Using Satellite Data for the Characterization of Local Animal Reservoir Populations of Hantaan Virus on the Weihe Plain, China
Abstract
1. Introduction
2. Materials and Methods
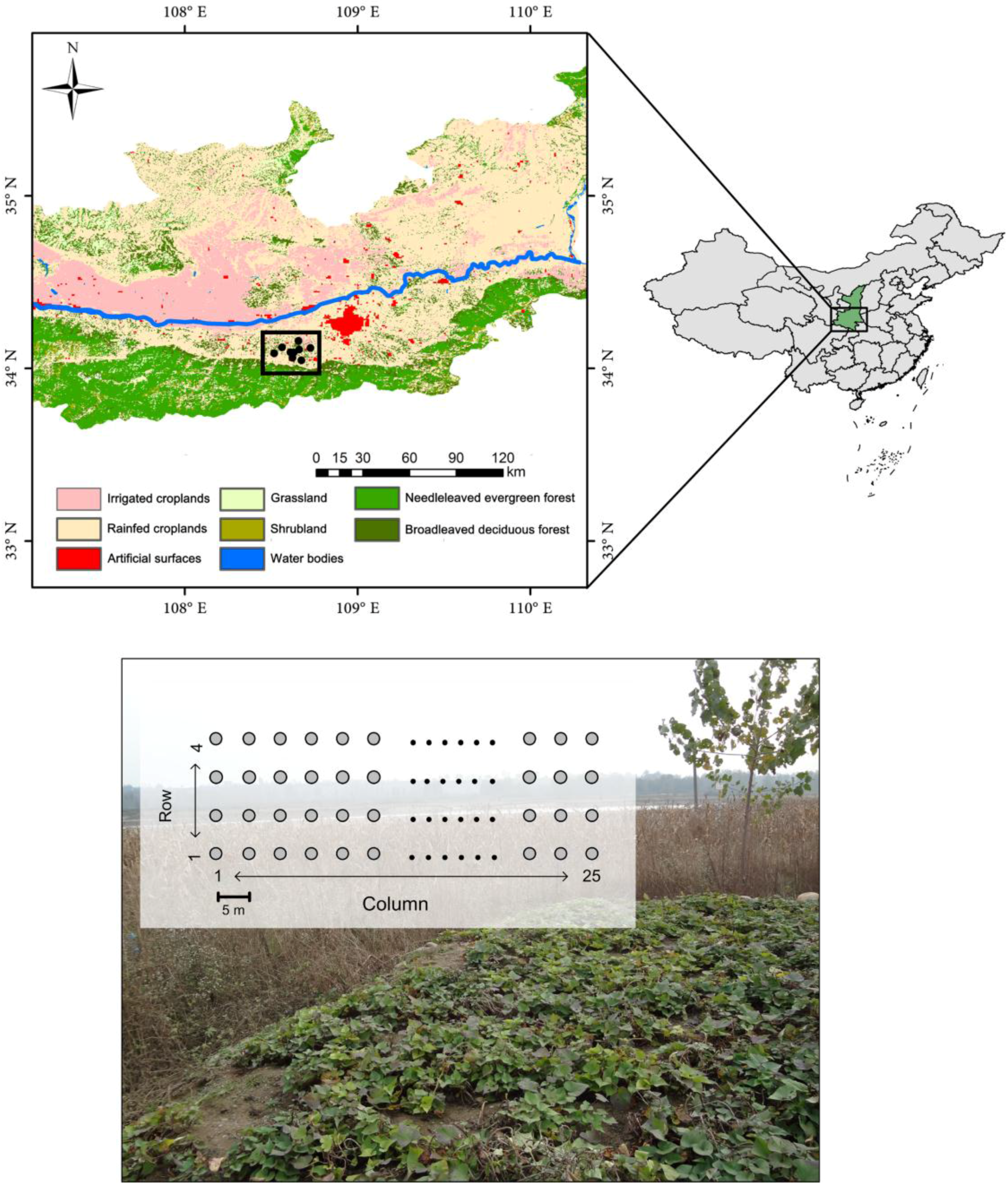
2.1. Rodent Sampling
2.2. Satellite Data
2.3. Statistical Analysis
2.4. Structure Equation Model
2.5. Ethical Review
3. Results
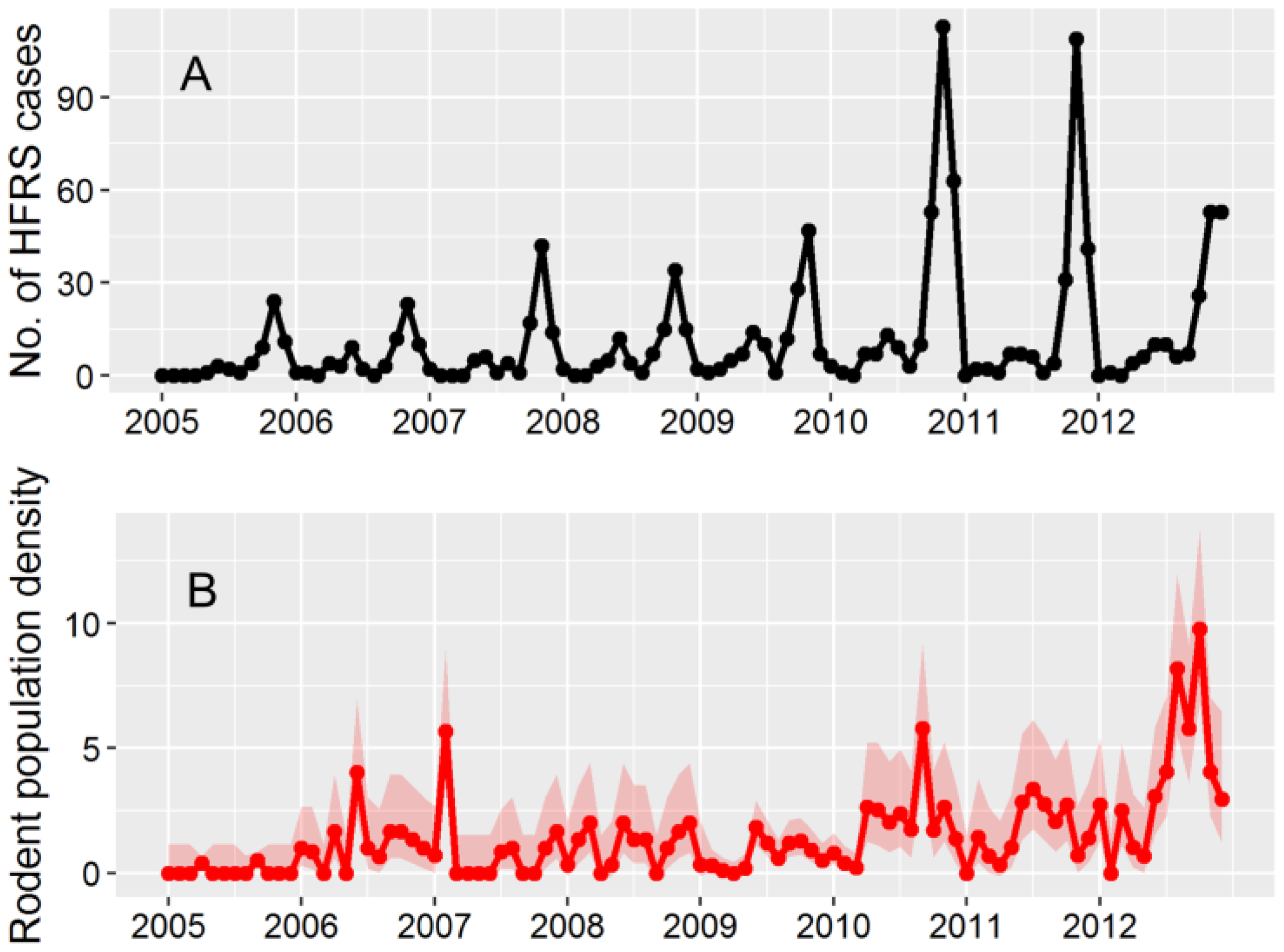
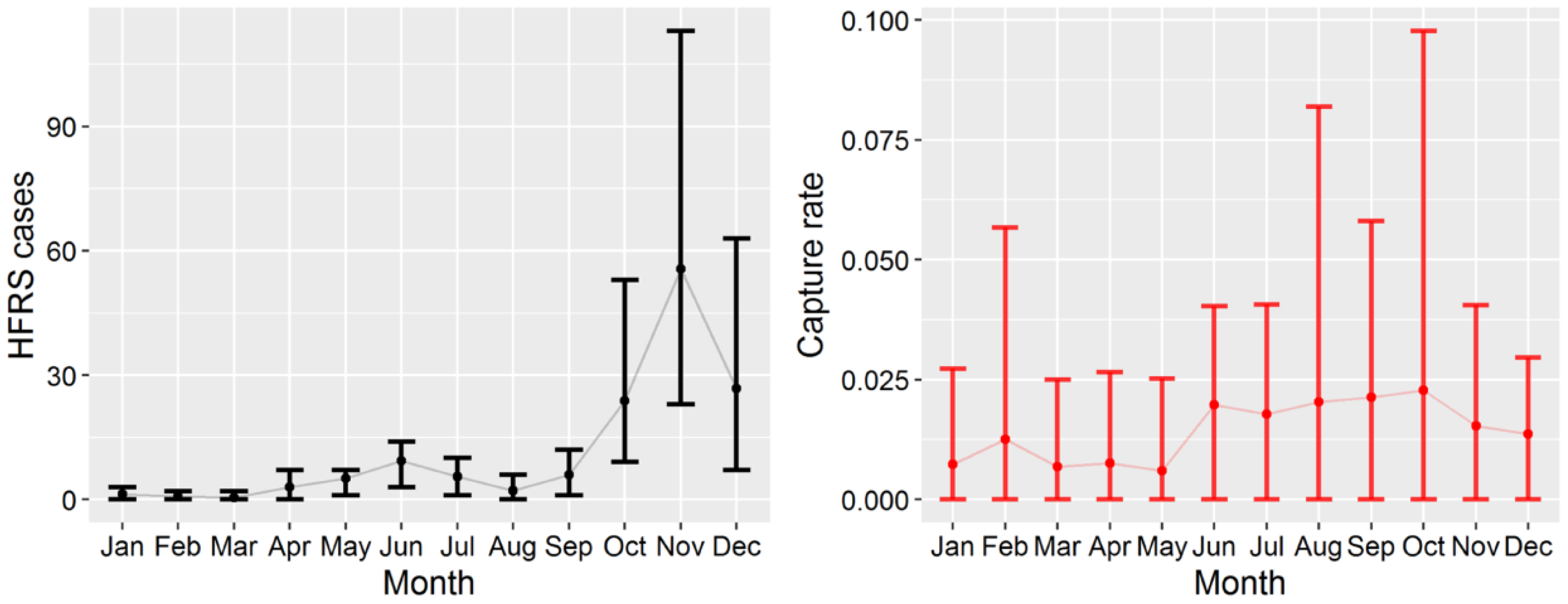
3.1. HFRS Epidemics in Hu County, 2005–2012
3.2. Apodemus Agrarius Population Dynamics and Temporal Pattern of HFRS
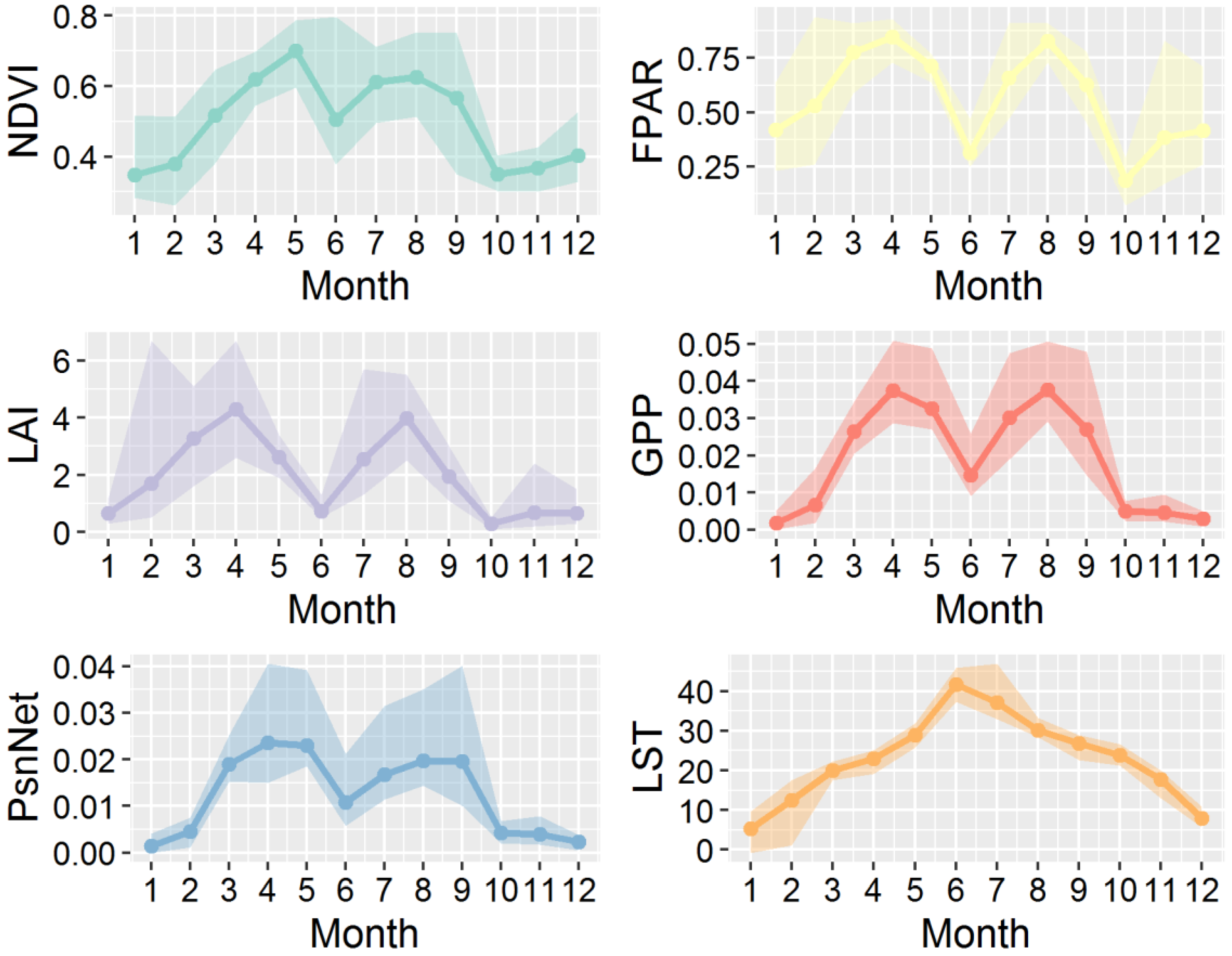
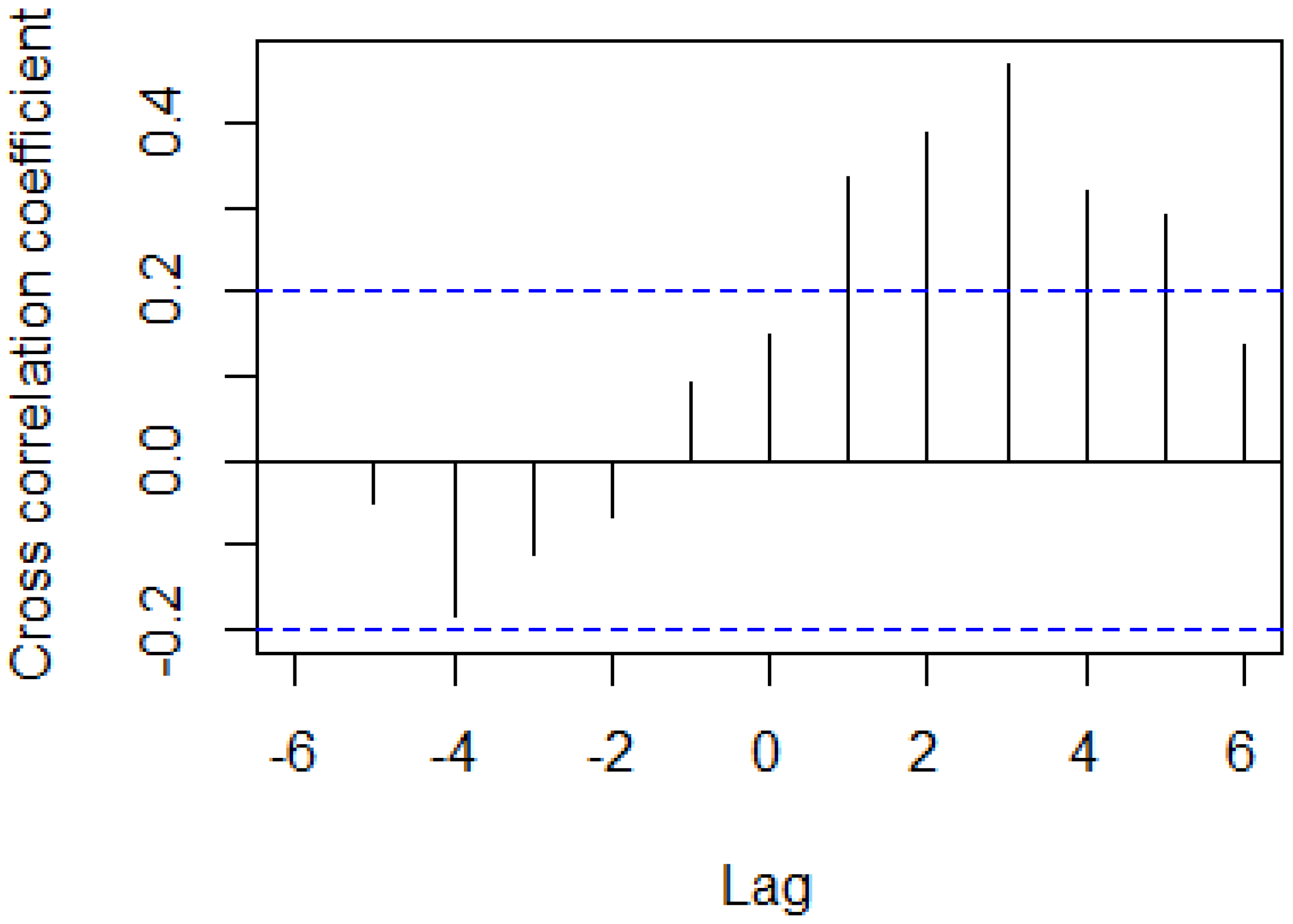
3.3. Correlations Between Remote Sensing-Based Indicators and Apodemus Agrarius Population Density
3.4. Correlations Between Remote Sensing-Based Indicators and Other Rodent Species
3.5. Structure Equation Model for Incidence of HFRS with Rodent Population Density and PsnNet
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of interest
References
- Tian, H.; Yu, P.; Bjørnstad, O.N.; Cazelles, B.; Yang, J.; Tan, H.; Huang, S.; Cui, Y.; Dong, L.; Ma, C. Anthropogenically driven environmental changes shift the ecological dynamics of hemorrhagic fever with renal syndrome. PLoS Pathog. 2017, 13, e1006198. [Google Scholar] [CrossRef] [PubMed]
- Yu, P.; Tian, H.; Ma, C.; Ma, C.; Wei, J.; Lu, X.; Wang, Z.; Zhou, S.; Li, S.; Dong, J. Hantavirus infection in rodents and haemorrhagic fever with renal syndrome in Shaanxi Province, China, 1984–2012. Epidemiol. Infect. 2015, 143, 405–411. [Google Scholar] [CrossRef] [PubMed]
- Jonsson, C.B.; Figueiredo, L.T.M.; Vapalahti, O. A global perspective on Hantavirus ecology, epidemiology, and disease. Clin. Microbiol. Rev. 2010, 23, 412–441. [Google Scholar] [CrossRef] [PubMed]
- Khaiboullina, S.F.; Morzunov, S.P.; St Jeor, S.C. Hantaviruses: Molecular biology, evolution and pathogenesis. Curr. Mol. Med. 2005, 5, 773–790. [Google Scholar] [CrossRef] [PubMed]
