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Abbreviations
| MP | Milk protein |
| BC | Body composition |
| MPC | Milk protein concentrate |
| WMD | Weighted mean difference |
| LBM | Lean body mass |
| WP | Whey protein |
| FFM | Fat-free mass |
| CI | Confidence interval |
| RT | Resistance training |
| WC | Waist circumference |
| FM | Fat mass |
| RCT | Randomized controlled trial |
| PROSPERO | Prospective Register of Systematic Reviews |
| BMI | Body mass index |
| PRISMA | Preferred Reporting Items for Systematic Reviews and Meta-Analyses |
| SD | Standard deviation |
| RoB | Risk of Bias |
| GRADE | Grading of Recommendations, Assessment, Development, and Evaluation |
| BFP | Body fat percentage |
| CP | Casein protein |
| BW | Body weight |
| AAs | Amino acids |
| BCAAs | Branched-chain amino acids |
| ALS | Amyotrophic lateral sclerosis |
| CF | Cystic fibrosis |
| CHD | Chronic heart disease |
| COPD | Chronic obstructive pulmonary disease |
| HIV | Human immunodeficiency virus |
| HTN | Hypertension |
| MetS | Metabolic syndrome |
| MM | Muscle mass |
| OAGB | One anastomosis gastric bypass |
| PCOS | Polycystic ovary syndrome |
| PICOS | Population, intervention, comparator, outcomes, study design |
| T2DM | Type 2 diabetes mellitus |
| WPH | Whey protein hydrolysates |
| GLP-1 | Glucagon-like peptide-1 |
| CCK | Cholecystokinin |
| BAT | Brown adipose tissue |
| WAT | White adipose tissue |
| OW | Overweight |
| OB | Obesity |
| AO | Abdominal obesity |
| BP | Blood pressure |
| WPI | Whey protein isolate |
| WPC | Whey protein concentrate |
| PL | Placebo |
| WPC-L | High-lactoferrin-containing WPC |
| ERD | Energy-restricted diet |
| CHO | Carbohydrate |
| MD | Maltodextrin |
| PRE | Progressive resistance exercise |
| ITF | Inulin-type fructans |
| SG | Sleeve gastrectomy |
| UK | United Kingdom |
| USA | United States of America |
| DPP-4 | Dipeptidyl peptidase 4 |
References
- Wirunsawanya, K.; Upala, S.; Jaruvongvanich, V.; Sanguankeo, A. Whey protein supplementation improves body composition and cardiovascular risk factors in overweight and obese patients: A systematic review and meta-analysis. J. Am. Coll. Nutr. 2018, 37, 60–70. [Google Scholar] [CrossRef]
- Miller, P.E.; Alexander, D.D.; Perez, V. Effects of whey protein and resistance exercise on body composition: A meta-analysis of randomized controlled trials. J. Am. Coll. Nutr. 2014, 33, 163–175. [Google Scholar] [CrossRef]
- Anderson, H.; Luhovyy, B.; Akhavan, T.; Panahi, S. Milk proteins in the regulation of body weight, satiety, food intake and glycemia. Milk Milk Prod. Hum. Nutr. 2011, 67, 147–159. [Google Scholar]
- Dougkas, A.; Reynolds, C.K.; Givens, I.D.; Elwood, P.C.; Minihane, A.M. Associations between dairy consumption and body weight: A review of the evidence and underlying mechanisms. Nutr. Res. Rev. 2011, 24, 72–95. [Google Scholar] [CrossRef]
- Aggarwal, R.; Bains, K.; Jain, R. Effect of milk or milk derived food supplementation on body composition of Young Indian Women. Asian J. Dairy Food Res. 2023, 42, 348–354. [Google Scholar] [CrossRef]
- Santos, G.D.O.; Costa e Silva, G.; Souza, R.B.d.; Medeiros, J.S.; Brito, I.S.d.; Cardoso, S.P.; Leão, P.V.T.; Nicolau, E.S.; Cappato, L.P.; Favareto, R. Effect of milk protein concentrate supplementation on body composition and biochemical markers during a resistance training program. Food Sci. Technol. 2022, 42, e67222. [Google Scholar] [CrossRef]
- Osuka, Y.; Fujita, S.; Kitano, N.; Kosaki, K.; Seol, J.; Sawano, Y.; Shi, H.; Fujii, Y.; Maeda, S.; Okura, T. Effects of aerobic and resistance training combined with fortified milk on muscle mass, muscle strength, and physical performance in older adults: A randomized controlled trial. J. Nutr. Health Aging 2017, 21, 1349–1357. [Google Scholar] [CrossRef]
- Li, M.-L.; Zhang, F.; Luo, H.-Y.; Quan, Z.-W.; Wang, Y.-F.; Huang, L.-T.; Wang, J.-H. Improving sarcopenia in older adults: A systematic review and meta-analysis of randomized controlled trials of whey protein supplementation with or without resistance training. J. Nutr. Health Aging 2024, 28, 100184. [Google Scholar] [CrossRef] [PubMed]
- Marangoni, F.; Pellegrino, L.; Verduci, E.; Ghiselli, A.; Bernabei, R.; Calvani, R.; Cetin, I.; Giampietro, M.; Perticone, F.; Piretta, L. Cow’s milk consumption and health: A health professional’s guide. J. Am. Coll. Nutr. 2019, 38, 197–208. [Google Scholar] [CrossRef]
- Antunes, I.; Bexiga, R.; Pinto, C.; Roseiro, L.; Quaresma, M. Cow’s Milk in Human Nutrition and the Emergence of Plant-Based Milk Alternatives. Foods 2022, 12, 99. [Google Scholar] [CrossRef]
- Pal, S.; Radavelli-Bagatini, S.; Hagger, M.; Ellis, V. Comparative effects of whey and casein proteins on satiety in overweight and obese individuals: A randomized controlled trial. Eur. J. Clin. Nutr. 2014, 68, 980–986. [Google Scholar] [CrossRef]
- Wilborn, C.D.; Taylor, L.W.; Outlaw, J.; Williams, L.; Campbell, B.; Foster, C.A.; Smith-Ryan, A.; Urbina, S.; Hayward, S. The effects of pre-and post-exercise whey vs. casein protein consumption on body composition and performance measures in collegiate female athletes. J. Sports Sci. Med. 2013, 12, 74. [Google Scholar] [CrossRef]
- Chandran, D.; Lejaniya, A.S.; Mahesh, S.V.; Maneesh, S.V.; Sureshkumar, R.; Buttar, H.S.; Kumar, H.; Sharun, K.; Yatoo, M.I.; Mohapatra, R.K.; et al. Major Health Effects of Casein and Whey Proteins Present in Cow Milk: A Narrative Review. Indian Vet. J. 2021, 98, 9–19. [Google Scholar]
- McGregor, R.A.; Poppitt, S.D. Milk protein for improved metabolic health: A review of the evidence. Nutr. Metab. 2013, 10, 46. [Google Scholar] [CrossRef] [PubMed]
- Hall, W.; Millward, D.; Long, S.; Morgan, L. Casein and whey exert different effects on plasma amino acid profiles, gastrointestinal hormone secretion and appetite. Br. J. Nutr. 2003, 89, 239–248. [Google Scholar] [CrossRef]
- Pennings, B.; Boirie, Y.; Senden, J.M.; Gijsen, A.P.; Kuipers, H.; van Loon, L.J. Whey protein stimulates postprandial muscle protein accretion more effectively than do casein and casein hydrolysate in older men. Am. J. Clin. Nutr. 2011, 93, 997–1005. [Google Scholar] [CrossRef]
- Devries, M.C.; McGlory, C.; Bolster, D.R.; Kamil, A.; Rahn, M.; Harkness, L.; Baker, S.K.; Phillips, S.M. Leucine, not total protein, content of a supplement is the primary determinant of muscle protein anabolic responses in healthy older women. J. Nutr. 2018, 148, 1088–1095. [Google Scholar] [CrossRef]
- Pal, S.; Radavelli-Bagatini, S. The effects of whey protein on cardiometabolic risk factors. Obes. Rev. 2013, 14, 324–343. [Google Scholar] [CrossRef]
- Singh, A.; Pezeshki, A.; Zapata, R.C.; Yee, N.J.; Knight, C.G.; Tuor, U.I.; Chelikani, P.K. Diets enriched in whey or casein improve energy balance and prevent morbidity and renal damage in salt-loaded and high-fat-fed spontaneously hypertensive stroke-prone rats. J. Nutr. Biochem. 2016, 37, 47–59. [Google Scholar] [CrossRef] [PubMed]
- Mohammadi, S.; Asbaghi, O.; Dolatshahi, S.; Omran, H.S.; Amirani, N.; Koozehkanani, F.J.; Garmjani, H.B.; Goudarzi, K.; Ashtary-Larky, D. Effects of supplementation with milk protein on glycemic parameters: A GRADE-assessed systematic review and dose–response meta-analysis. Nutr. J. 2023, 22, 49. [Google Scholar] [CrossRef] [PubMed]
- Mohammadi, S.; Ashtary-Larky, D.; Mehrbod, M.; Kouhi Sough, N.; Salehi Omran, H.; Dolatshahi, S.; Amirani, N.; Asbaghi, O. Impacts of supplementation with milk proteins on inflammation: A systematic review and meta-analysis. Inflammopharmacology 2025, 33, 1061–1083. [Google Scholar] [CrossRef]
- Mohammadi, S.; Ashtary-Larky, D.; Beyki, M.; Kouhi Sough, N.; Alaghemand, N.; Amirani, N.; Salehi Omran, H.; Dolatshahi, S.; Asbaghi, O. Impacts of Milk Protein Supplementation on Lipid Profile, Blood Pressure, Oxidative Stress, and Liver Enzymes: A Systematic Review and Meta-analysis. Nutr. Rev. 2025, nuaf068. [Google Scholar] [CrossRef]
- Lam, F.-C.; Khan, T.M.; Faidah, H.; Haseeb, A.; Khan, A.H. Effectiveness of whey protein supplements on the serum levels of amino acid, creatinine kinase and myoglobin of athletes: A systematic review and meta-analysis. Syst. Rev. 2019, 8, 130. [Google Scholar] [CrossRef] [PubMed]
- Tosukhowong, P.; Boonla, C.; Dissayabutra, T.; Kaewwilai, L.; Muensri, S.; Chotipanich, C.; Joutsa, J.; Rinne, J.; Bhidayasiri, R. Biochemical and clinical effects of Whey protein supplementation in Parkinson’s disease: A pilot study. J. Neurol. Sci. 2016, 367, 162–170. [Google Scholar] [CrossRef] [PubMed]
- Sousa, G.T.; Lira, F.S.; Rosa, J.C.; de Oliveira, E.P.; Oyama, L.M.; Santos, R.V.; Pimentel, G.D. Dietary whey protein lessens several risk factors for metabolic diseases: A review. Lipids Health Dis. 2012, 11, 67. [Google Scholar] [CrossRef]
- Huang, L.-P.; Condello, G.; Kuo, C.-H. Effects of milk protein in resistance training-induced lean mass gains for older adults aged ≥60 y: A systematic review and meta-analysis. Nutrients 2021, 13, 2815. [Google Scholar] [CrossRef]
- Sepandi, M.; Samadi, M.; Shirvani, H.; Alimohamadi, Y.; Taghdir, M.; Goudarzi, F.; Akbarzadeh, I. Effect of whey protein supplementation on weight and body composition indicators: A meta-analysis of randomized clinical trials. Clin. Nutr. ESPEN 2022, 50, 74–83. [Google Scholar] [CrossRef]
- Hidayat, K.; Chen, G.-C.; Wang, Y.; Zhang, Z.; Dai, X.; Szeto, I.; Qin, L.-Q. Effects of milk proteins supplementation in older adults undergoing resistance training: A meta-analysis of randomized control trials. J. Nutr. Health Aging 2018, 22, 237–245. [Google Scholar] [CrossRef]
- Hanach, N.I.; McCullough, F.; Avery, A. The impact of dairy protein intake on muscle mass, muscle strength, and physical performance in middle-aged to older adults with or without existing sarcopenia: A systematic review and meta-analysis. Adv. Nutr. 2019, 10, 59–69. [Google Scholar] [CrossRef] [PubMed]
- Khalafi, M.; Fatolahi, S.; Jafari, R.; Rosenkranz, S.K.; Symonds, M.E.; Abbaszadeh Bidgoli, Z.; Fernandez, M.L.; Dinizadeh, F.; Batrakoulis, A. Effects of Whey Protein Supplementation on Body Composition, Muscular Strength, and Cardiometabolic Health in Older Adults: A Systematic Review with Pairwise Meta-Analysis. Healthcare 2025, 13, 2814. [Google Scholar] [CrossRef]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. Int. J. Surg. 2021, 88, 105906. [Google Scholar] [CrossRef] [PubMed]
- Schardt, C.; Adams, M.B.; Owens, T.; Keitz, S.; Fontelo, P. Utilization of the PICO framework to improve searching PubMed for clinical questions. BMC Med. Inform. Decis. Mak. 2007, 7, 16. [Google Scholar] [CrossRef]
- Tagawa, R.; Watanabe, D.; Ito, K.; Otsuyama, T.; Nakayama, K.; Sanbongi, C.; Miyachi, M. Synergistic effect of increased total protein intake and strength training on muscle strength: A dose-response meta-analysis of randomized controlled trials. Sports Med.-Open 2022, 8, 110. [Google Scholar] [CrossRef]
- Sterne, J.A.; Savović, J.; Page, M.J.; Elbers, R.G.; Blencowe, N.S.; Boutron, I.; Cates, C.J.; Cheng, H.-Y.; Corbett, M.S.; Eldridge, S.M. RoB 2: A revised tool for assessing risk of bias in randomised trials. BMJ 2019, 366, l4898. [Google Scholar] [CrossRef]
- Guyatt, G.H.; Oxman, A.D.; Vist, G.E.; Kunz, R.; Falck-Ytter, Y.; Alonso-Coello, P.; Schünemann, H.J. GRADE: An emerging consensus on rating quality of evidence and strength of recommendations. BMJ 2008, 336, 924–926. [Google Scholar] [CrossRef] [PubMed]
- Borenstein, M.; Hedges, L.V.; Higgins, J.P.; Rothstein, H.R. Introduction to Meta-Analysis; John Wiley & Sons: Hoboken, NJ, USA, 2021. [Google Scholar]
- Higgins, J.P.; Thompson, S.G.; Deeks, J.J.; Altman, D.G. Measuring inconsistency in meta-analyses. Bmj 2003, 327, 557–560. [Google Scholar] [CrossRef]
- Begg, C.B.; Berlin, J.A. Publication bias: A problem in interpreting medical data. J. R. Stat. Soc. Ser. A 1988, 151, 419–445. [Google Scholar] [CrossRef]
- Egger, M.; Smith, G.D.; Schneider, M.; Minder, C. Bias in meta-analysis detected by a simple, graphical test. BMJ 1997, 315, 629–634. [Google Scholar] [CrossRef]
- Royston, P.; Altman, D.G. Regression using fractional polynomials of continuous covariates: Parsimonious parametric modelling. J. R. Stat. Soc. Ser. C Appl. Stat. 1994, 43, 429–453. [Google Scholar] [CrossRef]
- Mitchell, M.N. Interpreting and Visualizing Regression Models Using Stata; Stata Press College Station: College Station, TX, USA, 2012; Volume 558. [Google Scholar]
- Claessens, M.; Van Baak, M.; Monsheimer, S.; Saris, W. The effect of a low-fat, high-protein or high-carbohydrate ad libitum diet on weight loss maintenance and metabolic risk factors. Int. J. Obes. 2009, 33, 296–304. [Google Scholar] [CrossRef] [PubMed]
- Pal, S.; Ellis, V.; Dhaliwal, S. Effects of whey protein isolate on body composition, lipids, insulin and glucose in overweight and obese individuals. Br. J. Nutr. 2010, 104, 716–723. [Google Scholar] [CrossRef]
