Type-4 Resistant Starch in Substitution for Available Carbohydrate Reduces Postprandial Glycemic Response and Hunger in Acute, Randomized, Double-Blind, Controlled Study
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Design and Study Visits
2.2. Study Subjects
2.3. Study Foods
2.4. Measurements
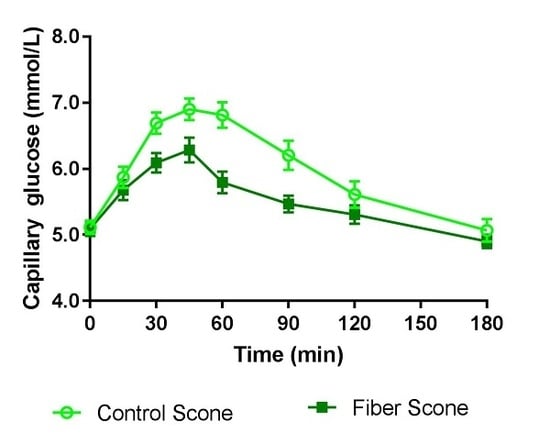
2.4.1. Capillary Glucose
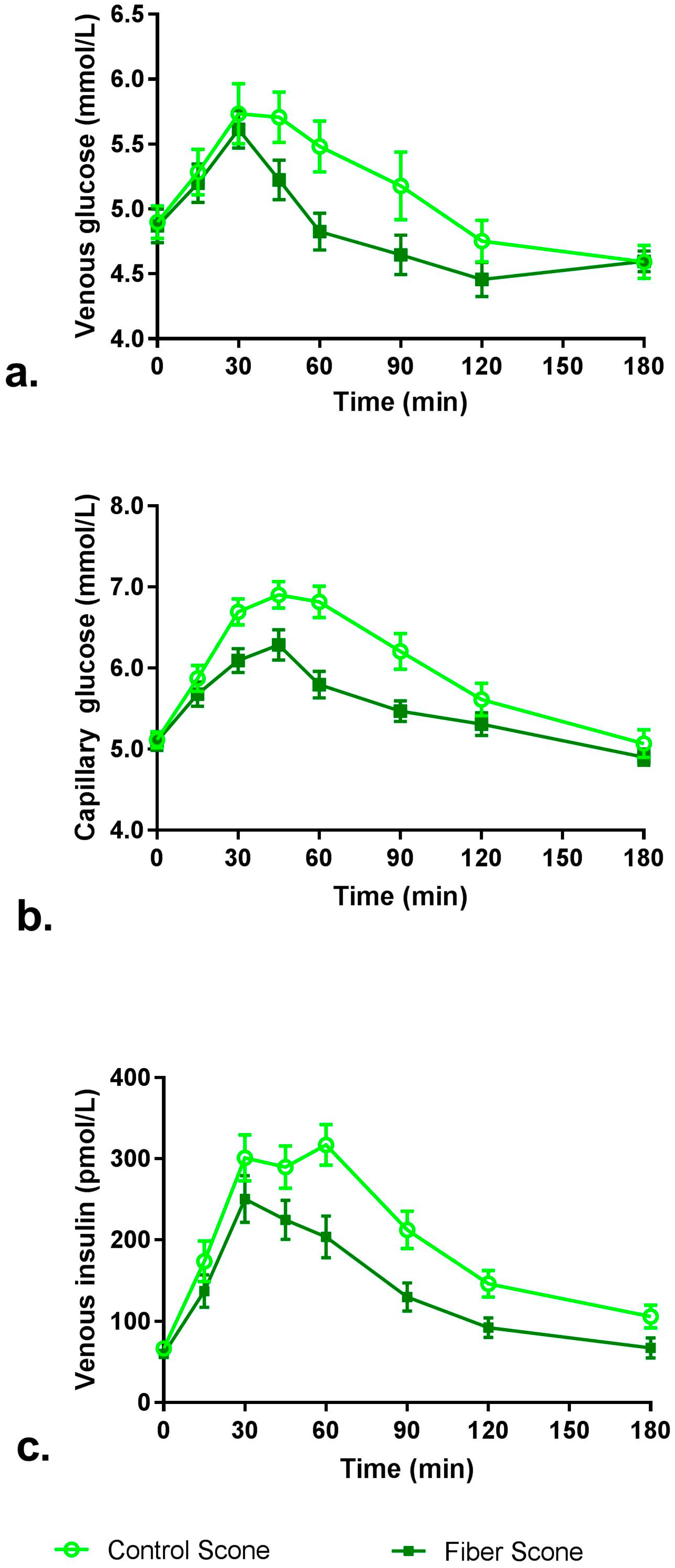
2.4.2. Venous Glucose and Insulin
2.4.3. Satiety Visual Analog Scales (VAS)
2.4.4. Gastrointestinal (GI) Tolerability Questionnaire
2.4.5. Palatability
2.5. Data Analysis
2.5.1. Outcome Variables
2.5.2. Sample Size
2.5.3. Statistical Analyses
3. Results
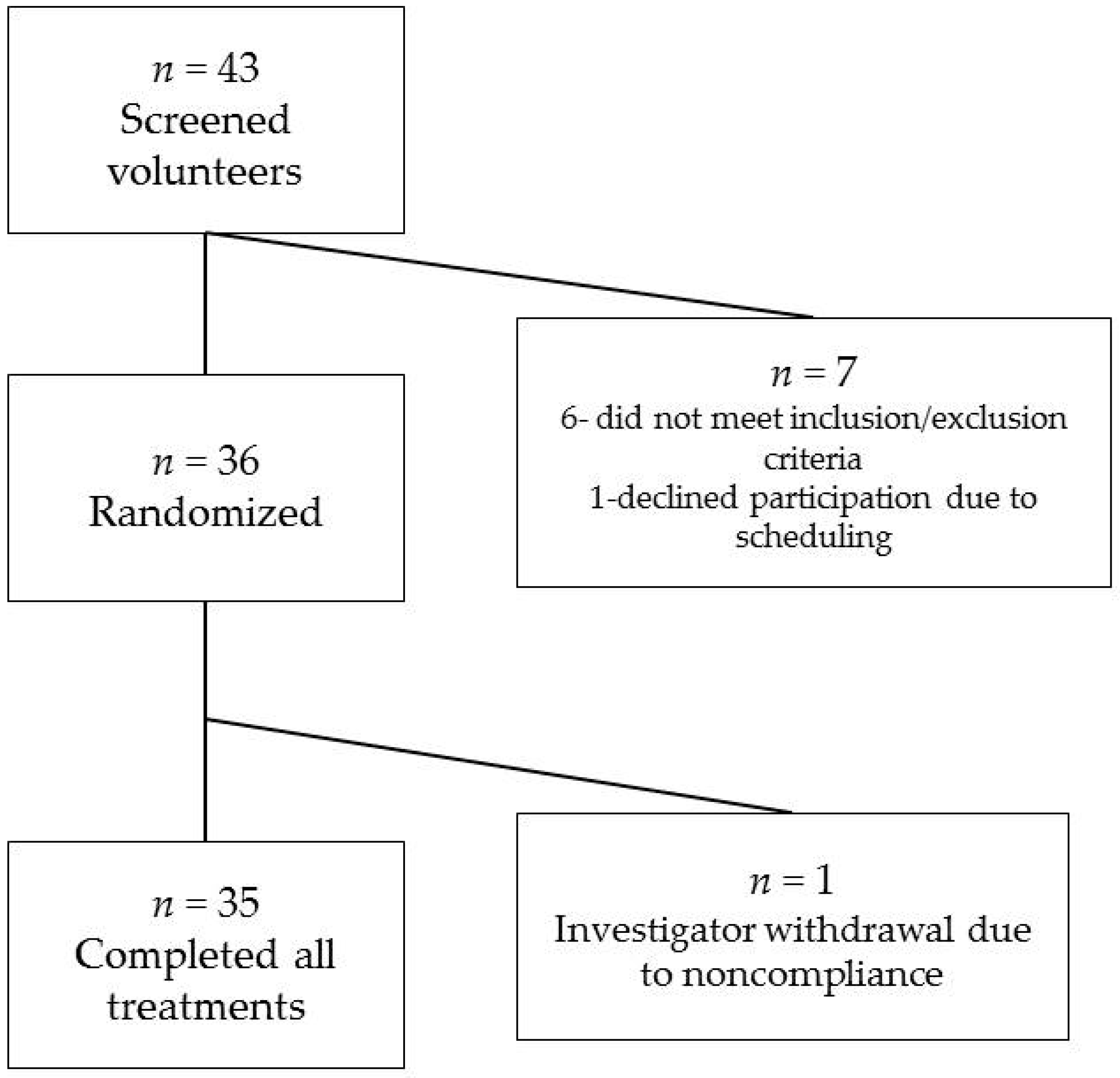
3.1. Study Subjects
3.2. Blood Glucose and Insulin
3.3. Satiety VAS Scores
3.4. GI Tolerability Questionnaire
3.5. Study Product Palatability
4. Discussion
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Dahl, W.J.; Stewart, M.L. Position of the academy of nutrition and dietetics: Health implications of dietary fiber. J. Acad. Nutr. Diet. 2015, 115, 1861–1870. [Google Scholar] [CrossRef] [PubMed]
- Stephen, A.M.; Champ, M.M.; Cloran, S.J.; Fleith, M.; van Lieshout, L.; Mejborn, H.; Burley, V.J. Dietary fibre in Europe: Current state of knowledge on definitions, sources, recommendations, intakes and relationships to health. Nutr. Res. Rev. 2017, 30, 1–42. [Google Scholar] [CrossRef] [PubMed]
- Tosh, S.M. Review of human studies investigating the post-prandial blood-glucose lowering ability of oat and barley food products. Eur. J. Clin. Nutr. 2013, 67, 310–317. [Google Scholar] [CrossRef] [PubMed]
- Robertson, M.D. Dietary-resistant starch and glucose metabolism. Curr. Opin. Clin. Nutr. Metab. Care 2012, 15, 362–367. [Google Scholar] [CrossRef] [PubMed]
