A Review of Medical Waste Management Systems in the Republic of Korea for Hospital and Medical Waste Generated from the COVID-19 Pandemic
Abstract
:1. Introduction
2. Materials and Methods
3. Results and Discussion
3.1. Definitions and Classification Systems of Medical Waste
3.2. Comparison of Collection, Storage, Transport, and Treatment of Medical Waste
3.2.1. Collection and Storage at Discharge Facilities
3.2.2. Transportation and Outsourced Incineration
3.2.3. Treatment Methods at the Discharge Facility
3.2.4. Treatment of General Medical Waste
3.3. The Korean Medical Waste Management System
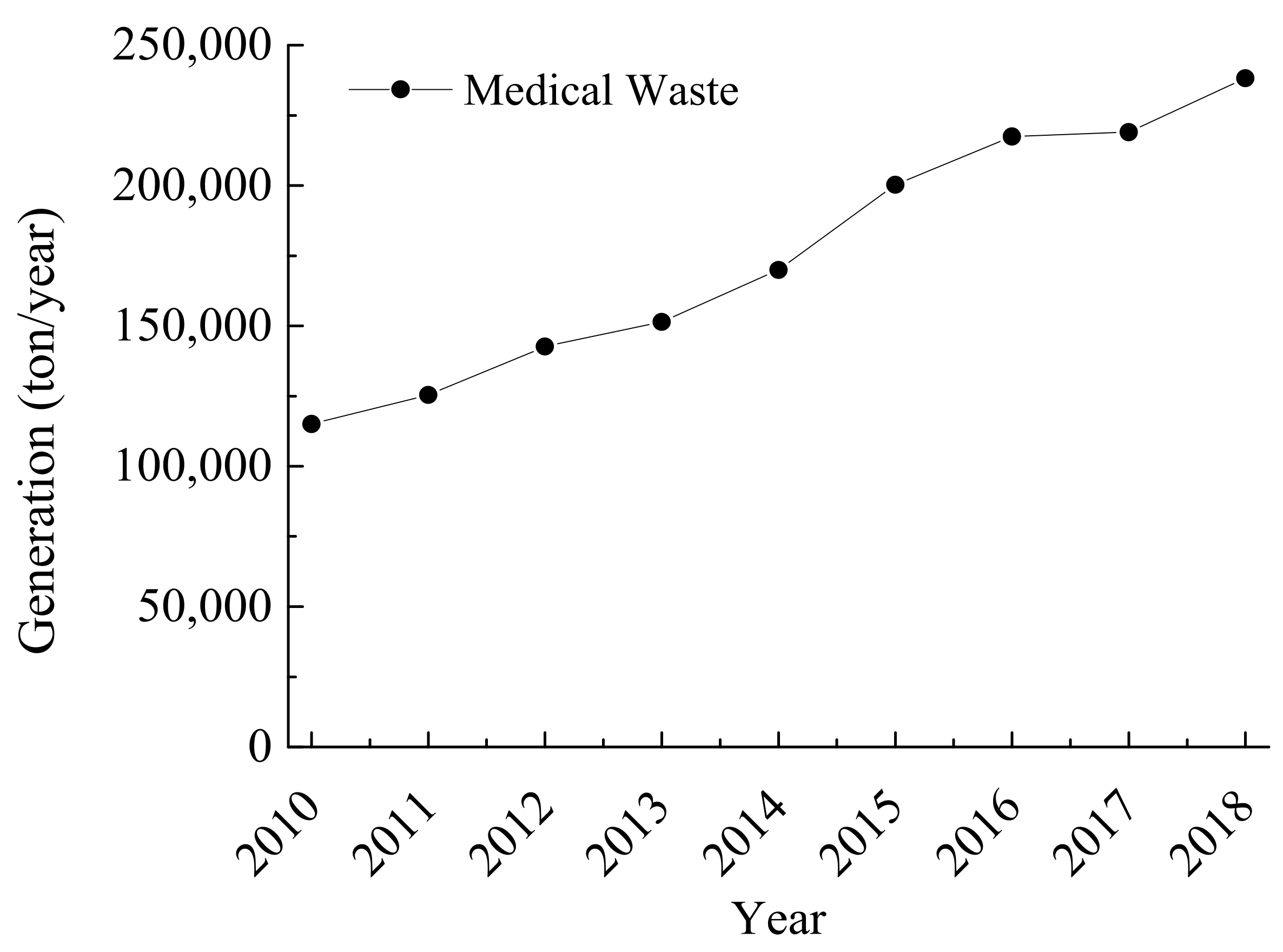
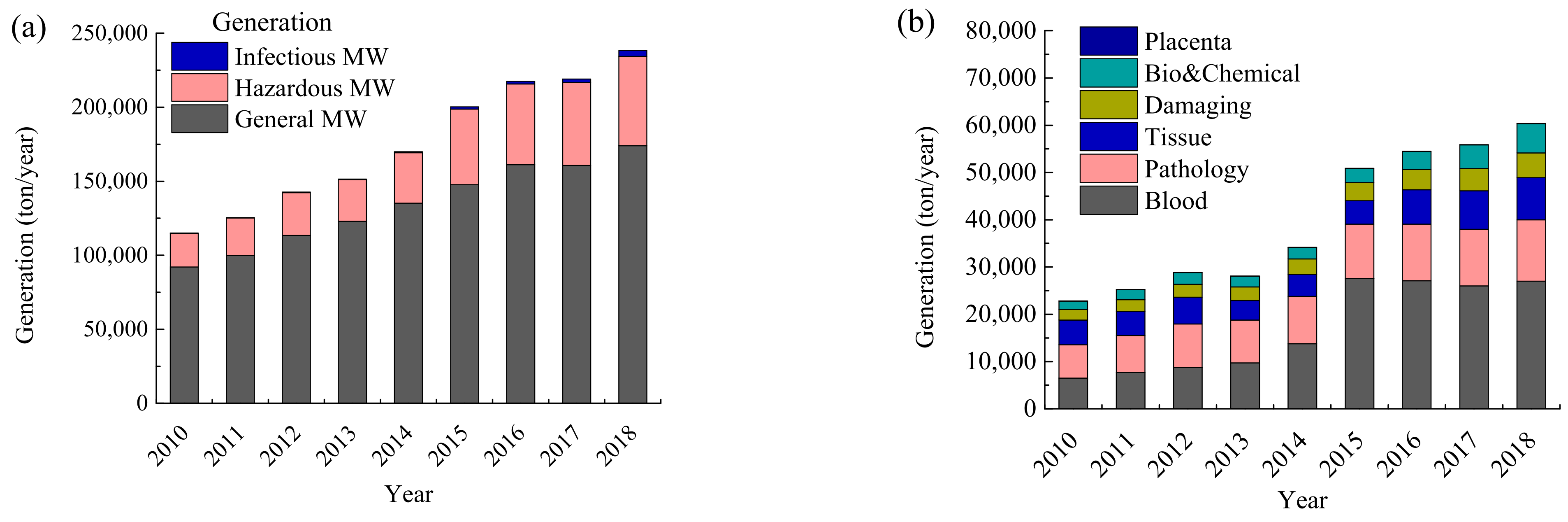
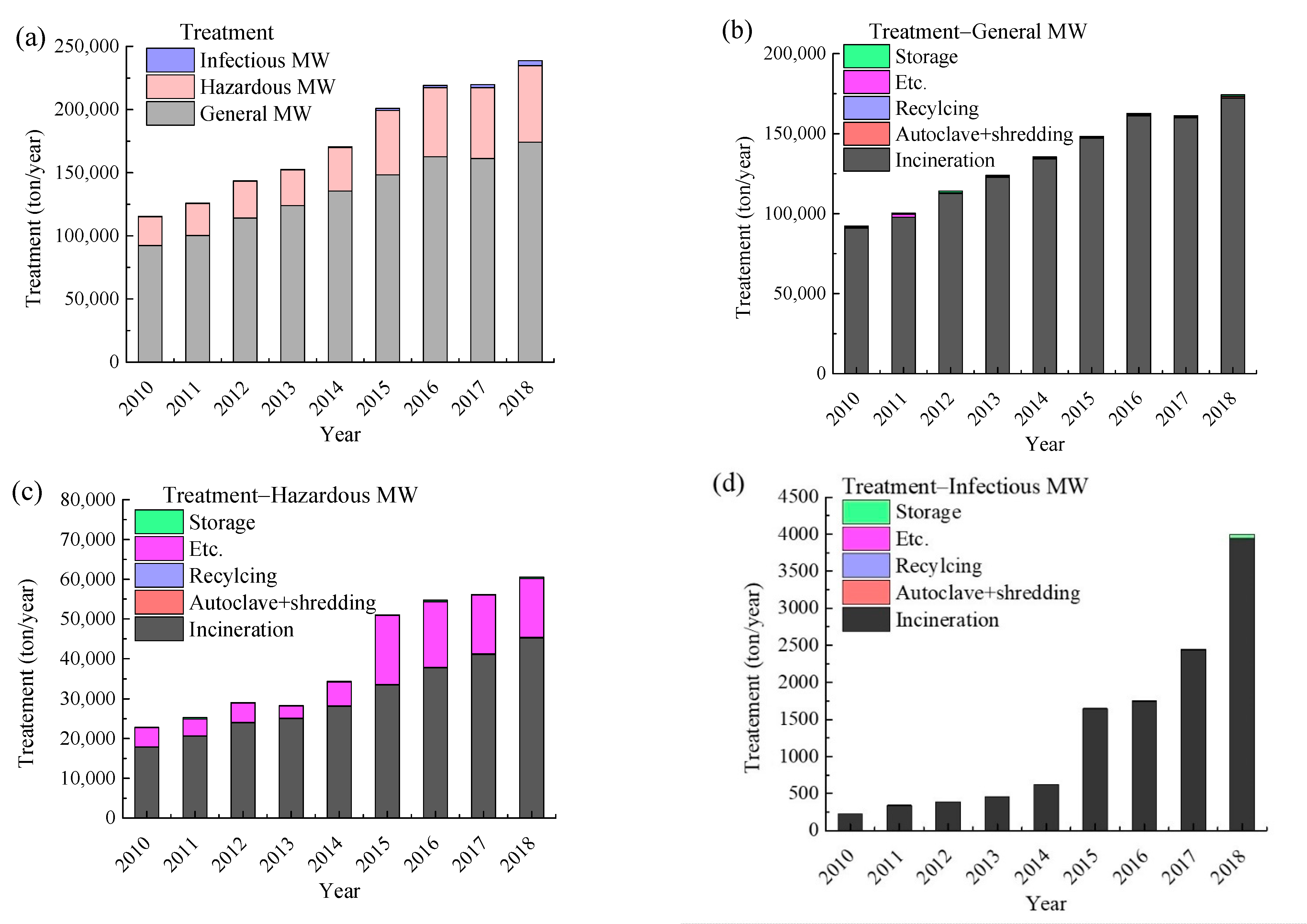
3.3.1. Status of Medical Waste in Korea
Generation and Treatment of Medical Waste in Korea
Medical Waste Treatment Methods and Status in Korea and Other Countries
3.4. Implications and Directions for Medical Waste Management in Korea
3.4.1. Reduction of Generation
Establishment of a Separate Medical Waste Collection System
Review of the Medical Waste Classification System
Efforts to Improve the Accuracy of Generation Statistics
3.4.2. Diversification of Medical Waste Treatment Methods
