Using Remote-Sensing Environmental and Fishery Data to Map Potential Yellowfin Tuna Habitats in the Tropical Pacific Ocean
Abstract
:1. Introduction
2. Data and Methods
2.1. Yellowfin Tuna Fishery Data
2.2. Remotely Sensed Environmental Data
2.3. Statistical Models for Spatial Predictions of Catch Rate
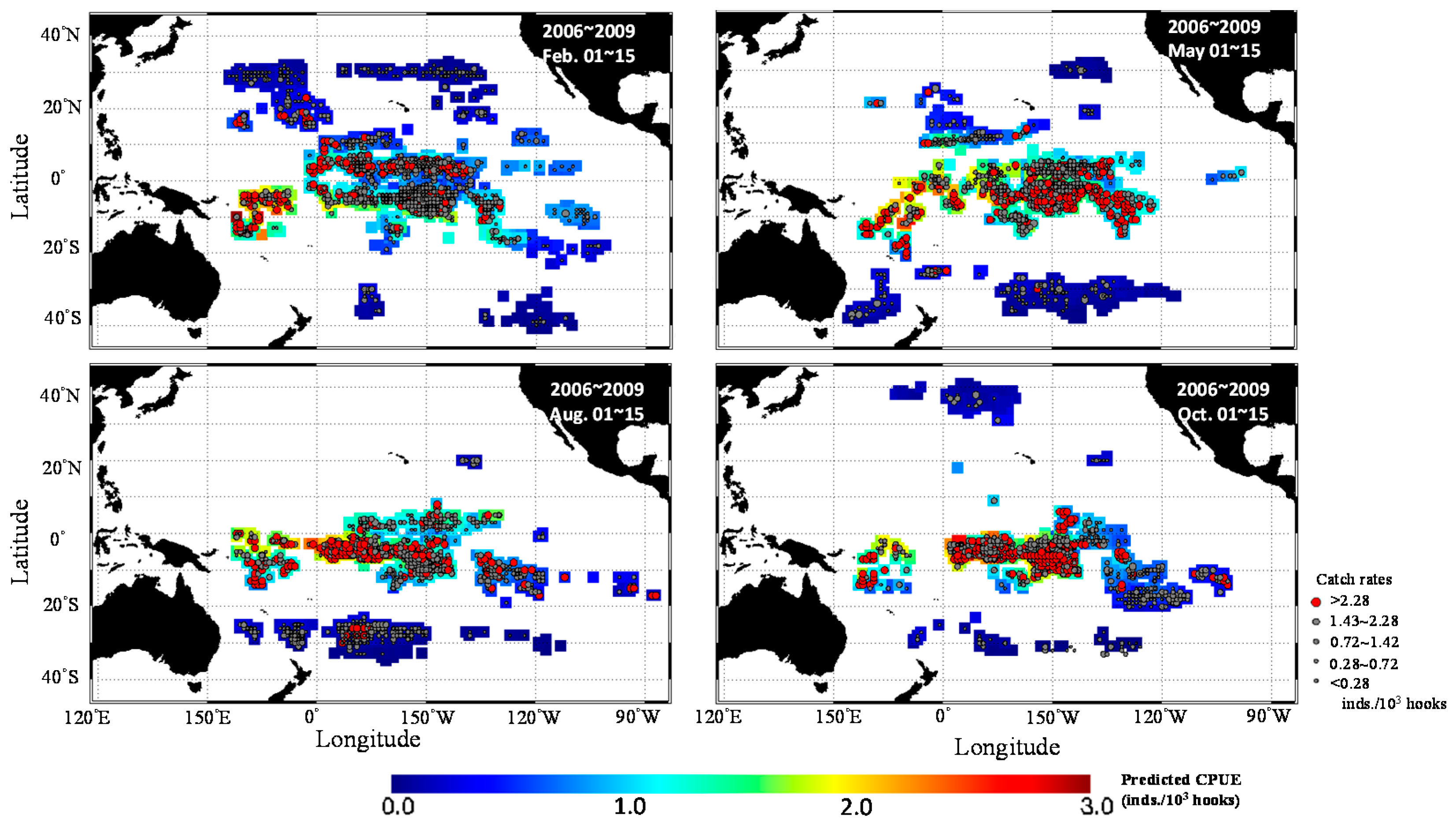
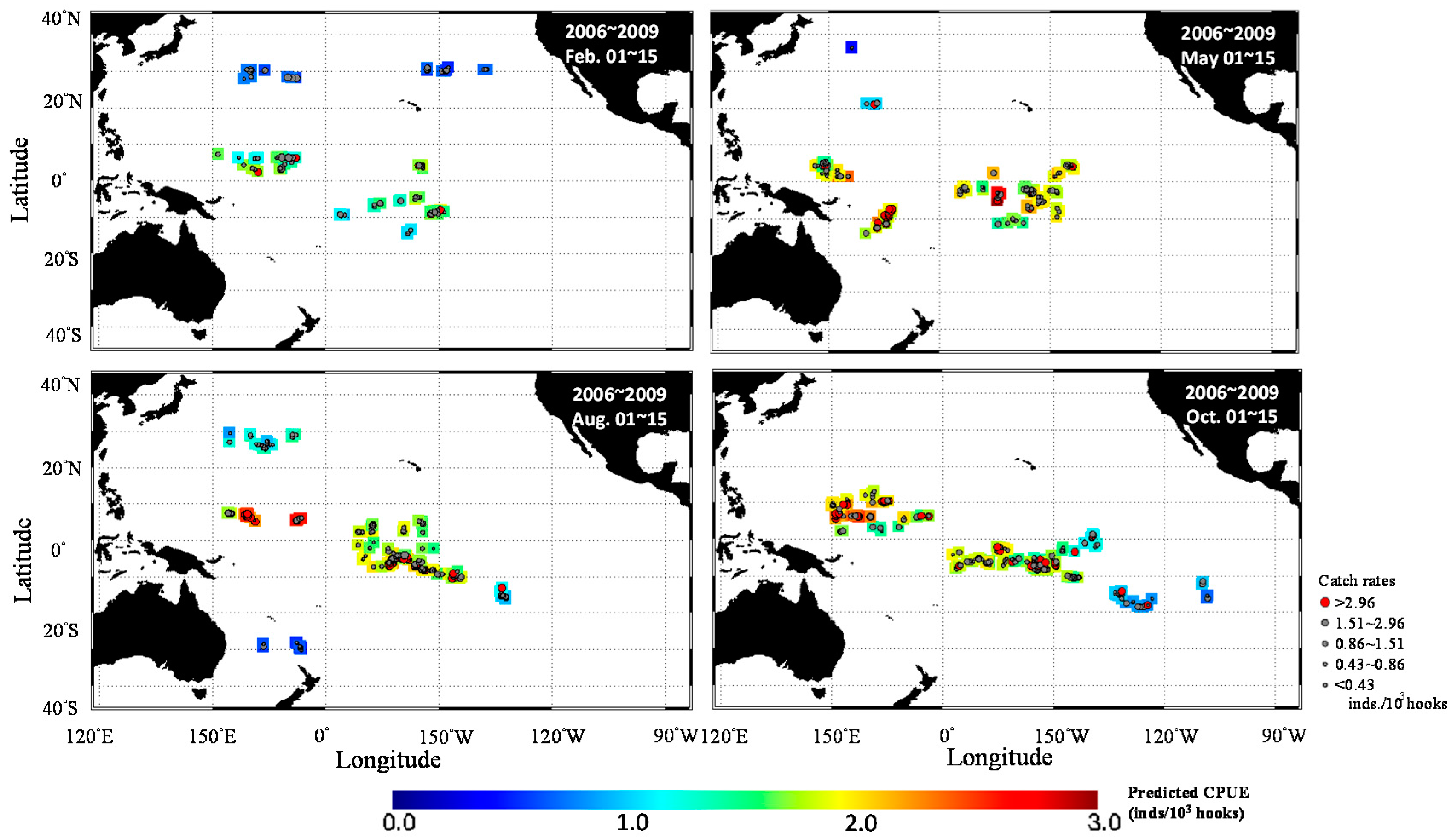
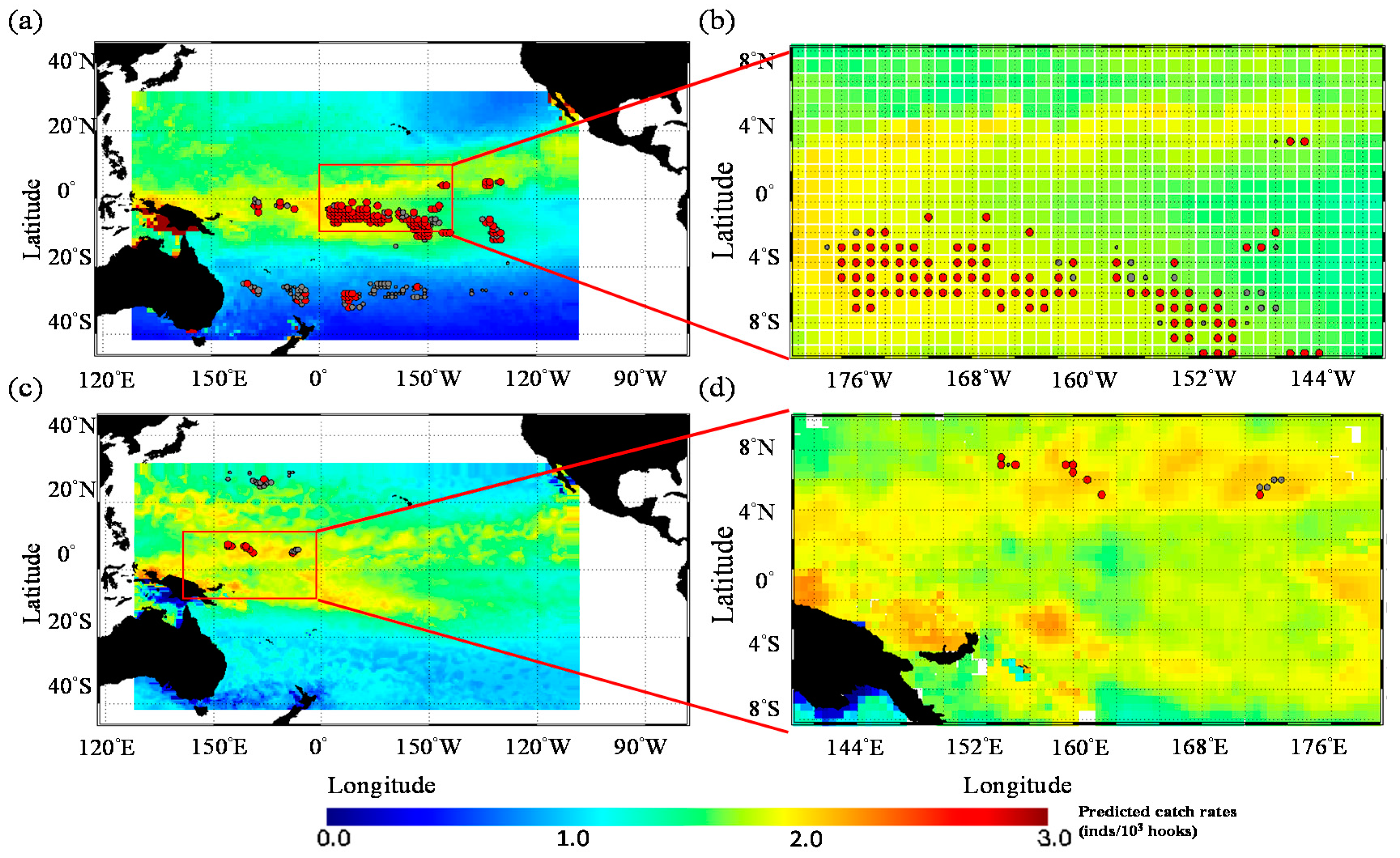
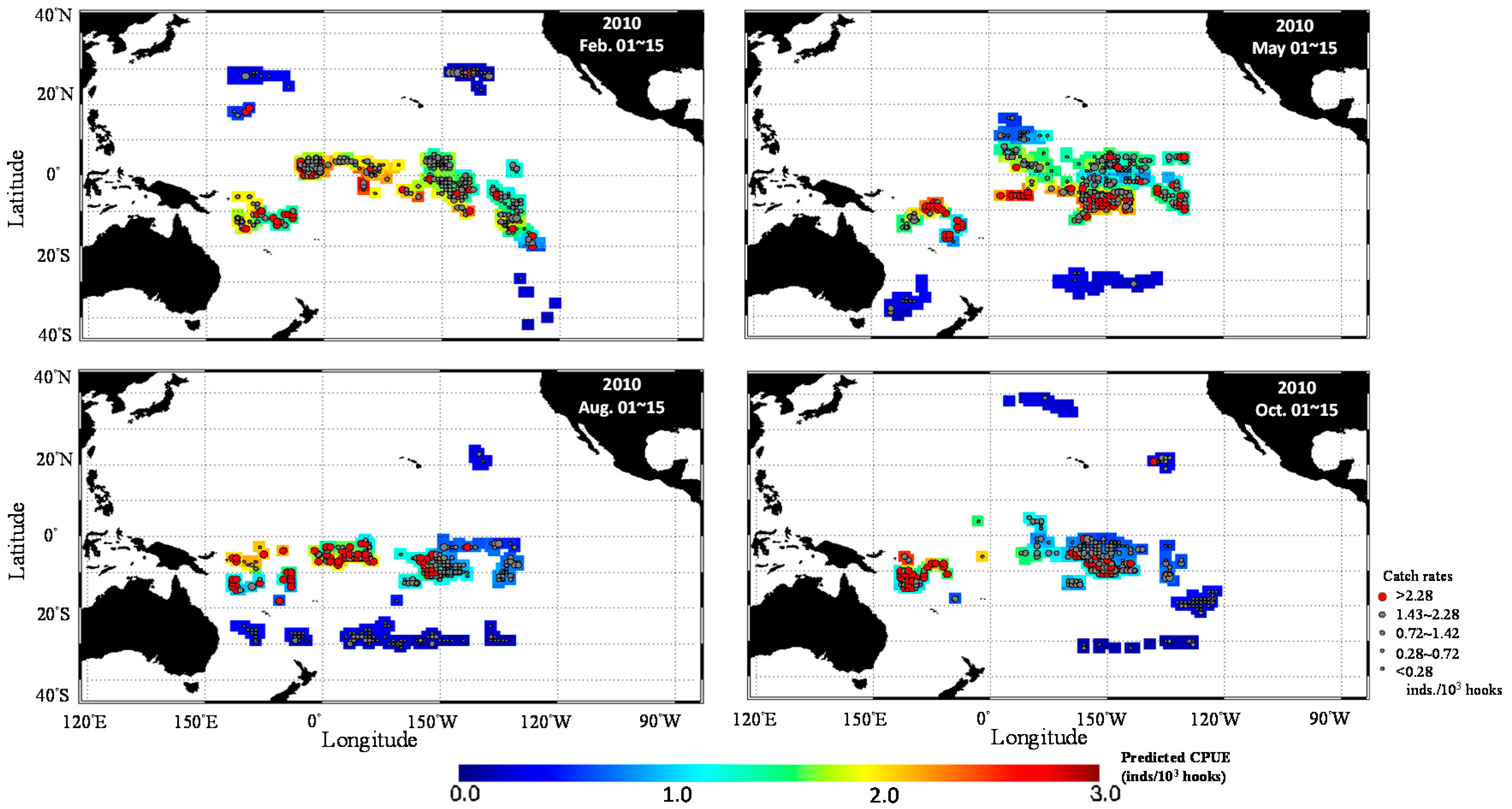
3. Results
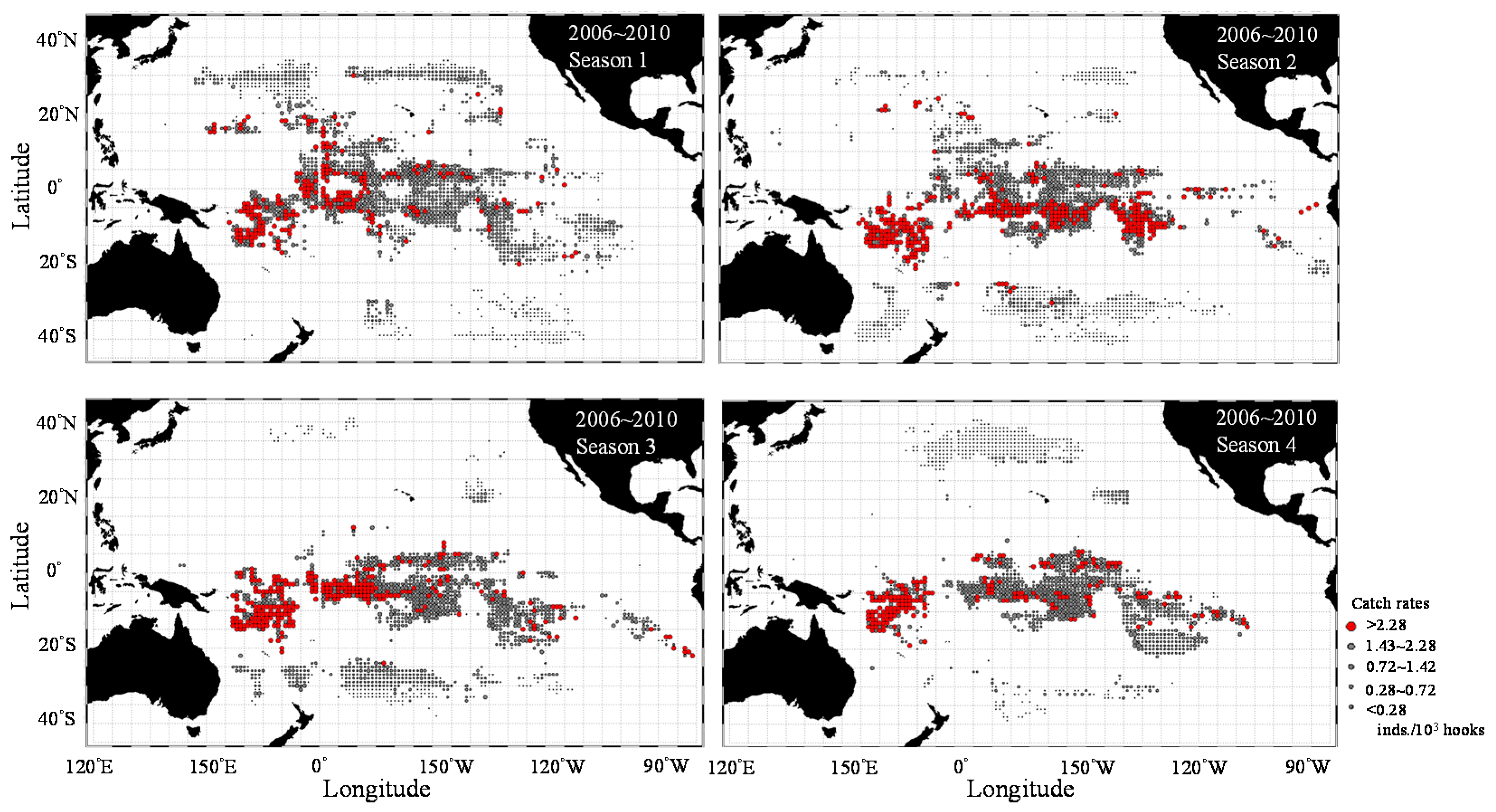
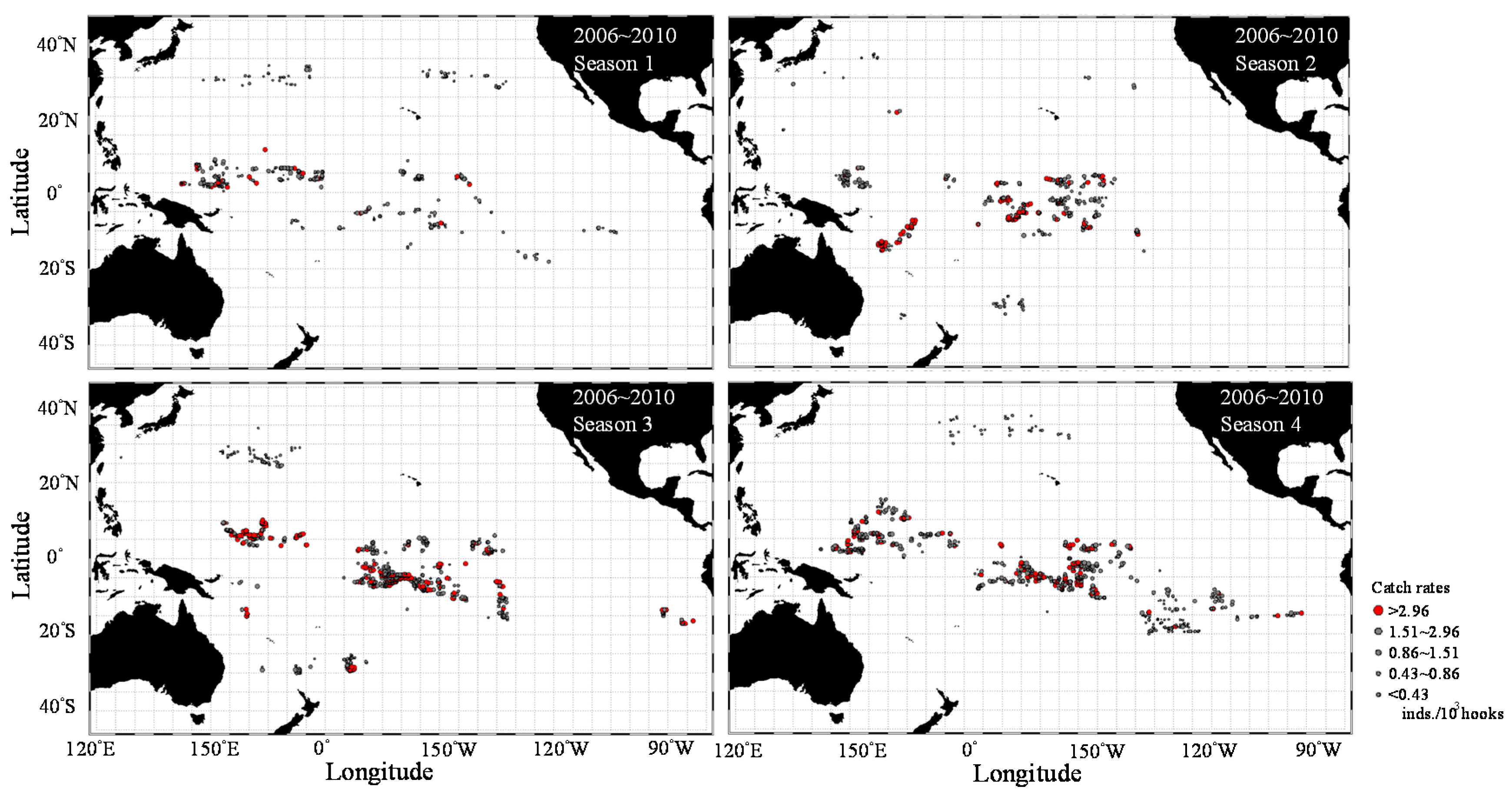
3.1. Catch Rate Patterns of Yellowfin Tuna in the TPO
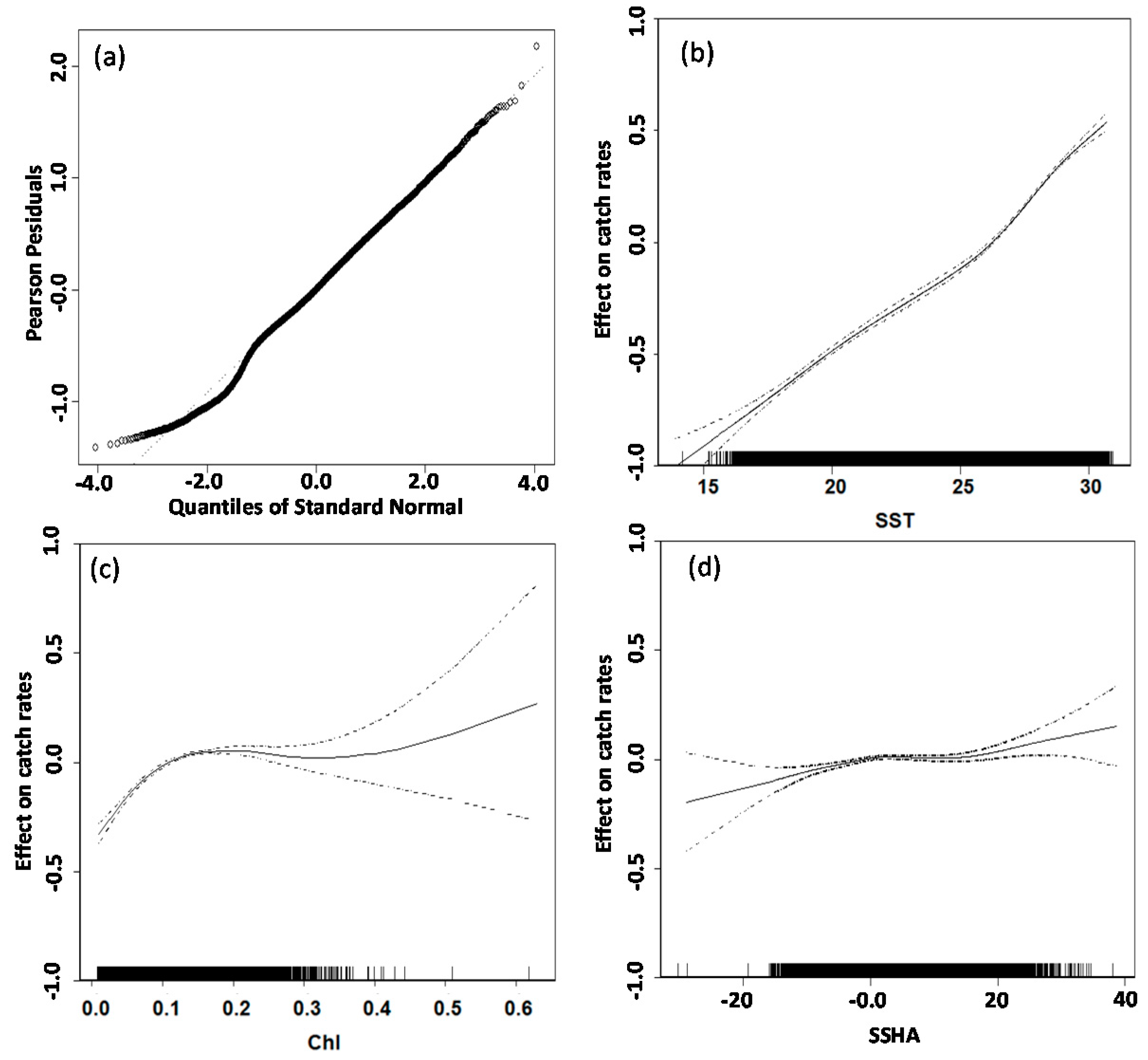
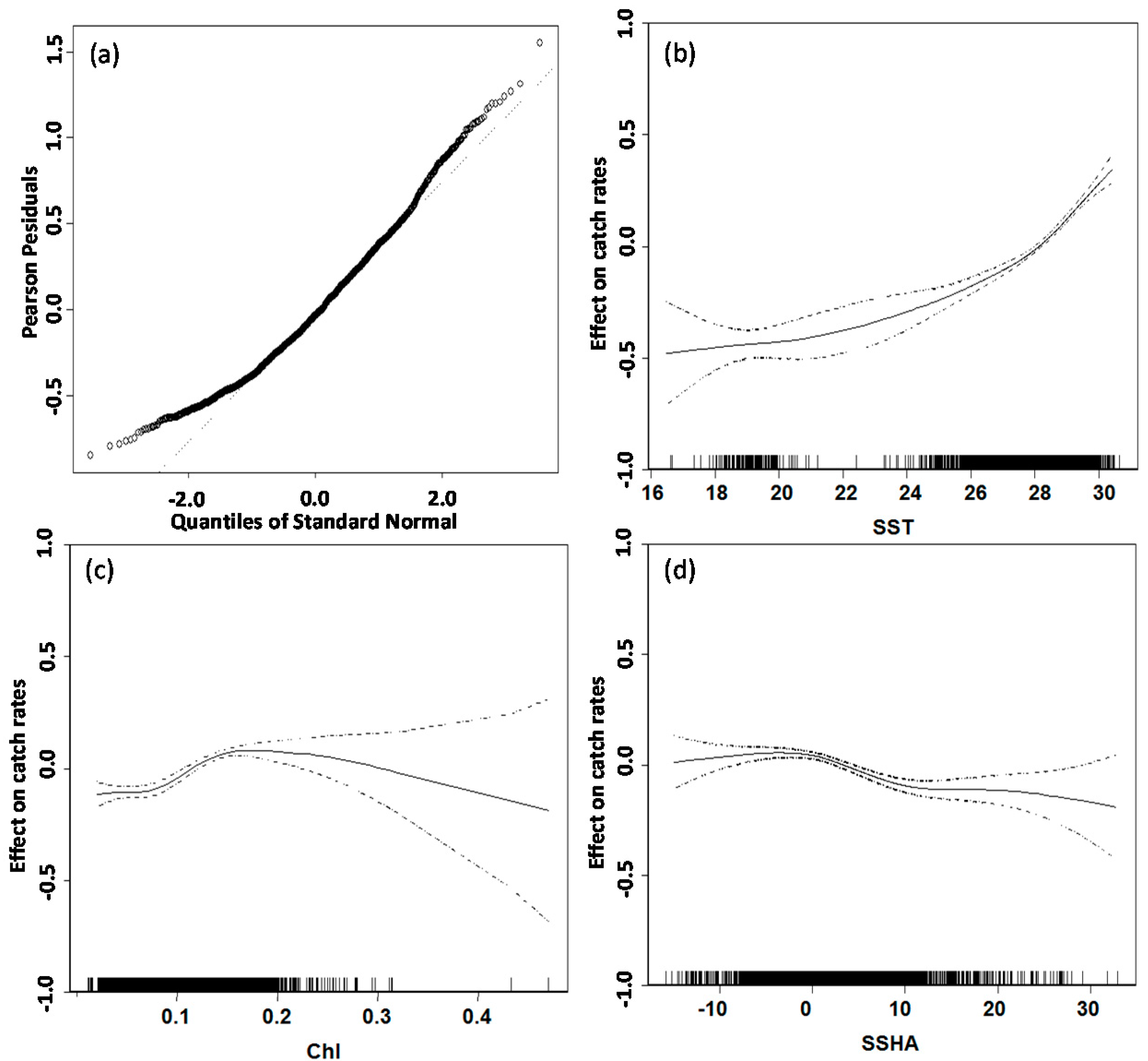
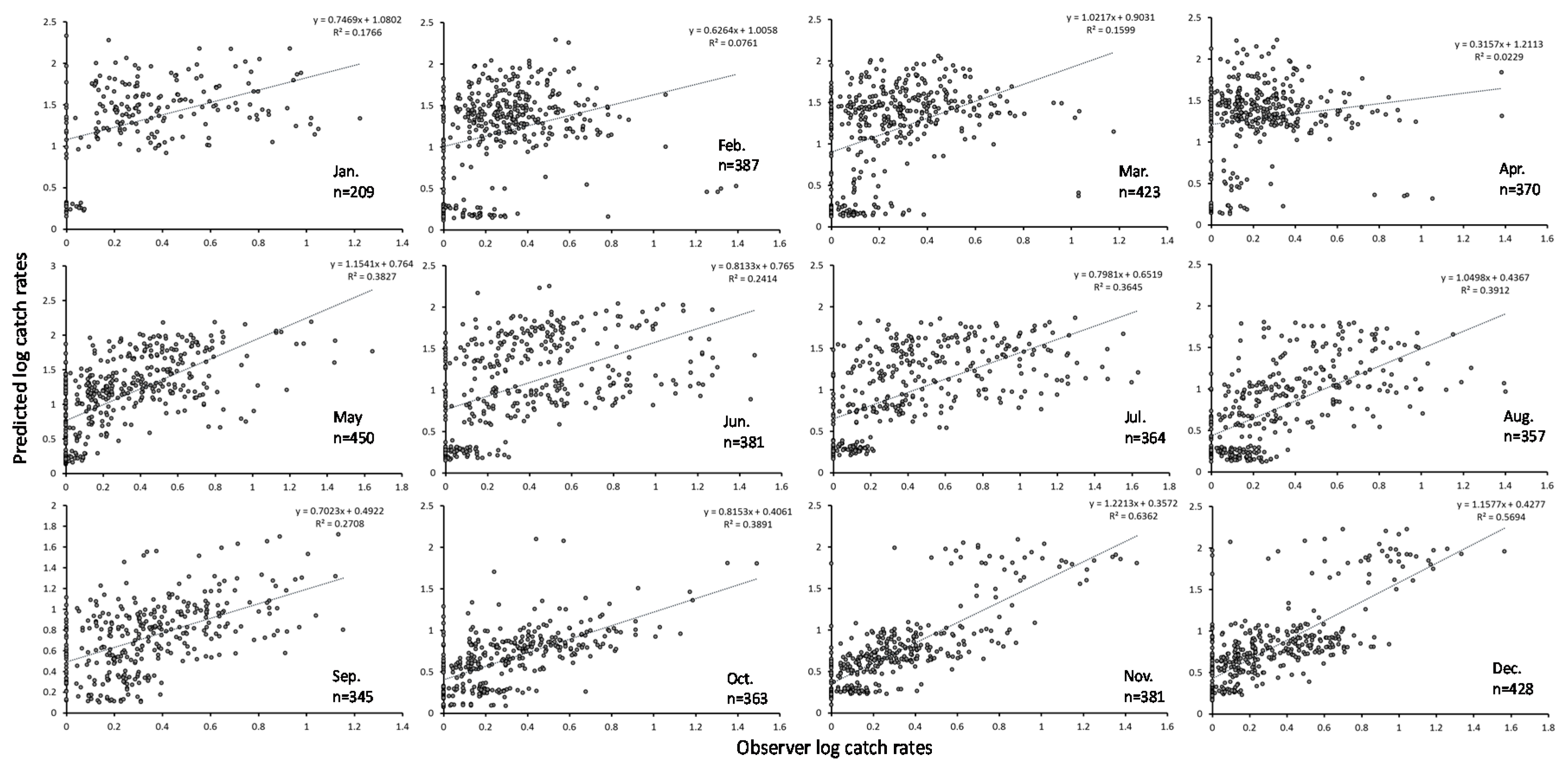
3.2. GAM Analysis and Selection of Error Model and Explanatory Variables
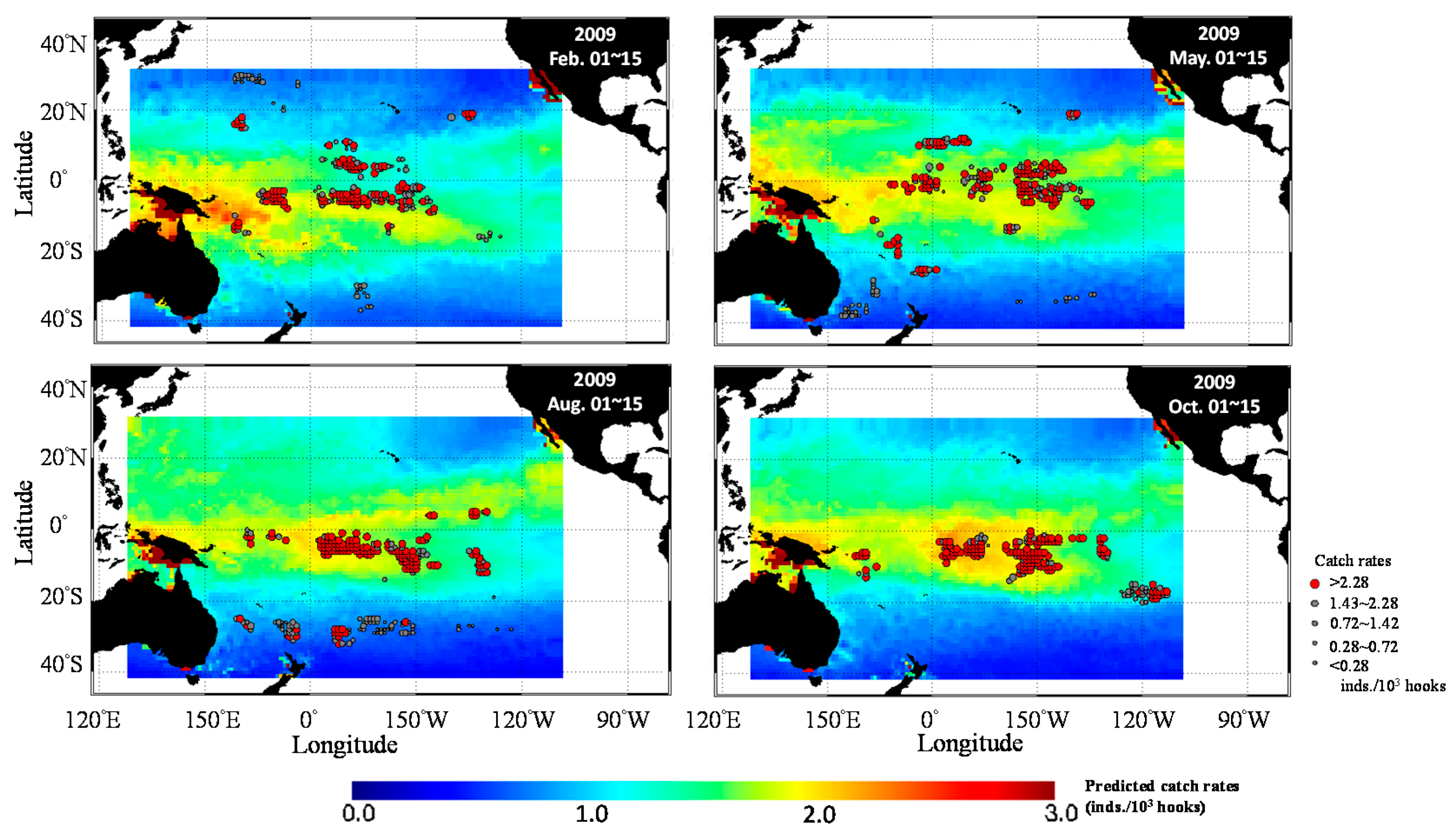
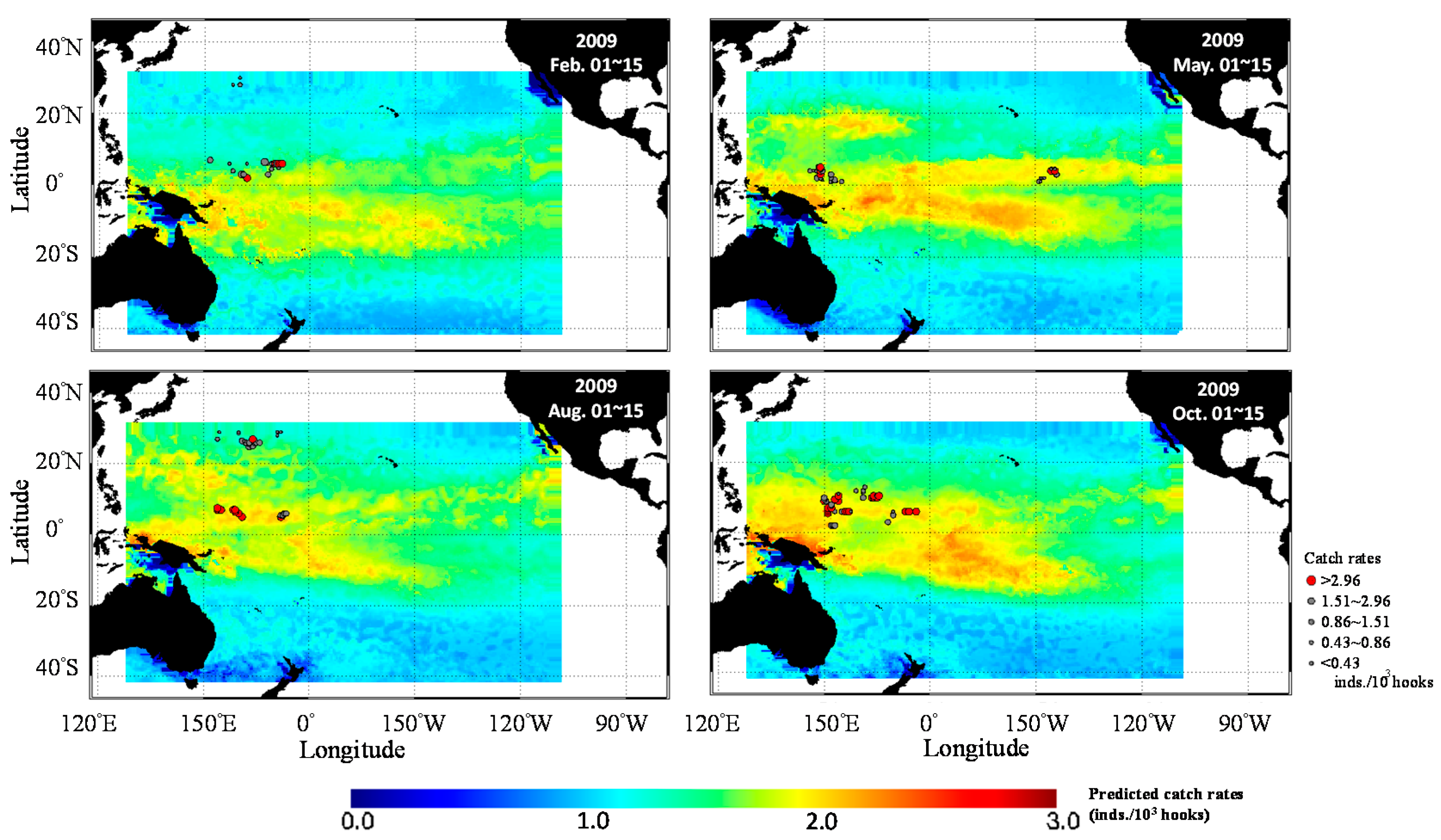
3.3. Predicted Spatial Distribution of Yellowfin Tuna in the TPO
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Schaefer, K.M.; Fuller, D.W.; Block, B.A. Movements, behavior, and habitat utilization of yellowfin tuna (Thunnus albacares) in the northeastern Pacific Ocean, ascertained through archival tag data. Mar. Biol. 2007, 152, 503–525. [Google Scholar] [CrossRef]
- Heithaus, M.R.; Frid, A.; Wirsing, A.J.; Worm, B. Predicting ecological consequences of marine top predator declines. Trends Ecol. Evol. 2008, 23, 202–210. [Google Scholar] [CrossRef] [PubMed]