- Childs, J.E.; Glass, G.E.; Korch, G.W.; LeDuc, J.W. Prospective seroepidemiology of Hantaviruses and population dynamics of small mammal communities of Baltimore, Maryland. Am. J. Trop. Med. Hyg. 1987, 37, 648–662. [Google Scholar] [CrossRef] [PubMed]
- Lee, P.; Amyx, H.L.; Gibbs, C.; Gajdusek, D.; Lee, H. Propagation of Korean hemorrhagic fever virus in laboratory rats. Infect. Immun. 1981, 31, 334–338. [Google Scholar] [PubMed]
- Yang, Z. The research progress of HFRS transmission routes. Pract. J. Med. Pharm. 2005, 22, 69–71. [Google Scholar]
- Mills, J.N.; Yates, T.L.; Ksiazek, T.G.; Peters, C.; Childs, J.E. Long-term studies of Hantavirus reservoir populations in the Southwestern United States: Rationale, potential, and methods. Emerg. Infect. Dis. 1999, 5, 95–101. [Google Scholar] [CrossRef] [PubMed]
- Luis, A.D.; Douglass, R.J.; Mills, J.N.; Bjørnstad, O.N. Environmental fluctuations lead to predictability in sin Nombre Hantavirus outbreaks. Ecology 2015, 96, 1691–1701. [Google Scholar] [CrossRef]
- Zhang, W.Y.; Fang, L.Q.; Jiang, J.F.; Hui, F.M.; Glass, G.E.; Yan, L.; Xu, Y.F.; Zhao, W.J.; Yang, H.; Liu, W.; et al. Predicting the risk of hantavirus infection in Beijing, People's Republic of China. Am. J. Trop. Med. Hyg. 2009, 80, 678–683. [Google Scholar] [PubMed]
- Lee, H.W.; Lee, P.W.; Johnson, K.M. Isolation of the etiologic agent of Korean hemorrhagic fever. J. Infect. Dis. 1978, 137, 298–308. [Google Scholar] [CrossRef] [PubMed]
- Yates, T.L.; Mills, J.N.; Parmenter, C.A.; Ksiazek, T.G.; Parmenter, R.R.; Vande Castle, J.R.; Calisher, C.H.; Nichol, S.T.; Abbott, K.D.; Young, J.C.; et al. The ecology and evolutionary history of an emergent disease: Hantavirus Pulmonary Syndromeevidence from two el niño episodes in the American southwest suggests that el niño–driven precipitation, the initial catalyst of a trophic cascade that results in a delayed density-dependent rodent response, is sufficient to predict heightened risk for human contraction of Hantavirus Pulmonary Syndrome. BioScience 2002, 52, 989–998. [Google Scholar]
