Intermittent Fasting—Short- and Long-Term Quality of Life, Fatigue, and Safety in Healthy Volunteers: A Prospective, Clinical Trial
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design and Setting
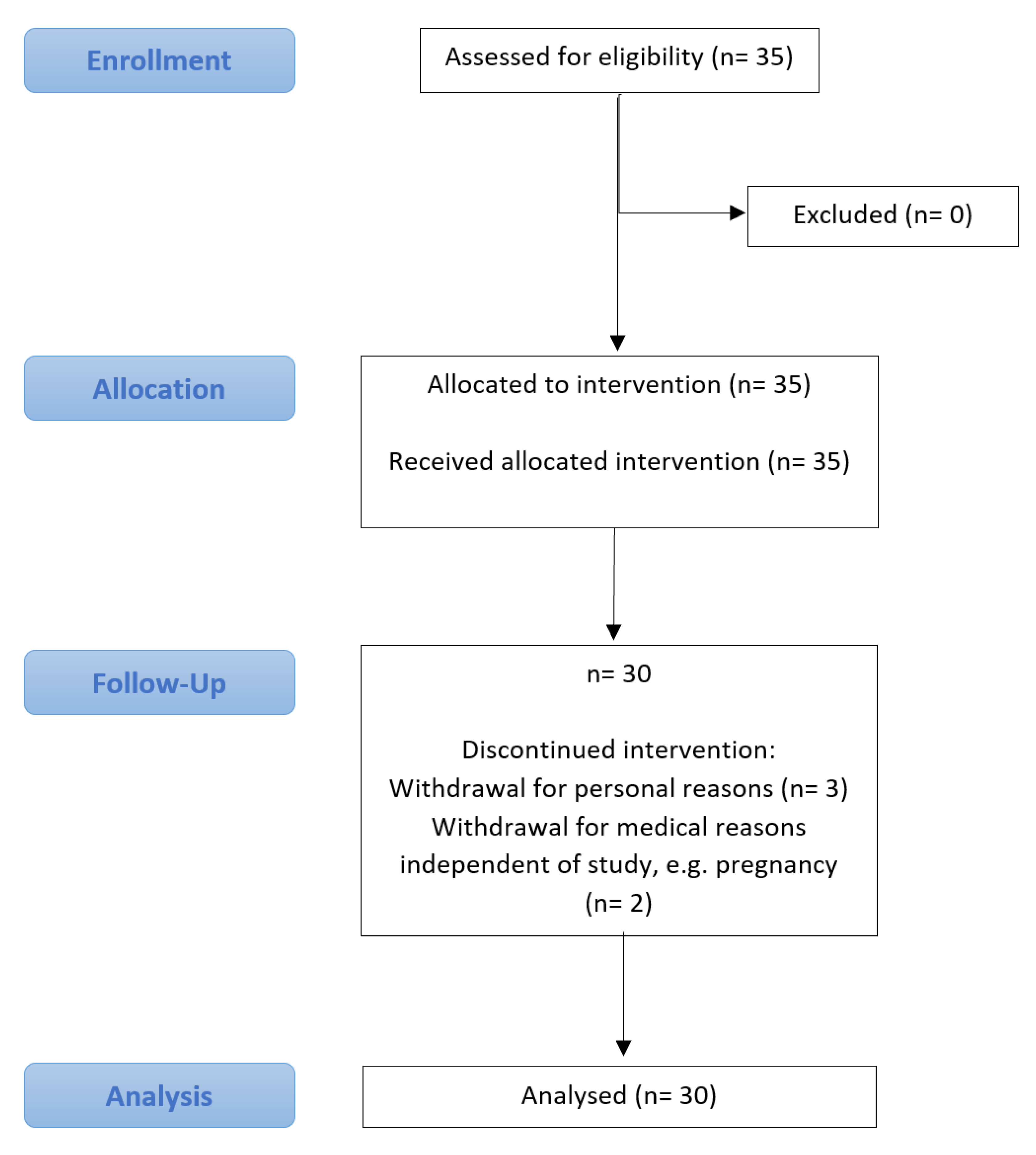
2.1.1. Participants
2.1.2. Intervention
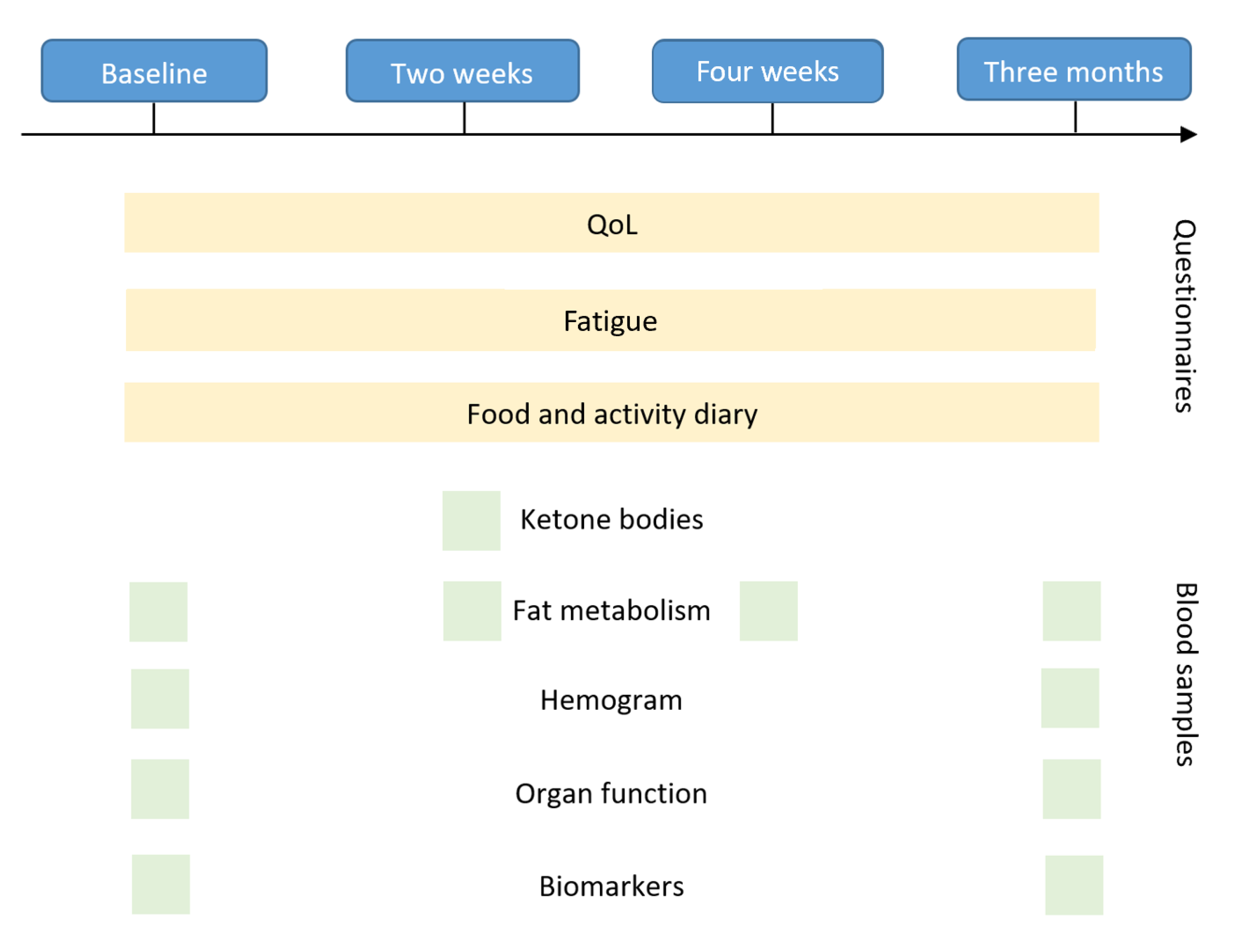
2.1.3. Outcomes
2.1.4. Quality of Life
2.2. WHO-5 Well-Being Index (WHO-5)
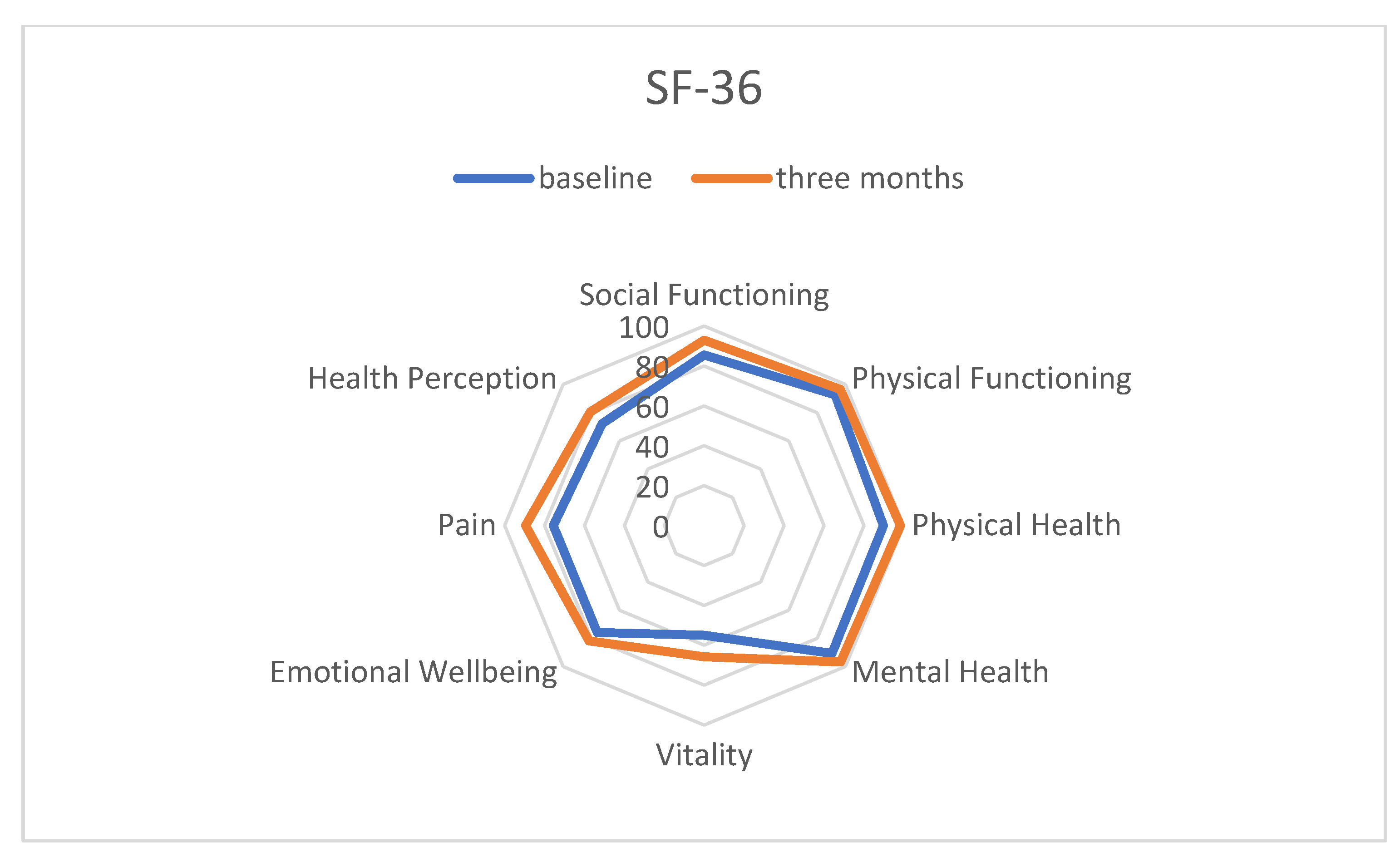
2.3. Short Form Health 36 (SF-36)
Fatigue
2.4. Fatigue Severity Scale (FSS)
2.5. Fatigue Assessment Scale (FAS)
2.5.1. Laboratory Changes
2.5.2. Adherence and Practicability
2.5.3. Statistical Analysis
3. Results
3.1. Quality of Life
3.2. Fatigue
3.3. Safety Profile and Metabolic Changes
3.4. Adherence and Practicability
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Patterson, R.E.; Sears, D.D. Metabolic effects of intermittent fasting. Annu. Rev. Nutr. 2017, 37, 371–393. [Google Scholar] [CrossRef] [PubMed]
- Trepanowski, J.F.; Bloomer, R.J. The impact of religious fasting on human health. Nutr. J. 2010, 9, 57. [Google Scholar] [CrossRef] [PubMed]
- Carlson, A.J.; Hoelzel, F. Apparent Prolongation of the Life Span of Rats by Intermittent Fasting: One Figure. J. Nutr. 1946, 31, 363–375. [Google Scholar] [CrossRef] [PubMed]
- De Toledo, F.W.; Grundler, F.; Sirtori, C.R.; Ruscica, M. Unravelling the health effects of fasting: A long road from obesity treatment to healthy life span increase and improved cognition. Ann. Med. 2020, 52, 147–161. [Google Scholar] [CrossRef]