- Kitchell, J.F.; Essington, T.E.; Boggs, C.H.; Schindler, D.E.; Walters, C.J. The role of sharks and longline fisheries in a pelagic ecosystem of the central Pacific. Ecosystems 2002, 5, 202–216. [Google Scholar] [CrossRef]
- Ward, P.; Myers, R.A. Shifts in open-ocean fish communities coinciding with the commencement of commercial fishing. Ecology 2005, 86, 835–847. [Google Scholar] [CrossRef]
- Polovina, A.J.J.; Woodworth-Jefcoats, P.A. Fishery-Induced Changes in the Subtropical Pacific Pelagic Ecosystem Size Structure: Observations and Theory. PLoS ONE 2013, 8, e62341. [Google Scholar] [CrossRef] [PubMed]
- Dickey, T.D. Emerging ocean observations for interdisciplinary data assimilation systems. J. Mar. Syst. 2003, 40, 5–48. [Google Scholar] [CrossRef]
- Alabia, I.D.; Saitoh, S.I.; Igarashi, H.; Ishikawa, Y.; Usui, N.; Kamachi, M.; Seito, M. Ensemble squid habitat model using three-dimensional ocean data. ICES J. Mar. Sci. J. Conseil 2016, 73, 1863–1874. [Google Scholar] [CrossRef]
- Damalas, D.; Megalofonou, P. Discovering where bluefin tuna, Thunnus thynnus (Linnaeus, 1785), might go: Using environmental and fishery data to map potential tuna habitat in the Eastern Mediterranean Sea. Sci. Mar. 2012, 76, 691–704. [Google Scholar]
- Lan, K.W.; Evans, K.; Lee, M.A. Effects of climate variability on the distribution and fishing conditions of yellowfin tuna (Thunnus albacares) in the western Indian Ocean. Clim. Chang. 2013, 119, 63–77. [Google Scholar] [CrossRef]
- Nieto, K.; Xu, Y.; Teo, S.L.H.; McClatchie, S.; Holmes, J. How important are coastal fronts to albacore tuna (Thunnus alalunga) habitat in the Northeast Pacific Ocean? Prog. Oceanogr. 2017, 150, 62–71. [Google Scholar] [CrossRef]