- Ken, D.A.; Thomas, G.K.; James, N.M. Long-term Hantavirus persistence in rodent populations in central Arizona. Emerg. Infect. Dis. J. 1999, 5, 102. [Google Scholar]
- Mills, J.N.; Ksiazek, T.G.; Ellis, B.A.; Rollin, P.E.; Nichol, S.T.; Yates, T.L.; Gannon, W.L.; Levy, C.E.; Engelthaler, D.M.; Davis, T. Patterns of association with host and habitat: antibody reactive with Sin Nombre virus in small mammals in the major biotic communities of the southwestern United States. Am. J. Trop. Med. Hyg. 1997, 56, 273–284. [Google Scholar] [CrossRef] [PubMed]
- Glass, G.E.; Cheek, J.E.; Patz, J.A.; Shields, T.M.; Doyle, T.J.; Thoroughman, D.A.; Hunt, D.K.; Enscore, R.E.; Gage, K.L.; Irland, C. Using remotely sensed data to identify areas at risk for Hantavirus Pulmonary Syndrome. Emerg. Infect. Dis. 2000, 6, 238–247. [Google Scholar] [CrossRef] [PubMed]
- Glass, G.E.; Yates, T.L.; Fine, J.B.; Shields, T.M.; Kendall, J.B.; Hope, A.G.; Parmenter, C.A.; Peters, C.; Ksiazek, T.G.; Li, C.H. Satellite imagery characterizes local animal reservoir populations of sin Nombre virus in the Southwestern United States. Proc. Natl. Acad. Sci. USA 2002, 99, 16817–16822. [Google Scholar] [CrossRef] [PubMed]
- Glass, G.E.; Shields, T.; Cai, B.; Yates, T.L.; Parmenter, R. Persistently highest risk areas for Hantavirus pulmonary syndrome: Potential sites for Refugia. Ecol. Appl. 2007, 17, 129–139. [Google Scholar] [CrossRef]
- Boone, J.D.; McGwire, K.C.; Otteson, E.W.; DeBaca, R.S.; Kuhn, E.A.; Villard, P.; Brussard, P.F.; St Jeor, S. Remote sensing and geographic information systems: Charting sin Nombre virus infections in deer mice. Emerg. Infect. Dis. 2000, 6, 248–258. [Google Scholar] [CrossRef] [PubMed]
