Residency, Site Fidelity, and Regional Movement of Tiger Sharks (Galeocerdo cuvier) at a Pupping Location in the Bahamas
Abstract
:1. Introduction
2. Methods
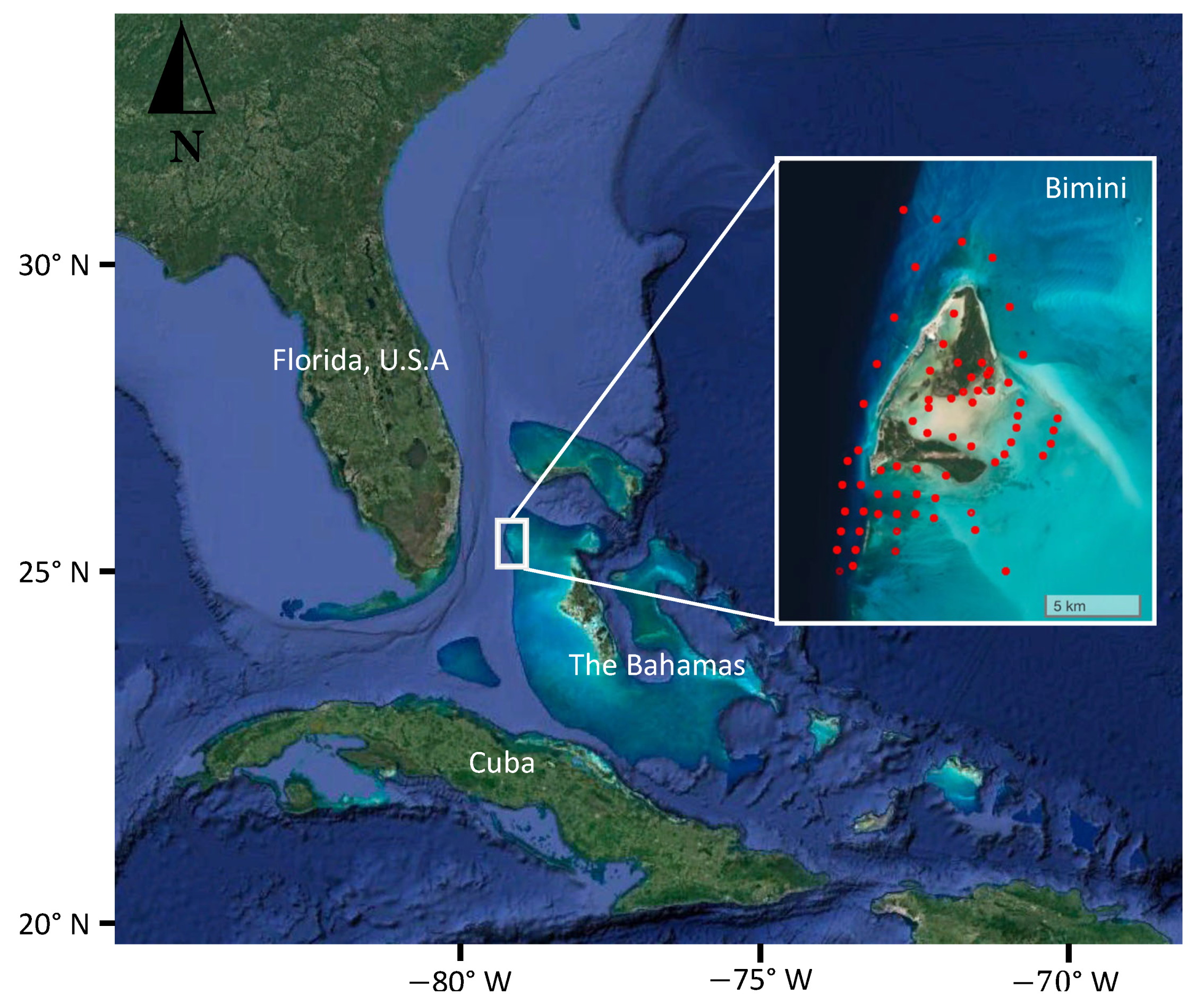
2.1. Study Location
2.2. Capture Methods
2.3. Tagging
2.4. Data Analysis
3. Results
3.1. Captures
