Amino Acid: Its Dual Role as Nutrient and Scavenger of Free Radicals in Soil
Abstract
:1. Introduction
2. Materials and Methods
2.1. Sampling and Processing
- (1)
- Middle-aged mixed stand of deciduous trees (33 years, oak 60%, hornbeam 30%, beech 10%; 415 m a.s.l., N 49°17′, E 16°38′, Haplic Cambisol) from the Ah horizon
- (2)
- Deciduous trees (95 years, beech 52%, European ash 15%, larch 15%, spruce 9%, fir 5%, oak 2%, Douglas fir 1%, hornbeam 1%; 490 m a.s.l., N 49°19′, E 16°40′, Rendzic Humic Leptosol) from the Oe, Ahk, and Bwk horizons;
- (3)
- Old-aged spruce stand (103 years, spruce 90%, beech 10%, 480 m a.s.l., N 49°19′, E 16°47′, Haplic Cambisol) from the Oe horizon
- (4)
- Old-aged beech stand (207 years, beech 99%, oak 1%, 480 m a.s.l., N 49°19′, E 16°47′, Dystric Luvisol) from the Oe and Aa horizon
2.2. Treatments and Soil Respiration
- A = amount of HCl for titration of control (mL);
- B = amount of HCl for titration of sample (mL);
- c(HCl) = exact concentration of HCl (mol/L);
- 6005 = coefficient for recalculation (from V (HCl) to a μg C-CO2).
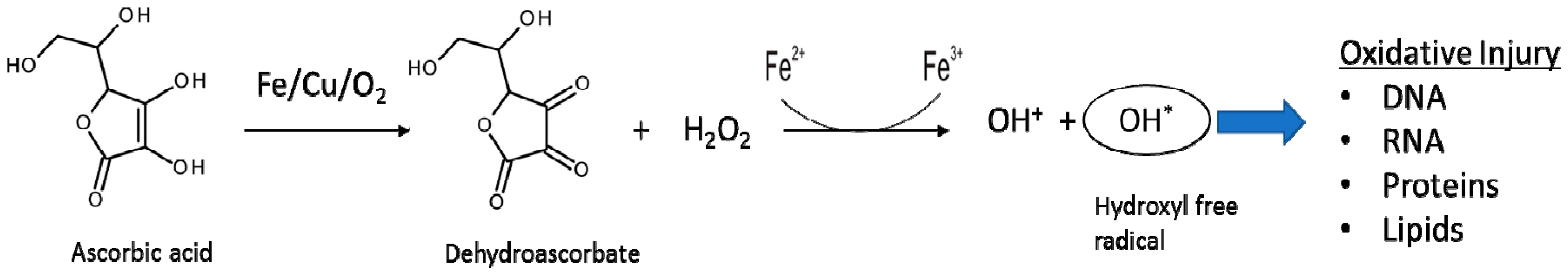
2.3. Reactions Involved
2.4. Statistical Analysis
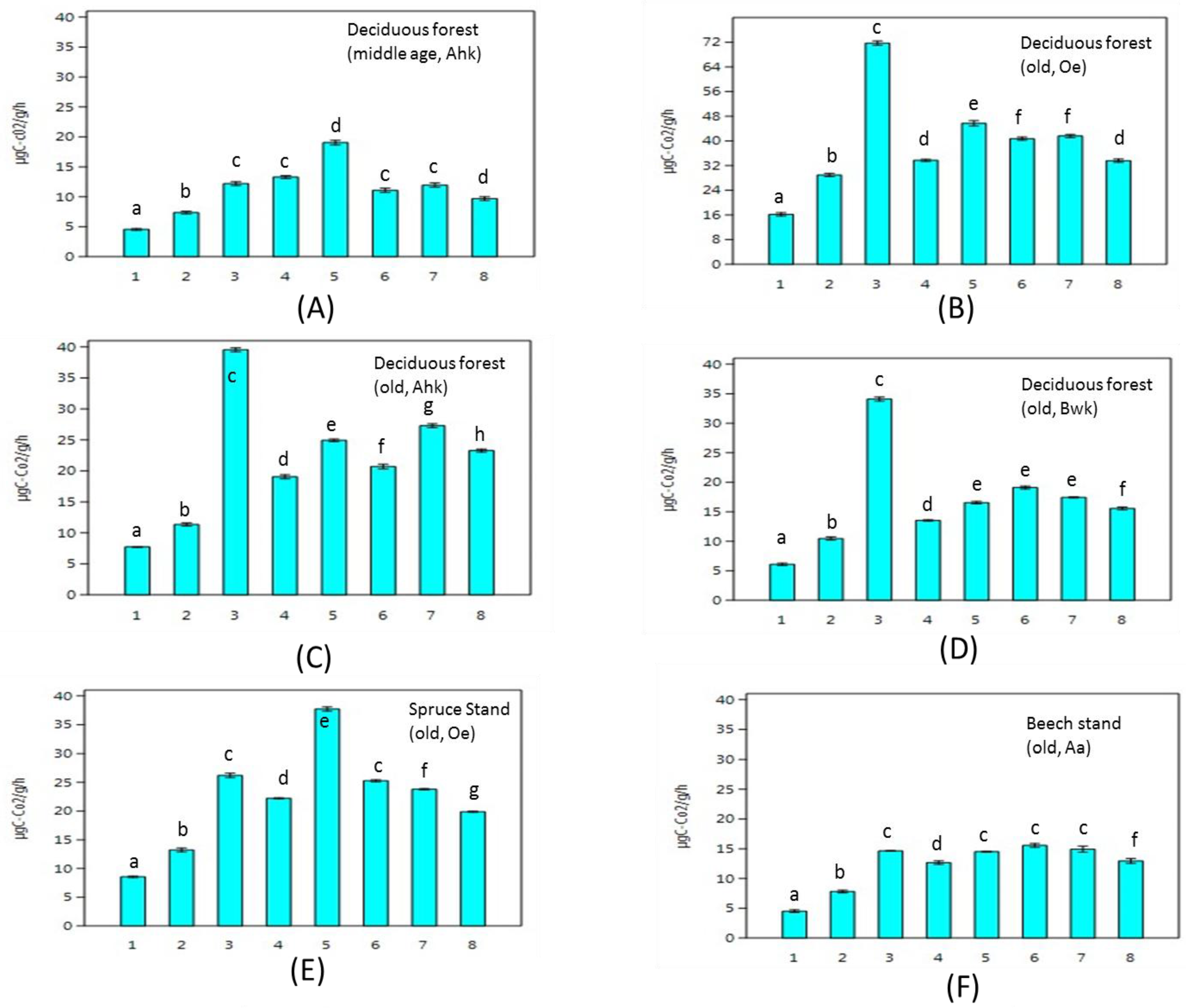
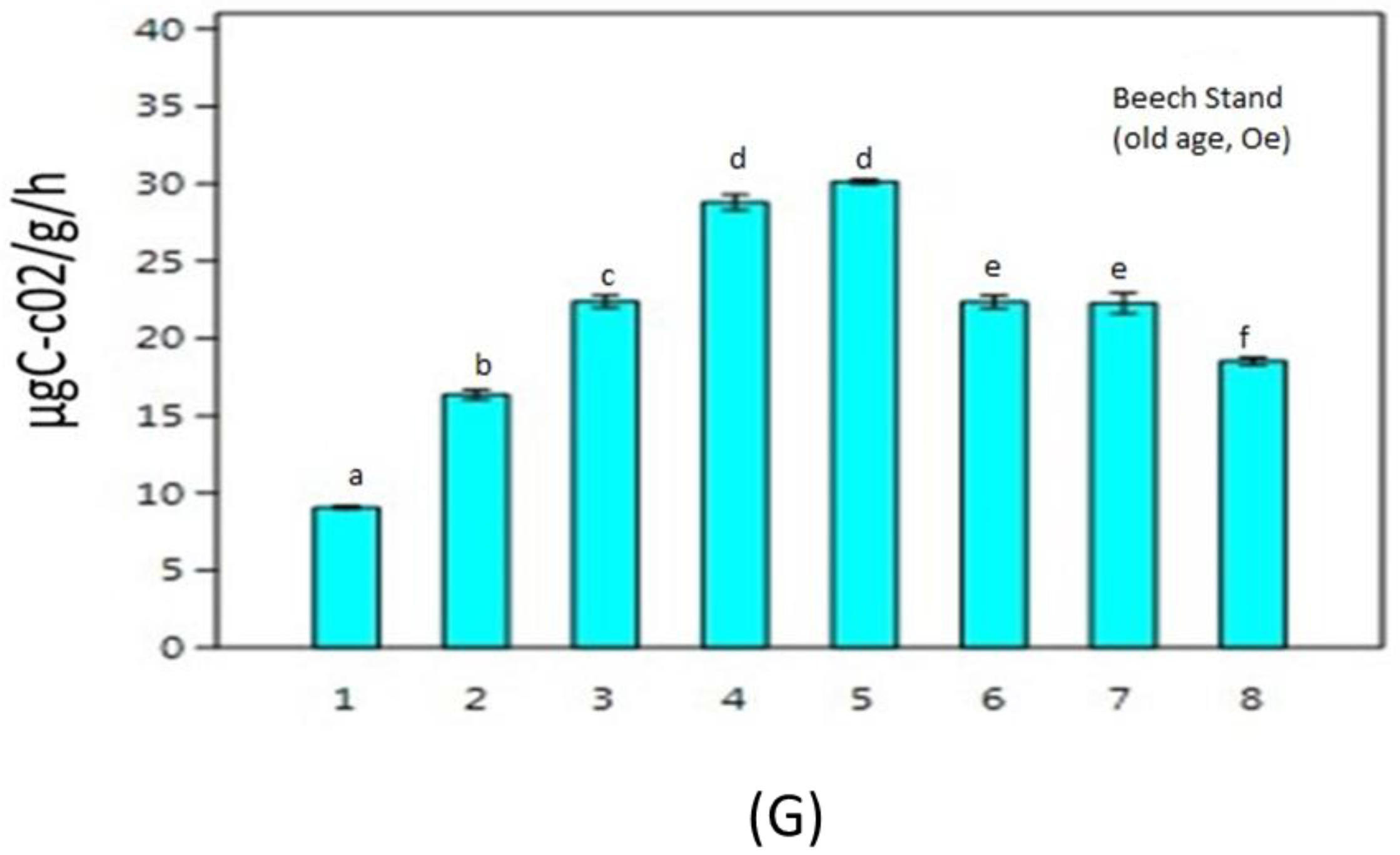
3. Results
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Nakashima, K.; Kiyosue, T.; Yamaguchi-Shinozaki, K.; Shinozaki, K. A nuclear gene, erd1, encoding a chloroplast-targeted Clp protease regulatory subunit homolog is not only induced by water stress but also developmentally up-regulated during senescence in Arabidopsis thaliana. Plant J. 1997, 12, 851–861. [Google Scholar] [CrossRef] [PubMed]
- Weaver, L.M.; Gan, S.; Quirino, B.; Amasino, R.M. A comparison of the expression patterns of several senescence-associated genes in response to stress and hormone treatment. Plant Mol. Biol. 1998, 37, 455–469. [Google Scholar] [CrossRef] [PubMed]
- Arrigoni, O.; de Tullio, M.C. Ascorbic acid: Much more than just an antioxidant. Biochim. Biophys. Acta 2002, 1569, 1–9. [Google Scholar] [CrossRef]