- Boone, J.D.; Otteson, E.W.; McGwire, K.C.; Villard, P.; Rowe, J.E.; St Jeor, S. Ecology and demographics of Hantavirus infections in rodent populations in the walker river basin of Nevada and California. Am. J. Trop. Med. Hyg. 1998, 59, 445–451. [Google Scholar] [CrossRef] [PubMed]
- Wei, L.; Qian, Q.; Wang, Z.Q.; Glass, G.E.; Song, S.X.; Zhang, W.Y.; Li, X.J.; Yang, H.; Wang, X.J.; Fang, L.Q. Using geographic information system-based ecologic niche models to forecast the risk of hantavirus infection in Shandong Province, China. Am. J. Trop. Med. Hyg. 2011, 84, 497–503. [Google Scholar] [CrossRef] [PubMed]
- Goodin, D.G.; Koch, D.E.; Owen, R.D.; Chu, Y.K.; Hutchinson, J.; Jonsson, C.B. Land cover associated with Hantavirus presence in Paraguay. Global Ecol. Biogeogr. 2006, 15, 519–527. [Google Scholar] [CrossRef]
- Yan, L.; Huang, H.; Zhang, W.; Wang, J.; Ren, Y.; Fang, L.; Huang, X.; Cao, W.; Yan, S.; Wang, S. The relationship between hemorrhagic fever with renal syndrome cases and time series of NDVI in Dayangshu district. J. Remote Sens. 2009, 13, 873–886. [Google Scholar]
- Chen, H.; Qiu, F. Epidemiologic surveillance on the hemorrhagic fever with renal syndrome in China. Chin. Med. J. 1993, 106, 857–863. [Google Scholar] [PubMed]
- Ministry of Health. Handbook of Epidemic Hemorrhagic Fever Prevention and Control; China People’s Health Publishing House: Beijing, China, 1998. [Google Scholar]
- Carlson, T.N.; Ripley, D.A. On the relation between NDVI, fractional vegetation cover, and leaf area index. Remote Sens. Environ. 1997, 62, 241–252. [Google Scholar] [CrossRef]
- Wiegand, C.; Richardson, A.; Escobar, D.; Gerbermann, A. Vegetation indices in crop assessments. Remote Sens. Environ. 1991, 35, 105–119. [Google Scholar] [CrossRef]
- Zhao, M.; Heinsch, F.A.; Nemani, R.R.; Running, S.W. Improvements of the MODIS terrestrial gross and net primary production global data set. Remote Sens. Environ. 2005, 95, 164–176. [Google Scholar] [CrossRef]