- Fluegel, S.M.; Shultz, T.D.; Powers, J.R.; Clark, S.; Barbosa-Leiker, C.; Wright, B.R.; Freson, T.S.; Fluegel, H.A.; Minch, J.D.; Schwarzkopf, L.K. Whey beverages decrease blood pressure in prehypertensive and hypertensive young men and women. Int. Dairy J. 2010, 20, 753–760. [Google Scholar] [CrossRef]
- Takahira, M.; Noda, K.; Fukushima, M.; Zhang, B.; Mitsutake, R.; Uehara, Y.; Ogawa, M.; Kakuma, T.; Saku, K. Randomized, double-blind, controlled, comparative trial of formula food containing soy protein vs. milk protein in visceral fat obesity. Circ. J. 2011, 75, 2235–2243. [Google Scholar] [CrossRef]
- Aldrich, N.D.; Reicks, M.M.; Sibley, S.D.; Redmon, J.B.; Thomas, W.; Raatz, S.K. Varying protein source and quantity do not significantly improve weight loss, fat loss, or satiety in reduced energy diets among midlife adults. Nutr. Res. 2011, 31, 104–112. [Google Scholar] [CrossRef]
- Gouni-Berthold, I.; Schulte, D.M.; Krone, W.; Lapointe, J.F.; Lemieux, P.; Predel, H.G.; Berthold, H.K. The whey fermentation product malleable protein matrix decreases TAG concentrations in patients with the metabolic syndrome: A randomised placebo-controlled trial. Br. J. Nutr. 2012, 107, 1694–1706. [Google Scholar] [CrossRef]
- Arciero, P.J.; Edmonds, R.C.; Bunsawat, K.; Gentile, C.L.; Ketcham, C.; Darin, C.; Renna, M.; Zheng, Q.; Zhang, J.Z.; Ormsbee, M.J. Protein-Pacing from Food or Supplementation Improves Physical Performance in Overweight Men and Women: The PRISE 2 Study. Nutrients 2016, 8, 288. [Google Scholar] [CrossRef]
- Larsen, A.E.; Bibby, B.M.; Hansen, M. Effect of a Whey Protein Supplement on Preservation of Fat Free Mass in Overweight and Obese Individuals on an Energy Restricted Very Low Caloric Diet. Nutrients 2018, 10, 1918. [Google Scholar] [CrossRef] [PubMed]
- Demling, R.H.; DeSanti, L. Effect of a hypocaloric diet, increased protein intake and resistance training on lean mass gains and fat mass loss in overweight police officers. Ann. Nutr. Metab. 2000, 44, 21–29. [Google Scholar] [CrossRef] [PubMed]
- Grey, V.; Mohammed, S.R.; Smountas, A.A.; Bahlool, R.; Lands, L.C. Improved glutathione status in young adult patients with cystic fibrosis supplemented with whey protein. J. Cyst. Fibros. 2003, 2, 195–198. [Google Scholar] [CrossRef] [PubMed]
- Lefferts, W.K.; Augustine, J.A.; Spartano, N.L.; Hughes, W.E.; Babcock, M.C.; Heenan, B.K.; Heffernan, K.S. Effects of Whey Protein Supplementation on Aortic Stiffness, Cerebral Blood Flow, and Cognitive Function in Community-Dwelling Older Adults: Findings from the ANCHORS A-WHEY Clinical Trial. Nutrients 2020, 12, 1054. [Google Scholar] [CrossRef]
- Hudson, J.L.; Zhou, J.; Kim, J.E.; Campbell, W.W. Incorporating Milk Protein Isolate into an Energy-Restricted Western-Style Eating Pattern Augments Improvements in Blood Pressure and Triglycerides, but Not Body Composition Changes in Adults Classified as Overweight or Obese: A Randomized Controlled Trial. Nutrients 2020, 12, 851. [Google Scholar] [CrossRef] [PubMed]
- Fuglsang-Nielsen, R.; Rakvaag, E.; Langdahl, B.; Knudsen, K.E.B.; Hartmann, B.; Holst, J.J.; Hermansen, K.; Gregersen, S. Effects of whey protein and dietary fiber intake on insulin sensitivity, body composition, energy expenditure, blood pressure, and appetite in subjects with abdominal obesity. Eur. J. Clin. Nutr. 2021, 75, 611–619. [Google Scholar] [CrossRef]
- Weinheimer, E.M.; Conley, T.B.; Kobza, V.M.; Sands, L.P.; Lim, E.; Janle, E.M.; Campbell, W.W. Whey Protein Supplementation Does Not Affect Exercise Training–Induced Changes in Body Composition and Indices of Metabolic Syndrome in Middle-Aged Overweight and Obese Adults. J. Nutr. 2012, 142, 1532–1539. [Google Scholar] [CrossRef]
- Kjølbæk, L.; Sørensen, L.B.; Søndertoft, N.B.; Rasmussen, C.K.; Lorenzen, J.K.; Serena, A.; Astrup, A.; Larsen, L.H. Protein supplements after weight loss do not improve weight maintenance compared with recommended dietary protein intake despite beneficial effects on appetite sensation and energy expenditure: A randomized, controlled, double-blinded trial. Am. J. Clin. Nutr. 2017, 106, 684–697. [Google Scholar] [CrossRef]
- Jeong, J.H.; Biruete, A.; Tomayko, E.J.; Wu, P.T.; Fitschen, P.; Chung, H.R.; Ali, M.; McAuley, E.; Fernhall, B.; Phillips, S.A. Results from the randomized controlled IHOPE trial suggest no effects of oral protein supplementation and exercise training on physical function in hemodialysis patients. Kidney Int. 2019, 96, 777–786. [Google Scholar] [CrossRef]
- Yang, J.; Wang, H.P.; Tong, X.; Li, Z.N.; Xu, J.Y.; Zhou, L.; Zhou, B.Y.; Qin, L.Q. Effect of whey protein on blood pressure in pre-and mildly hypertensive adults: A randomized controlled study. Food Sci. Nutr. 2019, 7, 1857–1864. [Google Scholar] [CrossRef]
- Frestedt, J.L.; Zenk, J.L.; Kuskowski, M.A.; Ward, L.S.; Bastian, E.D. A whey-protein supplement increases fat loss and spares lean muscle in obese subjects: A randomized human clinical study. Nutr. Metab. 2008, 5, 8. [Google Scholar] [CrossRef] [PubMed]
- Silva, L.B.d.C.; Mourão, L.F.; Silva, A.A.; Lima, N.M.F.V.; Almeida, S.R.; Franca, M.C., Jr.; Nucci, A.; Amaya-Farfán, J. Effect of nutritional supplementation with milk whey proteins in amyotrophic lateral sclerosis patients. Arq. Neuro-Psiquiatr. 2010, 68, 263–268. [Google Scholar] [CrossRef] [PubMed]
- Sohrabi, Z.; Eftekhari, M.H.; Eskandari, M.H.; Rezaianzadeh, A.; Sagheb, M.M. Intradialytic oral protein supplementation and nutritional and inflammation outcomes in hemodialysis: A randomized controlled trial. Am. J. Kidney Dis. 2016, 68, 122–130. [Google Scholar] [CrossRef]
- Sharp, M.H.; Lowery, R.P.; Shields, K.A.; Lane, J.R.; Gray, J.L.; Partl, J.M.; Hayes, D.W.; Wilson, G.J.; Hollmer, C.A.; Minivich, J.R. The effects of beef, chicken, or whey protein after workout on body composition and muscle performance. J. Strength Cond. Res. 2018, 32, 2233–2242. [Google Scholar] [CrossRef]
- Bumrungpert, A.; Pavadhgul, P.; Nunthanawanich, P.; Sirikanchanarod, A.; Adulbhan, A. Whey protein supplementation improves nutritional status, glutathione levels, and immune function in cancer patients: A randomized, double-blind controlled trial. J. Med. Food 2018, 21, 612–616. [Google Scholar] [CrossRef]
- Derosa, G.; D’angelo, A.; Maffioli, P. Change of some oxidative stress parameters after supplementation with whey protein isolate in patients with type 2 diabetes. Nutrition 2020, 73, 110700. [Google Scholar] [CrossRef] [PubMed]
- Ahmadi, A.; Eftekhari, M.H.; Mazloom, Z.; Masoompour, M.; Fararooei, M.; Eskandari, M.H.; Mehrabi, S.; Bedeltavana, A.; Famouri, M.; Zare, M. Fortified whey beverage for improving muscle mass in chronic obstructive pulmonary disease: A single-blind, randomized clinical trial. Respir. Res. 2020, 21, 216. [Google Scholar] [CrossRef]
- Gryson, C.; Ratel, S.; Rance, M.; Penando, S.; Bonhomme, C.; Le Ruyet, P.; Duclos, M.; Boirie, Y.; Walrand, S. Four-month course of soluble milk proteins interacts with exercise to improve muscle strength and delay fatigue in elderly participants. J. Am. Med. Dir. Assoc. 2014, 15, 958.e1–958.e9. [Google Scholar] [CrossRef] [PubMed]
- Keogh, J.B.; Clifton, P. The effect of meal replacements high in glycomacropeptide on weight loss and markers of cardiovascular disease risk. Am. J. Clin. Nutr. 2008, 87, 1602–1605. [Google Scholar] [CrossRef] [PubMed]
- Rambousková, J.; Procházka, B.; Binder, M.; Anděl, M. Effect of the liquid milk nutritional supplement with enhanced content of whey protein on the nutritional status of the elderly. Vnitr. Lek. 2014, 60, 556–561. [Google Scholar]
- Sattler, F.R.; Rajicic, N.; Mulligan, K.; Yarasheski, K.E.; Koletar, S.L.; Zolopa, A.; Smith, B.A.; Zackin, R.; Bistrian, B. Evaluation of high-protein supplementation in weight-stable HIV-positive subjects with a history of weight loss: A randomized, double-blind, multicenter trial. Am. J. Clin. Nutr. 2008, 88, 1313–1321. [Google Scholar] [CrossRef]
- Baer, D.J.; Stote, K.S.; Paul, D.R.; Harris, G.K.; Rumpler, W.V.; Clevidence, B.A. Whey protein but not soy protein supplementation alters body weight and composition in free-living overweight and obese adults. J. Nutr. 2011, 141, 1489–1494. [Google Scholar] [CrossRef]
- Ottestad, I.; Løvstad, A.; Gjevestad, G.O.; Hamarsland, H.; Benth, J.Š.; Andersen, L.F.; Bye, A.; Biong, A.S.; Retterstøl, K.; Iversen, P.O. Intake of a protein-enriched milk and effects on muscle mass and strength. A 12-week randomized placebo controlled trial among community-dwelling older adults. J. Nutr. Health Aging 2017, 21, 1160–1169. [Google Scholar] [CrossRef]
- Sugawara, K.; Takahashi, H.; Kashiwagura, T.; Yamada, K.; Yanagida, S.; Homma, M.; Dairiki, K.; Sasaki, H.; Kawagoshi, A.; Satake, M. Effect of anti-inflammatory supplementation with whey peptide and exercise therapy in patients with COPD. Respir. Med. 2012, 106, 1526–1534. [Google Scholar] [CrossRef]
- Björkman, M.; Finne-Soveri, H.; Tilvis, R. Whey protein supplementation in nursing home residents. A randomized controlled trial. Eur. Geriatr. Med. 2012, 3, 161–166. [Google Scholar] [CrossRef]
- Volek, J.S.; Volk, B.M.; Gómez, A.L.; Kunces, L.J.; Kupchak, B.R.; Freidenreich, D.J.; Aristizabal, J.C.; Saenz, C.; Dunn-Lewis, C.; Ballard, K.D. Whey protein supplementation during resistance training augments lean body mass. J. Am. Coll. Nutr. 2013, 32, 122–135. [Google Scholar] [CrossRef]
- Chalé, A.; Cloutier, G.J.; Hau, C.; Phillips, E.M.; Dallal, G.E.; Fielding, R.A. Efficacy of whey protein supplementation on resistance exercise–induced changes in lean mass, muscle strength, and physical function in mobility-limited older adults. J. Gerontol. Ser. A Biomed. Sci. Med. Sci. 2013, 68, 682–690. [Google Scholar] [CrossRef]
- Duff, W.R.; Chilibeck, P.D.; Rooke, J.J.; Kaviani, M.; Krentz, J.R.; Haines, D.M. The effect of bovine colostrum supplementation in older adults during resistance training. Int. J. Sport Nutr. Exerc. Metab. 2014, 24, 276–285. [Google Scholar] [CrossRef]
- Kerstetter, J.E.; Bihuniak, J.D.; Brindisi, J.; Sullivan, R.R.; Mangano, K.M.; Larocque, S.; Kotler, B.M.; Simpson, C.A.; Cusano, A.M.; Gaffney-Stomberg, E. The effect of a whey protein supplement on bone mass in older Caucasian adults. J. Clin. Endocrinol. Metab. 2015, 100, 2214–2222. [Google Scholar] [CrossRef] [PubMed]
- Hector, A.J.; Marcotte, G.R.; Churchward-Venne, T.A.; Murphy, C.H.; Breen, L.; von Allmen, M.; Baker, S.K.; Phillips, S.M. Whey protein supplementation preserves postprandial myofibrillar protein synthesis during short-term energy restriction in overweight and obese adults. J. Nutr. 2015, 145, 246–252. [Google Scholar] [CrossRef] [PubMed]
- Malekian, F.; Gebrelul, S.S.; Henson, J.F.; Cyrus, K.D.; Goita, M.; Kennedy, B.M. The efects of whey protein and resistant starch on body weight. Funct. Foods Health Dis. 2015, 5, 275–291. [Google Scholar] [CrossRef]
- Reimer, R.A.; Willis, H.J.; Tunnicliffe, J.M.; Park, H.; Madsen, K.L.; Soto-Vaca, A. Inulin-type fructans and whey protein both modulate appetite but only fructans alter gut microbiota in adults with overweight/obesity: A randomized controlled trial. Mol. Nutr. Food Res. 2017, 61, 1700484. [Google Scholar] [CrossRef]
- Hassan, K.; Hassan, F. Does whey protein supplementation affect blood pressure in hypoalbuminemic peritoneal dialysis patients? Ther. Clin. Risk Manag. 2017, 13, 989–997. [Google Scholar] [CrossRef]
- Hassan, K. Does whey protein supplementation improve the nutritional status in hypoalbuminemic peritoneal dialysis patients? Ther. Apher. Dial. 2017, 21, 485–492. [Google Scholar] [CrossRef]
- Dirks, M.L.; Tieland, M.; Verdijk, L.B.; Losen, M.; Nilwik, R.; Mensink, M.; de Groot, L.C.; van Loon, L.J. Protein supplementation augments muscle fiber hypertrophy but does not modulate satellite cell content during prolonged resistance-type exercise training in frail elderly. J. Am. Med. Dir. Assoc. 2017, 18, 608–615. [Google Scholar] [CrossRef]
- Gjevestad, G.O.; Ottestad, I.; Biong, A.S.; Iversen, P.O.; Retterstøl, K.; Raastad, T.; Skålhegg, B.S.; Ulven, S.M.; Holven, K.B. Consumption of protein-enriched milk has minor effects on inflammation in older adults—A 12-week double-blind randomized controlled trial. Mech. Ageing Dev. 2017, 162, 1–8. [Google Scholar] [CrossRef]
- Englund, D.A.; Kirn, D.R.; Koochek, A.; Zhu, H.; Travison, T.G.; Reid, K.F.; von Berens, Å.; Melin, M.; Cederholm, T.; Gustafsson, T. Nutritional supplementation with physical activity improves muscle composition in mobility-limited older adults, the VIVE2 study: A randomized, double-blind, placebo-controlled trial. J. Gerontol. Ser. A 2018, 73, 95–101. [Google Scholar] [CrossRef]
- Sahathevan, S.; Se, C.-H.; Ng, S.; Khor, B.-H.; Chinna, K.; Goh, B.L.; Gafor, H.A.; Bavanandan, S.; Ahmad, G.; Karupaiah, T. Clinical efficacy and feasibility of whey protein isolates supplementation in malnourished peritoneal dialysis patients: A multicenter, parallel, open-label randomized controlled trial. Clin. Nutr. ESPEN 2018, 25, 68–77. [Google Scholar] [CrossRef]
