Brown Adipose Tissue: New Challenges for Prevention of Childhood Obesity. A Narrative Review
Abstract
1. Introduction
2. Materials and Methods
2.1. Topic of the Review
2.2. Methodology
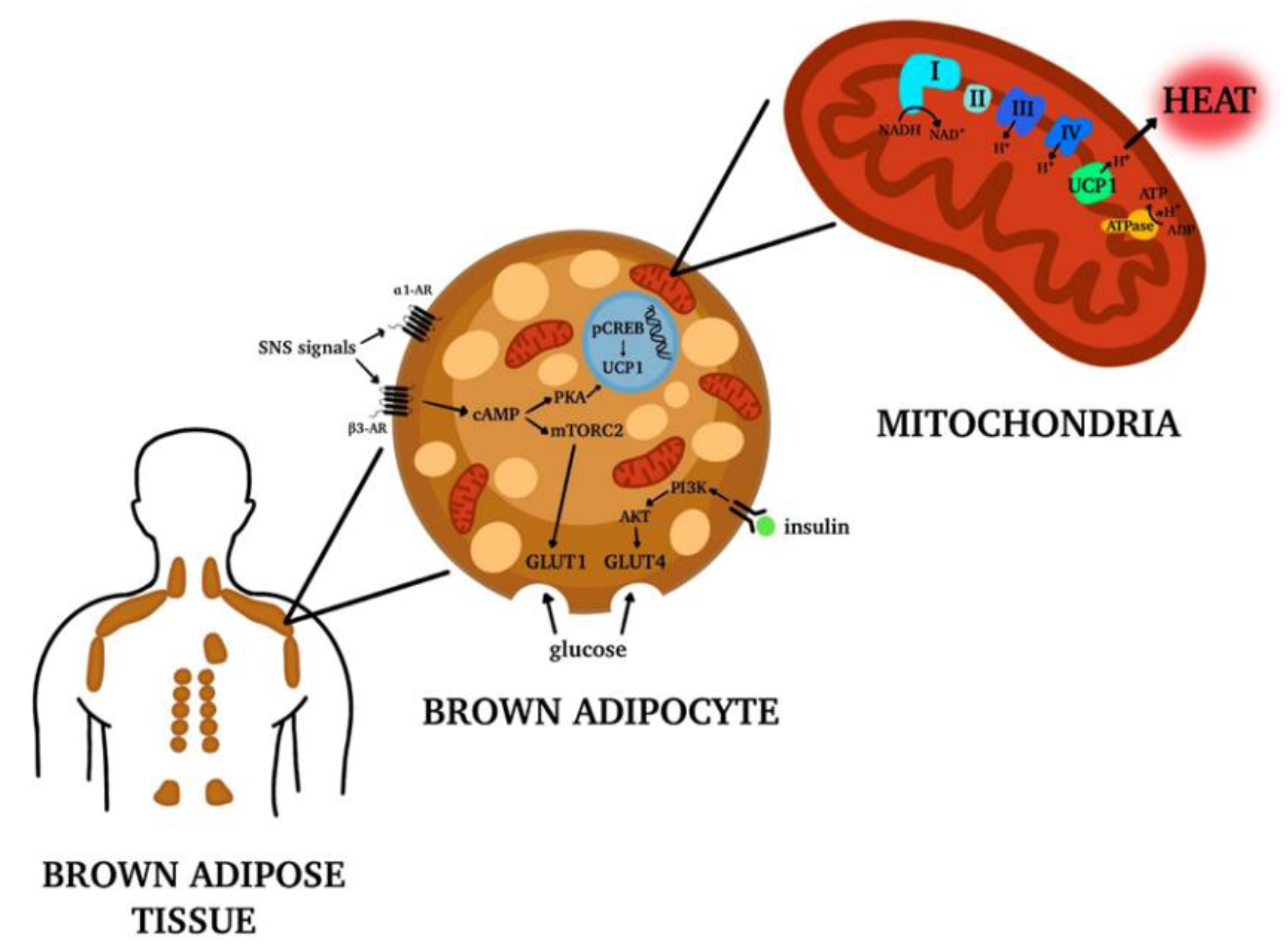
3. Brown Adipose Tissue (BAT): Localization and Functions
Brite or Beige Adipocytes
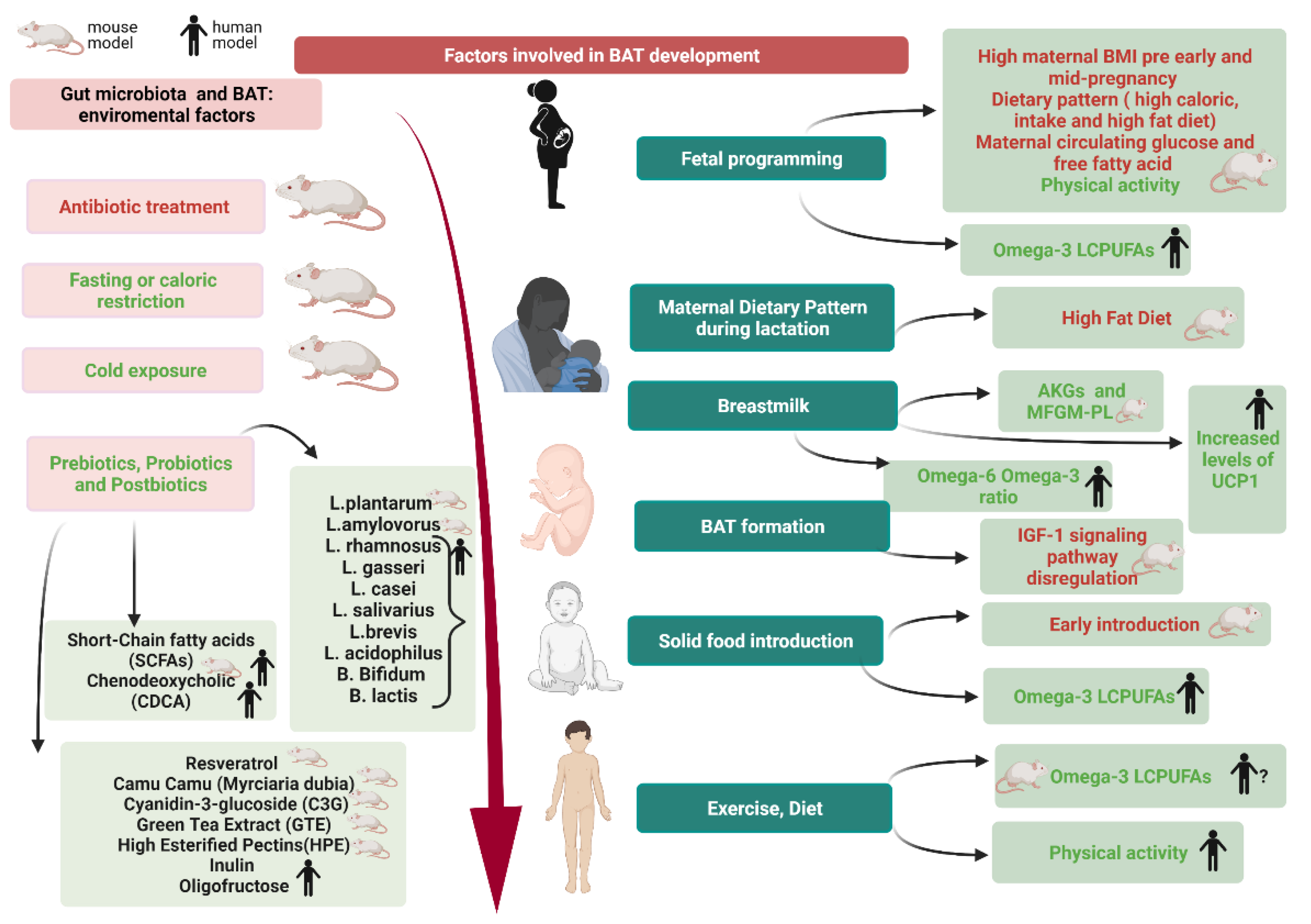
4. Pregnancy and BAT in Newborns: The Role of Fetal Programming
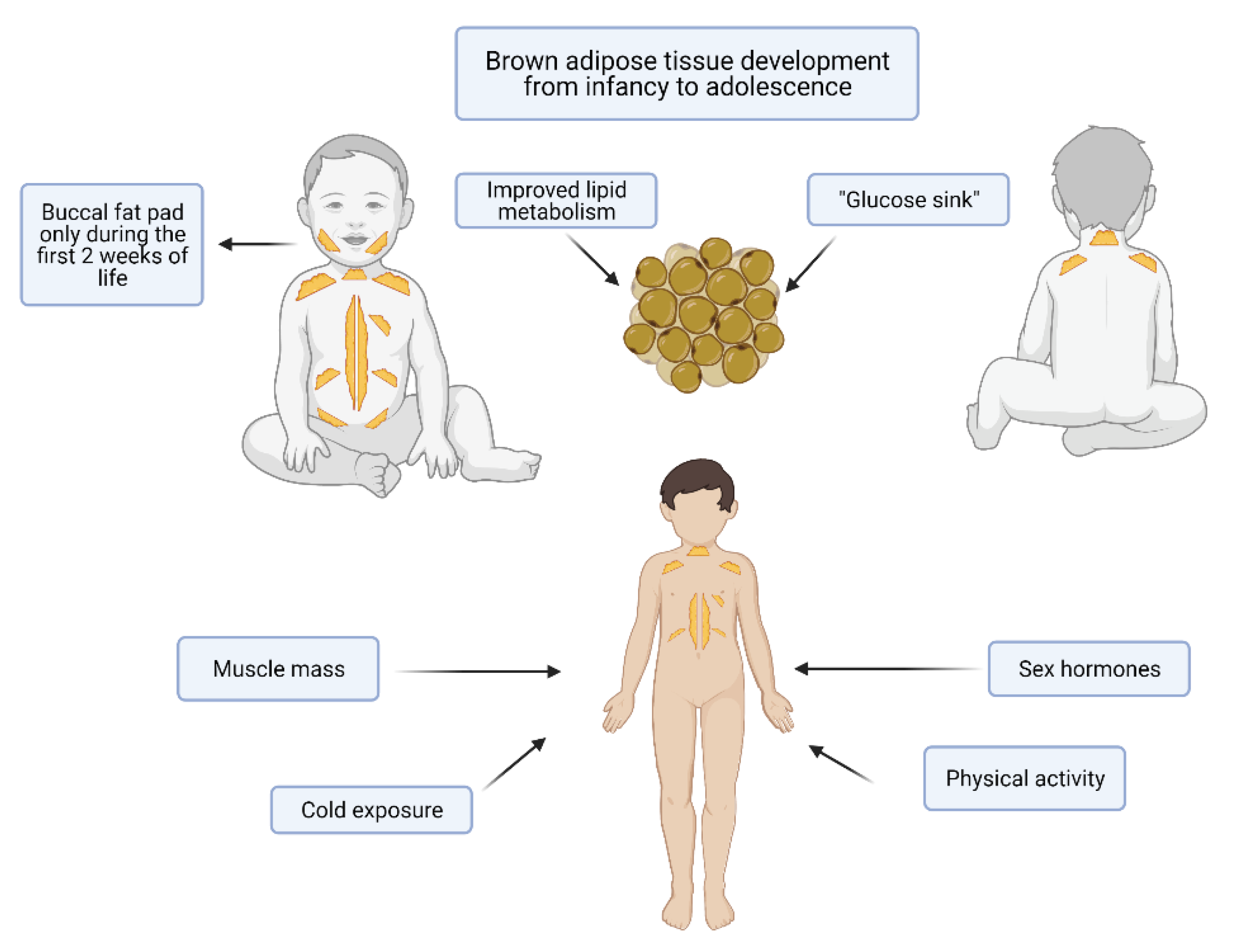
5. BAT in Term and Preterm Infants, Childhood and Adolescence
6. BAT Modulation
6.1. Breastfeeding and Solid Foods Introduction
6.2. Gut Microbiota and BAT
6.3. Environmental Factors Affecting Gut Microbiota and BAT
6.4. Modulating Factors of Gut Microbiota
6.4.1. Prebiotics
6.4.2. Probiotics
6.4.3. Postbiotics
6.5. Limitations of BAT Activation
7. Studies on Functional Nutrients on BAT: The Role of Omega-3 Fatty Acids
7.1. Effect of Omega-3 on WAT Function and Browning
7.2. Effect of Omega-3 on BAT Function
8. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Nobles, J.; Summerbell, C.; Brown, T.; Jago, R.; Moore, T. A Secondary Analysis of the Childhood Obesity Prevention Cochrane Review through a Wider Determinants of Health Lens: Implications for Research Funders, Researchers, Policymakers and Practitioners. Int. J. Behav. Nutr. Phys. Act. 2021, 18, 22. [Google Scholar] [CrossRef]
- Lauria, L.; Spinelli, A.; Buoncristiano, M.; Nardone, P. Decline of Childhood Overweight and Obesity in Italy from 2008 to 2016: Results from 5 Rounds of the Population-Based Surveillance System. Bmc. Public Health 2019, 19, 618. [Google Scholar] [CrossRef]
- An, R.; Shi, Y.; Shen, J.; Bullard, T.; Liu, G.; Yang, Q.; Chen, N.; Cao, L. Effect of Front-of-Package Nutrition Labeling on Food Purchases: A Systematic Review. Public Health 2021, 191, 59–67. [Google Scholar] [CrossRef]
- World Health Organization Regional Office for Europe. Mapping the Health System Response to Childhood Obesity in the WHO European Region. An Overview and Country Perspectives; World Health Organization: Copenhagen, Denmark, 2019. [Google Scholar]
- Coppack, S.W. Adipose Tissue Changes in Obesity. Biochem. Soc. Trans. 2005, 33 Pt 5, 1049–1052. [Google Scholar] [CrossRef]
- Greenberg, A.S.; Obin, M.S. Obesity and the Role of Adipose Tissue in Inflammation and Metabolism. Am. J. Clin. Nutr. 2006, 83, 461S–465S. [Google Scholar] [CrossRef] [PubMed]
- Fenzl, A.; Kiefer, F.W. Brown Adipose Tissue and Thermogenesis. Horm. Mol. Biol. Clin. Investig. 2014, 19, 25–37. [Google Scholar] [CrossRef] [PubMed]
- Klepac, K.; Georgiadi, A.; Tschöp, M.; Herzig, S. The Role of Brown and Beige Adipose Tissue in Glycaemic Control. Mol. Asp. Med. 2019, 68, 90–100. [Google Scholar] [CrossRef] [PubMed]
- Villarroya, F.; Cereijo, R.; Villarroya, J.; Giralt, M. Brown Adipose Tissue as a Secretory Organ. Nat. Rev. Endocrinol. 2017, 13, 26–35. [Google Scholar] [CrossRef]
- Speakman, J.R. Fifty Shades of Brown: The Functions, Diverse Regulation and Evolution of Brown Adipose Tissue. Mol. Asp. Med. 2019, 68, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Boström, P.; Sparks, L.M.; Ye, L.; Choi, J.H.; Giang, A.H.; Khandekar, M.; Virtanen, K.A.; Nuutila, P.; Schaart, G.; et al. Beige adipocytes are a distinct type of thermogenic fat cell in mouse and human. Cell 2012, 150, 366–376. [Google Scholar] [CrossRef] [PubMed]
- Zoico, E.; Rubele, S.; De Caro, A.; Nori, N.; Mazzali, G.; Fantin, F.; Rossi, A.; Zamboni, M. Brown and Beige Adipose Tissue and Aging. Front. Endocrinol. 2019, 10, 368. [Google Scholar] [CrossRef] [PubMed]
- Symonds, M.E.; Pope, M.; Budge, H. The Ontogeny of Brown Adipose Tissue. Annu. Rev. Nutr. 2015, 35, 295–320. [Google Scholar] [CrossRef] [PubMed]
- Symonds, M.E. Brown Adipose Tissue Growth and Development. Scientifica 2013, 2013, 305763. [Google Scholar] [CrossRef]
- Symonds, M.E.; Bloor, I.; Ojha, S.; Budge, H. The Placenta, Maternal Diet and Adipose Tissue Development in the Newborn. Ann. Nutr. Metab. 2017, 70, 232–235. [Google Scholar] [CrossRef]
- Symonds, M.E.; Bird, J.A.; Clarke, L.; Gate, J.J.; Lomax, M.A. Nutrition, Temperature and Homeostasis during Perinatal Development. Exp. Physiol. 1995, 80, 907–940. [Google Scholar] [CrossRef]
- Symonds, M.E.; Aldiss, P.; Dellschaft, N.; Law, J.; Fainberg, H.P.; Pope, M.; Sacks, H.; Budge, H. Brown Adipose Tissue Development and Function and Its Impact on Reproduction. J. Endocrinol. 2018, 238, R53–R62. [Google Scholar] [CrossRef]
- Symonds, M.E.; Stephenson, T.; Gardner, D.S.; Budge, H. Long-Term Effects of Nutritional Programming of the Embryo and Fetus: Mechanisms and Critical Windows. Reprod. Fertil. Dev. 2006, 19, 53–63. [Google Scholar] [CrossRef] [PubMed]
- Symonds, M.E.; Sebert, S.P.; Budge, H. The Impact of Diet during Early Life and Its Contribution to Later Disease: Critical Checkpoints in Development and Their Long-Term Consequences for Metabolic Health: Symposium on ‘Early Nutrition and Later Disease: Current Concepts, Research and Implicat. Proc. Nutr. Soc. 2009, 68, 416–421. [Google Scholar] [CrossRef] [PubMed]
- Kincaid, H.J.; Nagpal, R.; Yadav, H. Microbiome-immune-metabolic axis in the epidemic of childhood obesity: Evidence and opportunities. Obes. Rev. 2021, 21, 1–25. [Google Scholar] [CrossRef] [PubMed]
- Retnakaran, R.; Ye, C.; Hanley, A.J.G.; Connelly, P.W.; Sermer, M.; Zinman, B.; Hamilton, J.K. Effect of Maternal Weight, Adipokines, Glucose Intolerance and Lipids on Infant Birth Weight among Women without Gestational Diabetes Mellitus. Can. Med. Assoc. J. 2012, 184, 1353–1360. [Google Scholar] [CrossRef] [PubMed]
- Starling, A.P.; Brinton, J.T.; Glueck, D.H.; Shapiro, A.L.; Harrod, C.S.; Lynch, A.M.; Siega-Riz, A.M.; Dabelea, D. Associations of Maternal BMI and Gestational Weight Gain with Neonatal Adiposity in the Healthy Start Study. Am. J. Clin. Nutr. 2015, 101, 302–309. [Google Scholar] [CrossRef]
- Collado, M.C.; Isolauri, E.; Laitinen, K.; Salminen, S. Effect of Mother’s Weight on Infant’s Microbiota Acquisition, Composition, and Activity during Early Infancy: A Prospective Follow-up Study Initiated in Early Pregnancy. Am. J. Clin. Nutr. 2010, 92, 1023–1030. [Google Scholar] [CrossRef]
- Yu, Z.; Han, S.; Zhu, J.; Sun, X.; Ji, C.; Guo, X. Pre-Pregnancy Body Mass Index in Relation to Infant Birth Weight and Offspring Overweight/Obesity: A Systematic Review and Meta-Analysis. PLoS ONE 2013, 8, e61627. [Google Scholar] [CrossRef]
- Wang, H.; Chen, Y.; Mao, X.; Du, M. Maternal Obesity Impairs Fetal Mitochondriogenesis and Brown Adipose Tissue Development Partially via Upregulation of MiR-204-5p. Biochim. Biophys. Acta Mol. Basis Dis. 2019, 1865, 2706–2715. [Google Scholar] [CrossRef] [PubMed]
