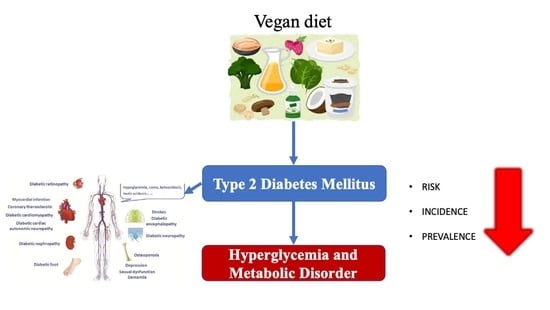
The Impact of Vegan Diet in the Prevention and Treatment of Type 2 Diabetes: A Systematic Review
Abstract
:1. Introduction
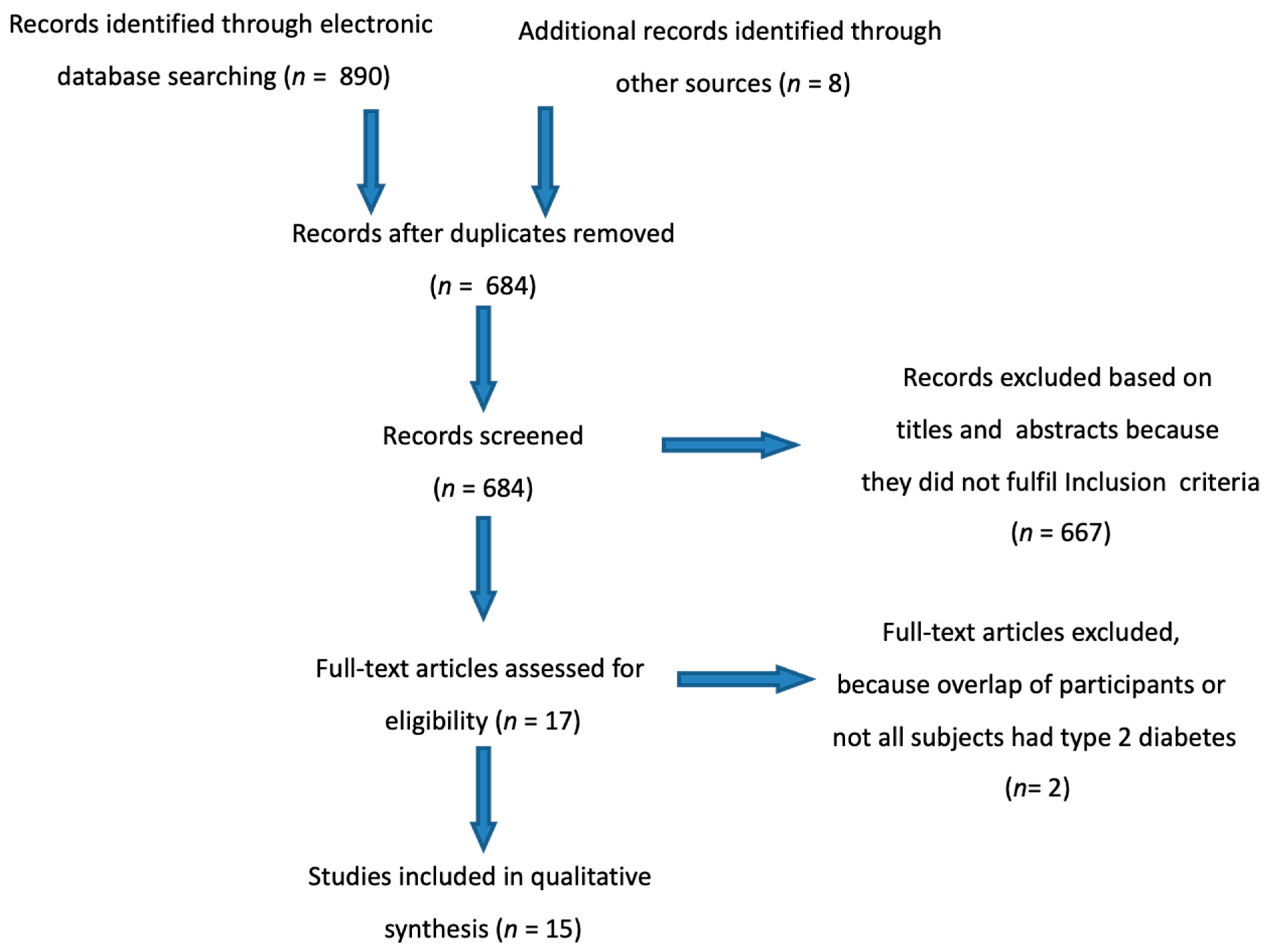
2. Materials and Methods
Methodological Quality Assessment of Studies
3. Results
3.1. Observational Studies
3.2. Studies Evaluating Prevalence of Type 2 Diabetes
3.3. Studies Evaluating Incidence of Type 2 Diabetes
3.4. Randomized Controlled Studies
3.5. Studies Evaluating Glycemic Control
3.6. Studies Evaluating Risk Factors for Diabetes Related Complications
4. Discussion
4.1. Potential Mechanisms
4.2. Potential Risk of Nutritional Deficiencies
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- The Vegan Society. UK: The Vegan Society. 2020. Available online: https://www.vegansociety.com/go-vegan/definition-veganism (accessed on 20 June 2020).
