Development and Optimization of a Fluorescent Differential Display PCR System for Analyzing the Stress Response in Lactobacillus sakei Strains
Abstract
:1. Introduction
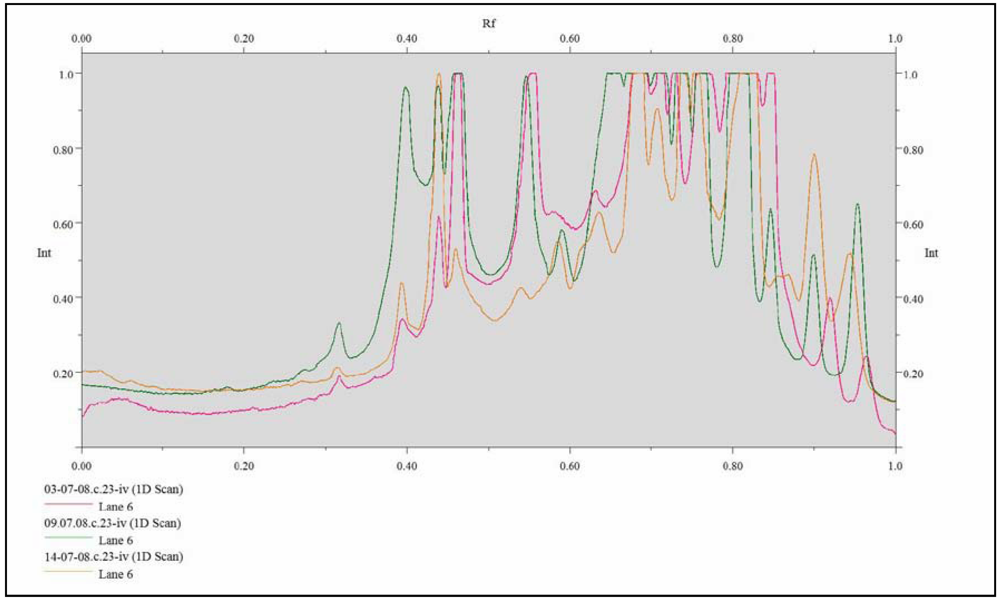
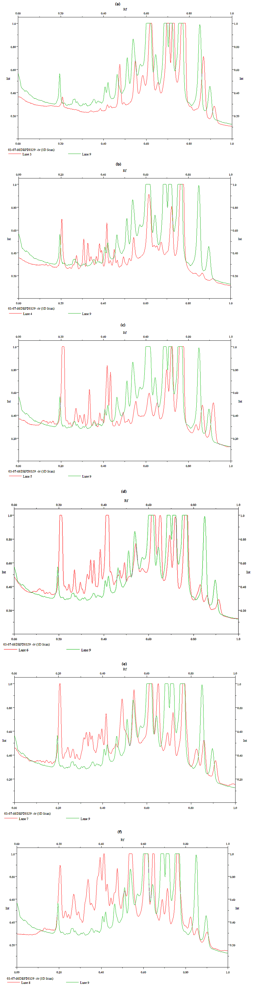
2. Results and Discussion
| DBPZ0098 | ||||||
|---|---|---|---|---|---|---|
| Control | 50°C | 55°C | 60°C | pH 2.5 | pH 3 | NaCl 9% |
| 1673.44 | 1673.44 | 1673.44 | 1673.44 | |||
| 1328.16 | 1328.16 | 1328.16 | ||||
| 1018.72 | ||||||
| 931.93 | 931.93 | 931.93 | ||||
| 882.75 | 882.75 | 882.75 | 882.75 | 882.75 | 882.75 | |
| 864.42 | 864.42 | 864.42 | ||||
| 834.89 | 834.89 | 834.89 | 834.89 | 834.89 | 834.89 | |
| 784.89 | 784.89 | 784.89 | 784.89 | 784.89 | 784.89 | |
| 749.74 | 749.74 | 749.74 | 749.74 | 749.74 | ||
| 693.97 | 693.97 | 693.97 | 693.97 | 693.97 | 693.97 | 693.97 |
| 663.45 | 663.45 | 663.45 | 663.45 | 663.45 | ||
| 632.92 | 632.92 | |||||
| 587.53 | 587.53 | 587.53 | 587.53 | 587.53 | 587.53 | |
| 550.05 | ||||||
| 530.28 | 530.28 | 530.28 | 530.28 | 530.28 | 530.28 | 530.28 |
| 504.74 | 504.74 | 504.74 | 504.74 | 504.74 | 504.74 | 504.74 |
| 486.83 | 486.83 | |||||
| 469.53 | 469.53 | 469.53 | 469.53 | 469.53 | 469.53 | 469.53 |
| 452.86 | 452.86 | 452.86 | 452.86 | 452.86 | 452.86 | 452.86 |
| 417.27 | 417.27 | 417.27 | 417.27 | 417.27 | 417.27 | 417.27 |
| DBPZ0338 | ||||||
| Control | 50°C | 55°C | 60°C | pH 2.5 | pH 3 | NaCl 9% |
| 2560.91 | 2560.91 | 2560.91 | 2560.91 | 2560.91 | 2560.91 | |
| 1750.63 | ||||||
| 1450.49 | ||||||
| 980.06 | 980.06 | 980.06 | ||||
| 970.73 | 970.73 | |||||
| 851.94 | ||||||
| 773.31 | 773.31 | 773.31 | 773.31 | |||
| 738.01 | ||||||
| 704.32 | 704.32 | 704.32 | 704.32 | 704.32 | 704.32 | |
| 674.71 | 674.71 | 674.71 | 674.71 | 674.71 | ||
| 600.51 | 600.51 | 600.51 | 600.51 | 600.51 | 600.51 | 600.51 |
| 571.08 | 571.08 | 571.08 | 571.08 | 571.08 | 571.08 | 571.08 |
| 532.01 | 532.01 | 532.01 | 532.01 | 532.01 | 532.01 | 532.01 |
| 513.04 | 513.04 | 513.04 | 513.04 | 513.04 | 513.04 | 513.04 |
| 336.57 | 336.57 | 336.57 | 336.57 | 336.57 | 336.57 | 336.57 |
| DBPZ0416 | ||||||
| Control | 50°C | 55°C | 60°C | pH 2.5 | pH 3 | NaCl 9% |
| 6032.41 | ||||||
| 5370.14 | 5370.14 | 5370.14 | 5370.14 | 5370.14 | ||
| 3653.56 | ||||||
| 3292.21 | 3292.21 | 3292.21 | 3292.21 | 3292.21 | ||
| 2721.33 | 2721.33 | 2721.33 | 2721.33 | 2721.33 | ||
| 2473.01 | 2473.01 | 2473.01 | 2473.01 | 2473.01 | 2473.01 | |
| 2383.11 | 2383.11 | 2383.11 | 2383.11 | |||
| 2276.34 | 2276.34 | 2276.34 | ||||
| 2184.14 | 2184.14 | 2184.14 | 2184.14 | 2184.14 | ||
| 2022.33 | 2022.33 | 2022.33 | 2022.33 | 2022.33 | 2022.33 | |
| 1875.22 | 1875.22 | 1875.22 | 1875.22 | 1875.22 | 1875.22 | 1875.22 |
