Development of a Biochar-Plant-Extract-Based Nitrification Inhibitor and Its Application in Field Conditions
Abstract
:1. Introduction
2. Materials and Methods
2.1. Selection and Handling of Plant Material and Soil
2.2. Biochar Production
Formation of Biochar-Plant Extract Complexes
| Identification | Description |
|---|---|
| BC * | Biochar |
| BCPi * | Biochar with ethanol extract from bark of pine |
| BCPe * | Biochar with water extract from bark of peumo |
| BCEu * | Biochar with water extract from bark of eucalyptus |
| Pine | Ethanol extract from pine |
| Peumo | Water extract from peumo |
| Eucalyptus | Water extract from eucalyptus |
2.3. Chemical Characterization of Biochar
2.4. Phytotoxicity Assay
2.5. Evaluation of Net N Mineralization in a Field Experiment
3. Results
3.1. Effect of Plant Extracts Adsorbed to Biochar on Nitrification
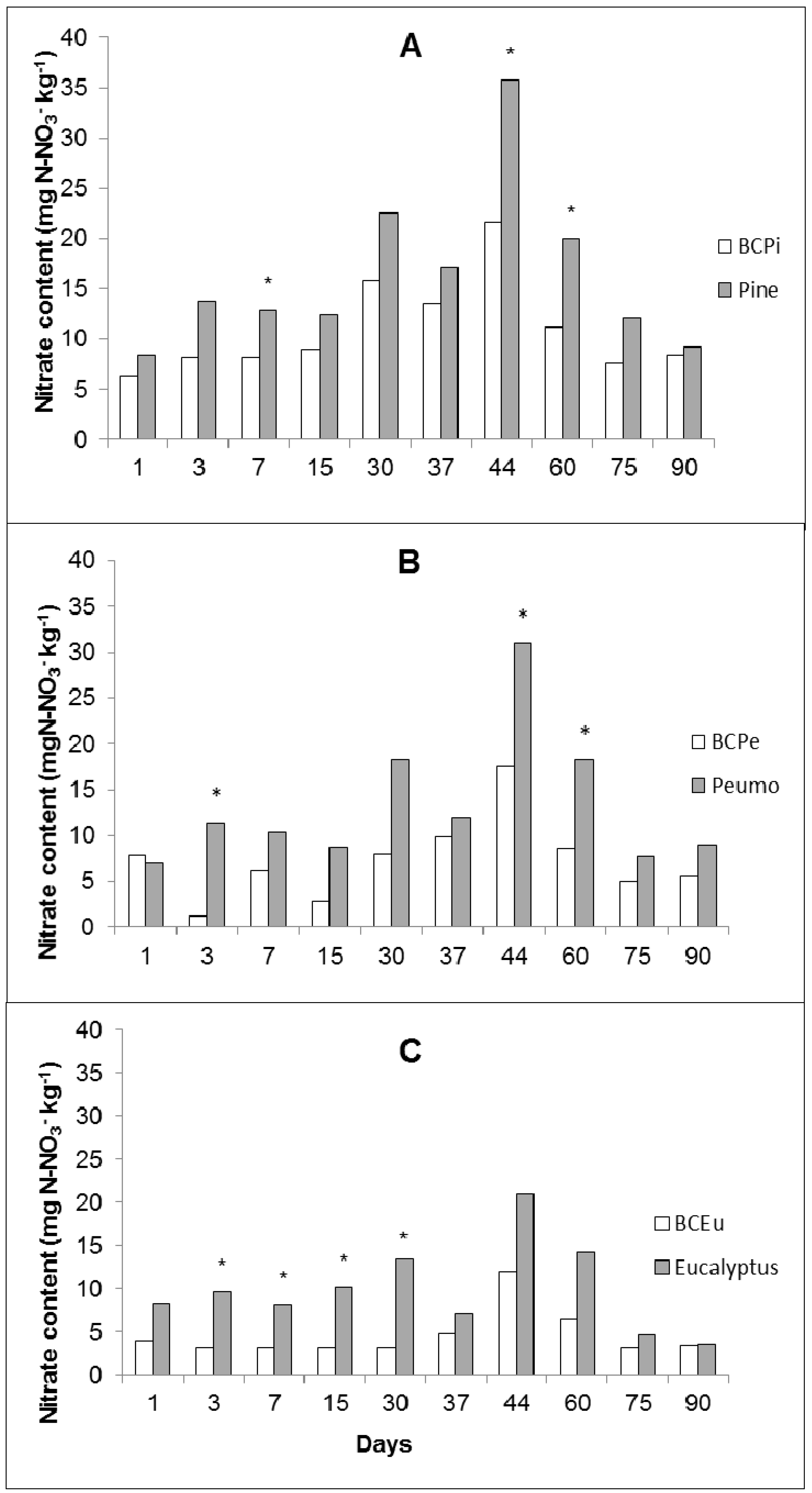
3.2. Chemical Characteristics of Biochar
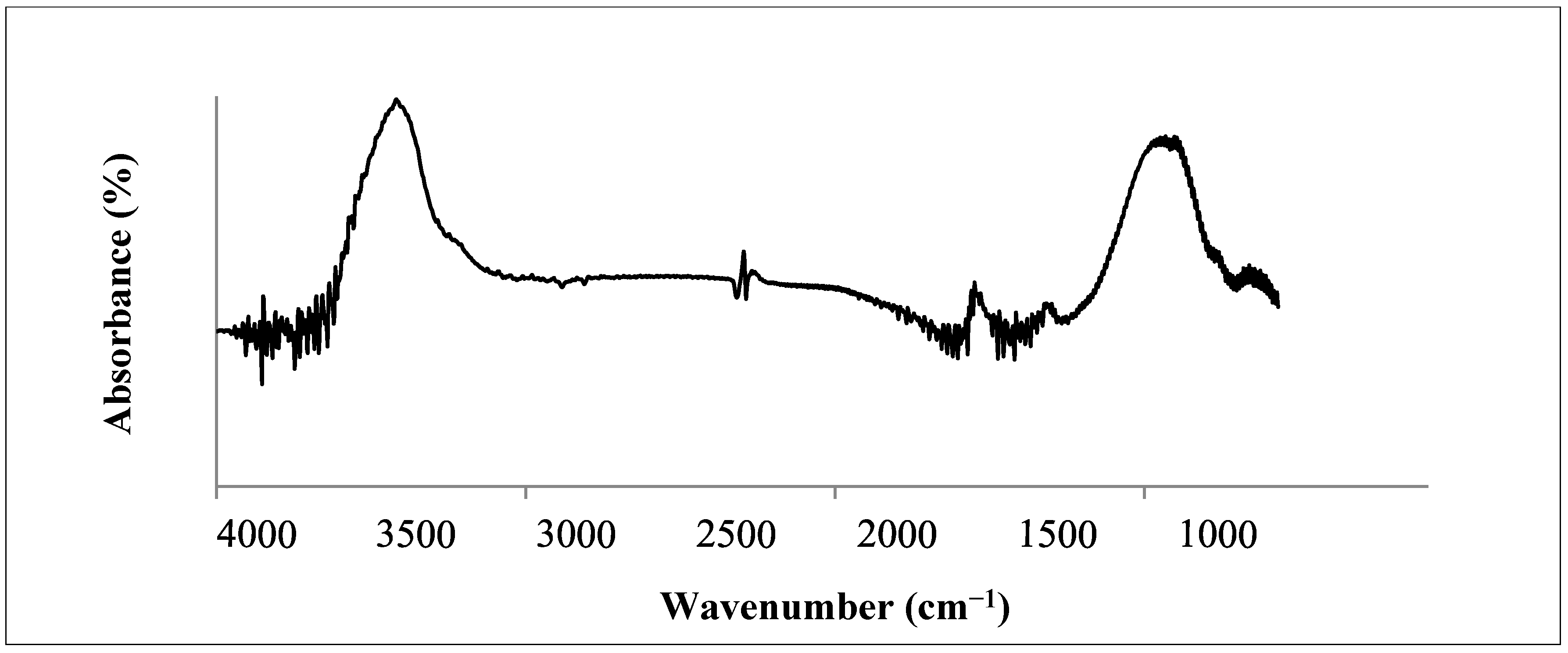
3.3. Phytotoxicity Assay
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Robertson, G.P.; Vitousek, P.M. Nitrogen in agriculture: Balancing the cost of an essential resource. Annu. Rev. Environ. Resour. 2009, 34, 97–125. [Google Scholar] [CrossRef]
- Subbarao, G.V.; Sahrawat, K.L.; Nakahara, K.; Ishikawa, T.; Kishii, M.; Rao, I.; Hash, C.T.; George, T.S.; Srinivasa Rao, P.; Nardi, P.; et al. Biological Nitrification Inhibition—A Novel Strategy to Regulate Nitrification in Agricultural Systems. Adv. Agron. 2012, 114, 249–301. [Google Scholar]
- Tilman, D.; Cassman, K.G.; Matson, P.A.; Naylor, R.; Polasky, S. Agricultural sustainability and intensive production practices. Nature 2002, 418, 671–677. [Google Scholar] [CrossRef] [PubMed]
- Subbarao, G.V; Ito, O.; Sahrawat, K.L; Berry, W.L; Nakahara, K.; Ishikawa, T.; Watanabe, T.; Suenaga, K.; Rondon, M.; Rao, I.M. Scope and strategies for regulation of nitrification in agricultural systems—Challenges and opportunities. Crit. Rev. Plant Sci. 2006, 25, 303–335. [Google Scholar] [CrossRef]
- Hofstra, N.; Bouwman, A.F. Denitrification in agricultural soils: Summarizing published data and estimating global annual rates. Nutr. Cycl. Agroecosyst. 2005, 72, 267–278. [Google Scholar] [CrossRef]
- Burney, J.A.; Davis, S.J.; Lobell, D.B. Greenhouse gas mitigation by agricultural intensification. Proc. Natl. Acad. Sci. USA 2010, 107, 12052–12057. [Google Scholar] [CrossRef] [PubMed]