- Dinu, M.; Abbate, R.; Gensini, G.F.; Casini, A.; Sofi, F. Vegetarian, vegan diets and multiple health outcomes: A systematic review with meta-analysis of observational studies. Crit. Rev. Food Sci. Nutr. 2017, 57, 3640–3649. [Google Scholar] [CrossRef]
- American Diabetes Association 5. Lifestyle Management: Standards of Medical Care in Diabetes—2019. Diabetes Care 2018, 42, S46–S60. [Google Scholar] [CrossRef] [Green Version]
- Liberati, A.; Altman, D.G.; Tetzlaff, J.; Mulrow, C.; Gøtzsche, P.C.; Ioannidis, J.P.A.; Clarke, M.; Devereaux, P.; Kleijnen, J.; Moher, D. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate healthcare interventions: Explanation and elaboration. BMJ 2009, 339, b2700. [Google Scholar] [CrossRef] [Green Version]
- National Heart, Lung, and Blood Institute. Study Quality Assessment Tools; National Institutes of Health: Bethesda, MD, USA, 2018. Available online: https://www.nhlbi.nih.gov/health-topics/study-quality-assessment-tools (accessed on 20 January 2021).
- Higgins, J.P.T.; Altman, D.G.; Gøtzsche, P.C.; Jüni, P.; Moher, D.; Oxman, A.D.; Savović, J.; Schulz, K.F.; Weeks, L.; Sterne, J.A.C.; et al. The Cochrane Collaboration’s tool for assessing risk of bias in randomised trials. BMJ 2011, 343, d5928. [Google Scholar] [CrossRef] [Green Version]
- Tonstad, S.; Stewart, K.; Oda, K.; Batech, M.; Herring, R.; Fraser, G. Vegetarian diets and incidence of diabetes in the Adventist Health Study-2. Nutr. Metab. Cardiovasc. Dis. 2013, 23, 292–299. [Google Scholar] [CrossRef] [Green Version]
- Satija, A.; Bhupathiraju, S.N.; Rimm, E.B.; Spiegelman, D.; Chiuve, S.E.; Borgi, L.; Willett, W.C.; Manson, J.E.; Sun, Q.; Hu, F.B. Plant-Based Dietary Patterns and Incidence of Type 2 Diabetes in US Men and Women: Results from Three Prospective Cohort Studies. PLoS Med. 2016, 13, e1002039. [Google Scholar] [CrossRef] [Green Version]
- Koloverou, E.; Panagiotakos, D.B.; Georgousopoulou, E.N.; Grekas, A.; Christou, A.; Chatzigeorgiou, M.; Chrysohoou, C.; Tousoulis, D.; Stefanadis, C.; Pitsavos, C.; et al. Dietary Patterns and 10-year (2002-2012) Incidence of Type 2 Diabetes: Results from the ATTICA Cohort Study. Rev. Diabet. Stud. 2016, 13, 246–256. [Google Scholar] [CrossRef] [Green Version]
- Chen, Z.; Zuurmond, M.G.; Van Der Schaft, N.; Nano, J.; Wijnhoven, H.; Ikram, M.A.; Franco, O.H.; Voortman, T. Plant versus animal based diets and insulin resistance, prediabetes and type 2 diabetes: The Rotterdam Study. Eur. J. Epidemiology 2018, 33, 883–893. [Google Scholar] [CrossRef] [Green Version]
- Papier, K.; Appleby, P.N.; Fensom, G.K.; Knuppel, A.; Perez-Cornago, A.; Schmidt, J.A.; Tong, T.Y.N.; Key, T.J. Vegetarian diets and risk of hospitalisation or death with diabetes in British adults: Results from the EPIC-Oxford study. Nutr. Diabetes 2019, 9, 7. [Google Scholar] [CrossRef]
- Tonstad, S.; Butler, T.; Yan, R.; Fraser, G.E. Type of Vegetarian Diet, Body Weight, and Prevalence of Type 2 Diabetes. Diabetes Care 2009, 32, 791–796. [Google Scholar] [CrossRef] [Green Version]
- Agrawal, S.; Millett, C.J.; Dhillon, P.K.; Subramanian, S.V.; Ebrahim, S. Type of vegetarian diet, obesity and diabetes in adult Indian population. Nutr. J. 2014, 13, 89. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nicholson, A.S.; Sklar, M.; Barnard, N.D.; Gore, S.; Sullivan, R.; Browning, S. Toward Improved Management of NIDDM: A Randomized, Controlled, Pilot Intervention Using a Lowfat, Vegetarian Diet. Prev. Med. 1999, 29, 87–91. [Google Scholar] [CrossRef]
