Understanding and Enhancing Soil Biological Health: The Solution for Reversing Soil Degradation
Abstract
:1. Introduction
2. What Constitutes a Healthy Soil?
2.1. Definition of Soil Health
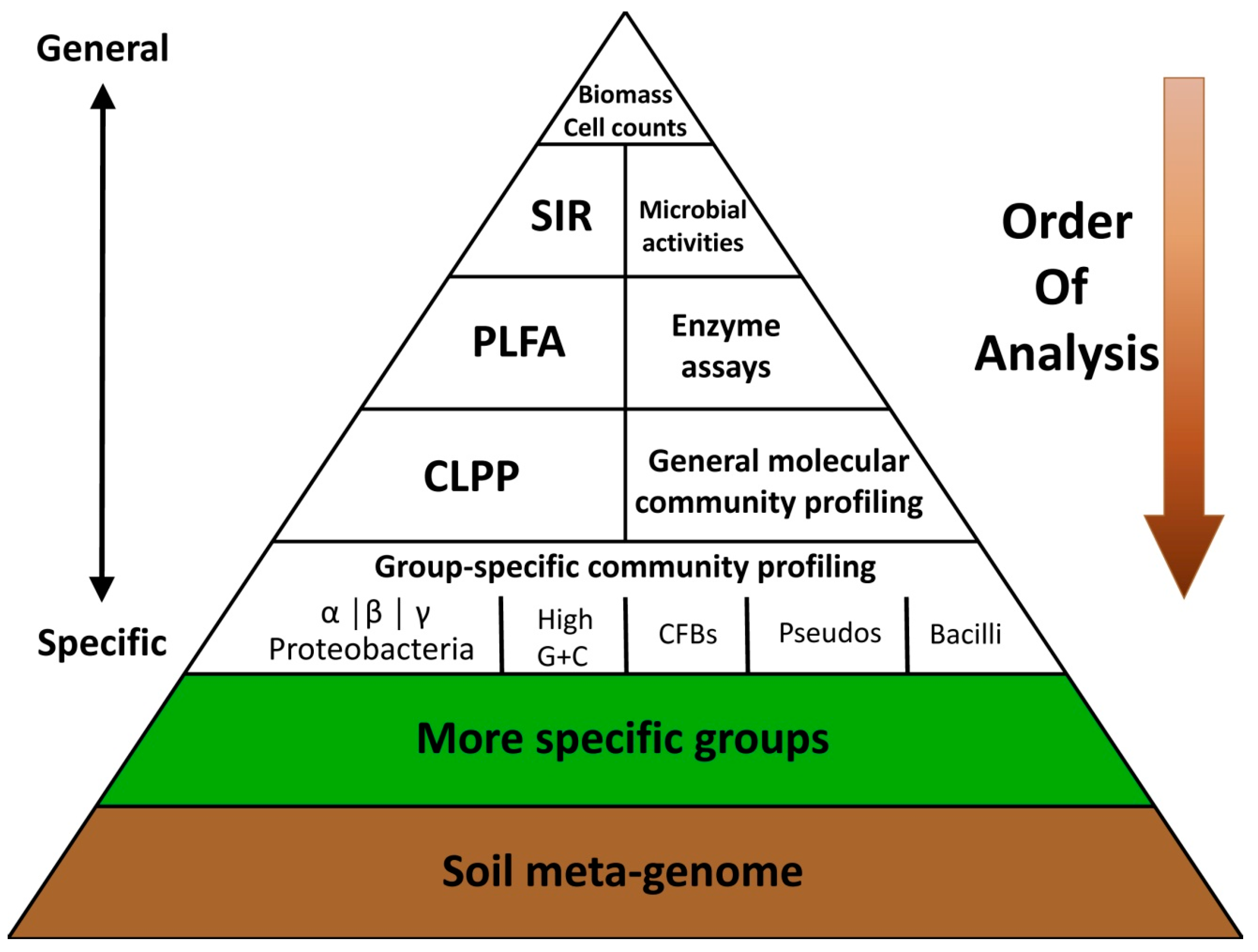
2.2. Existing Soil Quality/Health Assessment
2.3. The Significance of Soil Health to Agroecosystems and Soil Restoration
3. How Does Soil Biology Influence Soil Health, or What’s Missing in a Degraded Soil?
3.1. Soil Biology Overview
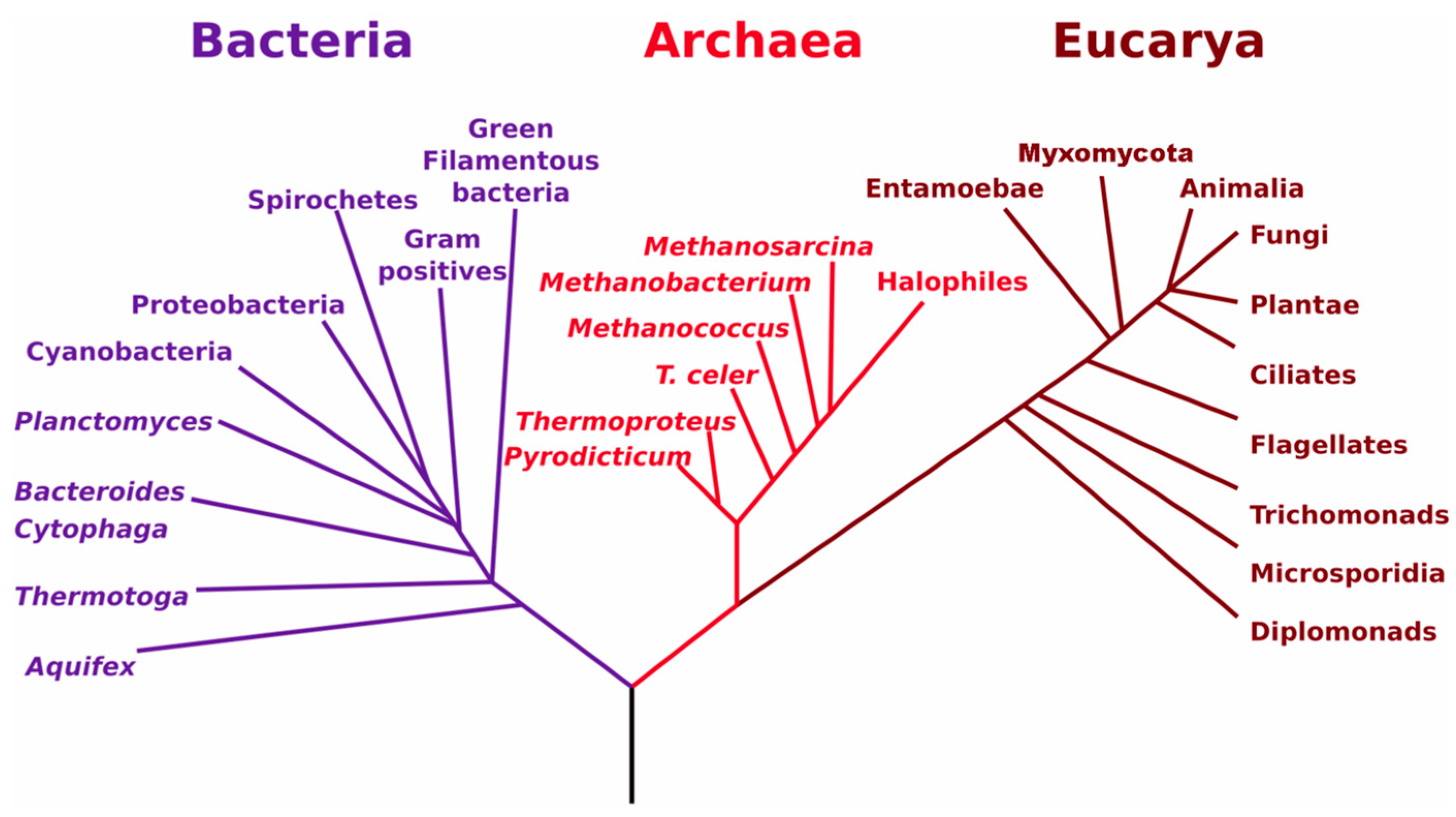
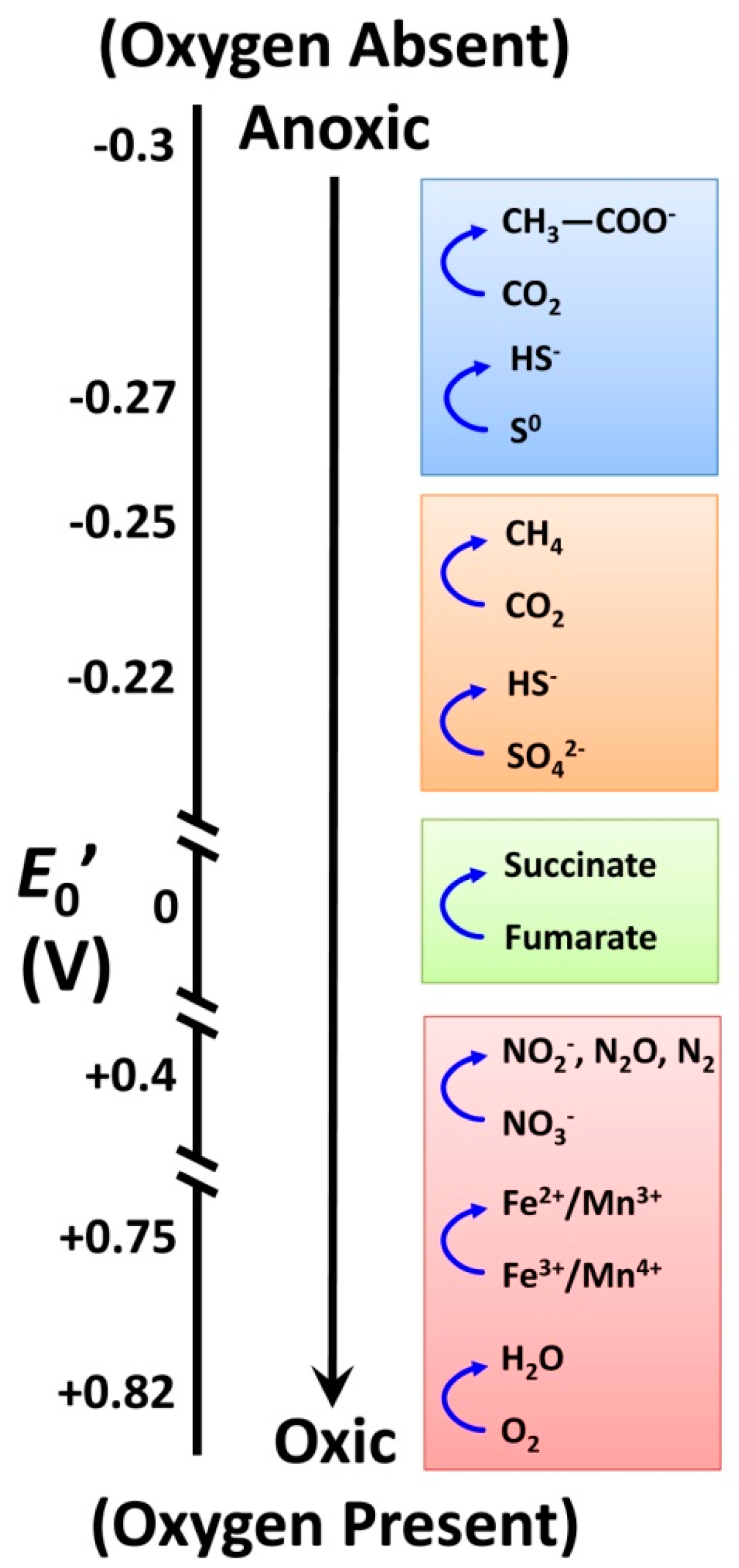
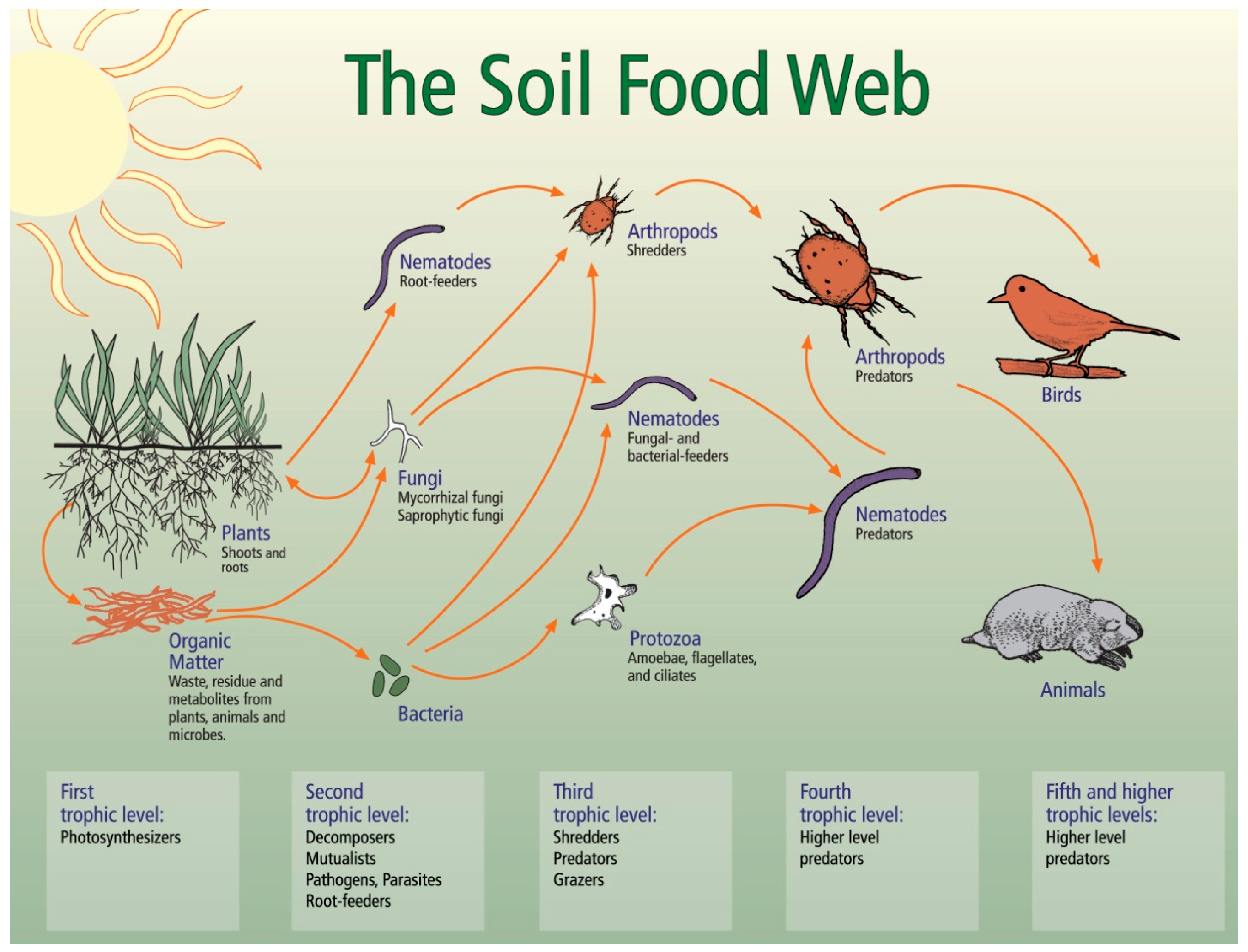
3.2. Relationships between Soil Biology, Soil Quality and Restoration Strategies
- (1)
- Accept, hold and release nutrients and other chemical constituents.
- (2)
- Accept, hold and release water to plants, streams and groundwater.
- (3)
- Promote and sustain root growth.
- (4)
- Maintain suitable soil biotic habitat.
- (5)
- Respond to management.
- (6)
- Resist degradation.
3.3. How does Soil Biology Influence Ecosystem Services that Are Crucial for Well-Functioning Soils?
| Ecosystem Services Provided by Soil Biota |
|---|
| Regulation of biogeochemical cycles |
| Retention and delivery of nutrients to primary producers |
| Maintenance of soil structure and fertility |
| Bioremediation of pollutants |
| Provision of clean drinking water |
| Mitigation of floods and droughts |
| Erosion control |
| Regulation of atmospheric trace gases |
| Pest and pathogen control |
| Regulation of plant production via non-nutrient biochemicals |
3.4. The Significance of Soil Biology to Sustaining Agriculture and Restoring Soil Health
4. How Can Soil Biology Be Used More Effectively to Mitigate Soil Degradation?
4.1. Strategies to Manipulate Soil Biology Focusing on Soil Microorganisms
4.2. Specific Approach: Plant Selection and Microbial Amendments
4.3. A General Approach: Modify the Whole Soil Community

5. What Are the Primary Knowledge Gaps Limiting Manipulation of Soil Biological Communities and Mitigation of Degraded Soils?