- Galloway, J.N.; Townsend, A.R.; Erisman, J.W.; Bekunda, M.; Cai, Z.; Freney, J.R.; Martinelli, L.A.; Seitzinger, S.P.; Sutton, M.A. Transformation of the nitrogen cycle: Recent trends, questions, and potential solutions. Science 2008, 320, 889–892. [Google Scholar] [CrossRef] [PubMed]
- Kahrl, F.; Li, Y.; Su, Y.; Tennigkeit, T.; Wilkes, A.; Xu, J. Greenhouse gas emissions from nitrogen fertilizer use in China. Environ. Sci. Policy 2010, 13, 688–694. [Google Scholar] [CrossRef]
- Luo, J.; Ledgard, S.F.; Lindsey, S.B. Nitrous oxide and greenhouse gas emissions from grazed pastures as affected by use of nitrification inhibitor and restricted grazing regime. Sci. Total Environ. 2013, 465, 107–114. [Google Scholar] [CrossRef] [PubMed]
- Singh, J.; Kunhikrishnan, A.; Bolan, N.S.; Saggar, S. Impact of urease inhibitor on ammonia and nitrous oxide emissions from temperate pasture soil cores receiving urea fertilizer and cattle urine. Sci. Total Environ. 2013, 465, 56–63. [Google Scholar] [CrossRef] [PubMed]
- González, M.E.; Cea, M.; Medina, J.; González, A.; Diez, M.C.; Cartes, P.; Monreal, C.; Navia, R. Evaluation of biodegradable polymers as encapsulating agents for the development of a urea controlled-release fertilizer using biochar as support material. Sci. Total Environ. 2015, 505, 446–453. [Google Scholar] [CrossRef] [PubMed]
- Guo, M.; Liu, M.; Liang, R.; Niu, A. Granular urea-formaldehyde slow-release fertilizer with superabsorbent and moisture preservation. J. Appl. Polym. Sci. 2006, 99, 3230–3235. [Google Scholar] [CrossRef]
- Subbarao, G.V.; Wang, H.Y.; Ito, O.; Nakahara, K.; Berry, W.L. NH4+ triggers the synthesis and release of biological nitrification inhibition compounds in Brachiaria humidicola roots. Plant Soil 2007, 290, 245–257. [Google Scholar] [CrossRef]
- Castaldi, S.; Carfora, A.; Fiorentino, A.; Natale, A.; Messere, A.; Miglietta, F.; Cotrufo, M.F. Inhibition of net nitrification activity in a Mediterranean woodland: Possible role of chemicals produced by Arbutus unedo. Plant Soil 2009, 315, 273–283. [Google Scholar] [CrossRef]
- Majumdar, D. Suppression of nitrification and N2O emission by karanjin––A nitrification inhibitor prepared from karanja (Pongamia glabra Vent.). Chemosphere 2002, 47, 845–850. [Google Scholar] [CrossRef]
- Suescun, F.; Paulino, L.; Zagal, E.; Ovalle, C.; Muñoz, C. Plant extracts from the Mediterranean zone of Chile potentially affect soil microbial activity related to N transformations: A laboratory experiment. Acta Agric. Scand. Sect. B Soil Plant Sci. 2012, 62, 556–564. [Google Scholar] [CrossRef]