Sterilization Grinding Treatment
Using Incinerators Not Dedicated to Medical Waste
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Jang, Y.-C.; Lee, C.; Yoon, O.-S.; Kim, H. Medical waste management in Korea. J. Environ. Manag. 2006, 80, 107–115. [Google Scholar] [CrossRef] [PubMed]
- Ghanimeh, S.; Gomez-Sanabria, A.; Tsydenova, N.; Kristina, S.; Maria, I.; Kumar, M. Two-Level Comparison of Waste Management Systems in Low-, Middle-, and High-Income Cities. Environ. Eng. Sci. 2019, 36, 1–15. [Google Scholar] [CrossRef]
- Bungau, S.; Tit, D.M.; Fodor, K.; Cioca, G.; Agop, M.; Iovan, C.; Cseppento, D.C.; Bumbu, A.; Bustea, C. Aspects regarding the pharmaceutical waste management in Romania. Sustainability 2018, 10, 2788. [Google Scholar] [CrossRef] [Green Version]
- Bungau, S.; Suciu, R.; Bumbu, A.; Cioca, G.; Tit, D.M. Study on hospital waste management in medical rehabilitation clinical hospital, Baile Felix. J. Environ. Prot. Ecol. 2015, 16, 980–987. [Google Scholar]
- Bungau, S.; Bungau, C.; Tit, D.M. Studies about last stage of product lifecycle management for a pharmaceutical product. J. Environ. Prot. Ecol. 2015, 16, 56–62. [Google Scholar]