- Amasene, M.; Besga, A.; Echeverria, I.; Urquiza, M.; Ruiz, J.R.; Rodriguez-Larrad, A.; Aldamiz, M.; Anaut, P.; Irazusta, J.; Labayen, I. Effects of Leucine-enriched whey protein supplementation on physical function in post-hospitalized older adults participating in 12-weeks of resistance training program: A randomized controlled trial. Nutrients 2019, 11, 2337. [Google Scholar] [CrossRef] [PubMed]
- Cereda, E.; Turri, A.; Klersy, C.; Cappello, S.; Ferrari, A.; Filippi, A.R.; Brugnatelli, S.; Caraccia, M.; Chiellino, S.; Borioli, V. Whey protein isolate supplementation improves body composition, muscle strength, and treatment tolerance in malnourished advanced cancer patients undergoing chemotherapy. Cancer Med. 2019, 8, 6923–6932. [Google Scholar] [CrossRef] [PubMed]
- Ten Haaf, D.S.; Eijsvogels, T.M.; Bongers, C.C.; Horstman, A.M.; Timmers, S.; de Groot, L.C.; Hopman, M.T. Protein supplementation improves lean body mass in physically active older adults: A randomized placebo-controlled trial. J. Cachexia Sarcopenia Muscle 2019, 10, 298–310. [Google Scholar] [CrossRef] [PubMed]
- Kang, L.; Gao, Y.; Liu, X.; Liang, Y.; Chen, Y.; Liang, Y.; Zhang, L.; Chen, W.; Pang, H.; Peng, L.-N. Effects of whey protein nutritional supplement on muscle function among community-dwelling frail older people: A multicenter study in China. Arch. Gerontol. Geriatr. 2019, 83, 7–12. [Google Scholar] [CrossRef]
- Rakvaag, E.; Fuglsang-Nielsen, R.; Bach Knudsen, K.E.; Landberg, R.; Johannesson Hjelholt, A.; Søndergaard, E.; Hermansen, K.; Gregersen, S. Whey protein combined with low dietary fiber improves lipid profile in subjects with abdominal obesity: A randomized, controlled trial. Nutrients 2019, 11, 2091. [Google Scholar] [CrossRef]
- Lynch, H.M.; Buman, M.P.; Dickinson, J.M.; Ransdell, L.B.; Johnston, C.S.; Wharton, C.M. No significant differences in muscle growth and strength development when consuming soy and whey protein supplements matched for leucine following a 12 week resistance training program in men and women: A randomized trial. Int. J. Environ. Res. Public Health 2020, 17, 3871. [Google Scholar] [CrossRef]
- Boutry-Regard, C.; Vinyes-Parés, G.; Breuillé, D.; Moritani, T. Supplementation with whey protein, omega-3 fatty acids and polyphenols combined with electrical muscle stimulation increases muscle strength in elderly adults with limited mobility: A randomized controlled trial. Nutrients 2020, 12, 1866. [Google Scholar] [CrossRef]
- Nakayama, K.; Saito, Y.; Sanbongi, C.; Murata, K.; Urashima, T. Effects of low-dose milk protein supplementation following low-to-moderate intensity exercise training on muscle mass in healthy older adults: A randomized placebo-controlled trial. Eur. J. Nutr. 2021, 60, 917–928. [Google Scholar] [CrossRef]
- Azhar, G.; Wei, J.Y.; Schutzler, S.E.; Coker, K.; Gibson, R.V.; Kirby, M.F.; Ferrando, A.A.; Wolfe, R.R. Daily consumption of a specially formulated essential amino acid-based dietary supplement improves physical performance in older adults with low physical functioning. J. Gerontol. Ser. A 2021, 76, 1184–1191. [Google Scholar] [CrossRef]
- Li, C.; Meng, H.; Wu, S.; Fang, A.; Liao, G.; Tan, X.; Chen, P.; Wang, X.; Chen, S.; Zhu, H. Daily supplementation with whey, soy, or whey-soy blended protein for 6 months maintained lean muscle mass and physical performance in older adults with low lean mass. J. Acad. Nutr. Diet. 2021, 121, 1035–1048.e1036. [Google Scholar] [CrossRef]
- Mizubuti, Y.; Vieira, E.; Silva, T.; d’Alessandro, M.; Generoso, S.; Teixeira, A.; Lima, A.; Correia, M. Comparing the effects of whey and casein supplementation on nutritional status and immune parameters in patients with chronic liver disease: A randomised double-blind controlled trial. Br. J. Nutr. 2021, 125, 768–779. [Google Scholar] [CrossRef] [PubMed]
- Mertz, K.H.; Reitelseder, S.; Bechshoeft, R.; Bulow, J.; Højfeldt, G.; Jensen, M.; Schacht, S.R.; Lind, M.V.; Rasmussen, M.A.; Mikkelsen, U.R. The effect of daily protein supplementation, with or without resistance training for 1 year, on muscle size, strength, and function in healthy older adults: A randomized controlled trial. Am. J. Clin. Nutr. 2021, 113, 790–800. [Google Scholar] [CrossRef] [PubMed]
- de Azevedo Bach, S.; Radaelli, R.; Schemes, M.B.; Neske, R.; Garbelotto, C.; Roschel, H.; Pinto, R.S.; Schneider, C.D. Can supplemental protein to low-protein containing meals superimpose on resistance-training muscle adaptations in older adults? A randomized clinical trial. Exp. Gerontol. 2022, 162, 111760. [Google Scholar] [CrossRef] [PubMed]
- Zong, M.; Shen, H.; Ren, L.; Han, T.; Chen, J.; Chen, Y.; Lu, J.; Zhang, Y.; Li, S.; Sun, J. Effects of whey protein complex combined with low-intensity exercise in elderly inpatients with COPD at a stable stage. Asia Pac. J. Clin. Nutr. 2023, 32, 375. [Google Scholar] [PubMed]
- de Carvalho Furtado, C.; Jamar, G.; Barbosa, A.C.B.; Dourado, V.Z.; do Nascimento, J.R.; de Oliveira, G.C.A.F.; Hi, E.M.B.; de Arruda Souza, T.; Parada, M.J.G.; de Souza, F.G. Whey Protein Supplementation in Older Adults with Type 2 Diabetes Undergoing a Resistance Training Program: A Double-Blind Randomized Controlled Trial. J. Aging Phys. Act. 2024, 33, 101–113. [Google Scholar]
- Dos Santos, E.M.; Moreira, A.S.; Huguenin, G.V.; Tibiriça, E.; De Lorenzo, A. Effects of Whey Protein Isolate on Body Composition, Muscle Mass, and Strength of Chronic Heart Failure Patients: A Randomized Clinical Trial. Nutrients 2023, 15, 2320. [Google Scholar] [CrossRef]
- Hodgson, J.M.; Zhu, K.; Lewis, J.R.; Kerr, D.; Meng, X.; Solah, V.; Devine, A.; Binns, C.W.; Woodman, R.J.; Prince, R.L. Long-term effects of a protein-enriched diet on blood pressure in older women. Br. J. Nutr. 2012, 107, 1664–1672. [Google Scholar] [CrossRef]
- Agin, D.; Gallagher, D.; Wang, J.; Heymsfield, S.B.; Pierson, R.N., Jr.; Kotler, D.P. Effects of whey protein and resistance exercise on body cell mass, muscle strength, and quality of life in women with HIV. Aids 2001, 15, 2431–2440. [Google Scholar] [CrossRef]
- Figueroa, A.; Wong, A.; Kinsey, A.; Kalfon, R.; Eddy, W.; Ormsbee, M.J. Effects of milk proteins and combined exercise training on aortic hemodynamics and arterial stiffness in young obese women with high blood pressure. Am. J. Hypertens. 2014, 27, 338–344. [Google Scholar] [CrossRef] [PubMed]
- Nabuco, H.C.G.; Tomeleri, C.M.; Fernandes, R.R.; Sugihara Junior, P.; Cavalcante, E.F.; Cunha, P.M.; Antunes, M.; Nunes, J.P.; Venturini, D.; Barbosa, D.S.; et al. Effect of whey protein supplementation combined with resistance training on body composition, muscular strength, functional capacity, and plasma-metabolism biomarkers in older women with sarcopenic obesity: A randomized, double-blind, placebo-controlled trial. Clin. Nutr. ESPEN 2019, 32, 88–95. [Google Scholar] [CrossRef] [PubMed]
- Ormsbee, M.J.; Kinsey, A.W.; Eddy, W.R.; Madzima, T.A.; Arciero, P.J.; Figueroa, A.; Panton, L.B. The influence of nighttime feeding of carbohydrate or protein combined with exercise training on appetite and cardiometabolic risk in young obese women. Appl. Physiol. Nutr. Metab. 2015, 40, 37–45. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Ling, C.; Liu, L.; Zhang, J.; Wang, J.; Tong, X.; Hidayat, K.; Chen, M.; Chen, X.; Zhou, H. Effects of whey protein or its hydrolysate supplements combined with an energy-restricted diet on weight loss: A randomized controlled trial in older women. Nutrients 2022, 14, 4540. [Google Scholar] [CrossRef]
- Nouri, M.; Tarighat-Esfanjani, A.; Sadra, V.; Ghasempour, Z.; Jafarabadi, M.A.; Tajfar, P.; Gargari, B.P. Effects of Whey Protein Concentrate on Glycemic Status, Lipid Profile, and Blood Pressure in Overweight/Obese Women with Type 2 Diabetes Mellitus: A Randomized Placebo-Controlled Clinical Trial. Turk. J. Endocrinol. Metab. 2022, 26, 148. [Google Scholar] [CrossRef]
- Nabuco, H.C.; Tomeleri, C.M.; Fernandes, R.R.; Junior, P.S.; Cavalcante, E.F.; Venturini, D.; Barbosa, D.S.; Silva, A.M.; Sardinha, L.B.; Cyrino, E.S. Effects of protein intake beyond habitual intakes associated with resistance training on metabolic syndrome-related parameters, isokinetic strength, and body composition in older women. J. Aging Phys. Act. 2019, 27, 545–552. [Google Scholar] [CrossRef]
- Fernandes, R.R.; Nabuco, H.C.; Junior, P.S.; Cavalcante, E.F.; Fabro, P.M.; Tomeleri, C.M.; Ribeiro, A.S.; Barbosa, D.S.; Venturini, D.; Schoenfeld, B.J. Effect of protein intake beyond habitual intakes following resistance training on cardiometabolic risk disease parameters in pre-conditioned older women. Exp. Gerontol. 2018, 110, 9–14. [Google Scholar] [CrossRef]
- Piccolo, B.D.; Comerford, K.B.; Karakas, S.E.; Knotts, T.A.; Fiehn, O.; Adams, S.H. Whey protein supplementation does not alter plasma branched-chained amino acid profiles but results in unique metabolomics patterns in obese women enrolled in an 8-week weight loss trial. J. Nutr. 2015, 145, 691–700. [Google Scholar] [CrossRef]
- Josse, A.R.; Tang, J.E.; Tarnopolsky, M.A.; Phillips, S.M. Body composition and strength changes in women with milk and resistance exercise. Med. Sci. Sports Exerc. 2010, 42, 1122–1130. [Google Scholar] [CrossRef]
- Mojtahedi, M.C.; Thorpe, M.P.; Karampinos, D.C.; Johnson, C.L.; Layman, D.K.; Georgiadis, J.G.; Evans, E.M. The effects of a higher protein intake during energy restriction on changes in body composition and physical function in older women. J. Gerontol. Ser. A Biomed. Sci. Med. Sci. 2011, 66, 1218–1225. [Google Scholar] [CrossRef]
- Elahikhah, M.; Haidari, F.; Khalesi, S.; Shahbazian, H.; Mohammadshahi, M.; Aghamohammadi, V. Milk protein concentrate supplementation improved appetite, metabolic parameters, adipocytokines, and body composition in dieting women with obesity: A randomized controlled trial. BMC Nutr. 2024, 10, 80. [Google Scholar] [CrossRef] [PubMed]
- Giglio, B.M.; Schincaglia, R.M.; da Silva, A.S.; Fazani, I.C.; Monteiro, P.A.; Mota, J.F.; Cunha, J.P.; Pichard, C.; Pimentel, G.D. Whey protein supplementation compared to collagen increases blood Nesfatin concentrations and decreases android fat in overweight women: A randomized double-blind study. Nutrients 2019, 11, 2051. [Google Scholar] [CrossRef] [PubMed]
- Haidari, F.; Aghamohammadi, V.; Mohammadshahi, M.; Ahmadi-Angali, K.; Asghari-Jafarabadi, M. Whey protein supplementation reducing fasting levels of anandamide and 2-AG without weight loss in pre-menopausal women with obesity on a weight-loss diet. Trials 2020, 21, 657. [Google Scholar] [CrossRef] [PubMed]
- Lopes Gomes, D.; Moehlecke, M.; Lopes da Silva, F.B.; Dutra, E.S.; D’Agord Schaan, B.; Baiocchi de Carvalho, K.M. Whey protein supplementation enhances body fat and weight loss in women long after bariatric surgery: A randomized controlled trial. Obes. Surg. 2017, 27, 424–431. [Google Scholar] [CrossRef]
- Zhu, K.; Kerr, D.A.; Meng, X.; Devine, A.; Solah, V.; Binns, C.W.; Prince, R.L. Two-year whey protein supplementation did not enhance muscle mass and physical function in well-nourished healthy older postmenopausal women. J. Nutr. 2015, 145, 2520–2526. [Google Scholar] [CrossRef]
- Taylor, L.W.; Wilborn, C.; Roberts, M.D.; White, A.; Dugan, K. Eight weeks of pre-and postexercise whey protein supplementation increases lean body mass and improves performance in Division III collegiate female basketball players. Appl. Physiol. Nutr. Metab. 2016, 41, 249–254. [Google Scholar] [CrossRef]
- Mori, H.; Tokuda, Y. Effect of whey protein supplementation after resistance exercise on the muscle mass and physical function of healthy older women: A randomized controlled trial. Geriatr. Gerontol. Int. 2018, 18, 1398–1404. [Google Scholar] [CrossRef]
- Brown, A.F.; Welsh, T.; Panton, L.B.; Moffatt, R.J.; Ormsbee, M.J. Higher-protein intake improves body composition index in female collegiate dancers. Appl. Physiol. Nutr. Metab. 2020, 45, 547–554. [Google Scholar] [CrossRef]
- Mori, H.; Tokuda, Y. Effect of whey protein supplementation after resistance exercise on the treatment of sarcopenia and quality of life among older women with sarcopenia: A randomized controlled trial. Jpn. J. Phys. Fit. Sports Med. 2021, 70, 207–218. [Google Scholar] [CrossRef]
- Biesek, S.; Vojciechowski, A.S.; Filho, J.M.; Menezes Ferreira, A.C.R.d.; Borba, V.Z.C.; Rabito, E.I.; Gomes, A.R.S. Effects of exergames and protein supplementation on body composition and musculoskeletal function of prefrail community-dwelling older women: A randomized, controlled clinical trial. Int. J. Environ. Res. Public Health 2021, 18, 9324. [Google Scholar] [CrossRef]
- Henriques, H.K.F.; Kattah, F.M.; Piccolo, M.S.; Rosa, C.d.O.B.; de Araújo Ventura, L.H.; Cerqueira, F.R.; Vieira, C.M.A.F.; Leite, J.I.A. Effect of whey protein supplementation on body composition of patients undergoing bariatric surgery. J. Arch. Health 2023, 4, 44–55. [Google Scholar] [CrossRef]
