Abandonment, Ecological Assembly and Public Health Risks in Counter-Urbanizing Cities
Abstract
:1. Introduction
2. Counter-Urbanization: Legacies, form, Ecosystem Structure and (Dis)services
2.1. Landscape Legacies and Urban Forms
2.2. Ecosystem Structure, Function, and (Dis)Services
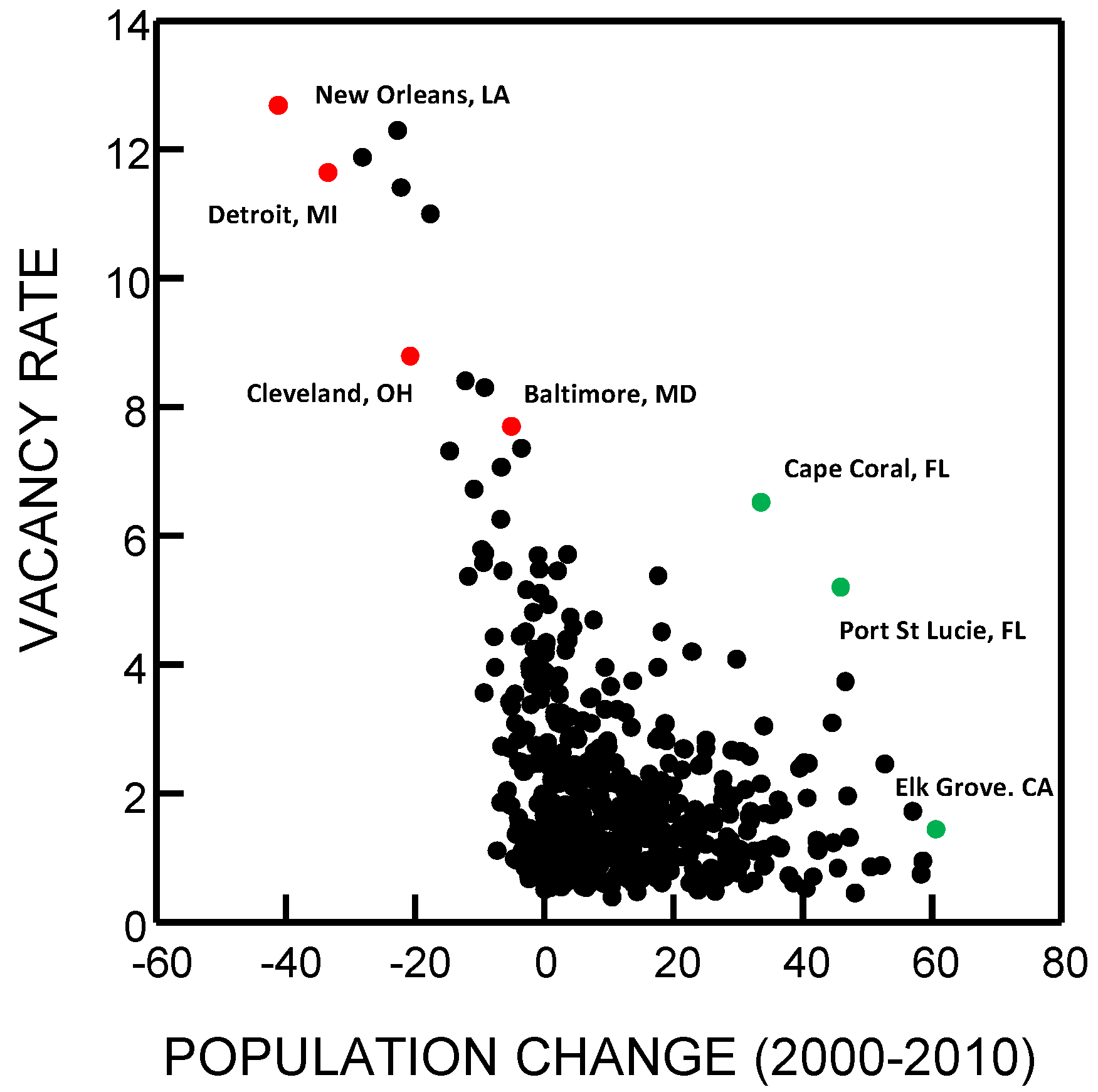
3. Counter-Urbanization and Abandonment
3.1. Urban Versus Counter-Urban “Greening”
3.2. Configurations and Perceptions of Abandonment
3.3. Abandonment and Assembly
3.4. Abandonment and Infrastructure Decline
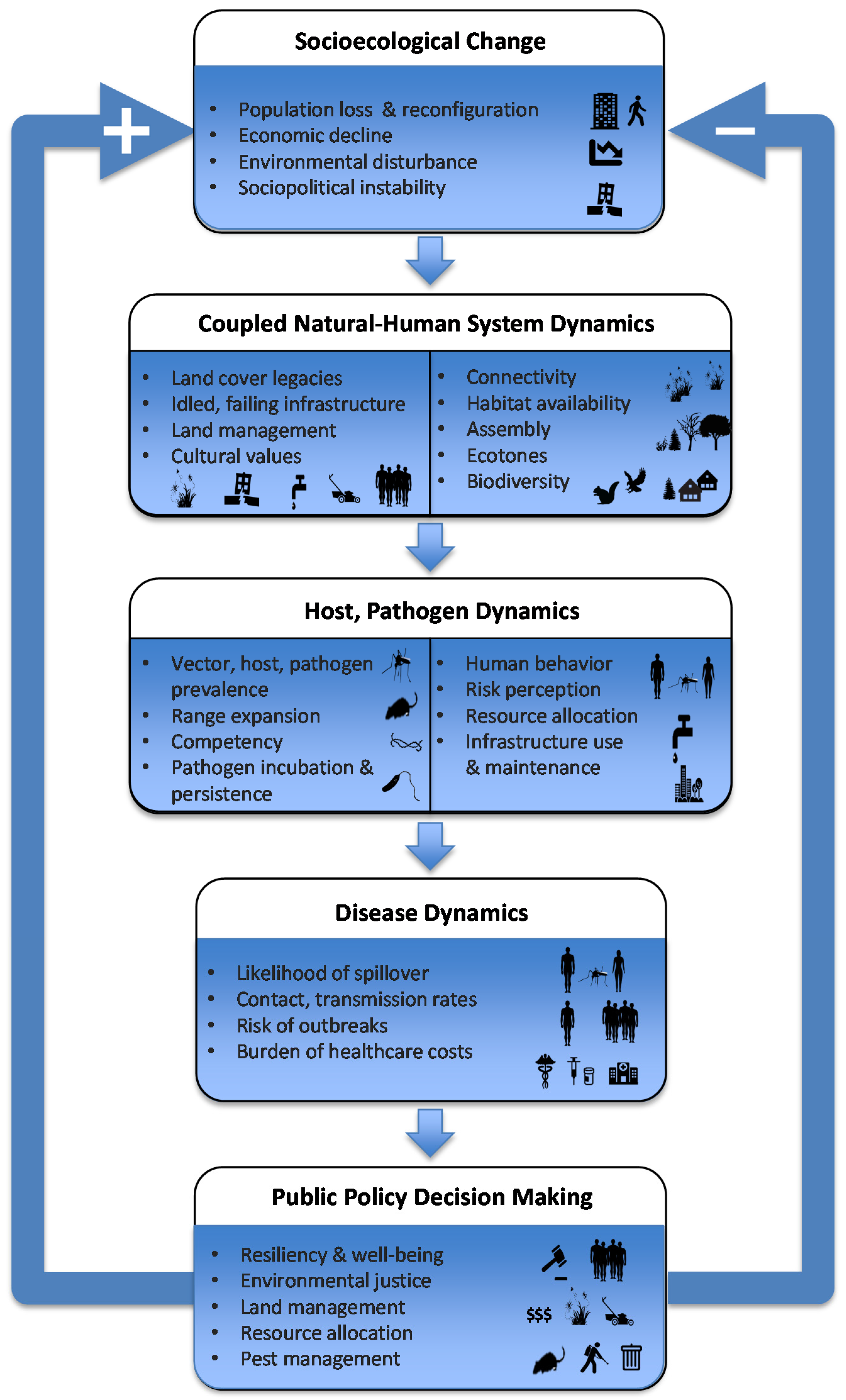
4. Counter-Urbanization, Abandonment and Human Health
4.1. Cities and Emerging Infectious Disease
4.2. Counter-Urbanization and Emerging Infectious Disease
4.3. Abandonment, Biodiversity and Emerging Infectious Disease
4.4. Abandonment and Exposure Risk
4.5. Infrastructure Decline and Exposure Risk
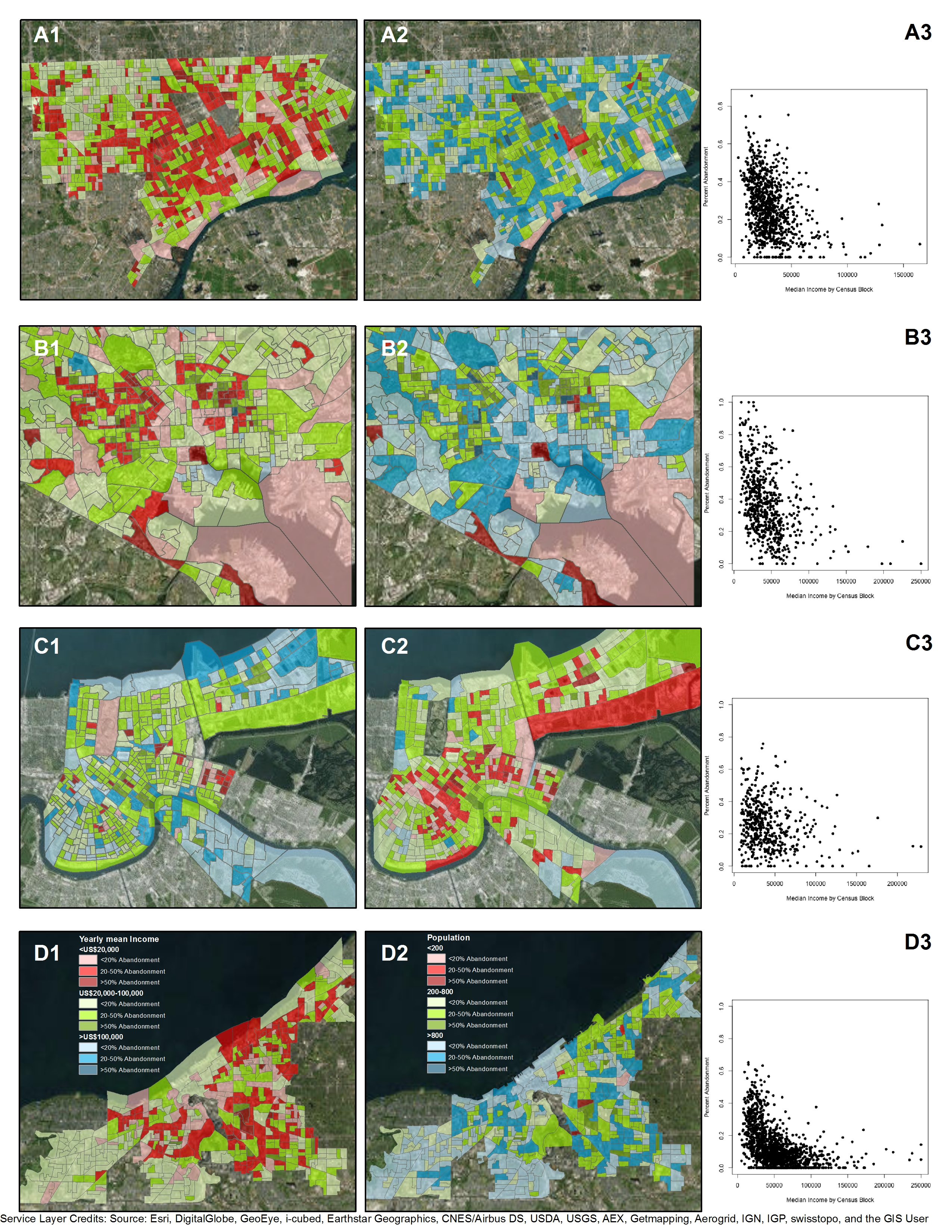
5. A Tale of Two Neighborhoods: Health Risks of Managing Abandonment and Depopulation in New Orleans
6. Implications
6.1. Socioecological Resilience and Environmental Justice
6.2. Planning for Decline?
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| BWA | Boil Water Advisory |
| EID | Emerging Infectious Disease |
| HPAI | highly pathogenic avian influenza |
| NORA | New Orleans Redevelopment Authority |
| PAM | primary amebic meningoencephalitis |
| ULI | Urban Land Institute |
| US | United States of America |
References
- Martinez-Fernandez, C.; Audirac, I.; Fol, S.; Cunningham-Sabot, E. Shrinking Cities: Urban Challenges of Globalization: Shrinking cities: urban challenges of globalization. Int. J. Urban Reg. Res. 2012, 36, 213–225. [Google Scholar] [CrossRef] [PubMed]
