Moderate Consumption of Beer (with and without Ethanol) and Menopausal Symptoms: Results from a Parallel Clinical Trial in Postmenopausal Women
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Population and Recruitment
2.2. Study Design
2.3. Intervention-Phytoestrogen Dose
Quantification of Prenylflavonoids in Beer Intervention Samples by LC-MS/MS
2.4. Measurements and Outcome Assessment
2.4.1. Medical History
2.4.2. Climacteric Symptoms
2.4.3. Anthropometric Measurements
2.4.4. Biological Samples and Biochemical Analyses
2.4.5. Dietary Intake and Physical Activity Assessments
2.5. Statistical Analyses
3. Results
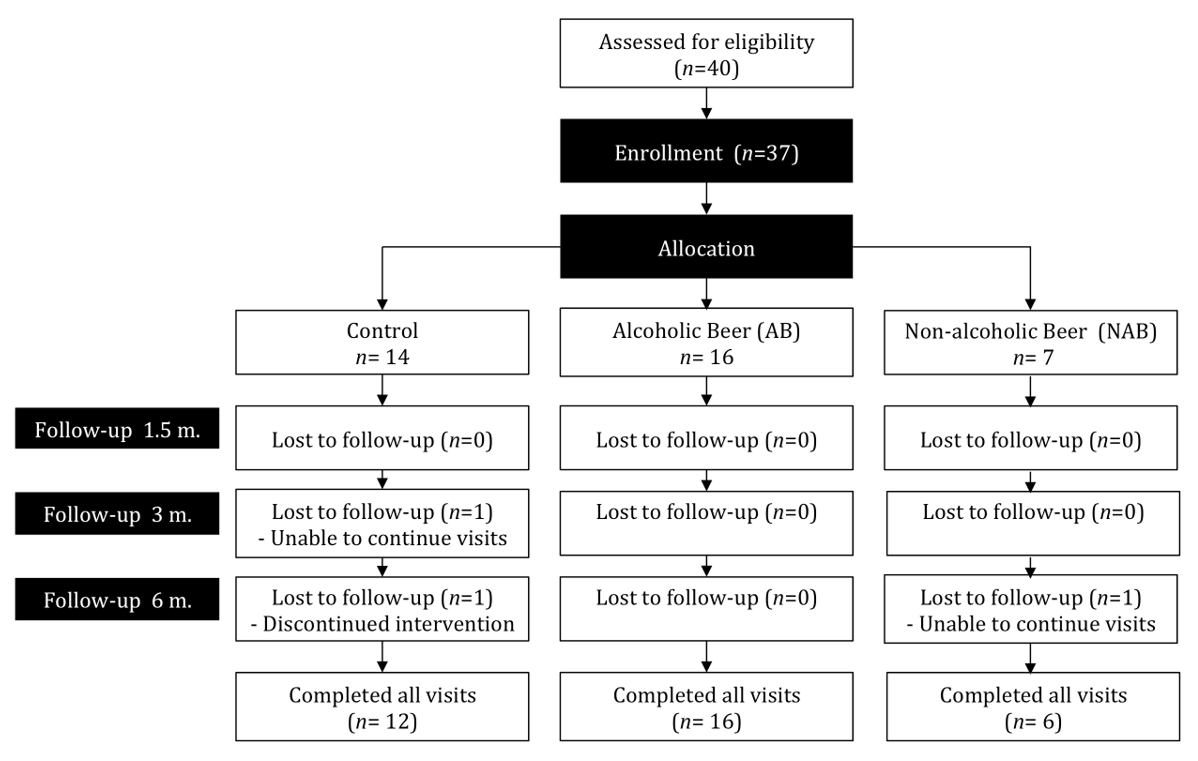
3.1. Study Subjects, Intervention, and Compliance
3.2. Participant Characteristics at Baseline
Covariates
3.3. Intervention Effects on Climacteric Symptoms
3.4. Intervention Effects on Sex Hormone Profile
3.5. Intervention Effects on CVRF
4. Discussion
4.1. Climacteric Symptoms
4.2. Sex Hormone Profile
4.3. Cardiovascular Risk Factors
4.3.1. Body Weight and Fat
4.3.2. Blood Pressure
4.3.3. Lipid Profile
4.3.4. Hepatic Profile
4.4. Strengths and Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Schneider, H.P.G. The quality of life in the post-menopausal woman. Best Pract. Res. Clin. Obstet. Gynaecol. 2002, 16, 395–409. [Google Scholar] [CrossRef] [PubMed]
- Jaspers, L.; Daan, N.M.P.; Van Dijk, G.M.; Gazibara, T.; Muka, T.; Wen, K.; Meun, C.; Zillikens, M.C.; Roeters, J.E.; Lennep, V.; et al. Maturitas Health in middle-aged and elderly women: A conceptual framework for healthy menopause. Maturitas 2015, 81, 93–98. [Google Scholar] [CrossRef]
- Kling, J.M.; Dowling, N.M.; Bimonte-Nelson, H.A.; Gleason, C.E.; Kantarci, K.; Manson, J.E.; Taylor, H.S.; Brinton, E.A.; Lobo, R.A.; Cedars, M.I.; et al. Impact of menopausal hormone formulations on pituitary-ovarian regulatory feedback. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2019, 317, R912–R920. [Google Scholar] [CrossRef] [PubMed]
- Van Gemert, W.A.; Monninkhof, E.M.; May, A.M.; Elias, S.G.; Van Der Palen, J.; Veldhuis, W.; Stapper, M.; Stellato, R.K.; Schuit, J.A.; Peeters, P.H. Association between changes in fat distribution and biomarkers for breast cancer. Endocr. Relat. Cancer 2017, 24, 297–305. [Google Scholar] [CrossRef]
- Rossouw, J.E.; Anderson, G.L.; Prentice, R.L.; LaCroix, A.Z.; Kooperberg, C.; Stefanick, M.L.; Jackson, R.D.; Beresford, S.A.A.; Howard, B.V.; Johnson, K.C.; et al. Risks and benefits of estrogen plus progestin in healthy postmenopausal women. principal results from the women’s health initiative randomized controlled trial. ACC Curr. J. Rev. 2002, 288, 321–333. [Google Scholar] [CrossRef]
- Manson, J.A.E.; Aragaki, A.K.; Rossouw, J.E.; Anderson, G.L.; Prentice, R.L.; LaCroix, A.Z.; Chlebowski, R.T.; Howard, B.V.; Thomson, C.A.; Margolis, K.L.; et al. Menopausal hormone therapy and long-term all-cause and cause-specific mortality: The Women’s Health Initiative randomized trials. JAMA J. Am. Med. Assoc. 2017, 318, 927–938. [Google Scholar] [CrossRef] [PubMed]
- Thaung Zaw, J.J.; Howe, P.R.C.; Wong, R.H.X. Postmenopausal health interventions: Time to move on from the Women’s Health Initiative? Ageing Res. Rev. 2018, 48, 79–86. [Google Scholar] [CrossRef]
- Tempfer, C.B.; Bentz, E.K.; Leodolter, S.; Tscherne, G.; Reuss, F.; Cross, H.S.; Huber, J.C. Phytoestrogens in clinical practice: A review of the literature. Fertil. Steril. 2007, 87, 1243–1249. [Google Scholar] [CrossRef]
- Clarkson, T.B.; Utian, W.H.; Barnes, S.; Gold, E.B.; Basaria, S.S.; Aso, T.; Kronenberg, F.; Frankenfeld, C.L.; Cline, J.M.A.; Landgren, B.M.; et al. The role of soy isoflavones in menopausal health: Report of the North American Menopause Society/Wulf, H. Utian Translational Science Symposium in Chicago, IL (October 2010). Menopause 2011, 18, 732–753. [Google Scholar] [CrossRef]
