A Phenological Approach to Spectral Differentiation of Low-Arctic Tundra Vegetation Communities, North Slope, Alaska
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Site and Low-Arctic Vegetation Types
2.2. Ground-Based Data and Sampling Protocol
2.3. EnMAP and Sentinel-2 Surface Reflectance Simulation
2.4. Stable Wavelength Identification Using the InStability Index
3. Results
3.1. Spectral Characteristics by Phenophase
3.2. InStability Index and Wavelength Selection of Ground-Based Spectra
3.3. InStability Index and Wavelength Selection of Simulated Satellite Reflectance Spectra
4. Discussion
4.1. Phenophase and Wavelength Stability of Ground-Based Spectra
4.2. Phenophase and Wavelength Stability of Satellite Resampled Spectra
4.3. Influence of Spatial Scale
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Bliss, L.; Heal, O.W.; Moore, J. Tundra Ecosystems: A Comparative Analysis; CUP Archive: Cambridge, UK, 1981; p. 25. [Google Scholar]
- Walker, D.A.; Raynolds, M.K.; Daniëls, F.J.; Einarsson, E.; Elvebakk, A.; Gould, W.A.; Katenin, A.E.; Kholod, S.S.; Markon, C.J.; Melnikov, E.S.; et al. The circumpolar Arctic vegetation map. J. Veg. 2005, 16, 267–282. [Google Scholar] [CrossRef]
- Walker, M.; Walker, D.; Auerbach, N. Plant communities of a tussock tundra landscape in the Brooks Range Foothills, Alaska. J. Veg. Sci. 1994, 5, 843–866. [Google Scholar] [CrossRef]
- Hope, A.S.; Kimball, J.S.; Stow, D.A. The relationship between tussock tundra spectral reflectance properties and biomass and vegetation composition. Int. J. Remote Sens. 1993, 14, 1861–1874. [Google Scholar] [CrossRef]
- Riedel, S.M.; Epstein, H.E.; Walker, D.A. Biotic controls over spectral reflectance of arctic tundra vegetation. Int. J. Remote Sens. 2005, 26, 2391–2405. [Google Scholar] [CrossRef]
- Buchhorn, M.; Walker, D.; Heim, B.; Raynolds, M.; Epstein, H.; Schwieder, M. Ground-based hyperspectral characterization of Alaska tundra vegetation along environmental gradients. Remote Sens. 2013, 5, 3971–4005. [Google Scholar] [CrossRef]
