Exogenous ABA Enhances the Antioxidant Defense System of Maize by Regulating the AsA-GSH Cycle under Drought Stress
Abstract
:1. Introduction
2. Materials and Methods
2.1. Plant Material, Growth Conditions, Experimental Design, and Sampling
2.2. Measurement of H2O2 Content
2.3. Measurement of MDA Content
2.4. Determination of Electrolyte Leakage
2.5. Measurement of Antioxidant Enzyme Activities
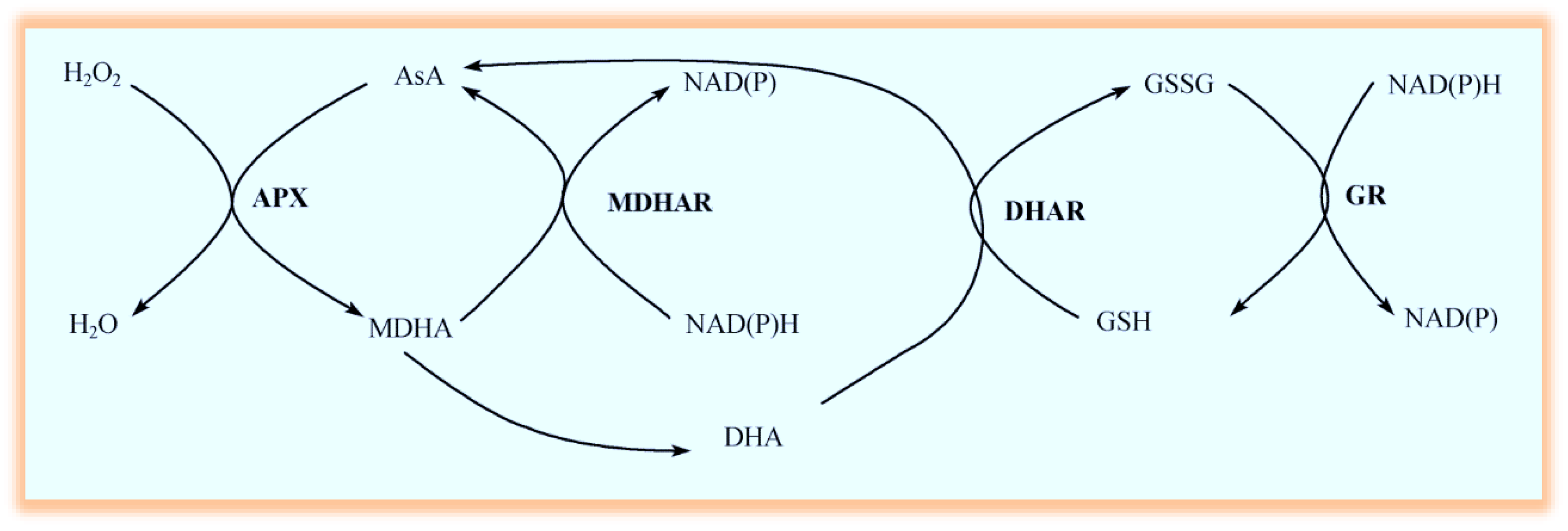
2.6. Measurements of AsA/DHA and GSH/GSSG in Leaves
2.7. RNA Isolation and Real-Time Quantitative Polymerase Chain Reaction
2.8. Statistical Analysis
3. Results
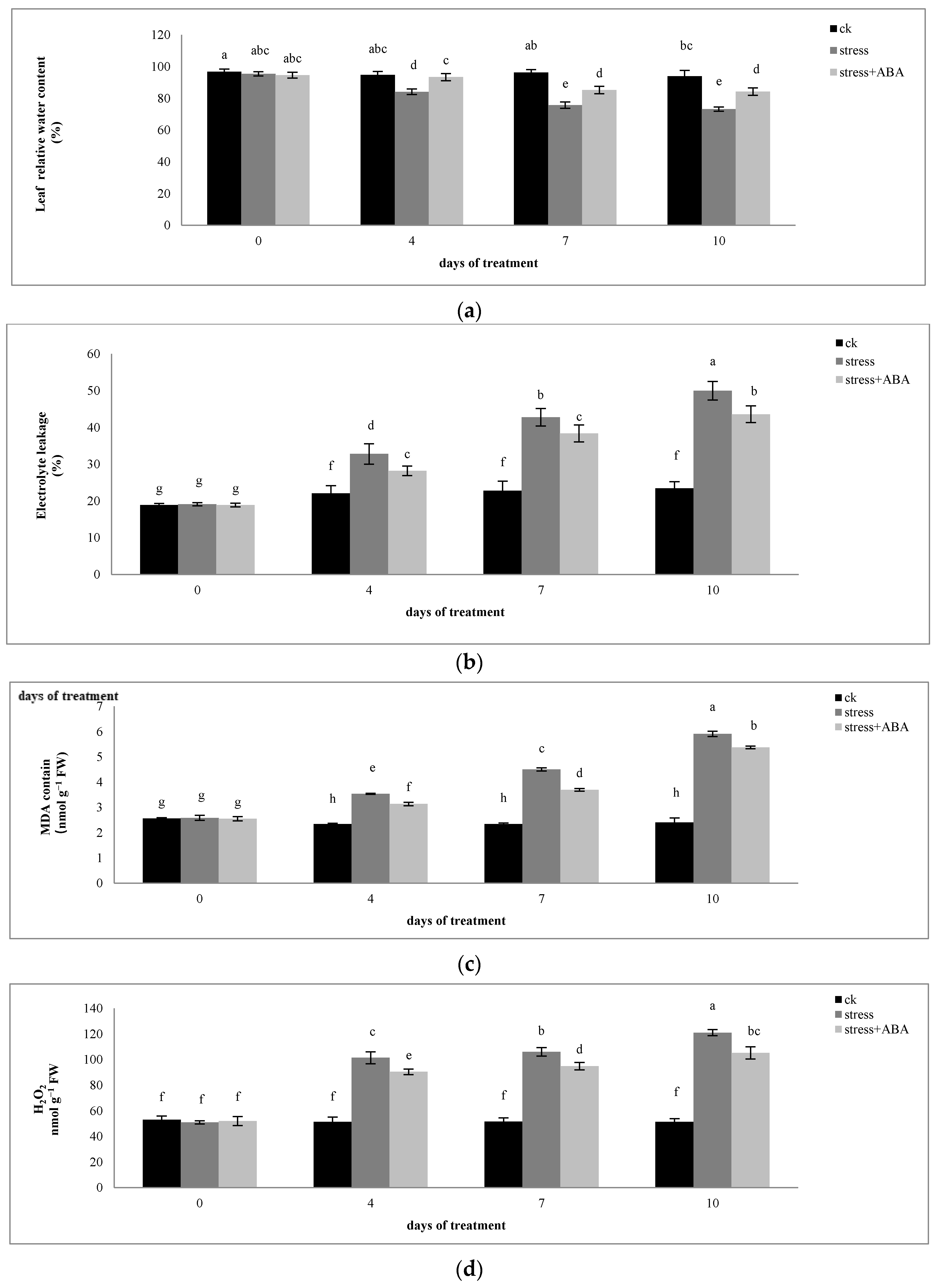
3.1. Effects of Drought Stress and/or ABA Application on the Relative Water Content, Relative Electrolyte Leakage, MDA Content, and H2O2 Content of Leaves
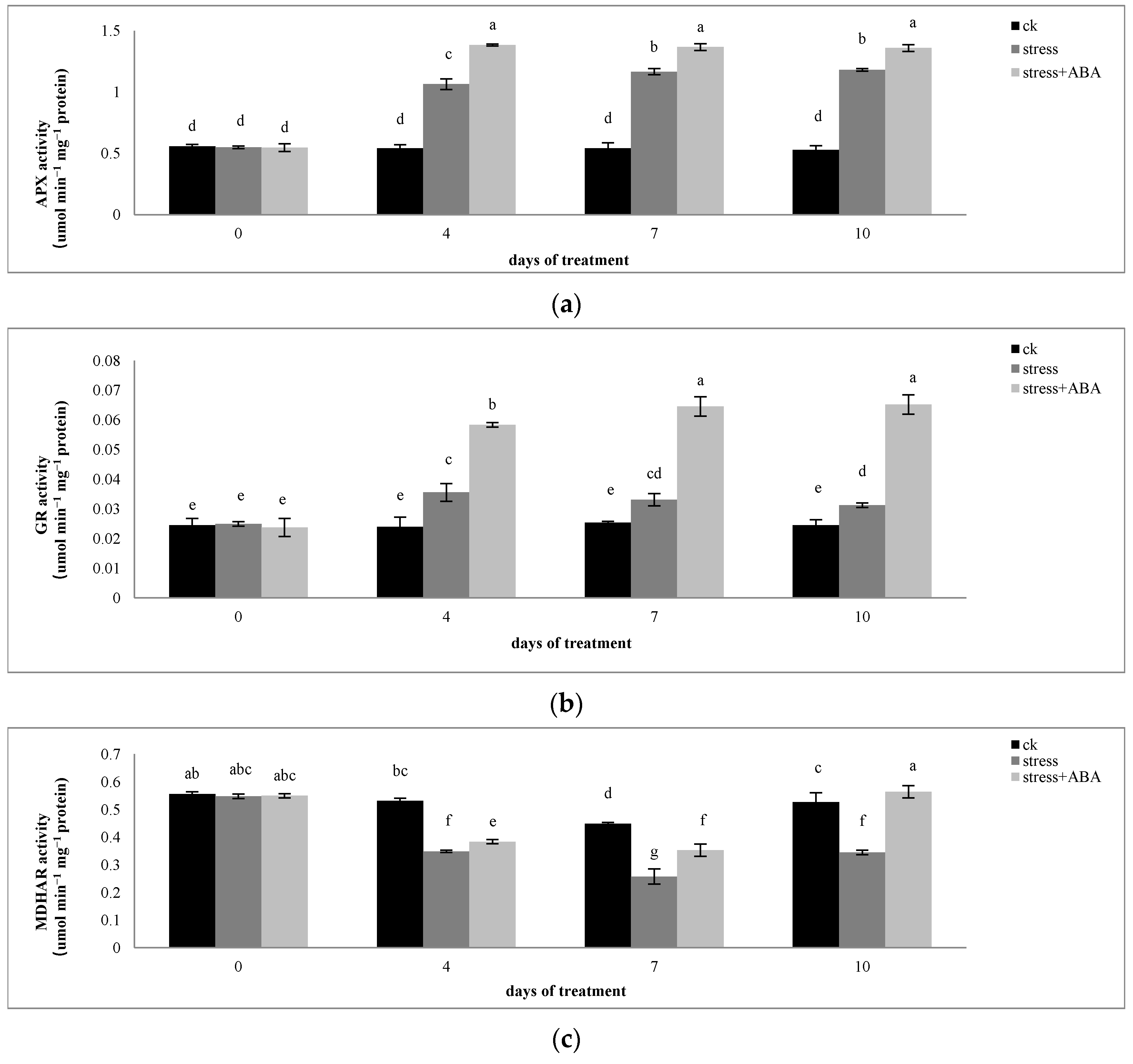
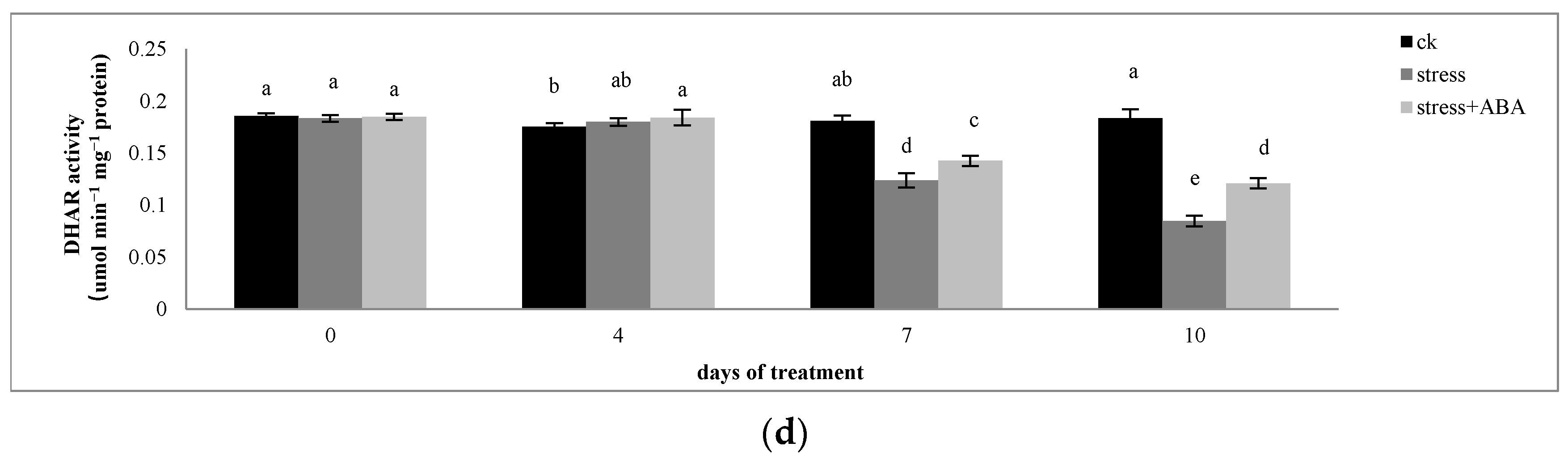
3.2. Effects of Drought Stress and/or ABA Application on APX, MDHAR, DHAR, and GR Activity
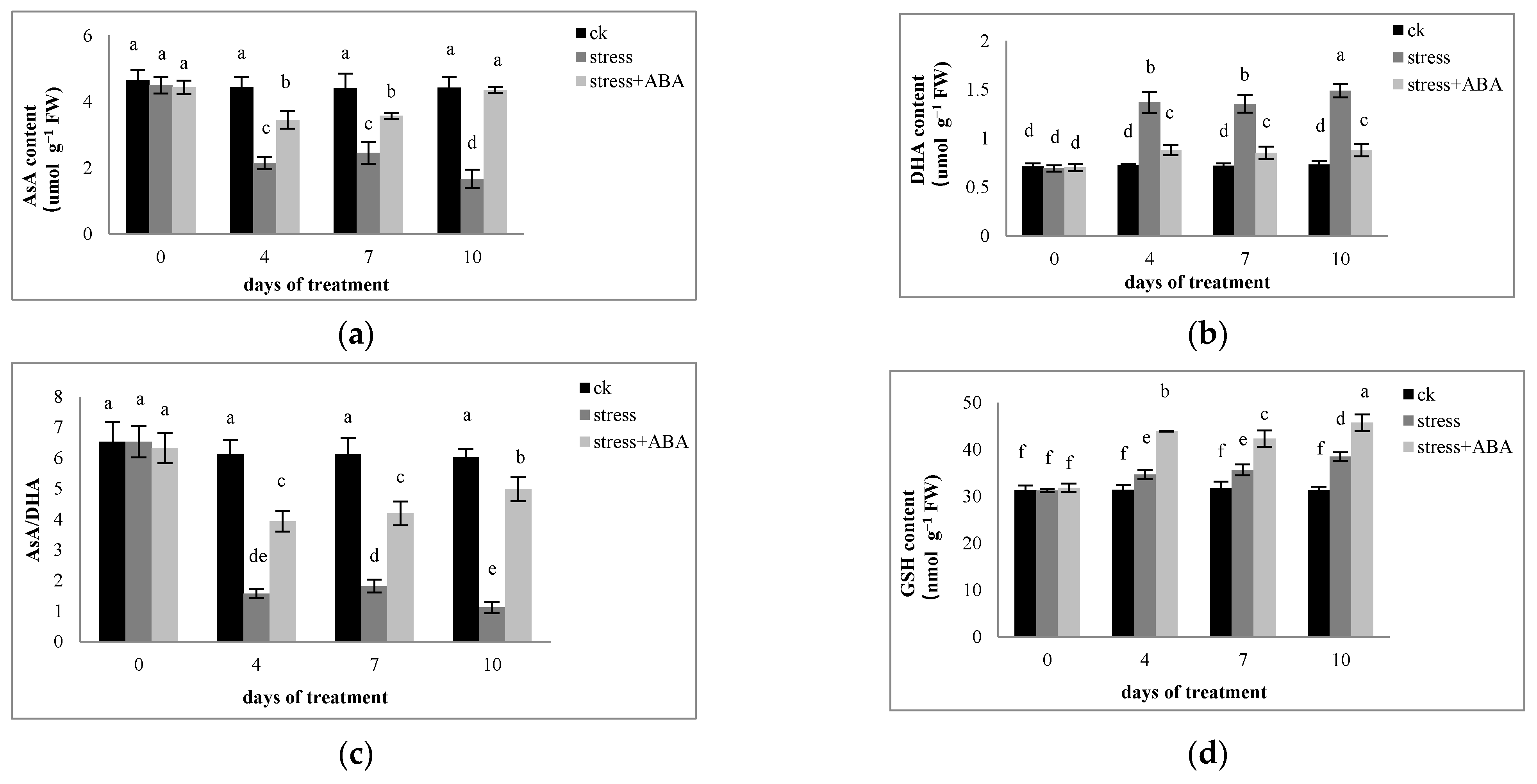
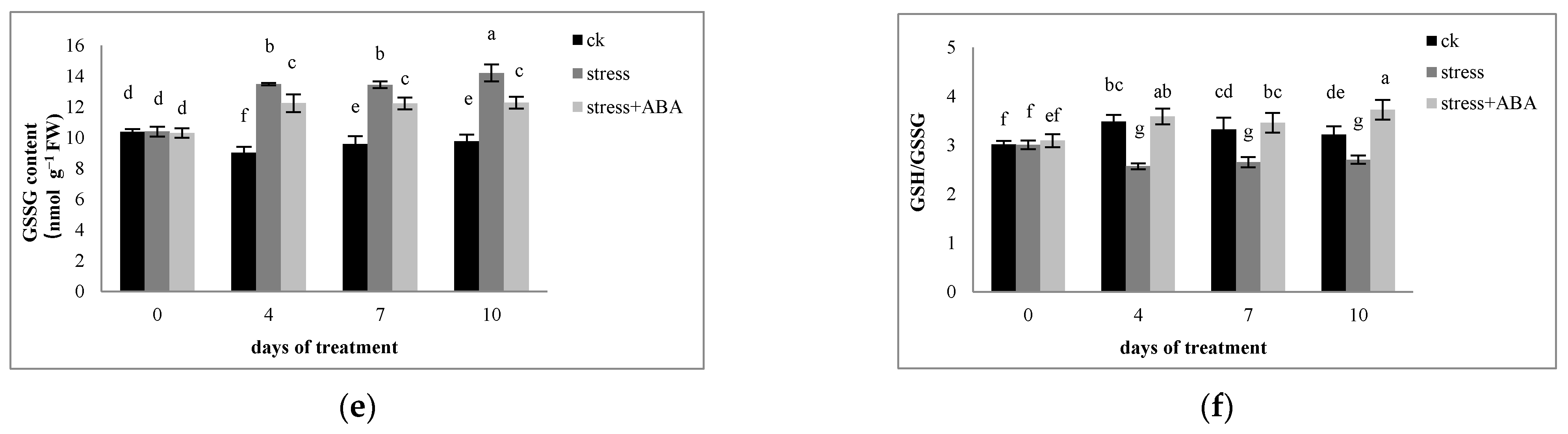
3.3. Effects of Drought Stress and/or ABA Application on Non-Enzymatic Antioxidants
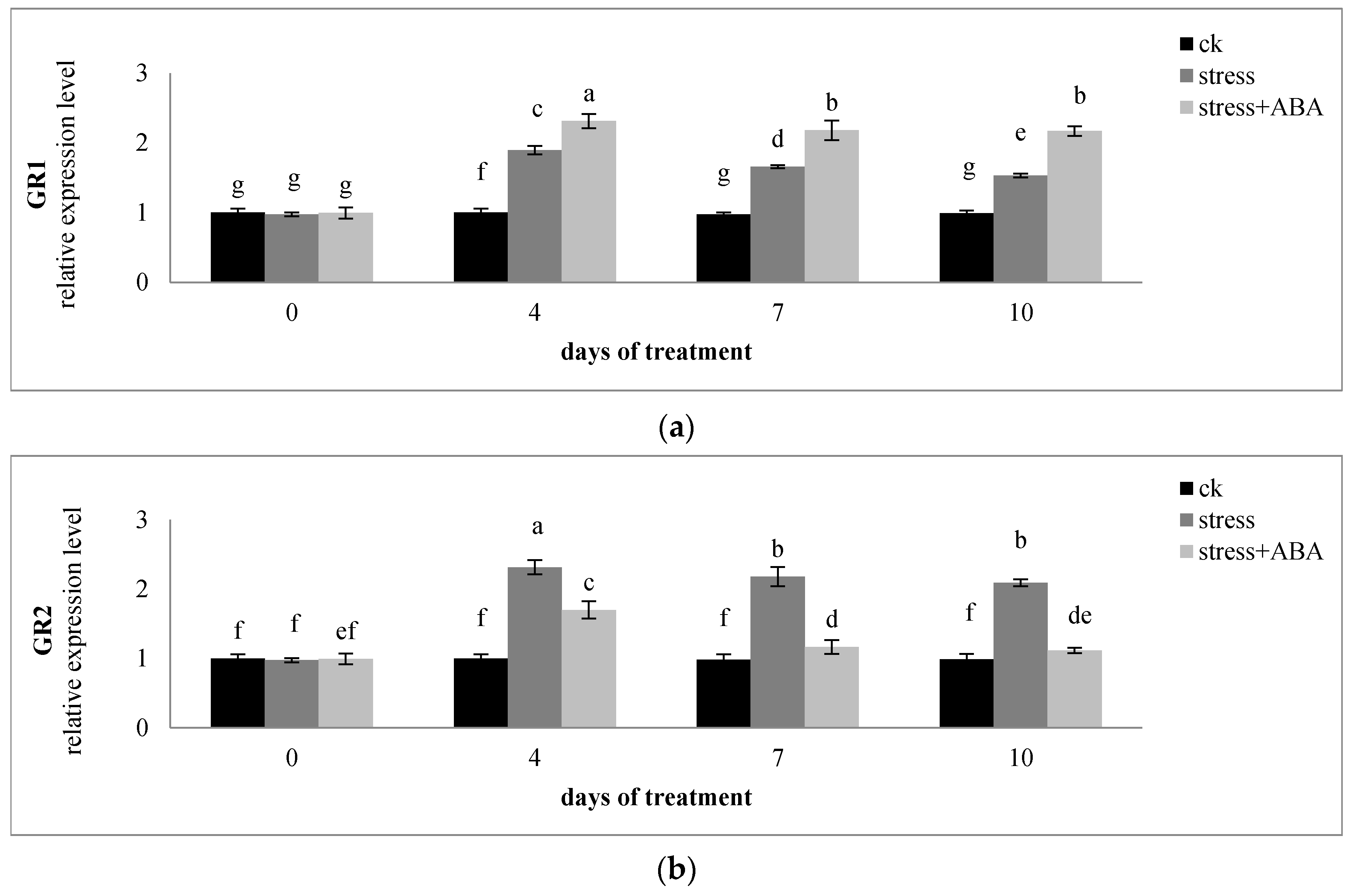
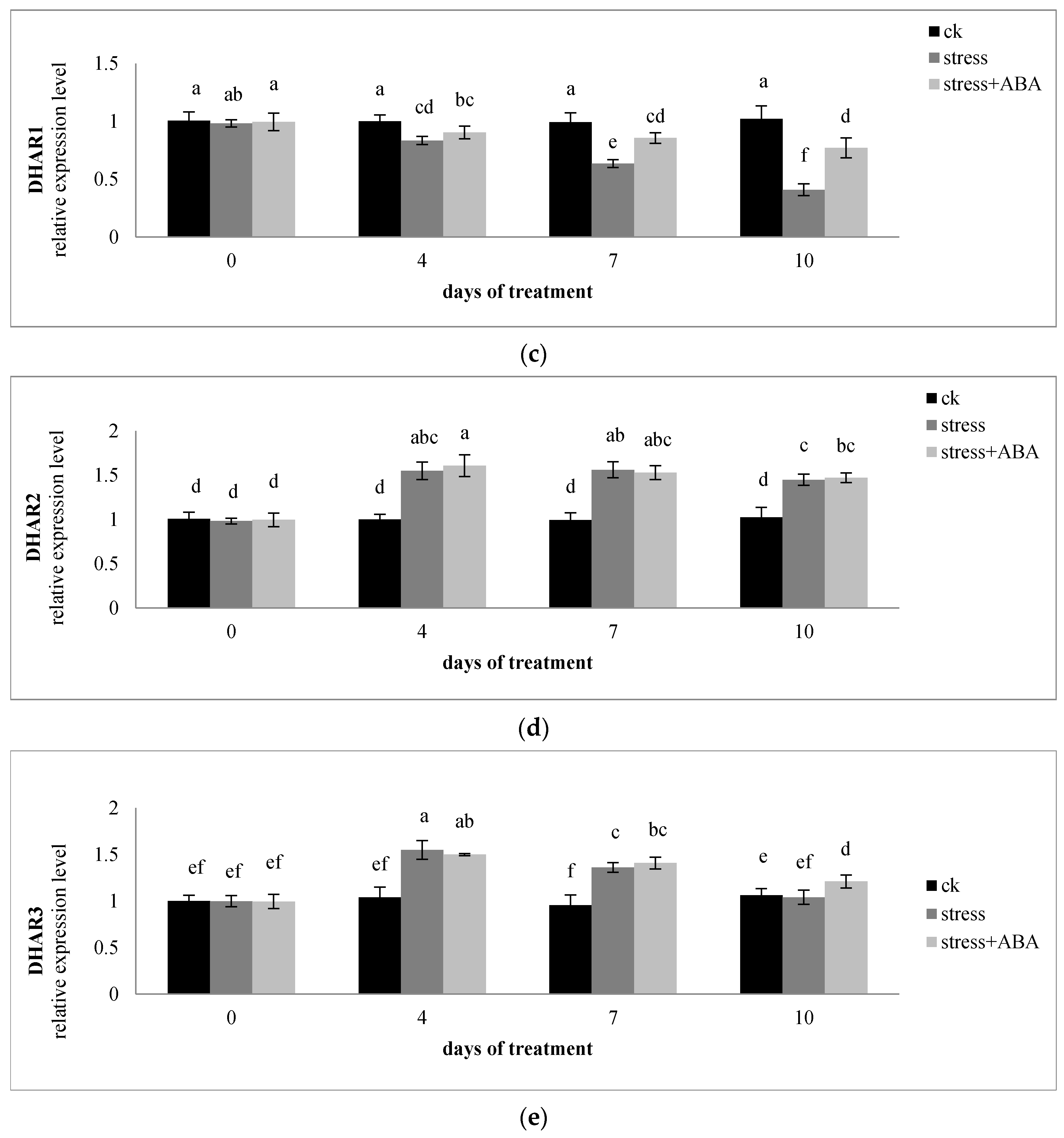
3.4. Effects of Drought Stress and/or ABA Application on the Expression of Genes Encoding AsA-GSH Cycle Enzymes
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Lee, R. The Outlook for Population Growth. Science 2011, 333, 569–573. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schiermeier, Q. Water risk as world warms. Nature 2014, 505, 10–11. [Google Scholar] [CrossRef] [PubMed]
- Ray, D.K.; Mueller, N.D.; West, P.C.; Foley, J.A. Yield Trends Are Insufficient to Double Global Crop Production by 2050. PLoS ONE 2013, 8, e66428. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Naveed, M.; Mitter, B.; Reichenauer, T.G.; Wieczorek, K.; Sessitsch, A. Increased drought stress resilience of maize through endophytic colonization by Burkholderia phytofirmans PsJN and Enterobacter sp. FD17. Environ. Exp. Bot. 2014, 97, 30–39. [Google Scholar] [CrossRef]