- Langley, A.; Briand, K.; Kirby, D.S.; Murtugudde, R. Influence of oceanographic variability on recruitment of yellowfin tuna (Thunnus albacares) in the western and central Pacific Ocean. Can. J. Fish. Aquat. Sci. 2009, 66, 1462–1477. [Google Scholar]
- Sund, P.N.; Blackburn, M.; Willians, F. Tunas and their environment in the Pacific Ocean: A review. Oceanogr. Mar. Biol. Ann. Rev. 1981, 19, 443–512. [Google Scholar]
- Dell, J.; Wilcox, C.; Hobday, A.J. Estimation of yellowfin tuna (Thunnus albacares) habitat in waters adjacent to Australia’s East Coast: Making the most of commercial catch data. Fish. Oceanogr. 2011, 20, 383–396. [Google Scholar] [CrossRef]
- Yen, K.W.; Lu, H.J.; Chang, Y.; Lee, M.A. Using remote-sensing data to detect habitat suitability for yellowfin tuna in the Western and Central Pacific Ocean. Int. J. Remote Sens. 2012, 33, 7507–7522. [Google Scholar] [CrossRef]
- Lan, K.W.; Nishida, T.; Lee, M.A.; Lu, H.J.; Huang, H.W.; Chang, S.K.; Lan, Y.C. Influence of the marine environment variability of the yellowfin tuna (Thunnus albacares) catch rate by the Taiwanese longline fishery in the Arabian Sea, with special reference to the high catch in 2004. J. Mar. Sci. Technol. 2012, 20, 514–524. [Google Scholar]
- Polovina, J.J.; Howell, E.A. Ecosystem indicators derived from satellite remotely sensed oeeanographie data for the North Pacific. ICES J. Mar. Sci. 2005, 62, 319–327. [Google Scholar] [CrossRef]