- Bratsch, S.; Epstein, H.; Buchhorn, M.; Walker, D. Differentiating among four Arctic Tundra plant communities at Ivotuk, Alaska using field spectroscopy. Remote Sens. 2016, 8, 51. [Google Scholar] [CrossRef]
- Laidler, G.J.; Treitz, P.M.; Atkinson, D.M. Remote sensing of arctic vegetation: Relations between the NDVI, spatial resolution and vegetation cover on Boothia Peninsula, Nunavut. Arctic 2008, 6, 1–13. [Google Scholar] [CrossRef]
- Olthof, I.; Latifovic, R. Short-term response of arctic vegetation NDVI to temperature anomalies. Int. J. Remote Sens. 2007, 28, 4823–4840. [Google Scholar] [CrossRef]
- Stow, D.A.; Hope, A.; McGuire, D.; Verbyla, D.; Gamon, J.; Huemmrich, F.; Houston, S.; Racine, C.; Sturm, M.; Tape, K.; et al. Remote sensing of vegetation and land-cover change in Arctic tundra ecosystems. Remote Sens. Environ. 2004, 89, 281–308. [Google Scholar] [CrossRef]
- Laidler, G.; Treitz, P. Biophysical remote sensing of arctic environments. Progress Phys. Geogr. 2003, 27, 44–68. [Google Scholar] [CrossRef]
- Boelman, N.; Stieglitz, M.; Rueth, H.; Sommerkorn, M.; Griffin, K.; Shaver, G.; Gamon, J. Response of NDVI, biomass, and ecosystem gas exchange to long-term warming and fertilization in wet sedge tundra. Oecologia 2003, 135, 414–421. [Google Scholar] [CrossRef] [PubMed]
- Muller, S.V.; Racoviteanu, A.E.; Walker, D.A. Landsat MSS-derived land-cover map of northern Alaska: Extrapolation methods and a comparison with photo-interpreted and AVHRR-derived maps. Int. J. Remote Sens. 1999, 20, 2921–2946. [Google Scholar] [CrossRef]
- Tucker, C. Red and photographic infrared linear combinations for monitoring vegetation. Remote Sens. Environ. 1979, 8, 127–150. [Google Scholar] [CrossRef]
- Van Leeuwen, W.J.; Huete, A.R. Effects of standing litter on the biophysical interpretation of plant canopies with spectral indices. Remote Sens. Environ. 1996, 55, 123–138. [Google Scholar] [CrossRef]
- Curran, P. Remote sensing of foliar chemistry. Remote Sens. Environ. 1989, 30, 271–278. [Google Scholar] [CrossRef]
- Bartley, G.E.; Scolnik, P.A. Plant carotenoids: Pigments for photoprotection, visual attraction, and human health. Plant Cell 1995, 7, 1027–1038. [Google Scholar] [CrossRef] [PubMed]
