Vitamin D Deficiency and Outcome of COVID-19 Patients
Abstract
1. Introduction
2. Materials and Methods
2.1. Patients and Data Collection
2.2. Diagnosis, Supportive Care and Treatment
2.3. Assessment of VitD Status and Cytokine Serum Levels
2.4. Statistics
3. Results
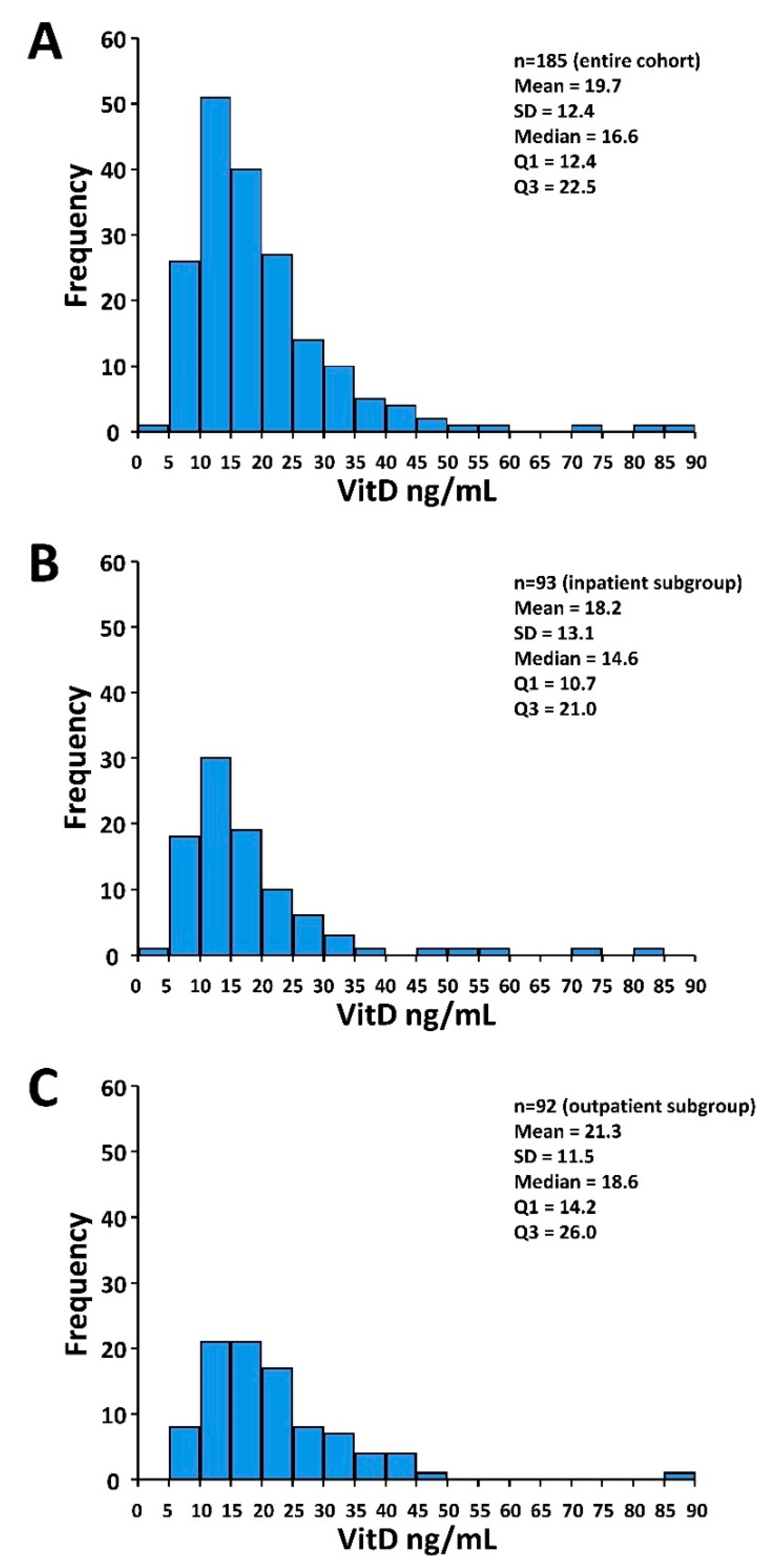
3.1. Patients and Treatment Characteristics and VitD Status
3.2. Patient and Treatment Characteristics According to VitD Status
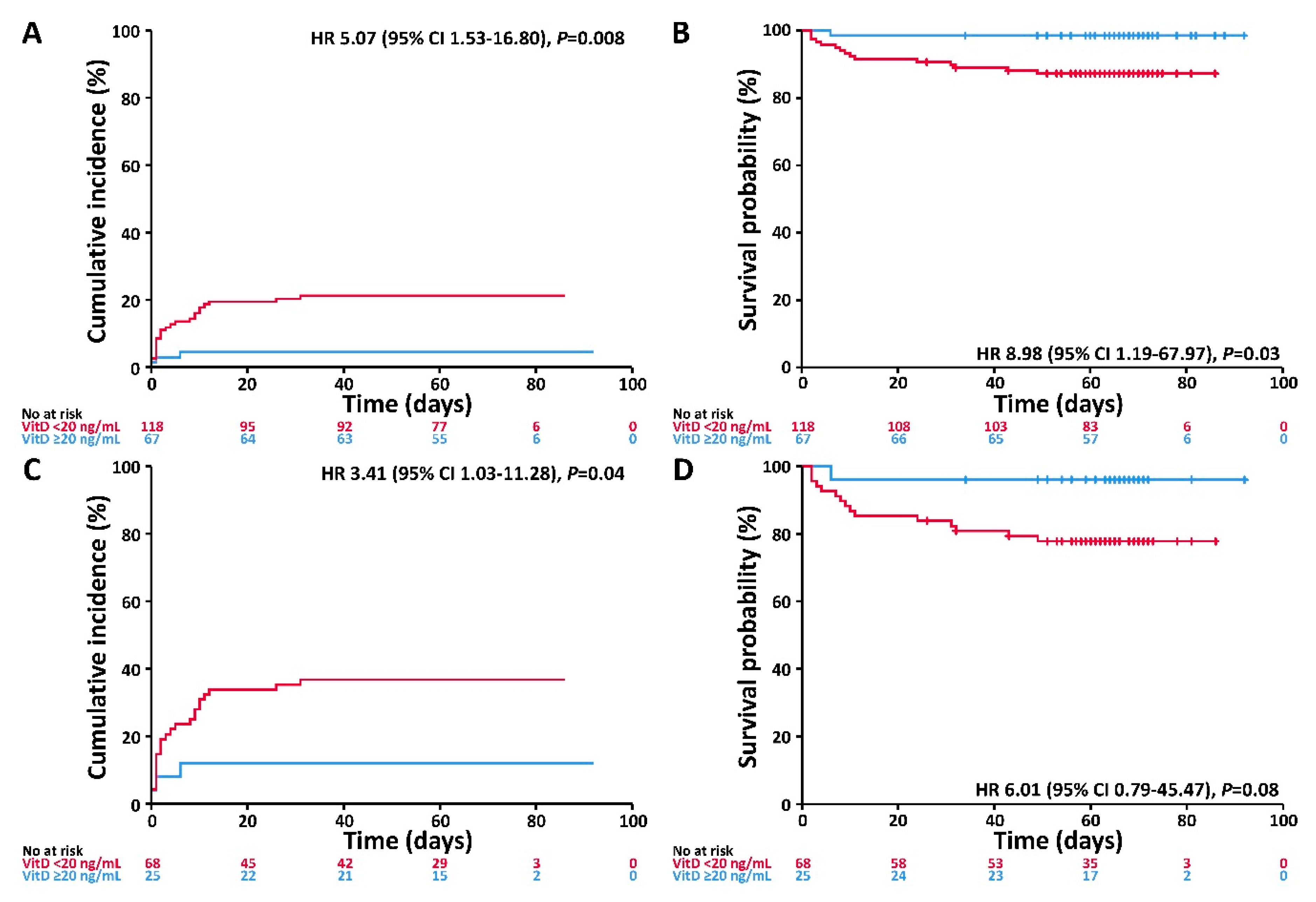
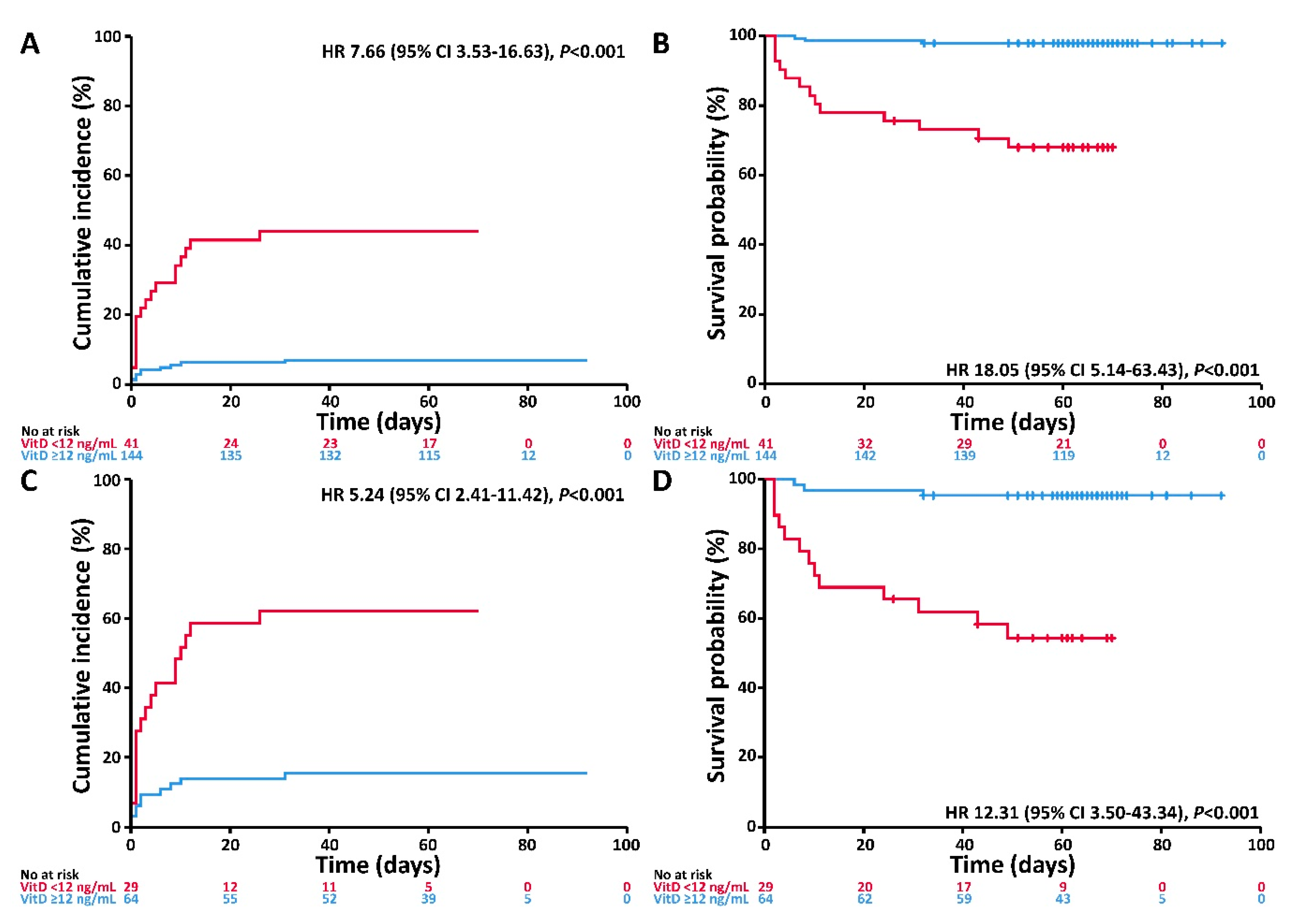
3.3. Associations of VitD Status with the Endpoints Invasive Mechanical Ventilation and/or Death and Death
4. Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A
| Entire Cohort (n = 185) | Inpatient Subgroup (n = 93) | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| IMV/D (Events, n = 28) | Death (Events, n = 16) | IMV/D (Events, n = 28) | Death (Events, n = 16) | ||||||
| HR (95% CI) | p | HR (95% CI) | p | HR (95% CI) | p | HR (95% CI) | p | ||
| Covariate | Effect | ||||||||
| VitD | <20 ng/mL | 5.75 (1.73–19.09) | 0.004 | 11.27 (1.48–85.55) | 0.02 | 3.99 (1.20–13.28) | 0.02 | 7.97 (1.05–60.60) | 0.04 |
| Age | ≥60 years | 4.03 (1.35–12.02) | 0.01 | 9.35 (1.21–72.45) | 0.03 | 3.91 (1.26–12.07) | 0.02 | 9.25 (1.16–73.88) | 0.04 |
| Gender | Male | 1.63 (0.73–3.61) | 0.23 | 2.31 (0.74–7.19) | 0.15 | 1.38 (0.61–3.11) | 0.44 | 2.04 (0.64–6.48) | 0.23 |
| Comorbidity | Any | 3.48 (1.36–8.87) | 0.009 | 5.90 (1.31–26.48) | 0.02 | 1.91 (0.72–5.02) | 0.19 | 3.15 (0.68–14.59) | 0.14 |
References
- Dong, E.; Du, H.; Gardner, L. An interactive web-based dashboard to track COVID-19 in real time. Lancet Infect. Dis. 2020, 20, 533–534. [Google Scholar] [CrossRef]