- Nouri, M.; Gargari, B.P.; Ghasempour, Z.; Sadra, V.; Jafarabadi, M.A.; Babaei, A.; Tajfar, P.; Tarighat-Esfanjani, A. The effects of whey protein on anthropometric parameters, resting energy expenditure, oxidative stress, and appetite in overweight/obese women with type 2 diabetes mellitus: A randomized placebo controlled clinical trial. Int. J. Diabetes Dev. Ctries. 2024, 44, 155–166. [Google Scholar] [CrossRef]
- Kasim-Karakas, S.E.; Almario, R.U.; Cunningham, W. Effects of protein versus simple sugar intake on weight loss in polycystic ovary syndrome (according to the National Institutes of Health criteria). Fertil. Steril. 2009, 92, 262–270. [Google Scholar] [CrossRef]
- Ahmadi Kani Golzar, F.; Sheikholeslami Vatani, D.; Mojtahedi, H.; Marandi, S.M. The effects of whey protein isolate supplementation and resistance training on cardiovascular risk factors in overweight young men. J. Isfahan Med. Sch. 2012, 30, 289–301. [Google Scholar]
- Sheikholeslami Vatani, D.; Ahmadi Kani Golzar, F. Changes in antioxidant status and cardiovascular risk factors of overweight young men after six weeks supplementation of whey protein isolate and resistance training. Appetite 2012, 59, 673–678. [Google Scholar] [CrossRef] [PubMed]
- Tahavorgar, A.; Vafa, M.R.; Shidfar, F.; Gohari, M.; Heydari, I. Beneficial Effects of Whey Protein Preloads on some Cardiovascular Diseases Risk Factors of Overweight and Obese Men are Stronger than Soy Protein Preloads—A Randomized Clinical Trial. J. Nutr. Intermed. Metab. 2015, 2, 69–75. [Google Scholar] [CrossRef]
- Moon, J.M.; Ratliff, K.M.; Blumkaitis, J.C.; Harty, P.S.; Zabriskie, H.A.; Stecker, R.A.; Currier, B.S.; Jagim, A.R.; Jäger, R.; Purpura, M. Effects of daily 24-gram doses of rice or whey protein on resistance training adaptations in trained males. J. Int. Soc. Sports Nutr. 2020, 17, 60. [Google Scholar] [CrossRef]
- Kataoka, Y.; Kamijo, Y.-i.; Ogawa, Y.; Sumiyoshi, E.; Nakae, M.; Ikegawa, S.; Manabe, K.; Morikawa, M.; Nagata, M.; Takasugi, S. Effects of hypervolemia by protein and glucose supplementation during aerobic training on thermal and arterial pressure regulations in hypertensive older men. J. Appl. Physiol. 2016, 121, 1021–1031. [Google Scholar] [CrossRef]
- Burke, D.G.; Chilibeck, P.D.; Davison, K.S.; Candow, D.C.; Farthing, J.; Smith-Palmer, T. The effect of whey protein supplementation with and without creatine monohydrate combined with resistance training on lean tissue mass and muscle strength. Int. J. Sport Nutr. Exerc. Metab. 2001, 11, 349–364. [Google Scholar] [CrossRef] [PubMed]
- Rankin, J.W.; Goldman, L.P.; Puglisi, M.J.; Nickols-Richardson, S.M.; Earthman, C.P.; Gwazdauskas, F.C. Effect of post-exercise supplement consumption on adaptations to resistance training. J. Am. Coll. Nutr. 2004, 23, 322–330. [Google Scholar] [CrossRef] [PubMed]
- Samadi, M.; Soleimani, D.; Moradi, M.; Kamari, N.; Rezaei, M.; Hozoori, M. The Effect of Whey Protein Supplementation on Inflammatory Markers and Muscle Damage in Basketball Athletes: A Double-Blinded Clinical Trial. J. Isfahan Med. Sch. 2021, 39, 659–664. [Google Scholar]
- Teixeira, F.J.; Matias, C.N.; Faleiro, J.; Giro, R.; Pires, J.; Figueiredo, H.; Carvalhinho, R.; Monteiro, C.P.; Reis, J.F.; Valamatos, M.J. A novel plant-based protein has similar effects compared to whey protein on body composition, strength, power, and aerobic performance in professional and semi-professional futsal players. Front. Nutr. 2022, 9, 934438. [Google Scholar] [CrossRef]
- Pettersson, S.; Edin, F.; Hjelte, C.; Scheinost, D.; Wagner, S.; Ekblom, B.; Jessen, N.; Madsen, K.; Andersson-Hall, U. Six weeks of aerobic exercise in untrained men with overweight/obesity improved training adaptations, performance and body composition independent of oat/potato or milk based protein-carbohydrate drink supplementation. Front. Nutr. 2021, 8, 617344. [Google Scholar] [CrossRef]
- Hulmi, J.J.; Laakso, M.; Mero, A.A.; Häkkinen, K.; Ahtiainen, J.P.; Peltonen, H. The effects of whey protein with or without carbohydrates on resistance training adaptations. J. Int. Soc. Sports Nutr. 2015, 12, 48. [Google Scholar] [CrossRef]
- Maltais, M.L.; Perreault, K.; Courchesne-Loyer, A.; Lagacé, J.-C.; Barsalani, R.; Dionne, I.J. Effect of resistance training and various sources of protein supplementation on body fat mass and metabolic profile in sarcopenic overweight older adult men: A pilot study. Int. J. Sport Nutr. Exerc. Metab. 2016, 26, 71–77. [Google Scholar] [CrossRef] [PubMed]
- Brown, E.C.; DiSilvestro, R.A.; Babaknia, A.; Devor, S.T. Soy versus whey protein bars: Effects on exercise training impact on lean body mass and antioxidant status. Nutr. J. 2004, 3, 22. [Google Scholar] [CrossRef]
- Hartman, J.W.; Tang, J.E.; Wilkinson, S.B.; Tarnopolsky, M.A.; Lawrence, R.L.; Fullerton, A.V.; Phillips, S.M. Consumption of fat-free fluid milk after resistance exercise promotes greater lean mass accretion than does consumption of soy or carbohydrate in young, novice, male weightlifters. Am. J. Clin. Nutr. 2007, 86, 373–381. [Google Scholar] [CrossRef]
- Cribb, P.J.; Williams, A.D.; Stathis, C.; Carey, M.F.; Hayes, A. Effects of whey isolate, creatine and resistance training on muscle hypertrophy. Med. Sci. Sports Exerc. 2007, 39, 298–307. [Google Scholar] [CrossRef]
- Eliot, K.; Knehans, A.; Bemben, D.; Witten, M.; Carter, J.; Bemben, M. The effects of creatine and whey protein supplementation on body composition in men aged 48 to 72 years during resistance training. J. Nutr. Health Aging 2008, 12, 208–212. [Google Scholar] [CrossRef]
- Arazi, H.; Hakimi, M.; Hoseini, K. The effects of Whey protein supplementation on Performance and hormonal adaptations following resistance training in Novice Men. Balt. J. Health Phys. Act. 2011, 3, 87–95. [Google Scholar] [CrossRef]
- DeNysschen, C.A.; Burton, H.W.; Horvath, P.J.; Leddy, J.J.; Browne, R.W. Resistance training with soy vs whey protein supplements in hyperlipidemic males. J. Int. Soc. Sports Nutr. 2009, 6, 8. [Google Scholar] [CrossRef]
- Hambre, D.; Vergara, M.; Lood, Y.; Bachrach-Lindström, M.; Lindström, T.; Nystrom, F.H. A randomized trial of protein supplementation compared with extra fast food on the effects of resistance training to increase metabolism. Scand. J. Clin. Lab. Investig. 2012, 72, 471–478. [Google Scholar] [CrossRef]
- Joy, J.M.; Lowery, R.P.; Wilson, J.M.; Purpura, M.; De Souza, E.O.; Wilson, S.M.; Kalman, D.S.; Dudeck, J.E.; Jäger, R. The effects of 8 weeks of whey or rice protein supplementation on body composition and exercise performance. Nutr. J. 2013, 12, 86. [Google Scholar] [CrossRef] [PubMed]
- Herda, A.A.; Herda, T.J.; Costa, P.B.; Ryan, E.D.; Stout, J.R.; Cramer, J.T. Muscle performance, size, and safety responses after eight weeks of resistance training and protein supplementation: A randomized, double-blinded, placebo-controlled clinical trial. J. Strength Cond. Res. 2013, 27, 3091–3100. [Google Scholar] [CrossRef] [PubMed]
- Babault, N.; Deley, G.; Le Ruyet, P.; Morgan, F.; Allaert, F.A. Effects of soluble milk protein or casein supplementation on muscle fatigue following resistance training program: A randomized, double-blind, and placebo-controlled study. J. Int. Soc. Sports Nutr. 2014, 11, 36. [Google Scholar] [CrossRef]
- Hulmi, J.J.; Tannerstedt, J.; Selanne, H.; Kainulainen, H.; Kovanen, V.; Mero, A.A. Resistance exercise with whey protein ingestion affects mTOR signaling pathway and myostatin in men. J. Appl. Physiol. 2009, 106, 1720–1729. [Google Scholar] [CrossRef]
- Reidy, P.T.; Borack, M.S.; Markofski, M.M.; Dickinson, J.M.; Deer, R.R.; Husaini, S.H.; Walker, D.K.; Igbinigie, S.; Robertson, S.M.; Cope, M.B. Protein supplementation has minimal effects on muscle adaptations during resistance exercise training in young men: A double-blind randomized clinical trial. J. Nutr. 2016, 146, 1660–1669. [Google Scholar] [CrossRef]
- Naclerio, F.; Seijo, M.; Larumbe-Zabala, E.; Ashrafi, N.; Christides, T.; Karsten, B.; Nielsen, B.V. Effects of supplementation with beef or whey protein versus carbohydrate in master triathletes. J. Am. Coll. Nutr. 2017, 36, 593–601. [Google Scholar] [CrossRef]
- Naclerio, F.; Seijo, M.; Larumbe-Zabala, E.; Earnest, C.P. Carbohydrates alone or mixing with beef or whey protein promote similar training outcomes in resistance training males: A double-blind, randomized controlled clinical trial. Int. J. Sport Nutr. Exerc. Metab. 2017, 27, 408–420. [Google Scholar] [CrossRef]
- Stojkovic, V.; Simpson, C.A.; Sullivan, R.R.; Cusano, A.M.; Kerstetter, J.E.; Kenny, A.M.; Insogna, K.L.; Bihuniak, J.D. The effect of dietary glycemic properties on markers of inflammation, insulin resistance, and body composition in postmenopausal American women: An ancillary study from a multicenter protein supplementation trial. Nutrients 2017, 9, 484. [Google Scholar] [CrossRef]
- Hwang, P.S.; Andre, T.L.; McKinley-Barnard, S.K.; Marroquín, F.E.M.; Gann, J.J.; Song, J.J.; Willoughby, D.S. Resistance training–induced elevations in muscular strength in trained men are maintained after 2 weeks of detraining and not differentially affected by whey protein supplementation. J. Strength Cond. Res. 2017, 31, 869–881. [Google Scholar] [CrossRef]
- Dudgeon, W.D.; Kelley, E.P.; Scheett, T.P. Effect of whey protein in conjunction with a caloric-restricted diet and resistance training. J. Strength Cond. Res. 2017, 31, 1353–1361. [Google Scholar] [CrossRef] [PubMed]
- Mobley, C.B.; Haun, C.T.; Roberson, P.A.; Mumford, P.W.; Romero, M.A.; Kephart, W.C.; Anderson, R.G.; Vann, C.G.; Osburn, S.C.; Pledge, C.D. Effects of whey, soy or leucine supplementation with 12 weeks of resistance training on strength, body composition, and skeletal muscle and adipose tissue histological attributes in college-aged males. Nutrients 2017, 9, 972. [Google Scholar] [CrossRef]
- Gaffney, K.A.; Lucero, A.; Stoner, L.; Faulkner, J.; Whitfield, P.; Krebs, J.; Rowlands, D.S. Nil Whey Protein Effect on Glycemic Control after Intense Mixed-Mode Training in Type 2 Diabetes. Med. Sci. Sports Exerc. 2018, 50, 11–17. [Google Scholar] [CrossRef] [PubMed]
- Holwerda, A.M.; Overkamp, M.; Paulussen, K.J.; Smeets, J.S.; Van Kranenburg, J.; Backx, E.M.; Gijsen, A.P.; Goessens, J.P.; Verdijk, L.B.; van Loon, L.J. Protein Supplementation after Exercise and before Sleep Does Not Further Augment Muscle Mass and Strength Gains during Resistance Exercise Training in Active Older Men. J. Nutr. 2018, 148, 1723–1732. [Google Scholar] [CrossRef]
- McAdam, J.S.; McGinnis, K.D.; Beck, D.T.; Haun, C.T.; Romero, M.A.; Mumford, P.W.; Roberson, P.A.; Young, K.C.; Lohse, K.R.; Lockwood, C.M. Effect of whey protein supplementation on physical performance and body composition in army initial entry training soldiers. Nutrients 2018, 10, 1248. [Google Scholar] [CrossRef]
- Forbes, S.C.; Bell, G.J. Whey protein isolate supplementation while endurance training does not Alter cycling performance or immune responses at rest or after exercise. Front. Nutr. 2019, 6, 19. [Google Scholar] [CrossRef]
- McAdam, J.S.; Lyons, K.D.; Beck, D.T.; Haun, C.T.; Romero, M.A.; Mumford, P.W.; Roberson, P.A.; Young, K.C.; Lohse, K.R.; Roberts, M.D. Whey protein supplementation effects on body composition, performance, and blood biomarkers during Army initial entry training. Front. Nutr. 2022, 9, 807928. [Google Scholar] [CrossRef]
- Obradović, J.; Vukadinović Jurišić, M.; Rakonjac, D. The effects of leucine and whey protein supplementation with eight weeks of resistance training on strength and body composition. J. Sports Med. Phys. Fit. 2020, 60, 864–869. [Google Scholar] [CrossRef] [PubMed]
- Dulac, M.; Pion, C.; Lemieux, F.; Carvalho, L.P.; Boutros, G.E.H.; Bélanger, M.; Gaudreau, P.; Chevalier, S.; Morais, J.; Noirez, P. Effects of slow-v. fast-digested protein supplementation combined with mixed power training on muscle function and functional capacities in older men. Br. J. Nutr. 2021, 125, 1017–1033. [Google Scholar] [CrossRef]
- Roberson, P.A.; Mobley, C.B.; Romero, M.A.; Haun, C.T.; Osburn, S.C.; Mumford, P.W.; Vann, C.G.; Greer, R.A.; Ferrando, A.A.; Roberts, M.D. LAT1 protein content increases following 12 weeks of resistance exercise training in human skeletal muscle. Front. Nutr. 2021, 7, 628405. [Google Scholar] [CrossRef]
- Zbinden-Foncea, H.; Ramos-Navarro, C.; Hevia-Larraín, V.; Castro-Sepulveda, M.; Saúl, M.J.; Kalazich, C.; Deldicque, L. Neither Chia Flour nor Whey Protein Supplementation Further Improves Body Composition or Strength Gains after a Resistance Training Program in Young Subjects with a Habitual High Daily Protein Intake. Nutrients 2023, 15, 1365. [Google Scholar] [CrossRef]
- Yapici, H.; Gülü, M.; Yagin, F.H.; Ugurlu, D.; Comertpay, E.; Eroglu, O.; Kocoğlu, M.; Aldhahi, M.I.; Karayigit, R.; Badri AL-Mhanna, S. The effect of 8-weeks of combined resistance training and chocolate milk consumption on maximal strength, muscle thickness, peak power and lean mass, untrained, university-aged males. Front. Physiol. 2023, 14, 1148494. [Google Scholar] [CrossRef]