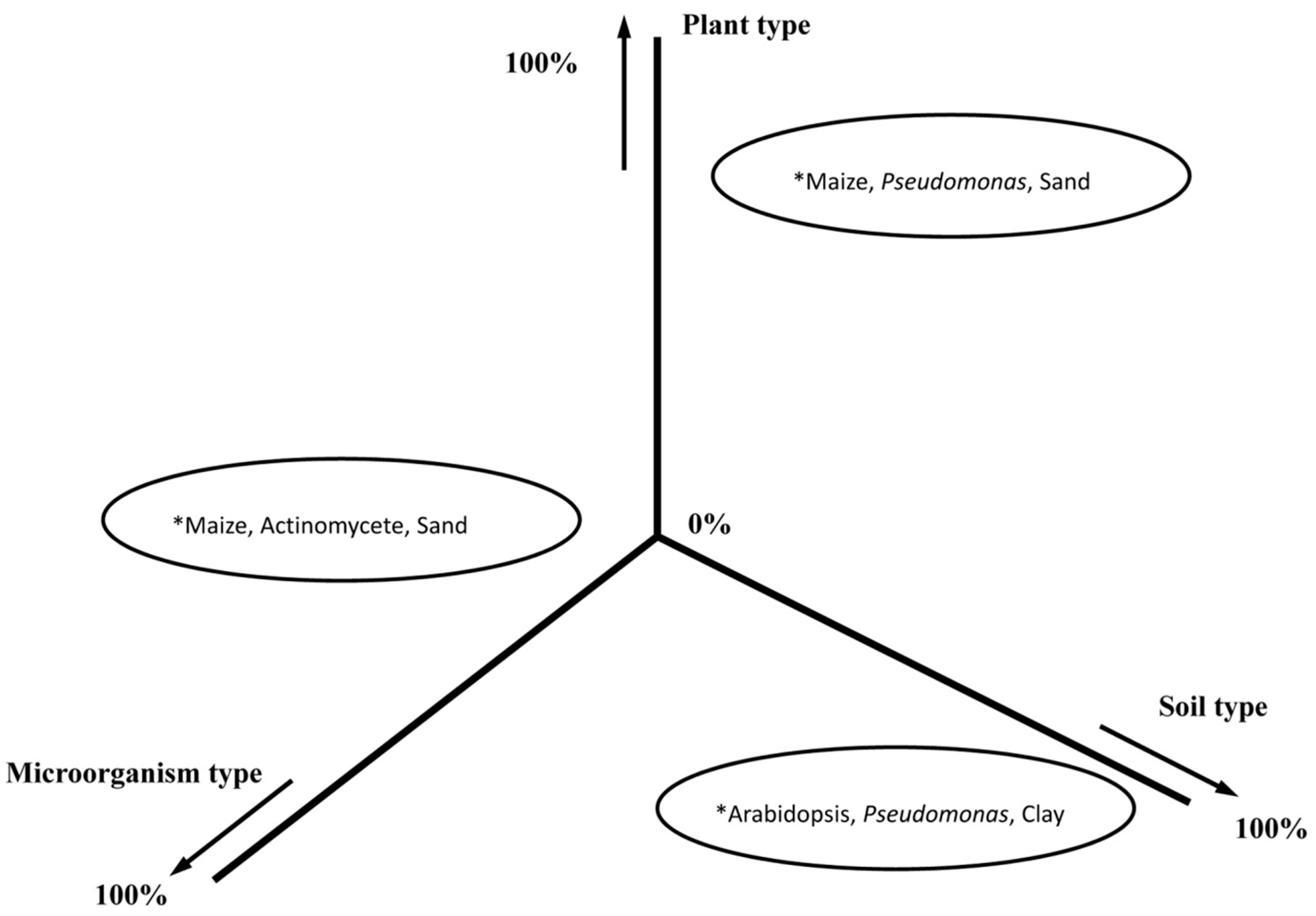
6. What Are the Highest Priority Research Needs to Improve Soil Health and Reverse Soil Degradation?
6.1. Framing High Priority Research Questions
6.2. Fundamental Information Lacking Regarding the Identities, Distributions, Ecology, and Functionality of Soil Biota
6.3. Defining Relationships among Climate, Edaphic Factors, and management with Respect to Soil Biota
6.4. Development of Improved Indicators of Soil Health
7. Soil Biology Research Investments Needed to Ensure Our Future by Promoting Soil Health and Mitigate Soil Degradation
8. Summary and Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Karlen, D.L.; Andrews, S.S.; Weinhold, B.J.; Zobeck, T.M. Soil quality assessment: Past, present, and future. Electron. J. Integr. Biosci. 2008, 6, 3–14. [Google Scholar]
- Karlen, D.L. Soil health: The concept, its role, and strategies for monitoring. In Soil Ecology and Ecosystem Services; Wall, D.H., Bardgett, R.D., Behan-Pelletier, V., Herrick, J.E., Jones, H., Ritz, K., Six, J., Strong, D.R., van der Putten, W.M., Eds.; Oxford University Press: New York, NY, USA, 2012; pp. 331–336. [Google Scholar]
- Doran, J.W.; Sarrantonio, M.; Liebig, M.A. Soil health and sustainability. Adv. Agron. 1996, 56, 1–54. [Google Scholar]
- Magdoff, F.; van Es, H. Building Soils for Better Crops; Sustainable Agriculture Network Publications: Burlington, VT, USA, 2000; p. 241. [Google Scholar]
- Harris, R.F.; Bezdicek, D.F. Descriptive aspects of soil quality/health. In Defining Soil Quality for a Sustainable Environment; Doran, J.W., Coleman, D.C., Bezdicek, D.F., Stewart, B.A., Eds.; Soil Science Society of America: Madison, WI, USA, 1994; pp. 23–35. [Google Scholar]
- Warkentin, B.P.; Fletcher, H.F. Soil quality for intensive agriculture. In Intensive Agriculture Society of Science, Soil and Manure. Proceedings of the International Seminar on Soil Environment and Fertilizer Management; National Institute of Agricultural Science: Tokyo, Japan, 1977; pp. 594–598. [Google Scholar]
- Doran, J.W.; Parkin, T.B. Defining and assessing soil quality. In Defining Soil Quality for a Sustainable Environment; Doran, J.W., Coleman, D.C., Bezdicek, D.F., Stewart, B.A., Eds.; Soil Science Society of America: Madison, WI, USA, 1994; pp. 3–21. [Google Scholar]
- Karlen, D.L.; Erbach, D.C.; Kaspar, T.C.; Colvin, T.S.; Berry, E.C.; Timmons, D.R. Soil tilth: A review of past perceptions and future needs. Soil Sci. Soc. Am. J. 1990, 54, 153–161. [Google Scholar] [CrossRef]
- Karlen, D.L.; Mausbach, M.J.; Doran, J.W.; Cline, R.G.; Harris, R.F.; Schuman, G.E. Soil quality: A concept, definition, and framework for evaluation. Soil Sci. Soc. Am. J. 1997, 61, 4–10. [Google Scholar] [CrossRef]
- Kaspar, T.C.; Radke, J.K.; Laflen, J.M. Small grain cover crops and wheel traffic effects on infiltration, runoff, and erosion. J. Soil Water Conserv. 2001, 56, 160–164. [Google Scholar]
- Larson, W.E.; Pierce, F.J. Conservation and enhancement of soil quality. In Evaluation for Sustainable Land Management in the Developing World, Proceedings of the International Workshop, Chiang Rai, Thailand, 15–21 September 1991; Dumanski, J., Pushparajah, E., Larson, M., Myers, R., Eds.; Int. Board for Soil Res. and Management: Bangkok, Thailand, 1991; Volume 2, pp. 175–203. [Google Scholar]
- National Research Council (NRC). Soil and Water Quality: An Agenda for Agriculture; National Academy Press: Washington, DC, USA, 1993. [Google Scholar]
- Doran, J.W.; Jones, A.J. Methods for Assessing Soil Quality; Soil Science Society of America: Madison, WI, USA, 1996. [Google Scholar]
- Andrews, S.S.; Karlen, D.L.; Cambardella, C.A. The soil management assessment framework: A quantitative soil quality evaluation method. Soil Sci. Soc. Am. J. 2004, 68, 1945–1962. [Google Scholar] [CrossRef]
- Karlen, D.L.; Stott, D.E. A framework for evaluating physical and chemical indicators of soil quality. In Defining Soil Quality for a Sustainable Environment; Doran, J.W., Coleman, D.C., Bezdicek, D.F., Stewart, B.A., Eds.; Soil Science Society of America: Madison, WI, USA, 1994; pp. 53–72. [Google Scholar]
- Smith, J.L.; Halvorson, J.J.; Papendick, R.I. Using multiple-variable indicator kriging for evaluating soil quality. Soil Sci. Soc. Am. J. 1993, 57, 743–749. [Google Scholar] [CrossRef]
- Parr, J.; Papendick, R.; Hornick, S.; Meyer, R. Soil quality: Attributes and relationship to alternative and sustainable agriculture. Am. J. Altern. Agric. 1992, 7, 5–11. [Google Scholar] [CrossRef]
- Daily, G.C.; Matson, P.A.; Vitousek, P.M. Ecosystem services supplied by soil. In Nature’s Services Societal Dependence on Natural Ecosystems; Daily, G.C., Ed.; Island Press: Washington, DC, USA, 1997; pp. 365–374. [Google Scholar]
- Romig, D.E.; Garlynd, M.J.; Harris, R.F.; McSweeney, K. How farmers assess soil health and quality. J. Soil Water Conserv. 1995, 50, 229–236. [Google Scholar]
- Jenny, H. Factors of Soil Formation; McGraw-Hill Book Co.: New York, NY, USA, 1941. [Google Scholar]
- Seybold, C.A.; Mausbach, M.J.; Karlen, D.L.; Rogers, H.H. Quantification of soil quality. In Soil Processes and the Carbon Cycle; Lal, R., Kimble, J.M., Follett, R.F., Stewart, B.A., Eds.; CRC Press Inc.: Boca Raton, FL, USA, 1998; pp. 387–404. [Google Scholar]
- Halvorson, J.J.; Smith, J.L.; Papendick, R.I. Issues of scale for evaluating soil quality. J. Soil Water Conserv. 1997, 52, 26–30. [Google Scholar]
- Doran, J.W.; Parkin, T.B. Quantitative indicators of soil quality: A minimum data set. In Methods for Assessing Soil Quality; Doran, J.W., Jones, A.D., Eds.; Soil Science Society of America: Madison, WI, USA, 1996; pp. 25–37. [Google Scholar]
- Gregorich, E.G.; Carter, M.R.; Angers, D.A.; Monreal, C.M.; Ellert, B.H. Towards a minimum data set to assess soil organic matter quality in agricultural soils. Can. J. Soil Sci. 1994, 74, 367–385. [Google Scholar] [CrossRef]
- Weil, R.R.; Magdoff, F. Significance of soil organic matter to soil quality and health. In Soil Organic Matter in Sustainable Agriculture; Magdoff, F., Weil, R.R., Eds.; CRC Press: Boca Raton, FL, USA, 2004; pp. 1–43. [Google Scholar]
- Wienhold, B.J.; Karlen, D.L.; Andrews, S.S.; Stott, D.E. Protocol for soil management assessment framework (SMAF) soil indicator scoring curve development. Renew. Agric. Food Syst. 2009, 24, 260–266. [Google Scholar] [CrossRef]
- Stott, D.E.; Andrews, S.S.; Liebig, M.A.; Wienhold, B.J.; Karlen, D.L. Evaluation of β-glucosidase activity as a soil quality indicator for the soil management assessment framework (SMAF). Soil Sci. Soc. Am. J. 2010, 74, 107–119. [Google Scholar] [CrossRef]
- Karlen, D.L.; Andrews, S.S.; Doran, J.W. Soil quality: Current concepts and applications. Adv. Agron. 2001, 74, 1–40. [Google Scholar]
- Karlen, D.L.; Gardner, J.C.; Rosek, M.J. A soil quality framework for evaluating the impact of CRP. J. Prod. Agric. 1998, 11, 56–60. [Google Scholar] [CrossRef]
- Potter, S.R.; Andrews, S.S.; Atwood, J.D.; Kellogg, R.L.; Lemunyon, J.; Norfleet, M.L.; Oman, D. Model Simulation of Soil Loss, Nutrient Loss, and Change in Soil Organic Carbon Associated with Crop Production; USDA-NRCS, Ed.; USDA Natural Resources Conservation Service: Washington, DC, USA, 2006. [Google Scholar]
- Marriott, E.E.; Wander, M. Qualitative and quantitative differences in particulate organic matter fractions in organic and conventional farming systems. Soil Biol. Biochem. 2006, 38, 1527–1536. [Google Scholar] [CrossRef]
- Pikul, J.L., Jr.; Johnson, J.M.F.; Schumacher, T.E.; Vigil, M.; Riedell, W.E. Change in surface soil carbon under rotated corn in eastern south Dakota. Soil Sci. Soc. Am. J. 2008, 72, 1738–1744. [Google Scholar] [CrossRef]
- Singer, J.W.; Kohler, K.A.; Liebman, M.; Richard, T.L.; Cambardella, C.A.; Buhler, D.D. Tillage and compost affect yield of corn, soybean, and wheat and soil fertility. Agron. J. 2004, 96, 531–537. [Google Scholar] [CrossRef]
- Teasdale, J.R. Strategies for soil conservation in no-tillage and organic farming systems. J. Soil Water Conserv. 2007, 62, 144A–147A. [Google Scholar]
- Marriott, E.E.; Wander, M.M. Total and labile soil organic matter in organic and conventional farming systems. Soil Sci. Soc. Am. J. 2006, 70, 950–959. [Google Scholar] [CrossRef]