- Running, S.W.; Nemani, R.R.; Heinsch, F.A.; Zhao, M.; Reeves, M.; Hashimoto, H. A continuous satellite-derived measure of global terrestrial primary production. Bioscience 2004, 54, 547–560. [Google Scholar] [CrossRef]
- Nachtigall, C.; Kroehne, U. (why) should we use SEM?—Pros and cons of Structural Equation Modeling. Methods Psychol. Res. 2003, 8, 1–22. [Google Scholar]
- Xu, L.; Schmid, B.V.; Liu, J.; Si, X.; Stenseth, N.C.; Zhang, Z. The trophic responses of two different rodent–vector–plague systems to climate change. Proc. R. Soc. B 2015, 282, 20141846. [Google Scholar] [CrossRef] [PubMed]
- Guan, P.; Huang, D.; He, M.; Shen, T.; Guo, J.; Zhou, B. Investigating the effects of climatic variables and reservoir on the incidence of hemorrhagic fever with renal syndrome in Huludao city, China: A 17-year data analysis based on structure equation model. BMC Infect. Dis. 2009, 9, 109. [Google Scholar] [CrossRef] [PubMed]
- PM, B. Comparative fit indexes in structural models. Psychol. Bull. 1990, 107, 238–246. [Google Scholar]
- Zeier, M.; Handermann, M.; Bahr, U.; Rensch, B.; Müller, S.; Kehm, R.; Muranyi, W.; Darai, G. New ecological aspects of Hantavirus infection: A change of a paradigm and a challenge of prevention—A review. Virus Genes 2005, 30, 157–180. [Google Scholar] [CrossRef] [PubMed]
- Kausrud, K.L.; Viljugrein, H.; Frigessi, A.; Begon, M.; Davis, S.; Leirs, H.; Dubyanskiy, V.; Stenseth, N.C. Climatically driven synchrony of gerbil populations allows large-scale plague outbreaks. Proc. Biol. Sci. 2007, 274, 1963–1969. [Google Scholar] [CrossRef] [PubMed]
- Morse, S.S.; Mazet, J.A.; Woolhouse, M.; Parrish, C.R.; Carroll, D.; Karesh, W.B.; Zambrana-Torrelio, C.; Lipkin, W.I.; Daszak, P. Prediction and prevention of the next pandemic zoonosis. Lancet 2012, 380, 1956–1965. [Google Scholar] [CrossRef]
- Xiao, H.; Huang, R.; Gao, L.-D.; Huang, C.-R.; Lin, X.-L.; Li, N.; Liu, H.-N.; Tong, S.-L.; Tian, H.-Y. Effects of humidity variation on the hantavirus infection and hemorrhagic fever with renal syndrome occurrence in Subtropical China. Am. J. Trop. Med. Hyg. 2016, 94, 420–427. [Google Scholar] [CrossRef] [PubMed]