- Moon, S.; Kang, J.; Kim, S.H.; Chung, H.S.; Kim, Y.J.; Yu, J.M.; Cho, S.T.; Oh, C.-M.; Kim, T. Beneficial effects of time-restricted eating on metabolic diseases: A systemic review and meta-analysis. Nutrients 2020, 12, 1267. [Google Scholar] [CrossRef]
- Rynders, C.A.; Thomas, E.A.; Zaman, A.; Pan, Z.; Catenacci, V.A.; Melanson, E.L. Effectiveness of Intermittent Fasting and Time-Restricted Feeding Compared to Continuous Energy Restriction for Weight Loss. Nutrients 2019, 11, 2442. [Google Scholar] [CrossRef] [PubMed]
- Swindell, W.R. Dietary restriction in rats and mice: A meta-analysis and review of the evidence for genotype-dependent effects on lifespan. Ageing Res. Rev. 2012, 11, 254–270. [Google Scholar] [CrossRef]
- Rassow, J.E.A. Biochemie, 3rd ed.; Duale Reihe; Thieme: New York, NY, USA, 2012. [Google Scholar]
- Horn, F. Biochemie des Menschen, 5th ed.; Thieme: New York, NY, USA, 2012. [Google Scholar]
- De Cabo, R.; Mattson, M.P. Effects of Intermittent Fasting on Health, Aging, and Disease. N. Engl. J. Med. 2019, 381, 2541–2551. [Google Scholar] [CrossRef]
- Sutton, E.F.; Beyl, R.; Early, K.S.; Cefalu, W.T.; Ravussin, E.; Peterson, C.M. Early Time-Restricted Feeding Improves Insulin Sensitivity, Blood Pressure, and Oxidative Stress Even without Weight Loss in Men with Prediabetes. Cell Metab. 2018, 27, 1212–1221.e3. [Google Scholar] [CrossRef]
- Furmli, S.; Elmasry, R.; Ramos, M.; Fung, J. Therapeutic use of intermittent fasting for people with type 2 diabetes as an alternative to insulin. BMJ. Case Rep. 2018, 2018, bcr-2017. [Google Scholar] [CrossRef]
- McNally, M.A.; Hartman, A.L. Ketone bodies in epilepsy. J. Neurochem. 2012, 121, 28–35. [Google Scholar] [CrossRef] [PubMed]