- Chen, M.N.; Lin, C.C.; Liu, C.F. Efficacy of phytoestrogens for menopausal symptoms: A meta-analysis and systematic review. Climacteric 2015, 18, 260–269. [Google Scholar] [CrossRef]
- Su, B.Y.W.; Tung, T.H.; Chien, W.H. Effects of phytoestrogens on depressive symptoms in climacteric women: A meta-analysis of randomized controlled trials. J. Altern. Complement. Med. 2018, 24, 850–851. [Google Scholar] [CrossRef]
- Tronina, T.; Popłonski, J.; Bartmanska, A. Flavonoids as Phytoestrogenic Components of Hops and Beer. Molecules 2020, 25, 4201. [Google Scholar] [CrossRef]
- Erkkola, R.; Vervarcke, S.; Vansteelandt, S.; Rompotti, P.; De Keukeleire, D.; Heyerick, A. A randomized, double-blind, placebo-controlled, cross-over pilot study on the use of a standardized hop extract to alleviate menopausal discomforts. Phytomedicine 2010, 17, 389–396. [Google Scholar] [CrossRef]
- Heyerick, A.; Vervarcke, S.; Depypere, H.; Bracke, M.; Keukeleire, D. De A first prospective, randomized, double-blind, placebo-controlled study on the use of a standardized hop extract to alleviate menopausal discomforts. Maturitas 2006, 54, 164–175. [Google Scholar] [CrossRef]
- Chen, X.; Mukwaya, E.; Wong, M.S.; Zhang, Y. A systematic review on biological activities of prenylated flavonoids. Pharm. Biol. 2014, 52, 655–660. [Google Scholar] [CrossRef]
- Bolca, S.; Possemiers, S.; Maervoet, V.; Huybrechts, I.; Heyerick, A.; Vervarcke, S.; Depypere, H.; De Keukeleire, D.; Bracke, M.; De Henauw, S.; et al. Microbial and dietary factors associated with the 8-prenylnaringenin producer phenotype: A dietary intervention trial with fifty healthy post-menopausal Caucasian women. Br. J. Nutr. 2007, 98, 950–959. [Google Scholar] [CrossRef]
- Possemiers, S.; Bolca, S.; Grootaert, C.; Heyerick, A.; Decroos, K.; Dhooge, W.; De Keukeleire, D.; Rabot, S.; Verstraete, W.; Van De Wiele, T. The prenylflavonoid isoxanthohumol from hops (Humulus lupulus L.) is activated into the potent phytoestrogen 8-prenylnaringenin in vitro and in the human intestine. J. Nutr. 2006, 136, 1862–1867. [Google Scholar] [CrossRef]
- Aghamiri, V.; Mirghafourvand, M.; Mohammad-Alizadeh-Charandabi, S.; Nazemiyeh, H. The effect of Hop (Humulus lupulus L.) on early menopausal symptoms and hot flashes: A randomized placebo-controlled trial. Complement. Ther. Clin. Pract. 2016, 23, 130–135. [Google Scholar] [CrossRef]
- Quifer-Rada, P.; Vallverdú-Queralt, A.; Martínez-Huélamo, M.; Chiva-Blanch, G.; Jáuregui, O.; Estruch, R.; Lamuela-Raventós, R. A comprehensive characterisation of beer polyphenols by high resolution mass spectrometry (LC–ESI-LTQ-Orbitrap-MS). Food Chem. 2015, 169, 336–343. [Google Scholar] [CrossRef]
- Hajirahimkhan, A.; Dietz, B.; Bolton, J. Botanical Modulation of Menopausal Symptoms: Mechanisms of Action? Planta Med. 2013, 79, 538–553. [Google Scholar] [CrossRef]
- Omoruyi, I.M.; Pohjanvirta, R. Estrogenic activities of food supplements and beers as assessed by a yeast bioreporter assay. J. Diet. Suppl. 2018, 15, 665–672. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Possemiers, S.; Heyerick, A.; Robbens, V.; De Keukeleire, D.; Verstraete, W. Activation of proestrogens from hops (Humulus lupulus L.) by intestinal microbiota; conversion of isoxanthohumol into 8-prenylnaringenin. J. Agric. Food Chem. 2005, 53, 6281–6288. [Google Scholar] [CrossRef] [PubMed]
- Guo, J.; Nikolic, D.; Chadwick, L.R.; Pauli, G.F.; Van Breemen, R.B. Identification of human hepatic cytochrome P450 enzymes involved in the metabolism of 8-prenylnaringenin and isoxanthohumol from hops (Humulus lupulus L.). Drug Metab. Dispos. 2006, 34, 1152–1159. [Google Scholar] [CrossRef] [PubMed]