- Fließbach, A.; Oberholzer, H.R.; Gunst, L.; Mader, P. Soil organic matter and biological soil quality indicators after 21 years of organic and conventional farming. Agric. Ecosyst. Environ. 2007, 118, 273–284. [Google Scholar] [CrossRef]
- Tu, C.; Louws, F.J.; Creamer, N.G.; Mueller, J.P.; Brownie, C.; Fager, K.; Bell, M.; Hu, S.J. Responses of soil microbial biomass and N availability to transition strategies from conventional to organic farming systems. Agric. Ecosyst. Environ. 2006, 113, 206–215. [Google Scholar] [CrossRef]
- Langdale, G.W.; Blevins, R.L.; Karlen, D.L.; McCool, D.K.; Nearing, M.A.; Skidmore, E.L.; Thomas, A.W.; Tyler, D.D.; Williams, J.R. Cover crop effects on soil erosion by wind and water. In Cover Crops for Clean Water; Hargrove, W.L., Ed.; Soil and Water Conservation Society: Ankeny, IA, USA, 1991; pp. 15–22. [Google Scholar]
- Kaspar, T.C.; Jaynes, D.B.; Parkin, T.B.; Moorman, T.B. Rye cover crop and gamagrass strip effects on NO3 concentration and load in tile drainage. J. Environ. Qual. 2007, 36, 1503–1511. [Google Scholar] [CrossRef] [PubMed]
- Kladivko, E.J.; Frankenberger, J.R.; Jaynes, D.B.; Meek, D.W.; Jenkinson, B.J.; Fausey, N.R. Nitrate leaching to subsurface drains as affected by drain spacing and changes in crop production system. J. Environ. Qual. 2004, 33, 1803–1813. [Google Scholar] [CrossRef] [PubMed]
- Strock, J.S.; Porter, P.M.; Russelle, M.P. Cover cropping to reduce nitrate loss through subsurface drainage in the northern US corn belt. J. Environ. Qual. 2004, 33, 1010–1016. [Google Scholar] [CrossRef] [PubMed]
- Snapp, S.S.; Swinton, S.M.; Labarta, R.; Mutch, D.; Black, J.R.; Leep, R.; Nyiraneza, J.; O’Neil, K. Evaluating cover crops for benefits, costs and performance within cropping system niches. Agron. J. 2005, 97, 322–332. [Google Scholar]
- Cambardella, C.A.; Johnson, J.M.F.; Varvel, G.E. Soil carbon sequestration in central U.S. Agroecosystems. In Managing Agricultural Greenhouse Gases: Coordinated Agricultural Research through Gracenet to Address Our Changing Climate; Liebig, M.A., Franzleubbers, A.J., Follet, R.F., Eds.; Academic Press, Elsevier: San Diego, CA, USA, 2012; pp. 41–58. [Google Scholar]
- Franzluebbers, A.J. Achieving soil organic carbon sequestration with conservation agricultural systems in the southeastern United States. Soil Sci. Soc. Am. J. 2010, 74, 347–357. [Google Scholar] [CrossRef]
- Johnson, D.W.; Verburg, P.S.J.; Amone, J.A. Soil extraction, ion exchange resin, and ion exchange membrane measures of soil mineral nitrogen during incubation of a tallgrass prairie soil. Soil Sci. Soc. Am. J. 2005, 69, 260–265. [Google Scholar] [CrossRef]
- Follett, R.F.; Vogel, K.P.; Varvel, G.E.; Mitchell, R.B.; Kimble, J. Soil carbon sequestration by switchgrass and no-till maize grown for bioenergy. BioEnergy Res. 2012, 5, 866–875. [Google Scholar] [CrossRef]
- Gál, A.; Vyn, T.J.; Michéli, E.; Kladivko, E.J.; McFee, W.W. Soil carbon and nitrogen accumulation with long-term no-till versus moldboard plowing overestimated with tilled-zone sampling depths. Soil Tillage Res. 2007, 96, 42–51. [Google Scholar] [CrossRef]
- Yang, X.; Drury, C.; Wander, M.; Kay, B. Evaluating the effect of tillage on carbon sequestration using the minimum detectable difference concept. Pedosphere 2008, 18, 421–430. [Google Scholar] [CrossRef]
- Clapp, C.E.; Allmaras, R.R.; Layese, M.F.; Linden, D.R.; Dowdy, R.H. Soil organic carbon and C-13 abundance as related to tillage, crop residue, and nitrogen fertilization under continuous corn management in minnesota. Soil Tillage Res. 2000, 55, 127–142. [Google Scholar] [CrossRef]
- Huggins, D.R.; Allmaras, R.R.; Clapp, C.E.; Lamb, J.A.; Randall, G.W. Corn-soybean sequence and tillage effects on soil carbon dynamics and storage. Soil Sci. Soc. Am. J. 2007, 71, 145–154. [Google Scholar] [CrossRef]
- Gans, J.; Wolinsky, M.; Dunbar, J. Computational improvements reveal great bacterial diversity and high metal toxicity in soil. Science 2005, 309, 1387–1390. [Google Scholar] [CrossRef] [PubMed]
- Gold, T. The deep, hot biosphere. Proc. Natl. Acad. Sci. USA 1992, 89, 6045–6049. [Google Scholar] [CrossRef] [PubMed]
- Pace, N.R. Mapping the tree of life: Progress and prospects. Microbiol. Mol. Biol. Rev. 2009, 73, 565–576. [Google Scholar] [CrossRef] [PubMed]
- Thauer, R.K. A fifth pathway of carbon fixation. Science 2007, 318, 1732–1733. [Google Scholar] [CrossRef] [PubMed]
- Beja, O.; Aravind, L.; Koonin, E.V.; Suzuki, M.T.; Hadd, A.; Nguyen, L.P.; Jovanovich, S.B.; Gates, C.M.; Feldman, R.A.; Spudich, J.L.; et al. Bacterial rhodospin: Evidence for a new type of phototrophy in the sea. Science 2000, 289, 1902–1906. [Google Scholar] [CrossRef] [PubMed]
- Boone, D.; Liu, Y.; Zhao, Z.; Balkwill, D.; Drake, G.; Stevens, T.; Aldrich, H. Bacillus infernus sp. Nov., an Fe(iii)- and Mn(iv)-reducing anaerobe from the deep terrestrial subsurface. Int. J. Syst. Bacteriol. 1995, 45, 441–448. [Google Scholar] [CrossRef] [PubMed]
- Leininger, S.; Urich, T.; Schloter, M.; Schwark, L.; Qi, J.; Nicol, G.; Prosser, J.; Schuster, S.; Schleper, C. Archaea predominate among ammonia-oxidizing prokaryotes in soils. Nature 2006, 442, 806–809. [Google Scholar] [CrossRef] [PubMed]
- Nelson, K.E.; Clayton, R.A.; Gill, S.R.; Gwinn, M.L.; Dodson, R.J.; Haft, D.H.; Hickey, E.K.; Peterson, J.D.; Nelson, W.C.; Ketchum, K.A.; et al. Evidence for lateral gene transfer between archaea and bacteria from genome sequence of Thermotoga maritima. Nature 1999, 399, 323–329. [Google Scholar] [CrossRef] [PubMed]
- Lavelle, P.; Decaëns, T.; Aubert, M.; Barot, S.; Blouin, M.; Bureau, F.; Margerie, P.; Mora, P.; Rossi, J.P. Soil invertebrates and ecosystem services. Eur. J. Soil Biol. 2006, 42, S3–S15. [Google Scholar] [CrossRef]
- Blouin, M.; Hodson, M.E.; Delgado, E.A.; Baker, G.; Brussaard, L.; Butt, K.R.; Dai, J.; Dendooven, L.; Pérès, G.; Tondoh, J. A review of earthworm impact on soil function and ecosystem services. Eur. J. Soil Sci. 2013, 64, 161–182. [Google Scholar] [CrossRef]
- Falkowski, P.G.; Fenchel, T.; DeLong, E.F. The microbial engines that drive earth’s biogeochemical cycles. Science 2008, 320, 1034–1039. [Google Scholar] [CrossRef] [PubMed]
- Kowalchuk, G.A.; Jones, S.E.; Blackall, L.L. Microbes orchestrate life on earth. ISME J. 2008, 2, 795–796. [Google Scholar] [CrossRef] [PubMed]
- Benayas, J.M.; Newton, A.C.; Diaz, A.; Bullock, J.M. Enhancement of biodiversity and ecosystem services by ecological restoration: A meta-analysis. Science 2009, 325, 1121–1124. [Google Scholar] [CrossRef] [PubMed]
- Pimental, D.; Wilson, C.; McCullum, C.; Huang, R.; Dwen, P.; Flack, J.; Tran, Q.; Saltman, T.; Cliff, B. Economic and environmental benefits of biodiversity. Bioscience 1997, 47, 747–757. [Google Scholar] [CrossRef]
- Nielsen, U.N.; Ayres, E.; Wall, D.H.; Bardgett, R.D. Soil biodiversity and carbon cycling: A review and synthesis of studies examining diversity-function relationships. Eur. J. Soil Sci. 2010, 62, 105–116. [Google Scholar] [CrossRef]
- Pritchard, S.G. Soil organisms and global climate change. Plant Pathol. 2011, 60, 82–89. [Google Scholar] [CrossRef]
- Cardinale, B.J. Biodiversity improves water quality through niche partitioning. Nature 2011, 472, 86–89. [Google Scholar] [CrossRef] [PubMed]
- Gessner, M.O.; Swan, C.M.; Dang, C.K.; McKie, B.G.; Bardgett, R.D.; Wall, D.H.; Hattenschwiler, S. Diversity meets decomposition. Trends Ecol. Evol. 2010, 25, 372–380. [Google Scholar] [CrossRef] [PubMed]
- Wagg, C.; Bender, S.F.; Widmer, F.; van der Heijden, M.G.A. Soil biodiversity and soil community composition determine ecosystem multifunctionality. Proc. Natl. Acad. Sci. USA 2014, 111, 5266–5270. [Google Scholar] [CrossRef] [PubMed]
- Van Elsas, J.D.; Chiurazzi, M.; Mallon, C.A.; Elhottova, D.; Kristufek, V.; Salles, J.F. Microbial diversity determines the invasion of soil by a bacterial pathogen. Proc. Natl. Acad. Sci. USA 2012, 109, 1159–1164. [Google Scholar] [CrossRef] [PubMed]
- Kremen, C. Managing ecosystem services, what do we need to know about their ecology? Ecol. Lett. 2005, 8, 468–479. [Google Scholar] [CrossRef] [PubMed]
- Hooper, D.U.; Chapin, F.S.; Ewel, J.J.; Hector, A.; Inchausti, P.; Lavorel, S.; Lawton, J.H.; Lodge, D.M.; Loreau, M.; Naeem, S.; et al. Effects of biodiversity on ecosystem functioning: A consensus of current knowledge. Ecol. Monogr. 2005, 75, 3–35. [Google Scholar] [CrossRef]