- Lan, K.W.; Lee, M.A.; Lu, H.J.; Shieh, W.J.; Lin, W.K.; Kao, S.C. Ocean variations associated with fishing conditions of yellowfin tuna (Thunnus albacares) in the equatorial Atlantic Ocean. ICES J. Mar. Sci. 2011, 68, 1063–1071. [Google Scholar] [CrossRef]
- Ishikawa, Y.; Awaji, T.; Toyoda, T.; In, T.; Nishina, K.; Nakayama, T.; Shima, S.; Masuda, S. High-resolution synthetic monitoring by a 4-dimensional variational data assimilation system in the northwestern North Pacific. J. Mar. Syst. 2009, 78, 237–248. [Google Scholar] [CrossRef]
- Nakada, S.; Hirose, N.; Senjyu, T.; Fukudome, K.I.; Tsuji, T.; Okei, N. Operational ocean prediction experiments for smart coastal fishing. Prog. Oceanogr. 2014, 121, 125–140. [Google Scholar] [CrossRef]
- Chang, S.K.; Hoyle, S.; Liu, H.I. Catch rate standardization for yellowfin tuna (Thunnus albacares) in Taiwan’s distant-water longline fishery in the Western and Central Pacific Ocean, with consideration of target change. Fish. Res. 2011, 107, 210–220. [Google Scholar] [CrossRef]
- Su, N.J.; Sun, C.L.; Punt, A.E.; Yeh, S.Z.; DiNardo, G. Environmental influences on seasonal movement patterns and regional fidelity of striped marlin Kajikia audax in the Pacific Ocean. Fish. Res. 2015, 166, 59–66. [Google Scholar] [CrossRef]
- Lehodey, P. Climate and fisheries: An insight from the Pacific Ocean. In Ecological Effects of Climate Variations in the North Atlantic; Stenseth, N.C., Ottersen, G., Hurrel, J., Belgrano, A., Eds.; Oxford University Press: Oxford, UK, 2004; pp. 137–146. [Google Scholar]