- Chen, N.; Zhou, M.; Dong, X.; Qu, J.; Gong, F.; Han, Y.; Qiu, Y.; Wang, J.; Liu, Y.; Wei, Y.; et al. Epidemiological and clinical characteristics of 99 cases of 2019 novel coronavirus pneumonia in Wuhan, China: A descriptive study. Lancet 2020, 395, 507–513. [Google Scholar] [CrossRef]
- Yang, J.; Zheng, Y.; Gou, X.; Pu, K.; Chen, Z.; Guo, Q.; Ji, R.; Wang, H.; Wang, Y.; Zhou, Y.-N. Prevalence of comorbidities and its effects in patients infected with SARS-CoV-2: A systematic review and meta-analysis. Int. J. Infect. Dis. 2020, 94, 91–95. [Google Scholar] [CrossRef] [PubMed]
- Shi, Y.; Yu, X.; Zhao, H.; Wang, H.; Zhao, R.; Sheng, J. Host susceptibility to severe COVID-19 and establishment of a host risk score: Findings of 487 cases outside Wuhan. Crit. Care 2020, 24, 1–4. [Google Scholar] [CrossRef]
- Charoenngam, N.; Holick, M.F. Immunologic Effects of Vitamin D on Human Health and Disease. Nutrients 2020, 12, 2097. [Google Scholar] [CrossRef]
- Holick, M.F. Vitamin D Deficiency. N. Engl. J. Med. 2007, 357, 266–281. [Google Scholar] [CrossRef]
- Martineau, A.R.; Jolliffe, D.A.; Hooper, R.L.; Greenberg, L.; Aloia, J.F.; Bergman, P.; Dubnov-Raz, G.; Esposito, S.; Ganmaa, D.; Ginde, A.A.; et al. Vitamin D supplementation to prevent acute respiratory tract infections: Systematic review and meta-analysis of individual participant data. BMJ 2017, 356, i6583. [Google Scholar] [CrossRef]
- Marik, P.E.; Kory, P.; Varon, J. Does vitamin D status impact mortality from SARS-CoV-2 infection? Med. Drug Discov. 2020, 6, 100041. [Google Scholar] [CrossRef]