- Li, C.; Goran, M.; Kaur, H.; Nollen, N.; Ahluwalia, J. Developmental Trajectories of Overweight During Childhood: Role of Early Life Factors. Obes. (Silver Springmd.) 2007, 15, 760–771. [Google Scholar] [CrossRef]
- Raipuria, M.; Bahari, H.; Morris, M. Effects of Maternal Diet and Exercise during Pregnancy on Glucose Metabolism in Skeletal Muscle and Fat of Weanling Rats. PLoS ONE 2015, 10, e0120980. [Google Scholar] [CrossRef] [PubMed]
- Galley, J.D.; Bailey, M.; Kamp Dush, C.; Schoppe-Sullivan, S.; Christian, L.M. Maternal Obesity Is Associated with Alterations in the Gut Microbiome in Toddlers. PLoS ONE 2014, 9, e113026. [Google Scholar] [CrossRef]
- Claycombe, K.J.; Vomhof-DeKrey, E.E.; Roemmich, J.N.; Rhen, T.; Ghribi, O. Maternal Low-Protein Diet Causes Body Weight Loss in Male, Neonate Sprague–Dawley Rats Involving UCP-1-Mediated Thermogenesis. J. Nutr. Biochem. 2015, 26, 729–735. [Google Scholar] [CrossRef]
- Ma, J.; Prince, A.L.; Bader, D. High-Fat Maternal Diet during Pregnancy Persistently Alters the Offspring Microbiome in a Primate Model. Nat. Commun. 2014, 5. [Google Scholar] [CrossRef]
- Chu, D.M.; Antony, K.M.; Ma, J. The Early Infant Gut Microbiome Varies in Association with a Maternal High-Fat Diet. Genome Med. 2016, 8, 77. [Google Scholar] [CrossRef]
- Almeida, M.M.; Dias-Rocha, C.P.; Souza, A.S.; Muros, M.F.; Mendonca, L.S.; Pazos-Moura, C.C.; Trevenzoli, I.H. Perinatal Maternal High-Fat Diet Induces Early Obesity and Sex-Specific Alterations of the Endocannabinoid System in White and Brown Adipose Tissue of Weanling Rat Offspring. Br. J. Nutr. 2017, 118, 788–803. [Google Scholar] [CrossRef] [PubMed]
- Lawlor, D.A.; Smith, G.D.; O’Callaghan, M.; Alati, R.; Mamun, A.A.; Williams, G.M.; Najman, J.M. Epidemiologic Evidence for the Fetal Overnutrition Hypothesis: Findings from the Mater-University Study of Pregnancy and Its Outcomes. Am. J. Epidemiol. 2007, 165, 418–424. [Google Scholar] [CrossRef] [PubMed]
- Salomäki, H.; Heinäniemi, M.; Vähätalo, L.H.; Ailanen, L.; Eerola, K.; Ruohonen, S.T.; Pesonen, U.; Koulu, M. Prenatal Metformin Exposure in a Maternal High Fat Diet Mouse Model Alters the Transcriptome and Modifies the Metabolic Responses of the Offspring. PLoS ONE 2014, 9, e115778. [Google Scholar] [CrossRef] [PubMed]
- Vanky, E.; Zahlsen, K.; Spigset, O.; Carlsen, S.M. Placental Passage of Metformin in Women with Polycystic Ovary Syndrome. Fertil. Steril. 2005, 83, 1575–1578. [Google Scholar] [CrossRef] [PubMed]
- Chiswick, C.; Reynolds, R.; Denison, F.; Drake, A.; Forbes, S.; Newby, D.; Walker, B.; Quenby, S.; Wray, S.; Weeks, A.; et al. Effect of Metformin on Maternal and Fetal Outcomes in Obese Pregnant Women (EMPOWaR): A Randomised, Double-Blind, Placebo-Controlled Trial. Lancet. Diabetes Endocrinol. 2015, 3. [Google Scholar] [CrossRef]
- Whitaker, R.C.; Dietz, W.H. Role of the Prenatal Environment in the Development of Obesity. J. Pediatrics 1998, 132, 768–776. [Google Scholar] [CrossRef]
- Martino, J.; Sebert, S.; Segura, M.T.; Garcia-Valdés, L.; Florido, J.; Padilla, M.C.; Marcos, A.; Rueda, R.; McArdle, H.J.; Budge, H.; et al. Maternal Body Weight and Gestational Diabetes Differentially Influence Placental and Pregnancy Outcomes. J. Clin. Endocrinol. Metab. 2016, 101, 59–68. [Google Scholar] [CrossRef]
- Keski-Nisula, L.; Kyynäräinen, H.-R.; Kärkkäinen, U.; Karhukorpi, J.; Heinonen, S.; Pekkanen, J. Maternal Intrapartum Antibiotics and Decreased Vertical Transmission of Lactobacillus to Neonates during Birth. Acta Paediatr. 2013, 102, 480–485. [Google Scholar] [CrossRef]
- Cassidy-Bushrow, A.E.; Burmeister, C.; Havstad, S.; Levin, A.M.; Lynch, S.V.; Ownby, D.R.; Rundle, A.G.; Woodcroft, K.J.; Zoratti, E.M.; Johnson, C.C.; et al. Prenatal Antimicrobial Use and Early-Childhood Body Mass Index. Int. J. Obes. 2018, 42, 1–7. [Google Scholar] [CrossRef]
- Mueller, N.T.; Whyatt, R.; Hoepner, L.; Oberfield, S.; Dominguez-Bello, M.G.; Widen, E.M.; Hassoun, A.; Perera, F.; Rundle, A. Prenatal Exposure to Antibiotics, Cesarean Section and Risk of Childhood Obesity. Int. J. obes. 2015, 39, 665–670. [Google Scholar] [CrossRef]
- Coscia, A.; Bardanzellu, F.; Caboni, E.; Fanos, V.; Peroni, D.G. When a Neonate Is Born, So Is a Microbiota. Life (Baseline.) 2021, 11, 148. [Google Scholar] [CrossRef] [PubMed]
- Kalliomäki, M.; Carmen Collado, M.; Salminen, S.; Isolauri, E. Early Differences in Fecal Microbiota Composition in Children May Predict Overweight. Am. J. Clin. Nutr. 2008, 87, 534–538. [Google Scholar] [CrossRef] [PubMed]
- Sandall, J.; Tribe, R.M.; Avery, L.; Mola, G.; Visser, G.H.; Homer, C.S.; Gibbons, D.; Kelly, N.M.; Kennedy, H.P.; Kidanto, H.; et al. Short-Term and Long-Term Effects of Caesarean Section on the Health of Women and Children. Lancet 2018, 392, 1349–1357. [Google Scholar] [CrossRef]
- Dhana, K.; Rd, J.; Liu, G.; Zhang, C.; Wang, X.; Field, A.; Chavarro, J.; Sun, Q. Association between Maternal Adherence to Healthy Lifestyle Practices and Risk of Obesity in Offspring: Results from Two Prospective Cohort Studies of Mother-Child Pairs in the United States. BMJ 2018, 362, k2486. [Google Scholar] [CrossRef]
- Mourtakos, S.P.; Tambalis, K.D.; Panagiotakos, D.B.; Antonogeorgos, G.; Arnaoutis, G.; Karteroliotis, K.; Sidossis, L.S. Maternal Lifestyle Characteristics during Pregnancy, and the Risk of Obesity in the Offspring: A Study of 5,125 Children. BMC Pregnancy Childbirth 2015, 15, 66. [Google Scholar] [CrossRef] [PubMed]
- Philips, E.M.; Santos, S.; Trasande, L.; Aurrekoetxea, J.J.; Barros, H.; von Berg, A.; Bergström, A.; Bird, P.K.; Brescianini, S.; Ní Chaoimh, C.; et al. Changes in Parental Smoking during Pregnancy and Risks of Adverse Birth Outcomes and Childhood Overweight in Europe and North America: An Individual Participant Data Meta-Analysis of 229,000 Singleton Births. PLoS Med. 2020, 17, e1003182. [Google Scholar] [CrossRef] [PubMed]
- Rzehak, P.; Saffery, R.; Reischl, E.; Covic, M.; Wahl, S.; Grote, V.; Xhonneux, A.; Langhendries, J.-P.; Ferre, N.; Closa-Monasterolo, R.; et al. European Childhood Obesity Trial Study group. Maternal Smoking during Pregnancy and DNA-Methylation in Children at Age 5.5 Years: Epigenome-Wide-Analysis in the European Childhood Obesity Project (CHOP)-Study. PLoS ONE 2016, 11, e0155554. [Google Scholar] [CrossRef]
- Tsai, P.-C.; Glastonbury, C.A.; Eliot, M.N.; Bollepalli, S.; Yet, I.; Castillo-Fernandez, J.E.; Carnero-Montoro, E.; Hardiman, T.; Martin, T.C.; Vickers, A.; et al. Smoking Induces Coordinated DNA Methylation and Gene Expression Changes in Adipose Tissue with Consequences for Metabolic Health. Clin. Epigenetics 2018, 10, 126. [Google Scholar] [CrossRef]
