Is There Association between Altered Adrenergic System Activity and Microvascular Endothelial Dysfunction Induced by a 7-Day High Salt Intake in Young Healthy Individuals
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Population
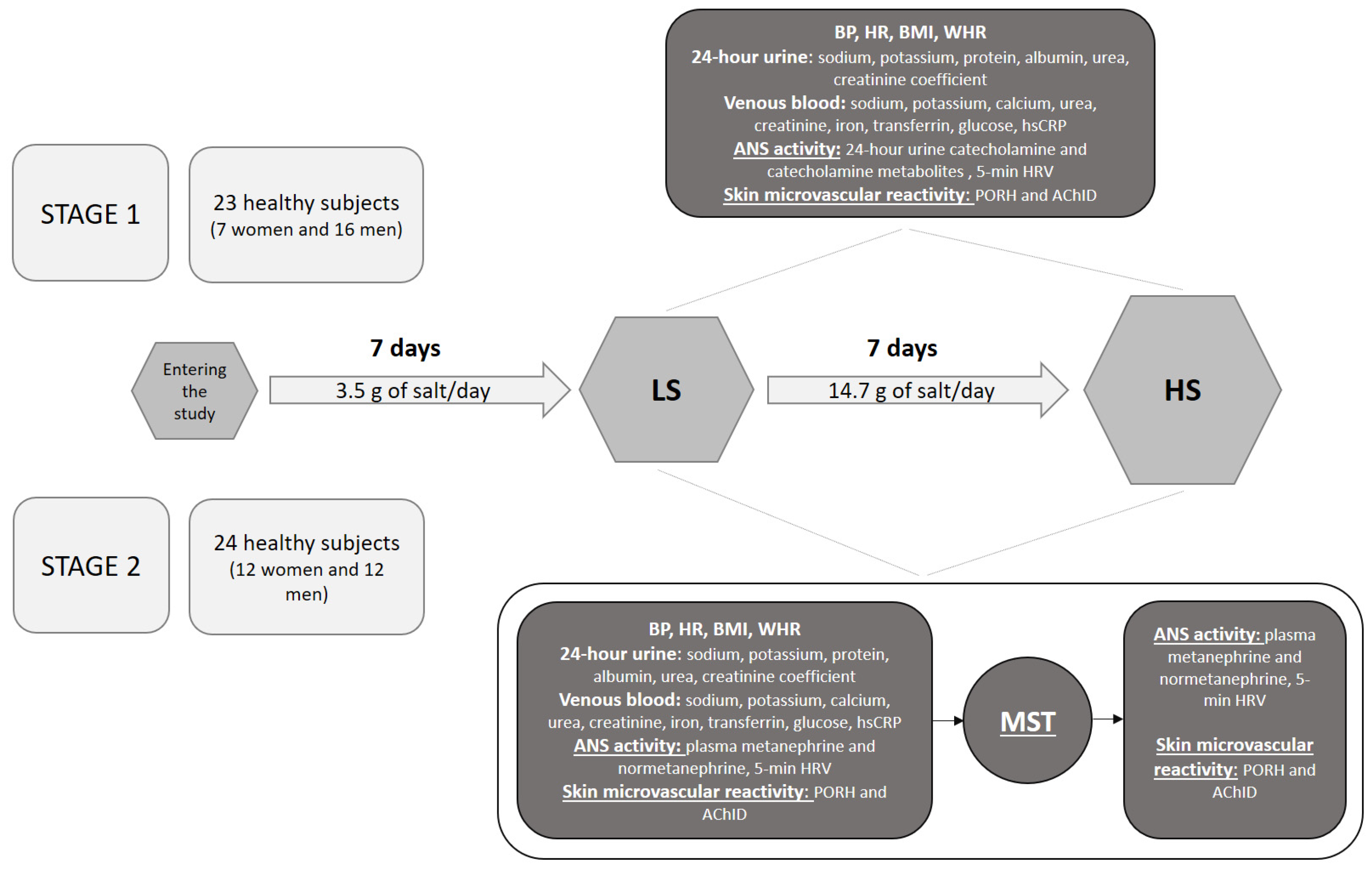
2.2. Study Protocol
2.3. Hemodynamic and Anthropometric Measurement
2.4. 24-h Urine Samples Analysis
2.5. Venous Blood Samples Analysis
2.6. Functional Assessment of Skin Microvascular Endothelial Function
2.7. Measurement of 5-min Heart Rate Variability
2.8. Mental Stress Test
2.9. Statistical Analysis
3. Results