- Ashraf, M. Biotechnological approach of improving plant salt tolerance using antioxidants as markers. Biotechnol. Adv. 2009, 27, 84–93. [Google Scholar] [CrossRef] [PubMed]
- Dalal, V.K.; Tripathy, B.C. Water-stress induced downsizing of light-harvesting antenna complex protects developing rice seedlings from photo-oxidative damage. Sci. Rep. 2018, 8, 5955. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Araújo, G.D.S.; Lopes, L.D.S.; Paula-Marinho, S.D.O.; Mesquita, R.O.; Gomes-Filho, E. H2O2 priming induces proteomic responses to defense against salt stress in maize. Plant Mol. Biol. 2021, 106, 33–48. [Google Scholar] [CrossRef]
- Bright, J.; Desikan, R.; Hancock, J.T.; Weir, I.S.; Neill, S.J. ABA-induced NO generation and stomatal closure in Arabidopsis are dependent on H2O2 synthesis. Plant J. 2006, 45, 113–122. [Google Scholar] [CrossRef] [PubMed]
- Santos, B.; Silva, P.; Matos, R.M.D.; Matos, R.; Neto, J.; Melo, Y. Induction of salt stress tolerance in chives by priming with H2O2 in hydroponic cultivation. Chil. J. Agric. Res. 2021, 81, 317–325. [Google Scholar] [CrossRef]
- Carvalho, F.; Silveira, J. H2O2-retrograde signaling as a pivotal mechanism to understand priming and cross stress tolerance in plants. In Priming-Mediated Stress and Cross-Stress Tolerance in Crop Plants; Academic Press: Cambridge, MA, USA, 2020; pp. 57–78. [Google Scholar]
- Cho, B.O.; Ryu, H.W.; Lee, C.W.; Lee, C.W.; Jin, C.H.; Seo, W.D.; Ryu, J.; Jeong, I.Y. Protective effects of new blackberry cultivar MNU-32 extracts against H2O2-induced oxidative stress in HepG2 cells. Food Sci. Biotechnol. 2015, 24, 643–650. [Google Scholar] [CrossRef]
- Selote, D.S.; Khanna, C.R. Drought acclimation confers oxidative stress tolerance by inducing coordinated antioxidant defense at cellular and subcellular level in leaves of wheat seedlings. Physiol. Plant. 2006, 127, 494–506. [Google Scholar] [CrossRef]
- Apel, K.; Hirt, H. Reactive oxygen species: Metabolism, oxidative stress, and signal transduction. Annu. Rev. Plant Biol. 2004, 55, 373–379. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Foyer, C.H. Reactive oxygen species, oxidative signaling and the regulation of photosynthesis. Environ. Exp. Bot. 2018, 154, 134–142. [Google Scholar] [CrossRef] [PubMed]