- Son, J.S.; Zhao, L.; Chen, Y.; Chen, K.; Chae, S.A.; de Avila, J.M.; Wang, H.; Zhu, M.-J.; Jiang, Z.; Du, M. Maternal Exercise via Exerkine Apelin Enhances Brown Adipogenesis and Prevents Metabolic Dysfunction in Offspring Mice. Sci. Adv. 2020, 6, eaaz0359. [Google Scholar] [CrossRef]
- Bae-Gartz, I.; Janoschek, R.; Kloppe, C.S.; Vohlen, C.; Roels, F.; Oberthür, A.; Alejandre Alcazar, M.A.; Lippach, G.; Muether, P.S.; Dinger, K.; et al. Running Exercise in Obese Pregnancies Prevents IL-6 Trans-Signaling in Male Offspring. Med. Sci. Sports Exerc. 2016, 48, 829–838. [Google Scholar] [CrossRef]
- Calkins, K.; Devaskar, S.U. Fetal Origins of Adult Disease. Curr. Probl. Pediatric Adolesc. Health Care 2011, 41, 158–176. [Google Scholar] [CrossRef] [PubMed]
- Maric-Bilkan, C.; Symonds, M.; Ozanne, S.; Alexander, B.T. Impact of Maternal Obesity and Diabetes on Long-Term Health of the Offspring. Exp. Diabetes Res. 2011, 2011, 2–3. [Google Scholar] [CrossRef] [PubMed]
- Lidell, M.E. Brown Adipose Tissue in Human Infants BT—Brown Adipose Tissue; Pfeifer, A., Klingenspor, M., Herzig, S., Eds.; Springer International Publishing: Cham, Switzerland, 2019. [Google Scholar] [CrossRef]
- Benador, I.Y.; Veliova, M.; Mahdaviani, K.; Petcherski, A.; Wikstrom, J.D.; Assali, E.A.; Acín-Pérez, R.; Shum, M.; Oliveira, M.F.; Cinti, S.; et al. Mitochondria Bound to Lipid Droplets Have Unique Bioenergetics, Composition, and Dynamics That Support Lipid Droplet Expansion. Cell Metab. 2018, 27, 869–885. [Google Scholar] [CrossRef]
- Harms, M.; Seale, P. Brown and Beige Fat: Development, Function and Therapeutic Potential. Nat. Med. 2013, 19, 1252–1263. [Google Scholar] [CrossRef] [PubMed]
- Kooijman, S.; van den Heuvel, J.K.; Rensen, P.C.N. Neuronal Control of Brown Fat Activity. Trends Endocrinol. Metab. 2015, 26, 657–668. [Google Scholar] [CrossRef]
- Symonds, M.E.; Pope, M.; Bloor, I.; Law, J.; Alagal, R.; Budge, H. Adipose Tissue Growth and Development: The Modulating Role of Ambient Temperature. J. Endocrinol. 2021, 248, R19–R28. [Google Scholar] [CrossRef]
- Silva, J.E. Thermogenic Mechanisms and Their Hormonal Regulation. Physiol. Rev. 2006, 86, 435–464. [Google Scholar] [CrossRef]
- de Jesus, L.A.; Carvalho, S.D.; Ribeiro, M.O.; Schneider, M.; Kim, S.-W.; Harney, J.W.; Larsen, P.R.; Bianco, A.C. The Type 2 Iodothyronine Deiodinase Is Essential for Adaptive Thermogenesis in Brown Adipose Tissue. J. Clin. Investig. 2001, 108, 1379–1385. [Google Scholar] [CrossRef]
- Sampath, S.C.; Sampath, S.C.; Bredella, M.A.; Cypess, A.M.; Torriani, M. Imaging of Brown Adipose Tissue: State of the Art. Radiology 2016, 280, 4–19. [Google Scholar] [CrossRef]
- Oelkrug, R.; Polymeropoulos, E.T.; Jastroch, M. Brown Adipose Tissue: Physiological Function and Evolutionary Significance. J. Comp. Physiol. B 2015, 185, 587–606. [Google Scholar] [CrossRef]
- Hui, S.C.N.; Ko, J.K.L.; Zhang, T.; Shi, L.; Yeung, D.K.W.; Wang, D.; Chan, Q.; Chu, W.C.W. Quantification of Brown and White Adipose Tissue Based on Gaussian Mixture Model Using Water–Fat and MRI in Adolescents. J. Magn. Reson. Imaging 2017, 46, 758–768. [Google Scholar] [CrossRef]
- Ponrartana, S.; Hu, H.H.; Gilsanz, V. On the Relevance of Brown Adipose Tissue in Children. Ann. N. Y. Acad. Sci. 2013, 1302, 24–29. [Google Scholar] [CrossRef] [PubMed]
- Ponrartana, S.; Patil, S.; Aggabao, P.C.; Pavlova, Z.; Devaskar, S.U.; Gilsanz, V. Brown Adipose Tissue in the Buccal Fat Pad during Infancy. PLoS ONE 2014, 9, e89533. [Google Scholar] [CrossRef] [PubMed]
- Plagge, A.; Gordon, E.; Dean, W.; Boiani, R.; Cinti, S.; Peters, J.; Kelsey, G. The Imprinted Signaling Protein XLαs Is Required for Postnatal Adaptation to Feeding. Nat. Genet. 2004, 36, 818–826. [Google Scholar] [CrossRef] [PubMed]
- Negron, S.G.; Ercan-Sencicek, A.G.; Freed, J.; Walters, M.; Lin, Z. Both Proliferation and Lipogenesis of Brown Adipocytes Contribute to Postnatal Brown Adipose Tissue Growth in Mice. Sci. Rep. 2020, 10, 20335. [Google Scholar] [CrossRef] [PubMed]
- Rui, L. Brown and Beige Adipose Tissues in Health and Disease. Compr. Physiol. 2017, 7, 1281–1306. [Google Scholar] [CrossRef] [PubMed]
- Kistner, A.; Rydén, H.; Anderstam, B.; Hellström, A.; Skorpil, M. Brown Adipose Tissue in Young Adults Who Were Born Preterm or Small for Gestational Age. J. Pediatric Endocrinol. Metab. 2018, 31, 641–647. [Google Scholar] [CrossRef] [PubMed]
- Boucher, J.; Mori, M.A.; Lee, K.Y.; Smyth, G.; Liew, C.W.; Macotela, Y.; Rourk, M.; Bluher, M.; Russell, S.J.; Kahn, C.R. Impaired Thermogenesis and Adipose Tissue Development in Mice with Fat-Specific Disruption of Insulin and IGF-1 Signalling. Nat. Commun. 2012, 3, 902. [Google Scholar] [CrossRef] [PubMed]
- Ponrartana, S.; Aggabao, P.C.; Chavez, T.A.; Dharmavaram, N.L.; Gilsanz, V. Changes in Brown Adipose Tissue and Muscle Development during Infancy. J. Pediatrics 2016, 173, 116–121. [Google Scholar] [CrossRef] [PubMed]
- Gilsanz, V.; Hu, H.H.; Kajimura, S. Relevance of Brown Adipose Tissue in Infancy and Adolescence. Pediatric Res. 2013, 73, 3–9. [Google Scholar] [CrossRef] [PubMed]
- Gilsanz, V.; Chung, S.A.; Jackson, H.; Dorey, F.J.; Hu, H.H. Functional Brown Adipose Tissue Is Related to Muscle Volume in Children and Adolescents. J. Pediatrics 2011, 158, 722–726. [Google Scholar] [CrossRef] [PubMed]
- Farmer, S.R. Brown Fat and Skeletal Muscle: Unlikely Cousins? Cell 2008, 134, 726–727. [Google Scholar] [CrossRef] [PubMed]
- Schulz, T.J.; Huang, T.L.; Tran, T.T.; Zhang, H.; Townsend, K.L.; Shadrach, J.L.; Cerletti, M.; McDougall, L.E.; Giorgadze, N.; Tchkonia, T.; et al. Identification of Inducible Brown Adipocyte Progenitors Residing in Skeletal Muscle and White Fat. Proc. Natl. Acad. Sci. USA 2011, 108, 143–148. [Google Scholar] [CrossRef] [PubMed]
- Dinas, P.C.; Nikaki, A.; Jamurtas, A.Z.; Prassopoulos, V.; Efthymiadou, R.; Koutedakis, Y.; Georgoulias, P.; Flouris, A.D. Association between Habitual Physical Activity and Brown Adipose Tissue Activity in Individuals Undergoing PET-CT Scan. Clin. Endocrinol. (Oxf.) 2015, 82, 147–154. [Google Scholar] [CrossRef] [PubMed]
- Widmann, M.; Nieß, A.M.; Munz, B. Physical Exercise and Epigenetic Modifications in Skeletal Muscle. Sports Med. 2019, 49, 509–523. [Google Scholar] [CrossRef] [PubMed]
- Boström, P.; Wu, J.; Jedrychowski, M.P.; Korde, A.; Ye, L.; Lo, J.C.; Rasbach, K.A.; Boström, E.A.; Choi, J.H.; Long, J.Z.; et al. A PGC1-α-Dependent Myokine That Drives Brown-Fat-like Development of White Fat and Thermogenesis. Nature 2012, 481, 463–468. [Google Scholar] [CrossRef]