3.1. Stage 1 Study Results—The Effects of 7-Day High-Salt (HS) Loading on Autonomic Nervous System (ANS) Activity and Microvascular Endothelial Function
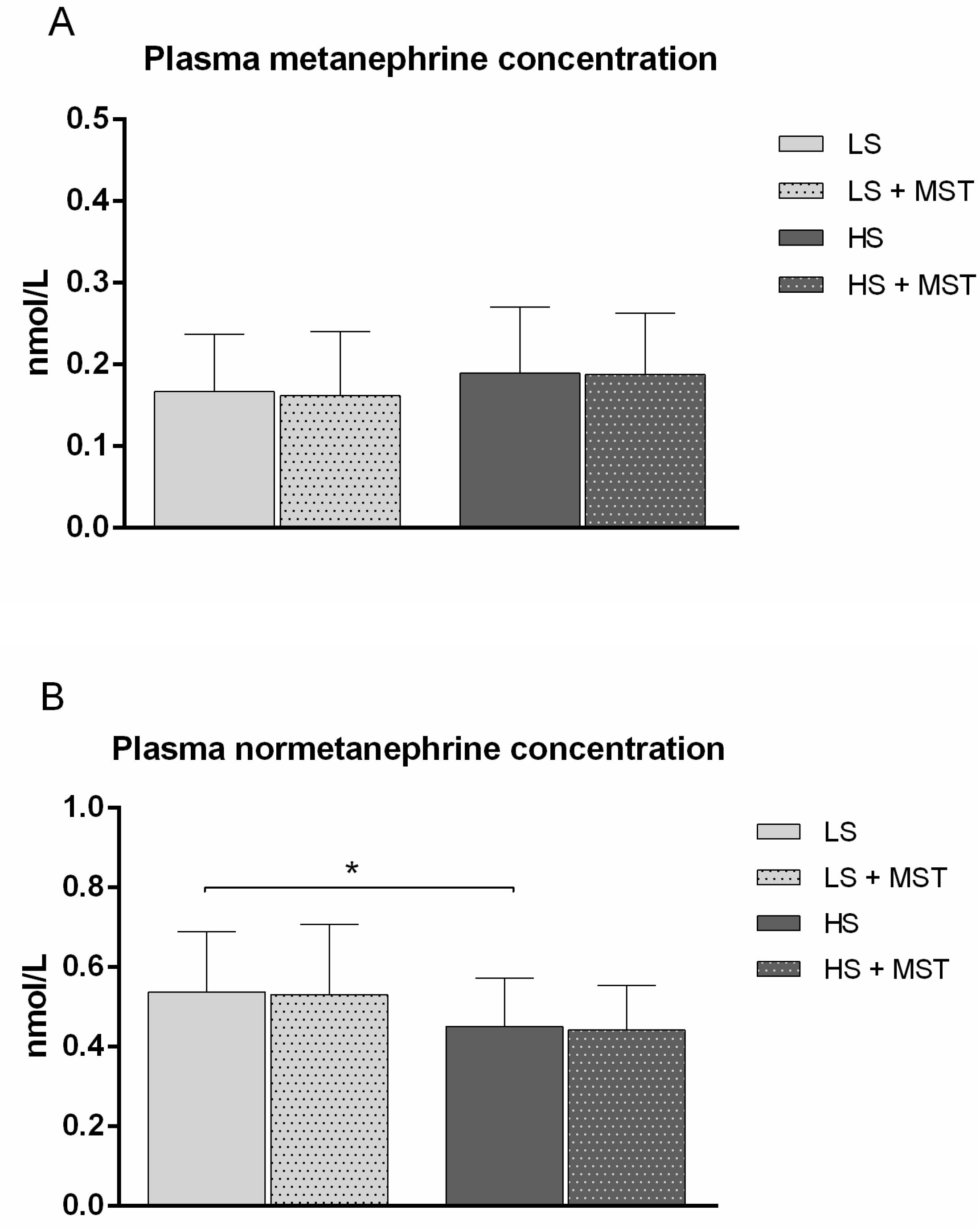
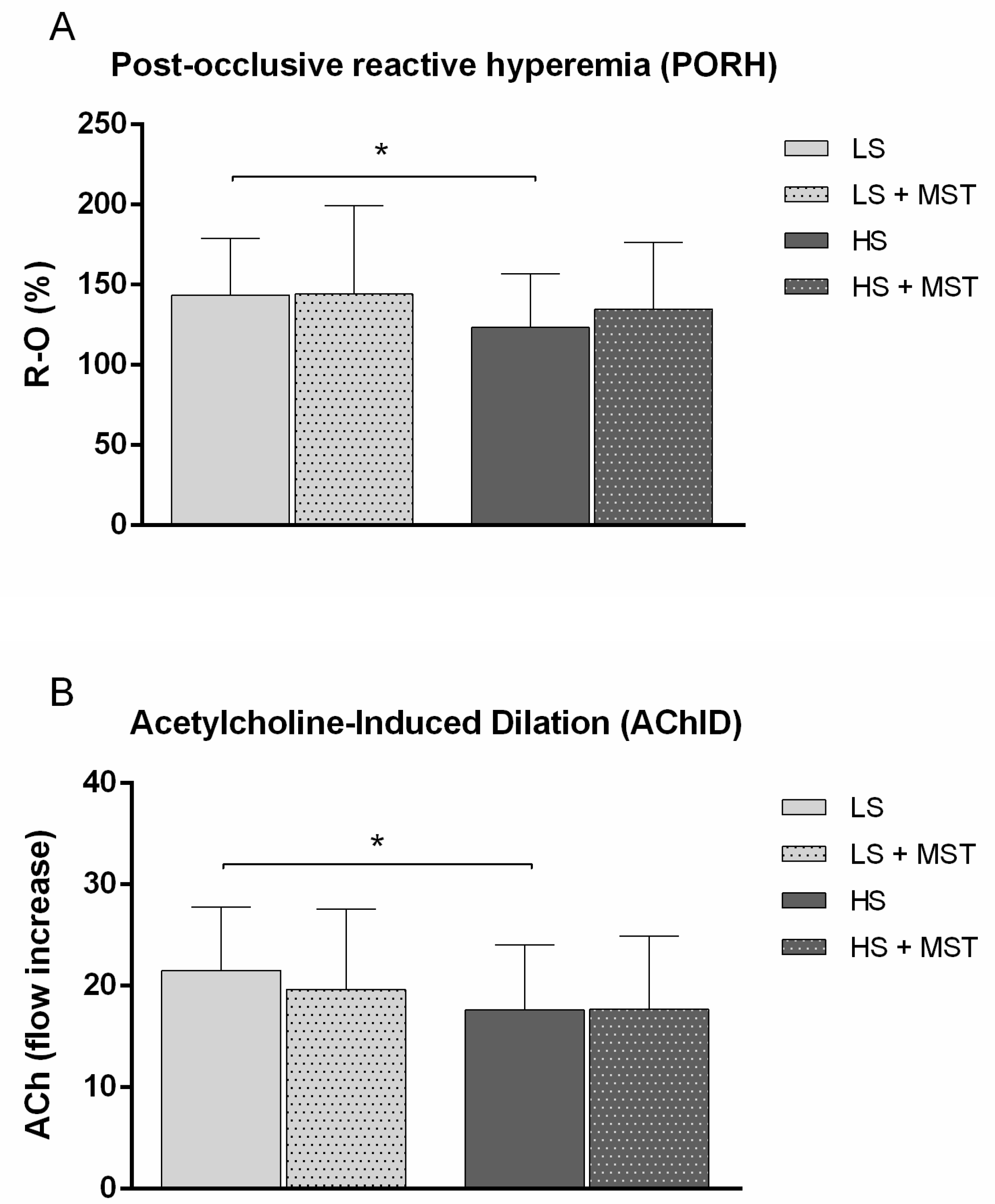
3.2. Stage 2 Study Results—The Effects of Increased Sympathetic Nervous System (SNS) Activity (by Mental Stress Test; MST) during 7-Day High-Salt (HS) Loading on Microvascular Endothelial Function
4. Discussion
4.1. High-Salt Diet and Autonomic Nervous System Activity
4.2. High-Salt Diet, Autonomic Nervous System and Microvascular Endothelial (Dys)function
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Jelakovic, B.; Vrdoljak, A.; Pecin, I.; Buzjak, V.; Karanovic, S.; Ivkovic, V.; Dapic, K.; Domislovic, V.; Reiner, Z. Less salt—More health. Croatian Action on Salt and Health (CRASH). J. Hypertens Res. 2016, 2, 61–68. [Google Scholar]
- Jelaković, B.; Reiner, Ž. Croatian national programme for reduction of excessive salt intake. Liječnički Vjesn. 2014, 136, 304–306. [Google Scholar]
- Jelakovic, B. Salt Intake in Croatia—EHUH 2 Report. In 41st Symposium Hypertension Highlights in 2020. Available online: https://kongresi.emed.hr/course/info.php?id=149 (accessed on 19 May 2021).