- World Health Organization. Safe Management of Wastes from Health-Care Activities, 2nd ed.; WHO: Geneva, Switzerland, 2014. [Google Scholar]
- United States Environmental Protection Agency. Model Guidelines for State Medical Waste Management. The Council of State Governments. Available online: https://www.epa.gov/rcra/model-guidelines-state-medical-waste-management (accessed on 31 May 2021).
- European Union. Directive 2008/98/EC of the European Parliament and of the Council of 19 November 2008 on Waste and Repealing Certain Directives. Available online: https://eur-lex.europa.eu/legal-content/EN/TXT/?uri=celex%3A32008L0098 (accessed on 11 January 2022).
- Ministry of Environment, Japan. Solid Waste Management and Recycling Technology of Japan: Towards a Sustainable Society. Available online: https://www.env.go.jp/en/recycle/smcs/attach/swmrt.pdf (accessed on 31 May 2021).
- Ministry of Environment, Japan. Waste Management and Public Cleansing Law, Japan. Available online: https://www.env.go.jp/en/laws/recycle/03.pdf (accessed on 31 May 2021).
- World Health Organization. Health-Care Waste. Available online: https://www.who.int/news-room/factsheets/detail/health-care-waste (accessed on 31 May 2021).
- Klangsin, P.; Harding, A.K. Medical Waste Treatment and Disposal Methods Used by Hospitals in Oregon, Washington, and Idaho. J. Air Waste Manag. Assoc. 1998, 48, 516–526. [Google Scholar] [CrossRef] [PubMed]
- Thakur, V.; Ramesh, A. Healthcare waste disposal strategy selection using grey-AHP approach. Benchmarking Int. J. 2017, 24, 735–749. [Google Scholar] [CrossRef]
- United Nations. Agreement Concerning the International Carriage of Dangerous Goods by Road. Available online: https://unece.org/transport/publications/agreement-concerning-international-carriage-dangerous-goods-road-adr-2021 (accessed on 31 May 2021).
- United Kingdom. Environmental protection, England and Wales: The Controlled Waste (England and Wales) Regulations 2012. Available online: https://www.legislation.gov.uk/uksi/2012/811/made (accessed on 31 May 2021).
- United Kingdom Department for Environment Food & Rural Affairs. Guidance on Prioritising Waste Collection Services during Coronavirus (COVID-19) Pandemic. Available online: https://www.gov.uk/government/publications/coronavirus-covid-19-advice-to-local-authorities-on-prioritising-waste-collections/guidance-on-prioritising-waste-collection-services-during-coronavirus-covid-19-pandemic (accessed on 31 May 2021).