- Rodriguez-Cuenca, S.; Monjo, M.; Frontera, M.; Gianotti, M.; Proenza, A.M.; Roca, P. Sex Steroid Receptor Expression Profile in Brown Adipose Tissue. Effects of Hormonal Status. Cell. Physiol. Biochem. 2007, 20, 877–886. [Google Scholar] [CrossRef]
- Betz, M.J.; Enerbäck, S. Targeting Thermogenesis in Brown Fat and Muscle to Treat Obesity and Metabolic Disease. Nat. Rev. Endocrinol 2018, 14, 77–87. [Google Scholar] [CrossRef]
- Fuller-Jackson, J.-P.; Dordevic, A.L.; Clarke, I.J.; Henry, B.A. Effect of Sex and Sex Steroids on Brown Adipose Tissue Heat Production in Humans. Eur. J. Endocrinol. 2020, 183, 343–355. [Google Scholar] [CrossRef]
- Ponrartana, S.; Aggabao, P.C.; Hu, H.H.; Aldrovandi, G.M.; Wren, T.A.L.; Gilsanz, V. Brown Adipose Tissue and Its Relationship to Bone Structure in Pediatric Patients. J. Clin. Endocrinol. Metab. 2012, 97, 2693–2698. [Google Scholar] [CrossRef]
- Zanotti, S.; Stadmeyer, L.; Smerdel-Ramoya, A.; Durant, D.; Canalis, E. Misexpression of CCAAT/Enhancer Binding Protein Beta Causes Osteopenia. J. Endocrinol. 2009, 201, 263–274. [Google Scholar] [CrossRef]
- Douglas, R.J. Could a Lowered Level of Uncoupling Protein in Brown Adipose Tissue Mitochondria Play a Role in SIDS Aetiology? Med. Hypotheses 1992, 37, 100–102. [Google Scholar] [CrossRef]
- Fatemi, A.; Item, C.; Stöckler-Ipsiroglu, S.; Ipsiroglu, O.; Sperl, W.; Patsch, W.; Strobl, W. Sudden Infant Death: No Evidence for Linkage to Common Polymorphisms in the Uncoupling Protein-1 and the Β3-Adrenergic Receptor Genes. Eur. J. Pediatrics 2002, 161, 337–339. [Google Scholar] [CrossRef]
- Furlong, M.A.; Fanburg–Smith, J.C.; Miettinen, M. The Morphologic Spectrum of Hibernoma: A Clinicopathologic Study of 170 Cases. Am. J. Surg. Pathol. 2001, 25, 809–814. [Google Scholar] [CrossRef]
- Klevos, G.; Jose, J.; Pretell-Mazzini, J.; Conway, S. Hibernoma. Am. J. Orthop. (Belle Mead Nj) 2015, 44, 284–287. [Google Scholar] [PubMed]
- Beals, C.; Rogers, A.; Wakely, P.; Mayerson, J.L.; Scharschmidt, T.J. Hibernomas: A Single-Institution Experience and Review of Literature. Med. Oncol 2014, 31, 769. [Google Scholar] [CrossRef] [PubMed]
- Aldiss, P.; Dellschaft, N.S.; Sacks, H.; Budge, H.; Symonds, M.E. Beyond Obesity—Thermogenic Adipocytes and Cardiometabolic Health. Horm. Mol. Biol. Clin. Investig. 2017, 31, 2. [Google Scholar] [CrossRef] [PubMed]
- Symonds, M.E.; Aldiss, P.; Pope, M.; Budge, H. Recent Advances in Our Understanding of Brown and Beige Adipose Tissue: The Good Fat That Keeps You Healthy. F1000Research 2018, 7, F1000 Faculty Rev-1129. [Google Scholar] [CrossRef]
- Dallner, O.S.; Chernogubova, E.; Brolinson, K.A.; Bengtsson, T. Beta3-Adrenergic Receptors Stimulate Glucose Uptake in Brown Adipocytes by Two Mechanisms Independently of Glucose Transporter 4 Translocation. Endocrinology 2006, 147, 5730–5739. [Google Scholar] [CrossRef] [PubMed]
- Olsen, J.M.; Sato, M.; Dallner, O.S.; Sandström, A.L.; Pisani, D.F.; Chambard, J.-C.; Amri, E.-Z.; Hutchinson, D.S.; Bengtsson, T. Glucose Uptake in Brown Fat Cells Is Dependent on MTOR Complex 2-Promoted GLUT1 Translocation. J. Cell Biol. 2014, 207, 365–374. [Google Scholar] [CrossRef]
- Chernogubova, E.; Cannon, B.; Bengtsson, T. Norepinephrine Increases Glucose Transport in Brown Adipocytes via Beta3-Adrenoceptors through a CAMP, PKA, and PI3-Kinase-Dependent Pathway Stimulating Conventional and Novel PKCs. Endocrinology 2004, 145, 269–280. [Google Scholar] [CrossRef]
- Berbée, J.F.P.; Boon, M.R.; Khedoe, P.P.S.J. Brown Fat Activation Reduces Hypercholesterolaemia and Protects from Atherosclerosis Development. Nat. Commun. 2015, 6, 6356. [Google Scholar] [CrossRef]
- Chondronikola, M.; Volpi, E.; Børsheim, E. Brown Adipose Tissue Improves Whole-Body Glucose Homeostasis and Insulin Sensitivity in Humans. Diabetes 2014, 63, 4099. [Google Scholar] [CrossRef]
- Chang, L.; Villacorta, L.; Li, R.; Hamblin, M.; Xu, W.; Dou, C.; Zhang, J.; Wu, J.; Zeng, R.; Chen, Y.E. Loss of Perivascular Adipose Tissue on Peroxisome Proliferator-Activated Receptor-γ Deletion in Smooth Muscle Cells Impairs Intravascular Thermoregulation and Enhances Atherosclerosis. Circulation 2012, 126, 1067–1078. [Google Scholar] [CrossRef]
- Robinson, L.; Ojha, S.; Symonds, M.; Budge, H. Body Mass Index as a Determinant of Brown Adipose Tissue Function in Healthy Children. J. Pediatrics 2014, 164, 318–322.e1. [Google Scholar] [CrossRef]
- Ouellet, V.; Routhier-Labadie, A.; Bellemare, W.; Lakhal-Chaieb, L.; Turcotte, E.; Carpentier, A.C.; Richard, D. Outdoor Temperature, Age, Sex, Body Mass Index, and Diabetic Status Determine the Prevalence, Mass, and Glucose-Uptake Activity of 18F-FDG-Detected BAT in Humans. J. Clin. Endocrinol. Metab. 2011, 96, 192–199. [Google Scholar] [CrossRef]
- Entringer, S.; Rasmussen, J.; Cooper, D.M.; Ikenoue, S.; Waffarn, F.; Wadhwa, P.D.; Buss, C. Association between Supraclavicular Brown Adipose Tissue Composition at Birth and Adiposity Gain from Birth to 6 Months of Age. Pediatric Res. 2017, 82, 1017–1021. [Google Scholar] [CrossRef]
- Koontz, M.B.; Gunzler, D.D.; Presley, L.; Catalano, P.M. Longitudinal Changes in Infant Body Composition: Association with Childhood Obesity. Pediatric Obes. 2014, 9, e141–e144. [Google Scholar] [CrossRef]
- Betz, M.J.; Enerbäck, S. Human Brown Adipose Tissue: What We Have Learned So Far. Diabetes 2015, 64, 2360. [Google Scholar] [CrossRef]
- Law, J.M.; Morris, D.E.; Astle, V. Brown Adipose Tissue Response to Cold Stimulation Is Reduced in Girls with Autoimmune Hypothyroidism. J. Endocr. Soc. 2019, 3, 2411–2426. [Google Scholar] [CrossRef]
- Lebastchi, J.; Ajluni, N.; Neidert, A.; Oral, E.A. A Report of Three Cases with Acquired Generalized Lipodystrophy With Distinct Autoimmune Conditions Treated With Metreleptin. J. Clin. Endocrinol. Metab. 2015, 100, 3967–3970. [Google Scholar] [CrossRef] [PubMed]
- Rzehak, P.; Hellmuth, C.; Uhl, O.; Kirchberg, F.F.; Peissner, W.; Harder, U.; Grote, V.; Weber, M.; Xhonneux, A.; Langhendries, J.P.; et al. Rapid Growth and Childhood Obesity Are Strongly Associated with LysoPC(14:0). Ann. Nutr. Metab. 2014, 64, 294–303. [Google Scholar] [CrossRef] [PubMed]
- Rito, A.I.; Buoncristiano, M.; Spinelli, A.; Salanave, B.; Kunešová, M.; Hejgaard, T.; García Solano, M.; Fijałkowska, A.; Sturua, L.; Hyska, J.; et al. Association between Characteristics at Birth, Breastfeeding and Obesity in 22 Countries: The WHO European Childhood Obesity Surveillance Initiative—COSI 2015/2017. Obes. Facts 2019, 12, 226–243. [Google Scholar] [CrossRef] [PubMed]