- Arrigoni, O.; de Tullio, M.C. The role of ascorbic acid in cell metabolism: Between gene-directed functions and unpredictable chemical reactions. J. Plant Physiol. 2000, 157, 481–488. [Google Scholar] [CrossRef]
- Kivirikko, K.I.; Pihlajaniemi, T. Collagen hydroxylases and the protein disulfide isomerase subunit of prolyl 4-hydroxylase. Adv. Enzym. Rel. Areas Mol. Biol 2009, 72, 325–398. [Google Scholar]
- Wojtaszek, P.; Smith, C.G.; Bolwell, G.P. Ultrastructural localisation and further biochemical characterisation of prolyl 4-hydroxylase from Phaseolus vulgaris: Comparative analysis. Int. J. Biochem. Cell Biol. 1999, 31, 463–477. [Google Scholar] [CrossRef]
- Eskling, M.; Arvidsson, P.; Åkerlund, H. The xanthophyll cycle, its regulation and components. Physiol. Plant 1997, 100, 806–816. [Google Scholar] [CrossRef]
- Davies, M.B.; Austin, J.; Partridge, D.A. Vitamin C: Its Chemistry and Biochemistry; Royal Society of Chemistry: London, UK, 1991. [Google Scholar]
- Smith, J.J.; Ververidis, P.; John, P. Characterization of the ethylene-forming enzyme partially purified from melon. Phytochemistry 1992, 31, 1485–1494. [Google Scholar] [CrossRef]
- Pastori, G.M.; Kiddle, G.; Antoniw, J.; Bernard, S.; Veljovic-Jovanovic, S.; Verrier, P.J.; Noctor, G.; Foyer, C.H. Leaf vitamin C contents modulate plant defense transcripts and regulate genes that control development through hormone signaling. Plant Cell 2003, 15, 939–951. [Google Scholar] [CrossRef] [PubMed]
- Barth, C.; Moeder, W.; Klessig, D.F.; Conklin, P.L. The timing of senescence and response to pathogens is altered in the ascorbate-deficient Arabidopsis mutant vitamin c-1. Plant Physiol. 2004, 134, 1784–1792. [Google Scholar] [CrossRef] [PubMed]
- Pavet, V.; Olmos, E.; Kiddle, G.; Mowla, S.; Kumar, S.; Antoniw, J.; Alvarez, M.E.; Foyer, C.H. Ascorbic acid deficiency activates cell death and disease resistance responses in Arabidopsis. Plant Physiol. 2005, 139, 1291–1303. [Google Scholar] [CrossRef] [PubMed]
