Wheat Straw Burial Enhances the Root Physiology, Productivity, and Water Utilization Efficiency of Rice under Alternative Wetting and Drying Irrigation
Abstract
1. Introduction
2. Materials and Methods
2.1. Experimental Location Characterization
2.2. Experimental Scheme, Treatments, and Cultural Practices
2.3. Determination of Root Physiological Characteristics
2.4. Determination of Dry Biomass Accumulation
2.5. Determination of Water Consumptive Use
2.6. Determination of WUE
2.7. Statistical Examination
3. Results
3.1. Consumption of Irrigation Water
3.2. Root Development and Physiological Activity Parameters
3.3. Dry Mass of Plant Parts Partitionings and Overall Plant Dry Mass
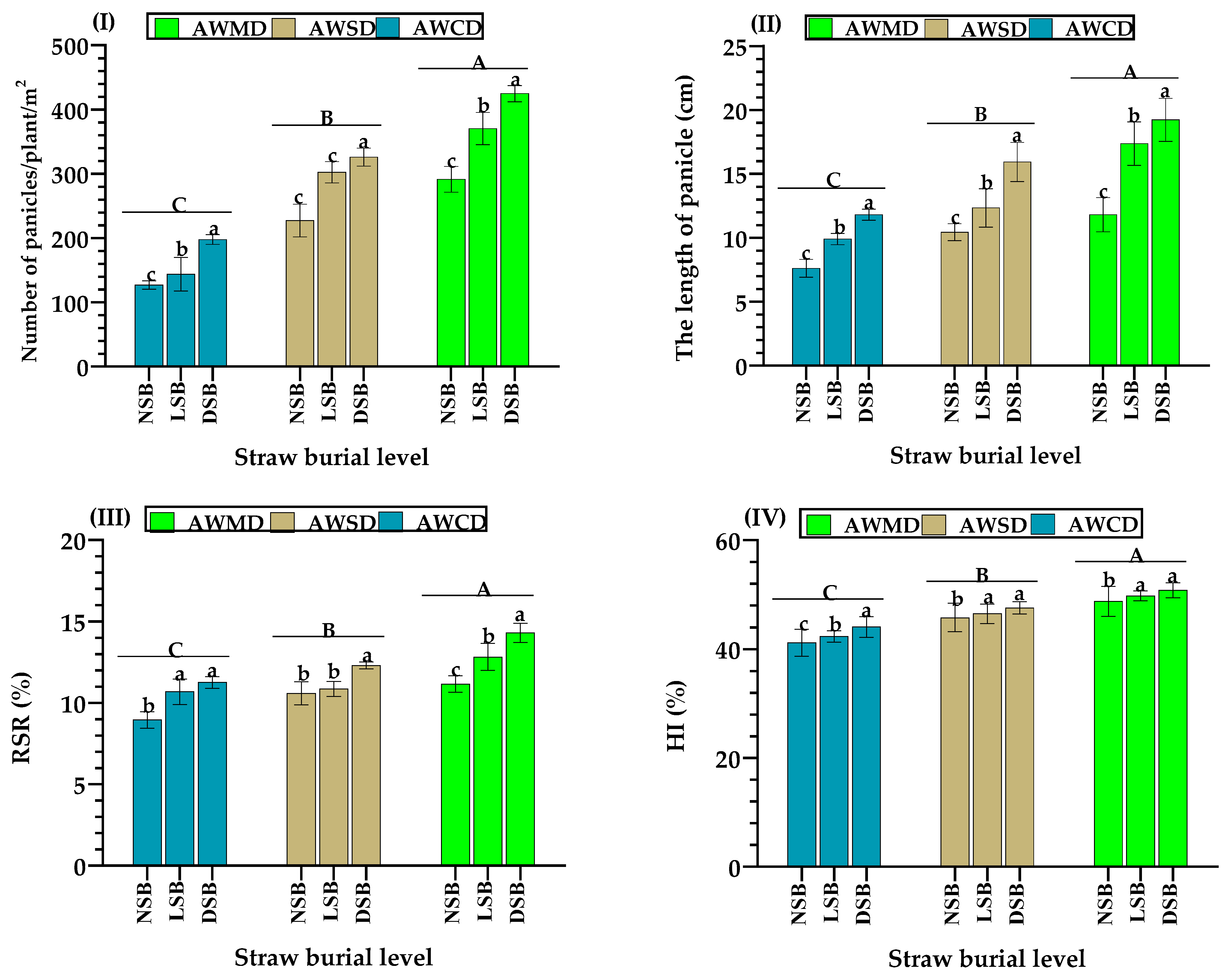
3.4. Indicators of the Grain and Dry Matter Production of Rice
3.5. WUE Indicators
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Jensen, C.R.; Battilani, A.; Plauborg, F.; Psarras, G.; Chartzoulakis, K.; Janowiak, F.; Stikic, R.; Jovanovic, Z.; Li, G.; Qi, X. Deficit irrigation based on drought tolerance and root signalling in potatoes and tomatoes. Agric. Water Manag. 2010, 98, 403–413. [Google Scholar] [CrossRef]
- Oumarou Abdoulaye, A.; Lu, H.; Zhu, Y.; Alhaj Hamoud, Y.; Sheteiwy, M. The global trend of the net irrigation water requirement of maize from 1960 to 2050. Climate 2019, 7, 124. [Google Scholar] [CrossRef]
- FAOSTAT. Agriculture data. In Agricultural Production; FAOSTAT: Rome, Italy, 2013. [Google Scholar]
- Buresh, R.; Haefele, S. Changes in Paddy Soils under Transition to Water-Saving and Diversified Cropping Systems; World Congress of Soil Science: Brisbane, Australia, 2010. [Google Scholar]
- Bouman, B. Water Management in Irrigated Rice: Coping with Water Scarcity; International Rice Research Institute: Manila, Philippines, 2007. [Google Scholar]
- Dibba, L.; Diagne, A.; Fialor, S.C.; Nimoh, F. Diffusion and adoption of new rice varieties for africa (NERICA) in the gambia. Afr. Crop Sci. J. 2012, 20, 141–153. [Google Scholar]
- Maclean, J.; Hardy, B.; Hettel, G. Rice Almanac: Source Book for One of the Most Important Economic Activities On Eazrth; IRRI: Los Baños, Philippines, 2013. [Google Scholar]
- Frolking, S.; Qiu, J.; Boles, S.; Xiao, X.; Liu, J.; Zhuang, Y.; Li, C.; Qin, X. Combining remote sensing and ground census data to develop new maps of the distribution of rice agriculture in china. Glob. Biogeochem. Cycles 2002, 16, 38. [Google Scholar] [CrossRef]
- Lin, Y.; Huabin, Z.; Jianxia, L. Current situation and prospect of rice water-saving irrigation technology in china. Chin. J. Ecol. 2014, 33, 1381–1387. [Google Scholar]
- Ye, Y.; Liang, X.; Chen, Y.; Liu, J.; Gu, J.; Guo, R.; Li, L. Alternate wetting and drying irrigation and controlled-release nitrogen fertilizer in late-season rice. Effects on dry matter accumulation, yield, water and nitrogen use. Field Crops Res. 2013, 144, 212–224. [Google Scholar] [CrossRef]