- Wheeler, M.L.; Fineberg, S.E.; Fineberg, N.S.; Gibson, R.G.; Hackward, L.L. Animal versus plant protein meals in individuals with type 2 diabetes and microalbuminuria: Effects on renal, glycemic, and lipid parameters. Diabetes Care 2002, 25, 1277–1282. [Google Scholar] [CrossRef] [Green Version]
- Ferdowsian, H.R.; Barnard, N.D.; Hoover, V.J.; Katcher, H.I.; Levin, S.M.; Green, A.A.; Cohen, J.L. A Multicomponent Intervention Reduces Body Weight and Cardiovascular Risk at a GEICO Corporate Site. Am. J. Health Promot. 2010, 24, 384–387. [Google Scholar] [CrossRef]
- Barnard, N.D.; Cohen, J.; Jenkins, D.J.A.; Turner-McGrievy, G.; Gloede, L.; Green, A.; Ferdowsian, H. A low-fat vegan diet and a conventional diabetes diet in the treatment of type 2 diabetes: A randomized, controlled, 74-wk clinical trial. Am. J. Clin. Nutr. 2009, 89, 1588S–1596S. [Google Scholar] [CrossRef]
- Mishra, S.; Xu, J.; Agarwal, U.; Gonzales, J.R.; Levin, S.A.; Barnard, N.D. A multicenter randomized controlled trial of a plant-based nutrition program to reduce body weight and cardiovascular risk in the corporate setting: The GEICO study. Eur. J. Clin. Nutr. 2013, 67, 718–724. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bunner, A.; Wells, C.L.; Gonzales, J.C.; Agarwal, U.; Bayat, E.; Barnard, N.D. A dietary intervention for chronic diabetic neuropathy pain: A randomized controlled pilot study. Nutr. Diabetes 2015, 5, e158. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lee, Y.-M.; Kim, S.-A.; Lee, I.-K.; Kim, J.-G.; Park, K.-G.; Jeong, J.-Y.; Jeon, J.-H.; Shin, J.-Y.; Lee, D.-H. Effect of a Brown Rice Based Vegan Diet and Conventional Diabetic Diet on Glycemic Control of Patients with Type 2 Diabetes: A 12-Week Randomized Clinical Trial. PLoS ONE 2016, 11, e0155918. [Google Scholar] [CrossRef] [Green Version]
- Barnard, N.D.; Levin, S.M.; Gloede, L.; Flores, R. Turning the Waiting Room into a Classroom: Weekly Classes Using a Vegan or a Portion-Controlled Eating Plan Improve Diabetes Control in a Randomized Translational Study. J. Acad. Nutr. Diet. 2018, 118, 1072–1079. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hu, E.A.; Pan, A.; Malik, V.; Sun, Q. White rice consumption and risk of type 2 diabetes: Meta-analysis and systematic review. BMJ 2012, 344, e1454. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Naghshi, S.; Sadeghi, O.; Willett, W.C.; Esmaillzadeh, A. Dietary intake of total, animal, and plant proteins and risk of all cause, cardiovascular, and cancer mortality: Systematic review and dose-response meta-analysis of prospective cohort studies. BMJ 2020, 370, m2412. [Google Scholar] [CrossRef] [PubMed]
- Ashley, J.M.; Jarvis, W.T. Position of the American Dietetic Association: Health Implications of Dietary Fiber. J. Am. Diet. Assoc. 2008, 108, 1716–1731. [Google Scholar] [CrossRef]
- Schulze, M.B.; Schulz, M.; Heidemann, C.; Schienkiewitz, A.; Hoffmann, K.; Boeing, H. Fiber and Magnesium Intake and Incidence of Type 2 Diabetes. Arch. Intern. Med. 2007, 167, 956–965. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- The InterAct Consortium Dietary fibre and incidence of type 2 diabetes in eight European countries: The EPIC-InterAct Study and a meta-analysis of prospective studies. Diabetology 2015, 58, 1394–1408. [CrossRef] [Green Version]