- Roig, M.G.; Rivera, Z.S.; Kennedy, J.F. L-ascorbic acid: An overview. Int. J. Food Sci. Nutr. 1993, 44, 59–72. [Google Scholar] [CrossRef]
- Gnarpe, H.; Michaelsson, M.; Dreborg, S. The in vitro effect of ascorbic acid on the bacterial growth in urine. Acta Pathol. Microbiol. Scand. 2009, 74, 41–50. [Google Scholar] [CrossRef]
- Rawal, B.D. Bactericidal Action of Ascorbic Acid on Pseudomonas aeruginosa: Alteration of Cell Surface as a Possible Mechanism. Chemotherapy 1978, 24, 166–171. [Google Scholar] [CrossRef] [PubMed]
- Datta, R.; Kelkar, A.; Baraniya, D.; Molaei, A.; Moulick, A.; Meena, R.S.; Formanek, P. Enzymatic Degradation of Lignin in Soil: A Review. Sustainability 2017, 9, 1163. [Google Scholar] [CrossRef]
- Arakawa, H.; Maeda, M.; Okubo, S.; Shimamura, T. Role of Hydrogen Peroxide in Bactericidal Action of Catechin. Biol. Pharm. Bull. 2004, 27, 277–281. [Google Scholar] [CrossRef] [PubMed]
- Carvalhais, L.C.; Dennis, P.G.; Fedoseyenko, D.; Hajirezaei, M.-R.; Borriss, R.; von Wirén, N. Root exudation of sugars, amino acids, and organic acids by maize as affected by nitrogen, phosphorus, potassium, and iron deficiency. J. Plant Nutr. Soil Sci. 2010, 174, 3–11. [Google Scholar] [CrossRef]
- Moe, L.A. Amino acids in the rhizosphere: From plants to microbes. Am. J. Bot. 2013, 100, 1692–1705. [Google Scholar] [CrossRef] [PubMed]
- Kuzyakov, Y. Sources of CO2 efflux from soil and review of partitioning methods. Soil Biol. Biochem. 2006, 38, 425–448. [Google Scholar] [CrossRef]
- Ferréa, C.; Zenone, T.; Comolli, R.; Seufert, G. Estimating heterotrophic and autotrophic soil respiration in a semi-natural forest of Lombardy, Italy. Pedobiologia 2012, 55, 285–294. [Google Scholar] [CrossRef]
- Liu, B.; Li, Y.; Zhang, X.; Wang, J.; Gao, M. Effects of chlortetracycline on soil microbial communities: Comparisons of enzyme activities to the functional diversity via Biolog EcoPlatesTM. Eur. J. Soil Biol. 2015, 68, 69–76. [Google Scholar] [CrossRef]
- Beverly, D.; Franklin, S. Heterotrophic and Autotrophic Soil Respiration under Simulated Dormancy Conditions. Open J. For. 2015, 5, 274–286. [Google Scholar] [CrossRef]