- Tian, H.; Huang, S.; Zhou, S.; Bi, P.; Yang, Z.; Li, X.; Chen, L.; Cazelles, B.; Yang, J.; Luo, L.; et al. Surface water areas significantly impacted 2014 dengue outbreaks in Guangzhou, China. Environ. Res. 2016, 150, 299–305. [Google Scholar] [CrossRef] [PubMed]
- Beck, L.R.; Lobitz, B.M.; Wood, B.L. Remote sensing and human health: New sensors and new opportunities. Emerg. Infect. Dis. 2000, 6, 217–227. [Google Scholar] [CrossRef] [PubMed]
- Addink, E.; De Jong, S.; Davis, S.; Dubyanskiy, V.; Burdelov, L.; Leirs, H. The use of high-resolution remote sensing for plague surveillance in Kazakhstan. Remote Sens. Environ. 2010, 114, 674–681. [Google Scholar] [CrossRef]
- Curran, P.J.; Atkinson, P.M.; Foody, G.M.; Milton, E. Linking remote sensing, land cover and disease. Adv. Parasitol. 2000, 47, 37–80. [Google Scholar] [PubMed]
- Ma, Z.C.; Liu, Z.Y.; Yang, Q.Y.; Ma, H.M.; Di, C.; Xie, X.M. Occurrence regularity of apodemus agrarius and its control technique in Guanzhong area, Shaanxi. Chin. Countrys. Well-off Technol. 2008, 14, 51–54. (In Chinese) [Google Scholar]
- Luo, C.W.; Chen, H.X. Study on the factors infl uenced epidemic of hemorrhagic fever with renal syndrome. Chin. J. Vector Biol. Control 2003, 14, 4. [Google Scholar]
- Yan, L.; Fang, L.Q.; Huang, H.G.; Zhang, L.Q.; Feng, D.; Zhao, W.J.; Zhang, W.Y.; Li, X.W.; Cao, W.C. Landscape elements and Hantaan virus–related hemorrhagic fever with renal syndrome, People’s Republic of China. Emerg. Infect. Dis. 2007, 13, 1301–1306. [Google Scholar] [CrossRef] [PubMed]
- Fang, L.; Yan, L.; Liang, S.; de Vlas, S.; Feng, D.; Han, X.; Zhao, W.; Xu, B.; Bian, L.; Yang, H. Spatial analysis of hemorrhagic fever with renal syndrome in China. BMC Infect. Dis. 2006, 6, 77. [Google Scholar] [CrossRef] [PubMed]