- Sparks, L.M.; Xie, H.; Koza, R.A.; Mynatt, R.; Hulver, M.W.; Bray, G.A.; Smith, S.R. A High-Fat Diet Coordinately Downregulates Genes Required for Mitochondrial Oxidative Phosphorylation in Skeletal Muscle. Diabetes 2005, 54, 1926–1933. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Meex, R.C.; Blaak, E.E.; Van Loon, L.J. Lipotoxicity plays a key role in the development of both insulin resistance and muscle atrophy in patients with type 2 diabetes. Obes. Rev. 2019, 20, 1205–1217. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Goff, L.M.; Bell, J.D.; So, P.-W.; Dornhorst, A.; Frost, G.S. Veganism and its relationship with insulin resistance and intramyocellular lipid. Eur. J. Clin. Nutr. 2004, 59, 291–298. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jehn, M.; Clark, J.M.; Guallar, E. Serum Ferritin and Risk of the Metabolic Syndrome in U.S. Adults. Diabetes Care 2004, 27, 2422–2428. [Google Scholar] [CrossRef] [Green Version]
- Kim, Y.; Keogh, J.; Clifton, P. A review of potential metabolic etiologies of the observed association between red meat consumption and development of type 2 diabetes mellitus. Metabolism 2015, 64, 768–779. [Google Scholar] [CrossRef] [PubMed]
- Agnoli, C.; Baroni, L.; Bertini, I.; Ciappellano, S.; Fabbri, A.; Papa, M.; Pellegrini, N.; Sbarbati, R.; Scarino, M.; Siani, V.; et al. Position paper on vegetarian diets from the working group of the Italian Society of Human Nutrition. Nutr. Metab. Cardiovasc. Dis. 2017, 27, 1037–1052. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Berrazaga, I.; Micard, V.; Gueugneau, M.; Walrand, S. The Role of the Anabolic Properties of Plant- versus Animal-Based Protein Sources in Supporting Muscle Mass Maintenance: A Critical Review. Nutrient 2019, 11, 1825. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pawlak, R.; Lester, S.E.; Babatunde, T. The prevalence of cobalamin deficiency among vegetarians assessed by serum vitamin B12: A review of literature. Eur. J. Clin. Nutr. 2014, 68, 541–548. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schüpbach, R.; Wegmuller, R.; Berguerand, C.; Bui, M.; Herteraeberli, I. Micronutrient status and intake in omnivores, vegetarians and vegans in Switzerland. Eur. J. Nutr. 2017, 56, 283–293. [Google Scholar] [CrossRef] [PubMed]
- Chapman, L.; Darling, A.; Brown, J. Association between metformin and vitamin B12 deficiency in patients with type 2 diabetes: A systematic review and meta-analysis. Diabetes Metab. 2016, 42, 316–327. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Iguacel, I.; Miguel-Berges, M.L.; Gómez-Bruton, A.; Moreno, L.A.; Julián, C. Veganism, vegetarianism, bone mineral density, and fracture risk: A systematic review and meta-analysis. Nutr. Rev. 2019, 77, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Janghorbani, M.; Van Dam, R.M.; Willett, W.C.; Hu, F.B. Systematic Review of Type 1 and Type 2 Diabetes Mellitus and Risk of Fracture. Am. J. Epidemiol. 2007, 166, 495–505. [Google Scholar] [CrossRef] [PubMed]
| Reference | Country | Cohort | Sex | Follow-Up (Year) | Population | QA |
|---|---|---|---|---|---|---|
| Tonstad (2013) [7] | US and Canada | Adventist Health Study-2 | M/F | 2 | 41,387 | fair |
| Satija (2015) [8] | US | Nurses Health Study + Nurses Health Study-2 + Health Professionals F–up Study | M/F | 20 | 200,727 | fair |
| Koloverou (2016) [9] | Greece | ATTICA Cohort Study | M/F | 10 | 1485 | good |