- Pan, J.; Liu, Y.; Zhong, X.; Lampayan, R.M.; Singleton, G.R.; Huang, N.; Liang, K.; Peng, B.; Tian, K. Grain yield, water productivity and nitrogen use efficiency of rice under different water management and fertilizer-n inputs in south china. Agric. Water Manag. 2017, 184, 191–200. [Google Scholar] [CrossRef]
- Alhaj Hamoud, Y.; Guo, X.; Wang, Z.; Shaghaleh, H.; Chen, S.; Hassan, A.; Bakour, A. Effects of irrigation regime and soil clay content and their interaction on the biological yield, nitrogen uptake and nitrogen-use efficiency of rice grown in southern china. Agric. Water Manag. 2019, 213, 934–946. [Google Scholar] [CrossRef]
- Xu, G.-w.; Lu, D.-K.; Wang, H.-Z.; Li, Y. Morphological and physiological traits of rice roots and their relationships to yield and nitrogen utilization as influenced by irrigation regime and nitrogen rate. Agric. Water Manag. 2018, 203, 385–394. [Google Scholar] [CrossRef]
- Alhaj Hamoud, Y.; Shaghaleh, H.; Sheteiwy, M.; Guo, X.; Elshaikh, N.A.; Khan, N.U.; Oumarou, A.; Rahim, S.F. Impact of alternative wetting and soil drying and soil clay content on the morphological and physiological traits of rice roots and their relationships to yield and nutrient use-efficiency. Agric. Water Manag. 2019, 223, 105706. [Google Scholar] [CrossRef]
- Tao, Y.; Zhang, Y.; Jin, X.; Saiz, G.; Jing, R.; Guo, L.; Liu, M.; Shi, J.; Zuo, Q.; Tao, H. More rice with less water–evaluation of yield and resource use efficiency in ground cover rice production system with transplanting. Eur. J. Agron. 2015, 68, 13–21. [Google Scholar] [CrossRef]
- Chu, G.; Wang, Z.; Zhang, H.; Yang, J.; Zhang, J. Agronomic and physiological performance of rice under integrative crop management. Agron. J. 2016, 108, 117–128. [Google Scholar] [CrossRef]
- Wang, Z.; Zhang, W.; Beebout, S.S.; Zhang, H.; Liu, L.; Yang, J.; Zhang, J. Grain yield, water and nitrogen use efficiencies of rice as influenced by irrigation regimes and their interaction with nitrogen rates. Field Crops Res. 2016, 193, 54–69. [Google Scholar] [CrossRef]
- Alhaj Hamoud, Y.; Wang, Z.; Guo, X.; Shaghaleh, H.; Sheteiwy, M.; Chen, S.; Qiu, R.; Elbashier, M.M.A. Effect of irrigation regimes and soil texture on the potassium utilization efficiency of rice. Agronomy 2019, 9, 100. [Google Scholar] [CrossRef]
- Islam, M.; Parul, S.; Pathan, A.; Islam, M.; Quasem, M. Influence of Cracking on Rice Seasons and Irrigation in Bangladesh; Asian Network for Scientific Information: Faisalabad, Pakistan, 2004. [Google Scholar]
- Stewart, R.D.; Abou Najm, M.R.; Rupp, D.E.; Lane, J.W.; Uribe, H.C.; Arumí, J.L.; Selker, J.S. Hillslope run-off thresholds with shrink–swell clay soils. Hydrol. Process. 2015, 29, 557–571. [Google Scholar] [CrossRef]
- Greve, A.; Andersen, M.; Acworth, R. Investigations of soil cracking and preferential flow in a weighing lysimeter filled with cracking clay soil. J. Hydrol. 2010, 393, 105–113. [Google Scholar] [CrossRef]
- Arnold, J.; Potter, K.; King, K.; Allen, P. Estimation of soil cracking and the effect on surface runoff in a texas blackland prairie watershed. Hydrol. Process. 2005, 19, 589–603. [Google Scholar] [CrossRef]