| 1771.13 | 1771.13 | 1771.13 | 1771.13 | 1771.13 | 1771.13 | 1771.13 |
| 1720.02 | 1720.02 | 1720.02 | 1720.02 | 1720.02 | 1720.02 | 1720.02 |
| 1642.11 | 1642.11 | 1642.11 | 1642.11 | 1642.11 | 1642.11 | 1642.11 |
| 1544.23 | 1544.23 | 1544.23 | 1544.23 | 1544.23 | ||
| 1454.23 | 1454.23 | 1454.23 | 1454.23 | 1454.23 | 1454.23 | 1454.23 |
| 1323.41 | 1323.41 | 1323.41 | 1323.41 | 1323.41 | 1323.41 | |
| 1151.11 | 1151.11 | 1151.11 | ||||
| 1074.03 | ||||||
| 880.12 | 880.12 | 880.12 | 880.12 | 880.12 | 880.12 | |
| 863.45 | 863.45 | 863.45 | 863.45 | 863.45 | 863.45 | |
| 740.53 | ||||||
| 643.75 | 643.75 | 643.75 | 643.75 | |||
| 600.21 | 600.21 | 600.21 | 600.21 | 600.21 | 600.21 | 600.21 |
| 576.43 | ||||||
| 512.01 | 512.01 | 512.01 | 512.01 | 512.01 | 512.01 | 512.01 |
| 486.52 | 486.52 | 486.52 | 486.52 | 486.52 | 486.52 | 486.52 |
| 478.59 | 478.59 | 478.59 | 478.59 | |||
| 441.67 | 441.67 | 441.67 | ||||
| 412.34 | 412.34 | 412.34 | 412.34 | |||
| 387.54 | ||||||
| 365.55 | 365.55 | 365.55 | 365.55 | 365.55 | 365.55 | |
| 354.22 | 354.22 | 354.22 | ||||
| 304.53 | 304.53 | |||||
| 291.23 | 291.23 | 291.23 | ||||
| DBPZ0062 | ||||||
| Control | 50°C | 55°C | 60°C | pH 2.5 | pH 3 | NaCl 9% |
| 5461.79 | ||||||
| 3862.88 | 3862.88 | 3862.88 | 3862.88 | |||
| 3333.04 | ||||||
| 2441.51 | 2441.51 | |||||
| 1685.79 | 1685.79 | |||||
| 1503.95 | 1503.95 | 1503.95 | 1503.95 | |||
| 1166.63 | 1166.63 | 1166.63 | 1166.63 | 1166.63 | 1166.63 | |
| 1039.42 | 1039.42 | |||||
| 964.55 | ||||||
| 901.32 | 901.32 | 901.32 | 901.32 | 901.32 | 901.32 | 901.32 |
| 870.74 | 870.74 | 870.74 | ||||
| 840.71 | 840.71 | |||||
| 796.05 | 796.05 | 796.05 | 796.05 | 796.05 | ||
| 705.81 | ||||||
| 687.53 | 687.53 | 687.53 | 687.53 | 687.53 | 687.53 | 687.53 |
| 649.93 | 649.93 | 649.93 | 649.93 | 649.93 | 649.93 | |
| 636.22 | 636.22 | |||||
| 615.23 | ||||||
| 607.29 | 607.29 | 607.29 | 607.29 | 607.29 | 607.29 | 607.29 |
| 584.36 | 584.36 | 584.36 | 584.36 | 584.36 | 584.36 | 584.36 |
| 543.07 | 543.07 | 543.07 | 543.07 | 543.07 | 543.07 | 543.07 |
| 511.96 | 511.96 | 511.96 | 511.96 | 511.96 | 511.96 | |
| 497.97 | 497.97 | |||||
| 472.33 | 472.33 | 472.33 | 472.33 | |||
| 451.2 | ||||||
| 433.57 | 433.57 | 433.57 | 433.57 | 433.57 | 433.57 | 433.57 |
| DBPZ0329 | ||||||
| Control | 50°C | 55°C | 60°C | pH 2.5 | pH 3 | NaCl 9% |
| 1722.17 | 1722.17 | |||||
| 1468.41 | ||||||
| 1422.94 | ||||||
| 1308.32 | ||||||
| 1022.48 | 1022.48 | 1022.48 | 1022.48 | 1022.48 | 1022.48 | 1022.48 |
| 965.71 | 965.71 | |||||
| 943.36 | 943.36 | 943.36 | 943.36 | 943.36 | ||
| 873.34 | 873.34 | 873.34 | 873.34 | 873.34 | 873.34 | |
| 840.95 | ||||||
| 805.84 | 805.84 | |||||
| 779.32 | 779.32 | |||||