- Young, A.; Britton, G. Carotenoids and stress. In Stress Responses in Plants: Adaptation and Acclimation Mechanisms; Alscher, R.G., Cummings, J.R., Eds.; Wiley: New York, NY, USA, 1990; pp. 87–112. [Google Scholar]
- Steyn, W.J.; Wand, S.J.E.; Holcroft, D.M.; Jacobs, G. Anthocyanins in vegetative tissues: A proposed unified function in photoprotection. New Phytol. 2002, 155, 349–361. [Google Scholar] [CrossRef]
- Close, D.C.; Beadle, C.L. The ecophysiology of foliar anthocyanin. Bot. Rev. 2003, 69, 149–161. [Google Scholar] [CrossRef]
- Chalker-Scott, L. Environmental significance of anthocyanins in plant stress responses. Photochem. Photobiol. 1999, 70, 1–9. [Google Scholar] [CrossRef]
- Gould, K.S.; McKelvie, J.; Markham, K.R. Do anthocyanins function as antioxidants in leaves? Imaging of H2O2 in red and green leaves after mechanical injury. Plant Cell Environ. 2002, 25, 1261–1269. [Google Scholar] [CrossRef]
- Guanter, L.; Kaufmann, H.; Segl, K.; Foerster, S.; Rogass, C.; Chabrillat, S.; Kuester, T.; Hollstein, A.; Rossner, G.; Chlebek, C.; et al. The EnMAP Spaceborne Imaging Spectroscopy Mission for earth observation. Remote Sens. 2015, 7, 8830–8857. [Google Scholar] [CrossRef]
- Drusch, M.; Del Bello, U.; Carlier, S.; Colin, O.; Fernandez, V.; Gascon, F.; Hoersch, B.; Isola, C.; Laberinti, P.; Martimort, P.; et al. Sentinel-2: ESA’s Optical High-Resolution Mission for GMES Operational Services. Remote Sens. Environ. 2012, 120, 25–36. [Google Scholar] [CrossRef]
- Chance, C.; Coops, N.; Crosby, K.; Aven, N. Spectral wavelength selection and detection of two invasive plant species in an urban area. Can. J. Remote Sens. 2016, 42, 27–40. [Google Scholar] [CrossRef]
- Somers, B.; Asner, G. Invasive species mapping in Hawaiian rainforests using multi-temporal hyperion spaceborne imaging spectroscopy. IEEE J. Sel. Top. Appl. Earth Observ. Remote Sens. 2013, 6, 351–359. [Google Scholar] [CrossRef]
- Somers, B.; Delalieux, S.; Verstraeten, W.W.; Van Aardt, J.A.N.; Albrigo, G.L.; Coppin, P. An automated waveband selection technique for optimized hyperspectral mixture analysis. Int. J. Remote Sens. 2010, 31, 5549–5568. [Google Scholar] [CrossRef]