- Huguenin-Elie, O.; Kirk, G.; Frossard, E. Phosphorus uptake by rice from soil that is flooded, drained or flooded then drained. Eur. J. Soil Sci. 2003, 54, 77–90. [Google Scholar] [CrossRef]
- Wissuwa, M.; Ismail, A.M.; Yanagihara, S. Effects of zinc deficiency on rice growth and genetic factors contributing to tolerance. Plant Physiol. 2006, 142, 731–741. [Google Scholar] [CrossRef]
- Sahrawat, K.L. Soil fertility in flooded and non-flooded irrigated rice systems. Arch. Agron. Soil Sci. 2012, 58, 423–436. [Google Scholar] [CrossRef]
- Belder, P.; Bouman, B.; Cabangon, R.; Guoan, L.; Quilang, E.; Yuanhua, L.; Spiertz, J.; Tuong, T. Effect of water-saving irrigation on rice yield and water use in typical lowland conditions in asia. Agric. Water Manag. 2004, 65, 193–210. [Google Scholar] [CrossRef]
- Carrijo, D.R.; Lundy, M.E.; Linquist, B.A. Rice yields and water use under alternate wetting and drying irrigation: A meta-analysis. Field Crops Res. 2017, 203, 173–180. [Google Scholar] [CrossRef]
- Bezborodov, G.; Shadmanov, D.; Mirhashimov, R.; Yuldashev, T.; Qureshi, A.S.; Noble, A.; Qadir, M. Mulching and water quality effects on soil salinity and sodicity dynamics and cotton productivity in central asia. Agric. Ecosyst. Environ. 2010, 138, 95–102. [Google Scholar] [CrossRef]
- Hamoud, Y.A.; Guo, X.; Wang, Z.; Chen, S.; Rasoul, G. Effects of irrigation water regime, soil clay content and their combination on growth, yield, and water use efficiency of rice grown in south china. Int. J. Agric. Biol. Eng. 2018, 11, 144–155. [Google Scholar]
- Shaghaleh, H.; Xu, X.; Wang, S. Current progress in production of biopolymeric materials based on cellulose, cellulose nanofibers, and cellulose derivatives. RSC Adv. 2018, 8, 825–842. [Google Scholar] [CrossRef] [PubMed]
- Shaghaleh, H.; Xu, X.; Liu, H.; Wang, S.; Hamoud, Y.A.; Dong, F.; Luo, J. The effect of atmospheric pressure plasma pretreatment with various gases on the structural characteristics and chemical composition of wheat straw and applications to enzymatic hydrolysis. Energy 2019, 176, 195–210. [Google Scholar] [CrossRef]
- García, C.; Montero, G.; Coronado, M.; Torres, R.; Vázquez, A.; Ayala, J.; León, J.; Sagaste, C. Power generation estimation from wheat straw in mexico. Energy Sustain. VI WIT Trans. Ecol. Environ. 2015, 195, 101–110. [Google Scholar]
- Oanh, N.T.K.; Ly, B.T.; Tipayarom, D.; Manandhar, B.R.; Prapat, P.; Simpson, C.D.; Liu, L.-J.S. Characterization of particulate matter emission from open burning of rice straw. Atmos. Environ. 2011, 45, 493–502. [Google Scholar] [CrossRef]
- Qu, C.; Li, B.; Wu, H.; Giesy, J.P. Controlling Air Pollution from Straw Burning in China Calls for Efficient Recycling; ACS Publications: Washington, DC, USA, 2012. [Google Scholar]
- Zhang, Y.; Liu, M.; Dannenmann, M.; Tao, Y.; Yao, Z.; Jing, R.; Zheng, X.; Butterbach-Bahl, K.; Lin, S. Benefit of using biodegradable film on rice grain yield and n use efficiency in ground cover rice production system. Field Crops Res. 2017, 201, 52–59. [Google Scholar] [CrossRef]
