SLAB51 Multi-Strain Probiotic Formula Increases Oxygenation in Oxygen-Treated Preterm Infants
Abstract
1. Introduction
2. Materials and Methods
2.1. Pilot Sample
2.2. In Vitro Study
2.2.1. Preparation of Bacterial Lysate for Cell Treatments
2.2.2. Caco-2 IECs
2.2.3. Western Blot
2.3. Statistical Analysis
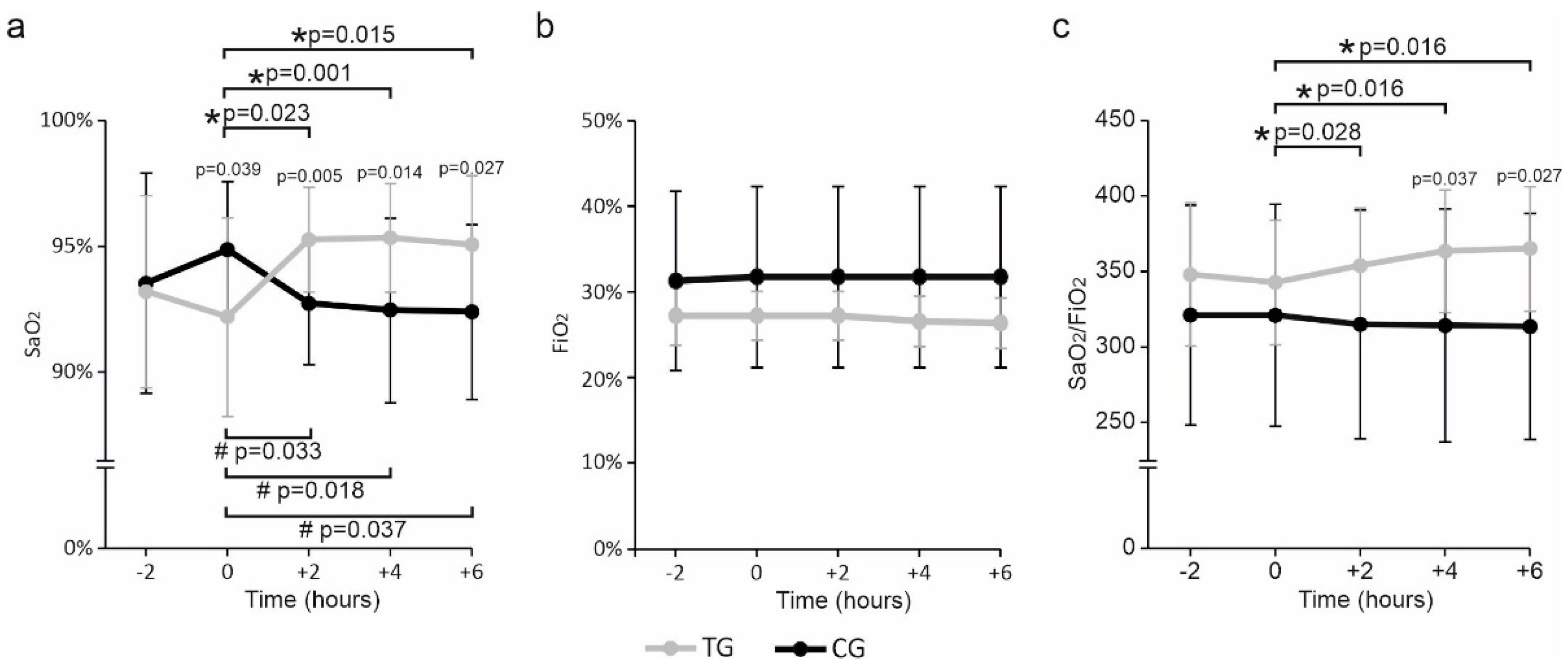
3. Results
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Liu, L.; Oza, S.; Hogan, D.; Chu, Y.; Perin, J.; Zhu, J.; Lawn, J.E.; Cousens, S.; Mathers, C.; Black, R.E. Global, regional, and national causes of under-5 mortality in 2000–15: An updated systematic analysis with implications for the Sustainable Development Goals. Lancet 2016, 388, 3027–3035. [Google Scholar] [CrossRef] [PubMed]
- Martin, R.J.; Wang, K.; Köroğlu, O.; Di Fiore, J.; Kc, P. Intermittent Hypoxic Episodes in Preterm Infants: Do They Matter? Neonatology 2011, 100, 303–310. [Google Scholar] [CrossRef] [PubMed]
- Di Fiore, J.M.; MacFarlane, P.M.; Martin, R.J. Intermittent Hypoxemia in Preterm Infants. Clin. Perinatol. 2019, 46, 553–565. [Google Scholar] [CrossRef] [PubMed]
- Mach, W.J.; Thimmesch, A.R.; Pierce, J.T. Consequences of Hyperoxia and the Toxicity of Oxygen in the Lung. Nurs. Res. Pract. 2011, 2011, 260482. [Google Scholar] [CrossRef]
- Perrone, S.; Giordano, M.; De Bernardo, G.; Lugani, P.; Sarnacchiaro, P.; Stazzoni, G.; Buonocore, G.; Esposito, S.; Tataranno, M.L.; National Study Group of Neonatal Clinical Biochemistry of the Italian Society of Neonatology. Management of oxygen saturation monitoring in preterm newborns in the NICU: The Italian picture. Ital. J. Pediatr. 2021, 47, 104. [Google Scholar] [CrossRef]
- Buonocore, G.; Perrone, S.; Tataranno, M.L. Oxygen toxicity: Chemistry and biology of reactive oxygen species. Semin. Fetal Neonatal Med. 2010, 15, 186–190. [Google Scholar] [CrossRef]
- Buonocore, G.; Perrone, S.; Vezzosi, P.; Longini, M.; Marzocchi, B.; Paffetti, P.; Bellieni, C.V.; Martinelli, S. Biomarkers of oxidative stress in babies at high risk for retinopathy of prematurity. Front. Biosci. 2009, E1, 547–552. [Google Scholar] [CrossRef][Green Version]
- Perrone, S.; Santacroce, A.; Longini, M.; Proietti, F.; Bazzini, F.; Buonocore, G. The Free Radical Diseases of Prematurity: From Cellular Mechanisms to Bedside. Oxidative Med. Cell. Longev. 2018, 2018, 7483062. [Google Scholar] [CrossRef]
- Clucas, L.; Doyle, L.W.; Dawson, J.; Donath, S.; Davis, P.G. Compliance with Alarm Limits for Pulse Oximetry in Very Preterm Infants. Pediatrics 2007, 119, 1056–1060. [Google Scholar] [CrossRef]