- Jiang, J.; Zhao, L.; Lin, L.; Gui, M.; Aleteng, Q.; Wu, B.; Wang, S.; Pan, B.; Ling, Y.; Gao, X. Postprandial blood glucose outweighs fasting blood glucose and hba1c in screening coronary heart disease. Sci. Rep. 2017, 7, 14212. [Google Scholar] [CrossRef] [PubMed]
- Lockyer, S.; Nugent, A.P. Health effects of resistant starch. Nutr. Bull. 2017, 42, 10–42. [Google Scholar] [CrossRef]
- Maningat, C.C.; Seib, P.A. RS4-type resistant starch: Chemistry, functionality and health benefits. In Resistant Starch: Sources, Applications and Health Benefits; Shi, Y.C., Maningat, C.C., Eds.; John Wiley & Sons: Chichester, UK, 2013; pp. 43–77. [Google Scholar]
- Haub, M.D.; Hubach, K.L.; Al-Tamimi, E.K.; Ornelas, S.; Seib, P.A. Different types of resistant starch elicit different glucose reponses in humans. J. Nutr. Metab. 2010, 2010, 230501. [Google Scholar] [CrossRef] [PubMed]
- Al-Tamimi, E.K.; Seib, P.A.; Snyder, B.S.; Haub, M.D. Consumption of cross-linked resistant starch (RS4XL) on glucose and insulin responses in humans. J. Nutr. Metab. 2010, 2010, 651063. [Google Scholar] [CrossRef] [PubMed]
- Martínez, I.; Kim, J.H.; Duffy, P.R.; Schlegel, V.L.; Walter, J. Resistant starches types 2 and 4 have differential effects on the composition of the fecal microbiota in human subjects. PLoS ONE 2010, 5, e15046. [Google Scholar] [CrossRef] [PubMed]
- Karalus, M.; Clark, M.; Greaves, K.A.; Thomas, W.; Vickers, Z.; Kuyama, M.; Slavin, J. Fermentable fibers do not affect satiety or food intake by women who do not practice restrained eating. J. Acad. Nutr. Diet. 2012, 112, 1356–1362. [Google Scholar] [CrossRef] [PubMed]
- Nichenametla, S.N.; Weidauer, L.A.; Wey, H.E.; Beare, T.M.; Specker, B.L.; Dey, M. Resistant starch type 4-enriched diet lowered blood cholesterols and improved body composition in a double blind controlled cross-over intervention. Mol. Nutr. Food Res. 2014, 58, 1365–1369. [Google Scholar] [CrossRef] [PubMed]