- Yaghubi, K.; Ghaderi, N.; Vafaee, Y.; Javadi, T. Potassium silicate alleviates deleterious effects of salinity on two strawberry cultivars grown under soilless pot culture. Sci. Hortic. 2016, 213, 87–95. [Google Scholar] [CrossRef]
- Sharma, P.; Dubey, R.S. Drought Induces Oxidative Stress and Enhances the Activities of Antioxidant Enzymes in Growing Rice Seedlings. Plant Growth Regul. 2005, 46, 209–221. [Google Scholar] [CrossRef]
- Scandalios, J.G. Oxidative stress: Molecular perception and transduction of signals triggering antioxidant gene defenses. Braz. J. Med. Biol. Res. 2005, 38, 995–1014. [Google Scholar] [CrossRef] [PubMed]
- Hameed, A.; Goher, M.; Iobal, N. Drought induced programmed cell death and associated changes in antioxidants, proteases, and lipid peroxidation in wheat leaves. Biol. Plant. 2013, 57, 370–374. [Google Scholar] [CrossRef]
- Hojati, M.; Modarres-Sanavy, S.A.M.; Karimi, M.; Ghanati, F. Responses of growth and antioxidant systems in Carthamus tinctorius L. under water deficit stress. Acta Physiol. Plant 2011, 33, 105–112. [Google Scholar] [CrossRef]
- Singh, R.; Parihar, P.; Prasad, S.M. Sulphur and calcium attenuate arsenic toxicity in Brassica by adjusting ascorbate–glutathione cycle and sulphur metabolism. Plant Growth Regul. 2020, 91, 221–235. [Google Scholar] [CrossRef]
- Zhou, W. Deteriorative effects of cadmium stress on antioxidant system and cellular structure in germinating seeds of Brassica napus L. J. Agric. Sci. Technol. 2018, 17, 63–74. [Google Scholar]
- Ma, J.; Zheng, G.; Pei, C.M.; Zhang, Z.Y. The function of ascorbate-glutathione cycle in salt tolerance of alfalfa mutant. Plant Physiol. 2015, 79, 287–297. [Google Scholar]
- Guo, P.; Baum, M.; Grando, S.; Ceccarelli, S.; Bai, G.; Li, R.; Korff, M.; Varshney, R.K.; Graner, A.; Valkoun, J. Differentially expressed genes between drought-tolerant and drought-sensitive barley genotypes in response to drought stress during the reproductive stage. Exp. Bot. 2009, 60, 3531–3544. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Liu, Y.; Zhang, J. Advances in the research on the AsA-GSH cycle in horticultural crops. Front. Agric. Chin. 2010, 4, 84–90. [Google Scholar] [CrossRef]