- Sola, A.; Golombek, S.G.; Bueno, M.T.M.; Lemus-Varela, L.; Zuluaga, C.; Domínguez, F.; Baquero, H.; Sarmiento, A.E.Y.; Natta, D.; Perez, J.M.R.; et al. Safe oxygen saturation targeting and monitoring in preterm infants: Can we avoid hypoxia and hyperoxia? Acta Paediatr. 2014, 103, 1009–1018. [Google Scholar] [CrossRef]
- Kapadia, V.; Oei, J.L. Optimizing oxygen therapy for preterm infants at birth: Are we there yet? Semin. Fetal Neonatal Med. 2020, 25, 101081. [Google Scholar] [CrossRef] [PubMed]
- Park, A.M.; Sanders, T.A.; Maltepe, E. Hypoxia-inducible factor (HIF) and HIF-stabilizing agents in neonatal care. Semin. Fetal Neonatal Med. 2010, 15, 196–202. [Google Scholar] [CrossRef] [PubMed]
- Amata, E.; Pittalà, V.; Marrazzo, A.; Parenti, C.; Prezzavento, O.; Arena, E.; Nabavi, S.M.; Salerno, L. Role of the Nrf2/HO-1 axis in bronchopulmonary dysplasia and hyperoxic lung injuries. Clin. Sci. 2017, 131, 1701–1712. [Google Scholar] [CrossRef] [PubMed]
- Kasai, S.; Shimizu, S.; Tatara, Y.; Mimura, J.; Itoh, K. Regulation of Nrf2 by Mitochondrial Reactive Oxygen Species in Physiology and Pathology. Biomolecules 2020, 10, 320. [Google Scholar] [CrossRef]
- Ceccarelli, G.; Marazzato, M.; Celani, L.; Lombardi, F.; Piccirilli, A.; Mancone, M.; Trinchieri, V.; Pugliese, F.; Mastroianni, C.M.; D’ettorre, G. Oxygen Sparing Effect of Bacteriotherapy in COVID-19. Nutrients 2021, 13, 2898. [Google Scholar] [CrossRef]
- Trinchieri, V.; Marazzato, M.; Ceccarelli, G.; Lombardi, F.; Piccirilli, A.; Santinelli, L.; Maddaloni, L.; Vassalini, P.; Mastroianni, C.M.; D’ettorre, G. Exploiting Bacteria for Improving Hypoxemia of COVID-19 Patients. Biomedicines 2022, 10, 1851. [Google Scholar] [CrossRef] [PubMed]
- Lombardi, F.; Augello, F.R.; Palumbo, P.; Bonfili, L.; Artone, S.; Altamura, S.; Sheldon, J.M.; Latella, G.; Cifone, M.G.; Eleuteri, A.M.; et al. Bacterial Lysate from the Multi-Strain Probiotic SLAB51 Triggers Adaptative Responses to Hypoxia in Human Caco-2 Intestinal Epithelial Cells under Normoxic Conditions and Attenuates LPS-Induced Inflammatory Response. Int. J. Mol. Sci. 2023, 24, 8134. [Google Scholar] [CrossRef]
- Castelli, V.; d’Angelo, M.; Lombardi, F.; Alfonsetti, M.; Antonosante, A.; Catanesi, M.; Benedetti, E.; Palumbo, P.; Cifone, M.G.; Giordano, A.; et al. Effects of the probiotic formulation SLAB51 in in vitro and in vivo Parkinson’s disease models. Aging 2020, 12, 4641–4659. [Google Scholar] [CrossRef]