- Tian, H.; Yu, P.; Luis, A.D.; Bi, P.; Cazelles, B.; Laine, M.; Huang, S.; Ma, C.; Zhou, S.; Wei, J. Changes in rodent abundance and weather conditions potentially drive hemorrhagic fever with renal syndrome outbreaks in Xi'an, China, 2005–2012. PLoS Negl. Trop. Dis. 2015, 9, e0003530. [Google Scholar] [CrossRef] [PubMed]
- Chelkowska, H.; Walkowa, W.; Adamczyk, K. Spatial relationships in sympatric populations of the rodents: Clethrionomys glareolus, microtus agrestis and apodemus agrarius. Acta Theriol. 1985, 30, 51–58. [Google Scholar] [CrossRef]
- Krebs, M.A. Effects of Understory Vegetation on the Photosynthesis and Leaf Water Potential of Young Douglas Fir Trees on Two Contrasting Sites in Northwestern Montana; The University of Montana: Missoula, MT, USA, 2003. [Google Scholar]
- Luis, A.D.; Douglass, R.J.; Mills, J.N.; Bjørnstad, O.N. The effect of seasonality, density and climate on the population dynamics of Montana deer mice, important reservoir hosts for sin Nombre Hantavirus. J. Anim. Ecol. 2010, 79, 462–470. [Google Scholar] [CrossRef] [PubMed]
- Stenseth, N.C.; Mysterud, A.; Ottersen, G.; Hurrell, J.W.; Chan, K.S.; Lima, M. Ecological effects of climate fluctuations. Science 2002, 297, 1292–1296. [Google Scholar] [CrossRef] [PubMed]
- Kausrud, K.L.; Mysterud, A.; Steen, H.; Vik, J.O.; Østbye, E.; Cazelles, B.; Framstad, E.; Eikeset, A.M.; Mysterud, I.; Solhøy, T. Linking climate change to lemming cycles. Nature 2008, 456, 93–97. [Google Scholar] [CrossRef] [PubMed]
- Yan, C.; Xu, L.; Xu, T.; Cao, X.; Wang, F.; Wang, S.; Hao, S.; Yang, H.; Zhang, Z. Agricultural irrigation mediates climatic effects and density dependence in population dynamics of Chinese striped hamster in North China plain. J. Anim. Ecol. 2013, 82, 334–344. [Google Scholar] [CrossRef] [PubMed]
| MODIS Land Product | Indicator | Spatial Resolution | Temporal Resolution | Temporal Period |
|---|---|---|---|---|
| MOD13A2 | NDVI | 1-km | 16-day | 2005–2012 |