- Liu, S.; Yang, J.; Zhang, X.; Drury, C.; Reynolds, W.; Hoogenboom, G. Modelling crop yield, soil water content and soil temperature for a soybean–maize rotation under conventional and conservation tillage systems in northeast china. Agric. Water Manag. 2013, 123, 32–44. [Google Scholar] [CrossRef]
- Jin, X.; Zuo, Q.; Ma, W.; Li, S.; Shi, J.; Tao, Y.; Zhang, Y.; Liu, Y.; Liu, X.; Lin, S. Water consumption and water-saving characteristics of a ground cover rice production system. J. Hydrol. 2016, 540, 220–231. [Google Scholar] [CrossRef]
- Liu, M.; Dannenmann, M.; Lin, S.; Saiz, G.; Yan, G.; Yao, Z.; Pelster, D.; Tao, H.; Sippel, S.; Tao, Y. Ground cover rice production system facilitates soil carbon and nitrogen stocks at regional scale. Biogeosci. Discuss. 2015, 12, 3647. [Google Scholar]
- Bunna, S.; Sinath, P.; Makara, O.; Mitchell, J.; Fukai, S. Effects of straw mulch on mungbean yield in rice fields with strongly compacted soils. Field Crops Res. 2011, 124, 295–301. [Google Scholar] [CrossRef]
- Chen, S.; Zhang, X.; Pei, D.; Sun, H.; Chen, S. Effects of straw mulching on soil temperature, evaporation and yield of winter wheat: Field experiments on the north china plain. Ann. Appl. Biol. 2007, 150, 261–268. [Google Scholar] [CrossRef]
- Humphreys, E.; Eberbach, P.; Katupitiya, A.; Kukal, S. Growth, yield and water productivity of zero till wheat as affected by rice straw mulch and irrigation schedule. Field Crops Res. 2011, 121, 209–225. [Google Scholar]
- Turmel, M.-S.; Speratti, A.; Baudron, F.; Verhulst, N.; Govaerts, B. Crop residue management and soil health: A systems analysis. Agric. Syst. 2015, 134, 6–16. [Google Scholar] [CrossRef]
- Chen, S.; Zhang, Z.; Wang, Z.; Guo, X.; Liu, M.; Hamoud, Y.A.; Zheng, J.; Qiu, R. Effects of uneven vertical distribution of soil salinity under a buried straw layer on the growth, fruit yield, and fruit quality of tomato plants. Sci. Hortic. 2016, 203, 131–142. [Google Scholar] [CrossRef]
- Chen, S.; Wang, Z.; Guo, X.; Rasool, G.; Zhang, J.; Xie, Y.; Yousef, A.H.; Shao, G. Effects of vertically heterogeneous soil salinity on tomato photosynthesis and related physiological parameters. Sci. Hortic. 2019, 249, 120–130. [Google Scholar] [CrossRef]
- Rasool, G.; Xiangping, G.; Zhenchang, W.; Sheng, C.; Alhaj Hamoud, Y.; Javed, Q. Response of fertigation under buried straw layer on growth, yield, and water-fertilizer productivity of chinese cabbage under greenhouse conditions. Commun. Soil Sci. Plant Anal. 2019, 50, 1030–1043. [Google Scholar] [CrossRef]
- Zhang, S.; Kong, D.; Chang, X.; Zhai, L. Effect of straw deep application on soil water storage capacity. J. Northeast Agric. Univ. 2010, 6, 034. [Google Scholar]
- Cao, J.; Liu, C.; Zhang, W.; Guo, Y. Effect of integrating straw into agricultural soils on soil infiltration and evaporation. Water Sci. Technol. 2012, 65, 2213–2218. [Google Scholar] [CrossRef]