- Moothoo-Padayachie, A.; Varghese, B.; Pammenter, N.W.; Govender, P.; Naidoo, S. Germination associated ROS production and glutathione redox capacity in two recalcitrant-seeded species differing in seed longevity. Botany 2016, 94, 1103–1114. [Google Scholar] [CrossRef]
- Liu, Y.J.; Yuan, Y.; Liu, Y.Y.; Liu, Y.; Fu, J.J.; Zheng, J.; Wang, G.Y. Gene families of maize glutathione–ascorbate redox cycle respond differently to abiotic stresses. J. Plant Physiol. 2012, 169, 183–192. [Google Scholar] [CrossRef] [PubMed]
- Kanchan, V.; Neha, U.; Nitin, K.; Gaurav, Y.; Jaspreet, S.; Mishra, R.K.; Vivek, K.; Rishi, V.; Upadhyay, R.G.; Mayank, P. Abscisic Acid Signaling and Abiotic Stress Tolerance in Plants: A Review on Current Knowledge and Future Prospects. Front. Plant Sci. 2017, 8, 161. [Google Scholar]
- Kuromori, T.; Seo, M.; Shinozaki, K. ABA Transport and Plant Water Stress Responses. Trends Plant Sci. 2018, 23, 513–522. [Google Scholar] [CrossRef] [PubMed]
- Thompson, A.J.; Andrews, J.; Mulholland, B.J.; McKee, J.M.T.; Hilton, H.W.; Horridge, J.S.; Farquhar, G.D.; Smeeton, R.C.; Smilie, I.R.A.; Black, C.R.; et al. Overproduction of abscisic acid in tomato increases transpiration efficiency and root hydraulic conductivity and influences leaf expansion. Plant Physiol. 2007, 143, 1905–1917. [Google Scholar] [CrossRef] [Green Version]
- Hu, X.L.; Jiang, M.Y.; Zhang, A.Y.; Lu, J. Abscisic acid-induced apoplastic H2O2 accumulation up-regulates the activities of chloroplastic and cytosolic antioxidant enzymes in maize leaves. Planta 2005, 223, 57–68. [Google Scholar] [CrossRef] [PubMed]
- Kellos, T.; Tímár, I.; Szilágyi, V.; Szalai, G.; Galiba, G.; Kocsy, G. Stress hormones and abiotic stress have different effects on antioxidants in maize lines with different sensitivity. Plant Biol. 2008, 10, 563–572. [Google Scholar] [CrossRef] [PubMed]