- Pallagst, K.; Martinez-Fernandez, C.; Wiechmann, T. Shrinking Cities: An Emerging Planning Phenomenon. In Shrinking Cities: International Perspectives and Policy Implications; Routledge: New York, NY, USA, 2014. [Google Scholar]
- Beyer, E.; Hagemann, A.; Rieniets, T.; Oswalt, P. Atlas of Shrinking Cities; Hatje Cantz Publishers: Berlin, Germany, 2006. [Google Scholar]
- Kabisch, S.; Haase, A.; Haase, D. Beyond Growth–Urban Development in Shrinking Cities as a Challenge for Modeling Approaches. Available online: http://www.iemss.org/iemss2006/sessions/all.html (accessed on 16 May 2016).
- Hollander, J.B.; Pallagst, K.; Schwarz, T.; Popper, F.J. Planning shrinking cities. Prog. Plan. 2009, 72, 223–232. [Google Scholar]
- Burkholder, S. The New Ecology of Vacancy: Rethinking Land Use in Shrinking Cities. Sustainability 2012, 4, 1154–1172. [Google Scholar] [CrossRef]
- U.S Census Bureau American FactFinder. Available online: http://factfinder.census.gov/faces/nav/jsf/pages/index.xhtml (accessed on 1 December 2015).
- Iverson Nassauer, J.; Raskin, J. Urban vacancy and land use legacies: A frontier for urban ecological research, design, and planning. Landsc. Urban Plan. 2014, 125, 245–253. [Google Scholar] [CrossRef]
- Lewis, J.A.; Zipperer, W.C.; Hazen, R.; Blum, M.J.; Elmqvist, T.; Henrik, E. Socioecological responses of urban vegetation to catastrophic flooding and land abandonment in New Orleans following Hurricane Katrina, 2015. Ecosphere. In review.