- Weinberger, M.H.; Fineberg, N.S.; Fineberg, S.E.; Weinberger, M. Salt Sensitivity, Pulse Pressure, and Death in Normal and Hypertensive Humans. Hypertension 2001, 37, 429–432. [Google Scholar] [CrossRef] [PubMed]
- Wong, M.M.Y.; Arcand, J.; Leung, A.A.; Ma, S.R.T.; Campbell, N.R.C.; Webster, J. The science of salt: A regularly updated systematic review of salt and health outcomes (December 2015–March 2016). J. Clin. Hypertens. 2017, 19, 322–332. [Google Scholar] [CrossRef] [PubMed]
- He, F.J.; MacGregor, G.A. A comprehensive review on salt and health and current experience of worldwide salt reduction programmes. J. Hum. Hypertens. 2009, 23, 363–384. [Google Scholar] [CrossRef] [PubMed]
- Bragulat, E.; de la Sierra, A. Salt intake, endothelial dysfunction, and salt-sensitive hypertension. J. Clin. Hypertens. 2020, 4, 41–46. [Google Scholar] [CrossRef]
- Grassi, G.; Cattaneo, B.M.; Seravalle, G.; Lanfranchi, A.; Mancia, G. Baroreflex Control of Sympathetic Nerve Activity in Essential and Secondary Hypertension. Hypertension 1998, 31, 68–72. [Google Scholar] [CrossRef]
- Boegehold, M.A.; Drenjancevic, I.; Lombard, J.H. Salt, Angiotensin II, Superoxide, and Endothelial Function. In Comprehensive Physiology; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2015; pp. 215–254. [Google Scholar]
- Cavka, A.; Jukic, I.; Ali, M.; Goslawski, M.; Bian, J.-T.; Wang, E.; Drenjancevic, I.; Phillips, S.A. Short-term high salt intake reduces brachial artery and microvascular function in the absence of changes in blood pressure. J. Hypertens. 2016, 34, 676–684. [Google Scholar] [CrossRef]
- DuPont, J.J.; Greaney, J.L.; Wenner, M.M.; Lennon-Edwards, S.L.; Sanders, P.W.; Farquhar, W.B.; Edwards, D.G. High dietary sodium intake impairs endothelium-dependent dilation in healthy salt-resistant humans. J. Hypertens. 2013, 31, 530–536. [Google Scholar] [CrossRef]
- Lennon-Edwards, S.; Ramick, M.G.; Matthews, E.L.; Brian, M.S.; Farquhar, W.B.; Edwards, D.G. Salt loading has a more deleterious effect on flow-mediated dilation in salt-resistant men than women. Nutr. Metab. Cardiovasc. Dis. 2014, 24, 990–995. [Google Scholar] [CrossRef]
- Dickinson, K.M.; Clifton, P.M.; Keogh, J.B. Endothelial function is impaired after a high-salt meal in healthy subjects. Am. J. Clin. Nutr. 2011, 93, 500–505. [Google Scholar] [CrossRef] [PubMed]
- Cavka, A.; Cosic, A.; Jukic, I.; Jelakovic, B.; Lombard, J.H.; Phillips, S.A.; Seric, V.; Mihaljevic, I.; Drenjancevic, I. The role of cyclo-oxygenase-1 in high-salt diet-induced microvascular dysfunction in humans. J. Physiol. 2015, 593, 5313–5324. [Google Scholar] [CrossRef] [PubMed]
- Barić, L.; Drenjančević, I.; Matić, A.; Stupin, M.; Kolar, L.; Mihaljević, Z.; Lenasi, H.; Šerić, V.; Stupin, A. Seven-day salt loading impairs microvascular endothelium-dependent vasodilation without changes in blood pressure, body composition and fluid status in healthy young humans. Kidney Blood Press Res. 2019, 44, 835–847. [Google Scholar] [CrossRef] [PubMed]
- Greaney, J.L.; DuPont, J.J.; Lennon-Edwards, S.L.; Sanders, P.W.; Edwards, D.G.; Farquhar, W.B. Dietary sodium loading impairs microvascular function independent of blood pressure in humans: Role of oxidative stress. J. Physiol. 2012, 590, 5519–5528. [Google Scholar] [CrossRef] [PubMed]