- Nikolic, D.; Li, Y.; Chadwick, L.R.; Grubjesic, S.; Schwab, P.; Metz, P.; Van Breemen, R.B. Metabolism of 8-prenylnaringenin, a potent phytoestrogen from hops (Humulus lupulus), by human liver microsomes. Drug Metab. Dispos. 2004, 32, 272–279. [Google Scholar] [CrossRef]
- Quifer-Rada, P.; Martínez-Huélamo, M.; Chiva-Blanch, G.; Jáuregui, O.; Estruch, R.; Lamuela-Raventós, R.M. Urinary isoxanthohumol is a specific and accurate biomarker of beer consumption. J. Nutr. 2014, 144, 484–488. [Google Scholar] [CrossRef][Green Version]
- Quifer-Rada, P.; Martínez-Huélamo, M.; Jáuregui, O.; Chiva-Blanch, G.; Estruch, R.; Lamuela-Raventós, R.M. Analytical condition setting a crucial step in the quantification of unstable polyphenols in acidic conditions: Analyzing prenylflavanoids in biological samples by liquid chromatography-electrospray ionization triple quadruple mass spectrometry. Anal. Chem. 2013, 85, 5547–5554. [Google Scholar] [CrossRef]
- Heinemann, L.A.J.; Potthoff, P.; Schneider, H.P.G. International versions of the Menopause Rating Scale (MRS). Health Qual. Life Outcomes 2003, 1, 1–4. [Google Scholar] [CrossRef]
- Zöllner, Y.; Acquadro, C.; Schaefer, M. Literature review of instruments to assess health-related quality of life during and after menopause. Qual. Life Res. 2005, 14, 309–327. [Google Scholar] [CrossRef]
- World Health Organization. WHO STEPS Surveillance Manual: The WHO STEPwise Approach to Chronic Disease Risk Factor Surveillance; World Health Organization: Geneva, Switzerland, 2008; ISBN 9241593830. [Google Scholar]
- Schilling, C.; Gallicchio, L.; Miller, S.R.; Langenberg, P.; Zacur, H.; Flaws, J.A. Genetic polymorphisms, hormone levels, and hot flashes in midlife women. Maturitas 2007, 57, 120–131. [Google Scholar] [CrossRef][Green Version]
- Juton, C.; Castro-Barquero, S.; Casas, R.; Freitas, T.; Ruiz-León, A.M.; Crovetto, F.; Domenech, M.; Crispi, F.; Vieta, E.; Gratacós, E.; et al. Reliability and Concurrent and Construct Validity of a Food Frequency Questionnaire for Pregnant Women at High Risk to Develop Fetal Growth Restriction. Nutrients 2021, 13, 1629. [Google Scholar] [CrossRef]
- Rothwell, J.A.; Perez-Jimenez, J.; Neveu, V.; Medina-Remón, A.; M’Hiri, N.; García-Lobato, P.; Manach, C.; Knox, C.; Eisner, R.; Wishart, D.S.; et al. Phenol-Explorer 3.0: A major update of the Phenol-Explorer database to incorporate data on the effects of food processing on polyphenol content. Database 2013, 2013. [Google Scholar] [CrossRef]
- Schröder, H.; Fitó, M.; Estruch, R.; Martínez-González, M.A.; Corella, D.; Salas-Salvadó, J.; Lamuela-Raventós, R.; Ros, E.; Salaverría, I.; Fiol, M.; et al. A Short screener is valid for assessing mediterranean diet adherence among older spanish men and women. J. Nutr. 2011, 141, 1140–1145. [Google Scholar] [CrossRef]
- Elosua, R.; Garcia, M.; Aguilar, A.; Molina, L.; Covas, M.-I.; Marrugat, J. Validation of the Minnesota Leisure Time Spanish Women. Med. Sci. Sports Exerc. 2000, 32, 1431–1437. [Google Scholar] [CrossRef]
- Ballinger, G.A. Using Generalized Estimating Equations for Longitudinal Data Analysis. Organ. Res. Methods 2004, 7, 127–150. [Google Scholar] [CrossRef]
- Chan, Y.H. Biostatistics ICorrelational analysis. Singapore Med. J. 2003, 44, 614–619. [Google Scholar]
- European Food Safety Authority. Dietary reference values for nutrients summary report. EFSA J. 2017, 14. [Google Scholar] [CrossRef]
- Ziv-Gal, A.; Flaws, J.A. Factors That May Influence the Experience of Hot Flushes by Healthy Middle-Aged Women. J. Women’s Health 2010, 19, 1905–1914. [Google Scholar] [CrossRef]