- Song, X.S.; Tiao, C.L.; Shi, K.; Mao, W.H.; Ogweno, J.O.; Zhou, Y.H.; Yu, J.Q. The response of antioxidant enzymes in cellular (Cucumissativus L.) leaves to methyl viologen-induced photo-oxidative stress. Plant Growth Regul. 2006, 49, 85–93. [Google Scholar] [CrossRef]
- Talaat, N.B.; Shawky, B.T.; Ibrahim, A.S. Alleviation of drought-induced oxidative stress in maize (Zeamays L.) plants by dual application of 24-epibrassinolide and spermine. Environ. Exp. Bot. 2015, 113, 47–58. [Google Scholar] [CrossRef]
- Hodges, D.M.; Delong, J.M.; Forney, C.F. Improving the thiobarbituric acid-reactive-substances assay for estimating lipid peroxidation in plant tissues containing anthocyanin and other interfering compounds. Planta 1999, 207, 604–611. [Google Scholar] [CrossRef]
- Zhu, Z.; Wei, G.; Li, J.; Qian, Q.Q.; Yu, J.Q. Silicon alleviates salt stress and increases antioxidant enzymes activity in leaves of salt-stressed cucumber (Cucumis sativus L.). Plant Sci. 2004, 167, 527–533. [Google Scholar] [CrossRef]
- Hoque, M.A.; Banu, M.N.; Okuma, E.; Amako, K.; Nakamura, Y.; Shimoishi, Y.; Murata, Y. Exogenous proline and glycinebetaine increase NaCl-induced ascorbate-glutathione cycle enzyme activities, and proline improves salt tolerance more than glycinebetaine in tobacco Bright Yellow-2suspension-cultured cells. J. Plant Physiol. 2007, 164, 1457–1468. [Google Scholar] [CrossRef]
- Doulis, A.G.; Debian, N.; Kingston Smith, A.H.; Foyer, C.H. Differential localization of antioxidants in maize leaves. Plant Physiol. 1997, 114, 1031–1037. [Google Scholar] [CrossRef] [Green Version]
- Ramiro, D.A.; Guerreiro-Filho, O.; Mazzafera, P. Phenol contents, oxidase activities, and the resistance of coffee to the leaf miner Leucoptera coffeella. J. Chem. Ecol. 2006, 32, 1977–1988. [Google Scholar] [CrossRef]
- Bradford, M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef]
- Hodges, D.M.; Andrews, C.J.; Johnson, D.A.; Hamilton, R.I. Antioxidant compound responses to chilling stress in differentially sensitive inbred maize lines. J. Exp. Bot. 1997, 48, 1105–1113. [Google Scholar] [CrossRef]
- Griffith, O.W. Determination of glutathione and glutathione disulfide using glutathione reductase and 2-vinyl pyridine. Anal. Biochem. 1980, 106, 207–212. [Google Scholar] [CrossRef]