- Mitchell, F. Vitamin-D and COVID-19: Do deficient risk a poorer outcome? Lancet Diabetes Endocrinol. 2020, 8, 570. [Google Scholar] [CrossRef]
- Rhodes, J.; Subramanian, S.; Laird, E.; Kenny, R.A. Editorial: Low population mortality from COVID-19 in countries south of latitude 35 degrees North supports vitamin D as a factor determining severity. Aliment. Pharmacol. Ther. 2020, 51, 1434–1437. [Google Scholar] [CrossRef]
- Grant, W.B.; Lahore, H.; McDonnell, S.; Baggerly, C.A.; French, C.B.; Aliano, J.L.; Bhattoa, H.P. Evidence that Vitamin D Supplementation Could Reduce Risk of Influenza and COVID-19 Infections and Deaths. Nutrients 2020, 12, 988. [Google Scholar] [CrossRef] [PubMed]
- Isaia, G.; Medico, E. Associations between hypovitaminosis D and COVID-19: A narrative review. Aging Clin. Exp. Res. 2020, 1–3. [Google Scholar] [CrossRef] [PubMed]
- Jakovac, H. COVID-19 and vitamin D—Is there a link and an opportunity for intervention? Am. J. Physiol. Metab. 2020, 318, E589. [Google Scholar] [CrossRef]
- Arboleda, J.F.; Urcuqui-Inchima, S. Vitamin D Supplementation: A Potential Approach for Coronavirus/COVID-19 Therapeutics? Front. Immunol. 2020, 11. [Google Scholar] [CrossRef]
- Hinweise zu Erkennung, Diagnostik und Therapie von Patienten mit COVID-19. Available online: https://www.rki.de/DE/Content/Kommissionen/Stakob/Stellungnahmen/Stellungnahme-Covid-19_Therapie_Diagnose.html (accessed on 1 August 2020).
- Verity, R.; Okell, L.C.; Dorigatti, I.; Winskill, P.; Whittaker, C.; Imai, N.; Cuomo-Dannenburg, G.; Thompson, H.; Walker, P.G.T.; Fu, H.; et al. Estimates of the severity of coronavirus disease 2019: A model-based analysis. Lancet Infect. Dis. 2020, 20, 669–677. [Google Scholar] [CrossRef]