- Upadhyaya, B.; McCormack, L.; Fardin-Kia, A.R.; Juenemann, R.; Nichenametla, S.; Clapper, J.; Specker, B.; Dey, M. Impact of dietary resistant starch type 4 on human gut microbiota and immunometabolic functions. Sci. Rep. 2016, 6, 28797. [Google Scholar] [CrossRef] [PubMed]
- Haub, M.D.; Louk, J.A.; Lopez, T.C. Novel resistant potato starches on glycemia and satiety in humans. J. Nutr. Metab. 2012, 2012, 478043. [Google Scholar] [CrossRef] [PubMed]
- Dahl, W.J.; Ford, A.L.; Ukhanova, M.; Radford, A.; Christman, M.C.; Waugh, S.; Mai, V. Resistant potato starches (type 4 RS) exhibit varying effects on laxation with and without phylum level changes in microbiota: A randomised trial in young adults. J. Funct. Foods 2016, 23, 1–11. [Google Scholar] [CrossRef]
- Stewart, M.L.; Zimmer, J.P. A high fiber cookie made with resistant starch type 4 reduces post-prandial glucose and insulin responses in healthy adults. Nutrients 2017, 9, 237. [Google Scholar] [CrossRef] [PubMed]
- Shimotoyodome, A.; Suzuki, J.; Kameo, Y.; Hase, T. Dietary supplementation with hydroxypropyl-distarch phosphate from waxy maize starch increases resting energy expenditure by lowering the postprandial glucose-dependent insulinotropic polypeptide response in human subjects. Br. J. Nutr. 2011, 106, 96–104. [Google Scholar] [CrossRef] [PubMed]
- Gentile, C.L.; Ward, E.; Holst, J.J.; Astrup, A.; Ormsbee, M.J.; Connelly, S.; Arciero, P.J. Resistant starch and protein intake enhances fat oxidation and feelings of fullness in lean and overweight/obese women. Nutr. J. 2015, 14, 113. [Google Scholar] [CrossRef] [PubMed]
- Stewart, M.L.; Zimmer, J.P. Post-prandial glucose and insulin response to high-fiber muffin top containing resistant starch type 4 in healthy adults: A double-blind, randomized, controlled trial. Nutrition 2018, in press. [Google Scholar]
- Cassidy, J.P.; Luzio, S.D.; Marino, M.T.; Baughman, R.A. Quantification of human serum insulin concentrations in clinical pharmacokinetic or bioequivalence studies: What defines the “best method”? Clin. Chem. Lab. Med. 2012, 50, 663–666. [Google Scholar] [CrossRef] [PubMed]