- Stafstrom, C.E.; Rho, J.M. The ketogenic diet as a treatment paradigm for diverse neurological disorders. Front. Pharmacol. 2012, 3, 59. [Google Scholar] [CrossRef] [PubMed]
- Henderson, C.G.; Turner, D.L.; Swoap, S.J. Health Effects of Alternate Day Fasting Versus Pair-Fed Caloric Restriction in Diet-Induced Obese C57Bl/6J Male Mice. Front. Physiol. 2021, 12, 641532. [Google Scholar] [CrossRef]
- Carvalho, L.F.; Samadder, A.N.; Agarwal, A.; Fernandes, L.F.; Abrao, M.S. Oxidative stress biomarkers in patients with endometriosis: Systematic review. Arch. Gynecol. Obstet. 2012, 286, 1033–1040. [Google Scholar] [CrossRef]
- Barbieri, R.L. Etiology and epidemiology of endometriosis. Am. J. Obstet. Gynecol. 1990, 162, 565–567. [Google Scholar] [CrossRef]
- Alimi, Y.; Iwanaga, J.; Loukas, M.; Tubbs, R.S. The Clinical Anatomy of Endometriosis: A Review. Cureus 2018, 10, e3361. [Google Scholar] [CrossRef] [PubMed]
- Ebert, A.D. Endometriose; De Gruyter: Berlin, Germany, 2019. [Google Scholar]
- Goerke, K.; Valet, A. 15-Uterus. In Klinikleitfaden Gynäkologie Geburtshilfe (Zehnte Ausgabe); Goerke, K., Steller, J., Valet, A., Eds.; Urban & Fischer: Munich, Germany, 2018; pp. 500–505. [Google Scholar]
- Laganà, A.S.; La Rosa, V.L.; Rapisarda, A.M.C.; Valenti, G.; Sapia, F.; Chiofalo, B.; Rossetti, D.; Frangez, H.B.; Bokal, E.V.; Vitale, S.G. Anxiety and depression in patients with endometriosis: Impact and management challenges. Int. J. Womens Health 2017, 9, 323–330. [Google Scholar] [CrossRef]
- Mattson, M.P.; Longo, V.D.; Harvie, M. Impact of intermittent fasting on health and disease processes. Ageing Res. Rev. 2017, 39, 46–58. [Google Scholar] [CrossRef]