- Bonfili, L.; Cecarini, V.; Cuccioloni, M.; Angeletti, M.; Berardi, S.; Scarpona, S.; Rossi, G.; Eleuteri, A.M. SLAB51 Probiotic Formulation Activates SIRT1 Pathway Promoting Antioxidant and Neuroprotective Effects in an AD Mouse Model. Mol. Neurobiol. 2018, 55, 7987–8000. [Google Scholar] [CrossRef]
- Campbell, N.K.; Fitzgerald, H.K.; Dunne, A. Regulation of inflammation by the antioxidant haem oxygenase 1. Nat. Rev. Immunol. 2021, 21, 411–425. [Google Scholar] [CrossRef]
- Dang, A.T.; Marsland, B.J. Microbes, metabolites, and the gut-lung axis. Mucosal Immunol. 2019, 12, 843–850. [Google Scholar] [CrossRef] [PubMed]
- Morais, L.H.; Schreiber, H.L., 4th; Mazmanian, S.K. The gut microbiota-brain axis in behaviour and brain disorders. Nat. Rev. Microbiol. 2021, 19, 241–255. [Google Scholar] [CrossRef] [PubMed]
- Mancin, L.; Wu, G.D.; Paoli, A. Gut microbiota-bile acid-skeletal muscle axis. Trends Microbiol. 2022, 31, 254–269. [Google Scholar] [CrossRef] [PubMed]
- Bonfili, L.; Gong, C.; Lombardi, F.; Cifone, M.G.; Eleuteri, A.M. Strategic Modification of Gut Microbiota through Oral Bacteriotherapy Influences Hypoxia Inducible Factor-1α: Therapeutic Implication in Alzheimer’s Disease. Int. J. Mol. Sci. 2021, 23, 357. [Google Scholar] [CrossRef] [PubMed]
- Pollard, P.J.; Yang, M.; Su, H.; Soga, T.; Kranc, K.R. Prolyl hydroxylase domain enzymes: Important regulators of cancer metabolism. Hypoxia 2014, 2, 127–142. [Google Scholar] [CrossRef]
- Taylor, C.T.; Scholz, C.C. The effect of HIF on metabolism and immunity. Nat. Rev. Nephrol. 2022, 18, 573–587. [Google Scholar] [CrossRef]
- Weidemann, A.; Johnson, R.S. Biology of HIF-1α. Cell Death Differ. 2008, 15, 621–627. [Google Scholar] [CrossRef]
- Singhal, R.; Shah, Y.M. Oxygen battle in the gut: Hypoxia and hypoxia-inducible factors in metabolic and inflammatory responses in the intestine. J. Biol. Chem. 2020, 295, 10493–10505. [Google Scholar] [CrossRef]
- Semenza, G.L. Regulation of Oxygen Homeostasis by Hypoxia-Inducible Factor 1. Physiology 2009, 24, 97–106. [Google Scholar] [CrossRef]
- Hoppe, G.; Yoon, S.; Gopalan, B.; Savage, A.R.; Brown, R.; Case, K.; Vasanji, A.; Chan, E.R.; Silver, R.B.; Sears, J.E. Comparative systems pharmacology of HIF stabilization in the prevention of retinopathy of prematurity. Proc. Natl. Acad. Sci. USA 2016, 113, E2516–E2525. [Google Scholar] [CrossRef]
- Wang, L.; Chen, Y.; Sternberg, P.; Cai, J. Essential Roles of the PI3 Kinase/Akt Pathway in Regulating Nrf2-Dependent Anti-oxidant Functions in the RPE. Investig. Ophthalmol. Vis. Sci. 2008, 49, 1671–1678. [Google Scholar] [CrossRef] [PubMed]