- Maki, K.C.; Rains, T.M.; Kelley, K.M.; Cook, C.M.; Schild, A.L.; Gietl, E. Fibermalt is well tolerated in healthy men and women at intakes up to 60 g/d: A randomized, double-blind, crossover trial. Int. J. Food Sci. Nutr. 2013, 64, 274–281. [Google Scholar] [CrossRef] [PubMed]
- Mialon, V.S.; Clark, M.R.; Leppard, P.I.; Cox, D.N. The effect of dietary fibre information on consumer responses to breads and “English” muffins: A cross-cultural study. Food Qual. Preference 2002, 13, 1–12. [Google Scholar] [CrossRef]
- Baixauli, R.; Salvador, A.; Martínez-Cervera, S.; Fiszman, S.M. Distinctive sensory features introduced by resistant starch in baked products. LWT Food Sci. Technol. 2008, 41, 1927–1933. [Google Scholar] [CrossRef]
- Brouns, F.; Bjorck, I.; Frayn, K.N.; Gibbs, A.L.; Lang, V.; Slama, G.; Wolever, T.M. Glycaemic index methodology. Nutr. Res. Rev. 2005, 18, 145–171. [Google Scholar] [CrossRef] [PubMed]
- Wolever, T.M.; Vorster, H.H.; Bjorck, I.; Brand-Miller, J.; Brighenti, F.; Mann, J.I.; Ramdath, D.D.; Granfeldt, Y.; Holt, S.; Perry, T.L.; et al. Determination of the glycaemic index of foods: Interlaboratory study. Eur. J. Clin. Nutr. 2003, 57, 475–482. [Google Scholar] [CrossRef] [PubMed]
- Augustin, L.S.; Kendall, C.W.; Jenkins, D.J.; Willett, W.C.; Astrup, A.; Barclay, A.W.; Bjorck, I.; Brand-Miller, J.C.; Brighenti, F.; Buyken, A.E.; et al. Glycemic index, glycemic load and glycemic response: An International Scientific Consensus Summit from the International Carbohydrate Quality Consortium (ICQC). Nutr. Metab. Cardiovasc. Dis. 2015, 25, 795–815. [Google Scholar] [CrossRef] [PubMed]
| Per Serving, As-Eaten | Control Scone | Fiber Scone |
|---|---|---|
| Weight (g) | 83.9 | 84.1 |
| Calories (kcal) | 328 | 270 |
| Fat (g) | 16.0 | 14.4 |