- Dorff, T.B.; Groshen, S.; Garcia, A.; Shah, M.; Tsao-Wei, D.; Pham, H.; Cheng, C.-W.; Brandhorst, S.; Cohen, P.; Wei, M.; et al. Safety and feasibility of fasting in combination with platinum-based chemotherapy. BMC Cancer 2016, 16, 360. [Google Scholar] [CrossRef]
- Bauersfeld, S.P.; Kessler, C.S.; Wischnewsky, M.; Jaensch, A.; Steckhan, N.; Stange, R.; Kunz, B.; Brückner, B.; Sehouli, J.; Michalsen, A. The effects of short-term fasting on quality of life and tolerance to chemotherapy in patients with breast and ovarian cancer: A randomized cross-over pilot study. BMC Cancer 2018, 18, 476. [Google Scholar] [CrossRef]
- Zajac, I.; Herreen, D.; Hunkin, H.; James-Martin, G.; Doyen, M.; Kakoschke, N.; Brindal, E. Modified Fasting Compared to True Fasting Improves Blood Glucose Levels and Subjective Experiences of Hunger, Food Cravings and Mental Fatigue, But Not Cognitive Function: Results of an Acute Randomised Cross-Over Trial. Nutrients 2020, 13, 65. [Google Scholar] [CrossRef] [PubMed]
- Plotti, F.; Terranova, C.; Luvero, D.; Bartolone, M.; Messina, G.; Feole, L.; Cianci, S.; Scaletta, G.; Marchetti, C.; Di Donato, V.; et al. Diet and Chemotherapy: The Effects of Fasting and Ketogenic Diet on Cancer Treatment. Chemotherapy 2020, 65, 77–84. [Google Scholar] [CrossRef] [PubMed]
- De Groot, S.; Vreeswijk, M.P.G.; Welters, M.J.P.; Gravesteijn, G.; Boei, J.J.W.A.; Jochems, A.; Houtsma, D.; Putter, H.; Van Der Hoeven, J.J.M.; Nortier, J.W.R.; et al. The effects of short-term fasting on tolerance to (neo) adjuvant chemotherapy in HER2-negative breast cancer patients: A randomized pilot study. BMC Cancer 2015, 15, 652. [Google Scholar] [CrossRef] [PubMed]