- Wall, D.H.; Bardgett, R.D.; Covich, A.P.; Snelgrove, P.V.R. The need for understanding how biodiversity and ecosystem functioning affect ecosystem service in soils and sediments. In Sustaining Biodiversity and Ecosystem Services in Soils and Sediments; Wall, D.H., Ed.; Island Press: Washington, DC, USA, 2004; Volume SCOPE 64, pp. 1–12. [Google Scholar]
- Montgomery, D.R. Dirt: The Erosion of Civilizations; Univ of California Press: Oakland, CA, USA, 2012. [Google Scholar]
- Altieri, M.A. The ecological role of biodiversity in agroecosystems. Agric. Ecosyst. Environ. 1999, 74, 19–31. [Google Scholar] [CrossRef]
- Wright, S.; Upadhyaya, A. A survey of soils for aggregate stability and glomalin, a glycoprotein produced by hyphae of arbuscular mycorrhizal fungi. Plant Soil 1998, 198, 97–107. [Google Scholar] [CrossRef]
- Six, J.; Bossuyt, H.; Degryze, S.; Denef, K. A history of research on the link between (micro)aggregates, soil biota, and soil organic matter dynamics. Soil Tillage Res. 2004, 79, 7–31. [Google Scholar] [CrossRef]
- Peck, S.L.; McQuaid, B.; Campbell, C.L. Using ant species (Hymenoptera: Formicidae) as a biological indicator of agroecosystem condition. Environ. Entomol. 1998, 27, 1102–1110. [Google Scholar] [CrossRef]
- Pinto-Tomás, A.A.; Anderson, M.A.; Suen, G.; Stevenson, D.M.; Chu, F.S.T.; Cleland, W.W.; Weimer, P.J.; Currie, C.R. Symbiotic nitrogen fixation in the fungus gardens of leaf-cutter ants. Science 2009, 326, 1120–1123. [Google Scholar] [CrossRef] [PubMed]
- Smith, S.E.; Read, D.J. Mycorrhizal Symbiosis, 3rd ed.; Academic Press: London, UK, 2008. [Google Scholar]
- Oberson, A.; Joner, E.J. Microbial turnover of phosphorus in soil. In Organic Phosphorus in the Environment; Turner, B.L., Frossard, E., Baldwin, D.S., Eds.; CABI International: Wallingford, CT, USA, 2005; pp. 133–165. [Google Scholar]
- Richardson, A.E.; Lynch, J.P.; Ryan, P.R.; Delhaize, E.; Smith, F.A.; Smith, S.E.; Harvey, P.R.; Ryan, M.H.; Veneklaas, E.J.; Lambers, H.; et al. Plant and microbial strategies to improve the phosphorus efficiency of agriculture. Plant Soil 2011, 349, 121–156. [Google Scholar] [CrossRef]
- McLaughlin, M.J.; McBeath, T.M.; Smernik, R.; Stacey, S.P.; Ajiboye, B.; Guppy, C. The chemical nature of P accumulation in agricultural soils—Implications for fertiliser management and design: An Australian perspective. Plant Soil 2011, 349, 69–87. [Google Scholar] [CrossRef]
- Simpson, R.J.; Oberson, A.; Culvenor, R.A.; Ryan, M.H.; Veneklaas, E.J.; Lambers, H.; Lynch, J.P.; Ryan, P.R.; Delhaize, E.; Smith, F.A.; et al. Strategies and agronomic interventions to improve the phosphorus-use efficiency of farming systems. Plant Soil 2011, 349, 89–120. [Google Scholar] [CrossRef]
- Drinkwater, L.E.; Snapp, S.S. Nutrients in agroecosystems: Rethinking the management paradigm. Adv. Agron. 2007, 92, 163–186. [Google Scholar]
- Dawson, J.C.; Huggins, D.R.; Jones, S.J. Characterizing nitrogen use efficiency in natural and agricultural ecosystems to improved the performance of cereal crops in low-input and organic agricultural systems. Field Crops Res. 2008, 107, 89–101. [Google Scholar] [CrossRef]
- Angus, T.A. Symposium on microbial insecticides. I. Bacterial pathogens of insects as microbial insecticides. Bacteriol. Rev. 1965, 29, 364–372. [Google Scholar] [PubMed]
- Vandenberg, J.D. Standardized bioassay and screening of Beauveria bassiana and Paecilomyces fumosoroseus against the Russian wheat aphid (Homoptera: Aphididae). J. Econ. Entomol. 1996, 89, 1418–1423. [Google Scholar] [CrossRef]
- Rillig, M.C. Arbuscular mycorrhizae and terrestrial ecosystems processes. Ecol. Lett. 2004, 7, 740–754. [Google Scholar] [CrossRef]
- Gianinazzi, S.; Gollotte, A.; Binet, M.N.; van Tuinen, D.; Redecker, D.; Wipf, D. Agroecology: The key role of arbuscular mycorrhizas in ecosystem services. Mycorrhiza 2010, 20, 519–530. [Google Scholar] [CrossRef] [PubMed]
- Lundgren, J.G.; Fergen, J.K. Predator community structure and trophic linkage strength to a focal prey. Mol. Ecol. 2014, 23, 3790–3798. [Google Scholar] [CrossRef] [PubMed]
- White, S.S.; Renner, K.A.; Menalled, F.D.; Landis, D.A. Feeding preferences of weed seed predators and effect on weed emergence. Weed Sci. 2007, 55, 606–612. [Google Scholar] [CrossRef]
- Shearin, A.F.; Reberg-Horton, S.C.; Gallandt, E.R. Direct effects of tillage on the activity density of ground beetle (Coleoptera: Carabidae) weed seed predators. Environ. Entomol. 2007, 36, 1140–1146. [Google Scholar] [CrossRef] [PubMed]
- Lundgren, J.G.; Shaw, J.T.; Zaborski, E.R.; Eastman, C.E. The influence of organic transition systems on beneficial ground-dwelling arthropods and predation of insects and weed seeds. Renew. Agric. Food Syst. 2006, 21, 227–237. [Google Scholar] [CrossRef]
- Chee-Sanford, J.C.; Williams II, M.M.; Davis, A.S.; Sims, G.K. Do microorganisms influence seed-bank dynamics? Weed Sci. 2009, 54, 575–587. [Google Scholar] [CrossRef]
- Lundgren, J.G.; Lehman, R.M. Bacterial gut symbionts contribute to seed digestion in an omnivorous beetle. PLoS One 2010, 5, e10831. [Google Scholar] [CrossRef] [PubMed]
- Kremer, R.J.; Li, J. Developing weed-suppressive soils through improved soil quality management. Soil Tillage Res. 2003, 72, 193–202. [Google Scholar] [CrossRef]
- Van Elsas, J.D.; Garbeva, P.; Salles, J.F. Effects of agronomic measures on the microbial diversity of soils as related to the suppression of soil-borne pathogens. Biodegradation 2002, 13, 29–40. [Google Scholar] [CrossRef] [PubMed]
- Garbeva, P.; van Veen, J.A.; van Elsas, J.D. Microbial diversity in soil: Selection of microbial populations by plant and soil type and implications for disease suppressiveness. Ann. Rev. Phytopathol. 2004, 42, 243–270. [Google Scholar] [CrossRef]
- Zahir, A.M.; Frankenberger, W.T. Plant growth promoting rhizobacteria: Applications and perspectives in agriculture. Adv. Agron. 2004, 81, 97–168. [Google Scholar]
- Kloepper, J.W.; Zablotowicz, R.M.; Tipping, E.M.; Lifshitz, R. Plant growth promotion mediated by bacterial rhizospheres colonizers. In The Rhizosphere and Plant Growth; Kleister, D.L., Cregan, P.B., Eds.; Kluwer: Dordrecht, The Netherland, 1991; pp. 315–326. [Google Scholar]
- Lutenberg, B.; Kamilova, F. Plant growth promoting rhizobacteria. Ann. Rev. Microbiol. 2009, 63, 541–556. [Google Scholar] [CrossRef]
- Sturz, A.V.; Christie, B.R.; Nowak, J. Bacterial endophytes: Potential role in developing sustainable systems of crop production. Crit. Rev. Plant Sci. 2000, 19, 1–30. [Google Scholar] [CrossRef]
- Harman, G.E.; Howell, C.R.; Viterbo, A.; Chet, I.; Lorito, M. Trichoderma species—Opportunistic, avirulent plant symbionts. Nat. Rev. Microbiol. 2004, 2, 43–56. [Google Scholar] [CrossRef] [PubMed]
- Taghavi, S.; van der Lelie, D.; Hoffman, A.; Zhang, Y.-B.; Walla, M.D.; Vangronsveld, J.; Newman, L.; Monchy, S. Genome sequence of the plant growth promoting endophytic bacterium Enterobacter sp. 638. PLoS Genet. 2010, 6, e1000943. [Google Scholar] [CrossRef] [PubMed]
- Haas, D.; Défago, G. Biological control of soil-borne pathogens by fluorescent pseudomonads. Nat. Rev. Microbiol. 2005, 3, 307–319. [Google Scholar] [CrossRef] [PubMed]
- Vallad, G.E.; Goodman, R.M. Systemic acquired resistance and induced systemic resistance in conventional agriculture. Crop Sci. 2004, 44, 1920–1934. [Google Scholar] [CrossRef]
- Compant, S.; Duffy, B.; Nowak, J.; Clément, C.; Barka, E.A. Use of plant growth-promoting bacteria for biocontrol of plant diseases: Principles, mechanisms of action, and future prospects. Appl. Environ. Microbiol. 2005, 71, 4951–4959. [Google Scholar] [CrossRef] [PubMed]
- Van Loon, L.C.; Bakker, P.A.H.M.; Pieterse, C.M.J. Systemic resistance induced by rhizosphere bacteria. Ann. Rev. Phytopathol. 1998, 36, 453–483. [Google Scholar] [CrossRef]
- Vessey, J.K. Plant growth promoting rhizobacteria as biofertilizers. Plant Soil 2003, 255, 571–586. [Google Scholar] [CrossRef]
- Glick, B.R.; Todorovic, B.; Czarny, J.; Cheng, Z.; Duan, J.; McConkey, B. Promotion of plant growth by bacterial ACC deaminase. Crit. Rev. Plant Sci. 2007, 26, 227–242. [Google Scholar] [CrossRef]
- Kim, Y.C.; Leveau, J.; Gardener, B.B.M.; Pierson, E.A.; Pierson, L.S.; Ryu, C.-M. The multifactorial basis for plant health promotion by plant-associated bacteria. Appl. Environ. Microbiol. 2011, 77, 1548–1555. [Google Scholar] [CrossRef] [PubMed]