- Schaefer, O.; Hümpel, M.; Fritzemeier, K.H.; Bohlmann, R.; Schleuning, W.D. 8-Prenyl naringenin is a potent ERα selective phytoestrogen present in hops and beer. J. Steroid Biochem. Mol. Biol. 2003, 84, 359–360. [Google Scholar] [CrossRef]
- Zołnierczyk, A.K.; Mączka, W.K.; Grabarczyk, M.; Wińska, K.; Woźniak, E.; Anioł, M. Isoxanthohumol–Biologically active hop flavonoid. Fitoterapia 2015, 103, 71–82. [Google Scholar] [CrossRef]
- Lethaby, A.; Marjoribanks, J.; Kronenberg, F.; Roberts, H.; Eden, J.; Brown, J. Phytoestrogens for menopausal vasomotor symptoms. Cochrane Database Syst. Rev. 2013, 2013. [Google Scholar] [CrossRef]
- Huber, R.; Gminski, R.; Tang, T.; Weinert, T.; Schulz, S.; Linke-Cordes, M.; Martin, I.; Fischer, H. Pomegranate (Punica granatum) seed oil for treating menopausal symptoms: An individually controlled cohort study. Altern. Ther. Health Med. 2017, 23, 28–34. [Google Scholar] [PubMed]
- Auerbach, L.; Rakus, J.; Bauer, C.; Gerner, C.; Ullmann, R.; Wimmer, H.; Huber, J. Pomegranate seed oil in women with menopausal symptoms: A prospective randomized, placebo-controlled, double-blinded trial. Menopause 2012, 19, 426–432. [Google Scholar] [CrossRef] [PubMed]
- Colli, M.C.; Bracht, A.; Soares, A.A.; De Oliveira, A.L.; Bôer, C.G.; De Souza, C.G.M.; Peralta, R.M. Evaluation of the efficacy of flaxseed meal and flaxseed extract in reducing menopausal symptoms. J. Med. Food 2012, 15, 840–845. [Google Scholar] [CrossRef] [PubMed]
- Dodin, S.; Lemay, A.; Jacques, H.; Légaré, F.; Forest, J.-C.; Mâsse, B. The Effects of Flaxseed Dietary Supplement on Lipid Profile, Bone Mineral Density, and Symptoms in Menopausal Women: A Randomized, Double-Blind, Wheat Germ Placebo-Controlled Clinical Trial. J. Clin. Endocrinol. Metab. 2005, 90, 1390–1397. [Google Scholar] [CrossRef]
- Ausmanas, M.K.; Tan, D.A.; Jaisamrarn, U.; Tian, X.W.; Holinka, C.F. Estradiol, FSH and LH profiles in nine ethnic groups of postmenopausal Asian women: The Pan-Asia Menopause (PAM) study. Climacteric 2007, 10, 427–437. [Google Scholar] [CrossRef]
- Soares, A.G.; Kilpi, F.; Fraser, A.; Nelson, S.M.; Sattar, N.; Welsh, P.I.; Tilling, K.; Lawlor, D.A. Longitudinal changes in reproductive hormones through the menopause transition in the Avon Longitudinal Study of Parents and Children (ALSPAC). Sci. Rep. 2020, 21258. [Google Scholar] [CrossRef]
- Rinaldi, S.; Peeters, P.H.M.; Bezemer, I.D.; Dossus, L.; Biessy, C.; Sacerdote, C.; Berrino, F.; Panico, S.; Palli, D.; Tumino, R.; et al. Relationship of alcohol intake and sex steroid concentrations in blood in pre- and post-menopausal women: The European Prospective Investigation into Cancer and Nutrition. Cancer Causes Control 2006, 17, 1033–1043. [Google Scholar] [CrossRef]
- Sierksma, A.; Sarkola, T.; Eriksson, C.J.P.; Van Der Gaag, M.S.; Grobbee, D.E.; Hendriks, H.F.J. Effect of moderate alcohol consumption on plasma dehydroepiandrosterone sulfate, testosterone, and estradiol levels in middle-aged men and postmenopausal women: A diet-controlled intervention study. Alcohol. Clin. Exp. Res. 2004, 28, 780–785. [Google Scholar] [CrossRef]
- Gavaler, J.S. Alcoholic beverages as a source of estrogens. Alcohol Res. Health 1998, 22, 220–227. [Google Scholar]
- Rad, M.; Hümpel, M.; Schaefer, O.; Schoemaker, R.C.; Schleuning, W.; Cohen, A.F.; Burggraaf, J. Pharmacokinetics and systemic endocrine effects of the phyto-oestrogen 8-prenylnaringenin after single oral doses to postmenopausal women. Br. J. Clin. Pharmacol. 2006. [Google Scholar] [CrossRef]
- Longnecker, M.P.; Tseng, M. Alcohol, hormones, and postmenopausal women. Alcohol Health Res. World 1998, 22, 185. [Google Scholar]