- Stockman, M.-C.; Thomas, D.; Burke, J.; Apovian, C.M. Intermittent fasting: Is the wait worth the weight? Curr. Obes. Rep. 2018, 7, 172–185. [Google Scholar] [CrossRef] [PubMed]
- Bener, A.; Al-Hamaq, A.O.A.A.; Öztürk, M.; Çatan, F.; Haris, P.I.; Rajput, K.U.; Ömer, A. Effect of ramadan fasting on glycemic control and other essential variables in diabetic patients. Ann. Afr. Med. 2018, 17, 196–202. [Google Scholar] [CrossRef] [PubMed]
- Shatila, H.; Baroudi, M.; Ahmad, R.E.S.; Chehab, R.; Forman, M.R.; Abbas, N.; Faris, M.; Naja, F. Impact of Ramadan fasting on dietary intakes among healthy adults: A year-round comparative study. Front. Nutr. 2021, 8, 689788. [Google Scholar] [CrossRef] [PubMed]
- Topp, C.W.; Østergaard, S.D.; Søndergaard, S.; Bech, P. The WHO-5 Well-Being Index: A systematic review of the literature. Psychother. Psychosom. 2015, 84, 167–176. [Google Scholar] [CrossRef]
- Hays, R.D.; Sherbourne, C.D.; Mazel, R.M. The rand 36-item health survey 1.0. Health Econ. 1993, 2, 217–227. [Google Scholar] [CrossRef]
- Drent, M.; Lower, E.E.; de Vries, J. Sarcoidosis-associated fatigue. Eur. Respir. J. 2012, 40, 255–263. [Google Scholar] [CrossRef]
- De Kleijn, W.P.; de Vries, J.; Wijnen, P.A.; Drent, M. Minimal (clinically) important differences for the Fatigue Assessment Scale in sarcoidosis. Respir. Med. 2011, 105, 1388–1395. [Google Scholar] [CrossRef]
- Fatigue Severity Scale (FSS). Springer Science+Business Media. Available online: https://link.springer.com/chapter/10.1007/978-1-4419-9893-4_35#citeas (accessed on 18 September 2022).