- Kim, C.-B.; Park, J.-H.; Park, H.-S.; Kim, H.-J.; Park, J.-J. Effects of whey protein supplement on 4-week resistance exercise-induced improvements in muscle mass and isokinetic muscular function under dietary control. Nutrients 2023, 15, 1003. [Google Scholar] [CrossRef]
- Kemmler, W.; Kohl, M.; Freiberger, E.; Sieber, C.; von Stengel, S. Effect of whole-body electromyostimulation and/or protein supplementation on obesity and cardiometabolic risk in older men with sarcopenic obesity: The randomized controlled FranSO trial. BMC Geriatr. 2018, 18, 70. [Google Scholar] [CrossRef]
- Lockwood, C.M.; Roberts, M.D.; Dalbo, V.J.; Smith-Ryan, A.E.; Kendall, K.L.; Moon, J.R.; Stout, J.R. Effects of hydrolyzed whey versus other whey protein supplements on the physiological response to 8 weeks of resistance exercise in college-aged males. J. Am. Coll. Nutr. 2017, 36, 16–27. [Google Scholar] [CrossRef]
- Park, Y.; Park, H.-Y.; Kim, J.; Hwang, H.; Jung, Y.; Kreider, R.; Lim, K. Effects of whey protein supplementation prior to, and following, resistance exercise on body composition and training responses: A randomized double-blind placebo-controlled study. J. Exerc. Nutr. Biochem. 2019, 23, 34. [Google Scholar] [CrossRef]
- Knuiman, P.; van Loon, L.J.; Wouters, J.; Hopman, M.; Mensink, M. Protein supplementation elicits greater gains in maximal oxygen uptake capacity and stimulates lean mass accretion during prolonged endurance training: A double-blind randomized controlled trial. Am. J. Clin. Nutr. 2019, 110, 508–518. [Google Scholar] [CrossRef]
- Mhamed, M.; Zarrouk, F.; Mrad, M.; Methnani, J.; Bahlous, A.; Zaouali, M.; Lindinger, M.; Bigard, X.; Bouhlel, E. Effects of whey protein on body composition, biochemical profile, and high intensity physical performances in well-trained endurance runners. Sci. Sports 2024, 39, 588–598. [Google Scholar] [CrossRef]
- Bodaghabadi, Z.; Mohammadi, L.O.; Halabchi, F.; Tavakol, Z.; Kluzek, S.; Tabesh, M.R.; Abolhasani, M.; Alizadeh, Z. Effects of high-dairy versus low-dairy, high-protein and low-calorie diets combined with aerobic exercise on central body fat in overweight women: A pragmatic randomized controlled trial. Obes. Med. 2023, 40, 100492. [Google Scholar] [CrossRef]
- Soares, A.L.d.S.; Machado-Lima, A.; Brech, G.C.; Greve, J.M.D.A.; Dos Santos, J.R.; Inojossa, T.R.; Rogero, M.M.; Salles, J.E.N.; Santarem-Sobrinho, J.M.; Davis, C.L. The influence of whey protein on muscle strength, glycemic control and functional tasks in older adults with type 2 diabetes mellitus in a resistance exercise program: Randomized and triple blind clinical trial. Int. J. Environ. Res. Public Health 2023, 20, 5891. [Google Scholar] [CrossRef]
- Ferguson-Stegall, L.; McCleave, E.; Ding, Z.; Doerner, P.G., III; Liu, Y.; Wang, B.; Healy, M.; Kleinert, M.; Dessard, B.; Lassiter, D.G. Aerobic exercise training adaptations are increased by postexercise carbohydrate-protein supplementation. J. Nutr. Metab. 2011, 2011, 623182. [Google Scholar] [CrossRef]
- Reljic, D.; Zieseniss, N.; Herrmann, H.J.; Neurath, M.F.; Zopf, Y. Protein supplementation does not maximize adaptations to low-volume high-intensity interval training in sedentary, healthy adults: A placebo-controlled double-blind randomized study. Nutrients 2022, 14, 3883. [Google Scholar] [CrossRef]
- Reljic, D.; Zieseniss, N.; Herrmann, H.J.; Neurath, M.F.; Zopf, Y. Protein Supplementation Increases Adaptations to Low-Volume, Intra-Session Concurrent Training in Untrained Healthy Adults: A Double-Blind, Placebo-Controlled, Randomized Trial. Nutrients 2024, 16, 2713. [Google Scholar] [CrossRef]
- Murray, M.; Vlietstra, L.; Best, A.M.; Sims, S.T.; Loehr, J.A.; Rehrer, N.J. Post-Exercise Whey Protein Supplementation: Effects on IGF-1, Strength, and Body Composition in Pre-Menopausal Women, a Randomised Controlled Trial. Nutrients 2025, 17, 2033. [Google Scholar] [CrossRef]
- Yıldız, N.; Coşkun, H.; Tanal, M.; Baş, M.; Sağlam, D. Presleep vs. Daytime Consumption of Casein-Enriched Milk: Effects on Muscle Function and Metabolic Health After Sleeve Gastrectomy. Nutrients 2025, 17, 2750. [Google Scholar] [CrossRef]
- Sabooni, K.; Farsi, F.; Safari, S.; Mahjoubi, M.; Davarpanah Jazi, A.H.; Moosavi, D.; Gholi, Z.; Kermansaravi, M.; Shahabi, S. The role of whey protein supplementation after one-anastomosis gastric bypass: Results of a double-blind randomized controlled trial. Clin. Nutr. ESPEN 2025, 69, 406–414. [Google Scholar] [CrossRef]
- Jonvik, K.L.; Paulussen, K.J.; Danen, S.L.; Ceelen, I.J.; Horstman, A.M.; Wardenaar, F.C.; Van Loon, L.J.; Van Dijk, J.-W. Protein supplementation does not augment adaptations to endurance exercise training. Med. Sci. Sports Exerc. 2019, 51, 2041. [Google Scholar] [CrossRef]
- Ormsbee, M.J.; Willingham, B.D.; Marchant, T.; Binkley, T.L.; Specker, B.L.; Vukovich, M.D. Protein supplementation during a 6-month concurrent training program: Effect on body composition and muscular strength in sedentary individuals. Int. J. Sport Nutr. Exerc. Metab. 2018, 28, 619–628. [Google Scholar] [CrossRef]
- Griffen, C.; Duncan, M.; Hattersley, J.; Weickert, M.O.; Dallaway, A.; Renshaw, D. Effects of resistance exercise and whey protein supplementation on skeletal muscle strength, mass, physical function, and hormonal and inflammatory biomarkers in healthy active older men: A randomised, double-blind, placebo-controlled trial. Exp. Gerontol. 2022, 158, 111651. [Google Scholar] [CrossRef]
- Arnarson, A.; Gudny Geirsdottir, O.; Ramel, A.; Briem, K.; Jonsson, P.; Thorsdottir, I. Effects of whey proteins and carbohydrates on the efficacy of resistance training in elderly people: Double blind, randomised controlled trial. Eur. J. Clin. Nutr. 2013, 67, 821–826. [Google Scholar] [CrossRef]
- Karelis, A.; Messier, V.; Suppère, C.; Briand, P.; Rabasa-Lhoret, R. Effect of cysteine-rich whey protein (immunocal®) supplementation in combination with resistance training on muscle strength and lean body mass in non-frail elderly subjects: A randomized, double-blind controlled study. J. Nutr. Health Aging 2015, 19, 531–536. [Google Scholar] [CrossRef] [PubMed]
- Kirk, B.; Mooney, K.; Amirabdollahian, F.; Khaiyat, O. Exercise and dietary-protein as a countermeasure to skeletal muscle weakness: Liverpool Hope University–Sarcopenia Aging Trial (LHU-SAT). Front. Physiol. 2019, 10, 445. [Google Scholar] [CrossRef]
- Michel, J.M.; Lievense, K.K.; Norton, S.C.; Costa, J.V.; Alphin, K.H.; Bailey, L.A.; Miller, G.D. The effects of graded protein intake in conjunction with progressive resistance training on skeletal muscle outcomes in older adults: A preliminary trial. Nutrients 2022, 14, 2739. [Google Scholar] [CrossRef] [PubMed]
- Kirk, B.; Mooney, K.; Cousins, R.; Angell, P.; Jackson, M.; Pugh, J.N.; Coyles, G.; Amirabdollahian, F.; Khaiyat, O. Effects of exercise and whey protein on muscle mass, fat mass, myoelectrical muscle fatigue and health-related quality of life in older adults: A secondary analysis of the Liverpool Hope University—Sarcopenia Ageing Trial (LHU-SAT). Eur. J. Appl. Physiol. 2020, 120, 493–503. [Google Scholar] [CrossRef]
- Koopmans, L.; Spoelder, M.; Bongers, C.C.; Eijsvogels, T.M.; Hopman, M.T. Daily supplementation of lesser mealworm protein for 11-weeks increases skeletal muscle mass in physically active older adults. J. Nutr. Health Aging 2024, 28, 100364. [Google Scholar] [CrossRef]
- Oikawa, S.Y.; Kamal, M.J.; Webb, E.K.; McGlory, C.; Baker, S.K.; Phillips, S.M. Whey protein but not collagen peptides stimulate acute and longer-term muscle protein synthesis with and without resistance exercise in healthy older women: A randomized controlled trial. Am. J. Clin. Nutr. 2020, 111, 708–718. [Google Scholar] [CrossRef] [PubMed]
- Phillips, S.M.; Tang, J.E.; Moore, D.R. The role of milk-and soy-based protein in support of muscle protein synthesis and muscle protein accretion in young and elderly persons. J. Am. Coll. Nutr. 2009, 28, 343–354. [Google Scholar] [CrossRef]
- Dangin, M.; Guillet, C.; Garcia-Rodenas, C.; Gachon, P.; Bouteloup-Demange, C.; Reiffers-Magnani, K.; Fauquant, J.; Ballèvre, O.; Beaufrère, B. The rate of protein digestion affects protein gain differently during aging in humans. J. Physiol. 2003, 549, 635–644. [Google Scholar] [CrossRef]
- Gilmartin, S.; O’Brien, N.; Giblin, L. Whey for sarcopenia; can whey peptides, hydrolysates or proteins play a beneficial role? Foods 2020, 9, 750. [Google Scholar] [CrossRef]
- Bendtsen, L.Q.; Lorenzen, J.K.; Bendsen, N.T.; Rasmussen, C.; Astrup, A. Effect of dairy proteins on appetite, energy expenditure, body weight, and composition: A review of the evidence from controlled clinical trials. Adv. Nutr. 2013, 4, 418–438. [Google Scholar] [CrossRef]
- Teegarden, D. The Influence of Dairy Product Consumption on BodyComposition. J. Nutr. 2005, 135, 2749–2752. [Google Scholar] [CrossRef]
- Fekete, Á.A.; Givens, D.I.; Lovegrove, J.A. Can milk proteins be a useful tool in the management of cardiometabolic health? An updated review of human intervention trials. Proc. Nutr. Soc. 2016, 75, 328–341. [Google Scholar] [CrossRef]
- Morifuji, M.; Aoyama, T.; Nakata, A.; Sambongi, C.; Koga, J.; Kurihara, K.; Kanegae, M.; Suzuki, K.; Higuchi, M. Post-exercise ingestion of different amounts of protein affects plasma insulin concentration in humans. Eur. J. Sport Sci. 2012, 12, 152–160. [Google Scholar] [CrossRef]
- Dangin, M.; Boirie, Y.; Guillet, C.; Beaufrère, B. Influence of the protein digestion rate on protein turnover in young and elderly subjects. J. Nutr. 2002, 132, 3228S–3233S. [Google Scholar] [CrossRef] [PubMed]
- Burd, N.A.; Tang, J.E.; Moore, D.R.; Phillips, S.M. Exercise training and protein metabolism: Influences of contraction, protein intake, and sex-based differences. J. Appl. Physiol. 2009, 106, 1692–1701. [Google Scholar] [CrossRef] [PubMed]
- Pesta, D.H.; Samuel, V.T. A high-protein diet for reducing body fat: Mechanisms and possible caveats. Nutr. Metab. 2014, 11, 53. [Google Scholar] [CrossRef] [PubMed]
- Ji, A.; Chen, W.; Zhang, T.; Shi, R.; Wang, X.; Wang, Y.; Xu, H.; Li, D. Whey protein and soy protein prevent obesity by upregulating uncoupling protein 1 to activate brown adipose tissue and promote white adipose tissue browning in high-fat diet-fed mice. Food Funct. 2022, 13, 12836–12851. [Google Scholar] [CrossRef]
- Zhou, J.; Keenan, M.J.; Losso, J.N.; Raggio, A.M.; Shen, L.; McCutcheon, K.L.; Tulley, R.T.; Blackman, M.R.; Martin, R.J. Dietary whey protein decreases food intake and body fat in rats. Obesity 2011, 19, 1568–1573. [Google Scholar] [CrossRef] [PubMed]
- Pezeshki, A.; Fahim, A.; Chelikani, P.K. Dietary whey and casein differentially affect energy balance, gut hormones, glucose metabolism, and taste preference in diet-induced obese rats. J. Nutr. 2015, 145, 2236–2244. [Google Scholar] [CrossRef] [PubMed]

| Reference | Country | Study Design | Participants | Sex | Sample Size | Trial Duration (Weeks) | Mean Age | Mean BMI | Intervention | |||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| IG | CG | IG | CG | IG | CG | Type | SupplementDose (g/day) | CG | ||||||
| Claessens et al., 2009 (a) [42] | Netherlands | R, P, SB, PC | Individuals with OW & OB | ♂/♀ | 14 | 16 | 12 | 45.4 ± 8.2 | 46 ± 8.8 | 32.9 ± 6 | 32.4 ± 4.8 | CP | 50 | MD |
| Claessens et al., 2009 (b) [42] | Netherlands | R, P, SB, PC | Individuals with OW & OB | ♂/♀ | 18 | 16 | 12 | 44.9 ± 8.5 | 46 ± 8.8 | 33.4 ± 4 | 32.4 ± 4.8 | WP | 50 | MD |
| Pal et al., 2010 (a) [43] | Australia | R, P, SB, PC | Individuals with OW & OB | ♂/♀ | 25 | 25 | 12 | 48.5 ± 1 | 48.4 ± 7.5 | 32 ± 4 | 30.6 ± 4 | WP | 54 | CHO |
| Pal et al., 2010 (b) [43] | Australia | R, P, SB, PC | Individuals with OW & OB | ♂/♀ | 20 | 25 | 12 | 48 ± 10.5 | 48.4 ± 7.5 | 31.3 ± 4.5 | 30.6 ± 4 | CP | 54 | CHO |
| Fluegel et al., 2010 [44] | USA | R, P, PC | Patients with HTN & pre-HTN | ♂/♀ | 36 | 35 | 6 | 20.4 ± 1.7 | 20.7 ± 1.9 | 25.1 ± 2.6 | 24.2 ± 2.4 | WP | 28 | Non-hydrolyzed WP beverage |
| Takahira et al., 2011 [45] | Japan | R, P, DB, PC | Individuals with visceral fat OB | ♂/♀ | 23 | 21 | 32 | 54.4 ± 13 | 56.8 ± 12.2 | 29.3 ± 3.8 | 29 ± 4.5 | MP | 22 | Soy PR |
| Aldrich et al., 2011 [46] | USA | R, P, CO | Midlife adults | ♂/♀ | 5 | 5 | 20 | 49.2 ± 3.9 | 51.3 ± 5.1 | 30.6 ± 1.5 | 29.9 ± 1.5 | WP | 45 | Control diet |
| Hodgson et al., 2012 [103] | Australia | R, P, DB, PC | Older women | ♀ | 93 | 87 | 96 | 74.3 ± 2.7 | 74.3 ± 2.6 | 26.3 ± 3.8 | 27.2 ± 3.9 | WP | 30 | Low-PR, high-CHO beverage |
| Gouni-Berthold et al., 2012 [47] | Germany | R, P, DB, PC | Patients with MetS | ♂/♀ | 83 | 88 | 12 | 52.9 ± 10.3 | 53.9 ± 9.5 | 30.8 ± 4.2 | 31.3 ± 4 | WP | 15.3 | Yogurt |
| Agin et al., 2001 [104] | USA | R, P, CO | Women with HIV | ♀ | 10 | 10 | 14 | 43.4 ± 10.6 | 41 ± 10.2 | 23 ± 2.3 | 24.8 ± 2.5 | WP | 57 | PRE |
| Ahmadi Kani Golzar et al., 2012 [128] | Iran | R, P, SB, PC | Young men with OW | ♂ | 10 | 10 | 6 | 22.7± 2.3 | 21.2± 1.0 | 26.5 ± 1.1 | 27.1 ± 1.5 | WP | 30 | Starch solution |