| Saturated fat (g) | 5.0 | 4.7 |
| Protein (g) | 7.1 | 6.1 |
| Total Carbohydrates (g) | 42.8 | 46.4 |
| Available Carbohydrates (g) | 38.8 | 28.9 |
| Dietary Fiber (g) * | 4.0 | 17.5 |
| Sugars (g) | 14.8 | 14.9 |
| Variable | All Participants (n = 35) 1 |
|---|---|
| Age (y) | 46.2 ± 2.2 |
| Sex (male/female) | 12/23 |
| Race (white/nonwhite) | 22/13 |
| Ethnicity (non-Hispanic/Hispanic) | 26/9 |
| Weight (kg) | 73.9 ± 2.1 |
| Body mass index (kg/m2) | 26.1 ± 0.5 |
| Fasting capillary glucose (mmol/L) | 5.04 ± 0.09 |
| Parameter | Control Scone | Fiber Scone | p-Value 3 |
|---|---|---|---|
| Venous blood glucose | |||
| iAUC0–120 min (min × mmol/L) | 69.65 ± 9.05 | 38.41 ± 5.77 | 0.0014 |
| iAUC0–180 min (min × mmol/L) 4 | 84.75 ± 11.43 | 48.29 ± 9.55 | 0.0039 |
| Cmax (mmol/L) 4 | 6.44 ± 0.18 | 5.88 ± 0.12 | 0.0039 |
| Capillary blood glucose | |||
| iAUC0–120 min (min × mmol/L) 4 | 139.14 ± 14.60 | 79.75 ± 8.10 | 0.0004 |
| iAUC0–180 min (min × mmol/L) 4 | 171.88 ± 19.65 | 94.63 ± 10.66 | 0.0003 |
| Cmax (mmol/L) 4 | 7.49 ± 0.19 | 6.72 ± 0.16 | 0.0002 |
| Venous insulin | |||
| iAUC0–120 min (min × pmol/L) | 19,229 ± 1865 | 12,592 ± 1686 | 0.0005 |
| iAUC0–180 min (min × pmol/L) | 23,850 ± 2138 | 14,192 ± 1901 | <0.0001 |
| Cmax (pmol/L) 4 | 392 ± 28 | 305 ± 26 | 0.0008 |
| Parameter | Control Scone | Fiber Scone | p-Value 3 |
|---|---|---|---|
| Hunger niAUC0–180 min (mm × min) | −1173 ± 896 | −2840 ± 780 | 0.0316 |
| Fullness niAUC0–180 min (mm × min) | 3926 ± 753 | 4028 ± 898 | 0.8858 |
| Desire to eat niAUC0–180 min (mm × min) | −1184 ± 918 | −3046 ± 773 | 0.0135 |
| Prospective consumption niAUC0–180 min (mm × min) | −2098 ± 799 | −2847 ± 672 | 0.1619 |
| Parameter | Control Scone | Fiber Scone | p-Value 4 |
|---|---|---|---|
| Nausea | 0 (0) 3 | 0 (0) | 1.000 |
| Bloating | 1 (2.9) | 0 (0) | 1.000 |
| GI Rumblings 4 | 4 (11.4) | 1 (2.9) | 0.2482 |
| Flatulence | 1 (2.9) | 0 (0.0) | 1.000 |