- Madan, J.C.; Hoen, A.G.; Lundgren, S.N.; Farzan, S.F.; Cottingham, K.L.; Morrison, H.G.; Sogin, M.L.; Li, H.; Moore, J.H.; Karagas, M.R. Association of Cesarean Delivery and Formula Supplementation with the Intestinal Microbiome of 6-Week-Old Infants. JAMA Pediatrics 2016, 170, 212–219. [Google Scholar] [CrossRef] [PubMed]
- Moreno, M.A.; Furtner, F.; Rivara, F.P. Breastfeeding as Obesity Prevention. Arch. Pediatrics Adolesc. Med. 2011. [Google Scholar] [CrossRef] [PubMed]
- Luoto, R.; Kalliomäki, M.; Laitinen, K.; Delzenne, N.M.; Cani, P.D.; Salminen, S.; Isolauri, E. Initial Dietary and Microbiological Environments Deviate in Normal-Weight Compared to Overweight Children at 10 Years of Age. J. Pediatric Gastroenterol. Nutr. 2011, 52, 90–95. [Google Scholar] [CrossRef]
- Yu, H.; Dilbaz, S.; Coßmann, J.; Hoang, A.C.; Diedrich, V.; Herwig, A.; Harauma, A.; Hoshi, Y.; Moriguchi, T.; Landgraf, K.; et al. Breast Milk Alkylglycerols Sustain Beige Adipocytes through Adipose Tissue Macrophages. J. Clin. Investig. 2019, 129, 2485–2499. [Google Scholar] [CrossRef]
- Gliniak, C.M.; Scherer, P.E. Critical Lipids Link Breastfeeding to Healthy Adipose Tissue in Infancy and Adulthood. J. Clin. Investig. 2019, 129, 2198–2200. [Google Scholar] [CrossRef]
- Sun, K.; Gao, Z.; Kolonin, M.G. Transient Inflammatory Signaling Promotes Beige Adipogenesis. Sci. Signal. 2018, 11, 527. [Google Scholar] [CrossRef]
- Ohta, T. Maternal High-Fat Diet Promotes Onset of Diabetes in Rat Offspring. Anim. Sci. J. 2017. [Google Scholar] [CrossRef]
- Taylor, P.D.; Samuelsson, A.M.; Poston, L. Maternal obesity and the developmental programming of hypertension: A role for leptin. Acta Physiol. 2014, 210, 508–523. [Google Scholar] [CrossRef] [PubMed]
- Blackmore, H.L. Maternal Diet-Induced Obesity Programs Cardiovascular Dysfunction in Adult Male Mouse Offspring Independent of Current Body Weight. Endocrinology 2014, 155, 3970–3980. [Google Scholar] [CrossRef] [PubMed]
- Kruse, M. High-Fat Intake during Pregnancy and Lactation Exacerbates High-Fat Diet-Induced Complications in Male Offspring in Mice. Endocrinology 2013, 154, 3565–3576. [Google Scholar] [CrossRef]
- Liang, X.; Yang, Q.; Zhang, L.; Maricelli, J.W.; Rodgers, B.D.; Zhu, M.J.; Du, M. Maternal High-Fat Diet during Lactation Impairs Thermogenic Function of Brown Adipose Tissue in Offspring Mice. Sci. Rep. 2016, 6, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Moreno-Navarrete, J.M.; Fernandez-Real, J.M. The Gut Microbiota Modulates Both Browning of White Adipose Tissue and the Activity of Brown Adipose Tissue. Rev. Endocr. Metab. Disord. 2019, 20, 387–397. [Google Scholar] [CrossRef]
- Li, T.; Gong, H.; Yuan, Q.; Du, M.; Ren, F.; Mao, X. Supplementation of Polar Lipids-Enriched Milk Fat Globule Membrane in High-Fat Diet-Fed Rats during Pregnancy and Lactation Promotes Brown/Beige Adipocyte Development and Prevents Obesity in Male Offspring. FASEB. J. 2020, 34, 4619–4634. [Google Scholar] [CrossRef]
- Rodríguez-Cano, A.M.; Mier-Cabrera, J.; Rodríguez-Hernández, C.; Allegre-Dávalos, A.L.; Muñoz-Manrique, C.; Perichart-Perera, O. Complementary Feeding Practices and Their Association with Adiposity Indicators at 12 Months of Age. J. Dev. Orig Health Dis. 2020, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Chiang, K.V.; Hamner, H.C.; Li, R.; Perrine, C.G. Timing of Introduction of Complementary Foods—United States, 2016-2018. Morb. Mortal. Wkly. Rep. 2020, 69, 1787–1791. [Google Scholar] [CrossRef]
- Peixoto, T.C.; Pietrobon, C.B.; Bertasso, I.M.; Caramez, F.A.H.; Calvino, C.; Santos, T.R.; Oliveira, E.; Moura, E.G.; Lisboa, P.C. Early Weaning Alters the Thermogenic Capacity of Brown Adipose Tissue in Adult Male and Female Rats. Eur. J. Nutr. 2019, 1–12. [Google Scholar] [CrossRef]
- Valle, A.; García-Palmer, F.J.; Oliver, J.; Roca, P. Sex Differences in Brown Adipose Tissue Thermogenic Features during Caloric Restriction. Cell. Physiol. Biochem. 2007, 19, 195–204. [Google Scholar] [CrossRef]
- Wu, C. Activating Brown Adipose Tissue for Weight Loss and Lowering of Blood Glucose Levels: A MicroPET Study Using Obese and Diabetic Model Mice. PLoS ONE 2014, 9, e113742. [Google Scholar] [CrossRef]
- Bonet, M.L.; Mercader, J.; Palou, A. A Nutritional Perspective on UCP1-Dependent Thermogenesis. Biochimie 2017, 134, 99–117. [Google Scholar] [CrossRef]
- Suárez-Zamorano, N.; Fabbiano, S.; Chevalier, C. Microbiota Depletion Promotes Browning of White Adipose Tissue and Reduces Obesity. Nat. Med. 2015, 21, 1497–1501. [Google Scholar] [CrossRef]
- Kincaid, H.J.; Nagpal, R.; Yadav, H. Microbiome-Immune-Metabolic Axis in the Epidemic of Childhood Obesity: Evidence and Opportunities. Obes. Rev. 2020, 21, e12963. [Google Scholar] [CrossRef]
- Mestdagh, R.; Dumas, M.E.; Rezzi, S. Gut Microbiota Modulate the Metabolism of Brown Adipose Tissue in Mice. J. Proteome Res. 2012, 11, 620–630. [Google Scholar] [CrossRef]
- Li, B.; Li, L.; Li, M. Microbiota Depletion Impairs Thermogenesis of Brown Adipose Tissue and Browning of White Adipose Tissue. Cell Rep. 2019, 26, 2720–2737. [Google Scholar] [CrossRef]
- Chevalier, C.; Stojanović, O.; Colin, D.J. Gut Microbiota Orchestrates Energy Homeostasis during Cold. Cell 2015, 163, 1360–1374. [Google Scholar] [CrossRef] [PubMed]
- Ziętak, M.; Kovatcheva-Datchary, P.; Markiewicz, L.H. Altered Microbiota Contributes to Reduced Diet-Induced Obesity upon Cold Exposure. Cell Metab. 2016, 23, 1216–1223. [Google Scholar] [CrossRef] [PubMed]
- Li, G.; Xie, C.; Lu, S. Intermittent Fasting Promotes White Adipose Browning and Decreases Obesity by Shaping the Gut Microbiota. Cell Metab. 2017, 26, 672–685. [Google Scholar] [CrossRef] [PubMed]
- Fabbiano, S.; Suárez-Zamorano, N.; Chevalier, C. Functional Gut Microbiota Remodeling Contributes to the Caloric Restriction-Induced Metabolic Improvements. Cell Metab. 2018, 28, 907–921. [Google Scholar] [CrossRef]
- Wankhade, U.D.; Shen, M.; Yadav, H. Novel Browning Agents, Mechanisms, and Therapeutic Potentials of Brown Adipose Tissue. Biomed. Res. Int. 2016, 2016, 2365609. [Google Scholar] [CrossRef]
- Liber, A.; Szajewska, H. Effect of Oligofructose Supplementation on Body Weight in Overweight and Obese Children: A Randomised, Double-Blind, Placebo-Controlled Trial. Br. J. Nutr. 2014, 112, 2068–2074. [Google Scholar] [CrossRef] [PubMed]
- Reynés, B.; Palou, M.; Rodríguez, A.M. Regulation of Adaptive Thermogenesis and Browning by Prebiotics and Postbiotics. Front. Physiol. 2019, 9, 1908. [Google Scholar] [CrossRef] [PubMed]