- Nugraha, B.; Riat, A.; Ghashang, S.K.; Eljurnazi, L.; Gutenbrunner, C. A Prospective Clinical Trial of Prolonged Fasting in Healthy Young Males and Females-Effect on Fatigue, Sleepiness, Mood and Body Composition. Nutrients 2020, 12, 2281. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Yang, Y.; Wang, Y.; Tang, H.; Zhang, F.; Zhang, Y.; Zhao, Y. Ketogenic diet for treatment of intractable epilepsy in adults: A meta-analysis of observational studies. Epilepsia Open 2018, 3, 9–17. [Google Scholar] [CrossRef] [PubMed]
- Chamari, K.; Briki, W.; Farooq, A.; Patrick, T.; Belfekih, T.; Herrera, C.P. Impact of Ramadan intermittent fasting on cognitive function in trained cyclists: A pilot study. Biol. Sport 2016, 33, 49. [Google Scholar] [CrossRef] [PubMed]
- Cherif, A.; Roelands, B.; Meeusen, R.; Chamari, K. Effects of intermittent fasting, caloric restriction, and Ramadan intermittent fasting on cognitive performance at rest and during exercise in adults. Sports Med. 2016, 46, 35–47. [Google Scholar] [CrossRef]
- Chaouachi, A.; Leiper, J.B.; Chtourou, H.; Aziz, A.R.; Chamari, K. The effects of Ramadan intermittent fasting on athletic performance: Recommendations for the maintenance of physical fitness. J. Sports Sci. 2012, 30, S53–S73. [Google Scholar] [CrossRef]
- Nugraha, B.; Ghashang, S.K.; Hamdan, I.; Gutenbrunner, C. Effect of Ramadan fasting on fatigue, mood, sleepiness, and health-related quality of life of healthy young men in summer time in Germany: A prospective controlled study. Appetite 2017, 111, 38–45. [Google Scholar] [CrossRef]
- Bragazzi, N.L.; Briki, W.; Khabbache, H.; Rammouz, I.; Chamari, K.; Demaj, T.; Re, T.S.; Zouhir, M. Ramadan fasting and patients with cancer: State-of-the-art and future prospects. Front. Oncol. 2016, 6, 27. [Google Scholar] [CrossRef]
- Alkandari, J.R.; Maughan, R.J.; Roky, R.; Aziz, A.R.; Karli, U. The implications of Ramadan fasting for human health and well-being. J. Sports Sci. 2012, 30, S9–S19. [Google Scholar] [CrossRef]
- Teong, X.T.; Hutchison, A.T.; Liu, B.; Wittert, G.A.; Lange, K.; Banks, S.; Heilbronn, L.K. Eight weeks of intermittent fasting versus calorie restriction does not alter eating behaviors, mood, sleep quality, quality of life and cognitive performance in women with overweight. Nutr. Res. 2021, 92, 32–39. [Google Scholar] [CrossRef]
- Solianik, R.; Sujeta, A. Two-day fasting evokes stress, but does not affect mood, brain activity, cognitive, psychomotor, and motor performance in overweight women. Behav. Brain Res. 2018, 338, 166–172. [Google Scholar] [CrossRef]
- Kesztyüs, D.; Fuchs, M.; Cermak, P.; Kesztyüs, T. Associations of time-restricted eating with health-related quality of life and sleep in adults: A secondary analysis of two pre-post pilot studies. BMC Nutr. 2020, 6, 76. [Google Scholar] [CrossRef] [PubMed]
- Anton, S.; Ezzati, A.; Witt, D.; McLaren, C.; Vial, P. The effects of intermittent fasting regimens in middle-age and older adults: Current state of evidence. Exp. Gerontol. 2021, 156, 111617. [Google Scholar] [CrossRef] [PubMed]