- Reddy, N.M.; Potteti, H.R.; Vegiraju, S.; Chen, H.-J.; Tamatam, C.M.; Reddy, S.P. PI3K-AKT Signaling via Nrf2 Protects against Hyperoxia-Induced Acute Lung Injury, but Promotes Inflammation Post-Injury Independent of Nrf2 in Mice. PLoS ONE 2015, 10, e0129676. [Google Scholar] [CrossRef] [PubMed]

| Clinical Variable | TG (No. 15) | CG (No. 15) | p Value |
|---|---|---|---|
| Gestational age at birth (weeks) (mean ± SD) | 29.7 ± 4.1 | 27.7 ± 3.6 | 0.16 |
| Gestational age at enrollment (weeks) (mean ± SD) | 33.3 ± 4.1 | 31.5 ± 5.1 | 0.43 |
| Gestational age at enrollment <32 weeks (no. -%) | 10–66.7% | 14–93.3% | 0.17 |
| Weight at birth (kg) (mean ± SD) | 1.6 ± 0.89 | 1.1 ± 0.72 | 0.11 |
| Weight at enrollment (kg) (mean ± SD) | 1.9 ± 0.82 | 1.5 ± 0.95 | 0.24 |
| Apgar score | |||
| 1 min | 6.6 ± 2.5 | 5.4 ± 1.7 | 0.14 |
| 5 min | 8.0 ± 1.6 | 8.0 ± 0.84 | 1 |
| Feeding (no. -%) | |||
| Breastfeeding | 0–0.0% | 1–6.7% | 1 |
| Breastfeeding + preterm formula | 11–73.3% | 11–73.3% | 0.68 |
| Breastfeeding + formula type 1 | 1–6.7% | 1–6.7% | 0.46 |
| Breastfeeding + hydrolyzed formula | 3–20.0% | 1–6.7% | 0.59 |
| Total parenteral nutrition | 0–0.0% | 1–6.7% | 1 |
| Ventilation mode (no. -%) | |||
| A/C | 0–0.0% | 1–6.7% | 1 |
| HFT | 15–100.0% | 12–80.0% | 0.22 |
| SIMV | 0–0.0% | 2–13.3% | 0.46 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Baldassarre, M.E.; Marazzato, M.; Pensa, M.; Loverro, M.T.; Quercia, M.; Lombardi, F.; Schettini, F.; Laforgia, N. SLAB51 Multi-Strain Probiotic Formula Increases Oxygenation in Oxygen-Treated Preterm Infants. Nutrients 2023, 15, 3685. https://doi.org/10.3390/nu15173685
Baldassarre ME, Marazzato M, Pensa M, Loverro MT, Quercia M, Lombardi F, Schettini F, Laforgia N. SLAB51 Multi-Strain Probiotic Formula Increases Oxygenation in Oxygen-Treated Preterm Infants. Nutrients. 2023; 15(17):3685. https://doi.org/10.3390/nu15173685
Chicago/Turabian StyleBaldassarre, Maria Elisabetta, Massimiliano Marazzato, Marta Pensa, Maria Teresa Loverro, Michele Quercia, Francesca Lombardi, Federico Schettini, and Nicola Laforgia. 2023. "SLAB51 Multi-Strain Probiotic Formula Increases Oxygenation in Oxygen-Treated Preterm Infants" Nutrients 15, no. 17: 3685. https://doi.org/10.3390/nu15173685
APA StyleBaldassarre, M. E., Marazzato, M., Pensa, M., Loverro, M. T., Quercia, M., Lombardi, F., Schettini, F., & Laforgia, N. (2023). SLAB51 Multi-Strain Probiotic Formula Increases Oxygenation in Oxygen-Treated Preterm Infants. Nutrients, 15(17), 3685. https://doi.org/10.3390/nu15173685