- Cerchiari, D.P.; de Moricz, R.D.; Sanjar, F.A.; Rapoport, P.B.; Moretti, G.; Guerra, M.M. Síndrome da boca ardente: Etiologia. Rev. Bras. Otorrinolaringol. 2006, 72, 419–424. [Google Scholar] [CrossRef]
- Khosla, S.; Melton, L.J.; Atkinson, E.J.; Fallon, W.M.O.; Klee, G.G.; Riggs, B.L. Relationship of Serum Sex Steroid Levels and Bone Turnover Markers with Bone Mineral Density in Men and Women: A Key Role for Bioavailable Estrogen. J. Clin. Endocrinol. Metab. 1998, 83, 2266–2274. [Google Scholar]
- Baer, D.J.; Judd, J.T.; Clevidence, B.A.; Muesing, R.A.; Campbell, W.S.; Brown, E.D.; Taylor, P.R. Moderate alcohol consumption lowers risk factors for cardiovascular disease in postmenopausal women fed a controlled diet. Am. J. Clin. Nutr. 2002, 75, 593–599. [Google Scholar] [CrossRef]
- Trius-Soler, M.; Vilas-Franquesa, A.; Tresserra-Rimbau, A.; Sasot, G.; Storniolo, C.E.; Estruch, R.; Lamuela-Raventós, R.M. Effects of the Non-Alcoholic Fraction of Beer on Abdominal Fat, Osteoporosis, and Body Hydration in Women. Molecules 2020, 25, 3910. [Google Scholar] [CrossRef]
- Chiva-Blanch, G.; Magraner, E.; Condines, X.; Valderas-Martínez, P.; Roth, I.; Arranz, S.; Casas, R.; Navarro, M.; Hervas, A.; Sisó, A.; et al. Effects of alcohol and polyphenols from beer on atherosclerotic biomarkers in high cardiovascular risk men: A randomized feeding trial. Nutr. Metab. Cardiovasc. Dis. 2015, 25, 36–45. [Google Scholar] [CrossRef]
- Husain, D.; Khanna, K.; Puri, S.; Haghighizadeh, M. Supplementation of soy isoflavones improved sex hormones, blood pressure, and postmenopausal symptoms. J. Am. Coll. Nutr. 2015, 34, 42–48. [Google Scholar] [CrossRef]
- Hooper, L.; Kroon, P.A.; Rimm, E.B.; Cohn, J.S.; Harvey, I.; Le Cornu, K.A.; Ryder, J.J.; Hall, W.L.; Cassidy, A. Flavonoids, flavonoid-rich foods, and cardiovascular risk: A meta-analysis of randomized controlled trials. Am. J. Clin. Nutr. 2008, 88, 38–50. [Google Scholar] [CrossRef]
- Lambert, M.N.T.; Thorup, A.C.; Hansen, E.S.S.; Jeppesen, P.B. Combined Red Clover isoflavones and probiotics potently reduce menopausal vasomotor symptoms. PLoS ONE 2017, 12, e0176590. [Google Scholar] [CrossRef]
- Borrelli, F.; Ernst, E. Alternative and complementary therapies for the menopause. Maturitas 2010, 66, 333–343. [Google Scholar] [CrossRef]
- Spaggiari, G.; Cignarelli, A.; Sansone, A.; Baldi, M.; Santi, D. To beer or not to beer: A meta-analysis of the effects of beer consumption on cardiovascular health. PLoS ONE 2020, 15, e0233619. [Google Scholar] [CrossRef]
- Sierksma, A.; Van Der Gaag, M.S.; Van Tol, A.; James, R.W.; Hendriks, H.F.J. Kinetics of HDL cholesterol and paraoxonase activity in moderate alcohol consumers. Alcohol. Clin. Exp. Res. 2002, 26, 1430–1435. [Google Scholar] [CrossRef]
- Xu, C.; Liu, Q.; Zhang, Q.; Jiang, Z.Y.; Gu, A. Urinary enterolactone associated with liver enzyme levels in US adults: National Health and Nutrition Examination Survey (NHANES). Br. J. Nutr. 2015, 114, 91–97. [Google Scholar] [CrossRef]
- Furtwængler, N.A.F.F.; Visser, R.O.D.E. Lack of international consensus in low-risk drinking guidelines. Drug Alcohol Rev. 2013, 32, 11–18. [Google Scholar] [CrossRef]
| Intervention Group | IX μg/Day | XN μg/Day | 8-PN μg/Day | 6-PN μg/Day | Total Amount μg/Day | Alcohol g/Day |
|---|---|---|---|---|---|---|
| AB | 302.7 ± 16.8 | 27.9 ± 0.6 | 5.5 ± 0.4 | 22.8 ± 0.3 | 358.9 ± 17.4 | 14 |
| NAB | 104.7 ± 3.8 | 81.3 ± 4.0 | 10.3 ± 0.8 | 62.7 ± 2.2 | 259.0 ± 10.3 | 0.0 |
| Control (n = 14) | AB (n = 16) | NAB (n = 7) | p-Value | |
|---|---|---|---|---|
| Weight, kg | 71.7 ± 13.0 | 64.7 ± 10.3 | 75.2 ± 20.3 | 0.324 |
| BMI, kg/m2 | 27.2 ± 4.4 | 25.3 ± 3.7 | 30.0 ± 9.0 | 0.634 |
| WC, cm | 89.4 ± 9.7 | 87.0 ± 10.3 | 90.6 ± 16.8 | 0.810 |
| DBP, mmHg | 74 ± 13 | 73 ± 6 | 74 ± 6 | 0.944 |