- Chugh, V.; Kaur, N.; Gupta, A.K. Evaluation of oxidative stress tolerance in maize (Zea mays L.) seedlings in response to drought. Indian J. Biochem. Biophys. 2011, 48, 47–53. [Google Scholar] [PubMed]
- Xing, X.; Xu, Z.; Tong, F.; Qi, Y.J.; Wang, X. Involvement of triadimefon induced early ABA-dependent H2O2 accumulation in soybean against water stress. Oil Crop Sci. 2020, 5, 41–47. [Google Scholar] [CrossRef]
- Queval, G.; Hager, J.; Gakiere, B.; Noctor, G. Why are literature data for H2O2 contents so variable? A discussion of potential difficulties in the quantitative assay of leaf extracts. J. Exp. Bot. 2008, 59, 135–146. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- De Souza, T.C.; Magalhães, P.C.; de Castro, E.M.; Carneiro, N.P.; Padilha, F.A.; JHYPER, C.C.G. ABA application to maize hybrids contrasting for drought tolerance: Changes in water parameters and in antioxidant enzyme activity. Plant Growth Regul. 2014, 73, 205–217. [Google Scholar] [CrossRef]
- Yang, J.; Zhang, J.; Ye, Y.; Yang, J.C.; Zhang, J.H.; Ye, Y.X.; Wang, Z.Q.; Zhu, Q.S.; Liu, L.J. Involvement of abscisic acid and ethylene in the responses of rice grains to water stress during filling. Plant Cell Environ. 2004, 27, 1055–1064. [Google Scholar] [CrossRef]
- Anjum, S.A.; Wang, L.; Farooq, M.; Xue, L.; Ali, S. Fulvic acid application improves the maize performance under well-watered and drought conditions. J. Agron. Crop Sci. 2011, 197, 409–417. [Google Scholar] [CrossRef]
- Moussa, H.R.; Abdel-Aziz, S.M. Comparative response of drought tolerant and sensitive maize genotypes to water stress. Aust. J. Crop Sci. 2008, 1, 31–36. [Google Scholar]
- Shan, C.; Wang, B.; Sun, H.; Gao, S.; Li, H. H2S induces NO in the regulation of AsA-GSH cycle in wheat seedlings by water stress. Protoplasma 2020, 257, 1487–1493. [Google Scholar] [CrossRef]
- Li, L.; Gu, W.; Li, C.; Li, W.; Li, C.; Li, J.; Wei, S. Exogenous spermidine improves drought tolerance in maize by enhancing the antioxidant defence system and regulating endogenous polyamine metabolism. Crop Pasture Sci. 2018, 69, 1076–1091. [Google Scholar] [CrossRef]