- Ramalho, C.E.; Hobbs, R.J. Time for a change: dynamic urban ecology. Trends Ecol. Evol. 2012, 27, 179–188. [Google Scholar] [CrossRef] [PubMed]
- Levy, M.Z.; Barbu, C.M.; Castillo-Neyra, R.; Quispe-Machaca, V.R.; Ancca-Juarez, J.; Escalante-Mejia, P.; Borrini-Mayori, K.; Niemierko, M.; Mabud, T.S.; Behrman, J.R.; et al. Urbanization, land tenure security and vector-borne Chagas disease. Proc. R. Soc. Lond. B Biol. Sci. 2014. [Google Scholar] [CrossRef] [PubMed]
- Pickett, S.T.A.; Cadenasso, M.L.; Grove, J.M.; Boone, C.G.; Groffman, P.M.; Irwin, E.; Kaushal, S.S.; Marshall, V.; McGrath, B.P.; Nilon, C.H.; et al. Urban ecological systems: Scientific foundations and a decade of progress. J. Environ. Manag. 2011, 92, 331–362. [Google Scholar] [CrossRef] [PubMed]
- Wallace, D.; Wallace, R. Urban systems during disasters: factors for resilience. Ecol. Soc. 2008, 13. Article 18. [Google Scholar]
- Kinzig, A.P.; Warren, P.; Martin, C.; Hope, D.; Katti, M. The effects of human socioeconomic status and cultural characteristics on urban patterns of biodiversity. Ecol. Soc. 2005, 10. Article 23. [Google Scholar]
- McDonnell, M.; Pickett, S.T. Ecosystem Structure and Function along Urban-Rural Gradients: An unexploited Opportunity for Ecology. Ecology 1990, 71, 1232–1237. [Google Scholar] [CrossRef]
- Shochat, E.; Warren, P.S.; Faeth, S.H.; McIntyre, N.E.; Hope, D. From patterns to emerging processes in mechanistic urban ecology. Trends Ecol. Evol. 2006, 21, 186–191. [Google Scholar] [CrossRef] [PubMed]
- Godefroid, S.; Koedam, N. Urban plant species patterns are highly driven by density and function of built-up areas. Landsc. Ecol. 2007, 22, 1227–1239. [Google Scholar] [CrossRef]
- Wu, J.; Loucks, O.L. From Balance of Nature to Hierarchical Patch Dynamics: A Paradigm Shift in Ecology. Q. Rev. Biol. 1995, 70, 439–466. [Google Scholar] [CrossRef]
- Wu, J.; Jenerette, G.D.; David, J.L. Linking land-use change with ecosystem processes: A hierarchical patch dynamic model. In Integrated Land Use and Environmental Models; Guhathakurta, F.S., Ed.; Springer: Berlin/Heidelberg, Germany, 2003; pp. 99–119. [Google Scholar]
- Grove, J.M.; William, R.B., Jr. A social ecology approach and applications of urban ecosystem and landscape analyses: A case study of Baltimore, Maryland. Urban Ecosyst. 1997, 1, 259–275. [Google Scholar] [CrossRef]
- Grove, M.; Cadenasso, M.; Pickett, S.; Machlis, G.; Burch, W.R.; Burch, W.R. Expanding the Landscape: applying Patch Dynamics to Social-Ecological Systems. In The Baltimore School of Urban Ecology: Space, Scale, and Time for the Study of Cities; Yale University Press: New Haven, CT, USA, 2015; pp. 38–76. [Google Scholar]
- Alberti, M. Advances in Urban Ecology Integrating Humans and Ecological Processes in Urban Ecosystems; Springer: New York, NY, USA, 2008. [Google Scholar]
- Liu, J.; Dietz, T.; Carpenter, S.R.; Folke, C.; Alberti, M.; Redman, C.L.; Schneider, S.H.; Ostrom, E.; Pell, A.N.; Lubchenco, J.; et al. Coupled human and natural systems. AMBIO J. Hum. Environ. 2007, 36, 639–649. [Google Scholar] [CrossRef]
- Kremer, P.; Hamstead, Z.A.; McPhearson, T. A social–ecological assessment of vacant lots in New York City. Landsc. Urban Plan. 2013, 120, 218–233. [Google Scholar] [CrossRef]
- Faschan, A.; Hoffer, R.; Daly, L.; Parish, S.B. Hurricane Katrina Impacts on Wastewater Infrastructure—Lessons Learned. Proc. Water Environ. Fed. 2007, 2007, 2859–2873. [Google Scholar] [CrossRef]
- Garvin, E.; Branas, C.; Keddem, S.; Sellman, J.; Cannuscio, C. More Than Just An Eyesore: Local Insights And Solutions on Vacant Land And Urban Health. J. Urban Health Bull. N. Y. Acad. Med. 2013, 90, 412–426. [Google Scholar] [CrossRef] [PubMed]
- Escobedo, F.; Varela, S.; Zhao, M.; Wagner, J.E.; Zipperer, W. Analyzing the efficacy of subtropical urban forests in offsetting carbon emissions from cities. Environ. Sci. Policy 2010, 13, 362–372. [Google Scholar] [CrossRef]
- Douglas, I. Urban ecology and urban ecosystems: Understanding the links to human health and well-being. Curr. Opin. Environ. Sustain. 2012, 4, 385–392. [Google Scholar] [CrossRef]
- Lyytimäki, J.; Sipilä, M. Hopping on one leg—The challenge of ecosystem disservices for urban green management. Urban For. Urban Green. 2009, 8, 309–315. [Google Scholar] [CrossRef]
- Becker, B.; Leisnham, P.; LaDeau, S.; LaDeau, S. A Tale of Two City Blocks: Differences in Immature and Adult Mosquito Abundances between Socioeconomically Different Urban Blocks in Baltimore (Maryland, USA). Int. J. Environ. Res. Public. Health 2014, 11, 3256–3270. [Google Scholar] [CrossRef] [PubMed]
- LaDeau, S.L.; Allan, B.F.; Leisnham, P.T.; Levy, M.Z. The ecological foundations of transmission potential and vector-borne disease in urban landscapes. Funct. Ecol. 2015, 29, 889–901. [Google Scholar] [CrossRef] [PubMed]
- Mackenstedt, U.; Jenkins, D.; Romig, T. The role of wildlife in the transmission of parasitic zoonoses in peri-urban and urban areas. Int. J. Parasitol. Parasites Wildl. 2015, 4, 71–79. [Google Scholar] [CrossRef] [PubMed]
- Forman, R.T.T.; Godron, M. Patches and Structural Components for A Landscape Ecology. BioScience 1981, 31, 733–740. [Google Scholar]
- Grimm, N.B.; Grove, J.G.; Pickett, S.T.A.; Redman, C.L. Integrated Approaches to Long-Term Studies of Urban Ecological Systems. BioScience 2000, 50, 571–584. [Google Scholar] [CrossRef]
- Grove, J.M.; Locke, D.H.; O’Neil-Dunne, J.P. An ecology of prestige in New York City: Examining the relationships among population density, socio-economic status, group identity, and residential canopy cover. Environ. Manage. 2014, 54, 402–419. [Google Scholar] [CrossRef] [PubMed]
- Alberti, M. The Effects of Urban Patterns on Ecosystem Function. Int. Reg. Sci. Rev. 2005, 28, 168–192. [Google Scholar] [CrossRef]
- Haase, D.; Seppelt, R.; Haase, A. Land use impacts of demographic change—lessons from Eastern German urban regions. In Use of Landscape Sciences for the Assessment of Environmental Security; Petrosillo, I., Müller, F., Jones, K.B., Zurlini, G., Krauze, K., Victorov, S., Li, B.-L., Kepner, W.G., Eds.; Springer Netherlands: Dordrecht, The Netherlands, 2007; pp. 329–344. [Google Scholar]
- Burkman, C.E.; Gardiner, M.M. Spider assemblages within greenspaces of a deindustrialized urban landscape. Urban Ecosyst. 2014, 18, 793–818. [Google Scholar] [CrossRef]
- Steenberg, J.W.N.; Millward, A.A.; Duinker, P.N.; Nowak, D.J.; Robinson, P.J. Neighbourhood-scale urban forest ecosystem classification. J. Environ. Manag. 2015, 163, 134–145. [Google Scholar] [CrossRef] [PubMed]
- Seto, K.; Parnell, S.; Elmqvist, T. A Global Outlook on Urbanization. In Urbanization, Biodiversity and Ecosystem Services: Challenges and Opportunities: A Global Assessment; Springer: New York, NY, USA, 2013. [Google Scholar]
- Bruno, J.F.; Stachowicz, J.J.; Bertness, M.D. Inclusion of facilitation into ecological theory. Trends Ecol. Evol. 2003, 18, 119–125. [Google Scholar] [CrossRef]
- Webb, C.O.; Ackerly, D.D.; McPeek, M.A.; Donoghue, M.J. Phylogenies and Community Ecology. Annu. Rev. Ecol. Syst. 2002, 33, 475–505. [Google Scholar] [CrossRef]
- Freudenberg, N.; Glaea, S.; Vlahov, D. Cities and the Health of the PublicFreudenberg, N., Glaea, S., Vlahov, D., Eds.; 1st ed.; Vanderbilt University Press: Nashville, TN, USA, 2006. [Google Scholar]
- Swan, C.M.; Pickett, S.T.; Szlavecz, K.; Warren, P.; Willey, K.T. Biodiversity and Community Composition in Urban Ecosystems: Coupled Human, Spatial, and Metacommunity Processes. Available online: https://www.researchgate.net/profile/Christopher_Swan3/publication/267857001_Biodiversity_and_Community_Composition_in_Urban_Ecosystems_Coupled_Human_Spatial_and_Metacommunity_Processes/links/54ad6e5d0cf2213c5fe3faa5.pdf (accessed on 17 May 2016).