- Weinberger, M.H. Salt sensitivity is associated with an increased mortality in both normal and hypertensive humans. J. Clin. Hypertens. 2002, 4, 274–276. [Google Scholar] [CrossRef]
- Barić, L.; Drenjančević, I.; Mihalj, M.; Matić, A.; Stupin, M.; Kolar, L.; Mihaljević, Z.; Mrakovčić-Šutić, I.; Šerić, V.; Stupin, A. enhanced antioxidative defense by vitamins C and E consumption prevents 7-day high-salt diet-induced microvascular endothelial function impairment in young healthy individuals. J. Clin. Med. 2020, 9, 843. [Google Scholar] [CrossRef]
- Schweda, F. Salt feedback on the renin-angiotensin-aldosterone system. Pflügers Arch. Eur. J. Physiol. 2015, 467, 565–576. [Google Scholar] [CrossRef] [PubMed]
- Stupin, A.; Drenjančević, I.; Rašić, L.; Ćosić, A.; Stupin, M. A cross-talk between renin-angiotensin and adrenergic system in cardiovascular health and disease. Southeast. Eur. Med. J. 2017, 1, 90–107. [Google Scholar]
- Campese, V.M.; Romoff, M.S.; Levitan, D.; Saglikes, Y.; Friedler, R.M.; Massry, S.G. Abnormal relationship between sodium intake and sympathetic nervous system activity in salt-sensitive patients with essential hypertension. Kidney Int. 1982, 21, 371–378. [Google Scholar] [CrossRef]
- Brooks, V.L.; Haywood, J.R.; Johnson, A.K. Translation of Salt Retention to Central Activation of The Sympathetic Nervous System In Hypertension. Clin. Exp. Pharmacol. Physiol. 2005, 32, 426–432. [Google Scholar] [CrossRef]
- O’Donaughy, T.L.; Qi, Y.; Brooks, V.L. Central Action of Increased Osmolality to Support Blood Pressure in Deoxycorticosterone Acetate–Salt Rats. Hypertension 2006, 48, 658–663. [Google Scholar] [CrossRef] [PubMed]
- Aldehni, F.; Tang, T.; Madsen, K.; Plattner, M.; Schreiber, A.; Friis, U.G.; Hammond, H.K.; Han, P.L.; Schweda, F. Stimulation of Renin Secretion by Catecholamines Is Dependent on Adenylyl Cyclases 5 and 6. Hypertension 2011, 57, 460–468. [Google Scholar] [CrossRef] [PubMed]
- Friis, U.G.; Madsen, K.; Stubbe, J.; Hansen, P.B.L.; Svenningsen, P.; Bie, P.; Skøtt, O.; Jensen, B.L. Regulation of renin secretion by renal juxtaglomerular cells. Pflügers Arch. Eur. J. Physiol. 2013, 465, 25–37. [Google Scholar] [CrossRef] [PubMed]
- Adams, J.M.; McCarthy, J.J.; Stocker, S.D. Excess Dietary Salt Alters Angiotensinergic Regulation of Neurons in the Rostral Ventrolateral Medulla. Hypertension 2008, 52, 932–937. [Google Scholar] [CrossRef]
- Jukic, I.; Mihaljevic, Z.; Matic, A.; Mihalj, M.; Kozina, N.; Selthofer-Relatic, K.; Mihaljevic, D.; Koller, A.; Tartaro Bujak, I.; Drenjancevic, I. Angiotensin II type 1 receptor is involved in flow-induced vasomotor responses of isolated middle cerebral arteries: Role of oxidative stress. Am. J. Physiol. Circ. Physiol. 2021, 320, H1609–H1624. [Google Scholar] [CrossRef]
- Cavka, A.; Cosic, A.; Grizelj, I.; Koller, A.; Jelakovic, B.; Lombard, J.H.; Phillips, S.A.; Drenjancevic, I. Effects of AT1 Receptor Blockade on Plasma Thromboxane A 2 (TXA 2) Level and Skin Microcirculation in Young Healthy Women on Low Salt Diet. Kidney Blood Press Res. 2013, 37, 432–442. [Google Scholar] [CrossRef] [PubMed]
- Stupin, A.; Rasic, L.; Matic, A.; Stupin, M.; Kralik, Z.; Kralik, G.; Grcevic, M.; Drenjancevic, I. Omega-3 polyunsaturated fatty acids-enriched hen eggs consumption enhances microvascular reactivity in young healthy individuals. Appl. Physiol. Nutr. Metab. 2018, 43, 988–995. [Google Scholar] [CrossRef]