- Kaur, J.; Bons, H.K. Mulching: A viable option to increase productivity of field and fruit crops. J. Appl. Nat. Sci. 2017, 9, 974–982. [Google Scholar] [CrossRef]
- Palta, J.A.; Yang, J. Crop root system behaviour and yield. Field Crops Res. 2014, 165, 1–149. [Google Scholar] [CrossRef]
- Petricka, J.J.; Winter, C.M.; Benfey, P.N. Control of arabidopsis root development. Annu. Rev. Plant Biol. 2012, 63, 563–590. [Google Scholar] [CrossRef]
- Hames, B.; Ruiz, R.; Scarlata, C.; Sluiter, A.; Sluiter, J.; Templeton, D. Preparation of samples for compositional analysis. Lab. Anal. Proced. (LAP) 2008, 1617, 65–71. [Google Scholar]
- Daly, P.; van Munster, J.M.; Blythe, M.J.; Ibbett, R.; Kokolski, M.; Gaddipati, S.; Lindquist, E.; Singan, V.R.; Barry, K.W.; Lipzen, A.; et al. Expression of aspergillus niger cazymes is determined by compositional changes in wheat straw generated by hydrothermal or ionic liquid pretreatments. Biotechnol. Biofuels 2017, 10, 35. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.; Yang, L.; Yang, Y.; Ouyang, Z. Rice root growth and nutrient uptake as influenced by organic manure in continuously and alternately flooded paddy soils. Agric. Water Manag. 2004, 70, 67–81. [Google Scholar] [CrossRef]
- Tennant, D. A test of a modified line intersect method of estimating root length. J. Ecol. 1975, 6, 995–1001. [Google Scholar] [CrossRef]
- Boero, G.; Thien, S. Phosphatase activity and phosphorus availability in the rhizosphere of corn roots. In The Soil–Root Interface; Elsevier: Amsterdam, The Netherlands, 1979; pp. 231–242. [Google Scholar]
- Zhang, X.; Tan, G.; Huang, Y. Experimental technology of plant physiology. Liaoning Sci. Technol. Press Shenyang China 1994, 21, 51–75. [Google Scholar]
- Bouman, B.; Peng, S.; Castaneda, A.; Visperas, R. Yield and water use of irrigated tropical aerobic rice systems. Agric. Water Manag. 2005, 74, 87–105. [Google Scholar] [CrossRef]
- Shao, G.-C.; Deng, S.; Liu, N.; Yu, S.-E.; Wang, M.-H.; She, D.-L. Effects of controlled irrigation and drainage on growth, grain yield and water use in paddy rice. Eur. J. Agron. 2014, 53, 1–9. [Google Scholar] [CrossRef]
- Zhao, Y.; Li, Y.; Wang, J.; Pang, H.; Li, Y. Buried straw layer plus plastic mulching reduces soil salinity and increases sunflower yield in saline soils. Soil Tillage Res. 2016, 155, 363–370. [Google Scholar] [CrossRef]
- Lampayan, R.M.; Rejesus, R.M.; Singleton, G.R.; Bouman, B.A. Adoption and economics of alternate wetting and drying water management for irrigated lowland rice. Field Crops Res. 2015, 170, 95–108. [Google Scholar] [CrossRef]
- Chu, G.; Wang, Z.; Zhang, H.; Liu, L.; Yang, J.; Zhang, J. Alternate wetting and moderate drying increases rice yield and reduces methane emission in paddy field with wheat straw residue incorporation. Food Energy Secur. 2015, 4, 238–254. [Google Scholar] [CrossRef]