- Pickett, S.T.; Cadenasso, M.L.; Grove, J.M.; Groffman, P.M.; Band, L.E.; Boone, C.G.; Burch, W.R.; Grimmond, C.S.B.; Hom, J.; Jenkins, J.C. Beyond urban legends: An emerging framework of urban ecology, as illustrated by the Baltimore ecosystem study. BioScience 2008, 58, 139–150. [Google Scholar] [CrossRef]
- Nowak, D.J.; Dwyer, J.F. Understanding the benefits and costs of urban forest ecosystems. In Urban and Community Forestry in the Northeast; Kuser, J.E., Ed.; Springer Netherlands: Dordrecht, The Netherlands, 2007; pp. 25–46. [Google Scholar]
- Schwarz, K.; Fragkias, M.; Boone, C.G.; Zhou, W.; McHale, M.; Grove, J.M.; O’Neil-Dunne, J.; McFadden, J.P.; Buckley, G.L.; Childers, D.; et al. Trees grow on money: Urban tree canopy cover and environmental justice. PLoS ONE 2015, 10, e0122051. [Google Scholar] [CrossRef] [PubMed]
- Berland, A.; Schwarz, K.; Herrmann, D.L.; Hopton, M.E. How environmental justice patterns are shaped by place: Terrain and tree canopy in cincinnati, Ohio, USA. Cities Environ. CATE 2015, 8. Article 1. [Google Scholar]
- Ernstson, H. The social production of ecosystem services: A framework for studying environmental justice and ecological complexity in urbanized landscapes. Landsc. Urban Plan. 2013, 109, 7–17. [Google Scholar] [CrossRef]
- Heynen, N. Green Urban Political Ecologies: Toward a Better Understanding of Inner-City Environmental Change. Environ. Plan. A 2006, 38, 499–516. [Google Scholar] [CrossRef]
- Heynen, N.C. The scalar production of injustice within the urban forest. Antipode 2003, 35, 980–998. [Google Scholar] [CrossRef]
- Pham, T.-T.-H.; Apparicio, P.; Séguin, A.-M.; Landry, S.; Gagnon, M. Spatial distribution of vegetation in Montreal: An uneven distribution or environmental inequity? Landsc. Urban Plan. 2012, 107, 214–224. [Google Scholar] [CrossRef]
- Bigsby, K.M.; McHale, M.R.; Hess, G.R. Urban Morphology Drives the Homogenization of Tree Cover in Baltimore, MD, and Raleigh, NC. Ecosystems 2013, 17, 212–227. [Google Scholar] [CrossRef]
- Brazel, A.; Selover, N.; Vose, R.; Heisler, G. The tale of two climates-Baltimore and Phoenix urban LTER sites. Clim. Res. 2000, 15, 123–135. [Google Scholar] [CrossRef]
- Shuster, W.D.; Dadio, S.; Drohan, P.; Losco, R.; Shaffer, J. Residential demolition and its impact on vacant lot hydrology: Implications for the management of stormwater and sewer system overflows. Landsc. Urban Plan. 2014, 125, 48–56. [Google Scholar] [CrossRef]
- Faeth, S.H.; Bang, C. Saari Urban biodiversity: Patterns and mechanisms. Ann. N. Y. Acad. Sci. 2011, 1223, 69–81. [Google Scholar] [CrossRef] [PubMed]
- Pándi, I.; Penksza, K.; Botta-Dukát, Z.; Kröel-Dulay, G. People move but cultivated plants stay: Abandoned farmsteads support the persistence and spread of alien plants. Biodivers. Conserv. 2014, 23, 1289–1302. [Google Scholar] [CrossRef]
- Otero, I.; Marull, J.; Tello, E.; Diana, G.L.; Pons, M.; Coll, F.; Boada, M. Land abandonment, landscape, and biodiversity: Questioning the restorative character of the forest transition in the Mediterranean. Ecol. Soc. 2015, 20. [Google Scholar] [CrossRef]
- Speldewinde, P.C.; Slaney, D.; Weinstein, P. Is restoring an ecosystem good for your health? Sci. Total Environ. 2015, 502, 276–279. [Google Scholar] [CrossRef] [PubMed]
- Benayas, J.M.R.; Newton, A.C.; Diaz, A.; Bullock, J.M. Enhancement of Biodiversity and Ecosystem Services by Ecological Restoration: A Meta-Analysis. Science 2009, 325, 1121–1124. [Google Scholar] [CrossRef] [PubMed]
- Dewar, T.; Manning, J. Introduction: The City after Abandonment. In The City after Abandonment; University of Pennsylvania Press: Philadelphia, PA, USA, 2012; pp. 133–150. [Google Scholar]
- Haase, D. Shrinking Cities, Biodiversity and Ecosystem Services. In Urbanization, Biodiversity and Ecosystem Services: Challenges and Opportunities: A Global Assessment; Springer: New York, NY, USA, 2013. [Google Scholar]
- Foo, K.; Martin, D. The Production of Urban Vacant Land: Relational Placemaking in Boston, Massachusetts Neighborhoods. Cities 2013, 35, 156–163. [Google Scholar] [CrossRef]
- Brownlow, A. An archaeology of fear and environmental change in Philadelphia. Geoforum 2006, 37, 227–245. [Google Scholar] [CrossRef]
- Gottdenker, N.L. Effects of Anthropogenic Land use Change on the Ecology of the Chagas Disease Agent Trypanosoma Cruzi, University of Georgia, 2009. Available online: http://purl.galileo.usg.edu/uga_etd/gottdenker_nicole_l_200908_phd (accessed on 17 January 2016).
- Berry, B.J.L. Urbanization and Counterurbanization in the United States. Ann. Am. Acad. Pol. Soc. Sci. 1980, 451, 13–20. [Google Scholar] [CrossRef]
- Tauzer, E. The Phytogeography of Vacant Lots in Baltimore, MD, USA: A Test of Island Biogeography. Available online: http://www.caryinstitute.org/sites/default/files/public/reprints/Tauzer_2009_REU.pdf (accessed on 16 May 2016).