| SBP, mmHg | 121 ± 15 | 120 ± 14 | 120 ± 16 | 0.929 |
| Heart rate, bpm | 70 ± 12 | 68 ± 11 | 71 ± 7 | 0.657 |
| Glucose, mg/dL | 90.9 ± 6.2 | 93.9 ± 7.7 | 97.1 ± 11.5 | 0.376 |
| Creatinine, mg/dL | 0.69 ± 0.12 | 0.64 ± 0.10 | 0.70 ± 0.11 | 0.483 |
| Uric acid, mg/dL | 4.9 ± 0.7 | 4.6 ± 1.3 | 5.3 ± 1.7 | 0.322 |
| Total cholesterol, mg/dL | 185 ± 30 | 206 ± 22 | 208 ± 27 | 0.060 |
| LDL-C, mg/dL | 114 ± 23 | 135 ± 25 | 142 ± 18 | 0.063 |
| HDL-C, mg/dL | 56 ± 13 | 57 ± 8 | 56 ± 13 | 0.895 |
| Triglycerides, mg/dL | 69 ± 32 | 77 ± 25 | 66 ± 18 | 0.663 |
| ApoA1, mg/dL | 150 ± 19 | 163 ± 14 | 158 ± 22 | 0.058 |
| ApoB mg/dL | 94 ± 17 | 106 ± 20 | 105 ± 15 | 0.188 |
| AST, U/L | 21 ± 12 a | 26 ± 10 b | 22 ± 5 ab | 0.025 |
| ALT, U/L | 19 ± 10 | 24 ± 14 | 18 ± 5 | 0.217 |
| GGT, U/L | 14 ± 5 | 22 ± 10 | 20 ± 15 | 0.057 |
| Total proteins, g/L | 70 ± 3 | 69 ± 5 | 71 ± 4 | 0.541 |
| Albumin, g/L | 42 ± 2 | 43 ± 3 | 42 ± 2 | 0.530 |
| TSH, ng/mL | 2.11 ± 1.50 | 2.63 ± 2.23 | 2.58 ± 0.50 | 0.290 |
| FT4, ng/mL | 1.16 ± 0.14 | 1.15 ± 0.16 | 1.07 ±0.07 | 0.318 |
| T3, ng/mL | 1.16 ± 0.21 | 1.15 ± 0.15 | 1.24 ± 0.40 | 0.961 |
| Cortisol, μg/dL | 13.3 ± 4.1 | 13.7 ± 5.1 | 14.2 ± 5.5 | 0.900 |
| PTH, ng/mL | 65.3 ± 24.2 | 61.6 ± 21.9 | 66.0 ± 17.8 | 0.653 |
| 25-hydroxy-vitamin D3, ng/mL | 23.2 ± 4.8 | 27.4 ± 10.5 | 24.8 ± 13.5 | 0.656 |
| FSH, U/L | 66.7 ± 21.5 a | 103.0 ± 44.4 b | 55.8 ± 22.5 a | 0.006 |
| E2, pg/mL | 24.1 ± 12.8 | 19.9 ± 8.1 | 22.5 ± 7.7 | 0.651 |
| Control (n = 14) | AB (n = 16) | NAB (n = 7) | p-Value | |
|---|---|---|---|---|
| Medical records | ||||
| Age, years | 55.6 ± 5.1 | 54.9 ± 3.6 | 56.4 ± 3.2 | 0.647 |
| Smoking habit | ||||
| Current, n (%) | 1 (7.1) | 6 (37.5) | 2 (28.6) | 0.170 |
| Former, n (%) | 3 (21.4) | 4 (25.0) | 0 (0.0) | |
| Never, n (%) | 10 (71.4) | 6 (37.5) | 5 (71.4) | |
| Sleeping time, hours | 6.4 ± 0.9 | 7.0 ± 1.3 | 6.8 ± 1.0 | 0.448 |
| Stress/depression from daily life1 | 2.6 ± 1.4ab | 3.4 ± 1.0a | 1.7± 1.1b | 0.013 |
| Stress/depression from work1 | 2.9 ± 1.5 | 2.7 ± 1.6 | 2.6 ± 1.3 | 0.881 |
| Time since the onset of menopause, months | 52.1 ± 35.5 | 36.7 ± 28.0 | 46.0 ± 55.5 | 0.432 |
| Physical activity, MET-min/day | 731 ± 449 | 681 ± 616 | 467 ± 118 | 0.587 |
| Dietary history | ||||
| Total energy intake, kcal/day | 2695 ± 517 | 2726 ± 673 | 2352 ± 264 | 0.189 |
| MedDiet, 14-item score | 8.7 ± 1.8 | 7.4 ± 1.6 | 7.9 ± 2.4 | 0.170 |
| Legumes, g/day | 62 ± 31 | 53 ± 23 | 43 ± 24 | 0.586 |
| Seeds, g/day | 5.0 ± 8.4 | 0.9 ± 1.5 | 1.7 ± 1.9 | 0.208 |
| Whole grains, g/day | 51 ± 39 | 95 ± 83 | 72 ± 69 | 0.419 |
| Isoflavonoids, mg/day | 6.4 ± 13.2 | 2.3 ± 5.3 | 10.3 ± 17.6 | 0.079 |
| Alcohol drinking habit | ||||
| Weekly, n (%) | 1 (7.1) | 9 (56.3) | 1 (14.3) | 0.025 |
| Occasionally, n (%) | 10 (71.4) | 7 (43.8) | 5 (71.4) | |
| Never, n (%) | 3 (21.4) | 0 (0.00) | 1 (14.3) | |
| Type of alcohol preferred | ||||
| Beer, n (%) | 3 (21.4) | 8 (50.0) | 3 (42.9) | 0.482 |
| Wine, n (%) | 7 (50.0) | 7 (43.8) | 3 (42.9) | |
| Spirits, n (%) | 1 (7.1) | 1 (6.3) | 0 (0.0) | |
| None, n (%) | 3 (21.4) | 0 (0.0) | 1 (14.3) | |
| Medication | ||||
| Antihypertensive agents, n (%) | 1 (7.1) | 3 (18.7) | 1 (14.3) | 0.649 |
| Lipid-lowering medication, n (%) | 0 (0.0) | 2 (12.5) | 0 (0.0) | 0.250 |
| Antidepressants, sedatives, anxiety pills, n (%) | 3 (21.4) | 3 (18.8) | 0 (0.0) | 0.425 |
| Sleeping pills, n (%) | 2 (14.3) | 2 (12.5) | 0 (0.0) | 0.585 |
| Dietary supplements, n (%) | 3 (21.4) | 8 (50.0) | 4 (57.1) | 0.172 |
| AB vs. Control | NAB vs. Control | AB vs. NAB | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Difference Time-Exposure (95% IC) | p-Value | p-Trend | Difference Time-Exposure (95% IC) | p-Value | p-Trend | Difference Time-Exposure (95% IC) | p-Value | p-Trend | |