- Gao, C.; El-Sawah, A.M.; Ali, D.F.I.; Hamoud, Y.A.; Shaghaleh, H.; Sheteiwy, M.S. The integration of bio and organic fertilizers improve plant growth, grain yield, quality and metabolism of hybrid maize (Zea mays L.). Agronomy 2020, 10, 319. [Google Scholar] [CrossRef]
- Sharma, P.K.; Verma, T.S.; Bhagat, R.M. Soil structural improvements with the addition of lantana camara biomass in rice-wheat cropping. Soil Use Manag. 1995, 11, 199–203. [Google Scholar] [CrossRef]
- Bandyopadhyay, K.; Mohanty, M.; Painuli, D.; Misra, A.; Hati, K.; Mandal, K.; Ghosh, P.; Chaudhary, R.; Acharya, C. Influence of tillage practices and nutrient management on crack parameters in a vertisol of central india. Soil Tillage Res. 2003, 71, 133–142. [Google Scholar] [CrossRef]
- Yang, J.-C.; Zhang, H.; Zhang, J.-H. Root morphology and physiology in relation to the yield formation of rice. J. Integr. Agric. 2012, 11, 920–926. [Google Scholar] [CrossRef]
- Kondhia, A.; Tabien, R.E.; Ibrahim, A. Evaluation and selection of high biomass rice (Oryza sativa L.) for drought tolerance. Am. J. Plant Sci. 2015, 6, 1962. [Google Scholar] [CrossRef]
- Sheteiwy, M.S.; Shao, H.; Qi, W.; Hamoud, Y.A.; Shaghaleh, H.; Khan, N.U.; Yang, R.; Tang, B. Gaba-alleviated oxidative injury induced by salinity, osmotic stress and their combination by regulating cellular and molecular signals in rice. Int. J. Mol. Sci. 2019, 20, 5709. [Google Scholar] [CrossRef]
- Colmer, T.D.; Pedersen, O. Oxygen dynamics in submerged rice (Oryza sativa). New Phytol. 2008, 178, 326–334. [Google Scholar] [CrossRef] [PubMed]
- Colmer, T. Long-distance transport of gases in plants: A perspective on internal aeration and radial oxygen loss from roots. Plant Cell Environ. 2003, 26, 17–36. [Google Scholar] [CrossRef]
- Evans, D.E. Aerenchyma formation. New Phytol. 2004, 161, 35–49. [Google Scholar] [CrossRef]
- Shiono, K.; Ogawa, S.; Yamazaki, S.; Isoda, H.; Fujimura, T.; Nakazono, M.; Colmer, T.D. Contrasting dynamics of radial o2-loss barrier induction and aerenchyma formation in rice roots of two lengths. Ann. Bot. 2010, 107, 89–99. [Google Scholar] [CrossRef] [PubMed]
- Kirk, G.J. Rice root properties for internal aeration and efficient nutrient acquisition in submerged soil. New Phytol. 2003, 159, 185–194. [Google Scholar] [CrossRef] [PubMed]
- Mishra, A.; Salokhe, V. Flooding stress: The effects of planting pattern and water regime on root morphology, physiology and grain yield of rice. J. Agron. Crop Sci. 2010, 196, 368–378. [Google Scholar] [CrossRef]
- Mishra, A.; Uphoff, N. Morphological and physiological responses of rice roots and shoots to varying water regimes and soil microbial densities. Arch. Agron. Soil Sci. 2013, 59, 705–731. [Google Scholar] [CrossRef]
- Ju, C.; Buresh, R.J.; Wang, Z.; Zhang, H.; Liu, L.; Yang, J.; Zhang, J. Root and shoot traits for rice varieties with higher grain yield and higher nitrogen use efficiency at lower nitrogen rates application. Field Crops Res. 2015, 175, 47–55. [Google Scholar] [CrossRef]