- Hippchen, T.; Kräusslich, H.-G.; Merle, U. Coronataxi brings outpatient care to covid-19 patients. Ann. Emerg. Med. 2020. [Google Scholar] [CrossRef]
- Schemper, M.; Smith, T.L. A note on quantifying follow-up in studies of failure time. Control. Clin. Trials 1996, 17, 343–346. [Google Scholar] [CrossRef]
- Ross, A.C.; Taylor, C.L.; Yaktine, A.L.; Del Valle, H.B. (Eds.) Institute of Medicine (US) Committee to Review Dietary Reference Intakes for Vitamin D and Calcium. In Dietary Reference Intakes for Calcium and Vitamin D; National Academies Press: Washington, DC, USA, 2011. [Google Scholar]
- Martineau, A.R.; Forouhi, N.G. Vitamin D for COVID-19: A case to answer? Lancet Diabetes Endocrinol. 2020, 8, 735–736. [Google Scholar] [CrossRef]
- D’avolio, A.; Avataneo, V.; Manca, A.; Cusato, J.; De Nicolò, A.; Lucchini, R.; Keller, F.; Cantù, M. 25-Hydroxyvitamin D Concentrations Are Lower in Patients with Positive PCR for SARS-CoV-2. Nutrients 2020, 12, 1359. [Google Scholar] [CrossRef]
- Munshi, R.; Hussein, M.H.; Toraih, E.A.; Elshazli, R.M.; Jardak, C.; Sultana, N.; Youssef, M.R.; Omar, M.; Attia, A.S.; Fawzy, M.S.; et al. Vitamin D insufficiency as a potential culprit in critical COVID-19 patients. J. Med. Virol. 2020. [Google Scholar] [CrossRef]
- Faul, J.L.; Kerley, C.P.; Love, B.; O’Neill, E.; Cody, C.; Tormey, W.; Hutchinson, K.; Cormican, L.J.; Burke, C.M. Vitamin D Deficiency and ARDS after SARS-CoV-2 Infection. Ir. Med. J. 2020, 113, 84. [Google Scholar] [PubMed]
- Rabenberg, M.; Scheidt-Nave, C.; Busch, M.A.; Rieckmann, N.; Hintzpeter, B.; Mensink, G.B.M. Vitamin D status among adults in Germany--results from the German Health Interview and Examination Survey for Adults (DEGS1). BMC Public Health 2015, 15, 641. [Google Scholar] [CrossRef] [PubMed]