| 759.71 | 759.71 | 759.71 | ||||
| 746.03 | 746.03 | 746.03 | ||||
| 708.68 | 708.68 | 708.68 | ||||
| 702.39 | 702.39 | 702.39 | 702.39 | |||
| 697.99 | 697.99 | 697.99 | 697.99 | 697.99 | ||
| 673.84 | ||||||
| 663.54 | 663.54 | 663.54 | 663.54 | 663.54 | ||
| 659.26 | 659.26 | 659.26 | 659.26 | 659.26 | ||
| 624.48 | 624.48 | 624.48 | 624.48 | 624.48 | 624.48 | 624.48 |
| 597.42 | 597.42 | 597.42 | ||||
| 570.58 | 570.58 | 570.58 | ||||
| 548.1 | 548.1 | 548.1 | 548.1 | 548.1 | 548.1 | 548.1 |
| 546.18 | 546.18 | |||||
| 527.78 | 527.78 | 527.78 | 527.78 | |||
| 522.14 | 522.14 | 522.14 | 522.14 | 522.14 | ||
| 501.79 | 501.79 | 501.79 | 501.79 | 501.79 | 501.79 | 501.79 |
| 494.21 | 494.21 | 494.21 | ||||
| 479.58 | 479.58 | 479.58 | 479.58 | 479.58 | 479.58 | 479.58 |
| 470.79 | ||||||
| 459.58 | 459.58 | 459.58 | 459.58 | 459.58 | 459.58 | 459.58 |
| 448.37 | ||||||
| 414.21 | 414.21 | 414.21 | 414.21 | 414.21 | 414.21 | |
| 389.06 | 389.06 | 389.06 | 389.06 | 389.06 | 389.06 | 389.06 |
3. Experimental Section
3.1. Bacterial Strains and Culture Conditions
3.2. Stress Treatments
3.3. Fluorescent Differential Display (FDD)
4. Conclusions
References and Notes
- Leroy, F.; Verluyten, J.; De Vuyst, L. Functional meat starter cultures for improved sausage fermentation. Int. J. Food Microbiol. 2006, 106, 270–285. [Google Scholar]
- Marceau, A.; Zagorec, M.; Chaillou, S.; Mera, T.; Champomier-Verges, M.C. Evidence for involvement of at least six proteins in adaptation of Lactobacillus sakei to cold temperatures and addition of NaCl. Appl. Environ. Microbiol. 2004, 70, 7260–7268. [Google Scholar] [CrossRef] [PubMed]
- Chaillou, S.; Champomier-Verges, M.C.; Cornet, M.; Crutz-Le Coq, A.M.; Dudez, A.M.; Martin, V.; Beaufils, S.; Darbon-Rongere, E. The complete genome sequence of the meat-borne lactic acid bacterium Lactobacillus sakei 23K. Nat. Biotechnol. 2005, 23, 1527–1533. [Google Scholar] [PubMed]
- Hufner, E.; Markieton, T.; Chaillou, S.; Crutz-Le Coq, A.M.; Zagorec, M.; Hertel, C. Identification of Lactobacillus sakei genes induced during meat fermentation and their role in survival and growth. Appl. Environ. Microbiol. 2007, 73, 2522–2531. [Google Scholar] [CrossRef] [PubMed]
- Liang, P.; Pardee, A.B. Differential display of eukaryotic messenger RNA by means of the polymerase chain reaction. Science 1992, 257, 967–971. [Google Scholar]
- Welsh, J.; Chada, K.; Dalal, S.S.; Cheng, R.; Ralph, D.; McClelland, M. Arbitrarily primed PCR fingerprinting of RNA. Nucleic Acids Res. 1992, 20, 4965–4970. [Google Scholar]
- McClelland, M.; Mathieu-Daude, F.; Welsh, J. RNA fingerprinting and differential display using arbitrarily primed PCR. Trends Genet. 1995, 11, 242–246. [Google Scholar] [CrossRef] [PubMed]