- Xie, T.; Gu, W.; Zhang, L.; Li, L.; Qu, D.; Li, C.; Meng, Y.; Li, J.; Wei, S.; Li, W. Modulating the antioxidant system by exogenous 2-(3, 4-dichlorophenoxy) triethylamine in maize seedlings exposed to polyethylene glycol-simulated drought stress. PLoS ONE 2018, 13, e0203626. [Google Scholar] [CrossRef]
- Ishikawa, T.; Shigeoka, S. Recent advances in ascorbate biosynthesis and the physiological significance of ascorbate peroxidase in photosynthesizing organisms. Biosci. Biotechnol. Biochem. 2008, 72, 1143–1154. [Google Scholar] [CrossRef] [PubMed]
- Sang, Q.Q.; Shu, S.; Shan, X.; Guo, S.R.; Sun, J. Effects of exogenous spermidine on antioxidant system of tomato seedlings exposed to high temperature stress. Russ. J. Plant Physiol. 2016, 63, 645–655. [Google Scholar] [CrossRef]
- Bai, J.; Tai, K.; Wu, H.; Lu, B.; Gong, C. Relative contribution of photorespiration and antioxidative mechanisms in Caragana korshinskii under drought conditions across the Loess Plateau. Funct. Plant Biol. 2017, 44, 1111–1123. [Google Scholar] [CrossRef] [PubMed]
- Gicek, N.; Gakirlar, H. Changes in some antioxidant enzyme activities in response to long-term salinity at two different temperatures. Gen. Appl. Plant Physiol. 2008, 34, 267–280. [Google Scholar]
- Li, N.; Cao, B.; Chen, Z.; Xu, K. Root morphology ion absorption and antioxidative defense system of two Chinese cabbage cultivars (Brassica rapa L.) reveal the different adaptation mechanisms to salt and alkali stress. Protoplasma 2021, 259, 385–398. [Google Scholar] [CrossRef]
- Shao, H.B.; Chu, L.Y.; Lu, Z.H.; Kang, C.M. Primary antioxidant free radical scavenging and redox signaling pathways in higher plant cells. Int. J. Biol. Sci. 2008, 4, 8–14. [Google Scholar] [CrossRef]
- Guo, Z.; Ou, W.; Lu, S.; Zhong, Q. Differential responses of antioxidative system to chilling and drought in four rice cultivars differing in sensitivity. Plant Physiol. Biochem. 2006, 44, 828–836. [Google Scholar] [CrossRef]
- Dash, S.; Mohanty, N. Response of seedlings to heat-stress in cultivars of wheat: Growth temperature-dependent differential modulation of photosystem 1 and 2 activity, and foliar antioxidant defense capacity. J. Plant Physiol. 2002, 159, 49–59. [Google Scholar] [CrossRef]