- Ryan, M.G.; Law, B.E. Interpreting, measuring, and modeling soil respiration. Biogeochemistry 2005, 73, 3–27. [Google Scholar] [CrossRef]
- Li, Y.; Xu, M.; Zou, X. Heterotrophic Soil Respiration in Relation to Environmental Factors and Microbial Biomass in Two Wet Tropical Forests. Plant Soil 2006, 281, 193–201. [Google Scholar] [CrossRef]
- Cardoso, E.J.B.N.; Vasconcellos, R.L.F.; Bini, D.; Miyauchi, M.Y.H.; dos Santos, C.A.; Alves, P.R.L.; de Paula, A.M.; Nakatani, A.S.; de Moraes Pereira, J.; Nogueira, M.A. Soil health: Looking for suitable indicators. What should be considered to assess the effects of use and management on soil health? Sci. Agric. 2013, 70, 274–289. [Google Scholar] [CrossRef]
- Laishram, J.; Saxena, K.G.; Maikhuri, R.K.; Rao, K.S. Soil quality and soil health: A review. Int. J. Ecol. Environ. Sci. 2012, 38, 19–37. [Google Scholar]
- Schinner, F.; Ohlinger, R.; Kandeler, D.E.; Margesin, R. Methods in Soil Biology; Springer: Berlin, Germany, 1996; pp. 406–410. [Google Scholar]
- Rajapaksha, R.M.C.P.; Tobor-Kaplon, M.A.; Baath, E. Metal Toxicity Affects Fungal and Bacterial Activities in Soil Differently. Appl. Environ. Microbiol. 2004, 70, 2966–2973. [Google Scholar] [CrossRef] [PubMed]
- Mazzetto, A.M.; Feigl, B.J.; Cerri, C.E.P.; Cerri, C.C. Comparing how land use change impacts soil microbial catabolic respiration in Southwestern Amazon. Braz. J. Microbiol. 2016, 47, 63–72. [Google Scholar] [CrossRef] [PubMed]
- Lorenz, K.; Lal, R. The Depth Distribution of Soil Organic Carbon in Relation to Land Use and Management and the Potential of Carbon Sequestration in Subsoil Horizons. Adv. Agron. 2005, 35–66. [Google Scholar]
- Pereira, M.G.; Valladares, G.S.; dos Anjos, L.H.C.; de Melo Benites, V.; Espíndula, A., Jr.; Ebeling, A.G. Organic carbon determination in histosols and soil horizons with high organic matter content from Brazil. Sci. Agric. 2006, 63, 187–193. [Google Scholar] [CrossRef]