| Chen (2018) [10] | Netherland | Rotterdam Study | M/F | 7.3 | 6798 | good |
| Papier (2019) [11] | UK | Oxford–EPIC Study | M/F | 17.6 | 45,314 | good |
| Tonstad (2009) [12] | US and Canada | Adventist Health Study-2 | M/F | Cross-sectional | 60,903 | fair |
| Agrawal (2014) [13] | India | National Family Health Survey-3 | M/F | Cross-sectional | 156,317 | good |
| Reference | Country | Participants | Design | Follow-up (Week) | Control Diet | QA (Score) |
|---|---|---|---|---|---|---|
| Nicholson (1999) [14]. | US | 11 (7I + 4C) | P | 12 | Conventional low–fat diet | 2.5 |
| Wheeler (2002) [15]. | US | 17 | CO | 6 | Animal–based protein diet | 3 |
| Barnard (2009) [16]. | US | 99 (49I + 50C) | P | 74 | Conventional diabetes diet | 4 |
| Ferdowsian (2010) [17]. | US | 19 (10I + 9C) | P | 22 | Usual diet | 2.5 |
| Mishra (2013) [18]. | US | 35 (17I + 18C) | P | 18 | Usual diet | 4 |
| Bunner (2015) [19]. | US | 33 (17I + 16C) | P | 20 | Usual diet | 4 |
| Lee (2016) [20]. | Korea | 93 (47I + 46C) | P | 12 | Conventional diabetes diet | 3.5 |
| Barnard (2018) [21]. | US | 40 (19I + 21C) | P | 20 | Portion-controlled diet | 4 |
| Reference | Intervention Group | Control Group | Between-Group Difference (Intervention–Control) Mean (95% CI) | p-Value | |||||
|---|---|---|---|---|---|---|---|---|---|
| Baseline | Final | Change | Baseline | Final | Change | ||||
| Nicholson (1999) [14] | 8.3 (1.7) | 6.9 (1.1) | −1.4 | 8.0 (1.1) | 7.0 (0.6) | −1.0 | |||
| Wheeler (2002) [15] | 8.1 (0.4) | 7.5 (0.3) | −0.6 ** | 7.9 (0.4) | 7.4 (0.3) | −0.5 ** | −0.1 | 0.75 | |
| Barnard (2009) [16] | 8.05 (0.16) | 7.71 (0.19) | −0.34 (0.19) | 7.93 (0.14) | 7.79 (0.18) | −0.14 (0.17) | −0.20 (−0.71 to 0.30) | 0.43 | |
| Ferdowsian (2010) [17] | 7.4 (0.3) | 7.1 (0.5) | −0.3 (0.6) | 7.0 (0.4) | 6.7 (0.4) | −0.3 (0.2) | 0 (−1.4 to 1.4) | 0.97 | |
| Mishra (2013) [18] | 7.52 (0.49) | 6.78 (0.44) | −0.74 (0.19) ** | 7.03 (0.36) | 7.13 (0.38) | 0.1 (0.12) | −0.84 (−0.37 to −1.1) ** | 0.003 | |
| Bunner (2015) [19] | 8.0 (1.7) | 7.2 (1.4) | −0.8 (1.2) * | 7.8 (1.6) | 7.8 (1.4) | 0.0 (0.9) | −0.7 (−1.5 to 0.1) | 0.07 | |
| Lee (2016) [20] | 7.7 (1.3) | 7.1 (0.9) | −0.5 (0.8) ** | 7.4 (1.0) | 7.2 (0.9) | −0.2 (0.7) * | −0.3 * | 0.017 | |
| Barnard (2018) [21] median | 6.7 | 6.2 | −0.4 * | 6.8 | 6.2 | −0.4 ** | 0.1 (−0.2 to 0.6) | 0.68 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pollakova, D.; Andreadi, A.; Pacifici, F.; Della-Morte, D.; Lauro, D.; Tubili, C. The Impact of Vegan Diet in the Prevention and Treatment of Type 2 Diabetes: A Systematic Review. Nutrients 2021, 13, 2123. https://doi.org/10.3390/nu13062123
Pollakova D, Andreadi A, Pacifici F, Della-Morte D, Lauro D, Tubili C. The Impact of Vegan Diet in the Prevention and Treatment of Type 2 Diabetes: A Systematic Review. Nutrients. 2021; 13(6):2123. https://doi.org/10.3390/nu13062123
Chicago/Turabian StylePollakova, Daniela, Aikaterini Andreadi, Francesca Pacifici, David Della-Morte, Davide Lauro, and Claudio Tubili. 2021. "The Impact of Vegan Diet in the Prevention and Treatment of Type 2 Diabetes: A Systematic Review" Nutrients 13, no. 6: 2123. https://doi.org/10.3390/nu13062123
APA StylePollakova, D., Andreadi, A., Pacifici, F., Della-Morte, D., Lauro, D., & Tubili, C. (2021). The Impact of Vegan Diet in the Prevention and Treatment of Type 2 Diabetes: A Systematic Review. Nutrients, 13(6), 2123. https://doi.org/10.3390/nu13062123