- Tinsley, G.M.; la Bounty, P.M. Effects of intermittent fasting on body composition and clinical health markers in humans. Nutr. Rev. 2015, 73, 661–674. [Google Scholar] [CrossRef] [PubMed]
- Jensen, N.J.; Wodschow, H.Z.; Nilsson, M.; Rungby, J. Effects of Ketone Bodies on Brain Metabolism and Function in Neurodegenerative Diseases. Int. J. Mol. Sci. 2020, 21, 8767. [Google Scholar] [CrossRef]
- Stekovic, S.; Hofer, S.J.; Tripolt, N.; Aon, M.A.; Royer, P.; Pein, L.; Stadler, J.T.; Pendl, T.; Prietl, B.; Url, J.; et al. Alternate Day Fasting Improves Physiological and Molecular Markers of Aging in Healthy, Non-obese Humans. Cell Metab. 2020, 31, 878–881. [Google Scholar] [CrossRef]
- Norgren, J.; Sindi, S.; Sandebring-Matton, A.; Kåreholt, I.; Daniilidou, M.; Akenine, U.; Nordin, K.; Rosenborg, S.; Ngandu, T.; Kivipelto, M. Ketosis After Intake of Coconut Oil and Caprylic Acid-With and Without Glucose: A Cross-Over Study in Healthy Older Adults. Front. Nutr. 2020, 7, 40. [Google Scholar] [CrossRef]
- Shen, J.; Botly, L.C.; Chung, S.A.; Gibbs, A.L.; Sabanadzovic, S.; Shapiro, C.M. Fatigue and shift work. J. Sleep Res. 2006, 15, 1–5. [Google Scholar] [CrossRef]
- Anvekar, A.P.; Nathan, E.A.; Doherty, D.A.; Patole, S.K. Effect of shift work on fatigue and sleep in neonatal registrars. PLoS ONE 2021, 16, e0245428. [Google Scholar] [CrossRef]
- Books, C.; Coody, L.C.; Kauffman, R.; Abraham, S. Night shift work and its health effects on nurses. Health Care Manag. 2017, 36, 347–353. [Google Scholar] [CrossRef]
- Thong, M.S.; van Noorden, C.J.; Steindorf, K.; Arndt, V. Cancer-related fatigue: Causes and current treatment options. Curr. Treat. Options Oncol. 2020, 21, 17. [Google Scholar] [CrossRef]
- Fox, R.S.; Ancoli-Israel, S.; Roesch, S.C.; Merz, E.L.; Mills, S.D.; Wells, K.J.; Sadler, G.R.; Malcarne, V.L. Sleep disturbance and cancer-related fatigue symptom cluster in breast cancer patients undergoing chemotherapy. Supportive Care Cancer 2020, 28, 845–855. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.; Safdie, F.M.; Raffaghello, L.; Wei, M.; Madia, F.; Parrella, E.; Hwang, D.; Cohen, P.; Bianchi, G.; Longo, V.D. Reduced levels of IGF-I mediate differential protection of normal and cancer cells in response to fasting and improve chemotherapeutic index. Cancer Res. 2010, 70, 1564–1572. [Google Scholar] [CrossRef] [PubMed]
- Carrillo-García, C.; Solano-Ruíz, M.d.C.; Martínez-Roche, M.E.; Gómez-García, C.I. Job satisfaction among health care workers: The role of gender and age. Rev. Latino-Am. Enferm. 2013, 21, 1314–1320. [Google Scholar] [CrossRef] [PubMed]


| Inclusion Criteria | ≥18 Years of Age |
|---|---|
| Written informed consent | |
| Ability to understand nature and effects of the trial participation | |
| Exclusion Criteria | Body mass index <18 kg/m2 |
| Weight loss of >10% of body weight in the last 6 months | |