| Sheikholeslami Vatani et al., 2012 [129] | Iran | R, P, SB, PC | Young men with OW | ♂ | 9 | 10 | 6 | 23 ± 2 | 21 ± 1 | 26.5 ± 1.2 | 27.2 ± 1.6 | WP | 90 | Starch solution |
| Figueroa et al., 2014 (a) [105] | USA | R, P, DB, PC | Young women with OB & high BP | ♀ | 11 | 11 | 4 | 31 ± 9.9 | 31 ± 6.6 | 37.9 ± 6.6 | 33.5 ± 4 | CP | 30 | MD |
| Figueroa et al., 2014 (b) [105] | USA | R, P, DB, PC | Young women with OB & high BP | ♀ | 11 | 11 | 4 | 28 ± 3.3 | 31 ± 6.6 | 34.3 ± 4.6 | 33.5 ± 4 | WP | 30 | MD |
| Tahavorgar et al., 2015 [130] | Iran | R, P, DB, PC | Men with OW & OB | ♂ | 26 | 19 | 12 | 39.4 ± 6.9 | 38.8 ± 8.8 | 32.1 ± 3.2 | 32.1 ± 2.7 | WP | 65 | Soy PR |
| Arciero et al., 2016 [48] | USA | R, P, CO | Individuals with OW | ♂/♀ | 12 | 9 | 16 | 48 ± 1 | 52 ± 4 | 32 ± 7 | 33 ± 3 | WP | 20–25 | Food PR |
| Larsen et al., 2018 [49] | Denmark | R, P, SB, PC | Individuals with OW & OB | ♂/♀ | 14 | 15 | 4 | 41 | 41 | 34.9 ± 5.4 | 35.1 ± 5.8 | WP | 41 | MD |
| Demling & DeSanti 2000 (a) [50] | USA | R, P, CO | Police officers with OW | ♂/♀ | 14 | 10 | 12 | 33 ± 4 | 35 ± 4 | 30 ± 3.9 | 29 ± 3.5 | CP | 74 | Hypocaloric diet |
| Demling & DeSanti 2000 (b) [50] | USA | R, P, CO | Police officers with OW | ♂/♀ | 14 | 10 | 12 | 34 ± 3 | 35 ± 4 | 31 ± 4.5 | 29 ± 3.5 | WP | 74 | Hypocaloric diet |
| Grey et al., 2003 (a) [51] | Canada | R, P, DB, PC | Patients with CF | ♂/♀ | 10 | 11 | 12 | 25.5 ± 6.3 | 24.2 ± 3.9 | 21 ± 4.3 | 20.6 ± 2.7 | WP | 20 | CP |
| Grey et al., 2003 (b) [51] | Canada | R, P, DB, PC | Patients with CF | ♂/♀ | 11 | 10 | 12 | 24.2 ± 3.9 | 25.5 ± 6.3 | 20.6 ± 2.7 | 21 ± 4.3 | CP | 20 | WP |
| Nabuco et al., 2019 [106] | Brazil | R, P, DB, PC | Older women with sarcopenic OB | ♀ | 13 | 13 | 12 | 68 ± 4.2 | 70.1 ± 3.9 | 26.4 ± 3 | 27.4 ± 3 | WP | 15 | MD |
| Moon et al., 2020 [131] | USA | R, P, DB, PC | Trained men | ♂ | 12 | 12 | 8 | 32.8 ± 6.7 | 32.8 ± 6.7 | 27.2 ± 1.9 | 27.8 ± 1.9 | WP | 24 | Rice |
| Lefferts et al., 2020 [52] | USA | R, P, DB, PC | Older adults | ♂/♀ | 53 | 46 | 12 | 69 ± 7 | 67 ± 6 | 27.9 ± 5.6 | 27 ± 3.9 | WP | 50 | MD |
| Hudson et al., 2020 [53] | USA | R, P, DB, PC | Individuals with OW & OB | ♂/♀ | 21 | 23 | 16 | 53 ± 9.2 | 52 ± 4.8 | 31 ± 3.2 | 30.3 ± 3.4 | MP | 64 | MD |
| Fuglsang-Nielsen et al., 2021 (a) [54] | Denmark | R, P, DB, PC | Individuals with abdominal OB | ♂/♀ | 15 | 16 | 12 | 64 | 64 | 29.7 ± 3.9 | 30.1 ± 3.7 | WP+ low fiber | 60 | MD |
| Fuglsang-Nielsen et al., 2021 (b) [54] | Denmark | R, P, DB, PC | Individuals with abdominal OB | ♂/♀ | 17 | 17 | 12 | 64 | 64 | 29.1 ± 3.4 | 28.7 ± 3.8 | WP+ high fiber | 60 | MD |
| Weinheimer et al., 2012 (a) [55] | USA | R, P, DB, PC | Individuals with OW & OB | ♂/♀ | 81 | 84 | 36 | 47± 8.1 | 49 ± 7 | 30.4 ± 2.6 | 29.9 ± 2.7 | WP | 20 | MD |
| Weinheimer et al., 2012 (b) [55] | USA | R, P, DB, PC | Individuals with OW & OB | ♂/♀ | 25 | 84 | 36 | 46 ± 9.4 | 49 ± 7 | 29.4 ± 2.3 | 29.9 ± 2.7 | WP | 40 | MD |
| Weinheimer et al., 2012 (c) [55] | USA | R, P, DB, PC | Individuals with OW & OB | ♂/♀ | 30 | 84 | 36 | 50 ± 7.1 | 49 ± 7 | 30.7 ± 3.4 | 29.9 ± 2.7 | WP | 60 | MD |
| Kjølbæk et al., 2017 [56] | Denmark | R, P, DB, PC | Individuals with OW & OB | ♂/♀ | 39 | 38 | 16 | 41.2 ± 10.2 | 38.3 ± 11.5 | 28.5 ± 3.1 | 28.9 ± 2.7 | WP | 45 | MD |
| Jeong et al., 2019 [57] | USA | R, P, PC | Hemodialysis patients | ♂/♀ | 38 | 34 | 48 | 56.6 ± 13.0 | 54.4 ± 12.3 | 30.6 ± 7.1 | 31.5 ± 7.6 | WP | 12.85 | Non-nutritive beverage |
| Yang et al., 2019 (a) [58] | China | R, P, PC | Individuals with pre- or mild HTN, and normal weight | ♂/♀ | 12 | 12 | 12 | 42.3 ± 11.6 | 43.8 ± 11.7 | 24.1 ± 3.1 | 24.3 ± 2.3 | WP | 30 | MD |
| Yang et al., 2019 (b) [58] | China | R, P, PC | Individuals with pre- or mild HTN, OW, and OB | ♂/♀ | 15 | 15 | 12 | 42.3 ± 11.6 | 43.8 ± 11.7 | 24.1 ± 3.1 | 24.3 ± 2.3 | WP | 30 | MD |
| Kataoka et al., 2016 [132] | Japan | R, P, PC | Patients with HTN | ♂ | 10 | 11 | 8 | 69 ± 3.1 | 69 ± 3.3 | 22 ± 3.1 | 23 ± 3.3 | WP | 4.28 | CHO |
| Ormsbee et al., 2015 (a) [107] | USA | R, P, DB, PC | Women with OW & OB | ♀ | 13 | 10 | 4 | 29.3 ± 4.3 | 27.7 ± 7.3 | 34.4 ± 4.7 | 33.1 ± 5.4 | WP | 30 | MD |
| Ormsbee et al., 2015 (b) [107] | USA | R, P, DB, PC | Women with OW & OB | ♀ | 14 | 10 | 4 | 30.0 ± 7.1 | 27.7 ± 7.3 | 36.5 ± 6.7 | 33.1 ± 5.4 | CP | 30 | MD |
| Sun et al., 2022 (a) [108] | China | R, P, SB, CO | Older women | ♀ | 16 | 18 | 8 | 61.3 ± 7.7 | 27.2 ± 1.6 | WP + ERD | 15.2 | ERD | ||
| Sun et al., 2022 (b) [108] | China | R, P, SB, CO | Older women | ♀ | 14 | 18 | 8 | 61.3 ± 7.7 | 27.2 ± 1.6 | WPH + RD | 16.8 | ERD | ||
| Nouri et al., 2022 [109] | Iran | R, P, DB, PC | Women with OW, OB & T2DM | ♀ | 18 | 17 | 12 | 44.0 ± 6.2 | 46.9 ± 5.1 | 32.5 ± 4.2 | 31.6 ± 5.0 | WP | 20 | Unfortified bread |
| Nabuco et al., 2019 [110] | Brazil | R, P, DB, PC | Older women | ♀ | 15 | 15 | 12 | 69.2 ± 4.1 | 68.4 ± 4.5 | 27.4 ± 5.1 | 26.6 ± 3.4 | WP | 15 | MD |
| Frestedt et al., 2008 [59] | USA | R, P, DB, PC | Individuals with OB | ♂/♀ | 31 | 28 | 12 | 43.6 ± 6.1 | 42 ± 6.3 | 35.7 ± 3.9 | 35.4 ± 3.7 | WP | 20 | MD |
| Silva et al., 2010 [60] | Brazil | R, P, DB, PC | Patients with ALS | ♂/♀ | 8 | 8 | 16 | 53 | 53 | 21.7 ± 1.1 | 22.9 ± 1.1 | WP | 22 | MD |
| Sohrabi et al., 2016 [61] | Iran | R, P, CO | Hemodialysis patients | ♂/♀ | 23 | 23 | 8 | 57 ± 9.6 | 55 ± 6.5 | 24.3 ± 4.2 | 22.4 ± 3.5 | WP | 6.42 | NI |
| Sharp et al., 2018 [62] | USA | R, P, DB, PC | Healthy individuals | ♂/♀ | 10 | 10 | 8 | 19 ± 2 | 21 ± 2 | 25.4 ± 4.8 | 25.2 ± 3.8 | WP | 46 | MD |
| Bumrungpert et al., 2018 [63] | Thailand | R, P, DB, PC | Patients with cancer | ♂/♀ | 23 | 19 | 12 | 54.1 ± 9.3 | 51.5 ± 9.6 | 24.9 ± 5.7 | 23.6 ± 3.7 | WP | 40 | MD |
| Derosa et al., 2020 (a) [64] | Italy | R, P, DB, PC | Patients with T2DM | ♂/♀ | 59 | 58 | 12 | 59.7 ± 9.1 | 58.6 ± 8.8 | 22.7 ± 2.1 | 22.7 ± 2.1 | WP | 5 | CP |
| Derosa et al., 2020 (b) [64] | Italy | R, P, DB, PC | Patients with T2DM | ♂/♀ | 58 | 59 | 12 | 58.6 ± 8.8 | 59.7 ± 9.1 | 22.7 ± 2.1 | 22.7 ± 2.1 | CP | 5 | WP |
| Ahmadi et al., 2020 [65] | Iran | R, P, SB, CO | Patients with COPD | ♂/♀ | 23 | 21 | 8 | 62.0 ± 7 | 63.4 ± 7.2 | 20.6 ± 3.4 | 21.5 ± 2.5 | WP | 15.9 | Dietary advice |
| Burke et al., 2001 [133] | Canada | R, P, DB, PC | Healthy men | ♂ | 10 | 5 | 6 | 18–31 | 18–31 | NR | NR | WP | 102 | MD |
| Rankin et al., 2004 [134] | USA | R, P, PC | Healthy men | ♂ | 10 | 9 | 10 | 20.5 ± 2 | 21 ± 1.4 | NR | NR | MP | 7.02 | CHO |
| Samadi et al., 2021 [135] | Iran | R, P, DB, PC | Basketball players | ♂ | 22 | 22 | 8 | 20–30 | 20–30 | 23.8 ± 2.3 | 22.8 ± 1.8 | WP | 25 | Starch |
| Teixeira et al., 2022 [136] | Portugal | R, P, DB, PC | Futsal players | ♂ | 20 | 20 | 8 | 18–35 | 18–35 | NR | NR | WP | 25 | Plant-based PR |
| Pettersson et al., 2021 [137] | Sweden | R, P, DB, PC | Individuals with OW & OB | ♂ | 10 | 10 | 6 | 28.2 ± 5.5 | 27.9 ± 5 | 29.8 ± 2.3 | 30.4 ± 1.8 | MP | 8.57 | CHO |
| Gryson et al., 2014 [66] | France | R, P, DB, PC | Older adults | ♂/♀ | 9 | 9 | 16 | 60.9± 0.5 | 60.5± 0.7 | 26.2 ± 1.8 | 26.8 ± 2.7 | MP | 10 | 4g MP |
| Hulmi et al., 2015 [138] | Finland | R, P, DB, PC | Healthy man | ♂ | 22 | 21 | 12 | 31.4 ± 6.6 | 36.4 ± 19.2 | 25.6 ± 0.9 | 25.4 ± 0.9 | WP | 13 | MD |
| Maltais et al., 2016 [139] | Canada | R, P, DB, PC | Patients with sarcopenia | ♂ | 8 | 8 | 16 | 68 ± 5.1 | 64 ± 4.9 | 25.8 ± 3 | 27 ± 2.7 | MP | 13.53 | Soy milk |
| Keogh & Clifton 2008 [67] | Australia | R, P, DB, PC | Individuals with OW & OB | ♂/♀ | 34 | 38 | 52 | 49.6 ± 12.3 | 50.3 ± 12.4 | 34.4 ± 3.7 | 34.4 ± 3.7 | WP | 15 | Skim milk |
| Fernandes et al., 2018 [111] | Brazil | R, P, DB, PC | Older women | ♀ | 16 | 16 | 12 | 67.3 ± 4.1 | 67.8 ± 4 | 25.9 ± 2.7 | 25.4 ± 2.6 | WP | 15 | MD |
| Rambousková et al., 2014 [68] | Czech Republic | R, P, CO | Older adults | ♂/♀ | 23 | 24 | 8 | 84.2 ± 9.7 | 85.3 ± 9.2 | 20.3 ± 2.9 | 20.4 ± 2.8 | MP | 18.2 | NI |
| Piccolo et al., 2015 [112] | USA | R, P, DB, PC | Women with OB | ♀ | 16 | 11 | 8 | 41 ± 9.8 | 41 ± 9.8 | 36.9 ± 3.1 | 36 ± 4.8 | WP | 20 | Gelatin |
| Brown et al., 2004 (a) [140] | USA | R, P, DB, PC | Healthy men | ♂ | 9 | 9 | 9 | 20.3 ± 1 | 21.6± 0.2 | 25.0 ± 2.7 | 24.7 ± 2.4 | WP | 33 | Soy PR |
| Brown et al., 2004 (b) [140] | USA | R, P, DB, CO | Healthy men | ♂ | 9 | 9 | 9 | 20.3 ± 1 | 20.4 ± 1.9 | 25.0 ± 2.7 | 24.9 ± 0.8 | WP | 33 | Training |
| Hartman et al., 2007 [141] | Canada | R, P, PC | Healthy men | ♂ | 18 | 19 | 12 | 18–30 | 18–30 | 25.6 ± 3.6 | 23.9 ± 3.0 | MP | 12.5 | CHO |
| Cribb et al., 2007 [142] | Australia | R, P, DB, PC | Male bodybuilders | ♂ | 5 | 7 | 11 | 24 ± 5 | 24 ± 7 | 21.4 ± 3.9 | 24.3 ± 4.0 | WP | 105 | CHO |
| Sattler et al., 2008 [69] | USA | R, P, DB, PC | Patients with HIV | ♂/♀ | 29 | 30 | 12 | 41 ± 25.9 | 41 ± 23.7 | 20.7 ± 2.3 | 21.1 ± 2.8 | WP | 80 | CHO |
| Eliot et al., 2008 [143] | USA | R, P, DB, PC | Middle-aged healthy men | ♂ | 11 | 10 | 14 | 48–72 | 48–72 | NR | NR | WP | 15 | Gatorade |
| Josse et al., 2010 [113] | Canada | R, P, SB, PC | Healthy women | ♀ | 10 | 10 | 12 | 23.2 ± 8.9 | 22.4 ± 7.6 | 26.2 ± 13.3 | 25.2 ± 12 | MP | 25.71 | MD |
| Mojtahedi et al., 2011 [114] | USA | R, P, DB, PC | Older women | ♀ | 13 | 13 | 24 | 64.7 ± 4.4 | 64.6 ± 5.2 | 32.3 ± 3.9 | 32.7 ± 4.2 | WP | 50 | MD |
| Arazi et al., 2011 [144] | Iran | R, P, DB, PC | Healthy men | ♂ | 20 | 20 | 8 | 21.3 ± 1.2 | 22.5 ± 3.4 | 24.1 ± 1.3 | 23.9 ± 1.4 | WP | 131 | Starch |
| Baer et al., 2011 [70] | USA | R, P, DB, PC | Individuals with OW & OB | ♂/♀ | 23 | 25 | 23 | 49 ± 43.2 | 51 ± 45 | 31 ± 10.6 | 31.1 ± 12.5 | WP | 55 | CHO |
| Elahikhah et al., 2024 [115] | Iran | R, P, SB, CO | Women with OB | ♀ | 21 | 20 | 8 | 37.1 ± 5.7 | 36.7 ± 9.0 | 33.6 ± 2.9 | 35.0 ± 3.0 | MPC | 20 | WLD |
| Giglio et al., 2019 [116] | Brazil | R, P, DB, PC | Women with OW | ♀ | 17 | 20 | 8 | 37.8 ± 12 | 43 ± 8 | 31.1 ± 4 | 30.9 ± 3.6 | WP | 25 | Collagen |
| DeNysschen et al., 2009 [145] | USA | R, P, DB, PC | Men with hyperlipidemia | ♂ | 10 | 9 | 12 | 38 | 28.5 ± 2.1 | 27.9 ± 1.2 | WP | 26.6 | CHO | |
| Haidari et al., 2020 [117] | Iran | R, P, CO | Pre-menopausal women with OB | ♀ | 30 | 30 | 8 | 31 ± 6.2 | 32.2 ± 5.1 | 33.5 ± 3.1 | 33.3 ± 2.6 | WP+ WLD | 30 | WLD |
| Hambre et al., 2012 [146] | Sweden | R, P, CO | Healthy men | ♂ | 12 | 12 | 12 | 24.2 ± 3.7 | 23.2 ± 3.4 | 22.6 ± 2.5 | 22.3 ± 1.9 | WP | 33 | A meal of fast food |
| Ottestad et al., 2017 [71] | Norway | R, P, DB, PC | Older adults | ♂/♀ | 17 | 19 | 12 | 76.8 ± 6.2 | 77.1 ± 4.7 | 27.6 ± 4.2 | 25.9 ± 4.9 | MP | 40 | CHO |
| Lopes Gomes et al., 2017 [118] | Brazil | R, P, CO | Postmenopausal women | ♀ | 15 | 15 | 16 | 41 ± 10 | 49 ± 10 | 36 ± 6 | 35 ± 4 | WP | 69 | Hypocaloric diet |
| Sugawara et al., 2012 [72] | Japan | R, P, DB, CO | Patients with COPD | ♂/♀ | 17 | 14 | 12 | 77.4 ± 5.2 | 77.1 ± 5.8 | NR | NR | WP | 20 | Normal diet |
| Björkman et al., 2012 [73] | Finland | R, P, CO | Nursing home residents | ♂/♀ | 46 | 51 | 24 | 84.1 ± 7.6 | 83 ± 8.7 | 24.8 ± 4.3 | 24 ± 5.5 | WP | 20 | Regular fruit juice |
| Joy et al., 2013 [147] | USA | R, P, DB, PC | Resistance-trained men | ♂ | 12 | 12 | 8 | 21.3 ± 19 | 21.3 ± 1.9 | NR | NR | WP | 28.57 | MD |
| Herda et al., 2013 [148] | USA | R, P, DB, PC | Trained men | ♂ | 22 | 21 | 8 | 21.0 ± 1.6 | 20.9 ± 1.7 | 23.6 ± 1.9 | 24.6 ± 4.3 | WP | 28.57 | MD |
| Volek et al., 2013 [74] | USA | R, P, DB, PC | Non-resistance-trained men | ♂/♀ | 19 | 22 | 36 | 22.8 ± 3.7 | 22.3 ± 3.1 | 25.1 ± 6.2 | 24.5 ± 5.8 | WP | 22 | MD |
| Chalé et al., 2013 [75] | USA | R, P, DB, PC | Older adults | ♂/♀ | 42 | 38 | 28 | 78 ± 4 | 77.3 ± 3.9 | 27 ± 3.2 | 26.9 ± 3.1 | WP | 40 | MD |
| Babault et al., 2014 (a) [149] | France | R, P, DB, PC | Physically active men | ♂ | 22 | 24 | 10 | 22.2 ± 3.9 | 22 ± 3.9 | 23.7 ± 3.5 | 23.4 ± 3.7 | CP | 24.8 | MD |
| Babault et al., 2014 (b) [149] | France | R, P, DB, PC | Physically active men | ♂ | 22 | 24 | 10 | 22.5 ± 4.1 | 22 ± 3.9 | 22.7 ± 2.4 | 23.4 ± 3.7 | MP | 24.8 | MD |
| Duff et al., 2014 [76] | Canada | R, P, DB, PC | Individuals with AO | ♂/♀ | 21 | 19 | 8 | 57.5 ± 6.3 | 61.8 ± 4.8 | 25.9 ± 7.3 | 26.9 ± 6.4 | WP | 38 | Bovine colostrum |
| Zhu et al., 2015 [119] | Australia | R, P, DB, PC | Older women | ♀ | 101 | 95 | 96 | 74.2 ± 2.8 | 74.3 ± 2.6 | 26.1 ± 3.8 | 27.2 ± 4 | WP | 30 | Skim MP |
| Hulmi et al., 2009 [150] | Finland | R, P, DB, PC | Young men | ♂ | 9 | 9 | 21 | 24.7 ± 5 | 27.4 ± 3.1 | 23.2 ± 2.5 | 23.2 ± 2.5 | WP | 8.5 | PL |
| Kerstetter et al., 2015 [77] | USA | R, P, DB, PC | Older adults | ♂/♀ | 106 | 102 | 72 | 69.9 ± 6.1 | 70.5 ± 6.4 | 26.1 ± 3.4 | 26.4 ± 4 | WP | 45 | MD |
| Hector et al., 2015 [78] | Canada | R, P, DB, PC | Adults with OW & OB | ♂/♀ | 7 | 12 | 2 | 52 ± 7.5 | 48 ± 10.4 | 34.7 ± 4.1 | 36.9 ± 4.1 | WP | 54 | MD |
| Malekian et al., 2015 [79] | USA | R, P, PC | African American men & women | ♂/♀ | 15 | 13 | 24 | 35 ± 4 | 32 ± 8 | 43 ± 8 | 43 ± 8 | WP | 56 | Starch |
| Taylor et al., 2016 [120] | USA | R, P, DB, PC | Basketball players | ♀ | 8 | 6 | 8 | 20 ± 2 | 21 ± 3 | 22.8 ± 1.9 | 23.8 ± 3.1 | WP | 27.4 | MD |