- Bunnag, S.; Pongthai, P. Selection of rice (Oryza sativa L.) cultivars tolerant to drought stress at the vegetative stage under field conditions. Am. J. Plant Sci. 2013, 4, 1701. [Google Scholar] [CrossRef]
- Dou, F.; Soriano, J.; Tabien, R.E.; Chen, K. Soil texture and cultivar effects on rice (Oryza sativa, L.) grain yield, yield components and water productivity in three water regimes. PLoS ONE 2016, 11, e0150549. [Google Scholar] [CrossRef] [PubMed]
- Carmeis Filho, A.C.d.A.; Crusciol, C.A.C.; Nascente, A.S.; Mauad, M.; Garcia, R.A. Influence of potassium levels on root growth and nutrient uptake of upland rice cultivars. Rev. Caatinga 2017, 30, 32–44. [Google Scholar] [CrossRef]
- Huang, M.; Zou, Y.-b.; Jiang, P.; Bing, X.; Md, I.; Ao, H.-J. Relationship between grain yield and yield components in super hybrid rice. Agric. Sci. China 2011, 10, 1537–1544. [Google Scholar] [CrossRef]
- Belder, P.; Spiertz, J.; Bouman, B.; Lu, G.; Tuong, T. Nitrogen economy and water productivity of lowland rice under water-saving irrigation. Field Crops Res. 2005, 93, 169–185. [Google Scholar] [CrossRef]
- Bouman, B.; Tuong, T.P. Field water management to save water and increase its productivity in irrigated lowland rice. Agric. Water Manag. 2001, 49, 11–30. [Google Scholar] [CrossRef]
- Huguenin-Elie, O.; Kirk, G.; Frossard, E. The effects of water regime on phosphorus responses of rainfed lowland rice cultivars. Ann. Bot. 2008, 103, 211–220. [Google Scholar] [CrossRef]
- Siavoshi, M.; Laware, S.L. Effect of organic fertilizer on growth and yield components in rice (Oryza sativa L.). J. Agric. Sci. 2011, 3, 217. [Google Scholar] [CrossRef]
- Xiang, J.; Haden, V.R.; Peng, S.; Bouman, B.A.; Huang, J.; Cui, K.; Visperas, R.M.; Zhu, D.; Zhang, Y.; Chen, H. Effect of deep placement of nitrogen fertilizer on growth, yield, and nitrogen uptake of aerobic rice. Aust. J. Crop Sci. 2013, 7, 870. [Google Scholar]
- Ding, C.; You, J.; Chen, L.; Wang, S.; Ding, Y. Nitrogen fertilizer increases spikelet number per panicle by enhancing cytokinin synthesis in rice. Plant Cell Rep. 2014, 33, 363–371. [Google Scholar] [CrossRef]



| Average Data | May | Jun | Jul | Aug | Sep | Oct |
|---|---|---|---|---|---|---|
| TMax (°C) | 30.7 | 31.8 | 32.1 | 39.3 | 29.8 | 27.4 |
| TMin (°C) | 18.4 | 20.9 | 22.5 | 28.5 | 16.7 | 12.9 |
| Max. Relative humidity % | 95 | 84.5 | 100 | 84.5 | 88.8 | 73.8 |
| Min. Relative humidity % | 68.3 | 61.9 | 68.3 | 61.9 | 67.5 | 66.5 |
| Sunshine (h) | 8 | 9 | 11 | 11 | 9 | 7 |
| Solar Rad. MJ m−2 day−1 | 96.3 | 113.6 | 96.5 | 113.2 | 78 | 72.8 |
| Rainfall (mm) | 104.6 | 188.5 | 172.1 | 135.4 | 85.3 | 65.9 |
| Treatment | Water Use Indicator | Irrigations Number | |||
|---|---|---|---|---|---|
| AWD Regime | Straw Burial | ETc (mm) | Dp (mm) | TI (mm) | IN (Events) |
| AWMD | NSB | 579.5 ± 19.51 Aa | 273.6 ± 10.7 Aa | 853.1 ± 9.07 Aa | 17 ± 0.47 Aa |
| LSB | 523.2 ± 13.79 Aa | 277.0 ± 3.7 Aa | 800.2 ± 10.28 Aa | 16 ± 0.47 Aa | |
| DSB | 501.1 ± 4.13 Aa | 240.8 ± 7.6 Ab | 751.9 ± 11.73 Ab | 14 ± 0.24 Ab | |