- Torres, L. Disminución de las Pérdidas de Nitrógeno Desde un Suelo Andisol Mediante el Uso de Extractos Vegetales. Tesis de Grado; Universidad de Concepción, Facultad de Agronomía: Chillán, Chile, 2012; p. 47. (In Spanish) [Google Scholar]
- Muñoz, C.; Quilodrán, C.; Navia, R. Evaluation of Biochar-Plant Extracts Complexes on Soil Nitrogen Dynamics. J. Biobased Mater. Bioenergy 2014, 8, 377–385. [Google Scholar] [CrossRef]
- Lehmann, J.; Joseph, S. Biochar for Environmental Management: Science and Technology; Earthscan: New York, NY, USA, 2009. [Google Scholar]
- Lehmann, J.; da Silva, J.P., Jr.; Steiner, C.; Nehls, T.; Zech, W.; Glaser, B. Nutrient availability and leaching in an archaeological Anthrosol and a Ferralsol of the Central Amazon basin: Fertilizer, manure and charcoal amendments. Plant Soil 2003, 249, 343–357. [Google Scholar] [CrossRef]
- Lehmann, J.; Gaunt, J.; Rondon, M. Biochar sequestration in terrestrial ecosystems—A review. Mitig. Adapt. Strategy. Glob. Chang. 2006, 11, 403–427. [Google Scholar] [CrossRef]
- Woolf, D.; Amonette, J.E.; Street-Perrott, F.A.; Lehmann, J.; Joseph, S. Sustainable biochar to mitigate global climate change. Nat. Commun. 2010. [Google Scholar] [CrossRef] [PubMed]
- Stolpe, N.B. Descripciones de los Principales Suelos de la VIII Región de Chile; Departamento de Suelos y Recursos Naturales, Universidad de Concepción: Chillán, Chile, 2006. (In Spanish) [Google Scholar]
- Celi, L.; Schnitzer, M.; Nègre, M. Analysis of carboxyl groups in soil humic acids by a wet chemical method, Fourier-transform infrared spectrophotometry, and solution-state carbon-13 nuclear magnetic resonance. A comparative study. Soil Sci. 1997, 162, 189–197. [Google Scholar] [CrossRef]
- Sobrero, M.C.; Ronco, A. Ensayo de toxicidad aguda con semillas de lechuga (Lactuca sativa L.). In Ensayos Toxicológicos y Métodos de Evaluación de Calidad de Aguas. Estandarización, Intercalibración, Resultados y Aplicaciones; Castillo, G., Ed.; Centro Internacional de Investigaciones para el Desarrollo: Ottawa, ON, Canada, 2004; pp. 71–79. (In Spanish) [Google Scholar]
- Tiquia, S.M.; Tam, N.F.; Hodgkiss, I.J. Effects of composting on phytotoxicity of spent pig-manure sawdust litter. Environ. Pollut. 1996, 93, 249–256. [Google Scholar] [CrossRef]