- Rantoa, N.R.; van Huyssteen, C.W.; du Preez, C.C. Organic Carbon Content in the Soil Master Horizons of South Africa. Vadose Zo. J. 2015, 14. Available online: http://hdl.handle.net/11660/1757 (accessed on 8 August 2017). [CrossRef]
- Asgharipour, M.R.; Rafiei, M. The effects of land use on biomass and catabolic diversity of soil microbial communities. Afr. J. Agric. Res. 2011, 6, 4607–4612. [Google Scholar]
- Kourtev, P.S.; Ehrenfeld, J.G.; Häggblom, M. Experimental analysis of the effect of exotic and native plant species on the structure and function of soil microbial communities. Soil Biol. Biochem. 2003, 35, 895–905. [Google Scholar] [CrossRef]
- Frey, S.D.; Drijber, R.; Smith, H.; Melillo, J. Microbial biomass, functional capacity, and community structure after 12 years of soil warming. Soil Biol. Biochem. 2008, 40, 2904–2907. [Google Scholar] [CrossRef]
- Sinsabaugh, R.L.; Belnap, J.; Rudgers, J.; Kuske, C.R.; Martinez, N.; Sandquist, D. Soil microbial responses to nitrogen addition in arid ecosystems. Front. Microbiol. 2015, 6, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Broughton, R.C.I.; Newsham, K.K.; Hill, P.W.; Stott, A.; Jones, D.L. Differential acquisition of amino acid and peptide enantiomers within the soil microbial community and its implications for carbon and nitrogen cycling in soil. Soil Biol. Biochem. 2015, 88, 83–89. [Google Scholar] [CrossRef]
- Farrell, M.; Prendergast-Miller, M.; Jones, D.L.; Hill, P.W.; Condron, L.M. Soil microbial organic nitrogen uptake is regulated by carbon availability. Soil Biol. Biochem. 2014, 77, 261–267. [Google Scholar] [CrossRef]
- Hopkins, D.W. D- and L-amino acid metabolism in soil. Prog. Nitrogen Cycl. Stud. 1996, 57–61. [Google Scholar]
- Kemmitt, S.J.; Wright, D.; Murphy, D.V.; Jones, D.L. Regulation of amino acid biodegradation in soil as affected by depth. Biol. Fertil. Soils 2008, 44, 933–941. [Google Scholar] [CrossRef]
- Eddy, B.P.; Ingram, M. Interactions between ascorbic acid and bacteria. Bacteriol. Rev. 1953, 17, 93. [Google Scholar] [PubMed]