- Anaya, A.L. Allelopathy as a tool in the management of biotic resources in agroecosystems. Crit. Rev. Plant Sci. 1999, 18, 697–739. [Google Scholar] [CrossRef]
- Kloepper, J.W.; Hu, C.-H.; Burkett-Cadena, M.; Liu, K.; Xu, J.; McInroy, J. Increased populations of deleterious fluorescent pseudomonads colonizing rhizomes of leatherleaf fern (Rumohra adiantiformis) and expression of symptoms of fern distortion syndrome after application of benlate systemic fungicide. Appl. Soil Ecol. 2012, 61, 236–246. [Google Scholar] [CrossRef]
- Nehl, D.B.; Allen, S.J.; Brown, J.F. Deleterious rhizosphere bacteria: An integrating perspective. Appl. Soil Ecol. 1996, 5, 1–20. [Google Scholar] [CrossRef]
- Kremer, R.J. Deleterious rhizobacteria. In Plant-Associated Bacteria; Gnanamanickam, S.S., Ed.; Springer: Dordrecht, The Netherland, 2006; pp. 335–357. [Google Scholar]
- Badri, D.V.; Vivanco, J.M. Regulation and function of root exudates. Plant Cell Environ. 2009, 32, 666–681. [Google Scholar] [CrossRef] [PubMed]
- Bais, H.P.; Weir, T.L.; Perry, L.G.; Gilroy, S.; Vivanco, J.M. The role of root exudates in rhizosphere interactions with plants and other organisms. Ann. Rev. Plant Biol. 2006, 57, 233–266. [Google Scholar] [CrossRef]
- Kowalchuk, G.A.; Buma, D.S.; de Boer, W.; Klinkhamer, P.G.; van Veen, J.A. Effects of above-ground plant species composition and diversity on the diversity of soil-borne microorganisms. Antonie Leeuwenhoek 2002, 81, 509–520. [Google Scholar] [CrossRef] [PubMed]
- Högberg, M.N.; Högberg, P.; Myrold, D.D. Is microbial community composition in boreal forest soils determined by pH, C-to-N ratio, the trees, or all three? Oecologia 2007, 150, 590–601. [Google Scholar] [CrossRef] [PubMed]
- Micallef, S.A.; Shiaris, M.P.; Colón-Carmona, A. Influence of arabidopsis thaliana accessions on rhizobacterial communities and natural variation in root exudates. J. Exp. Bot. 2009, 60, 1729–1742. [Google Scholar] [CrossRef] [PubMed]
- Grayston, S.J.; Wang, S.; Campbell, C.D.; Edwards, A.C. Selective influence of plant species on microbial diversity in the rhizosphere. Soil Biol. Biochem. 1998, 30, 369–378. [Google Scholar] [CrossRef]
- Kuklinsky-Sobral, J.; Araújo, W.L.; Mendes, R.; Geraldi, I.O.; Pizzirani-Kleiner, A.A.; Azevedo, J.L. Isolation and characterization of soybean—Associated bacteria and their potential for plant growth promotion. Environ. Microbiol. 2004, 6, 1244–1251. [Google Scholar] [CrossRef] [PubMed]
- Salles, J.F.; van Veen, J.A.; van Elsas, J.D. Multivariate analyses of Burkholderia species in soil: Effect of crop and land use history. Appl. Environ. Microbiol. 2004, 70, 4012–4020. [Google Scholar] [CrossRef] [PubMed]
- Badri, D.V.; Loyola-Vargas, V.M.; Broeckling, C.D.; De-la-Peña, C.; Jasinski, M.; Santelia, D.; Martinoia, E.; Sumner, L.W.; Banta, L.M.; Stermitz, F. Altered profile of secondary metabolites in the root exudates of Arabidopsis ATP-binding cassette transporter mutants. Plant Physiol. 2008, 146, 762–771. [Google Scholar] [CrossRef] [PubMed]
- Xie, X.; Zhang, H.; Paré, P.W. Sustained growth promotion in arabidopsis with long-term exposure to the beneficial soil bacterium Bacillus subtilis (gb03). Plant Signal. Behav. 2009, 4, 948–953. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Xie, X.; Kim, M.S.; Kornyeyev, D.A.; Holaday, S.; Pare, P.W. Soil bacteria augment Arabidopsis photosynthesis by decreasing glucose sensing and abscisic acid levels in planta. Plant J. 2008, 56, 264–273. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Kim, M.-S.; Sun, Y.; Dowd, S.E.; Shi, H.; Paré, P.W. Soil bacteria confer plant salt tolerance by tissue-specific regulation of the sodium transporter HKT1. Mol. Plant-Microbe Interact. 2008, 21, 737–744. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Sun, Y.; Xie, X.; Kim, M.-S.; Dowd, S.E.; Paré, P.W. A soil bacterium regulates plant acquisition of iron via deficiency-inducible mechanisms. Plant J. 2009, 58, 568–577. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Murzello, C.; Sun, Y.; Kim, M.-S.; Xie, X.; Jeter, R.M.; Zak, J.C.; Dowd, S.E.; Paré, P.W. Choline and osmotic-stress tolerance induced in Arabidopsis by the soil microbe Bacillus subtilis (GB03). Mol. Plant-Microbe Interact. 2010, 23, 1097–1104. [Google Scholar] [CrossRef] [PubMed]
- Adesemoye, A.; Torbert, H.; Kloepper, J. Enhanced plant nutrient use efficiency with PGPR and AMF in an integrated nutrient management system. Can. J. Microbiol. 2008, 54, 876–886. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Subler, S.; Edwards, C.A. Effects of agricultural biostimulants on soil microbial activity and nitrogen dynamics. Appl. Soil Ecol. 2002, 19, 249–259. [Google Scholar] [CrossRef]
- Kinnersley, A.M. The role of phytochelates in plant growth and productivity. Plant Growth Regul. 1993, 12, 207–218. [Google Scholar] [CrossRef]
- Subler, S.; Dominguez, J.; Edwards, C.A. Assessing biological activity of agricultural biostimulants: Bioassays for plant growth regulators in three soil additives. Commun. Soil Sci. Plant Anal. 1998, 29, 859–866. [Google Scholar] [CrossRef]
- Batie, S.; Nowak, P.; Schnepf, M. Taking Conservation Seriously as a Wicked Problem. Managing Agricultural Landscapes for Environmental Quality II. Achieving More Effective Conservation; Soil and Water Conservation Society: Ankeny, IA, USA, 2010; pp. 143–155. [Google Scholar]
- Tate, R., III. Soil Microbiology; John Wiley & Sons: New York, NY, USA, 2000. [Google Scholar]
- Fierer, N.; Jackson, R.B. The diversity and biogeography of soil bacterial communities. Proc. Natl. Acad. Sci. USA 2006, 103, 626–631. [Google Scholar] [CrossRef] [PubMed]
- Lauber, C.L.; Hamady, M.; Knight, R.; Fierer, N. Pyrosequencing-based assessment of soil pH as a predictor of soil bacterial community structure at the continental scale. Appl. Environ. Microbiol. 2009, 75, 5111–5120. [Google Scholar] [CrossRef] [PubMed]
- Rousk, J.; Bååth, E.; Brookes, P.C.; Lauber, C.L.; Lozupone, C.; Caporaso, J.G.; Knight, R.; Fierer, N. Soil bacterial and fungal communities across a pH gradient in an arable soil. ISME J. 2010, 4, 1340–1351. [Google Scholar] [CrossRef] [PubMed]
- Jansa, J.; Wiemken, A.; Frossard, E. The Effects of Agricultural Practices on Arbuscular Mycorrhizal Fungi; Geological Society of London: London, UK, 2006. [Google Scholar]
- Moonen, A.; Barberi, P. Functional biodiversity: An agroecosystem approach. Agric. Ecosyst. Environ. 2008, 127, 7–21. [Google Scholar] [CrossRef]
- Dias, T.; Dukes, A.; Antunes, P.M. Accounting for soil biotic effects on soil health and crop productivity in the design of crop rotations. J. Sci. Food Agric. 2014. [Google Scholar] [CrossRef]
- Young, I.; Ritz, K. Tillage, habitat space and function of soil microbes. Soil Tillage Res. 2000, 53, 201–213. [Google Scholar] [CrossRef]
- Kladivko, E.J. Tillage systems and soil ecology. Soil Tillage Res. 2001, 61, 61–76. [Google Scholar] [CrossRef]
- Köhl, L.; Oehl, F.; van der Heijden, M.G.A. Agricultural practices indirectly influence plant productivity and ecosystem services through effects on soil biota. Ecol. Appl. 2014, 24, 1842–1853. [Google Scholar] [CrossRef]
- Triplett, G.B.; Dick, W.A. No-tillage crop production: A revolution in agriculture! Agron. J. 2008, 100, S153–S165. [Google Scholar] [CrossRef]
- Helgason, T.; Daniell, T.J.; Husband, R.; Fitter, A.H.; Young, J.P.W. Ploughing up the wood-wide web? Nature 1998. [Google Scholar] [CrossRef]
- Bailey, V.L.; Smith, J.L.; Bolton, H. Fungal-to-bacterial ratios in soil investigated for enhanced C sequestration. Soil Biol. Biochem. 2002, 34, 997–1007. [Google Scholar] [CrossRef]
- Helgason, B.; Walley, F.; Germida, J. No-till soil management increases microbial biomass and alters community profiles in soil aggregates. Appl. Soil Ecol. 2010, 46, 390–397. [Google Scholar] [CrossRef]
- Schippers, B.; Bakker, A.W.; Bakker, P.A. Interactions of deleterious and beneficial rhizosphere microorganisms and the effect of cropping practices. Ann. Rev. Phytopathol. 1987, 25, 339–358. [Google Scholar] [CrossRef]
- Davinic, M.; Moore-Kucera, J.; Acosta-Martinez, V.; Zak, J.; Allen, V. Soil fungal distribution and functionality as affected by grazing and vegetation components of integrated crop–livestock agroecosystems. Appl. Soil Ecol. 2013, 66, 61–70. [Google Scholar] [CrossRef]