- Wong, K.K.; McClelland, M. Stress-inducible gene of Salmonella typhimurium identified by arbitrarily primed PCR of RNA. Proc. Natl. Acad. Sci. USA 1994, 91, 639–643. [Google Scholar] [CrossRef]
- Shepard, B.D.; Gilmore, M.S. Identification of aerobically and anaerobically induced genes in Enterococcus faecalis by random arbitrarily primed PCR. Appl. Environ. Microbiol. 1999, 65, 1470–1476. [Google Scholar] [PubMed]
- Du, L.D.; Kolenbrander, P.E. Identification of saliva-regulated genes of Streptococcus gordonii DL1 by differential display using random arbitrarily primed PCR. Infec. Immunity 2000, 68, 4834–4837. [Google Scholar] [CrossRef]
- Chia, J.S.; Lee, Y.Y.; Huang, P.T.; Chen, J.Y. Identification of stress-responsive genes in Streptococcus mutans by differential display reverse transcription-PCR. Infec. Immunity 2001, 69, 2493–2501. [Google Scholar]
- Le Breton, Y.; Muller, C.; Auffray, Y.; Rincé, A. New insights into the Enterococcus faecalis CroRS two-component system obtained using a differential-display random arbitrarily primed PCR approach. Appl. Environ. Microbiol. 2007, 73, 3738–3741. [Google Scholar] [CrossRef] [PubMed]
- Lechiancole, T.; Blaiotta, G.; Messina, D.; Fusco, V.; Villani, F.; Salzano, G. Evaluation of intra-specific diversities in Oenococcus oeni through analysis of genomic and expressed DNA. Syst. Appl. Microbiol. 2006, 29, 375–381. [Google Scholar] [CrossRef] [PubMed]
- Reinhardt, B.; Frank, U.; Gellner, K.; Lohmann, J.U. High-resolution, fluorescence based differential display on a DNA sequencer followed by band excision. Biotechniques 1999, 27, 268–271. [Google Scholar]
- Cho, Y.J.; Meade, J.D.; Walden, J.C.; Chen, X. Multicolor fluorescent differential display. Biotechnique 2001, 30, 562–572. [Google Scholar]
- Liang, P.; Meade, D.; Pardee, A.B. A protocol for differential display of mRNA expression using either fluorescent or radioactive labeling. Nature Protocols 2007, 2, 457–470. [Google Scholar]
- Lockyer, A.E.; Noble, L.R.; Rollinson, D.; Jones, C.S. Schistosoma mansoni: resistant specific infection-induced gene expression in Biomphalaria glabrata identified by fluorescent-based differential display. Exp. Parasitol. 2004, 107, 97–104. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.P.; Chen, K.P.; Yao, Q.; Liu, X.Y. Fluorescent differential display analysis of gene expression for NPV resistance in Bombyx mori L. JEN 2005, 129, 27–31. [Google Scholar]
- Shimizu, M.; Kosaka, N.; Shimada, T.; Nagahata, T.; Iwasaki, H.; Nagai, H.; Shiba, T.; Emi, M. Universal fluorescent labeling (UFL) method for automated microsatellite analysis. DNA Res. 2002, 9, 173–178. [Google Scholar]