| Entire Cohort | Inpatients | Outpatients | pa | |
|---|---|---|---|---|
| n = 185 | n = 93 | n = 92 | ||
| Median Age [years] (IQR) | 60 (49–70) | 63 (52–74) | 56 (42–64) | <0.001 |
| Age, n (%) | 0.02 | |||
| <60 years | 88 (48) | 36 (39) | 52 (57) | |
| ≥60 years | 97 (52) | 57 (61) | 40 (43) | |
| Gender, n (%) | 0.003 | |||
| Male | 95 (51) | 59 (63) | 36 (39) | |
| Female | 90 (49) | 34 (37) | 56 (61) | |
| Cardiovascular disease b, n (%) | <0.001 | |||
| Yes | 58 (31) | 45 (48) | 13 (14) | |
| No | 127 (69) | 48 (52) | 79 (86) | |
| Diabetes, n (%) | 0.14 | |||
| Yes | 19 (10) | 13 (14) | 6 (7) | |
| No | 166 (90) | 80 (86) | 86 (93) | |
| Chronic kidney disease, n (%) | 0.006 | |||
| Yes | 8 (4) | 8 (9) | 0 (0) | |
| No | 177 (96) | 85 (91) | 92 (100) | |
| Chronic lung disease, n (%) | 0.28 | |||
| Yes | 15 (8) | 10 (11) | 5 (5) | |
| No | 170 (92) | 83 (89) | 87 (95) | |
| Active or history of malignancy, n (%) | 1 | |||
| Yes | 17 (9) | 9 (10) | 8 (9) | |
| No | 168 (91) | 84 (90) | 84 (91) | |
| Comorbidity, n (%) | <0.001 | |||
| Any | 77 (42) | 52 (56) | 25 (27) | |
| None | 108 (58) | 41 (44) | 67 (73) | |
| Median VitD level [ng/mL] (IQR) | 16.6 (12.4–22.5) | 14.6 (10.7–21.0) | 18.6 (14.2–26.0) | 0.001 |
| Median IL-6 c [pg/mL] (IQR) | 35.8 (14.4–80.0) | 35.8 (14.4–80.0) | - | - |
| VitD, n (%) | 0.004 | |||
| <12 ng/mL | 41 (22) | 29 (31) | 12 (13) | |
| ≥12 ng/mL | 144 (78) | 64 (69) | 80 (87) | |
| Maximum oxygen therapy c, n (%) | <0.001 | |||
| None | 105 (57) | 13 (14) | 92 (100) | |