- Acosta-Martinez, V.; Lascano, R.; Calderon, F.; Booker, J.D.; Zobeck, T.M.; Upchurch, D.R. Dryland cropping systems influence the microbial biomass and enzyme activities in a semiarid sandy soil. Biol. Fertil. Soils 2011, 47, 655–667. [Google Scholar] [CrossRef]
- Reeves, D.W. The role of soil organic matter in maintaining soil quality in continuous cropping systems. Soil Tillage Res. 1997, 43, 131–167. [Google Scholar] [CrossRef]
- Schipanski, M.E.; Barbercheck, M.; Douglas, M.R.; Finney, D.M.; Haider, K.; Kaye, J.P.; Kemanian, A.R.; Mortensen, D.A.; Ryan, M.R.; Tooker, J.; et al. A framework for evaluating ecosystem services provided by cover crops in agroecosystems. Agric. Syst. 2014, 125, 12–22. [Google Scholar] [CrossRef]
- Boswell, E.P.; Koide, R.T.; Shumway, D.L.; Addy, H.D. Winter wheat cover cropping, VA mycorrhizal fungi and maize growth and yield. Agric. Ecosyst. Environ. 1998, 67, 55–65. [Google Scholar] [CrossRef]
- White, C.M.; Weil, R.R. Forage radish and cereal rye cover crop effects on mycorrhizal fungus colonization of maize roots. Plant Soil 2010, 328, 507–521. [Google Scholar] [CrossRef]
- Lehman, R.M.; Taheri, W.I.; Osborne, S.L.; Buyer, J.S.; Douds, D.D., Jr. Fall cover cropping can increase arbuscular mycorrhizae in soils supporting intensive agricultural production. Appl. Soil Ecol. 2012, 61, 300–304. [Google Scholar] [CrossRef]
- Horst, W.J.; Kamh, M.; Jibrin, J.M.; Chude, V.O. Agronomic measures for increasing P availability to crops. Plant Soil 2001, 237, 211–223. [Google Scholar] [CrossRef]
- Dabney, S.M.; Delgado, J.A.; Reeves, D.W. Using winter cover crops to improve soil and water quality. Commun. Soil Sci. Plant Anal. 2001, 32, 1221–1250. [Google Scholar] [CrossRef]
- Wilhelm, W.W.; Johnson, J.M.; Karlen, D.L.; Lightle, D.T. Corn stover to sustain soil organic carbon further constrains biomass supply. Agron. J. 2007, 99, 1665–1667. [Google Scholar] [CrossRef]
- Cotton, J.; Acosta-Martínez, V.; Moore-Kucera, J.; Burow, G. Early changes due to sorghum biofuel cropping systems in soil microbial communities and metabolic functioning. Biol. Fertil. Soils 2012, 49, 403–413. [Google Scholar] [CrossRef]
- Lehman, R.M.; Ducey, T.F.; Jin, V.L.; Acosta-Martinez, V.; Ahlschwede, C.M.; Jeske, E.S.; Drijber, R.A.; Cantrell, K.B.; Frederick, J.R.; Fink, D.M. Soil microbial community response to corn stover harvesting under rain-fed, no-till conditions at multiple US locations. BioEnergy Res. 2014, 7, 540–550. [Google Scholar] [CrossRef]
- Bullock, D.G. Crop rotation. Crit. Rev. Plant Sci. 1992, 11, 309–326. [Google Scholar] [CrossRef]
- McDaniel, M.D.; Grandy, A.S.; Tiemann, L.K.; Weintraub, M.N. Crop rotation complexity regulates the decomposition of high and low quality residues. Soil Biol. Biochem. 2014, 78, 243–254. [Google Scholar] [CrossRef]
- Bunemann, E.K.; Bossio, D.A.; Smithson, P.C.; Frossard, E.; Oberson, A. Microbial community composition and substrate use in a highly weathered soil as affected by crop rotation and P fertilization. Soil Biol. Biochem. 2004, 36, 889–901. [Google Scholar] [CrossRef]
- Oehl, F.; Sieverding, E.; Ineichen, K.; Mäder, P.; Boller, T.; Wiemken, A. Impact of land use intensity on the species diversity of arbuscular mycorrhizal fungi in agroecosystems of central Europe. Appl. Environ. Microbiol. 2003, 69, 2816–2824. [Google Scholar] [CrossRef] [PubMed]
- Lupwaya, N.Z.; Blackshaw, R.E. Soil microbial properties in Bt (Bacillus thuringiensis) corn cropping systems. Appl. Soil Ecol. 2013, 63, 127–133. [Google Scholar] [CrossRef]
- Barazani, O.; Friedman, J. Allelopathic bacteria and their impact on higher plants. Crit. Rev. Microbiol. 2001, 27, 41–45. [Google Scholar] [CrossRef] [PubMed]
- Bunemann, E.; Smithson, P.C.; Jama, B.; Frossard, E.; Oberson, A. Maize productivity and nutrient dynamics in maize-fallow rotations in western Kenya. Plant Soil 2004, 264, 195–208. [Google Scholar] [CrossRef]
- Rosendahl, S.; Matzen, H.B. Genetic structure of arbuscular mycorrhizal populations in fallow and cultivated soils. New Phytol. 2008, 179, 1154–1161. [Google Scholar] [CrossRef] [PubMed]
- Wetterauer, D.; Killorn, R. Fallow-and flooded-soil syndromes: Effects on crop production. J. Prod. Agric. 1996, 9, 39–41. [Google Scholar] [CrossRef]
- Anderson, R.L. Possible causes of dry pea synergy to corn. Weed Technol. 2012, 26, 438–442. [Google Scholar] [CrossRef]
- Calbrix, R.; Barray, S.; Chabrerie, O.; Fourrie, L.; Laval, K. Impact of organic amendments on the dynamics of soil microbial biomass and bacterial communities in cultivated land. Appl. Soil Ecol. 2007, 35, 511–522. [Google Scholar] [CrossRef]
- Chaudhry, V.; Rehman, A.; Mishra, A.; Chauhan, P.S.; Nautiyal, C.S. Changes in bacterial community structure of agricultural land due to long-term organic and chemical amendments. Microb. Ecol. 2012, 64, 450–460. [Google Scholar] [CrossRef] [PubMed]
- Sun, H.Y.; Deng, S.P.; Raun, W.R. Bacterial community structure and diversity in a century-old manure-treated agroecosystem. Appl. Environ.Microbiol. 2004, 70, 5868–5874. [Google Scholar] [CrossRef] [PubMed]
- Saison, C.; Degrange, V.; Oliver, R.; Millard, P.; Commeaux, C.; Montange, D.; le Roux, X. Alteration and resilience of the soil microbial community following compost amendment: Effects of compost level and compost-borne microbial community. Environ. Microbiol. 2006, 8, 247–257. [Google Scholar] [CrossRef] [PubMed]
- Kallenbach, C.; Grandy, A.S. Controls over soil microbial biomass responses to carbon amendments in agricultural systems: A meta-analysis. Agric. Ecosyst. Environ. 2011, 144, 241–252. [Google Scholar] [CrossRef]
- Cheng, Y.; Ishimoto, K.; Kuriyama, Y.; Osaki, M.; Ezawa, T. Ninety-year-, but not single, application of phosphorus fertilizer has a major impact on arbuscular mycorrhizal fungi communities. Plant Soil 2012, 365, 397–407. [Google Scholar] [CrossRef]
- Geisseler, D.; Scow, K.M. Long-term effects of mineral fertilizers on soil microorganisms: A review. Soil Biol. Biochem. 2014, 75, 54–63. [Google Scholar] [CrossRef]
- Carlisle, S.; Trevors, J. Glyphosate in the environment. Water Air Soil Pollut. 1988, 39, 409–420. [Google Scholar]
- Means, N.E.; Kremer, R.J.; Ramsier, C. Effects of glyphosate and foliar amendments on activity of microorganisms in the soybean rhizosphere. J. Environ. Sci. Health Part B 2007, 42, 125–132. [Google Scholar] [CrossRef]
- Wardle, D.; Parkinson, D. Influence of the herbicide glyphosate on soil microbial community structure. Plant Soil 1990, 122, 29–37. [Google Scholar] [CrossRef]
- Haney, R.; Senseman, S.; Hons, F.; Zuberer, D. Effect of glyphosate on soil microbial activity and biomass. Weed Sci. 2009, 48, 89–93. [Google Scholar]
- Liphadzi, K.B.; Al-Khatib, K.; Bensch, C.N.; Stahlman, P.W.; Dille, J.A.; Todd, T.; Rice, C.W.; Horak, M.J.; Head, G. Soil microbial and nematode communities as affected by glyphosate and tillage practices in a glyphosate-resistant cropping system. Weed Sci. 2005, 53, 536–545. [Google Scholar] [CrossRef]
- Mijangos, I.; Becerril, J.M.; Albizu, I.; Epelde, L.; Garbisu, C. Effects of glyphosate on rhizosphere soil microbial communities under two different plant compositions using cultivation-dependent and -independent methodologies. Soil Biol. Biochem. 2009, 41, 505–513. [Google Scholar] [CrossRef]
- Zablotowicz, R.M.; Reddy, K.N. Impact of glyphosate on the Bradyrhizobium japonicum symbiosis with glyphosate-resistant trangenic soybean: A minireview. J. Environ. Qual. 2004, 33, 825–831. [Google Scholar] [CrossRef] [PubMed]
- Johal, G.; Huber, D. Glyphosate effects on diseases of plants. Eur. J. Agron. 2009, 31, 144–152. [Google Scholar] [CrossRef]
- Kremer, R.J.; Means, N.E. Glyphosate and glyphosate-resistant crop interactions with rhizosphere microorganisms. Eur. J. Agron. 2009, 31, 153–161. [Google Scholar] [CrossRef]
- Duke, S.O.; Lydon, J.; Koskinen, W.C.; Moorman, T.B.; Chaney, R.L.; Hammerschmidt, R. Glyphosate effects on plant mineral nutrition, crop rhizosphere microbiota, and plant disease in glyphosate-resistant crops. J. Agric. Food Chem. 2012, 60, 10375–10397. [Google Scholar] [CrossRef] [PubMed]
- Marshall, E.; Brown, V.; Boatman, N.; Lutman, P.; Squire, G.; Ward, L. The role of weeds in supporting biological diversity within crop fields. Weed Res. 2003, 43, 77–89. [Google Scholar] [CrossRef]