- Ripamonte, P.; Krempel Merighe, G.; Watanabe, Y.F.; Caetano, A.R.; Vieira Meirelles, F. Development and optimization of a fluorescent differential display PCR system for studying bovine embryo development in vitro. Gen. Mol. Res. 2005, 4, 726–733. [Google Scholar]
- Keiji, K.; Takashi, I.; Yoshiyuki, S. Fluorescent differential display analysis of expression in differentiating neuroblastoma cells. Gene 1997, 184, 73–81. [Google Scholar]
- Dong-Kug, C.; Takashi, I.; Yuko, M.; Yoshiyuki, S. Fluorescent differential display analysis of expression inapoptotic neuroblastoma cells. Gene 1997, 223, 21–31. [Google Scholar]
- Gevers, D.; Danielsen, M.; Huys, G.; Swings, J. Molecular characterization of tet(M) genes in Lactobacillus isolates from different types of fermented dry sausage. Appl. Environ. Microbiol. 2003, 69, 1270–1275. [Google Scholar] [CrossRef] [PubMed]
- Champomier-Verges, M.C.; Marceau, A.; Mera, T.; Zagorec, M. The pepR gene of Lactobacillus sakei is positively regulated by anaerobiosis at the transcriptional level. Appl. Environ. Microbiol. 2002, 68, 3873–3877. [Google Scholar] [CrossRef] [PubMed]
- Axelsson, L.; Lindstad, G.; Naterstad, K. Development of an inducible gene expression system for Lactobacillus sakei. Lett. Appl. Microbiol. 2003, 37, 115–120. [Google Scholar] [CrossRef] [PubMed]
- Bonomo, M.G.; Ricciardi, A.; Zotta, T.; Parente, E.; Salzano, G. Molecular and technological characterization of lactic acid bacteria from traditional fermented sausages of Basilicata region (Southern Italy). Meat Sci. 2008, 80, 1238–1248. [Google Scholar]
© 2009 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Bonomo, M.G.; Sico, M.A.; Grieco, S.; Salzano, G. Development and Optimization of a Fluorescent Differential Display PCR System for Analyzing the Stress Response in Lactobacillus sakei Strains. Nutrients 2009, 1, 210-223. https://doi.org/10.3390/nu1020210
Bonomo MG, Sico MA, Grieco S, Salzano G. Development and Optimization of a Fluorescent Differential Display PCR System for Analyzing the Stress Response in Lactobacillus sakei Strains. Nutrients. 2009; 1(2):210-223. https://doi.org/10.3390/nu1020210
Chicago/Turabian StyleBonomo, Maria Grazia, Maria Anna Sico, Simona Grieco, and Giovanni Salzano. 2009. "Development and Optimization of a Fluorescent Differential Display PCR System for Analyzing the Stress Response in Lactobacillus sakei Strains" Nutrients 1, no. 2: 210-223. https://doi.org/10.3390/nu1020210
APA StyleBonomo, M. G., Sico, M. A., Grieco, S., & Salzano, G. (2009). Development and Optimization of a Fluorescent Differential Display PCR System for Analyzing the Stress Response in Lactobacillus sakei Strains. Nutrients, 1(2), 210-223. https://doi.org/10.3390/nu1020210