- United Kingdom Department for Environment Food & Rural Affairs. Managing Household Waste and Recycling Centres (HWRCs) in England during the Coronavirus (COVID-19) Pandemic. Available online: https://www.gov.uk/government/publications/coronavirus-covid-19-advice-to-local-authorities-on-prioritising-waste-collections/managing-household-waste-and-recycling-centres-hwrcs-in-england-during-the-coronavirus-covid-19-pandemic (accessed on 31 May 2021).
- United States Environmental Protection Agency. Does RCRA Regulate Wastes that may Contain the Virus that Causes COVID-19, Such as Used Medical Equipment or Personal Protective Equipment? Available online: https://www.epa.gov/coronavirus/does-rcra-regulate-wastes-may-contain-virus-causes-covid-19-such-used-medical-equipment (accessed on 31 May 2021).
- Healthcare Environmental Resource Center. Types of Regulated Medical Waste (RMW). Available online: https://www.hercenter.org/rmw/rmwtypes.php (accessed on 31 May 2021).
- Miyazaki, M.; Une, H. Infectious waste management in Japan: A revised regulation and a management process in medical institutions. Waste Manag. 2005, 25, 616–621. [Google Scholar] [CrossRef] [PubMed]
- Umweltbundesamt. Best Practice Municipal Waste Management. 2018. Available online: https://www.umweltbundesamt.de/en/publikationen/best-practice-municipal-waste-management (accessed on 31 May 2021).
- European Environment Agency. Overview of National Waste Prevention Programmes in Europe–Belgium–Flanders, Country Fact Sheet. Available online: https://www.eea.europa.eu/themes/waste/waste-prevention (accessed on 11 January 2022).
- European Union. General Classification and Labelling Requirements for Dangerous Substances and Preparations. Available online: https://ec.europa.eu/environment/archives/dansub/pdfs/annex6_en.pdf (accessed on 11 January 2022).
- Yong, Z.; Gang, X.; Guanxing, W.; Tao, Z.; Dawei, J. Medical waste management in China: A case study of Nanjing. Waste Manag. 2009, 29, 1376–1382. [Google Scholar] [CrossRef] [PubMed]
- Manga, V.E.; Forton, O.T.; Mofor, L.A.; Woodard, R. Health care waste management in Cameroon: A case study from the Southwestern Region. Resour. Conserv. Recycl. 2011, 57, 108–116. [Google Scholar] [CrossRef]
| Category | Republic of Korea [1] | WHO [6] | USA [7] | EU [8] | Japan [9,10] | China [11] |
|---|---|---|---|---|---|---|
| Terminology | Medical Waste | Healthcare Waste | Medical Waste | Healthcare waste | Infectious Waste | Medical Waste |
| Sharps | Sharps | Sharps | Contaminated sharps/Unused sharps | Sharps | Infectious Industrial | Sharps |
| Body parts and organic including blood bags and blood preserves | Body parts and fluids/Blood contaminated/ Pathological test | Pathological | Bulk human blood/Pathological wastes | Body parts and organs including blood bags and blood preserves | Infectious Municipal | Human or animal pathological wastes, tissues, organs, blood, pus, body parts, and fluids |
| Waste whose collection and disposal is subject to special requirements to prevent infections | Infectious | Infectious | Isolation/Cultures and stocks of infectious agents and associated biologicals/animal wastes | Human and Animal Infectious | Infectious Industrial /Infectious Municipal | Infectious |
| Waste whose collection and disposal are not subject to special requirements to prevent infection (e.g., dressings, plaster casts, linen, disposable clothing, diapers contaminated with blood, etc.) | General | Infectious | - | Infectious | Infectious Municipal | Infectious |
| Chemicals consisting of or containing dangerous substances | Biological/ Chemical | Chemical | - | Chemical | Chemical | |
| Chemicals other than those mentioned in 18 01 06 | Biological/ Chemical | Chemical | Small volumes of chemical hazardous waste | Chemical/ Unused hazardous medicines | Chemical | |
| Cytotoxic and cytostatic medicines | Cytotoxic | Antineoplastic drug | Unused hazardous medicines | Chemical | ||