- Stupin, A.; Mihalj, M.; Kolobarić, N.; Šušnjara, P.; Kolar, L.; Mihaljević, Z.; Matić, A.; Stupin, M.; Jukić, I.; Kralik, Z.; et al. Anti-inflammatory potential of n-3 polyunsaturated fatty acids enriched hen eggs consumption in improving microvascular endothelial function of healthy individuals—Clinical trial. Int. J. Mol. Sci. 2020, 21, 4149. [Google Scholar] [CrossRef]
- Anderson, E.A.; Sinkey, C.A.; Lawton, W.J.; Mark, A.L. Elevated sympathetic nerve activity in borderline hypertensive humans. Evidence from direct intraneural recordings. Hypertension 1989, 14, 177–183. [Google Scholar] [CrossRef]
- De La Sierra, A.; Lluch, M.D.M.; Coca, A.; Aguilera, M.T.; Giner, V.; Bragulat, E.; Urbano-Márquez, A. Fluid, Ionic and hormonal changes induced by high salt intake in salt-sensitive and salt-resistant hypertensive patients. Clin. Sci. 1996, 91, 155–161. [Google Scholar] [CrossRef]
- Lohmeier, T.E.; Hildebrandt, D.A.; Hood, W.A. Renal nerves promote sodium excretion during long-term increases in salt intake. Hypertension 1999, 33, 487–492. [Google Scholar] [CrossRef][Green Version]
- Campese, V.M.; Karubian, F.; Chervu, I.; Parise, M.; Sarkies, N.; Bigazzi, R. Pressor reactivity to norepinephrine and angiotensin in salt-sensitive hypertensive patients. Hypertension 1993, 21, 301–307. [Google Scholar] [CrossRef]
- Koolen, M.I.; van Brummelen, P. Adrenergic activity and peripheral hemodynamics in relation to sodium sensitivity in patients with essential hypertension. Hypertension 1984, 6, 820–825. [Google Scholar] [CrossRef] [PubMed]
- Miyajima, E. Reduced sympathetic inhibition in salt-sensitive Japanese young adults. Am. J. Hypertens. 1999, 12, 1195–1200. [Google Scholar] [CrossRef][Green Version]
- Graudal, N.A.; Hubeck-Graudal, T.; Jurgens, G. Effects of low sodium diet versus high sodium diet on blood pressure, renin, aldosterone, catecholamines, cholesterol, and triglyceride. Cochrane Database Syst. Rev. 2017. [Google Scholar] [CrossRef] [PubMed]
- Malik, M. Heart rate variability: Standards of measurement, physiological interpretation and clinical use. Task Force of the European Society of Cardiology and the North American Society of Pacing and Electrophysiology. Circulation 1996, 93, 1043–1065. [Google Scholar] [CrossRef]
- Young, H.A.; Benton, D. Heart-rate variability: A biomarker to study the influence of nutrition on physiological and psychological health? Behav. Pharmacol. 2018, 29, 140–151. [Google Scholar] [CrossRef] [PubMed]
- Drenjancevic, I.; Grizelj, I.; Harsanji-Drenjancevic, I.; Cavka, A.; Selthofer-Relatic, K. The interplay between sympathetic overactivity, hypertension and heart rate variability (Review, invited). Acta Physiol. Hung. 2014, 101, 129–142. [Google Scholar] [CrossRef]
- McNeely, J.D.; Windham, B.G.; Anderson, D.E. Dietary sodium effects on heart rate variability in salt sensitivity of blood pressure. Psychophysiology 2008, 45, 405–411. [Google Scholar] [CrossRef]
- Pogson, Z.E.K.; McKeever, T.M.; Lewis, S.A.; Pacey, S.J.; Antoniak, M.D.; Britton, J.R.; Fogarty, A.W. Does a low sodium diet modify heart rate variability? A randomised placebo-controlled double-blind trial. Int. J. Cardiol. 2009, 135, 390–393. [Google Scholar] [CrossRef]