| Current or recent diet aimed at reducing weight | |
| Current or past eating disorders | |
| Acute medical conditions (including ulcus/gastritis) | |
| Chronic or acute heart, liver, or kidney disease | |
| Diabetes mellitus type I | |
| Large surgical procedures in the last 6 months | |
| Pregnancy and breastfeeding period | |
| Participation in another interventional trial likely to interfere with the study outcome |
| Frequency (n) Percentage (%) | Frequency (n) Percentage (%) | ||
|---|---|---|---|
| Gender Female Male | 27 (90.0%) 3 (10.0%) | Eating habits None in particular Vegetarian Vegan Organic products only | 21 (70.0%) 5 (16.7%) 3 (10.0%) 1 (3.3%) |
| Age [years] <20 20–29 30–39 40–49 50–59 | 1 (3.3%) 17 (56.7%) 4 (13.3%) 4 (13.3%) 4 (13.3%) | Meal preparation Rarely self-prepared Often homemade Mostly home cooked Always self-prepared | 0 (0.0%) 10 (33.3%) 20 (66.7%) 0 (0.0%) |
Working hours [hours/week] <40 40–50 >50 | 14 (46.7%) 13 (43.3%) 3 (10.0%) | Consumption of ready-made products Never Rarely Often | 9 (30.0%) 20 (66.7%) 1 (3.3%) |
| Movement at work Predominantly sedentary Relatively plenty of exercise | 17 (56.7%) 13 (43.3%) | Physical activity [hours/week] <0.5 0.5 1–2 >2 | 6 (20.7%) 2 (6.9%) 6 (20.7%) 15 (51.7%) |
| Nutritional awareness Yes No | 17 (60.7%) 11 (39.3%) | Weight fluctuations yes no | 4 (13.3%) 26 (86.7%) |
| Diet Rather one-sided Varies Balanced | 1 (3.3%) 16 (53.3%) 13 (43.3%) | First diet yes no | 11 (40.7%) 16 (59.3%) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Anic, K.; Schmidt, M.W.; Furtado, L.; Weidenbach, L.; Battista, M.J.; Schmidt, M.; Schwab, R.; Brenner, W.; Ruckes, C.; Lotz, J.; et al. Intermittent Fasting—Short- and Long-Term Quality of Life, Fatigue, and Safety in Healthy Volunteers: A Prospective, Clinical Trial. Nutrients 2022, 14, 4216. https://doi.org/10.3390/nu14194216
Anic K, Schmidt MW, Furtado L, Weidenbach L, Battista MJ, Schmidt M, Schwab R, Brenner W, Ruckes C, Lotz J, et al. Intermittent Fasting—Short- and Long-Term Quality of Life, Fatigue, and Safety in Healthy Volunteers: A Prospective, Clinical Trial. Nutrients. 2022; 14(19):4216. https://doi.org/10.3390/nu14194216
Chicago/Turabian StyleAnic, Katharina, Mona W. Schmidt, Larissa Furtado, Lina Weidenbach, Marco J. Battista, Marcus Schmidt, Roxana Schwab, Walburgis Brenner, Christian Ruckes, Johannes Lotz, and et al. 2022. "Intermittent Fasting—Short- and Long-Term Quality of Life, Fatigue, and Safety in Healthy Volunteers: A Prospective, Clinical Trial" Nutrients 14, no. 19: 4216. https://doi.org/10.3390/nu14194216
APA StyleAnic, K., Schmidt, M. W., Furtado, L., Weidenbach, L., Battista, M. J., Schmidt, M., Schwab, R., Brenner, W., Ruckes, C., Lotz, J., Lackner, K. J., Hasenburg, A., & Hasenburg, A. (2022). Intermittent Fasting—Short- and Long-Term Quality of Life, Fatigue, and Safety in Healthy Volunteers: A Prospective, Clinical Trial. Nutrients, 14(19), 4216. https://doi.org/10.3390/nu14194216