| Somatic Model 1 Model 2 Model 3 | −1.1 (−2.7, 0.6) −1.2 (−2.7, 0.3) −1.3 (−2.8, 0.2) | 0.199 0.120 0.088 | 0.223 0.126 0.083 | −1.0 (−3.2, 1.2) −1.5 (−3.7, 0.7) −1.7 (−4.0, 0.5) | 0.354 0.184 0.128 | 0.367 0.230 0.138 | −0.0 (−1.9, 1.0) 0.3 (−1.7, 2.2) 0.5 (−1.6, 2.5) | 0.988 0.779 0.665 | 0.993 0.890 0.737 |
| Psychological Model 1 Model 2 Model 3 | −2.1 (−3.8, −0.5) −2.1 (−3.5, −0.7) −2.1 (−3.5, −0.6) | 0.011 0.004 0.004 | 0.021 0.007 0.010 | −1.1 (−2.8, 0.6) −1.1 (−2.8, 0.7) −1.3 (−2.9, 0.3) | 0.196 0.236 0.120 | 0.254 0.321 0.157 | −1.0 (−2.9, 0.9) −1.0 (−3.1, 1.0) −0.8 (−2.6, 1.0) | 0.290 0.313 0.396 | 0.377 0.331 0.415 |
| Urogenital Model 1 Model 2 Model 3 | −0.4 (−1.3, 0.5) −0.4 (−1.3, 0.4) −0.5 (−1.4, 0.3) | 0.367 0.309 0.214 | 0.354 0.252 0.170 | −0.1 (−1.2, 1.0) −0.5 (−1.7, 0.7) −0.7 (−1.9, 9.5) | 0.861 0.397 0.236 | 0.898 0.459 0.288 | −0.3 (−1.3, 0.7) 0.1 (−1.1, 1.2) 0.2 (−1.0, 1.3) | 0.544 0.915 0.783 | 0.488 0.949 0.933 |
| Total MRS Model 1 Model 2 Model 3 | −3.5 (−6.8, −0.3) −3.8 (−6.8, −0.9) −3.9 (−6.9, −1.0) | 0.031 0.011 0.009 | 0.041 0.013 0.011 | −2.2 (−5.3, 0.9) −3.0 (−5.9, −0.1) −3.1 (−6.0, −0.2) | 0.160 0.040 0.033 | 0.191 0.073 0.062 | −1.3 (−4.2 1.6) −0.8 (−3.7, 2.2) −0.8 (−3.8, 2.2) | 0.376 0.602 0.601 | 0.354 0.465 0.456 |
| AB vs. Control | NAB vs. Control | AB vs. NAB | ||||
|---|---|---|---|---|---|---|
| Difference Time-Exposure (95% IC) | p-Value | Difference Time-Exposure (95% IC) | p-Value | Difference Time-Exposure (95% IC) | p-Value | |
| LH Model 1 Model 2 Model 3 | −5.02 (−9.30, −0.73) −4.77 (−9.03, −0.51) −4.74 (−8.92, −0.56) | 0.022 0.028 0.026 | −1.44 (−9.14, 6.27) −1.25 (−9.18, 6.68) −1.09 (−8.99, 6.80) | 0.714 0.758 0.786 | −3.55 (−11.26, 4.17) −3.52 (−11.35, 4.34) −3.71 (−11.68, 4.26) | 0.367 0.382 0.361 |
| FSH Model 1 Model 2 Model 3 | −7.70 (−17.23, 1.83) −7.20 (−16.85, 2.45) −6.25 (−16.26, 3.76) | 0.113 0.144 0.221 | 2.29 (−8.37, 12.95) 2.89 (−7.88, 13.66) 3.76 (−7.23, 14.76) | 0.674 0.599 0.502 | −9.99 (−18.80, −1.18) −10.09 (−19.01, −1.17) −10.01 (−14.76, −1.36) | 0.026 0.027 0.023 |
| E2 Model 1 Model 2 Model 3 | −2.61 (−17.88, 12.66) −2.32 (−17.64, 13.00) −2.22 (−17.40, 12.96) | 0.738 0.766 0.774 | −7.53 (−21.61, 6.55) −7.39 (−21.55, 6.76) −7.30 (−21.44, 6.84) | 0.295 0.306 0.312 | 4.92 (−6.64, 16.47) 5.07 (−6.47, 16.61) 5.08 (−6.45, 16.60) | 0.404 0.389 0.388 |
| Progesterone Model 1 Model 2 Model 3 | 0.03 (−0.13, 0.18) 0.02 (−0.12, 0.16) 0.02 (−0.11, 0.15) | 0.742 0.802 0.803 | −0.00 (−0.17, 0.16) −0.01 (−0.17, 0.14) −0.02 (−0.16, 0.12) | 0.984 0.861 0.822 | 0.03 (−0.05, 0.11) 0.03 (−0.05, 0.11) 0.03 (−0.05, 0.11) | 0.502 0.446 0.414 |
| SHBG Model 1 Model 2 Model 3 | −10.08 (−20.77, 0.61) −10.16 (−20.87, 0.55) −10.00 (−20.82, 0.83) | 0.065 0.063 0.070 | −11.37 (−26.60, 3.86) −11.28 (−26.72, 4.16) −10.60 (−26.26, 5.07) | 0.143 0.152 0.185 | 1.29 (−14.31, 16.90) 1.11 (−14.64, 16.87) 0.60 (−15.52, 16.72) | 0.871 0.890 0.942 |
| T-total Model 1 Model 2 Model 3 | −2.91 (−8.72, 2.90) −3.08 (−8.82, 2.66) −2.70 (−8.31, 2.91) | 0.327 0.292 0.345 | −6.21 (−12.31, −0.11) −6.70 (−12.75, −0.66) −5.56 (−11.94, 0.82) | 0.046 0.030 0.088 | 3.30 (−0.53, 7.13) 3.62 (−0.16, 7,41) 2.86 (−1.50, 7.21) | 0.091 0.061 0.199 |
| TFI Model 1 Model 2 Model 3 | −0.23 (−0.71 0.24) −0.25 (−0.72, 0.21) −0.24 (−0.69, 0.22) | 0.335 0.282 0.312 | −0.42 (−0.86, 0.01) −0.47 (−0.90, −0.05) −0.43 (−0.86, 0.01) | 0.054 0.029 0.046 | 0.19 (−0.14, 0.53) 0.22 (−0.08, 0.52) 0.20 (−0.10, 0.50) | 0.259 0.155 0.194 |