- Allen, A.R.; Gullixson, L.R.; Wolhart, S.C.; Kost, S.L.; Schroeder, D.R.; Eisenach, J.H. Dietary sodium influences the effect of mental stress on heart rate variability. J. Hypertens. 2014, 32, 374–382. [Google Scholar] [CrossRef] [PubMed]
- Minami, J.; Kawano, Y.; Ishimitsu, T.; Takishita, S. Blunted parasympathetic modulation in salt-sensitive patients with essential hypertension. J. Hypertens. 1997, 15, 727–735. [Google Scholar] [CrossRef] [PubMed]
- Roustit, M.; Cracowski, J.-L. Assessment of endothelial and neurovascular function in human skin microcirculation. Trends Pharmacol. Sci. 2013, 34, 373–384. [Google Scholar] [CrossRef] [PubMed]
- Carter, J.R.; Goldstein, D.S. Sympathoneural and Adrenomedullary Responses to Mental Stress. In Comprehensive Physiology; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2014; pp. 119–146. [Google Scholar]
- Pussard, E. Plasma metanephrines responses to adreno-sympathetic stress. J. Investig. Biochem. 2014, 3, 154. [Google Scholar] [CrossRef]
- Lenders, J.W.M.; Eisenhofer, G. Normetanephrine and Metanephrine. In Encyclopedia of Endocrine Diseases; Elsevier: Amsterdam, The Netherlands, 2017; pp. 420–424. [Google Scholar]
- Grillo, A.; Salvi, L.; Coruzzi, P.; Salvi, P.; Parati, G. Sodium Intake and Hypertension. Nutrients 2019, 11, 1970. [Google Scholar] [CrossRef]
- DeLalio, L.J.; Sved, A.F.; Stocker, S.D. Sympathetic Nervous System Contributions to Hypertension: Updates and Therapeutic Relevance. Can. J. Cardiol. 2020, 36, 712–720. [Google Scholar] [CrossRef]
| Parameter | LS | HS |
|---|---|---|
| N (W/M) | 47 (19/28) | |
| Age (years) | 21 ± 2 | |
| Anthropometric Parameters | ||
| BMI (kg/m2) | 23.9 ± 3.1 | 24.2 ± 3.2 |
| WHR | 0.82 ± 0.06 | 0.82 ± 0.05 |
| Hemodynamic Parameters | ||
| SBP (mmHg) | 116 ± 12 | 117 ± 14 |
| DBP (mmHg) | 73 ± 9 | 72 ± 8 |
| MAP (mmHg) | 87 ± 8 | 87 ± 8 |
| HR (beats per minute) | 82 ± 15 | 79 ± 13 |
| Serum Biochemical Parameters | ||
| urea (mmol/L) | 4.7 ± 1.3 | 4.7 ± 1.0 |
| creatinine (µmol/L) | 82 ± 14 | 77 ± 14 |
| sodium (mmol/L) | 138.4 ± 2.1 | 140.2 ± 2.5 * |
| potassium (mmol/L) | 4.0 ± 0.2 | 4.2 ± 0.3 * |
| calcium (mmol/L) | 2.47 ± 0.07 | 2.42 ± 0.09 * |
| iron (μmol/L) | 20.2 ± 7.5 | 19.2 ± 6.4 |
| transferrin (g/L) | 2.71 ± 0.32 | 2.59 ± 0.31 * |
| glucose (mmol/L) | 4.9 ± 0.5 | 4.9 ± 0.7 |
| hsCRP (mg/L) | 1.2 ± 2.4 | 0.9 ± 1.0 |
| 24-h Urine Biochemical Parameters | ||
| 24h urine volume (mL) | 1618 ± 654 | 1642 ± 657 |
| 24h creatinine coefficient (µmol/24 h/kg) | 180.5 ± 66.3 | 177.0 ± 47.0 |
| 24 h urine urea (mmol/dU) | 318.5 ± 196.2 | 293.0 ± 90.1 |
| 24 h urine protein (mg/dU) | 98.9 ± 53.1 | 100.6 ± 42.6 |
| 24 h urine albumin (mg/dU) | 10.4 ± 17.5 | 7.0 ±4.9 |
| 24 h sodium (mmol/dU) | 109.1 ± 51.6 | 247.6 ± 107.0 * |
| 24 h potassium (mmol/dU) | 46.0 ± 18.5 | 51.9 ± 19.9 |
| calculated salt intake (g/day) | 6.4 ± 3.0 | 14.5 ± 6.3 * |
| LS | HS | |
|---|---|---|
| N (W/M) | 23 (7/16) | |
| Dopamine, µmol/dU | 1.47 ± 0.050 | 1.40 ± 0.70 |
| Noradrenaline, µmol/dU | 0.71 ± 2.46 | 0.18 ± 0.08 * |
| Adrenaline, µmol/dU | 0.03 ± 0.02 | 0.02 ± 0.02 |
| Homovanillic acid, µmol/dU | 23.25 ± 7.95 | 21.08 ± 8.17 |
| Normetanephrine, µmol/dU | 1.05 ± 0.34 | 0.93 ± 0.34 * |
| Metanephrine, µmol/dU | 0.54 ± 0.16 | 0.45 ± 0.17 * |
| Vanillylmandelic acid, µmol/dU | 16.98 ± 5.80 | 15.68 ± 6.50 |
| Parameter | LS | HS |
|---|---|---|
| N (W/M) | 23 (7/16) | |
| Time Domain Analysis | ||
| Mean HR (bpm) | 68 ± 10 | 69 ± 9 |