- Udenigwe, C.C.; Aluko, R.E. Chemometric analysis of the amino acid requirements of antioxidant food protein hydrolysates. Int. J. Mol. Sci. 2011, 12, 3148–3161. [Google Scholar] [CrossRef] [PubMed]
- Masek, A.; Chrzescijanska, E.; Zaborski, M. Estimation of the antioxidative properties of amino acids—An electrochemical approach. Int. J. Electrochem. Sci. 2014, 9, 7904. [Google Scholar]
- Nussbaum, M.A.; Przedwiecki, J.E.; Staerk, D.U.; Lunte, S.M.; Riley, C.M. Electrochemical characteristics of amino acids and peptides derivatized with naphthalene-2, 3-dicarboxaldehyde: pH effects and differences in oxidation potentials. Anal. Chem. 1992, 64, 1259–1263. [Google Scholar] [CrossRef] [PubMed]
- Dobson, C.M.; Winter, N.S. The Identification of Amino Acids by Interpretation of Titration Curves: An Undergraduate Experiment for Biochemistry. World J. Chem. Educ. 2014, 2, 59–61. [Google Scholar]
- Nelson, D.L.; Lehninger, A.L.; Cox, M.M. Lehninger Principles of Biochemistry; Worth Publishers: Basingstoke, UK, 2008. [Google Scholar]
- Fontaine, S.; Mariotti, A.; Abbadie, L. The priming effect of organic matter: A question of microbial competition? Soil Biol. Biochem. 2003, 35, 837–843. [Google Scholar] [CrossRef]
| Sampling site | Figureure abbr. | Corg (%) | Nt (%) | C/N | pH/H2O | pH/CaCl2 | Clay (%) | Silt (%) | Sand (%) |
|---|---|---|---|---|---|---|---|---|---|
| Deciduous forest (middle age, Haplic Cambisol, Ah horizon) | Soil A | 4.3 | 0.3 | 14.8 | 5.1 | 3.8 | 6.8 | 50 | 43.2 |
| Deciduous forest (old age, Rendzic Humic Leptosol, Oe horizon) | Soil B | 26.2 | 2.0 | 13.1 | 7.7 | 7.2 | NA | NA | NA |
| Deciduous forest (old age, Rendzic Humic Leptosol, Ahk horizon) | Soil C | 11.1 | 1.0 | 11.4 | 7.5 | 7.1 | 12.5 | 65 | 22.5 |
| Deciduous forest (old age, Rendzic Humic Leptosol, Bwk horizon) | Soil D | 2.2 | 0.2 | 10.5 | 7.1 | 6.6 | 23.2 | 60.3 | 16.5 |
| Spruce stand (old age, Haplic Cambisol, Oe horizon) | Soil E | 33.6 | 1.5 | 23 | 4.8 | 3.9 | NA | NA | NA |