- Somers, B.; Asner, G.P.; Tits, L.; Coppin, P. Endmember variability in spectral mixture analysis: A review. Remote Sens. Environ. 2011, 115, 1603–1616. [Google Scholar] [CrossRef]
- Peterson, S.; Roberts, D.; Beland, M.; Kokaly, R.; Ustin, S. Oil detection in the coastal marshes of Louisiana using MESMA applied to band subsets of AVIRIS data. Remote Sens. Environ. 2015, 159, 222–231. [Google Scholar] [CrossRef]
- Walker, D.A.; Binnian, E.; Evans, B.M.; Lederer, N.D. Terrain, vegetation and landscape evolution of the R4D research site, Brooks Range Foothills, Alaska. Ecography 1989, 12, 238–261. [Google Scholar] [CrossRef]
- Bliss, L.C.; Matveyeva, N.V. Circumpolar arctic vegetation. In Arctic Ecosystems in a Changing Climate: An Ecophysiological Perspective; Academic Press: San Diego, CA, USA, 1992; pp. 59–89. [Google Scholar]
- Tenhunen, J.D.; Lange, O.L.; Hahn, S.; Siegwolf, R.; Oberbauer, S.F. The ecosystem role of poikilohydric tundra plants. In Arctic Ecosystems in a Changing Climate: An Ecophysiological Perspective; Academic Press: San Diego, CA, USA, 1992. [Google Scholar]
- Lehnert, L.W.; Meyer, H.; Bendix, J. Hsdar: Manage, Analyse and Simulate Hyperspectral Data in R; R Package: Marburg, Germany, 2016. [Google Scholar]
- Clark, R.N.; Roush, T.L. Reflectance spectroscopy: Quantitative analysis techniques for remote sensing applications. J. Geophys. Res. Solid Earth 1984, 89, 6329–6340. [Google Scholar] [CrossRef]
- Clark, R.N.; King, T. Automatic Continuum Analysis of Reflectance Spectra. In Proceedings of the 3rd Airborne Imaging Spectrometer Data Analysis Workshop, Denver, CO, USA, 15 August 1987; NASA: Pasadena, CA, USA. [Google Scholar]
- Somers, B.; Cools, K.; Delalieux, S.; Stuckens, J.; Van der Zande, D.; Verstraeten, W.W.; Coppin, P. Nonlinear hyperspectral mixture analysis for tree cover estimates in orchards. Remote Sens. Environ. 2009, 113, 1183–1193. [Google Scholar] [CrossRef]
- Rock, B.N.; Hoshizaki, T.; Miller, J.R. Comparison of in situ and airborne spectral measurements of the blue shift associated with forest decline. Remote Sens. Environ. 1988, 24, 109–127. [Google Scholar] [CrossRef]