| Reidy et al., 2016 [151] | USA | R, P, DB, PC | Young men | ♂ | 18 | 18 | 12 | 25 ± 4.7 | 25 ± 4.8 | 25.8 ± 3.3 | 24.6 ± 2.9 | WP | 21.5 | MD |
| Naclerio et al., 2017 [152] | UK | R, P, DB, PC | Master triathletes | ♂ | 8 | 8 | 10 | 45.3 ± 8.9 | 46.2 ± 7 | 25.2 ± 4.4 | 23.8 ± 2.6 | WP | 20 | MD |
| Naclerio et al., 2017 [153] | UK | R, P, DB, PC | Resistance-trained men | ♂ | 8 | 8 | 8 | 26 ± 5 | 29 ± 9 | 23.1 ± 3.9 | 24.9 ± 5.1 | WP | 20 | MD |
| Stojkovic et al., 2017 [154] | USA | R, P, DB, PC | Post-menopausal women | ♂ | 38 | 46 | 72 | 68.9 ± 5.5 | 69.3 ± 6.1 | 26 ± 3.7 | 25.8 ± 4.1 | WP | 20 | MD |
| Hwang et al., 2017 [155] | USA | R, P, DB, PC | Resistance-trained men | ♂ | 11 | 9 | 10 | 20.9 ± 1.3 | 21 ± 1.1 | 25.1 ± 3.7 | 24.4 ± 3.1 | WP | 25 | MD |
| Dudgeon et al., 2017 [156] | USA | R, P, SB, PC | Resistance-trained men | ♂ | 8 | 8 | 8 | 24 ± 1.6 | 24 ± 1.6 | NR | NR | WP | 32 | CHO |
| Mobley et al., 2017 (a) [157] | USA | R, P, DB, PC | College-aged men | ♂ | 17 | 15 | 12 | 21 ± 4.1 | 21 ± 3.9 | NR | NR | WP | 50 | MD |
| Mobley et al., 2017 (b) [157] | USA | R, P, DB, PC | College-aged men | ♂ | 14 | 15 | 12 | 21 ± 3.7 | 21 ± 3.9 | NR | NR | WP | 50 | MD |
| Reimer et al., 2017 (a) [80] | Canada | R, P, DB, PC | Adults with OW& OB | ♂/♀ | 22 | 26 | 12 | 38.7 ± 12.1 | 40.4 ± 13.6 | 31.5 ± 6.1 | 31.1 ± 4.5 | WP | 10 | Prebiotic bar (ITF) |
| Reimer et al., 2017 (b) [80] | Canada | R, P, DB, CO | Adults with OW& OB | ♂/♀ | 21 | 27 | 12 | 40.7 ± 15.5 | 39.8 ± 12.6 | 31.7 ± 5.3 | 31.3 ± 6.1 | WP | 10 | Control snack bar |
| Hassan & Hassan 2017 [81] | Israel | R, P, CO | Peritoneal dialysis patients | ♂/♀ | 18 | 18 | 12 | 59.7 ± 11.5 | 58.1 ± 12.3 | 28.7 ± 3.3 | 28.6 ± 3.5 | WP | 26.3 | PR without WP |
| Hassan 2017 [82] | Israel | R, P, CO | Peritoneal dialysis patients | ♂/♀ | 19 | 17 | 12 | 58.4 ± 11.8 | 56.9 ± 12.6 | 28.3 ± 3.3 | 27.9 ± 4.3 | WP | 28.1 | PR without WP |
| Dirks et al., 2017 [83] | Netherland | R, P, DB, PC | Frail elderly | ♂/♀ | 17 | 17 | 24 | 76 ± 8 | 77 ± 8 | 29.5 ± 4.8 | 28.6 ± 3.6 | MP | 30 | PL |
| Gjevestad et al., 2017 [84] | Norway | R, P, DB, PC | Older adults | ♂/♀ | 14 | 17 | 12 | 76.9 ± 4.9 | 77.7 ± 4.8 | 27.1 ± 3.8 | 26.4 ± 4.9 | MP | 40 | CHO |
| Mori et al., 2018 [121] | Japan | R, P, SB, CO | Older adults | ♀ | 25 | 25 | 24 | 70.6 ± 4.6 | 70.6 ± 4.2 | 22.1 ± 2.1 | 22.9 ± 2.9 | WP | 6.37 | Exercise |
| Gaffney et al., 2018 [158] | New Zealand | R, P, DB, PC | Patients with T2DM | ♂ | 12 | 12 | 10 | 53.5 ± 5.6 | 57.8 ± 5.2 | 29.6 ± 2.7 | 30.1 ± 4.9 | WP | 20 | CHO |
| Holwerda et al., 2018 [159] | Netherlands | R, P, DB, PC | Active older men | ♂ | 21 | 20 | 12 | 69 ± 4.6 | 71 ± 4.5 | 25.5 ± 2.7 | 245.1 ± 2.2 | WP | 30 | PL |
| Englund et al., 2018 [85] | USA & Sweden | R, P, DB, PC | Older adults | ♂/♀ | 60 | 57 | 24 | 78.1 ± 5.8 | 76.9 ± 4.9 | 27.9 ± 3.3 | 28.4 ± 3.9 | WP | 20 | Nonnutritive sweetened drink |
| Sahathevan et al., 2018 [86] | Malaysia | R, P, CO | Peritoneal dialysis patients | ♂/♀ | 37 | 37 | 24 | 50.8 ± 15.2 | 42.1 ± 14.5 | 21.6 ± 2.8 | 21.2 ± 2.3 | WP | 27.4 | Dietary counseling |
| Park et al., 2019 [171] | South Korea | R, P, DB, PC | Young men | ♂ | 10 | 8 | 12 | 37.8 ± 12 | 43 ± 8 | 22.6 ± 2.9 | 25.1 ± 2.6 | WP | 17.4 | PL |
| Forbes et al., 2019 [161] | Canada | R, P, DB, PC | Healthy men | ♂ | 9 | 9 | 6 | 27 ± 7 | 27 ± 7 | NR | NR | WP | 79 | CHO |
| Amasene et al., 2019 [87] | Spain | R, P, DB, PC | Post-hospitalized older adults | ♂/♀ | 15 | 13 | 12 | 82.9 ± 5.5 | 81.7 ± 6.4 | 27.4 ± 3.5 | 30.8 ± 6.5 | WP | 5.7 | PL |
| Cereda et al., 2019 [88] | Italy | R, P, CO | Malnourished advanced cancer patients | ♂/♀ | 82 | 84 | 12 | 65.1 ± 11.7 | 65.7 ± 11.4 | 22 ± 4.1 | 22.3 ± 3.9 | WP | 20 | Nutritional counseling |
| Ten Haaf et al., 2019 [89] | Netherlands | R, P, DB, PC | Physically active older adults | ♂/♀ | 58 | 56 | 12 | 69 ± 3.7 | 69 ± 4.4 | 27.2 ± 2.6 | 26.3 ± 2.5 | MP | 31 | PL |
| Kang et al., 2019 [90] | China | R, P, CO | Frail older adults | ♂/♀ | 66 | 49 | 12 | 76.7 ± 7.11 | 78.0 ± 6.8 | 21.0 ± 3.4 | 22.7 ± 4.4 | WP | 32.4 | Resistance exercise |
| Rakvaag et al., 2019 (a) [91] | Denmark | R, P, DB, PC | Adults with AO | ♂/♀ | 15 | 16 | 12 | 67 ± 6.7 | 62 ± 7.4 | 28.4 ± 4.1 | 30.3 ± 4.5 | WP + low fiber | 60 | MD + low fiber |
| Rakvaag et al., 2019 (b) [91] | Denmark | R, P, DB, PC | Adults with AO | ♂/♀ | 17 | 17 | 12 | 65 ± 6.7 | 64 ± 8.1 | 29.6 ± 2.3 | 29.1 ± 3.6 | WP + high fiber | 60 | MD+ high fiber |
| Brown et al., 2020 [122] | USA | R, P, DB, PC | Female collegiate dancers | ♀ | 10 | 11 | 12 | 19.9 ± 0.7 | 19.4 ± 1.5 | 21.7 ± 2.4 | 21.8 ± 1.9 | WP | 75 | MD |
| McAdam et al., 2022 [162] | USA | R, P, DB, PC | Army soldiers | ♂ | 39 | 42 | 9 | 21 ± 3 | 23 ± 4 | 25.7 ± 4.4 | 25.4 ± 5.1 | WP | 38.6 | CHO |
| McAdam et al., 2018 [160] | USA | R, P, DB, PC | Army soldiers | ♂ | 34 | 35 | 8 | 19 ± 1 | 19 ± 1 | 24.5 ± 4.2 | 24.1 ± 3.7 | WP | 77 | CHO |
| Obradović et al., 2020 [163] | Serbia | R, P, PC | Male college athletes | ♂ | 10 | 10 | 8 | 23 ± 4 | 23 ± 4 | 25.0 ± 1.7 | 24.7 ± 1.4 | WP | 45.4 | MD |
| Lynch et al., 2020 [92] | USA | R, P, DB, PC | Untrained young individuals | ♂/♀ | 19 | 26 | 12 | 18–35 | 18–35 | 18.5–29.9 | 18.5–29.9 | WP | 19 | Soy PR |
| Boutry-Regard et al., 2020 [93] | Japan | R, P, DB, PC | Elderly adults | ♂/♀ | 15 | 12 | 12 | 78 ± 3.9 | 78 ± 6.9 | 21.3 ± 3.5 | 20.8 ± 2.8 | WP | 20 | MD |
| Mori et al., 2021 [123] | Japan | R, P, CO | Older women with sarcopenia | ♀ | 20 | 19 | 24 | 78.1 ± 2.7 | 78.1 ± 4.6 | 20.3 ± 2.5 | 19.6 ± 2.2 | WP | 3.14 | Exercise |
| Biesek et al., 2021 [124] | Brazil | R, P, SB, PC | Older women | ♀ | 16 | 15 | 12 | 73.1 ± 5.3 | 70.4 ± 3.9 | 28.1 ± 3.8 | 27.1 ± 4.3 | WP | 21 | MD |
| Dulac et al., 2021 (a) [164] | Canada | R, P, DB, PC | Older men | ♂ | 21 | 19 | 12 | 68·3 ± 5.3 | 70.7 ± 8.6 | 26.7 ± 3 | 25.4 ± 3.4 | WP | 30 | MD |
| Dulac et al., 2021 (b) [164] | Canada | R, P, DB, PC | Older men | ♂ | 20 | 19 | 12 | 69 ± 6.1 | 70.7 ± 8.6 | 26 ± 3.5 | 25.4 ± 3.4 | CP | 30 | MD |
| Roberson et al., 2021 [165] | USA | R, P, DB, PC | College-aged men | ♂ | 17 | 12 | 12 | 21 ± 2 | 21 ± 1 | NR | NR | WP | 52.6 | MD |
| Nakayama et al., 2021 [94] | Japan | R, P, DB, PC | Healthy older adults | ♂/♀ | 61 | 61 | 24 | 71.4 ± 6.2 | 70.4 ± 5.5 | 23.1 ± 3.1 | 22.8 ± 3.1 | MP | 10 | PL |
| Koopmans et al., 2024 [190] | Netherlands | R, P, DB, PC | Physically active older adults | ♂/♀ | 23 | 20 | 11 | 70 ±5 | 68 ± 5 | 24.4 ± 2.3 | 23.8 ± 2.7 | WP | 30 | MD |
| Azhar et al., 2021 [95] | USA | R, P, DB, CO | Older adults | ♂/♀ | 32 | 29 | 12 | 66–86 | 66–86 | 31.7 ± 6.1 | 32.7 ± 1.5 | WP | 15 | Nutrition education |
| Li et al., 2021 [96] | China | R, P, CO | Older adults with low lean mass | ♂/♀ | 16 | 30 | 24 | 71 ± 4 | 71 ± 4 | 21.8 ± 2 | 20.8 ± 2.2 | WP | 16 | NI |
| Mizubuti et al., 2021 (a) [97] | Brazil | R, P, DB, PC | Patients with chronic liver disease | ♂/♀ | 35 | 40 | 2 | 51.6 ± 9.4 | 52.6 ± 11.4 | NR | NR | WP | 40 | CP |
| Mizubuti et al., 2021 (b) [97] | Brazil | R, P, DB, PC | Patients with chronic liver disease | ♂/♀ | 40 | 35 | 2 | 52.6 ± 11.4 | 51.6 ± 9.4 | NR | NR | CP | 40 | WP |
| Mertz et al., 2021 [98] | Denmark | R, P, DB, PC | Healthy older adults | ♂/♀ | 44 | 34 | 48 | 70.3 ± 4.3 | 69.6 ± 3.9 | 25.2 ± 3.6 | 26 ± 3.9 | WP | 40 | MD+ sucrose |
| Bach et al., 2022 [99] | Brazil | R, P, DB, PC | Older adults | ♂/♀ | 15 | 16 | 12 | 66.9 ± 4.3 | 65.8 ± 5.0 | 26.3 ± 2.2 | 25.4 ± 2.0 | WP | 40 | MD |
| Henriques et al., 2023 [125] | Brazil | R, P, DB, PC | Patients underwent bariatric surgery | ♀ | 17 | 15 | 8 | 46 ± 8.26 | 47.6 ± 7.4 | 31.2 ± 3.1 | 32.9 ± 6.3 | WP | 30 | MD |
| Zbinden-Foncea et al., 2023 [166] | Chile | R, P, SB, PC | Untrained young men | ♂ | 6 | 6 | 8 | 22.4 ± 3.1 | 22.3 ± 1.9 | WP | 9.85 | Sugar-free orange juice | ||
| Yapici et al., 2023 [167] | Saudi Arabia | R, P, CO | Untrained young men | ♂ | 11 | 11 | 8 | 20.9 ± 0.9 | 19.8 ± 0.7 | 21.7 ± 1.4 | 23.8 ± 1.1 | MP | 12.85 | Resistance training program |
| Kim et al., 2023 [168] | South Korea | R, P, DB, PC | Healthy sedentary men | ♂ | 17 | 15 | 12 | 23.5 ± 2.7 | 24.5 ± 3.3 | 24 ± 1.2 | 24.3 ± 1.8 | WP | 60 | CHO |
| Zong et al., 2023 [100] | China | R, P, CO | Elderly inpatients with COPD | ♂/♀ | 27 | 29 | 12 | 80.5 ± 7.7 | 81.1 ± 12 | 21.6 ± 3.6 | 24.6 ± 3.8 | WP | 20 | Low-intensity exercise |
| Nouri et al., 2024 [126] | Iran | R, P, DB, PC | Women with T2DM, OW, & OB | ♀ | 18 | 17 | 12 | 44 ± 6.2 | 46.9 ± 5.1 | 32.5 ± 4.2 | 31.6 ± 5.0 | WP | 20 | Unfortified bread |
| Furtado et al., 2024 [101] | Brazil | R, P, DB, PC | Older adults with T2DM | ♂/♀ | 19 | 20 | 12 | 68.0 ± 5.7 | 66.6 ± 6.3 | 30.3 ± 6.1 | 30.7 ± 6.1 | WP | 9.42 | MD |
| Kemmler et al., 2018 [169] | Germany | R, P, CO | Patients with sarcopenic OB | ♂ | 33 | 34 | 16 | 78.1 ± 5.4 | 76.9 ± 5.2 | 26.3 ± 2.5 | 26 ± 2.5 | WP | 137 | NI |
| Kirk et al., 2020 (a) [189] | UK | R, P, CO | Older adults | ♂/♀ | 22 | 24 | 16 | 69 ± 6 | 66 ± 4 | 27.4 ± 4.9 | 28.1 ± 7.4 | WP | 111 | Exercise |
| Kirk et al., 2020 (b) [189] | UK | R, P, CO | Older adults | ♂/♀ | 23 | 31 | 16 | 72 ± 6 | 68 ± 6 | 27.1 ± 4.1 | 26.2 ± 4.5 | WP | 111 | NI |
| Santos et al., 2023 [102] | Brazil | R, P, SB, PC | Patients with CHD | ♂/♀ | 15 | 10 | 12 | 64 ± 4.4 | 61 ± 14.8 | 28.6 ± 4.6 | 26.8 ± 3.5 | WPI | 30 | MD |
| Kasim-Karakas et al., 2009 [127] | USA | R, P, SB, PC | Women with PCOS, OW& OB | ♀ | 11 | 13 | 8 | 28 ± 3 | 38.9 ± 1.6 | 35.4 ± 1.2 | WP | 60 | CHO | |
| Lockwood et al., 2017 (a) [170] | USA | R, P, DB, PC | Healthy men | ♂ | 15 | 15 | 8 | 21.8 ± 3.5 | 20.9 ± 1.5 | 24.9 ± 8.5 | 23.8 ± 8.5 | WPC-L | 60 | CHO |
| Lockwood et al., 2017 (b) [170] | USA | R, P, DB, PC | Healthy men | ♂ | 13 | 15 | 8 | 21.3 ± 2.5 | 20.9 ± 1.5 | 25.9 ± 5.8 | 23.8 ± 8.5 | WPC | 60 | CHO |
| Lockwood et al., 2017 (c) [170] | USA | R, P, DB, PC | Healthy men | ♂ | 13 | 15 | 8 | 21.5 ± 3.2 | 20.9 ± 1.5 | 25.1 ± 6.5 | 23.8 ± 8.5 | WPH | 60 | CHO |
| Knuiman et al., 2019 [172] | Netherlands | R, P, DB, PC | Recreationally active men | ♂ | 19 | 21 | 10 | 21.5 ± 1.7 | 22.5 ± 2.3 | 22.3 ± 1.7 | 22.4 ± 1.4 | CP | 41 | CHO |
| Mhamed et al., 2024 [173] | Tunisia | R, P, CO | Well-trained endurance athletes | ♂ | 20 | 9 | 8 | NR | NR | 19.7 ± 0.6 | 20.2 ± 0.9 | WP | 30 | NI |
| Bodaghabadi et al., 2023 [174] | Iran | R, P, CO | Women with OW | ♀ | 26 | 21 | 2 | 37.8 ± 6.5 | 37.8 ± 6.5 | 26.9 ± 1.6 | 27.8 ± 1.7 | CP | 40 | High PR, low-dairy diet |
| Soares et al., 2023 [175] | Brazil | R, P, TB, PC | Men with T2DM | ♂ | 13 | 13 | 12 | 68.1 ± 4.5 | 68.9 ± 4.1 | 29.3 ± 2.6 | 26.8 ± 3.8 | WPI | 5.71 | MD |
| Ferguson-Stegall et al., 2011 [176] | USA | R, P, PC | Untrained individuals | ♂/♀ | 11 | 10 | 4.5 | 22.1 ± 2.3 | 21.3 ± 3.2 | 24.8 ± 1.5 | 25.7 ± 1.6 | MP | 5.24 | Isocaloric fat |
| Wilborn et al., 2013 (a) [12] | USA | R, P, DB, PC | Collegiate female athletes | ♀ | 8 | 8 | 8 | 20.0 ± 1.9 | 21.0 ± 2.8 | 26.4 ± 9.2 | 29.0 ± 11.0 | WP | 13.71 | CP |
| Wilborn et al., 2013 (b) [12] | USA | R, P, DB, PC | Collegiate female athletes | ♀ | 8 | 8 | 8 | 21.0 ± 2.8 | 20.0 ± 1.9 | 29.0 ± 11.0 | 26.4 ± 9.2 | CP | 13.71 | WP |
| Reljic et al., 2022 [177] | Germany | R, P, DB, PC | Sedentary, healthy adults | ♂/♀ | 19 | 20 | 8 | 30.0 ± 7.8 | 32.5 ± 8.0 | 24.4 ± 3.2 | 24.9± 3.8 | WP | 18.6 | MD |
| Reljic et al., 2024 [178] | Germany | R, P, DB, PC | Untrained healthy adults | ♂/♀ | 19 | 17 | 8 | 26 ± 4 | 27 ± 6 | 21.8 ± 2.2 | 25.0 ± 4.3 | WP | 12.4 | MD |
| Murray et al., 2025 [179] | New Zealand | R, P, DB, PC | Pre-menopausal women | ♀ | 15 | 12 | 12 | 34.2 ±9.1 | 32.8 ± 9.7 | 24 ± 3.9 | 27.1 ± 3.1 | WP | 17.14 | Milo powder |
| Yıldız et al., 2025 (a) [180] | Turkey | R, P, CO | Individuals underwent laparoscopic SG | ♂/♀ | 15 | 15 | 12 | 35.1 ± 9.7 | 35.1 ± 9.7 | 42.3 ± 6.1 | 41.0 ± 3.1 | CP | 15 | Standard PR diet |
| Yıldız et al., 2025 (b) [180] | Turkey | R, P, CO | Individuals underwent laparoscopic SG | ♂/♀ | 15 | 15 | 12 | 35.1 ± 9.7 | 35.1 ± 9.7 | 41.0 ± 5.2 | 41.0 ± 3.1 | CP | 15 | Standard PR diet |
| Sabooni et al., 2025 [181] | Iran | R, P, DB, PC | Patients underwent OAGB | ♂/♀ | 39 | 39 | 12 | 40.5 ± 10.7 | 41.4 ± 8.6 | 46.4 ± 5.6 | 43.4 ± 2.9 | WP | 22.6 | PL (no PR) |
| Ormsbee et al., 2018 [183] | USA | R, P, PC | Sedentary individuals | ♂/♀ | 29 | 22 | 24 | 21.0 ± 3.2 | 20.3 ± 2.3 | 23.6 ± 1.5 | 25.7 ± 1.6 | MP | 84 | CHD |
| Jonvik et al., 2019 [182] | Netherlands | R, P, DB, PC | Active men | ♂ | 30 | 26 | 12 | 26 ± 6 | 26 ± 6 | 23.8 ± 2.9 | 24.3 ± 2.3 | CP | 41 | CHD |
| Griffen et al., 2022 (a) [184] | UK | R, P, DB, PC | Healthy, active older men | ♂ | 9 | 9 | 12 | 68 ± 3 | 67 ± 3 | 26.6 ± 2.4 | 25.1 ± 2.7 | WP | 50 | MD |
| Griffen et al., 2022 (b) [184] | UK | R, P, DB, PC | Healthy, active older men | ♂ | 9 | 9 | 12 | 66 ± 6 | 67 ± 6 | 25.0 ± 1.8 | 25.1 ± 3 | WP | 50 | MD |
| Arnarson et al., 2013 [185] | Iceland | R, P, DB, PC | Elderly people | ♂/♀ | 75 | 66 | 12 | 73.3 ± 6.0 | 74.6 ± 5.8 | 28.1 ± 4.4 | 29.4 ± 4.8 | WP | 8.57 | CHO |