| MOD15A2 | LAI | 1-km | 8-day | 2005–2012 |
| MOD15A2 | FPAR | 1-km | 8-day | 2005–2012 |
| MOD17A2 | GPP | 1-km | 8-day | 2005–2012 |
| MOD17A2 | PsnNet | 1-km | 8-day | 2005–2012 |
| MOD11A2 | LST | 1-km | 8-day | 2005–2012 |
| Year | 2005 | 2006 | 2007 | 2008 | 2009 | 2010 | 2011 | 2012 |
|---|---|---|---|---|---|---|---|---|
| Incidence of HFRS (1/10, 000) | 0.90 | 1.11 | 1.51 | 1.61 | 2.23 | 4.62 | 3.46 | 2.64 |
| Lag (month) | 0 | 1 | 2 | 3 | 4 | 5 | 6 |
|---|---|---|---|---|---|---|---|
| r | 0.19 | 0.31 ** | 0.41 ** | 0.42 ** | 0.39 ** | 0.31 ** | 0.13 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yu, P.; Li, Y.; Xu, B.; Wei, J.; Li, S.; Dong, J.; Qu, J.; Xu, J.; Huang, Z.Y.X.; Ma, C.; et al. Using Satellite Data for the Characterization of Local Animal Reservoir Populations of Hantaan Virus on the Weihe Plain, China. Remote Sens. 2017, 9, 1076. https://doi.org/10.3390/rs9101076
Yu P, Li Y, Xu B, Wei J, Li S, Dong J, Qu J, Xu J, Huang ZYX, Ma C, et al. Using Satellite Data for the Characterization of Local Animal Reservoir Populations of Hantaan Virus on the Weihe Plain, China. Remote Sensing. 2017; 9(10):1076. https://doi.org/10.3390/rs9101076
Chicago/Turabian StyleYu, Pengbo, Yidan Li, Bo Xu, Jing Wei, Shen Li, Jianhua Dong, Jianhui Qu, Jing Xu, Zheng Y.X. Huang, Chaofeng Ma, and et al. 2017. "Using Satellite Data for the Characterization of Local Animal Reservoir Populations of Hantaan Virus on the Weihe Plain, China" Remote Sensing 9, no. 10: 1076. https://doi.org/10.3390/rs9101076
APA StyleYu, P., Li, Y., Xu, B., Wei, J., Li, S., Dong, J., Qu, J., Xu, J., Huang, Z. Y. X., Ma, C., Yang, J., Zhang, G., Chen, B., Huang, S., Shi, C., Gao, H., Liu, F., Tian, H., Stenseth, N. C., ... Wang, J. (2017). Using Satellite Data for the Characterization of Local Animal Reservoir Populations of Hantaan Virus on the Weihe Plain, China. Remote Sensing, 9(10), 1076. https://doi.org/10.3390/rs9101076