- Vallianou, N.; Stratigou, T.; Christodoulatos, G.S.; Tsigalou, C.; Dalamaga, M. Probiotics, Prebiotics, Synbiotics, Postbiotics, and Obesity: Current Evidence, Controversies, and Perspectives. Curr. Obes. Rep. 2020, 9, 179–192. [Google Scholar] [CrossRef] [PubMed]
- Hill, C.; Guarner, F.; Reid, G.; Gibson, G.R.; Merenstein, D.J.; Pot, B.; Morelli, L.; Canani, R.B.; Flint, H.J.; Salminen, S.; et al. Expert Consensus Document. The International Scientific Association for Probiotics and Prebiotics Consensus Statement on the Scope and Appropriate Use of the Term Probiotic. Nat. Rev. Gastroenterol. Hepatol. 2014, 11, 506–514. [Google Scholar] [CrossRef] [PubMed]
- Chen, M.; Hou, P.; Zhou, M.; Ren, Q.; Wang, X.; Huang, L.; Hui, S.; Yi, L.; Mi, M. Resveratrol Attenuates High-Fat Diet-Induced Non-Alcoholic Steatohepatitis by Maintaining Gut Barrier Integrity and Inhibiting Gut Inflammation through Regulation of the Endocannabinoid System. Clin. Nutr. 2020, 39, 1264–1275. [Google Scholar] [CrossRef]
- Anhê, F.F.; Roy, D.; Pilon, G.; Dudonné, S.; Matamoros, S.; Varin, T.V.; Garofalo, C.; Moine, Q.; Desjardins, Y.; Levy, E.; et al. A Polyphenol-Rich Cranberry Extract Protects from Diet-Induced Obesity, Insulin Resistance and Intestinal Inflammation in Association with Increased Akkermansia Spp. Population in the Gut Microbiota of Mice. Gut 2015, 64, 872–883. [Google Scholar] [CrossRef] [PubMed]
- Serrano, A.; Asnani-Kishnani, M.; Rodríguez, A.M. Programming of the Beige Phenotype in White Adipose Tissue of Adult Mice by Mild Resveratrol and Nicotinamide Riboside Supplementations in Early Postnatal Life. Mol. Nutr. Food Res. 2018, 62, e1800463. [Google Scholar] [CrossRef]
- Hui, S.; Liu, Y.; Huang, L. Resveratrol Enhances Brown Adipose Tissue Activity and White Adipose Tissue Browning in Part by Regulating Bile Acid Metabolism via Gut Microbiota Remodeling. Int. J. Obes. (Lond.) 2020, 44, 1678–1690. [Google Scholar] [CrossRef]
- You, Y.; Yuan, X.; Liu, X. Cyanidin-3-Glucoside Increases Whole Body Energy Metabolism by Upregulating Brown Adipose Tissue Mitochondrial Function. Mol. Nutr. Food Res. 2017, 61, 1700261. [Google Scholar] [CrossRef]
- Huang, F.; Zhao, R.; Xia, M.; Shen, G.X. Impact of Cyanidin-3-Glucoside on Gut Microbiota and Relationship with Metabolism and Inflammation in High Fat-High Sucrose Diet-Induced Insulin Resistant Mice. Microorganisms 2020, 8, 1238. [Google Scholar] [CrossRef]
- Anhê, F.F.; Nachbar, R.T.; Varin, T.V. Treatment with Camu Camu (Myrciaria Dubia) Prevents Obesity by Altering the Gut Microbiota and Increasing Energy Expenditure in Diet-Induced Obese Mice. Gut 2019, 68, 453–464. [Google Scholar] [CrossRef] [PubMed]
- Neyrinck, A.M.; Bindels, L.B.; Geurts, L.; Van Hul, M.; Cani, P.D.; Delzenne, N.M. A Polyphenolic Extract from Green Tea Leaves Activates Fat Browning in High-Fat-Diet-Induced Obese Mice. J. Nutr. Biochem. 2017, 49, 15–21. [Google Scholar] [CrossRef] [PubMed]
- Jeong, H.W.; Kim, J.K.; Kim, A.Y.; Cho, D.; Lee, J.-H.; Choi, J.K.; Park, M.; Kim, W. Green Tea Encourages Growth of Akkermansia Muciniphila. J. Med. Food 2020, 23, 841–851. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Luo, L.; Luo, Y.; Zhang, J.; Wang, X.; Sun, K.; Zeng, L. Prebiotic Properties of Green and Dark Tea Contribute to Protective Effects in Chemical-Induced Colitis in Mice: A Fecal Microbiota Transplantation Study. J. Agric. Food Chem. 2020, 68, 6368–6380. [Google Scholar] [CrossRef]
- Jung, E.S.; Park, J.I.; Park, H.; Holzapfel, W.; Hwang, J.S.; Lee, C.H. Seven-Day Green Tea Supplementation Revamps Gut Microbiome and Caecum/Skin Metabolome in Mice from Stress. Sci. Rep. 2019, 9, 18418. [Google Scholar] [CrossRef]
- García-Carrizo, F.; Cannon, B.; Nedergaard, J. Regulation of Thermogenic Capacity in Brown and White Adipocytes by the Prebiotic High-Esterified Pectin and Its Postbiotic Acetate. Int. J. Obes. (Lond.) 2020, 44, 715–726. [Google Scholar] [CrossRef]
- Dewulf, E.M.; Cani, P.D.; Claus, S.P. Insight into the Prebiotic Concept: Lessons from an Exploratory, Double Blind Intervention Study with Inulin-Type Fructans in Obese Women. Gut 2013, 62, 1112–1121. [Google Scholar] [CrossRef]
- Nicolucci, A.C.; Hume, M.P.; Martínez, I. Prebiotics Reduce Body Fat and Alter Intestinal Microbiota in Children Who Are Overweight or With Obesity. Gastroenterology 2017, 153, 711–722. [Google Scholar] [CrossRef]
- Xiao, X.; Bai, J.; Li, M.S.; Zhang, J.Y.; Sun, X.J.; Dong, Y. Supplementation of Fermented Barley Extracts with Lactobacillus Plantarum Dy-1 Inhibits Obesity via a UCP1-Dependent Mechanism. Biomed. Environ. Sci. 2019, 32, 578–591. [Google Scholar] [CrossRef]
- Fang, C.; Kim, H.; Yanagisawa, L.; Bennett, W.; Sirven, M.A.; Alaniz, R.C.; Talcott, S.T.; Mertens-Talcott, S.U. Gallotannins and Lactobacillus Plantarum WCFS1 Mitigate High-Fat Diet-Induced Inflammation and Induce Biomarkers for Thermogenesis in Adipose Tissue in Gnotobiotic Mice. Mol. Nutr. Food Res. 2019, 63, e1800937. [Google Scholar] [CrossRef]
- Park, S.-S.; Lee, Y.-J.; Kang, H.; Yang, G.; Hong, E.J.; Lim, J.Y.; Oh, S.; Kim, E. Lactobacillus Amylovorus KU4 Ameliorates Diet-Induced Obesity in Mice by Promoting Adipose Browning through PPARγ Signaling. Sci. Rep. 2019, 9, 20152. [Google Scholar] [CrossRef]
- Stojanov, S.; Berlec, A.; Štrukelj, B. The Influence of Probiotics on the Firmicutes/Bacteroidetes Ratio in the Treatment of Obesity and Inflammatory Bowel Disease. Microorganisms 2020, 8, 1715. [Google Scholar] [CrossRef] [PubMed]
- Cerdó, T.; García-Santos, J.A.; Bermúdez, M.G.; Campoy, C. The Role of Probiotics and Prebiotics in the Prevention and Treatment of Obesity. Nutrients 2019, 11, 635. [Google Scholar] [CrossRef]
- Luoto, R.; Kalliomäki, M.; Laitinen, K.; Isolauri, E. The Impact of Perinatal Probiotic Intervention on the Development of Overweight and Obesity: Follow-up Study from Birth to 10 Years. Int. J. Obes. (Lond.) 2010, 34, 1531–1537. [Google Scholar] [CrossRef] [PubMed]
- Alisi, A.; Bedogni, G.; Baviera, G.; Giorgio, V.; Porro, E.; Paris, C.; Giammaria, P.; Reali, L.; Anania, F.; Nobili, V. Randomised Clinical Trial: The Beneficial Effects of VSL#3 in Obese Children with Non-Alcoholic Steatohepatitis. Aliment. Pharm. 2014, 39, 1276–1285. [Google Scholar] [CrossRef]
- Sanchis-Chordà, J.; Del Pulgar, E.M.G.; Carrasco-Luna, J.; Benítez-Páez, A.; Sanz, Y.; Codoñer-Franch, P. Bifidobacterium Pseudocatenulatum CECT 7765 Supplementation Improves Inflammatory Status in Insulin-Resistant Obese Children. Eur. J. Nutr. 2019, 58, 2789–2800. [Google Scholar] [CrossRef] [PubMed]