| Chemicals other than those mentioned in 18 01 08 | Pharmaceutical | Small volumes of chemical hazardous waste | Unused non-hazardous medicines | Medical | ||
| Amalgam waste from dental care | Amalgam waste from dental care | - | Amalgam waste from dental care | Amalgam waste from dental care |
| ROK | USA | Germany | UK | Japan | China | |
|---|---|---|---|---|---|---|
| Infectious medical waste | All waste from medical practices for persons isolated to protect others from the infectious diseases specified under Article 2-1 of the Infectious Disease Control and Prevention Act | Isolated waste/infectious pathogens and related organism media | Wastes whose collection and disposal is subject to special requirements to prevent infection | Infectious waste/contaminated infectious clinical waste | Place of discharge: Material discharged after being used for treatment, examination, etc. in infectious disease beds, tuberculosis beds, operating rooms, emergency outpatient rooms, intensive care units, and examination rooms Types of infectious diseases: Class I, II, III, IV, and V Infectious Diseases, Novel Influenza Infection, etc., Designated Infectious Disease, New Infectious Disease, Tuberculosis Examination, etc. under the Act on the Prevention of Infectious Diseases and Medical Care for Patients with Infectious Diseases | Material that is carrying a variety of pathogenic microorganisms and spreading the risk of infectious disease induced (blood, blood products, and objects that are polluted with them; quarantine waste; microbiology laboratory waste; dialysis waste; used surgical operation clothes; infectious organ pieces, blood and anything contaminated with these materials) |
| Tissue waste | Human or animal tissues, organs, body parts, animal cadavers, blood, pus, and blood products (serum, plasma, blood derivatives) | Animal waste | Body parts and organs, including blood bags and blood products | - | Form: Pathological wastes (organs, tissue, skin, etc.) from surgery, etc. | Human or animal pathological waste, including tissues, organs, blood, pus, and body parts and fluids |
| Pathological waste | Culture media used for testing/examination, culture vessels, stored strains, waste test tubes, slides, cover glass, waste media, waste gloves | Pathological waste | - | - | Form: Material used for tests, examinations, etc. related to pathogenic microorganisms (media, laboratory animal cadavers, test tubes, Petri dishes) | Human body waste and medical experimental animal carcasses produced during processes of diagnosis and treatment |
| Injury waste | Injection needles, suture needles, surgical blades, acupuncture needles, dental needles, broken glass of testers | Injury waste/unused injury waste | Pointed or sharp objects | Non-contaminated injury waste | Form: Sharp objects stained with blood (including broken glass fragments, etc.) | Medical sharp waste that can hurt or cut the human body (needles, syringes, broken glass, blades, and other items that could cause a cut or puncture) |
| Biological and chemical waste | Lung vaccines, lung cancer drugs, lung chemotherapy drugs | Anticancer drugs/small amounts of chemically harmful substances | Chemicals/cytotoxic drugs and mitogens consisting of or containing dangerous toxic substances | Cytotoxic and cytostatic waste | - | Medicine waste: Medicine waste of expired, obsolete, deteriorated, or contaminated (common medicines that are expired or are no longer required or are discarded; other medicines discarded that could cause cancers or genetic diseases; the discarded vaccine products) Chemical waste: toxic, corrosive, flammable, and explosive chemical goods (hazardous chemicals; heavy metal-containing waste; pharmaceutical waste; amalgam wastes; gynotoxic waste; genotoxic waste |