| Abdominal Pain | 0 (0.0) | 0 (0.0) | 1.000 |
| Diarrhea | 0 (0.0) | 0 (0.0) | 1.000 |
| Parameter | Control Scone | Fiber Scone | p-Value 6 |
|---|---|---|---|
| Appearance 1 | 7.74 ± 0.32 | 7.63 ± 0.32 | 0.4875 |
| Texture 1 | 6.97 ± 0.34 | 6.40 ± 0.36 | 0.0743 |
| Flavor 1 | 7.11 ± 0.04 | 6.69 ± 0.43 | 0.1185 7 |
| Acceptance 1 | 7.17 ± 0.36 | 6.77 ± 0.43 | 0.2271 7 |
| Healthy 2 | 5.91 ± 0.46 | 5.71 ± 0.44 | 0.4205 |
| Nutritious 3 | 5.57 ± 0.44 | 5.23 ± 0.44 | 0.2092 7 |
| Recommend 4 | 0.17 ± 0.20 | 0.09 ± 0.21 | 0.5712 |
| Tolerated 4 | 0.89 ± 0.20 | 0.97 ± 0.16 | 0.8601 7 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Stewart, M.L.; Wilcox, M.L.; Bell, M.; Buggia, M.A.; Maki, K.C. Type-4 Resistant Starch in Substitution for Available Carbohydrate Reduces Postprandial Glycemic Response and Hunger in Acute, Randomized, Double-Blind, Controlled Study. Nutrients 2018, 10, 129. https://doi.org/10.3390/nu10020129
Stewart ML, Wilcox ML, Bell M, Buggia MA, Maki KC. Type-4 Resistant Starch in Substitution for Available Carbohydrate Reduces Postprandial Glycemic Response and Hunger in Acute, Randomized, Double-Blind, Controlled Study. Nutrients. 2018; 10(2):129. https://doi.org/10.3390/nu10020129
Chicago/Turabian StyleStewart, Maria L., Meredith L. Wilcox, Marjorie Bell, Mary A. Buggia, and Kevin C. Maki. 2018. "Type-4 Resistant Starch in Substitution for Available Carbohydrate Reduces Postprandial Glycemic Response and Hunger in Acute, Randomized, Double-Blind, Controlled Study" Nutrients 10, no. 2: 129. https://doi.org/10.3390/nu10020129
APA StyleStewart, M. L., Wilcox, M. L., Bell, M., Buggia, M. A., & Maki, K. C. (2018). Type-4 Resistant Starch in Substitution for Available Carbohydrate Reduces Postprandial Glycemic Response and Hunger in Acute, Randomized, Double-Blind, Controlled Study. Nutrients, 10(2), 129. https://doi.org/10.3390/nu10020129