- Kadooka, Y.; Sato, M.; Imaizumi, K.; Ogawa, A.; Ikuyama, K.; Akai, Y.; Okano, M.; Kagoshima, M.; Tsuchida, T. Regulation of Abdominal Adiposity by Probiotics (Lactobacillus Gasseri SBT2055) in Adults with Obese Tendencies in a Randomized Controlled Trial. Eur. J. Clin. Nutr. 2010, 64, 636–643. [Google Scholar] [CrossRef] [PubMed]
- Szulińska, M.; Łoniewski, I.; van Hemert, S.; Sobieska, M.; Bogdański, P. Dose-Dependent Effects of Multispecies Probiotic Supplementation on the Lipopolysaccharide (LPS) Level and Cardiometabolic Profile in Obese Postmenopausal Women: A 12-Week Randomized Clinical Trial. Nutrients 2018, 10, 773. [Google Scholar] [CrossRef]
- Canfora, E.E.; Beek, C.M.; Jocken, J.W.E. Colonic Infusions of Short-Chain Fatty Acid Mixtures Promote Energy Metabolism in Overweight/Obese Men: A Randomized Crossover Trial. Sci. Rep. 2017, 7, 1–12. [Google Scholar] [CrossRef]
- Chambers, E.S.; Preston, T.; Frost, G. Role of Gut Microbiota-Generated Short-Chain Fatty Acids in Metabolic and Cardiovascular Health. Curr. Nutr. Rep. 2018, 7, 198–206. [Google Scholar] [CrossRef]
- Gao, Z.; Yin, J.; Zhang, J. Butyrate improves insulin sensitivity and increases energy expenditure in mice. Diabetes 2009, 58, 1509–1517. [Google Scholar] [CrossRef] [PubMed]
- Sahuri-Arisoylu, M.; Brody, L.P.; Parkinson, J. Reprogramming of Hepatic Fat Accumulation and “browning” of Adipose Tissue by the Short-Chain Fatty Acid Acetate. Int. J. Obes. 2016, 40, 955–963. [Google Scholar] [CrossRef]
- Moreno-Navarrete, J.M.; Serino, M.; Blasco-Baque, V. Gut Microbiota Interacts with Markers of Adipose Tissue Browning, Insulin Action and Plasma Acetate in Morbid Obesity. Mol. Nutr. Food Res. 2018, 62, 1700721. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.-X.; Chen, Y.-M.; Chen, W.-Q.; Deng, X.-Q.; Jiang, Z.-Q. Energy Expenditure and Energy Intake in 10-12 Years Obese and Non-Obese Chinese Children in a Guangzhou Boarding School. Asia Pac. J. Clin. Nutr. 2008, 17, 235–242. [Google Scholar] [PubMed]
- Stensel, D.J.; Lin, F.P.; Nevill, A.M. Resting Metabolic Rate in Obese and Nonobese Chinese Singaporean Boys Aged 13-15 y. Am. J. Clin. Nutr. 2001, 74, 369–373. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Broeders, E.P.; Nascimento, E.B.; Havekes, B. The Bile Acid Chenodeoxycholic Acid Increases Human Brown Adipose Tissue Activity. Cell Metab. 2015, 22, 418–426. [Google Scholar] [CrossRef] [PubMed]
- Worthmann, A.; John, C.; Rühlemann, M.C. Cold-Induced Conversion of Cholesterol to Bile Acids in Mice Shapes the Gut Microbiome and Promotes Adaptive Thermogenesis. Nat. Med. 2017, 23, 839–849. [Google Scholar] [CrossRef] [PubMed]
- Nutrizione Umana, S.I. Livelli di Assunzione di Riferimento di Nutrienti ed Energia per la Popolazione Italiana IV Revisione; SICS: Milano, Italy, 2014. [Google Scholar]
- Salvini, S.; Parpinel, M.; Gnagnarella, P.; Maisonneuve, P.; Turrini, A. Banca Dati di Composizione Degli Alimenti per Studi Epidemiologici in ITALIA; Europeo di Oncologia: Milano, Italy, 1998. [Google Scholar]
- Albracht-Schulte, K.; Kalupahana, N.S.; Ramalingam, L.; Wang, S.; Rahman, S.M. Omega-3 fatty acids in obesity and metabolic syndrome: A mechanistic update. J. Nutr. Biochem. 2018, 58, 1–16. [Google Scholar] [CrossRef]
- Kalupahana, N.S.; Goonapienuwala, B.L.; Moustaid-Moussa, N. Omega-3 Fatty Acids and Adipose Tissue: Inflammation and Browning. Annu. Rev. Nutr. 2020, 40, 25–49. [Google Scholar] [CrossRef]
- Lund, J.; Larsen, L.H.; Lauritzen, L. Fish Oil as a Potential Activator of Brown and Beige Fat Thermogenesis. Adipocyte 2018, 7, 88–95. [Google Scholar] [CrossRef]
- Flachs, P.; Rühl, R.; Hensler, M. Synergistic Induction of Lipid Catabolism and Anti-Inflammatory Lipids in White Fat of Dietary Obese Mice in Response to Calorie Restriction and n-3 Fatty Acids. Diabetologia 2011, 54, 2626–2638. [Google Scholar] [CrossRef] [PubMed]
- LeMieux, M.J.; Kalupahana, N.S.; Scoggin, S. Eicosapentaenoic acid reduces adipocyte hypertrophy and inflammation in diet-induced obese mice in an adiposity-independent manner. J. Nutr. 2015, 145, 411–417. [Google Scholar] [CrossRef]
- Soni, N.; Ross, A.B.; Scheers, N.; Nookaew, I.; Gabrielsson, B.G.; Sandberg, A.S. The Omega-3 Fatty Acids EPA and DHA, as a Part of a Murine High-Fat Diet, Reduced Lipid Accumulation in Brown and White Adipose Tissues. Int. J. Mol. Sci. 2019, 20, 5895. [Google Scholar] [CrossRef]
- Bargut, T.C.; Souza-Mello, V.; Mandarim-de-Lacerda, C.A. Fish Oil Diet Modulates Epididymal and Inguinal Adipocyte Metabolism in Mice. Food Funct. 2016, 7, 1468–1476. [Google Scholar] [CrossRef]
- Browning, L.M.; Krebs, J.D.; Moore, C.S. The Impact of Long Chain N-3 Polyunsaturated Fatty Acid Supplementation on Inflammation, Insulin Sensitivity and CVD Risk in a Group of Overweight Women with an Inflammatory Phenotype. Diabetes Obes. Metab. 2007, 9, 70–80. [Google Scholar] [CrossRef] [PubMed]
- Spencer, M.; Finlin, B.S.; Unal, R. Omega-3 Fatty Acids Reduce Adipose Tissue Macrophages in Human Subjects with Insulin Resistance. Diabetes 2013, 62, 1709–1717. [Google Scholar] [CrossRef]
- Ferguson, J.F.; Xue, C.; Hu, Y. Adipose Tissue RNASeq Reveals Novel Gene-Nutrient Interactions Following n-3 PUFA Supplementation and Evoked Inflammation in Humans. J. Nutr. Biochem. 2016, 30, 126–132. [Google Scholar] [CrossRef] [PubMed]
- Domingo, P.; Gallego-Escuredo, J.M.; Fernández, I. Effects of docosahexanoic acid supplementation on inflammatory and subcutaneous adipose tissue gene expression in HIV-infected patients on combination antiretroviral therapy (cART). A sub-study of a randomized, double-blind, placebo-controlled study. Cytokine 2018, 105, 73–79. [Google Scholar] [CrossRef]
- Noumi, Y.; Kawamura, R.; Tabara, Y. An Inverse Association between Serum Resistin Levels and N-3 Polyunsaturated Fatty Acids Intake Was Strongest in the SNP-420 G/G Genotype in the Japanese Cohort: The Toon Genome Study. Clin. Endocrinol. 2018, 88, 51–57. [Google Scholar] [CrossRef] [PubMed]
- Bahreini, M.; Ramezani, A.H.; Shishehbor, F. The Effect of Omega-3 on Circulating Adiponectin in Adults with Type 2 Diabetes Mellitus: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. Can. J. Diabetes 2018, 42, 553–559. [Google Scholar] [CrossRef]
- Hames, K.C.; Morgan-Bathke, M.; Harteneck, D.A. Very-long-chain ω-3 fatty acid supplements and adipose tissue functions: A randomized controlled trial. Am. J. Clin. Nutr. 2017, 105, 1552–1558. [Google Scholar] [CrossRef] [PubMed]
- Huerta, A.E.; Navas-Carretero, S.; Prieto-Hontoria, P.L. Effects of α-Lipoic Acid and Eicosapentaenoic Acid in Overweight and Obese Women during Weight Loss. Obesity 2015, 23, 313–321. [Google Scholar] [CrossRef] [PubMed]