- Lehodey, P.; Hampton, J.; Brill, R.W.; Nicol, S.; Senina, I.; Calmettes, B.; Pørtner, H.O.; Bopp, L.; Ilyina, T.; Bell, J.D.; et al. Vulnerability of Tropical Pacific Fisheries and Aquaculture to Climate Change; Bell, J.D., Johnson, J.E., Hobday, A.J., Eds.; Secretariat of the Pacific Community: Noumea, New Caledonia, 2011; pp. 433–492. [Google Scholar]
- Maunder, M.N.; Punt, A.E. Standardizing catch and effort data: A review of recent approaches. Fish. Res. 2004, 70, 141–159. [Google Scholar] [CrossRef]
- Mugo, R.; Saitoh, S.I.; Nihira, A.; Kuroyama, T. Habitat characteristics of skipjack tuna (Katsuwonus pelamis) in the western North Pacific: A remote sensing perspective. Fish. Oceanogr. 2015, 19, 382–396. [Google Scholar] [CrossRef]
- Wood, S.M. Generalized Additive Models, an Introduction with R; Chapman and Hall: London, UK, 2006; p. 392. [Google Scholar]
- Pearman, P.B.; Guisan, A.; Broennimann, O.; Randin, C.F. Niche dynamics in space and time. Trends Ecol. Evol. 2008, 23, 149–158. [Google Scholar] [CrossRef] [PubMed]
- Vincent, D.G. The South Pacific Convergence Zone (SPCZ): A review. Mon. Weather Rev. 1994, 122, 1949–1970. [Google Scholar] [CrossRef]
- Rayner, N.A.; Brohan, P.; Parker, D.E.; Folland, C.K.; Hardwick, J.; Kennedy, J.; Vanicek, M.; Ansell, T.; Tett, S.F.; Kent, E.C. Improved analyses of changes and uncertainties in marine temperature measured in situ since the mid-nineteenth century: The HadSST2 dataset. J. Clim. 2005, 19, 446–469. [Google Scholar] [CrossRef]