- Winter, M.; Schweiger, O.; Klotz, S.; Nentwig, W.; Andriopoulos, P.; Arianoutsou, M.; Basnou, C.; Delipetrou, P.; Didžiulis, V.; Hejda, M.; et al. Plant extinctions and introductions lead to phylogenetic and taxonomic homogenization of the European flora. Proc. Natl. Acad. Sci. USA 2009, 106, 21721–21725. [Google Scholar] [CrossRef] [PubMed]
- Alonso, D.; Etienne, R.; Mckane, A. The merits of neutral theory. Trends Ecol. Evol. 2006, 21, 451–457. [Google Scholar] [CrossRef] [PubMed]
- Bertness, M.D.; Leonard, G. The Role of Positive Interactions in Communities: Lessons from Intertidal Habitats. Ecology 1997, 78, 1976–1989. [Google Scholar] [CrossRef]
- Connor, E.F.; Simberloff, D. The Assembly of Species Communities: Chance or Competition? Ecology 1979, 60, 1132–1140. [Google Scholar] [CrossRef]
- Diamond, J.M. Assembly of species communities. In Ecology and Evolution of Communities; Cody, M., Diamond, J.M., Eds.; The belknap press of Harvard Univerisity: Cambridge, UK, 1975; pp. 342–444. [Google Scholar]
- Gotelli, N.J.; McCabe, D.J. Species Co-Occurrence: A Meta-Analysis of J.M. Diamond’s Assembly Rules Model. Ecology 2002, 83, 2091–2096. [Google Scholar] [CrossRef]
- Hubbell, S.P. The Unified Neutral Theory of Biodiversity and Biogeography; Princeton University Press: Princeton, NJ, USA, 2001. [Google Scholar]
- Weiher, E.; Keddy, P. Ecological Assembly Rules: Perspectives, Advances, Retreats; Cambridge University Press: Cambridge, UK, 1999. [Google Scholar]
- Freudenburg, W.R.; Frickel, S.; Gramling, R. Beyond the Nature/Society Divide: Learning to Think about a Mountain. Sociol. Forum 1995, 10, 361–392. [Google Scholar] [CrossRef]
- Grimm, N.B.; Redman, C.L. Approaches to the study of urban ecosystems: The case of Central Arizona—Phoenix. Urban Ecosyst. 2004, 7, 199–213. [Google Scholar] [CrossRef]
- Kowarik, I.; Körner, S.; NetLibrary, I. Wild Urban Woodlands New Perspectives for Urban Forestry; Springer: Berlin, Germany; New York, NY, USA, 2005. [Google Scholar]
- Burkman, C.E.; Gardiner, M.M. Urban greenspace composition and landscape context influence natural enemy community composition and function. Biol. Control 2014, 75, 58–67. [Google Scholar] [CrossRef]
- Hanks, L.; Denno, R.F. Natural Enemies and Plant Water Relations Influence the Distribution of An Armored Scale Insect. Ecology 1993, 74, 1081–1091. [Google Scholar] [CrossRef]
- Concepción, E.D.; OBrist, M.K. Impacts of urban sprawl on species richness of plants, butterflies, gastropods and birds: not only built-up area matters. Urban Ecosyst. 2016, 19, 225–242. [Google Scholar] [CrossRef]
- Paula, M.; Shrewsbury, M.J.R. Evaluation of Components of Vegetational Texture for Predicting Azalea Lace Bug, Stephanitis pyrioides (Heteroptera: Tingidae), Abundance in Managed Landscapes. Environ. Entomol. 2000, 29, 919–926. [Google Scholar]
- Burkman, C.E. The Influence of Habitat Management and Landscape on Spider Assemblages within Urban Greenspaces of Cleveland, Ohio. Master’s Thesis, The Ohio State University, Columbus, OH, USA, 2013. [Google Scholar]
- Caillouët, K.A.; Michaels, S.R.; Xiong, X.; Foppa, I.; Wesson, D.M. Increase in West Nile neuroinvasive disease after hurricane Katrina. Emerg. Infect. Dis. 2008, 14, 804. [Google Scholar] [CrossRef] [PubMed]
- Caillouët, K.A.; Carlson, J.C.; Wesson, D.; Jordan, F. Colonization of abandoned swimming pools by larval mosquitoes and their predators following Hurricane Katrina. J. Vector Ecol. 2008, 33, 166–172. [Google Scholar] [CrossRef]
- Himsworth, C.G.; Parsons, K.L.; Jardine, C.; Patrick, D.M. Rats, cities, people, and pathogens: A systematic review and narrative synthesis of literature regarding the ecology of rat-associated zoonoses in urban centers. Vector Borne Zoonotic Dis. 2013, 13, 349–359. [Google Scholar] [CrossRef] [PubMed]
- Keating, D. Redevelopment of Vacant Land in the Blighted Neighborhoods of Cleveland, Ohio, Resulting from the Housing Foreclosure Crisis. J. Regen. Renew. 2010, 4, 39–52. [Google Scholar]
- Margulis, H.L. Rat Fields, Neighborhood Sanitation, and Rat Complaints in Newark, New Jersey. Geogr. Rev. 1977, 67, 221. [Google Scholar] [CrossRef]
- Feng, A.Y.T.; Himsworth, C.G. The secret life of the city rat: a review of the ecology of urban Norway and black rats (Rattus norvegicus and Rattus rattus). Urban Ecosyst. 2013, 17, 149–162. [Google Scholar] [CrossRef]
- Wilcox, B.A.; Gubler, D.J. Disease ecology and the global emergence of zoonotic pathogens. Environ. Health Prev. Med. 2005, 10, 263–272. [Google Scholar] [CrossRef] [PubMed]
- Kilpatrick, A.M.; Randolph, S.E. Drivers, dynamics, and control of emerging vector-borne zoonotic diseases. Lancet 2012, 380, 1946–1955. [Google Scholar] [CrossRef]
- Dobson, A. Population dynamics of pathogens with multiple host species. Am. Nat. 2004, 164, S64–S78. [Google Scholar] [CrossRef] [PubMed]
- Daszak, P.; Cunningham, A.A.; Hyatt, A.D. Anthropogenic environmental change and the emergence of infectious diseases in wildlife. Acta Trop. 2001, 78, 103–116. [Google Scholar] [CrossRef]
- Engering, A.; Hogerwerf, L.; Slingenbergh, J. Pathogen–host–environment interplay and disease emergence. Emerg. Microbes Infect. 2013, 2, e5. [Google Scholar] [CrossRef] [PubMed]
- Brearley, G.; Rhodes, J.; Bradley, A.; Baxter, G.; Seabrook, L.; Lunney, D.; Liu, Y.; McAlpine, C. Wildlife disease prevalence in human-modified landscapes. Biol. Rev. Camb. Philos. Soc. 2013, 88, 427–442. [Google Scholar] [CrossRef] [PubMed]
- Finucane, M.L.; Fox, J.; Saksena, S.; Spencer, J.H. A Conceptual Framework for Analyzing Social-Ecological Models of Emerging Infectious Diseases. In Understanding Society and Natural Resources; Manfredo, M.J., Vaske, J.J., Rechkemmer, A., Duke, E.A., Eds.; Springer Netherlands: Dordrecht, The Netherlands, 2014; pp. 93–109. [Google Scholar]
- Patz, J.A.; Daszak, P.; Tabor, G.M.; Aguirre, A.A.; Pearl, M.; Epstein, J.; Wolfe, N.D.; Kilpatrick, A.M.; Foufopoulos, J.; Molyneux, D.; et al. Unhealthy Landscapes: Policy Recommendations on Land Use Change and Infectious Disease Emergence. Environ. Health Perspect. 2004, 112, 1092–1098. [Google Scholar] [CrossRef] [PubMed]
- Gottdenker, N.L.; Streicker, D.G. Anthropogenic land use change and infectious diseases: A review of the evidence. EcoHealth 2014, 11, 619–632. [Google Scholar] [CrossRef] [PubMed]
- Bradley, C.A.; Altizer, S. Urbanization and the ecology of wildlife diseases. Trends Ecol. Evol. 2007, 22, 95–102. [Google Scholar] [CrossRef] [PubMed]
- Randolph, S.E.; Dobson, A.D.M. Pangloss revisited: A critique of the dilution effect and the biodiversity-buffers-disease paradigm. Parasitology 2012, 139, 847–863. [Google Scholar] [CrossRef] [PubMed]
- Saksena, S.; Fox, J.; Epprecht, M.; Tran, C.C.; Castrence, M.; Nong, D.; Spencer, J.; Nguyen, L.; Finucane, M.; Vien, T.D.; et al. Role of Urbanization, Land-Use Diversity, and Livestock Intensification in Zoonotic Emerging Infectious Diseases; East-West Center: Honolulu, HI, USA, 2014. [Google Scholar]
- McGee, T. The emergence of desakota regions in Asia: Expanding a hypothesis. In The Extended Metropolis: Settlement Transition in Asia; University of Hawaii Press: Honolulu, HI, USA, 1991; pp. 3–25. [Google Scholar]
- Alvar, J.; Yactayo, S.; Bern, C. Leishmaniasis and poverty. Trends Parasitol. 2006, 22, 552–557. [Google Scholar] [CrossRef] [PubMed]
- Chaves, L.F.; Cohen, J.M.; Pascual, M.; Wilson, M.L. Social Exclusion Modifies Climate and Deforestation Impacts on a Vector-Borne Disease. PLoS Negl. Trop. Dis. 2008, 2, e176. [Google Scholar] [CrossRef] [PubMed]
- Costa, C.H.N.; Werneck, G.L.; Rodrigues, L.; Santos, M.V.; Araújo, I.B.; Moura, L.S.; Moreira, S.; Gomes, R.B.B.; Lima, S.S. Household structure and urban services: Neglected targets in the control of visceral leishmaniasis. Ann. Trop. Med. Parasitol. 2005, 99, 229–236. [Google Scholar] [CrossRef] [PubMed]
- Beers, S.A.; Novosil’ts ev, G.I.; Mel’nikova, L.I. The role of the water factor in the dissemination of Toxocara eggs and the spread of toxocariasis in a megalopolis. Parazitologiia 1998, 33, 129–135. [Google Scholar]
- Paquet-Durand, I.; Hernández, J.; Dolz, G.; Zuñiga, J.J.R.; Schnieder, T.; Epe, C. Prevalence of Toxocara spp., Toxascaris leonina and ancylostomidae in public parks and beaches in different climate zones of Costa Rica. Acta Trop. 2007, 104, 30–37. [Google Scholar] [CrossRef] [PubMed]
- Walsh, M.G. Forest Fragmentation and Risk of Giardiasis in New York State. EcoHealth 2013, 10, 405–414. [Google Scholar] [CrossRef] [PubMed]
- Giardiasis Surveillance—United States, 2009–2010. Available online: http://www.cdc.gov/mmwr/preview/mmwrhtml/ss6105a2.htm (accessed on 1 April 2016).