- Walker, D.A.; Auerbach, N.A.; Shippert, M.M. NDVI, biomass, and landscape evolution of glaciated terrain in northern Alaska. Polar Rec. 1995, 31, 169–178. [Google Scholar] [CrossRef]
- Chuvieco, E.; Martin, M.P.; Palacios, A. Assessment of different spectral indices in the red-near-infrared spectral domain for burned land discrimination. Int. J. Remote Sens. 2002, 23, 5103–5110. [Google Scholar] [CrossRef]
- Asner, G. P. Biophysical and biochemical sources of variability in canopy reflectance. Remote Sens. Environ. 1998, 64, 234–253. [Google Scholar] [CrossRef]
- Sims, D.; Gamon, J. Relationships between leaf pigment content and spectral reflectance across a wide range of species, leaf structures and developmental stages. Remote Sens. Environ. 2002, 81, 337–354. [Google Scholar] [CrossRef]
- Filella, I.; Penuelas, J. The red edge position and shape as indicators of plant chlorophyll content, biomass and hydric status. Int. J. Remote Sens. 1994, 15, 1459–1470. [Google Scholar] [CrossRef]
- Ustin, S.; Curtiss, B. Spectral characteristics of ozone-treated conifers. Environ. Exp. Bot. 1990, 30, 293–308. [Google Scholar] [CrossRef]
- Gitelson, A.A.; Merzlyak, M.N. Remote estimation of chlorophyll content in higher plant leaves. Int. J. Remote Sens. 1997, 18, 2691–2697. [Google Scholar] [CrossRef]
- Gitelson, A.A.; Zur, Y.; Chivkunova, O.B.; Merzlyak, M.N. Assessing Carotenoid Content in Plant Leaves with Reflectance Spectroscopy. Photochem. Photobiol. 2002, 75, 272–281. [Google Scholar] [CrossRef]
- Stylinski, C.; Gamon, J.; Oechel, W. Seasonal patterns of reflectance indices, carotenoid pigments and photosynthesis of evergreen chaparral species. Oecologia 2002, 131, 366–374. [Google Scholar] [CrossRef] [PubMed]

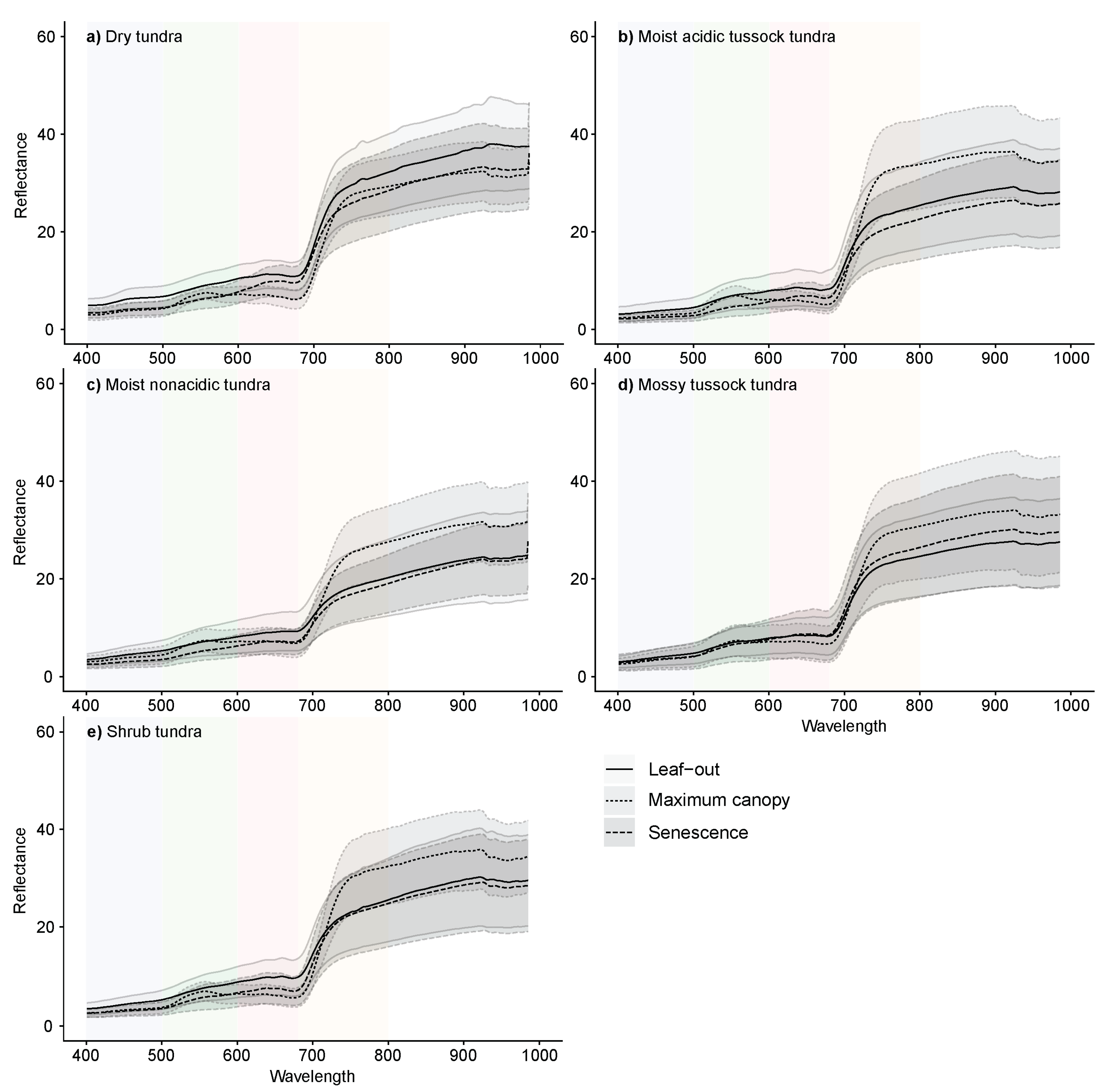
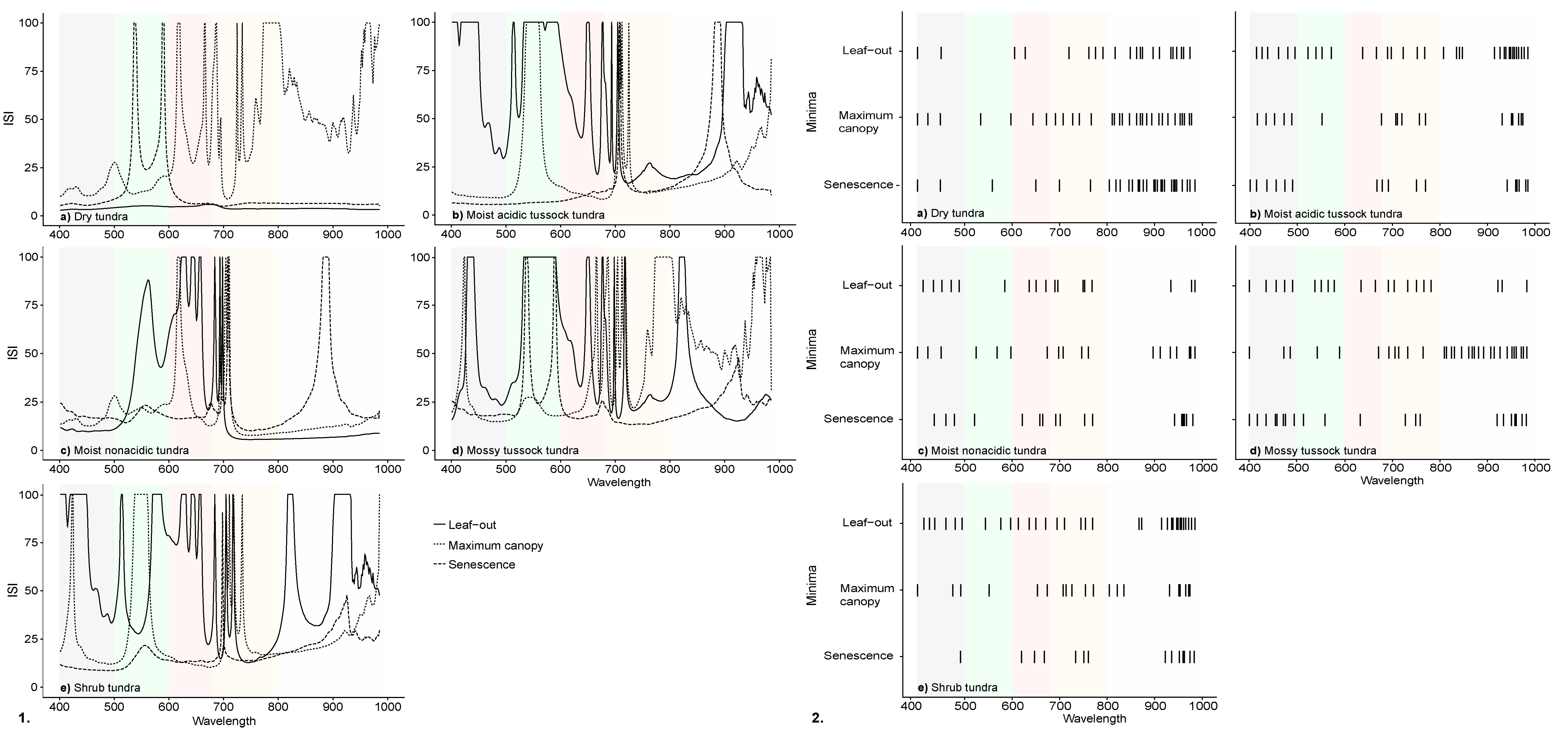
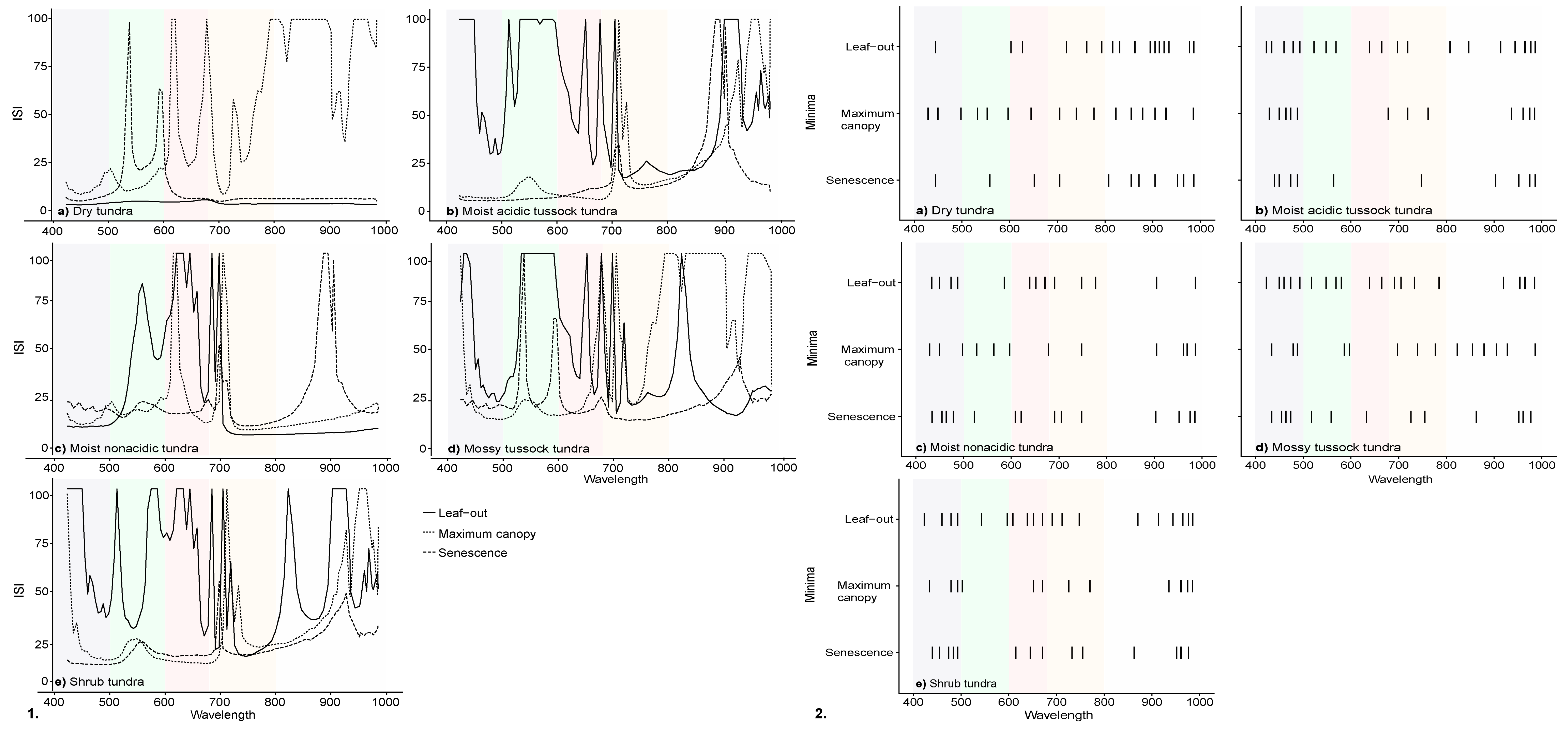
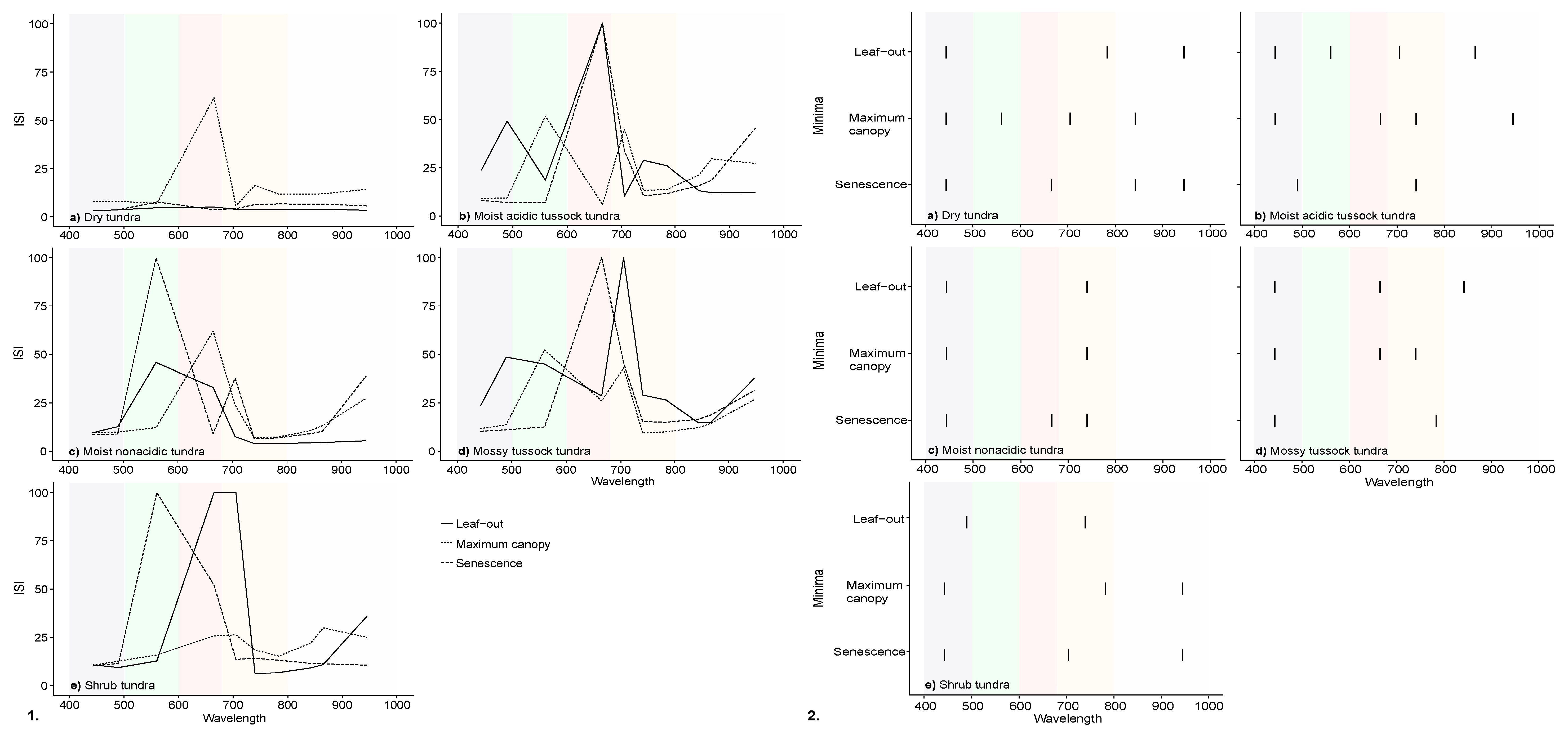
| Community | Description | Sub-Community | Description | % Cover |
|---|---|---|---|---|