| Mean RR (ms) | 902 ± 119 | 875 ± 103 |
| SDNN (ms) | 92 ± 32 | 79 ± 38 |
| RMS-SD (ms) | 82 ± 35 | 73 ± 43 |
| pNN50 (%) | 38 ± 18 | 35 ± 20 |
| Frequency Domain Analysis | ||
| TP (ms2/Hz) | 2755 ± 1995 | 1848 ± 1580 |
| VLF (ms2/Hz) | 843 ± 557 | 774 ± 669 |
| LF (ms2/HZ) | 1205 ± 1051 | 687 ± 666 * |
| HF (ms2/Hz) | 708 ± 683 | 598 ± 677 |
| LF/HF | 2.19 ± 1.40 | 1.77 ± 0.92 |
| Parameter | LS | LS+MST | LS | HS+MST |
|---|---|---|---|---|
| N (W/M) | 24 (12/12) | |||
| Time Domain Analysis | ||||
| Mean HR (bpm) | 73 ± 12 | 72 ± 10 | 75 ± 9 | 71 ± 10 |
| Mean RR (ms) | 844 ± 144 | 844 ± 119 | 813 ± 98 | 854 ± 114 |
| SDNN (ms) | 84 ± 43 | 79 ± 35 | 84 ± 43 | 93 ± 50 |
| RMS-SD (ms) | 82 ± 58 | 71 ± 44 | 76 ± 60 | 89 ± 68 |
| pNN50 (%) | 35 ± 24 | 34 ± 21 | 32 ± 25 | 37 ± 25 |
| Frequency Domain Analysis | ||||
| TP (ms2/Hz) | 2905 ± 3593 | 2111 ± 1954 | 2399 ± 2504 | 3314 ± 3842 |
| VLF (ms2/Hz) | 803 ± 726 | 716 ± 537 | 768 ± 715 | 908 ± 753 |
| LF (ms2/HZ) | 1086 ± 1331 | 888 ± 1121 | 813 ± 922 | 1344 ± 1801 |
| HF (ms2/Hz) | 1016 ± 1659 | 507 ± 400 | 818 ± 1172 | 1062 ± 1586 |
| LF/HF | 1.84 ± 1.38 | 2.72 ± 3.20 | 1.74 ± 1.02 | 1.88 ± 1.27 |
| Parameter | LS | LS+MST | HS | HS+MST |
|---|---|---|---|---|
| N (W/M) | 24 (12/12) | |||
| % of total power within frequency band | ||||
| Cardiac (0.6–2.0 Hz) | 6.39 ± 5.51 | 11.75 ± 11.46 | 5.58 ± 5.07 | 9.19 ± 9.58 |
| Respiratory (0.15–0.6 Hz) | 3.47 ± 3.28 | 6.84 ± 7.52 | 3.10 ± 3.25 | 5.86 ± 5.94 |
| Myogenic (0.05–0.15 Hz) | 3.65 ± 2.71 | 4.95 ± 4.95 | 3.09 ± 2.28 | 4.25 ± 3.43 |
| Sympathetic (0.02–0.05 Hz) | 14.40 ± 3.81 | 14.31 ± 4.60 | 14.59 ± 4.09 | 13.48 ± 4.40 |
| Endothelial (0.008–0.02 Hz) | 72.49 ± 9.99 | 61.92 ± 18.78 | 73.63 ± 9.66 | 67.86 ± 13.80 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Stupin, A.; Drenjančević, I.; Šušnjara, P.; Debeljak, Ž.; Kolobarić, N.; Jukić, I.; Mihaljević, Z.; Martinović, G.; Selthofer-Relatić, K. Is There Association between Altered Adrenergic System Activity and Microvascular Endothelial Dysfunction Induced by a 7-Day High Salt Intake in Young Healthy Individuals. Nutrients 2021, 13, 1731. https://doi.org/10.3390/nu13051731
Stupin A, Drenjančević I, Šušnjara P, Debeljak Ž, Kolobarić N, Jukić I, Mihaljević Z, Martinović G, Selthofer-Relatić K. Is There Association between Altered Adrenergic System Activity and Microvascular Endothelial Dysfunction Induced by a 7-Day High Salt Intake in Young Healthy Individuals. Nutrients. 2021; 13(5):1731. https://doi.org/10.3390/nu13051731
Chicago/Turabian StyleStupin, Ana, Ines Drenjančević, Petar Šušnjara, Željko Debeljak, Nikolina Kolobarić, Ivana Jukić, Zrinka Mihaljević, Goran Martinović, and Kristina Selthofer-Relatić. 2021. "Is There Association between Altered Adrenergic System Activity and Microvascular Endothelial Dysfunction Induced by a 7-Day High Salt Intake in Young Healthy Individuals" Nutrients 13, no. 5: 1731. https://doi.org/10.3390/nu13051731
APA StyleStupin, A., Drenjančević, I., Šušnjara, P., Debeljak, Ž., Kolobarić, N., Jukić, I., Mihaljević, Z., Martinović, G., & Selthofer-Relatić, K. (2021). Is There Association between Altered Adrenergic System Activity and Microvascular Endothelial Dysfunction Induced by a 7-Day High Salt Intake in Young Healthy Individuals. Nutrients, 13(5), 1731. https://doi.org/10.3390/nu13051731