- Prince, E.D.; Goodyear, C.P. Hypoxia-based habitat compression of tropical pelagic fishes. Fish. Oceanogr. 2006, 15, 451–464. [Google Scholar] [CrossRef]
- Yen, K.W.; Lu, H.J. Spatial–temporal variations in primary productivity and population dynamics of skipjack tuna Katsuwonus pelamis in the western and central Pacific Ocean. Fish. Sci. 2016, 82, 563–571. [Google Scholar] [CrossRef]
- Menard, F.; Marsac, F.; Bellier, E.; Cazelles, B. Climatic Oscillations and tuna catch rates in the Indian Ocean: A wavelet approach of time series analysis. Fish. Oceanogr. 2007, 16, 95–104. [Google Scholar] [CrossRef]
- Rebert, J.P.; Donguy, J.R.; Eldin, G.; Wyrtki, K. Relations between sea level, thermocline depth, heat content, and dynamic height in the tropical Pacific Ocean. J. Geophys. Res. Oceans 1985, 90, 11719–11725. [Google Scholar] [CrossRef]
- Le Traon, P.Y.; Dibarboure, G. Mesoscale mapping capabilities from multiple altimeter missions. J. Atmos. Ocean. Technol. 1999, 16, 1208–1223. [Google Scholar] [CrossRef]
- Ducet, N.; Le Traon, P.Y.; Reverdin, G. Global high-resolution mapping of ocean circulation from the combination of T/P and ERS-1/2. J. Geophys. Res. 2000, 105, 19477–19498. [Google Scholar] [CrossRef]
- Sournia, A. Pelagic biogeography and fronts. Prog. Oceanogr. 1994, 34, 109–120. [Google Scholar] [CrossRef]
- Bertrand, A.; Josse, E.; Bach, P.; Gros, P.; Dagorn, L. Hydrological and trophic characteristics of tuna habitat: Consequences on tuna distribution and longline catchability. Can. J. Fish. Aquat. Sci. 2002, 59, 1002–1013. [Google Scholar] [CrossRef]