| FEI Model 1 Model 2 Model 3 | 0.004 (−0.113, 0.122) 0.011 (−0.109, 0.131) 0.011 (−0.108, 0.131) | 0.943 0.857 0.852 | −0.031 (−0.154, 0.092) −0.026 (−0.151, −0.100) −0.025 (−0.149, 0.099) | 0.619 0.688 0.689 | 0.035 (−0.043, 0.114) 0.037 (−0.041, 0.114) 0.037 (−0.040, 0.113) | 0.377 0.351 0.349 |
| AB vs. Control | NAB vs. Control | AB vs. NAB | ||||
|---|---|---|---|---|---|---|
| Difference Time-Exposure (95% IC) | p-Value | Difference Time-Exposure (95% IC) | p-Value | Difference Time-Exposure (95% IC) | p-Value | |
| Weight, kg | −0.4 (−3.0, 2.1) | 0.742 | −6.0 (−16.6, 4.6) | 0.267 | 5.6 (−4.8, 16.0) | 0.293 |
| BMI, kg/m2 | −0.4 (−1.4, 0.7) | 0.487 | −2.8 (−7.1, 1.6) | 0.218 | 2.4 (−1.9, 6.7) | 0.275 |
| WC, cm | −0.2 (−3.1, 2.7) | 0.887 | −5.2 (−12.3, 1.9) | 0.150 | 5.0 (−2.0, 12.3) | 0.160 |
| DBP, mmHg | 1.4 (−3.6, 6.3) | 0.590 | −6.3 (−12.9, 0.2) | 0.057 | 7.7 (2.1, 13.3) | 0.007 |
| SDP, mmHg | −1.7 (−8.8, 5.4) | 0.639 | −10.8 (−22.5, 0.9) | 0.070 | 9.1 (−2.2, 20.5) | 0.115 |
| Heart rate, bpm | 3.6 (−2.3, 9.5) | 0.233 | −0.4 (−5.9, 5.1) | 0.886 | 4.0 (−1.1, 9.1) | 0.125 |
| Glucose, mg/dL | 0.7 (−3.3, 4.7) | 0.735 | 3.1 (−5.9, 12.1) | 0.496 | −2.4 (−11.5, 6.7) | 0.601 |
| Total cholesterol, mg/dL | −6.0 (−19.6, 7.6) | 0.386 | −10.1 (−26.1, 5.8) | 0.212 | 4.1 (−9.7, 17.9) | 0.558 |
| LDL-C, mg/dL | −12.8 (−26.4, 0.8) | 0.064 | −16.1 (−29.2, −3.1) | 0.016 | 3.3 (−9.3, 15.9) | 0.600 |
| HDL-C, mg/dL | 3.5 (−3.6, 10.6) | 0.403 | 1.3 (−6.0, 8.6) | 0.734 | 2.2 (−6.0, 10.5) | 0.518 |
| Triglycerides, mg/dL | 7.2 (−11.3, 25.8) | 0.446 | 5.3 (−12.4, 23.0) | 0.558 | 1.9 (−14.5, 18.3) | 0.817 |
| ApoA1, mg/dL | 4.4 (−13.5, 22.2) | 0.633 | −16.6 (−33.3, 0.29) | 0.054 | 20.9 (5.1, 36.6) | 0.010 |
| ApoB, mg/dL | −2.3 (−12.8, 8.1) | 0.663 | −3.8 (−17.7, 10.2) | 0.598 | 1.4 (−13.8, 16.7) | 0.853 |
| Lpa, mg/dL | 18.1 (−6.5, 42.8) | 0.149 | 13.4 (−11.7, 38.4) | 0.295 | 4.8 (−5.6, 14.1) | 0.319 |
| AST, U/L | 0.4 (−7.8, 8.6) | 0.922 | −0.9 (−8.6, 6.8) | 0.821 | 1.3 (−5.5, 8.1) | 0.706 |
| ALT, U/L | 1.2 (−5.0, 7.5) | 0.705 | −0.7 (−6.6, 5.2) | 0.813 | 1.9 (−3.0, 6.9) | 0.445 |
| GGT, U/L | 7.2 (0.3, 14.2) | 0.042 | 6.4 (1−1, 11.6) | 0.018 | 0.9 (−6.5, 8.2) | 0.817 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Trius-Soler, M.; Marhuenda-Muñoz, M.; Laveriano-Santos, E.P.; Martínez-Huélamo, M.; Sasot, G.; Storniolo, C.E.; Estruch, R.; Lamuela-Raventós, R.M.; Tresserra-Rimbau, A. Moderate Consumption of Beer (with and without Ethanol) and Menopausal Symptoms: Results from a Parallel Clinical Trial in Postmenopausal Women. Nutrients 2021, 13, 2278. https://doi.org/10.3390/nu13072278
Trius-Soler M, Marhuenda-Muñoz M, Laveriano-Santos EP, Martínez-Huélamo M, Sasot G, Storniolo CE, Estruch R, Lamuela-Raventós RM, Tresserra-Rimbau A. Moderate Consumption of Beer (with and without Ethanol) and Menopausal Symptoms: Results from a Parallel Clinical Trial in Postmenopausal Women. Nutrients. 2021; 13(7):2278. https://doi.org/10.3390/nu13072278
Chicago/Turabian StyleTrius-Soler, Marta, María Marhuenda-Muñoz, Emily P. Laveriano-Santos, Miriam Martínez-Huélamo, Gemma Sasot, Carolina E. Storniolo, Ramon Estruch, Rosa M. Lamuela-Raventós, and Anna Tresserra-Rimbau. 2021. "Moderate Consumption of Beer (with and without Ethanol) and Menopausal Symptoms: Results from a Parallel Clinical Trial in Postmenopausal Women" Nutrients 13, no. 7: 2278. https://doi.org/10.3390/nu13072278
APA StyleTrius-Soler, M., Marhuenda-Muñoz, M., Laveriano-Santos, E. P., Martínez-Huélamo, M., Sasot, G., Storniolo, C. E., Estruch, R., Lamuela-Raventós, R. M., & Tresserra-Rimbau, A. (2021). Moderate Consumption of Beer (with and without Ethanol) and Menopausal Symptoms: Results from a Parallel Clinical Trial in Postmenopausal Women. Nutrients, 13(7), 2278. https://doi.org/10.3390/nu13072278