- Greaves, M.P.; Sargent, J.A. Herbicide-induced microbial invasion of plant roots. Weed Sci. 1986, 34, 50–53. [Google Scholar]
- Kuklinsky-Sobral, J.; Welingon, L.A.; Mendes, R.; Pizzirani-Kleiner, A.A.; Azavedo, J.L. Isolation and characterization of endophytic bacteria from soybean (Glycine max) grown in soil treated with glyphosate herbicide. Plant Soil 2005, 273, 91–99. [Google Scholar] [CrossRef]
- Ipsilantis, I.; Samourelis, C.; Karpouzas, D.G. The impact of biological pesticides on arbuscular mycorrhizal fungi. Soil Biol. Biochem. 2012, 45, 147–155. [Google Scholar] [CrossRef]
- Griffiths, B.S.; Philippot, L. Insights into the resistance and resilience of the soil microbial community. FEMS Microbiol. Rev. 2013, 37, 112–129. [Google Scholar] [PubMed]
- Morales, S.E.; Holben, W.E. Linking bacterial identities and ecosystem processes: Can “omic” analyses be more than the sum of their parts? FEMS Microbiol. Ecol. 2011, 75, 2–16. [Google Scholar] [CrossRef] [PubMed]
- Acosta-Martinez, V.; Dowd, S.E.; Sun, Y.; Allen, V.G. Tag-encoded pyrosequencing analysis of bacterial diversity in a single soil type as affected by management and land use. Soil Biol. Biochem. 2008, 40, 2762–2770. [Google Scholar] [CrossRef]
- Acosta-Martinez, V.; Dowd, S.E.; Sun, Y.; Wester, D.; Allen, V.G. Pyrosequencing analysis for characterization of soil bacterial populations as affected by an integrated livestock-cotton production system. Appl. Soil Ecol. 2010, 45, 13–25. [Google Scholar] [CrossRef]
- Sugiyama, A.; Vivanco, J.M.; Jayanty, S.S.; Manter, D.K. Pyrosequencing assessment of soil microbial communities in organic and conventional potato farms. Plant Discuss. 2010. [Google Scholar] [CrossRef]
- Douds, D., Jr.; Nagahashi, G.; Pfeffer, P.; Reider, C.; Kayser, W. On-farm production of AM fungus inoculum in mixtures of compost and vermiculite. Bioresour. Technol. 2006, 97, 809–818. [Google Scholar] [CrossRef] [PubMed]
- Orwin, K.H.; Wardle, D.A.; Greenfield, L.G. Ecological consequences of carbon substrate identity and diversity in a laboratory study. Ecology 2006, 87, 580–593. [Google Scholar] [CrossRef] [PubMed]
- Loreau, M.; Naeem, S.; Inchausti, P.; Bengtsson, J.; Grime, J.; Hector, A.; Hooper, D.; Huston, M.; Raffaelli, D.; Schmid, B. Biodiversity and ecosystem functioning: Current knowledge and future challenges. Science 2001, 294, 804–808. [Google Scholar] [CrossRef] [PubMed]
- Pennanen, T.; Caul, S.; Daniell, T.; Griffiths, B.; Ritz, K.; Wheatley, R. Community-level responses of metabolically-active soil microorganisms to the quantity and quality of substrate inputs. Soil Biol. Biochem. 2004, 36, 841–848. [Google Scholar] [CrossRef]
- Larkin, R.P.; Honeycutt, C.W. Effects of different 3-year cropping systems on soil microbial communities and Rhizoctonia diseases of potato. Phytopathology 2006, 96, 68–79. [Google Scholar] [CrossRef] [PubMed]
- Ochiai, N.; Powelson, M.L.; Crowe, F.J.; Dick, R.P. Green manure effects on soil quality in relation to suppression of Verticillium wilt of potatoes. Biol. Fertil. Soils 2008, 44, 1013–1023. [Google Scholar] [CrossRef]
- Postma, J.; Schilder, M.T.; Bloem, J.; van Leeuwen-Haagsma, W.K. Soil suppressivenss and functional diversity of the soil microflora in organic farming systems. Soil Biol. Biochem. 2008, 40, 2394–2406. [Google Scholar] [CrossRef]
- Wittebolle, L.; Marzorati, M.; Clement, L.; Balloi, A.; Daffonchio, D.; Heylen, K.; de Vos, P.; Verstraete, W.; Boon, N. Initial community evenness favours functionality under selective stress. Nature 2009, 458, 623–626. [Google Scholar] [CrossRef] [PubMed]
- Tracy, B.F.; Sanderson, M.A. Forage productivity, species evenness, and weed invasion in pasture communities. Agric. Ecosyst. Environ. 2004, 102, 175–183. [Google Scholar] [CrossRef]
- Crowder, D.W.; Northfield, T.D.; Strand, M.R.; Snyder, W.E. Organic agriculture promotes evenness and natural pest control. Nature 2010, 466, 109–112. [Google Scholar] [CrossRef] [PubMed]
- Bossio, D.A.; Scow, K.M.; Gunapala, N.; Graham, K.J. Determinants of soil microbial communities: Effects of agricultural management, season, and soil type on phospholipid fatty acid profiles. Microb. Ecol. 1998, 36, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Smith, J.L. Soil quality: The role of microorganisms. In Encyclopedia of Environmental Microbiology; Bitton, G., Ed.; John Wiley and Sons: New York, NY, USA, 2002; pp. 2944–2957. [Google Scholar]
- Kuchenbuch, R.O.; Buczko, U. Re-visiting potassium- and phosphate-fertilizer responses in field experiments and soil-test interpretations by means of data mining. J. Plant Nutr. Soil Sci. 2011, 174, 171–185. [Google Scholar] [CrossRef]
- Frossard, E.; Condron, L.M.; Oberson, A.; Sinaj, S.; Fardeau, J.C. Processes governing phosphorus availability in temperate soils. J. Environ. Qual. 2000, 29, 15–23. [Google Scholar] [CrossRef]
- Condron, L.M.; Newman, S. Revisiting the fundamentals of phosphorus fractionation of sediments and soils. J. Soils Sediments 2011, 11, 830–840. [Google Scholar] [CrossRef]
- Hinsinger, P.; Brauman, A.; Devau, N.; Gerard, F.; Jourdan, C.; Laclau, J.-P.; le Cadre, E.; Jaillard, B.; Plassard, C. Acquisition of phosphorus and other poorly mobile nutrients by roots. Where do plant nutrition models fail? Plant Soil 2011, 348, 29–61. [Google Scholar] [CrossRef]
- American Academy of Microbiology (AAM). Incorporating Microbial Processes into Climate Change Models; AAM: Washington, DC, USA, 2011. [Google Scholar]
- Smith, J.L.; Collins, H.P. Managing soil microorganisms and their processes. In Soil Microbiology, Ecology and Biochemistry; Paul, E.A., Ed.; Academic Press: Burlington, MA, USA, 2007; pp. 471–500. [Google Scholar]
- Smith, R.G.; Gross, K.L.; Robertson, G.P. Effects of crop diversity on agroecosystem function: Crop yield response. Ecosystems 2008, 11, 355–366. [Google Scholar] [CrossRef]
- Cook, R.J. Toward cropping systems that enhance productivity and sustainability. Proc. Natl. Acad. Sci. USA 2006, 103, 18389–19384. [Google Scholar] [CrossRef] [PubMed]
- American Academy of Microbiology (AAM). How Microbes Can Feed the World; AAM: Washington, DC, USA, 2013; p. 33. [Google Scholar]
- Gupta, V.V.S.R.; Rovira, A.D.; Roger, D.K. Principles and management of soil biological factors for sustainable rainfed farming systems. In Rainfed Farming Systems; Tow, P., Cooper, I., Partridge, I., Birch, C., Eds.; Springer: Dordrecht, The Netherland, 2011; pp. 149–184. [Google Scholar]
- Liebig, M.; Carpenter-Boggs, L.; Johnson, J.; Wright, S.; Barbour, N. Cropping system effects on soil biological characteristics in the Great Plains. Renew. Agric. Food Syst. 2006, 21, 36–48. [Google Scholar] [CrossRef]
- Kowalchuk, G.A.; Bruinsma, M.; van Veen, J.A. Assessing responses of soil microorganisms to GM plants. Trends Ecol. Evol. 2003, 18, 403–410. [Google Scholar] [CrossRef]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lehman, R.M.; Cambardella, C.A.; Stott, D.E.; Acosta-Martinez, V.; Manter, D.K.; Buyer, J.S.; Maul, J.E.; Smith, J.L.; Collins, H.P.; Halvorson, J.J.; et al. Understanding and Enhancing Soil Biological Health: The Solution for Reversing Soil Degradation. Sustainability 2015, 7, 988-1027. https://doi.org/10.3390/su7010988
Lehman RM, Cambardella CA, Stott DE, Acosta-Martinez V, Manter DK, Buyer JS, Maul JE, Smith JL, Collins HP, Halvorson JJ, et al. Understanding and Enhancing Soil Biological Health: The Solution for Reversing Soil Degradation. Sustainability. 2015; 7(1):988-1027. https://doi.org/10.3390/su7010988
Chicago/Turabian StyleLehman, R. Michael, Cynthia A. Cambardella, Diane E. Stott, Veronica Acosta-Martinez, Daniel K. Manter, Jeffrey S. Buyer, Jude E. Maul, Jeffrey L. Smith, Harold P. Collins, Jonathan J. Halvorson, and et al. 2015. "Understanding and Enhancing Soil Biological Health: The Solution for Reversing Soil Degradation" Sustainability 7, no. 1: 988-1027. https://doi.org/10.3390/su7010988
APA StyleLehman, R. M., Cambardella, C. A., Stott, D. E., Acosta-Martinez, V., Manter, D. K., Buyer, J. S., Maul, J. E., Smith, J. L., Collins, H. P., Halvorson, J. J., Kremer, R. J., Lundgren, J. G., Ducey, T. F., Jin, V. L., & Karlen, D. L. (2015). Understanding and Enhancing Soil Biological Health: The Solution for Reversing Soil Degradation. Sustainability, 7(1), 988-1027. https://doi.org/10.3390/su7010988