- Longeri, L.; Etchevers, J.; Venegas, J. Metodología de perfusión para estudios de nitrificación de suelos. Cienc. Investig. Agrar. (Chile) 1979, 4, 295–299. (In Spanish) [Google Scholar]
- Robarge, W.; Edwards, A.; Johnson, B. Water and waste water analysis for nitrate via nitration of salicylic acid 1. Commun. Soil Sci. Plant Anal. 1983, 14, 1207–1215. [Google Scholar] [CrossRef]
- Paris, O.; Zollfrank, C.; Zickler, G.A. Decomposition and carbonisation of wood biopolymers—A microstructural study of softwood pyrolysis. Carbon 2005, 43, 53–66. [Google Scholar] [CrossRef]
- Baldock, J.A.; Smernik, R.J. Chemical composition and bioavailability of thermally altered Pinus resinosa (Red pine) wood. Org. Geochem. 2002, 33, 1093–1109. [Google Scholar] [CrossRef]
- Schmidt, M.W.I.; Noack, A.G. Black carbon in soils and sediments: Analysis, distribution, implications, and current challenges. Glob. Biogeochem. Cycles 2000, 14, 777–793. [Google Scholar] [CrossRef]
- Emino, E.R.; Warman, P.R. Biological assay for compost quality. Compost Sci. Util. 2004, 12, 342–348. [Google Scholar] [CrossRef]
- Smolander, A.; Kanerva, S.; Adamczyk, B.; Kitunen, V. Nitrogen transformations in boreal forest soils—Does composition of plant secondary compounds give any explanations? Plant Soil 2012, 350, 1–26. [Google Scholar] [CrossRef]
- Hättenschwiler, S.; Vitousek, P.M. The role of polyphenols in terrestrial ecosystem nutrient cycling. Trends Ecol. Evol. 2000, 15, 238–243. [Google Scholar] [CrossRef]
- Kraus, T.E.C.; Dahlgren, R.A.; Zasoski, R.J. Tannins in nutrient dynamics of forest ecosystems—A review. Plant Soil 2003, 256, 41–66. [Google Scholar] [CrossRef]
- Zhang, L.-L.; Wu, Z.-J.; Shi, Y.-F.; Chen, L.-J.; Song, Y.-C.; Juan, Y.-H. Inhibitory effects of aromatic compounds on soil nitrification. Pedosphere 2010, 20, 326–333. [Google Scholar] [CrossRef]
- Downie, A.; Crosky, A.; Munroe, P. Physical properties of biochar. In Biochar for Environmental Management: Science and Technology; Earthscan: New York, NY, USA, 2009; Chapter 2; pp. 13–32. [Google Scholar]
- Major, J.; Steiner, C.; Downie, A.; Lehmann, J. Biochar effects on nutrient leaching. In Biochar for Environmental Management: Science and Technology; Earthscan: New York, NY, USA, 2009; Chapter 15; pp. 271–287. [Google Scholar]
- Williams, P.T.; Reed, A.R. Development of activated carbon pore structure via physical and chemical activation of biomass fibre waste. Biomass Bioenergy 2006, 30, 144–152. [Google Scholar] [CrossRef]