| Blood-contaminated waste | Waste blood bags, waste used in hemodialysis, and other waste requiring special control because they contain enough blood to leak | Blood | - | - | Form: Blood, etc. (blood, serum, plasma, and body fluid) | - |
| General medical waste | Sanitary cotton, bandages, gauze, disposable diapers, sanitary napkins, disposable syringes, and infusion sets containing blood, body fluids, secretions, and excrement | - | Wastes whose collection and disposal is not subject to special requirements to prevent infection/amalgam waste from dental care | Sanitary/diaper waste, etc. | - | - |
| Other medical waste | - | Low-concentration radioactive waste | Pharmaceutical waste | If there are no applicable items from Step 1 to Step 3, it is classified as non-infectious waste * | ||
| Remarks | Wastes Control Act Enforcement Decree (see Supplementary Material) Types of Medical Waste | US EPA (https://www.epa.gov/sites/default/files/2016-02/documents/model_guidelines_for_state_medical_waste_management.pdf (accessed on 31 May 2021)) | EU Waste Framework Directive 2008/98/EC | UK, Waste control regulations (England and Wales regulations 2012;2012;811) | Japan Ministry of the Environment, Infectious Waste Disposal Manual | Healthcare Waste Management regulation |
| Country | Discharge/Container | Storage | Transport | Disposal |
|---|---|---|---|---|
| USA |
|
|
|
|
| Japan |
|
|
|
|
| Germany |
|
Stored at temperatures of −15 °C or below (can be stored for up to one week)∙
|
|
|
| UK |
| Follows ADR regulations |
| |
| Republic of Korea |
|
|
|
|
| China |
|
|
|
|
| Disposal Technology | WHO | Basel Convention | USA | Japan | China | Republic of Korea | ||
|---|---|---|---|---|---|---|---|---|
| California | Florida | |||||||
| Physical disposal | Shredding |
| ||||||
| Grinding | ||||||||
| Mixing | ||||||||
| Chemical disposal | Disinfectant disposal |
|
|
|
|
|
|
|
| Alkali hydrolysis |
| |||||||
| Biological disposal | Composting |
| ||||||
| Earthworm farming | ||||||||
| Thermal disposal | Steam sterilization (steam disposal) |
|
|
|
|
|
|
|
| ||||||||
| Dry heat sterilization |
|
|
|
|
| |||
| Microwave |
|
|
|
| ||||
| Radiation (irradiation) disposal | Cobalt 60 |
|
| |||||
| UV-rays |
| ✕ | ||||||
| Other disposals |
|
| ||||||
| Reference | [6] | [8] | [7] | [9] | [11] | [1] | ||
| Category | Discharger Storage | Transport | Treatment |
|---|---|---|---|
| Current regulations on infectious waste |
|
|
|
| Reinforced COVID-19 infectious waste management |
|
|
|
| Country | ROK | Japan | UK | USA | China |
|---|---|---|---|---|---|
| Governance | Government-led waste treatment | Government-led application of existing novel influenza guidelines | Decided by local governments | Guidelines of private companies | Decided by local governments under the guidance of the central government |
| Content |
|
|
|
|
|
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yoon, C.-W.; Kim, M.-J.; Park, Y.-S.; Jeon, T.-W.; Lee, M.-Y. A Review of Medical Waste Management Systems in the Republic of Korea for Hospital and Medical Waste Generated from the COVID-19 Pandemic. Sustainability 2022, 14, 3678. https://doi.org/10.3390/su14063678
Yoon C-W, Kim M-J, Park Y-S, Jeon T-W, Lee M-Y. A Review of Medical Waste Management Systems in the Republic of Korea for Hospital and Medical Waste Generated from the COVID-19 Pandemic. Sustainability. 2022; 14(6):3678. https://doi.org/10.3390/su14063678
Chicago/Turabian StyleYoon, Cheol-Woo, Min-Jung Kim, Yoon-Su Park, Tae-Wan Jeon, and Min-Yong Lee. 2022. "A Review of Medical Waste Management Systems in the Republic of Korea for Hospital and Medical Waste Generated from the COVID-19 Pandemic" Sustainability 14, no. 6: 3678. https://doi.org/10.3390/su14063678
APA StyleYoon, C.-W., Kim, M.-J., Park, Y.-S., Jeon, T.-W., & Lee, M.-Y. (2022). A Review of Medical Waste Management Systems in the Republic of Korea for Hospital and Medical Waste Generated from the COVID-19 Pandemic. Sustainability, 14(6), 3678. https://doi.org/10.3390/su14063678