| Dry tundra | Upland sparsely vegetated areas with prostrate dwarf shrubs (Salix glauca) and abundant lichens | Dry tundra | Dryas octopetala-Selaginella sibirica, Dryas octopetala-Salix glauca: Prostrate dwarf-shrub, forb, fruticose-lichen tundra. Xeric, acidic, windblown or shallow winter snow cover, stony or with considerable bare soil. Ridge crests, dry river terraces. Dominated by Dryas octopetala, occasionally with Salix glauca. | 4.2 |
| Moist acidic tussock tundra | Occurs on soils with pH < 5.0–5.5 and is dominated by dwarf erect shrubs such as Betula nana and Salix pulchra, graminoids species (Eriophorum vaginatum) and acidophilous mosses | Shrubby tussock tundra dominated by Betula nana | Betula nana-Eriophorum vaginatum. Dwarf-shrub, sedge, moss tundra (shrubby tussock tundra dominated by dwarf birch, Betula nana). Mesic to subhygric, acidic, moderate snow. Lower slopes and water-track margins. Mostly on Itkillik I glacial surfaces. | 3.1 |
| Shrubby tussock tundra dominated by Salix pulchra | Salix pulchra-Carex bigelowii. Dwarf-shrub, sedge, moss tundra (shrubby tussock tundra dominated by diamond-leaf willow, Salix pulchra). Subhygric, moderate snow, lower slopes with solifluction. | 3.0 | ||
| Moist non-acidic tundra | Dominated by mosses, graminoids (Carex bigilowii), and prostrate dwarf shrubs (Dryas integriolia) | Moist non-acidic tundra | Carex bigelowii-Dryas integrifolia, typical subtype; Tomentypnum nitens-Carex bigelowii, Salix glauca subtype: Nontussock sedge, dwarf-shrub, moss tundra (moist nonacidic tundra). Mesic to subhygric, nonacidic (pH > 5.5), shallow to moderate snow. Solifluction areas and somewhat unstable slopes. Some south-facing slopes have scattered glaucous willow (Salix glauca). | 5.8 |
| Mossy tussock tundra | A moist acidic tussock tundra-type community dominated by sedges (E. vaginatum) and abundant Sphagnum spp. | Moist acidic tussock tundra | Eriophorum vaginatum-Sphagnum; Carex bigelowii-Sphagnum: Tussock sedge, dwarf-shrub, moss tundra (tussock tundra, moist acidic tundra). Mesic to subhygric, acidic, shallow to moderate snow, stable. This unit is the zonal vegetation on fine-grained substrates with ice-rich permafrost. Some areas on steeper slopes with solifluction are dominated by Bigelow sedge (Carex bigelowii). | 54.2 |