- Allan, B.F.; Langerhans, R.B.; Ryberg, W.A.; Landesman, W.J.; Griffin, N.W.; Katz, R.S.; Oberle, B.J.; Schutzenhofer, M.R.; Smyth, K.N.; de St. Maurice, A.; et al. Ecological correlates of risk and incidence of West Nile virus in the United States. Oecologia 2009, 158, 699–708. [Google Scholar] [CrossRef] [PubMed]
- Bradley, C.A.; Gibbs, S.E.J.; Altizer, S. Urban land use predicts West Nile virus exposure in songbirds. Ecol. Appl. 2008, 18, 1083–1092. [Google Scholar] [CrossRef] [PubMed]
- Johnson, B.J.; Munafo, K.; Shappell, L.; Tsipoura, N.; Robson, M.; Ehrenfeld, J.; Sukhdeo, M.V. The roles of mosquito and bird communities on the prevalence of West Nile virus in urban wetland and residential habitats. Urban Ecosyst. 2012, 15, 513–531. [Google Scholar] [CrossRef] [PubMed]
- Reisen, W.K.; Takahashi, R.M.; Carroll, B.D.; Quiring, R. Delinquent mortgages, neglected swimming pools, and West Nile virus, California. Emerg. Infect. Dis. 2008, 14, 1747–1749. [Google Scholar] [CrossRef] [PubMed]
- LaDeau, S.L.; Leisnham, P.T.; Biehler, D.; Bodner, D. Higher mosquito production in low-income neighborhoods of Baltimore and Washington, DC: Understanding ecological drivers and mosquito-borne disease risk in temperate cities. Int. J. Environ. Res. Public. Health 2013, 10, 1505–1526. [Google Scholar] [CrossRef] [PubMed]
- Weterings, R.; Umponstira, C.; Buckley, H.L. Container-breeding mosquitoes and predator community dynamics along an urban-forest gradient: The effects of habitat type and isolation. Basic Appl. Ecol. 2014, 15, 486–495. [Google Scholar] [CrossRef]
- Diaz, J.H. Chagas disease in the United States: A cause for concern in Louisiana? J. La. State Med. Soc. 2007, 159, 21–23, 25–29. [Google Scholar] [PubMed]
- Ramsey, J.M.; Alvear, A.L.; Ordoñez, R.; Muñoz, G.; Garcia, A.; Lopez, R.; Leyva, R. Risk factors associated with house infestation by the Chagas disease vector Triatoma pallidipennis in Cuernavaca metropolitan area, Mexico. Med. Vet. Entomol. 2005, 19, 219–228. [Google Scholar] [CrossRef] [PubMed]
- Mizgajska, H. Eggs of Toxocara spp. in the environment and their public health implications. J. Helminthol. 2001, 75, 147–151. [Google Scholar] [PubMed]
- Despommier, D. Toxocariasis: Clinical Aspects, Epidemiology, Medical Ecology, and Molecular Aspects. Clin. Microbiol. Rev. 2003, 16, 265–272. [Google Scholar] [CrossRef] [PubMed]
- Macpherson, C.N.L. The epidemiology and public health importance of toxocariasis: A zoonosis of global importance. Int. J. Parasitol. 2013, 43, 999–1008. [Google Scholar] [CrossRef] [PubMed]
- Hotez, P.J. Neglected Infections of Poverty in the United States of America. PLoS Negl. Trop. Dis. 2008, 2, e256. [Google Scholar] [CrossRef] [PubMed]
- Gibbs, S.E.J.; Wimberly, M.C.; Madden, M.; Masour, J.; Yabsley, M.J.; Stallknecht, D.E. Factors affecting the geographic distribution of West Nile virus in Georgia, USA: 2002–2004. Vector Borne Zoonotic Dis. 2006, 6, 73–82. [Google Scholar] [CrossRef] [PubMed]
- Gage, K.L.; Dennis, D.T.; Orloski, K.A.; Ettestad, P.; Brown, T.L.; Reynolds, P.J.; Pape, W.J.; Fritz, C.L.; Carter, L.G.; Stein, J.D. Cases of cat-associated human plague in the Western US, 1977–1998. Clin. Infect. Dis. 2000, 30, 893–900. [Google Scholar] [CrossRef] [PubMed]
- Stenseth, N.C.; Atshabar, B.B.; Begon, M.; Belmain, S.R.; Bertherat, E.; Carniel, E.; Gage, K.L.; Leirs, H.; Rahalison, L. Plague: Past, present, and future. PLoS Med. 2008, 5, e3. [Google Scholar] [CrossRef] [PubMed]
- Jones, M.E.; Curns, A.T.; Krebs, J.W.; Childs, J.E. Environmental and human demographic features associated with epizootic raccoon rabies in Maryland, Pennsylvania, and Virginia. J. Wildl. Dis. 2003, 39, 869–874. [Google Scholar] [CrossRef] [PubMed]
- Duke, J.E. Land Use and Urbanization Patterns in An Established Enzootic Raccoon Rabies Area; Auburn University: Atlanta, GA, USA, 2012. [Google Scholar]
- Duke, J.E.; Blanton, J.D.; Ivey, M.; Rupprecht, C. Modeling enzootic raccoon rabies from land use patterns—Georgia (USA) 2006–2010. F1000Research 2014, 2. [Google Scholar] [CrossRef] [PubMed]
- Levin, S.A. Ecosystems and the biosphere as complex adaptive systems. Ecosystems 1998, 1, 431–436. [Google Scholar] [CrossRef]
- Tilman, D.; Knops, J.; Wedin, D.; Reich, P.; Ritchie, M.; Siemann, E. The influence of functional diversity and composition on ecosystem processes. Science 1997, 277, 1300–1302. [Google Scholar] [CrossRef]
- Gunderson, L. Ecology and society: Ecological and human community resilience in response to natural disasters. Ecol. Soc. 2010, 15. Article 18. [Google Scholar]
- Folke, C. Resilience: The emergence of a perspective for social–ecological systems analyses. Glob. Environ. Chang. 2006, 16, 253–267. [Google Scholar] [CrossRef]
- Adger, W.N. Social and ecological resilience: Are they related? Prog. Hum. Geogr. 2000, 24, 347–364. [Google Scholar] [CrossRef]
- Holling, C.S. Understanding the complexity of economic, ecological, and social systems. Ecosystems 2001, 4, 390–405. [Google Scholar] [CrossRef]
- Holling, C.S. Resilience and Stability of Ecological Systems. Annu. Rev. Ecol. Syst. 1973, 4, 1–23. [Google Scholar] [CrossRef]
- Pickett, S.T.A.; Cadenasso, M.L.; Grove, J.M. Resilient cities: Meaning, models, and metaphor for integrating the ecological, socio-economic, and planning realms. Landsc. Urban Plan. 2004, 69, 369–384. [Google Scholar] [CrossRef]
- Redman, C.L. Resilience theory in archaeology. Am. Anthropol. 2005, 107, 70–77. [Google Scholar] [CrossRef]
- Pickett, S.T.A.; Cadenasso, M.L.; Grove, J.M.; Nilon, C.H.; Pouyat, R.V.; Zipperer, W.C.; Costanza, R. Urban ecological systems: Linking terrestrial ecological, physical, and socioeconomic components of metropolitan areas. Annu. Rev. Ecol. Syst. 2001, 32, 127–157. [Google Scholar] [CrossRef]
- Putnam, R.D. Bowling Alone: The Collapse and Revival of American Community; Simon and Schuster: New York, NY, USA, 2001. [Google Scholar]
- Putnam, R.D. E pluribus unum: Diversity and community in the twenty-first century the 2006 Johan Skytte Prize Lecture. Scand. Polit. Stud. 2007, 30, 137–174. [Google Scholar] [CrossRef]