| Low-dose oxygen (NC) | 45 (24) | 45 (48) | 0 (0) | |
| HFNO | 12 (6) | 12 (13) | 0 (0) | |
| IMV | 23 (12) | 23 (25) | 0 (0) | |
| Vitamin D supplementation d, n (%) | 0.36 | |||
| Yes | 6 (5) | 6 (6) | 0 (0) | |
| No | 108 (95) | 87 (94) | 21 (100) | |
| Unknown | 71 | 71 |
| Entire Cohort | Inpatient Subgroup | Outpatient Subgroup | |||||||
|---|---|---|---|---|---|---|---|---|---|
| (n = 185) | (n = 93) | (n = 92) | |||||||
| VitD <12 ng/mL | VitD ≥12 ng/mL | Pa | VitD < 12 ng/mL | VitD ≥ 12 ng/mL | Pa | VitD < 12 ng/mL | VitD ≥ 12 ng/mL | pa | |
| n = 41 | n = 144 | n = 29 | n = 64 | n = 12 | n = 80 | ||||
| Median age [years] (IQR) | 66 (53–78) | 58 (47–67) | 0.002 | 71 (54–79) | 62 (50–70) | 0.03 | 60 (48–77) | 55 (42–63) | 0.22 |
| Age, n (%) | 0.12 | 0.36 | 0.76 | ||||||
| <60 years | 15 (37) | 73 (51) | 9 (31) | 27 (42) | 6 (50) | 46 (58) | |||
| ≥60 years | 26 (63) | 71 (49) | 20 (69) | 37 (58) | 6 (50) | 34 (42) | |||
| Gender, n (%) | 0.6 | 0.82 | 0.76 | ||||||
| Male | 23 (56) | 72 (50) | 19 (66) | 40 (62) | 4 (33) | 32 (40) | |||
| Female | 18 (44) | 72 (50) | 10 (34) | 24 (38) | 8 (67) | 48 (60) | |||
| Cardiovascular disease b, n (%) | 0.06 | 0.26 | 0.69 | ||||||
| Yes | 18 (44) | 40 (28) | 17 (59) | 28 (44) | 1 (8) | 12 (15) | |||
| No | 23 (56) | 104 (72) | 12 (41) | 36 (56) | 11 (92) | 68 (85) | |||
| Diabetes, n (%) | 0.04 | 0.33 | 0.17 | ||||||
| Yes | 8 (20) | 11 (8) | 6 (21) | 7 (11) | 2 (17) | 4 (5) | |||
| No | 33 (80) | 133 (92) | 23 (79) | 57 (89) | 10 (83) | 76 (95) | |||
| Chronic kidney disease, n (%) | 1 | 1 | 1 | ||||||
| Yes | 2 (5) | 6 (4) | 2 (7) | 6 (9) | 0 (0) | 0 (0) | |||