| Shrub tundra | Dominated by dwarf shrubs Betula nana and Salix pulchra interspersed with graminoids, forbs, lichens and mosses | Dwarf shrub tundra dominated by Betula nana | Betula nana-Rubus chamaemorus. Dwarf-shrub, moss tundra dominated by dwarf birch (Betula nana). Subhygric to hygric, acidic, moderate to moderately deep snow. Upland water tracks, margins of water tracks and lower slope areas. Somewhat drier areas have abundant lichens. | 1.7 |
| Dwarf shrub tundra dominated by Salix pulchra | Salix pulchra-Sphagnum warnstorfii. Dwarf-shrub or low-shrub tundra dominated by willows (Salix pulchra). Subhygric, acidic, moderate to moderately deep snow. Margins of upland water tracks, palsas and high-centered polygons. | 1.1 |
| Community | Spectral Measurements | Distinct Plots |
|---|---|---|
| Dry tundra | 63 | 7 |
| Moist acidic tussock tundra | 126 | 17 |
| Moist non-acidic tundra | 144 | 14 |
| Mossy tussock tundra | 153 | 16 |
| Shrub tundra | 126 | 14 |
| Red Edge Inflection Point | Max Reflectance | Max Continuum Removed Band Depth | |||||
|---|---|---|---|---|---|---|---|
| Vegetation Community | Date | Wavelength (nm) | 550 nm (%) | 750 nm (%) | 920 nm (%) | Blue (400–550 nm) | Red (550–750 nm) |
| Dry tundra | Leaf-out | 706 | 9.28 | 32.35 | 40.54 | 0.13 | 0.38 |
| Maximum canopy | 710 | 9.01 | 31.54 | 37.10 | 0.26 | 0.64 | |
| Senescence | 704 | 7.33 | 36.28 | 44.19 | 0.21 | 0.37 | |
| Moist acidic tussock tundra | Leaf-out | 706 | 10.08 | 32.19 | 37.70 | 0.20 | 0.41 |
| Maximum canopy | 714 | 10.35 | 39.45 | 46.08 | 0.45 | 0.74 | |
| Senescence | 705 | 6.82 | 28.03 | 37.37 | 0.28 | 0.42 | |
| Moist non-acidic tundra | Leaf-out | 706 | 7.59 | 21.38 | 26.79 | 0.14 | 0.23 |
| Maximum canopy | 712 | 9.33 | 28.97 | 35.08 | 0.25 | 0.56 | |
| Senescence | 704 | 6.52 | 22.49 | 33.42 | 0.24 | 0.31 | |
| Mossy tussock tundra | Leaf-out | 708 | 8.78 | 27.68 | 32.71 | 0.16 | 0.38 |
| Maximum canopy | 713 | 8.94 | 32.20 | 39.76 | 0.29 | 0.62 | |