- Leitki, N. Does Diversity Erode Social Cohesion? Social Capital and Race in British Neighbourhoods. Polit. Stud. 2008, 56, 99–126. [Google Scholar] [CrossRef]
- Stolle, D.; Soroka, S.; Johnston, R. When Does Diversity Erode Trust? Neighborhood Diversity, Interpersonal Trust and the Mediating Effect of Social Interactions. Polit. Stud. 2008, 56, 57–75. [Google Scholar] [CrossRef]
- Walsh, M.G. Rat sightings in New York City are associated with neighborhood sociodemographics, housing characteristics, and proximity to open public space. PeerJ 2014, 2, e533. [Google Scholar] [CrossRef] [PubMed]
- Begon, M. Effects of Host Diversity on Disease Dynamics. In Infectious Disease Ecology: Effects of Ecosystems on Disease and of Disease on Ecosystems; Princeton University Press: Princeton, NJ, USA, 2010. [Google Scholar]
- Roche, B.; Dobson, A.P.; Guegan, J.-F.; Rohani, P. Linking community and disease ecology: The impact of biodiversity on pathogen transmission. Philos. Trans. R. Soc. B 2012, 367, 2807–2813. [Google Scholar] [CrossRef] [PubMed]
- Roche, B.; Guégan, J.-F. Ecosystem dynamics, biological diversity and emerging infectious diseases. C. R. Biol. 2011, 334, 385–392. [Google Scholar] [CrossRef] [PubMed]
- Keesing, F.; Holt, R.D.; Ostfeld, R.S. Effects of species diversity on disease risk: Effects of species diversity on disease risk. Ecol. Lett. 2006, 9, 485–498. [Google Scholar] [CrossRef] [PubMed]
- Wood, C.L.; Lafferty, K.D.; DeLeo, G.; Young, H.S.; Hudson, P.J.; Kuris, A.M. Does biodiversity protect humans against infectious disease? Ecology 2014, 95, 817–832. [Google Scholar] [CrossRef] [PubMed]
- Blum, M.J. Diversity and Disease in a Post-Trauma Urban Landscape; Tulane University: New Orleans, LA, USA, 2014. [Google Scholar]
- Plyusnina, A.; Krajinović, L.C.; Margaletić, J.; Niemimaa, J.; Nemirov, K.; Lundkvist, Å.; Markotić, A.; Miletić-Medved, M.; Avšič-Županc, T.; Henttonen, H.; et al. Genetic evidence for the presence of two distinct hantaviruses associated with Apodemus mice in Croatia and analysis of local strains. J. Med. Virol. 2011, 83, 108–114. [Google Scholar] [CrossRef] [PubMed]
- Tadin, A.; Turk, N.; Korva, M.; Margaletić, J.; Beck, R.; Vucelja, M.; Habuš, J.; Svoboda, P.; Županc, T.A.; Henttonen, H.; et al. Multiple Co-infections of Rodents with Hantaviruses, Leptospira, and Babesia in Croatia. Vector Borne Zoonotic Dis. 2012, 12, 388–392. [Google Scholar] [CrossRef] [PubMed]
- Riley, S.; Serieys, L.E.K.; Moriarty, J. Infectious disease and contaminats in urban wildlife: Unseen and often overlooked threats. In Urban Wildlife Conservation: Theory and Practice; McCleery, R.A., Moorman, C.E., Peterson, M.N., Eds.; Springer US: Boston, MA, USA, 2014. [Google Scholar]
- Linard, C.; Lamarque, P.; Heyman, P.; Ducoffre, G.; Luyasu, V.; Tersago, K.; Vanwambeke, S.O.; Lambin, E.F. Determinants of the geographic distribution of Puumala virus and Lyme borreliosis infections in Belgium. Int. J. Health Geogr. 2007, 6, 15. [Google Scholar] [CrossRef] [PubMed]
- Becker, D.J.; Streicker, D.G.; Altizer, S. Linking anthropogenic resources to wildlife-pathogen dynamics: A review and meta-analysis. Ecol. Lett. 2015, 18, 483–495. [Google Scholar] [CrossRef] [PubMed]
- Wright, A.N.; Gompper, M.E. Altered parasite assemblages in raccoons in response to manipulated resource availability. Oecologia 2005, 144, 148–156. [Google Scholar] [CrossRef] [PubMed]
- Haque, R. Human Intestinal Parasites. J. Health Popul. Nutr. 2007, 25, 387–391. [Google Scholar] [PubMed]
- Ogrzewalska, M.; Uezu, A.; Jenkins, C.N.; Labruna, M.B. Effect of Forest Fragmentation on Tick Infestations of Birds and Tick Infection Rates by Rickettsia in the Atlantic Forest of Brazil. EcoHealth 2011, 8, 320–331. [Google Scholar] [CrossRef] [PubMed]
- Choffnes, E.R.; Rapporteurs, A.M. Global Issues in Water, Sanitation, and Health: Workshop Summary; National Academies Press: Washington, DC, USA, 2009. [Google Scholar]
- Rainey, R. Boil Water Advisories May be Common in New Orleans, but the Risk is Real, Experts Say. Available online: http://www.nola.com/politics/index.ssf/2013/03/boil_water_advisories_may_be_c.html (accessed on 16 May 2016).
- Lin, J.C.F.; Rutter, J.; Park, H. Events That Led to Flint’s Water Crisis. Available online: http://www.nytimes.com/interactive/2016/01/21/us/flint-lead-water-timeline.html?_r=0 (accessed on 16 May 2016).
- Smith, M.; Goodnough, A. Michigan Officials Defend Handling of Legionnaires’ Disease Outbreak in Flint. N. Y. Times. 2016. Available online: http://www.nytimes.com/2016/02/10/us/michigan-officials-defend-handling-of-legionnaires-disease-outbreak-in-flint.html?_r=0 (accessed on 16 May 2016).
- Michigan Department of Health and Human Services. Summary of Legionellosis Outbreak-Genesee County, June 2014–March 2015. Available online: http://www.michigan.gov/documents/mdhhs/Genesee_Co_Legionnaires_Disease_Investigation_Ex_Summary_FINAL_510722_7.pdf (accessed on 16 May 2016).
- Beer, K.D.; Gargano, J.W.; Roberts, V.A.; Hill, V.R.; Garrison, L.E.; Kutty, P.; Hilborn, E.D.; Wade, T.J.; Fullerton, K.E.; Yoder, J.S. Surveillance for waterborne disease outbreaks associated with drinking water—United States, 2011–2012. Morb. Mortal. Wkly. Rep. 2015, 64, 842–848. [Google Scholar] [CrossRef]
- The Brookings Institute. New Orleans after the Storm: Lessons from the Past, a Plan for the Future; The Brookings Institute: Washington, DC, USA, 2005. [Google Scholar]
- Ehrehfeucht, R.; Nelson, M. Recovery in a shrinking city: Challenges to rightsizing post- Katrina New Orleans. In The City After Abandonment; City in the twenty-first century book series; University of Pennsylvania Press: Philadelphia, PA, USA, 2012. [Google Scholar]
- Gotham, K.F.; Blum, M.; Campanella, R. Toward a new normal: Trauma, diversity, and the New Orleans urban long-term research area exploratory (ULTRA-Ex) project. Cities Environ. CATE 2014, 7, 4. [Google Scholar]
- City Business Staff Reports, St. Bernard Prez: Smaller Population Straining Parish’s Budget; New Orleans CityBusiness: New Orleans, LA, USA, 2010.