| Beech stands (old age, Dystric Luvisol, Aa horizon) | Soil F | 7.6 | 0.32 | 23.7 | 4.1 | 3.4 | 6.5 | 48.8 | 44.7 |
| Beech stands (old age, Dystric Luvisol, Oe horizon) | Soil G | 25.1 | 0.86 | 29.2 | 4.4 | 3.3 | NA | NA | NA |
| Treatment | Soil Type | Highest % Increase in Respiration |
|---|---|---|
| Ascorbic acid (AA) | Deciduous forest (old age, Rendzic Humic Leptosol, Oe horizon) | 78 |
| AA + Beta Alanine | Deciduous forest (old age, Rendzic Humic Leptosol, Oe horizon) | 147 |
| AA + D-glutamic acid | Beech stand (old age, Dystric Luvisol, Oe horizon) | 184 |
| AA + L-glutamic acid | Spruce stand (old age, Haplic Cambisol, Oe horizon) | 185 |
| AA + L-Citrulline | Spruce stand (old age, Haplic Cambisol, Oe horizon) | 188 |
| AA + L-Ornithine | Deciduous forest (old age, Rendzic Humic Leptosol, Ahk horizon) | 140 |
| AA + D-Ornithine | Deciduous forest (old age, Rendzic Humic Leptosol, Ahk horizon) | 105 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Datta, R.; Baraniya, D.; Wang, Y.-F.; Kelkar, A.; Meena, R.S.; Yadav, G.S.; Teresa Ceccherini, M.; Formanek, P. Amino Acid: Its Dual Role as Nutrient and Scavenger of Free Radicals in Soil. Sustainability 2017, 9, 1402. https://doi.org/10.3390/su9081402
Datta R, Baraniya D, Wang Y-F, Kelkar A, Meena RS, Yadav GS, Teresa Ceccherini M, Formanek P. Amino Acid: Its Dual Role as Nutrient and Scavenger of Free Radicals in Soil. Sustainability. 2017; 9(8):1402. https://doi.org/10.3390/su9081402
Chicago/Turabian StyleDatta, Rahul, Divyashri Baraniya, Yong-Feng Wang, Aditi Kelkar, Ram Swaroop Meena, Gulab Singh Yadav, Maria Teresa Ceccherini, and Pavel Formanek. 2017. "Amino Acid: Its Dual Role as Nutrient and Scavenger of Free Radicals in Soil" Sustainability 9, no. 8: 1402. https://doi.org/10.3390/su9081402
APA StyleDatta, R., Baraniya, D., Wang, Y.-F., Kelkar, A., Meena, R. S., Yadav, G. S., Teresa Ceccherini, M., & Formanek, P. (2017). Amino Acid: Its Dual Role as Nutrient and Scavenger of Free Radicals in Soil. Sustainability, 9(8), 1402. https://doi.org/10.3390/su9081402