- Szalai, G.; Hellos, T.; Galiba, G.; Kocsy, G. Glutathione as an antioxidant and regulatory molecule in plants under abiotic stress conditions. J. Plant Growth Regul. 2009, 28, 66–80. [Google Scholar] [CrossRef]
- Verma, G.; Mishra, S.; Sangwan, N.; Sharma, S. Reactive oxygen species mediate axis-cotyledon signaling to induce reserve mobilization during germination and seedling establishment in Vigna radiata. J. Plant Physiol. 2015, 184, 79–88. [Google Scholar] [CrossRef]
| Gene | Forward Sequence | Reverse Sequence |
|---|---|---|
| GR1 | 5′-CGGTGCAATAGTGGTTGATG-3′ | 5′-CCTATTGGTGGTTGGGAGAA-3′ |
| GR2 | 5′-CGATATTGCGGTTAAATGTG-3′ | 5′-AAGTTCGTCTTTGGCTTGGA-3′ |
| DHAR1 | 5′-CATCAAGACTAAGCCCACCAA-3′ | 5′-TAGAAACATGGCCACCACAA-3′ |
| DHAR2 | 5′-CAATGTCCATGCCTACACCA-3′ | 5′-CAGGTAGCACCAAAGCACAA-3′ |
| DHAR3 | 5′-CGAGGAAAAATGGATTGGTG-3′ | 5′-TGTTCCATCGCTTGGATCTT-3′ |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jiang, Z.; Zhu, H.; Zhu, H.; Tao, Y.; Liu, C.; Liu, J.; Yang, F.; Li, M. Exogenous ABA Enhances the Antioxidant Defense System of Maize by Regulating the AsA-GSH Cycle under Drought Stress. Sustainability 2022, 14, 3071. https://doi.org/10.3390/su14053071
Jiang Z, Zhu H, Zhu H, Tao Y, Liu C, Liu J, Yang F, Li M. Exogenous ABA Enhances the Antioxidant Defense System of Maize by Regulating the AsA-GSH Cycle under Drought Stress. Sustainability. 2022; 14(5):3071. https://doi.org/10.3390/su14053071
Chicago/Turabian StyleJiang, Zizhu, Hengguang Zhu, Hanyu Zhu, Yuzhao Tao, Changzhuang Liu, Jiaqi Liu, Fuqiang Yang, and Ming Li. 2022. "Exogenous ABA Enhances the Antioxidant Defense System of Maize by Regulating the AsA-GSH Cycle under Drought Stress" Sustainability 14, no. 5: 3071. https://doi.org/10.3390/su14053071
APA StyleJiang, Z., Zhu, H., Zhu, H., Tao, Y., Liu, C., Liu, J., Yang, F., & Li, M. (2022). Exogenous ABA Enhances the Antioxidant Defense System of Maize by Regulating the AsA-GSH Cycle under Drought Stress. Sustainability, 14(5), 3071. https://doi.org/10.3390/su14053071