- Kasozi, G.N.; Zimmerman, A.R.; Nkedi-Kizza, P.; Gao, B. Catechol and humic acid sorption onto a range of laboratory-produced black carbons (biochars). Environ. Sci. Technol. 2010, 44, 6189–6195. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Chen, G.; Chen, L.; Chen, Y.; Lehmann, J.; McBride, M.B.; Hay, A.G. Adsorption of copper and zinc by biochars produced from pyrolysis of hardwood and corn straw in aqueous solution. Bioresour. Technol. 2011, 102, 8877–8884. [Google Scholar] [CrossRef] [PubMed]
- González, M.E; Cea, M.; Sangaletti, N.; González, A.; Toro, C.; Diez, M.C.; Moreno, N.; Querol, X.; Navia, R. Biochar derived from agricultural and forestry residual biomass: Characterization and potential application for enzymes immobilization. J. Biobased Mater. Bioenergy 2013, 7, 724–732. [Google Scholar]
- Zeng, Z.; Zhang, S.-D.; Li, T.-Q.; Zhao, F.-L.; He, Z.-L.; Zhao, H.-P.; Yang, X.-E.; Wang, H.-L.; Zhao, J.; Rafiq, M.T. Sorption of ammonium and phosphate from aqueous solution by biochar derived from phytoremediation plants. J. Zhejiang Univ. Sci. B 2013, 14, 1152–1161. [Google Scholar] [CrossRef] [PubMed]
- Zhu, D.; Kwon, S.; Pignatello, J.J. Adsorption of single-ring organic compounds to wood charcoals prepared under different thermochemical conditions. Environ. Sci. Technol. 2005, 39, 3990–3998. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Reyes-Escobar, J.; Zagal, E.; Sandoval, M.; Navia, R.; Muñoz, C. Development of a Biochar-Plant-Extract-Based Nitrification Inhibitor and Its Application in Field Conditions. Sustainability 2015, 7, 13585-13596. https://doi.org/10.3390/su71013585
Reyes-Escobar J, Zagal E, Sandoval M, Navia R, Muñoz C. Development of a Biochar-Plant-Extract-Based Nitrification Inhibitor and Its Application in Field Conditions. Sustainability. 2015; 7(10):13585-13596. https://doi.org/10.3390/su71013585
Chicago/Turabian StyleReyes-Escobar, Jhónatan, Erick Zagal, Marco Sandoval, Rodrigo Navia, and Cristina Muñoz. 2015. "Development of a Biochar-Plant-Extract-Based Nitrification Inhibitor and Its Application in Field Conditions" Sustainability 7, no. 10: 13585-13596. https://doi.org/10.3390/su71013585
APA StyleReyes-Escobar, J., Zagal, E., Sandoval, M., Navia, R., & Muñoz, C. (2015). Development of a Biochar-Plant-Extract-Based Nitrification Inhibitor and Its Application in Field Conditions. Sustainability, 7(10), 13585-13596. https://doi.org/10.3390/su71013585