| No | 39 (95) | 138 (96) | 27 (93) | 58 (91) | 12 (100) | 80 (100) | |||
| Chronic lung disease, n (%) | 0.1 | 0.07 | 0.61 | ||||||
| Yes | 6 (15) | 9 (6) | 6 (21) | 4 (6) | 0 (0) | 5 (6) | |||
| No | 35 (85) | 135 (94) | 23 (79) | 60 (94) | 12 (100) | 75 (94) | |||
| Active or history of malignancy, n (%) | 0.54 | 0.45 | 1 | ||||||
| Yes | 5 (12) | 12 (8) | 4 (14) | 5 (8) | 1 (8) | 7 (9) | |||
| No | 36 (88) | 132 (92) | 25 (86) | 59 (92) | 11 (92) | 73 (91) | |||
| Comorbidity, n (%) | 0.11 | 0.26 | 1 | ||||||
| Any | 22 (54) | 55 (38) | 19 (66) | 33 (52) | 3 (25) | 22 (27) | |||
| None | 19 (46) | 89 (62) | 10 (34) | 31 (48) | 9 (75) | 58 (73) | |||
| Median IL-6 c [pg/mL] (IQR) | 70.5 (32.0–326.3) | 29.7 (14.3–59.9) | 0.01 | 70.5 (32.0–326.3) | 29.7 (14.3–59.9) | 0.01 | - | - | - |
| Treatment mode, n (%) | 0.004 | 1 | 1 | ||||||
| Inpatient | 29 (71) | 64 (44) | 29 (100) | 64 (100) | 0 (0) | 0 (0) | |||
| Outpatient | 12 (29) | 80 (56) | 0 (0) | 0 (0) | 12 (100) | 80 (100) | |||
| Maximum oxygen therapy d, n (%) | <0.001 | 0.004 | 1 | ||||||
| None | 15 (37) | 90 (63) | 3 (10) | 10 (16) | 12 (100) | 80 (100) | |||
| Low-dose oxygen (NC) | 8 (20) | 37 (26) | 8 (28) | 37 (58) | 0 (0) | 0 (0) | |||
| HFNO | 4 (10) | 8 (6) | 4 (14) | 8 (13) | 0 (0) | 0 (0) | |||
| IMV | 14 (34) | 9 (6) | 14 (48) | 9 (14) | 0 (0) | 0 (0) | |||
| Vitamin D supplementation e, n (%) | 0.19 | 0.17 | 1 | ||||||
| Yes | 0 (0) | 6 (7) | 0 (0) | 6 (9) | 0 (0) | 0 (0) | |||
| No | 30 (100) | 78 (93) | 29 (100) | 58 (91) | 1 (100) | 20 (100) | |||
| Unknown | 11 | 60 | 11 | 60 | |||||
| Entire Cohort (n = 185) | Inpatient Subgroup (n = 93) | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| IMV/D (Events, n = 28) | Death (Events, n = 16) | IMV/D (Events, n = 28) | Death (Events, n = 16) | ||||||