| Senescence | 707 | 8.86 | 29.31 | 34.59 | 0.24 | 0.40 | |
| Shrub tundra | Leaf-out | 704 | 8.75 | 25.16 | 31.87 | 0.22 | 0.33 |
| Maximum canopy | 714 | 8.10 | 30.96 | 37.61 | 0.38 | 0.70 | |
| Senescence | 705 | 6.09 | 26.24 | 33.65 | 0.26 | 0.46 | |
| Vegetation Community | Leaf-Out | Maximum Canopy | Senescence |
|---|---|---|---|
| ISI | ISI | ISI | |
| Dry tundra | 1532 | 148,112 | 8477 |
| Moist acidic tussock tundra | 69,256 | 15,867 | 7730 |
| Moist non-acidic tundra | 26,104 | 7393 | 9835 |
| Mossy tussock tundra | 38,139 | 150,136 | 13,047 |
| Shrub tundra | 71,140 | 17,715 | 6623 |
| Vegetation | Leaf-Out | Maximum Canopy | Senescence | |||
|---|---|---|---|---|---|---|
| ISI | EnMAP | Sentinel-2 | EnMAP | Sentinel-2 | EnMAP | Sentinel-2 |
| Dry tundra | 401 | 38 | 7795 | 155 | 1125 | 53 |
| Moist acidic tussock tundra | 16,789 | 684 | 4570 | 225 | 2050 | 297 |
| Moist non-acidic tundra | 2834 | 129 | 1883 | 182 | 2687 | 339 |
| Mossy tussock tundra | 12,958 | 485 | 7933 | 218 | 2346 | 311 |
| Shrub tundra | 9694 | 807 | 4928 | 200 | 1724 | 351 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Beamish, A.L.; Coops, N.; Chabrillat, S.; Heim, B. A Phenological Approach to Spectral Differentiation of Low-Arctic Tundra Vegetation Communities, North Slope, Alaska. Remote Sens. 2017, 9, 1200. https://doi.org/10.3390/rs9111200
Beamish AL, Coops N, Chabrillat S, Heim B. A Phenological Approach to Spectral Differentiation of Low-Arctic Tundra Vegetation Communities, North Slope, Alaska. Remote Sensing. 2017; 9(11):1200. https://doi.org/10.3390/rs9111200
Chicago/Turabian StyleBeamish, Alison Leslie, Nicholas Coops, Sabine Chabrillat, and Birgit Heim. 2017. "A Phenological Approach to Spectral Differentiation of Low-Arctic Tundra Vegetation Communities, North Slope, Alaska" Remote Sensing 9, no. 11: 1200. https://doi.org/10.3390/rs9111200
APA StyleBeamish, A. L., Coops, N., Chabrillat, S., & Heim, B. (2017). A Phenological Approach to Spectral Differentiation of Low-Arctic Tundra Vegetation Communities, North Slope, Alaska. Remote Sensing, 9(11), 1200. https://doi.org/10.3390/rs9111200