- Hariri, M.; Ghiasvand, R.; Shiranian, A. Does Omega-3 Fatty Acids Supplementation Affect Circulating Leptin Levels? A Systematic Review and Meta-Analysis on Randomized Controlled Clinical Trials. Clin. Endocrinol. (Oxf.) 2015, 82, 221–228. [Google Scholar] [CrossRef] [PubMed]
- Donahue, S.M.; Rifas-Shiman, S.L.; Gold, D.R. Prenatal Fatty Acid Status and Child Adiposity at Age 3 y: Results from a US Pregnancy Cohort. Am. J. Clin. Nutr. 2011, 93, 780–788. [Google Scholar] [CrossRef] [PubMed]
- Rudolph, M.C.; Young, B.E.; Lemas, D.J. Early Infant Adipose Deposition Is Positively Associated with the N-6 to n-3 Fatty Acid Ratio in Human Milk Independent of Maternal BMI. Int. J. Obes. (Lond.) 2017, 41, 510–517. [Google Scholar] [CrossRef]
- Zhao, M.; Chen, X. Eicosapentaenoic Acid Promotes Thermogenic and Fatty Acid Storage Capacity in Mouse Subcutaneous Adipocytes. Biochem. Biophys. Res. Commun. 2014, 450, 1446–1451. [Google Scholar] [CrossRef] [PubMed]
- Laiglesia, L.M.; Lorente-Cebrián, S.; Prieto-Hontoria, P.L. Eicosapentaenoic Acid Promotes Mitochondrial Biogenesis and Beige-like Features in Subcutaneous Adipocytes from Overweight Subjects. J. Nutr. Biochem. 2016, 37, 76–82. [Google Scholar] [CrossRef]
- Kim, M.; Goto, T.; Yu, R. Fish Oil Intake Induces UCP1 Upregulation in Brown and White Adipose Tissue via the Sympathetic Nervous System. Sci. Rep. 2015, 5, 1–12. [Google Scholar] [CrossRef]
- Ghandour, R.A.; Colson, C.; Giroud, M. Impact of Dietary Ω3 Polyunsaturated Fatty Acid Supplementation on Brown and Brite Adipocyte Function. J. Lipid Res. 2018, 59, 452–461. [Google Scholar] [CrossRef]
- Mendonça, A.M.; Cayer, L.G.J.; Pauls, S.D. Distinct Effects of Dietary ALA, EPA and DHA on Rat Adipose Oxylipins Vary by Depot Location and Sex. Prostaglandins Leukot. Essent. Fat. Acids 2018, 129, 13–24. [Google Scholar] [CrossRef] [PubMed]
- Caesar, R.; Tremaroli, V.; Kovatcheva-Datchary, P. Crosstalk between Gut Microbiota and Dietary Lipids Aggravates WAT Inflammation through TLR Signaling. Cell Metab. 2015, 22, 658–668. [Google Scholar] [CrossRef]
- Scheele, C.; Nielsen, S. Metabolic Regulation and the Anti-Obesity Perspectives of Human Brown Fat. Redox Biol. 2017, 12, 770–775. [Google Scholar] [CrossRef]
- Rossignoli, C.P.; Dechandt, C.R.P.; Souza, A.O.; Sampaio, I.H.; Vicentini, T.M.; Teodoro, B.G.; Neto, M.P.C.; Ferrari, G.D.; Couto-Lima, C.A.; Alberici, L.C. Effects of Intermittent Dietary Supplementation with Conjugated Linoleic Acid and Fish Oil (EPA/DHA) on Body Metabolism and Mitochondrial Energetics in Mice. J. Nutr. Biochem. 2018, 60, 16–23. [Google Scholar] [CrossRef]
- Im, D.S. Functions of Omega-3 Fatty Acids and FFA4 (GPR120) in Macrophages. Eur. J. Pharm. 2016, 785, 36–43. [Google Scholar] [CrossRef] [PubMed]
- Andersen, A.D.; Michaelsen, K.F.; Hellgren, L.I. A Randomized Controlled Intervention with Fish Oil versus Sunflower Oil from 9 to 18 Months of Age: Exploring Changes in Growth and Skinfold Thicknesses. Pediatr. Res. 2011, 70, 368–374. [Google Scholar] [CrossRef] [PubMed]
- Couet, C.; Delarue, J.; Ritz, P. Effect of Dietary Fish Oil on Body Fat Mass and Basal Fat Oxidation in Healthy Adults. Int. J. Obes. Relat. Metab. Disord. 1997, 21, 637–643. [Google Scholar] [CrossRef]
- Du, S.; Jin, J.; Fang, W. Does Fish Oil Have an Anti-Obesity Effect in Overweight/Obese Adults? A Meta-Analysis of Randomized Controlled Trials. PLoS ONE 2015, 10, e0142652. [Google Scholar]
- Bender, N.; Portmann, M.; Heg, Z. Fish or n3-PUFA intake and body composition: A systematic review and meta-analysis. Obes. Rev. 2014, 15, 657–665. [Google Scholar] [CrossRef]
- Madsen, L.; Kristiansen, K. Of Mice and Men: Factors Abrogating the Antiobesity Effect of Omega-3 Fatty Acids. Adipocyte 2012, 1, 173–176. [Google Scholar] [CrossRef][Green Version]
- Xiang, A.S.; Giles, C.; Loh, R.K.C.; Formosa, M.F.; Eikelis, N.; Lambert, G.W.; Meikle, P.J.; Kingwell, B.A.; Carey, A.L. Plasma Docosahexaenoic Acid and Eicosapentaenoic Acid Concentrations Are Positively Associated with Brown Adipose Tissue Activity in Humans. Metabolites 2020, 10, 388. [Google Scholar] [CrossRef] [PubMed]
- Huang, T.; Wang, T.; Heianza, Y.; Zheng, Y.; Sun, D.; Kang, J.H.; Pasquale, L.R.; Rimm, E.B.; Manson, J.E.; Hu, F.B.; et al. Habitual Consumption of Long-Chain n-3 PUFAs and Fish Attenuates Genetically Associated Long-Term Weight Gain. Am. J. Clin. Nutr. 2019, 109, 665–673. [Google Scholar] [CrossRef] [PubMed]
- Fleckenstein-Elsen, M.; Dinnies, D.; Jelenik, T.; Roden, M.; Romacho, T.; Eckel, J. Eicosapentaenoic Acid and Arachidonic Acid Differentially Regulate Adipogenesis, Acquisition of a Brite Phenotype and Mitochondrial Function in Primary Human Adipocytes. Mol. Nutr. Food Res. 2016, 60, 2065–2075. [Google Scholar] [CrossRef] [PubMed]
- Worsch, S.; Heikenwalder, M.; Hauner, H.; Bader, B.L. Dietary N-3 Long-Chain Polyunsaturated Fatty Acids Upregulate Energy Dissipating Metabolic Pathways Conveying Anti-Obesogenic Effects in Mice. Nutr. Metab. 2018, 15, 65. [Google Scholar] [CrossRef]
- Fan, R.; Toney, A.M.; Jang, Y.; Ro, S.-H.; Chung, S. Maternal N-3 PUFA Supplementation Promotes Fetal Brown Adipose Tissue Development through Epigenetic Modifications in C57BL/6 Mice. Biochim. Biophys Acta Mol. Cell Biol. Lipids 2018, 1863, 1488–1497. [Google Scholar] [CrossRef]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Verduci, E.; Calcaterra, V.; Di Profio, E.; Fiore, G.; Rey, F.; Magenes, V.C.; Todisco, C.F.; Carelli, S.; Zuccotti, G.V. Brown Adipose Tissue: New Challenges for Prevention of Childhood Obesity. A Narrative Review. Nutrients 2021, 13, 1450. https://doi.org/10.3390/nu13051450
Verduci E, Calcaterra V, Di Profio E, Fiore G, Rey F, Magenes VC, Todisco CF, Carelli S, Zuccotti GV. Brown Adipose Tissue: New Challenges for Prevention of Childhood Obesity. A Narrative Review. Nutrients. 2021; 13(5):1450. https://doi.org/10.3390/nu13051450
Chicago/Turabian StyleVerduci, Elvira, Valeria Calcaterra, Elisabetta Di Profio, Giulia Fiore, Federica Rey, Vittoria Carlotta Magenes, Carolina Federica Todisco, Stephana Carelli, and Gian Vincenzo Zuccotti. 2021. "Brown Adipose Tissue: New Challenges for Prevention of Childhood Obesity. A Narrative Review" Nutrients 13, no. 5: 1450. https://doi.org/10.3390/nu13051450
APA StyleVerduci, E., Calcaterra, V., Di Profio, E., Fiore, G., Rey, F., Magenes, V. C., Todisco, C. F., Carelli, S., & Zuccotti, G. V. (2021). Brown Adipose Tissue: New Challenges for Prevention of Childhood Obesity. A Narrative Review. Nutrients, 13(5), 1450. https://doi.org/10.3390/nu13051450