| HR (95% CI) | p | HR (95% CI) | p | HR (95% CI) | p | HR (95% CI) | p | ||
| Covariate | Effect | ||||||||
| VitD | <12 ng/mL | 6.12 (2.79–13.42) | <0.001 | 14.73 (4.16–52.19) | <0.001 | 4.65 (2.11–10.25) | <0.001 | 11.51 (3.24–40.92) | <0.001 |
| Age | ≥60 years | 3.20 (1.05–9.71) | 0.04 | 7.70 (0.99–58.81) | 0.05 | 3.40 (1.11–10.41) | 0.03 | 8.71 (1.11–68.18) | 0.04 |
| Gender | Male | 1.69 (0.76–3.76) | 0.20 | 2.50 (0.80–7.81) | 0.12 | 1.40 (0.62–3.19) | 0.41 | 2.15 (0.68–6.78) | 0.19 |
| Comorbidity | Any | 2.70 (1.04–7.04) | 0.04 | 5.30 (0.96–19.27) | 0.06 | 1.55 (0.59–4.09) | 0.37 | 2.60 (0.58–11.73) | 0.21 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Radujkovic, A.; Hippchen, T.; Tiwari-Heckler, S.; Dreher, S.; Boxberger, M.; Merle, U. Vitamin D Deficiency and Outcome of COVID-19 Patients. Nutrients 2020, 12, 2757. https://doi.org/10.3390/nu12092757
Radujkovic A, Hippchen T, Tiwari-Heckler S, Dreher S, Boxberger M, Merle U. Vitamin D Deficiency and Outcome of COVID-19 Patients. Nutrients. 2020; 12(9):2757. https://doi.org/10.3390/nu12092757
Chicago/Turabian StyleRadujkovic, Aleksandar, Theresa Hippchen, Shilpa Tiwari-Heckler, Saida Dreher, Monica Boxberger, and Uta Merle. 2020. "Vitamin D Deficiency and Outcome of COVID-19 Patients" Nutrients 12, no. 9: 2757. https://doi.org/10.3390/nu12092757
APA StyleRadujkovic, A., Hippchen, T., Tiwari-Heckler, S., Dreher, S., Boxberger, M., & Merle, U. (2020). Vitamin D Deficiency and Outcome of COVID-19 Patients. Nutrients, 12(9), 2757. https://doi.org/10.3390/nu12092757