| Karelis et al., 2015 (a) [186] | Canada | R, P, DB, PC | Non-frail elderly individuals | ♂/♀ | 34 | 33 | 19.3 | 69.9 ± 3.6 | 71.0 ± 4.6 | 24.9 ± 2.8 | 25.4 ± 2.8 | WP | 20 | CP |
| Karelis et al., 2015 (b) [186] | Canada | R, P, DB, PC | Non-frail elderly individuals | ♂/♀ | 33 | 34 | 19.3 | 71.0 ± 4.6 | 69.9 ± 3.6 | 25.4 ± 2.8 | 24.9 ± 2.8 | CP | 20 | Cysteine-rich WP |
| Kirk et al., 2019 [187] | UK | R, P, CO | Older adults | ♂/♀ | 22 | 24 | 16 | 69 ± 6 | 66 ± 4 | 27.4 ± 4.9 | 28.1 ± 7.4 | WP | 111.3 | Exercise |
| Michel et al., 2022 [188] | USA | R, P, CO | Older adults | ♂/♀ | 9 | 9 | 10 | 67.3 ± 8.9 | 72.1 ± 7.1 | 24.3 ± 4.3 | 27.2 ± 5.4 | WP | 75 | Regular diet |
| Sub-Groups | Number of Effect Sizes | WMD (95%CI) | p-Value | Heterogeneity | ||
|---|---|---|---|---|---|---|
| p-Value Heterogeneity | I2 (%) | p-Value Between Sub-Groups | ||||
| BW (kg) | ||||||
| Overall effect | 133 | −22 (−0.52, 0.09) | 0.160 | <0.001 | 38.3 | |
| Trial duration (weeks) | ||||||
| >8 | 92 | −0.14 (−0.53, 0.24) | 0.459 | <0.001 | 50.7 | 0.780 |
| ≤8 | 41 | −0.22 (−0.63, 0.18) | 0.279 | 0.900 | 0 | |
| Intervention type | ||||||
| CP | 11 | −0.32 (−1.08, 0.44) | 0.408 | 0.999 | 0 | 0.902 |
| WP | 104 | −0.22 (−0.61, 0.15) | 0.248 | <0.001 | 48.9 | |
| MP | 18 | −0.14 (−0.53, 0.25) | 0.489 | 0.877 | 0 | |
| Supplement dose (g/day) | ||||||
| >30 | 53 | −0.13 (−0.42, 0.15) | 0.356 | 0.530 | 0 | 0.995 |
| ≤30 | 80 | −0.13 (−0.58, 0.31) | 0.547 | <0.001 | 51.6 | |
| Baseline BMI | ||||||
| Normal | 56 | 0.13 (−0.14, 0.40) | 0.346 | 0.335 | 6.6 | 0.005 |
| OW | 40 | 0.09 (−0.24, 0.44) | 0.585 | 0.863 | 0.0 | |
| OB | 37 | −1.50 (−2.46, −0.55) | 0.002 | <0.001 | 50.1 | |
| Sex | ||||||
| Both | 67 | −0.06 (−0.54, 0.41) | 0.793 | <0.001 | 60.0 | 0.239 |
| Female | 25 | −0.55 (−1.01, −0.09) | 0.019 | 0.811 | 0 | |
| Male | 41 | −0.07 (−0.50, 0.34) | 0.714 | 0.931 | 0 | |
| Health status | ||||||
| Healthy | 110 | −0.31 (−0.67, 0.05) | 0.092 | <0.001 | 39.9 | 0.290 |
| Unhealthy | 23 | 0.04 (−0.50, 0.59) | 0.876 | 0.071 | 32.0 | |
| Age | ||||||
| ≤60 | 93 | −0.50 (−0.89, −0.11) | 0.011 | <0.001 | 40.7 | 0.001 |
| >60 | 40 | 0.42 (0.03, 0.80) | 0.031 | 0.318 | 8.5 | |
| BMI (kg/m2) | ||||||
| Overall effect | 70 | −0.03 (−0.14, 0.09) | 0.626 | 0.309 | 7.2 | |
| Trial duration (weeks) | ||||||
| >8 | 45 | −0.07 (−0.19, 0.05) | 0.268 | 0.872 | 0 | 0.988 |
| ≤8 | 25 | −0.06 (−0.31, 0.17) | 0.586 | 0.022 | 39.9 | |
| Intervention type | ||||||
| CP | 9 | −0.02 (−0.33, 0.27) | 0.853 | 0.951 | 0 | 0.047 |
| WP | 54 | 0.01 (−0.11, 0.14) | 0.802 | 0.286 | 9.1 | |
| MP | 7 | −0.55 (−0.99, −0.12) | 0.012 | 0.389 | 4.9 | |
| Supplement dose (g/day) | ||||||
| >30 | 21 | −0.06 (−0.29, 0.17) | 0.609 | 0.339 | 9.2 | 0.782 |
| ≤30 | 49 | −0.02 (−0.15, 0.11) | 0.739 | 0.315 | 8.0 | |
| Baseline BMI | ||||||
| Normal | 20 | 0.05 (−0.18, 0.29) | 0.655 | 0.002 | 53.7 | 0.462 |
| OW | 25 | 0.01 (−0.18, 0.20) | 0.902 | 0.999 | 0 | |
| OB | 25 | −0.16 (−0.42, 0.10) | 0.237 | 0.436 | 1.8 | |
| Sex | ||||||
| Both | 38 | 0.08 (−0.05, 0.23) | 0.219 | 0.301 | 9.6 | 0.010 |
| Female | 16 | −0.36 (−0.65, −0.08) | 0.012 | 0.811 | 0 | |
| Male | 16 | −0.17 (−0.42, 0.08) | 0.187 | 0.451 | 0 | |
| Health status | ||||||
| Healthy | 50 | −0.05 (−0.20, 0.09) | 0.477 | 0.964 | 0 | 0.390 |
| Unhealthy | 20 | 0.08 (−0.19, 0.35) | 0.552 | 0.002 | 54.2 | |
| Age | ||||||
| ≤60 | 47 | −0.02 (−0.18, 0.13) | 0.779 | 0.040 | 28.2 | 0.800 |
| >60 | 23 | −0.05 (−0.28, 0.17) | 0.612 | 0.984 | 0 | |
| WC (cm) | ||||||
| Overall effect | 45 | −0.69 (−1.16, −0.22) | 0.004 | <0.001 | 47.9 | |
| Trial duration (weeks) | ||||||
| >8 | 31 | −0.84 (−1.40, −0.29) | 0.003 | <0.001 | 56.6 | 0.216 |
| ≤8 | 14 | −0.19 (−1.06, 0.66) | 0.652 | 0.326 | 11.6 | |
| Intervention type | ||||||
| CP | 6 | −0.25 (−0.73, 0.21) | 0.286 | 0.955 | 0 | 0.481 |
| WP | 36 | −0.71 (−1.29, −0.13) | 0.016 | <0.001 | 54.8 | |
| MP | 3 | −0.69 (−3.33, 1.94) | 0.605 | 0.096 | 57.4 | |
| Supplement dose (g/day) | ||||||
| >30 | 17 | −1.19 (−2.22, −0.15) | 0.024 | <0.001 | 67.8 | 0.096 |
| ≤30 | 28 | −0.27 (−0.59, 0.05) | 0.105 | 0.404 | 4.0 | |
| Baseline BMI | ||||||
| Normal | 6 | −0.42 (−1.05, 0.20) | 0.183 | 0.030 | 59.7 | 0.461 |
| OW | 14 | −0.48 (−1.04, 0.07) | 0.092 | 0.974 | 0 | |
| OB | 25 | −1.15 (−2.18, −0.13) | 0.027 | <0.001 | 61.1 | |
| Sex | ||||||
| Both | 25 | −0.28 (−0.54, −0.02) | 0.035 | 0.620 | 0 | 0.514 |
| Female | 15 | −0.44 (−1.28, 0.38) | 0.294 | 0.583 | 0 | |
| Male | 5 | −2.06 (−5.24, 1.10) | 0.202 | <0.001 | 90.1 | |
| Health status | ||||||
| Healthy | 35 | −0.77 (−1.45, −0.09) | 0.026 | <0.001 | 48.5 | 0.416 |
| Unhealthy | 10 | −0.42 (−0.94, 0.10) | 0.114 | 0.110 | 37.3 | |
| Age | ||||||
| ≤60 | 34 | −0.73 (−1.29, −0.17) | 0.011 | <0.001 | 59.6 | 0.981 |
| >60 | 11 | −0.72 (−1.38, −0.06) | 0.032 | 0.998 | 0 | |
| FM (kg) | ||||||
| Overall effect | 111 | −0.66 (−0.91, −0.41) | <0.001 | <0.001 | 42.1 | |
| Trial duration (weeks) | ||||||
| >8 | 74 | −0.44 (−0.70, −0.19) | 0.001 | 0.098 | 18.0 | 0.042 |
| ≤8 | 37 | −1.03 (−1.53, −0.52) | <0.001 | <0.001 | 58.9 | |
| Intervention type | ||||||
| CP | 12 | −0.06 (−0.69, 0.55) | 0.830 | 0.866 | 0 | 0.197 |
| WP | 85 | −0.70 (−1.02, −0.38) | <0.001 | <0.001 | 50.2 | |
| MP | 14 | −0.52 (−0.81, −0.24) | <0.001 | 0.658 | 0 | |
| Supplement dose (g/day) | ||||||
| >30 | 51 | −0.82 (−1.21, −0.43) | <0.001 | <0.001 | 54.3 | 0.173 |
| ≤30 | 60 | −0.47 (−0.78, −0.16) | 0.003 | 0.054 | 23.8 | |
| Baseline BMI | ||||||
| Normal | 35 | −0.70 (−1.16, −0.24) | 0.003 | <0.001 | 63.8 | 0.014 |
| OW | 47 | −0.25 (−0.50, −0.00) | 0.049 | 0.965 | 0 | |
| OB | 29 | −1.20 (−1.86, −0.54) | <0.001 | 0.022 | 37.8 | |
| Sex | ||||||
| Both | 49 | −0.28 (−0.50, −0.07) | 0.010 | 0.833 | 0 | 0.017 |
| Female | 21 | −1.09 (−1.71, −0.46) | 0.001 | 0.005 | 50.2 | |
| Male | 41 | −0.79 (−1.28, −0.31) | 0.001 | <0.001 | 55.8 | |
| Health status | ||||||
| Healthy | 99 | −0.72 (−0.98, −0.46) | <0.001 | <0.001 | 42.7 | 0.109 |
| Unhealthy | 12 | 0.01 (−0.85, 0.88) | 0.977 | 0.176 | 27.3 | |
| Age | ||||||
| ≤60 | 78 | −0.85 (−1.18, −0.53) | <0.001 | <0.001 | 49.8 | 0.001 |
| >60 | 33 | −0.10 (−0.39, 0.18) | 0.470 | 0.963 | 0 | |
| BFP (%) | ||||||
| Overall effect | 80 | −0.66 (−1.03, −0.28) | 0.001 | <0.001 | 71.2 | |
| Trial duration (weeks) | ||||||
| >8 | 55 | −0.69 (−1.19, −0.19) | 0.006 | <0.001 | 76.8 | 0.466 |
| ≤8 | 25 | −0.44 (−0.88, −0.00) | 0.046 | 0.108 | 26.9 | |
| Intervention type | ||||||
| CP | 9 | −0.77 (−3.08, 1.53) | 0.510 | <0.001 | 94.5 | 0.475 |
| WP | 61 | −0.73 (−1.09, −0.37) | <0.001 | <0.001 | 45.4 | |
| MP | 10 | −0.44 (−0.74, −0.15) | 0.003 | 0.968 | 0 | |
| Supplement dose (g/day) | ||||||
| >30 | 27 | −1.05 (−1.81, −0.29) | 0.006 | <0.001 | 88.5 | 0.268 |
| ≤30 | 53 | −0.59 (−0.87, −0.32) | <0.001 | 0.716 | 0 | |
| Baseline BMI | ||||||
| Normal | 28 | −0.63 (−0.88, −0.39) | <0.001 | 0.860 | 0 | 0.100 |
| OW | 32 | −0.28 (−0.57, 0.00) | 0.051 | 0.659 | 0 | |
| OB | 20 | −1.27 (−2.63, 0.08) | 0.066 | <0.001 | 89.1 | |
| Sex | ||||||
| Both | 37 | −0.75 (−1.36, −0.15) | 0.014 | <0.001 | 80.3 | 0.671 |
| Female | 14 | −0.75 (−1.41, −0.09) | 0.026 | 0.075 | 37.8 | |
| Male | 29 | −0.41 (−1.02, 0.20) | 0.188 | <0.001 | 58.3 | |
| Health status | ||||||
| Healthy | 70 | −0.64 (−1.05, −0.22) | 0.003 | <0.001 | 73.7 | 0.344 |
| Unhealthy | 10 | −0.98 (−1.57, −0.40) | 0.001 | 0.328 | 12.4 | |
| Age | ||||||
| ≤60 | 59 | −0.69 (−1.16, −0.22) | 0.004 | <0.001 | 77.1 | 0.342 |
| >60 | 21 | −0.39 (−0.79, 0.00) | 0.053 | 0.680 | 0 | |
| FFM (Kg) | ||||||
| Overall effect | 40 | 0.67 (0.40, 0.94) | <0.001 | 0.483 | 0 | |
| Trial duration (weeks) | ||||||
| >8 | 26 | 0.81 (0.45, 1.17) | <0.001 | 0.881 | 0 | 0.194 |
| ≤8 | 14 | 0.34 (−0.26, 0.95) | 0.272 | 0.087 | 36.1 | |
| Intervention type | ||||||
| CP | 5 | 0.38 (−0.59, 1.36) | 0.455 | 0.429 | 0 | 0.031 |
| WP | 32 | 0.84 (0.54, 1.15) | <0.001 | 0.805 | 0 | |
| MP | 3 | −0.40 (−1.31, 0.50) | 0.385 | 0.318 | 12.6 | |
| Supplement dose (g/day) | ||||||
| >30 | 15 | 0.43 (−0.25, 1.12) | 0.213 | 0.099 | 33.6 | 0.682 |
| ≤30 | 25 | 0.59 (0.26, 0.93) | 0.001 | 0.845 | 0 | |
| Baseline BMI | ||||||
| Normal | 18 | 0.26 (−0.25, 0.78) | 0.314 | 0.257 | 16.4 | 0.059 |
| OW | 13 | 0.54 (−0.04, 1.12) | 0.069 | <0.001 | 0 | |
| OB | 9 | 1.09 (0.62, 1.57) | <0.001 | 0.386 | 5.9 | |
| Sex | ||||||
| Both | 16 | 0.64 (0.11, 1.18) | 0.018 | 0.458 | 0 | 0.004 |
| Female | 6 | 1.15 (0.70, 1.60) | <0.001 | 0.385 | 5.0 | |
| Male | 18 | 0.05 (−0.41, 0.53) | 0.816 | 0.987 | 0 | |
| Health status | ||||||
| Healthy | 30 | 0.66 (0.37, 0.95) | <0.001 | 0.606 | 0 | 0.901 |
| Unhealthy | 10 | 0.72 (−0.24, 1.69) | 0.141 | 0.197 | 26.9 | |
| Age | ||||||
| ≤60 | 29 | 0.44 (0.02, 0.86) | 0.040 | 0.149 | 21.7 | 0.311 |
| >60 | 11 | 0.79 (0.25, 1.33) | 0.004 | 0.988 | 0 | |
| LBM (kg) | ||||||
| Overall effect | 65 | 0.41 (0.19, 0.62) | <0.001 | 0.036 | 25.5 | |
| Trial duration (weeks) | ||||||
| >8 | 46 | 0.32 (0.05, 0.58) | 0.018 | 0.048 | 27.3 | 0.011 |
| ≤8 | 19 | 0.77 (0.55, 0.99) | <0.001 | 0.911 | 0 | |
| Intervention type | ||||||
| CP | 7 | 1.10 (−0.62, 2.83) | 0.211 | 0.002 | 71.6 | 0.560 |
| WP | 47 | 0.33 (0.09, 0.57) | 0.006 | 0.132 | 19.0 | |
| MP | 11 | 0.46 (0.21, 0.72) | <0.001 | 0.906 | 0 | |
| Supplement dose (g/day) | ||||||
| >30 | 29 | 0.59 (0.18, 1.01) | 0.005 | 0.012 | 41.1 | 0.271 |
| ≤30 | 36 | 0.34 (0.14, 0.53) | <0.001 | 0.399 | 4.1 | |
| Baseline BMI | ||||||
| Normal | 20 | 0.64 (0.46, 0.81) | <0.001 | 0.607 | 0 | 0.002 |
| OW | 25 | 0.12 (−0.10, 0.35) | 0.293 | 0.814 | 0 | |
| OB | 20 | 0.61 (−0.20, 1.42) | 0.142 | 0.005 | 51.1 | |
| Sex | ||||||
| Both | 37 | 0.27 (−0.02, 0.56) | 0.069 | 0.008 | 39.7 | 0.026 |
| Female | 7 | 0.62 (0.19, 1.06) | 0.005 | 0.844 | 0 | |
| Male | 21 | 0.86 (0.54, 1.17) | <0.001 | 0.842 | 0 | |
| Health status | ||||||
| Healthy | 59 | 0.38 (0.14, 0.62) | 0.002 | 0.037 | 26.2 | 0.072 |
| Unhealthy | 6 | 0.73 (0.44, 1.02) | <0.001 | 0.795 | 0 | |
| Age | ||||||
| ≤60 | 41 | 0.65 (0.34, 0.96) | <0.001 | 0.073 | 25.4 | 0.024 |
| >60 | 24 | 0.23 (0.03, 0.42) | 0.021 | 0.433 | 2.1 | |
| MM (kg) | ||||||
| Overall effect | 17 | −0.07 (−0.33, 0.19) | 0.588 | 1.000 | 0 | |
| Trial duration (weeks) | ||||||
| >8 | 12 | −0.07 (−0.34, 0.19) | 0.596 | 0.995 | 0 | 0.970 |
| ≤8 | 5 | −0.05 (−0.96, 0.85) | 0.906 | 0.995 | 0 | |
| Intervention type | ||||||
| CP | 2 | 2.47 (−1.55, 6.49) | 0.229 | 0.846 | 0 | 0.450 |
| WP | 14 | −0.02 (−0.54, 0.49) | 0.922 | 1.000 | 0 | |
| MP | 1 | −0.10 (−0.39, 0.19) | 0.511 | - | - | |
| Supplement dose (g/day) | ||||||
| >30 | 11 | −0.08 (−0.34, 0.18) | 0.542 | 1.000 | 0 | 0.708 |
| ≤30 | 6 | 0.14 (−1.03, 1.32) | 0.805 | 0.858 | 0 | |
| Baseline BMI | ||||||
| Normal | 7 | −0.21 (−1.08, 0.66) | 0.635 | 1.000 | 0 | 0.500 |
| OW | 7 | −0.07 (−0.35, 0.19) | 0.575 | 0.997 | 0 | |
| OB | 3 | 1.15 (−0.96, 3.27) | 0.285 | 0.738 | 0 | |
| Sex | ||||||
| Both | 10 | −0.09 (−0.37, 0.18) | 0.521 | 0.988 | 0 | 0.711 |
| Male | 7 | 0.04 (−0.63, 0.73) | 0.888 | 0.998 | 0 | |
| Health status | ||||||
| Healthy | 13 | −0.09 (−0.35, 0.17) | 0.492 | 1.000 | 0 | 0.329 |
| Unhealthy | 4 | 0.74 (−0.91, 2.39) | 0.379 | 0.806 | 0 | |
| Age | ||||||
| ≤60 | 11 | 0.03 (−0.68, 0.75) | 0.929 | 0.995 | 0 | 0.761 |
| >60 | 6 | −0.08 (−0.36, 0.19) | 0.539 | 0.992 | 0 | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mohammadi, S.; Ashtary-Larky, D.; Alaghemand, N.; Alnsour, A.F.; Shokouhifar, S.; Borzabadi, A.; Mehrbod, M.; Candow, D.G.; Forbes, S.C.; Antonio, J.; et al. Effects of Supplementation with Milk Proteins on Body Composition and Anthropometric Parameters: A Systematic Review and Dose–Response Meta-Analysis. Nutrients 2025, 17, 3877. https://doi.org/10.3390/nu17243877
Mohammadi S, Ashtary-Larky D, Alaghemand N, Alnsour AF, Shokouhifar S, Borzabadi A, Mehrbod M, Candow DG, Forbes SC, Antonio J, et al. Effects of Supplementation with Milk Proteins on Body Composition and Anthropometric Parameters: A Systematic Review and Dose–Response Meta-Analysis. Nutrients. 2025; 17(24):3877. https://doi.org/10.3390/nu17243877
Chicago/Turabian StyleMohammadi, Shooka, Damoon Ashtary-Larky, Navid Alaghemand, Amneh F. Alnsour, Shokoufeh Shokouhifar, Aida Borzabadi, Milad Mehrbod, Darren G. Candow, Scott C. Forbes, Jose Antonio, and et al. 2025. "Effects of Supplementation with Milk Proteins on Body Composition and Anthropometric Parameters: A Systematic Review and Dose–Response Meta-Analysis" Nutrients 17, no. 24: 3877. https://doi.org/10.3390/nu17243877
APA StyleMohammadi, S., Ashtary-Larky, D., Alaghemand, N., Alnsour, A. F., Shokouhifar, S., Borzabadi, A., Mehrbod, M., Candow, D. G., Forbes, S. C., Antonio, J., Suzuki, K., & Asbaghi, O. (2025). Effects of Supplementation with Milk Proteins on Body Composition and Anthropometric Parameters: A Systematic Review and Dose–Response Meta-Analysis. Nutrients, 17(24), 3877. https://doi.org/10.3390/nu17243877