- Lee, Y.C.; Liu, H.C. The tuna statistics procedures of Taiwan longline and gillnet Fisheries in the Indian Ocean. IPTP Collectiv. Vol. 1996, 9, 368–369. [Google Scholar]
- Austin, M. Species distribution models and ecological theory: A critical assessment and some possible new approaches. Ecol. Model. 2007, 200, 1–19. [Google Scholar] [CrossRef]
- Zhang, C.I.; Hollowed, A.B.; Lee, J.B.; Kim, D.H. An IFRAME approach for assessing impacts of climate change on fisheries. ICES J. Mar. Sci. 2011, 68, 1318–1328. [Google Scholar] [CrossRef]
- Cheung, W.W.L.; Dunne, J.; Sarmiento, J.L.; Pauly, D. Integrating ecophysiology and plankton dynamics into projected maximum fisheries catch potential under climate change in the Northeast Atlantic. ICES J. Mar. Sci. 2011, 68, 1008–1018. [Google Scholar] [CrossRef]
- Flierl, G.; Grünbaum, D.; Levin, S.A.; Olson, D. From individuals to aggregations: The interplay between behavior and physics. J. Theor. Biol. 1999, 196, 397–454. [Google Scholar] [CrossRef] [PubMed]
- Humston, R.; Olson, D.B.; Ault, J.S. Behavioral assumptions in models of fish movement and their influence on population dynamics. Trans. Am. Fish. Soc. 2004, 133, 1304–1328. [Google Scholar] [CrossRef]
| (a) | |||
|---|---|---|---|
| Deviance Explained | AIC | p Value | |
| (SST) | 32.30% | 54,340.69 | <0.01 |
| (CHL) | 6.20% | 60,466.75 | <0.01 |
| (SSHA) | 0.16% | 61,644.42 | <0.01 |
| (SST) + (CHL) | 33.50% | 54,023.75 | <0.01 |
| (SST) + (SSHA) | 32.40% | 54,332.00 | <0.01 |