| AWSD | NSB | 418.3 ± 14.54 Bb | 160.7 ± 6.3 Ba | 579.0 ± 8.60 Ba | 13 ± 0.48 Ba |
| LSB | 361.7 ± 9.73 Bc | 153.1 ± 2.7 Ba | 514.8 ± 7.54 Ba | 11 ± 0.32 Ba | |
| DSB | 352.3 ± 13.79 Bc | 135.9 ± 3.8 Bb | 478.3 ± 10.28 Bb | 11 ± 0.77 Ba | |
| AWCD | NSB | 231.8 ± 6.57 Cd | 128.5 ± 1.25 Ca | 365.4 ± 7.21 Ca | 11 ± 0.41 Ca |
| LSB | 214.6 ± 6.73 Cd | 118.6 ± 4.2 Ca | 333.2 ± 3.6 Ca | 9 ± 0.35 Ca | |
| DSB | 207.4 ± 3.61 Cd | 104.4 ± 4.3 Cb | 307.8 ± 7.90 Cb | 7 ± 0.68 Cb | |
| ANOVA | |||||
| AWD regime | * | * | ** | * | |
| Straw burial | ** | *** | *** | *** | |
| AWD × straw | ** | ** | * | ns | |
| Treatment | Water Use Efficiency Indicator | ||||
|---|---|---|---|---|---|
| AWD Regime | Straw Burial | CWUEY (kg/m3) | IWUEY (kg /m3) | CWUEB (kg/m3) | IWUEB (kg/m3) |
| AWMD | NSB | 0.81 ± 0.02 Cb | 0.52 ± 0.01 Cc | 2.13 ± 0.09 Cb | 1.40 ± 0.05 Cb |
| LSB | 1.15 ± 0.02 Ca | 0.82 ± 0.03 Cb | 2.80 ± 0.10 Ca | 1.83 ± 0.04 Cb | |
| DSB | 1.46 ± 0.04 Ca | 1.02 ± 0.02 Ca | 3.05 ± 0.07 Ca | 2.51 ± 0.06 Ca | |
| AWSD | NSB | 0.96 ± 0.01 Ab | 0.70 ± 0.01 Ab | 2.54 ± 0.11 Ac | 1.69 ± 0.07 Ab |
| LSB | 1.39 ± 0.08 Aa | 0.98 ± 0.05 Ab | 3.53 ± 0.12 Ab | 2.44 ± 0.10 Aa | |
| DSB | 1.87 ± 0.05 Aa | 1.47 ± 0.07 Aa | 4.31 ± 0.09 Aa | 3.05 ± 0.06 Aa | |
| AWCD | NSB | 0.87 ± 0.04 Bb | 0.61 ± 0.03 Bb | 2.22 ± 0.10 Bb | 1.51 ± 0.02 Bb |
| LSB | 1.25 ± 0.07 Ba | 0.87 ± 0.02 Bb | 3.33 ± 0.09 Ba | 2.13 ± 0.04 Ba | |
| DSB | 1.62 ± 0.09 Ba | 1.11 ± 0.06 Ba | 3.49 ± 0.11 Ba | 2.62 ± 0.08 Ba | |
| ANOVA | |||||
| AWD regime | * | * | ** | * | |
| Straw burial | *** | ** | ** | *** | |
| AWD × Straw | *** | *** | * | * | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Franz Gouertoumbo, W.; Alhaj Hamoud, Y.; Guo, X.; Shaghaleh, H.; Ali Adam Hamad, A.; Elsadek, E. Wheat Straw Burial Enhances the Root Physiology, Productivity, and Water Utilization Efficiency of Rice under Alternative Wetting and Drying Irrigation. Sustainability 2022, 14, 16394. https://doi.org/10.3390/su142416394
Franz Gouertoumbo W, Alhaj Hamoud Y, Guo X, Shaghaleh H, Ali Adam Hamad A, Elsadek E. Wheat Straw Burial Enhances the Root Physiology, Productivity, and Water Utilization Efficiency of Rice under Alternative Wetting and Drying Irrigation. Sustainability. 2022; 14(24):16394. https://doi.org/10.3390/su142416394
Chicago/Turabian StyleFranz Gouertoumbo, Willy, Yousef Alhaj Hamoud, Xiangping Guo, Hiba Shaghaleh, Amar Ali Adam Hamad, and Elsayed Elsadek. 2022. "Wheat Straw Burial Enhances the Root Physiology, Productivity, and Water Utilization Efficiency of Rice under Alternative Wetting and Drying Irrigation" Sustainability 14, no. 24: 16394. https://doi.org/10.3390/su142416394
APA StyleFranz Gouertoumbo, W., Alhaj Hamoud, Y., Guo, X., Shaghaleh, H., Ali Adam Hamad, A., & Elsadek, E. (2022). Wheat Straw Burial Enhances the Root Physiology, Productivity, and Water Utilization Efficiency of Rice under Alternative Wetting and Drying Irrigation. Sustainability, 14(24), 16394. https://doi.org/10.3390/su142416394