- Alexander-Bloch, B. St. Bernard Water System Tests Positive for Rare Brain-Eating Amoeba, CDC Confirms; Times-Picayune New Orleans: New Orleans, LA, USA, 2013. [Google Scholar]
- Yoder, J.S.; Straif-Bourgeois, S.; Roy, S.L.; Moore, T.A.; Visvesvara, G.S.; Ratard, R.C.; Hill, V.R.; Wilson, J.D.; Linscott, A.J.; Crager, R.; et al. Primary amebic meningoencephalitis deaths associated with sinus irrigation using contaminated tap water. Clin. Infect. Dis. 2012, 55, 79–85. [Google Scholar] [CrossRef] [PubMed]
- Wilcox, B.A.; Colwell, R.R. Emerging and Reemerging Infectious Diseases: Biocomplexity as an Interdisciplinary Paradigm. EcoHealth 2005, 2, 244–257. [Google Scholar] [CrossRef]
- Waltner-Toews, D. An ecosystem approach to health and its applications to tropical and emerging diseases. Cad. Saúde Pública 2001, 17 Suppl., 7–22. [Google Scholar] [CrossRef] [PubMed]
- Gallopín, G.C. Linkages between vulnerability, resilience, and adaptive capacity. Glob. Environ. Chang. 2006, 16, 293–303. [Google Scholar] [CrossRef]
- Gotham, K.F.; Campanella, R. Coupled vulnerability and resilience: The dynamics of cross-scale interactions in post-Katrina New Orleans. Ecol. Soc. 2011, 16. [Google Scholar] [CrossRef]
- Meerow, S.; Newell, J.P.; Stults, M. Defining urban resilience: A review. Landsc. Urban Plan. 2016, 147, 38–49. [Google Scholar] [CrossRef]
- Holling, C.S. Surprise for Science, Resilience for Ecosystems, and Incentives for People. Ecol. Appl. 1996, 6, 733–735. [Google Scholar] [CrossRef]
- Beisner, D.T.H. Alternative Stable States in Ecology. Front. Ecol. Environ. 2003, 1, 376–382. [Google Scholar] [CrossRef]
- Environemental Justice. Available online: https://www.epa.gov/sites/production/files/widgets/ejscreenwidget.html (accessed on 4 April 2016).
- Sanders, R. Some determinants of urban forest structure. Elsevier 1984, 8, 13–27. [Google Scholar] [CrossRef]
- Pataki, D.E.; Carreiro, M.M.; Cherrier, J.; Grulke, N.E.; Jennings, V.; Pincetl, S.; Pouyat, R.V.; Whitlow, T.H.; Zipperer, W.C. Coupling biogeochemical cycles in urban environments: Ecosystem services, green solutions, and misconceptions. Front. Ecol. Environ. 2011, 9, 27–36. [Google Scholar] [CrossRef]
- Blanco, H.; Alberti, M.; Olshansky, R.; Chang, S.; Wheeler, S.M.; Randolph, J.; London, J.B.; Hollander, J.B.; Pallagst, K.M.; Schwarz, T.; et al. Shaken, shrinking, hot, impoverished and informal: Emerging research agendas in planning. Prog. Plan. 2009, 72, 195–250. [Google Scholar] [CrossRef]
- Campanella, R. The Great Footprint Debate, Updated; Times-Picayune New Orleans: New Orleans, LA, USA, 2015. [Google Scholar]

| Disease | Pathogen and Type | Transmission | Main Host | Mechanism | Outcome | Study | Location |
|---|---|---|---|---|---|---|---|
| Socioeconomic Transition | |||||||
| American Cutaneous Leishmaniasis | Protozoa: Leishmania spp. | Vector; sandflies | Wild and domestic mammals, humans | Poor housing conditions and environmental sanitation, lack of personal protective measures, disruption of services like trash collection and sewage system, and human migration | Increased contact between hosts and infected sand flies. | [103,104,105] | North and South America |
| Toxocariasis | Ascarid nematodes: Toxocara canis and Toxocara cati | Indirect by contact with soil or water contaminated with infected eggs | Dogs, cats, raccoons. | Greater soil humidity, greater feral animal movement. | Faster incubation, increased human-pathogen contact, especially in neighborhoods with poor irrigation systems and that are prone to flooding events. | [106,107] | Various |
| Habitat Expansion | |||||||
| Giardiasis | Protazoa: Giardia lamblia | Indirect; waterborne | Humans, mammals | Increased heterogeneity in environment, edge effect between vegetative habitat and managed landscape, more standing water | Increased human-pathogen contact | [108,109] | New York (US) |
| West Nile Neuroinvasive Disease, Dengue Fever and Chikungunya | West Nile Virus, Dengue Virus and Chikungunya Virus | Vector; mosquito (Culex spp., Anopheles spp.) | Mammals, birds | Increased breeding habitat through standing water in abandoned tires, pools of water associated with land use change and demolition, abandoned swimming pools | Increased abundance of mosquito vectors | [85,110,111,112,113,114,115] | North America |
| Chagas Disease | Parasite: Trypanosoma cruzi | Vector; triatomine bug | Wild and domestic mammals, humans | Forest fragmentation, an increase in edge habitat, and built infrastructure decay | Increased vector abundance and prevalence of parasite and association between competent host species diversity and infection prevalence. | [65,116,117] | South America, Louisiana (US) |
| Contact Rate | |||||||
| Toxocariasis | Ascarid nematodes: Toxocara canis and Toxocara cati | Direct, indirect; sometimes vector (flies) | Domestic cats, dogs, foxes, raccoons | Playgrounds and sandboxes in low income neighborhoods with more feral dog and cat populations | Increased human-pathogen contact, especially children | [118,119,120,121] | North America |
| West Nile Neuroinvasive Disease | Virus: flavivirus | Vector-mosquito (Culex spp.) | Passerine birds, mammals | Anthropogenic land-use, temperature, and housing density favoring bird host density increases | Expansion of pathogen and spillover into human population. | [122] | Georgia (US) |
| Leptospirosis | Bacteria: Leptiospira | Direct; water, food or soil contaminated with infected animal urine | Domestic and feral mammals, primarily rodents. | Household environmental factors such as residence in flood-risk regions with open sewers, proximity to accumulated refuse, and presence of chickens and rats. | Increased prevalence in neighborhoods especially those with low socioeconomic status, increased refuse, standing water | [23,24] | Salvador, Brazil |
| Connectivity and Species Movement | |||||||
| Plague | Bacteria: Yersina pestis | Direct; vector; flea | Rodents including prairie dogs, peri-domestic animals | Urban infringement of vegetated and forested environments and associated increases in connectivity. | Habitat expansion of pathogen via species movement, increased contact rate where rodents more readily interact with peri-domestic animals | [123,124] | US, Tanzania |
| Rabies | Virus: Rhabdoviridae | Direct; saliva | Mammals, raccoons, dogs, bats, etc. | Heterogeneous land use, increases in green space mixed with resource clumping, and low human population density | Increased contact between humans and infected hosts | [125,126,127] | North America |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gulachenski, A.; Ghersi, B.M.; Lesen, A.E.; Blum, M.J. Abandonment, Ecological Assembly and Public Health Risks in Counter-Urbanizing Cities. Sustainability 2016, 8, 491. https://doi.org/10.3390/su8050491
Gulachenski A, Ghersi BM, Lesen AE, Blum MJ. Abandonment, Ecological Assembly and Public Health Risks in Counter-Urbanizing Cities. Sustainability. 2016; 8(5):491. https://doi.org/10.3390/su8050491
Chicago/Turabian StyleGulachenski, Alexandra, Bruno M. Ghersi, Amy E. Lesen, and Michael J. Blum. 2016. "Abandonment, Ecological Assembly and Public Health Risks in Counter-Urbanizing Cities" Sustainability 8, no. 5: 491. https://doi.org/10.3390/su8050491
APA StyleGulachenski, A., Ghersi, B. M., Lesen, A. E., & Blum, M. J. (2016). Abandonment, Ecological Assembly and Public Health Risks in Counter-Urbanizing Cities. Sustainability, 8(5), 491. https://doi.org/10.3390/su8050491