| (CHL) + (SSHA) | 5.65% | 6058.38 | <0.01 |
| (SST) + (CHL) + (SSHA) | 33.60% | 54,003.60 | <0.01 |
| (b) | |||
| Deviance Explained | AIC | p Value | |
| (SST) | 11.00% | 5924.31 | <0.01 |
| (CHL) | 2.65% | 6150.07 | <0.01 |
| (SSHA) | 1.46% | 6175.01 | <0.01 |
| (SST) + (CHL) | 14.60% | 5843.11 | <0.01 |
| (SST) + (SSHA) | 13.10% | 5873.93 | <0.01 |
| (CHL) + (SSHA) | 3.75% | 6131.81 | <0.01 |
| (SST) + (CHL) + (SSHA) | 16.50% | 5796.74 | <0.01 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lan, K.-W.; Shimada, T.; Lee, M.-A.; Su, N.-J.; Chang, Y. Using Remote-Sensing Environmental and Fishery Data to Map Potential Yellowfin Tuna Habitats in the Tropical Pacific Ocean. Remote Sens. 2017, 9, 444. https://doi.org/10.3390/rs9050444
Lan K-W, Shimada T, Lee M-A, Su N-J, Chang Y. Using Remote-Sensing Environmental and Fishery Data to Map Potential Yellowfin Tuna Habitats in the Tropical Pacific Ocean. Remote Sensing. 2017; 9(5):444. https://doi.org/10.3390/rs9050444
Chicago/Turabian StyleLan, Kuo-Wei, Teruhisa Shimada, Ming-An Lee, Nan-Jay Su, and Yi Chang. 2017. "Using Remote-Sensing Environmental and Fishery Data to Map Potential Yellowfin Tuna Habitats in the Tropical Pacific Ocean" Remote Sensing 9, no. 5: 444. https://doi.org/10.3390/rs9050444
APA StyleLan, K.-W., Shimada, T., Lee, M.-A., Su, N.-J., & Chang, Y. (2017). Using Remote-Sensing Environmental and Fishery Data to Map Potential Yellowfin Tuna Habitats in the Tropical Pacific Ocean. Remote Sensing, 9(5), 444. https://doi.org/10.3390/rs9050444