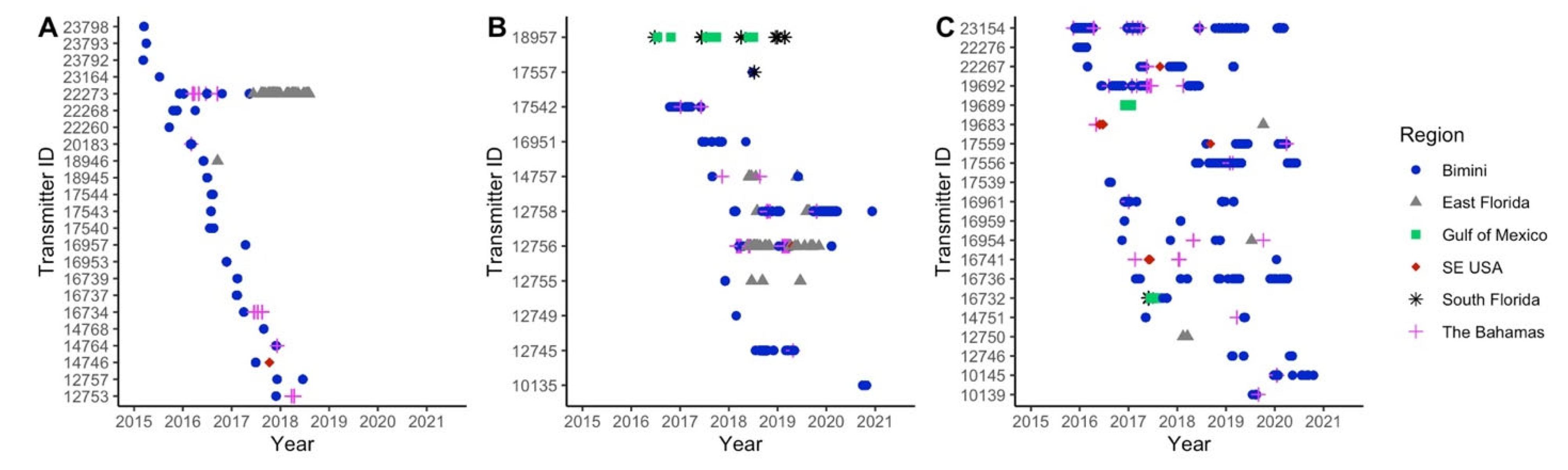
3.2. Local and Regional Recaptures
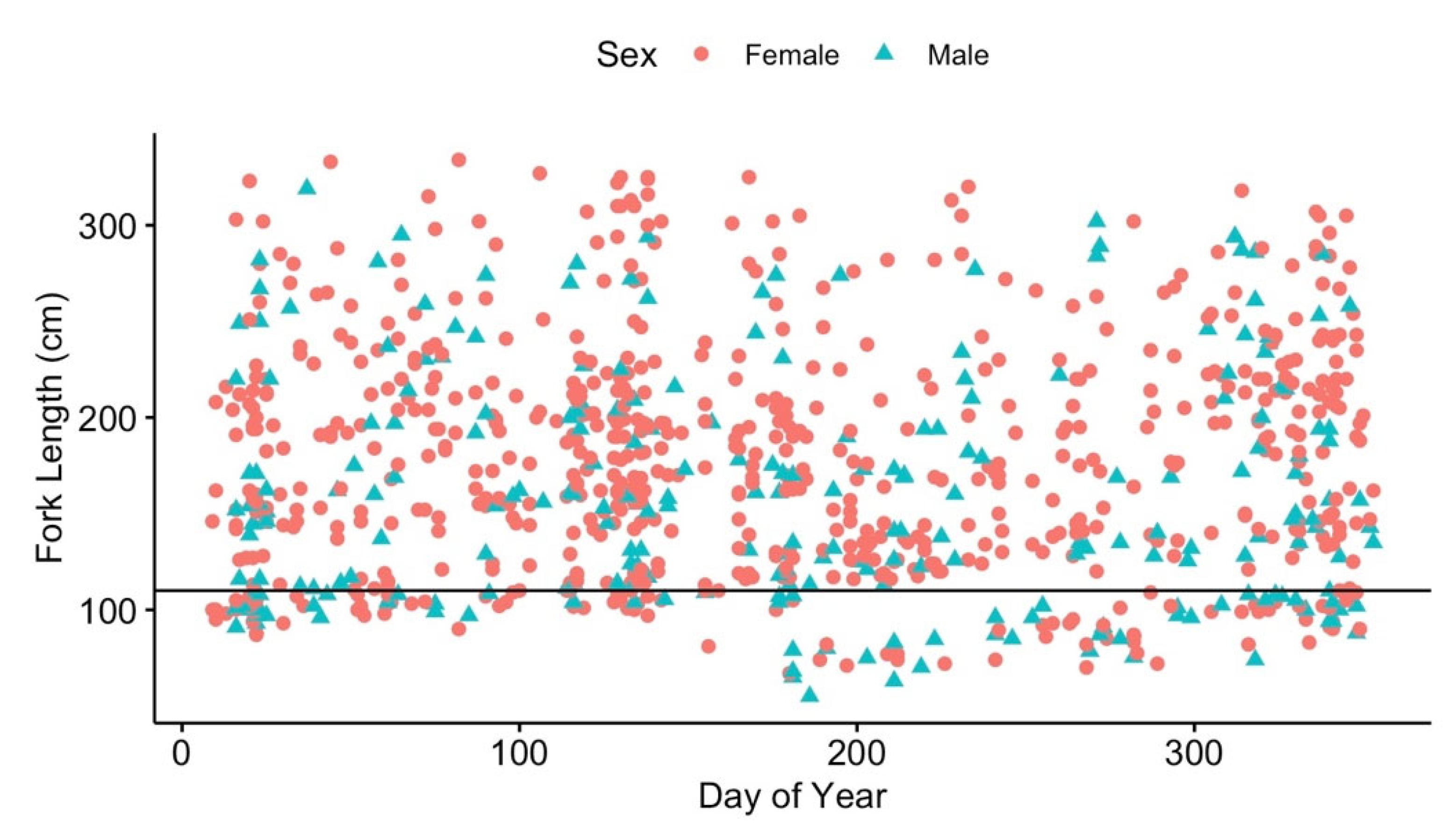
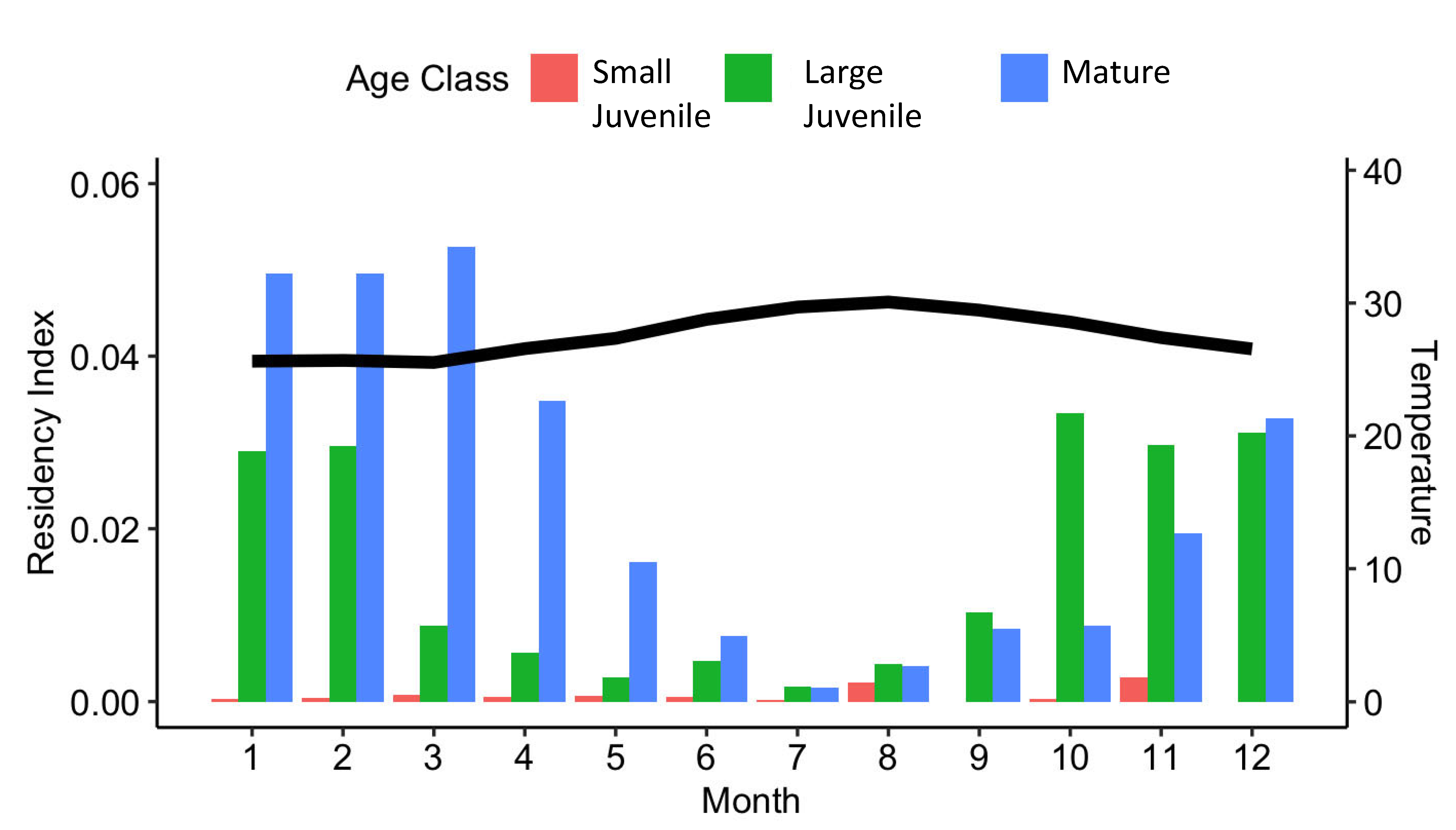
3.3. Local Residency
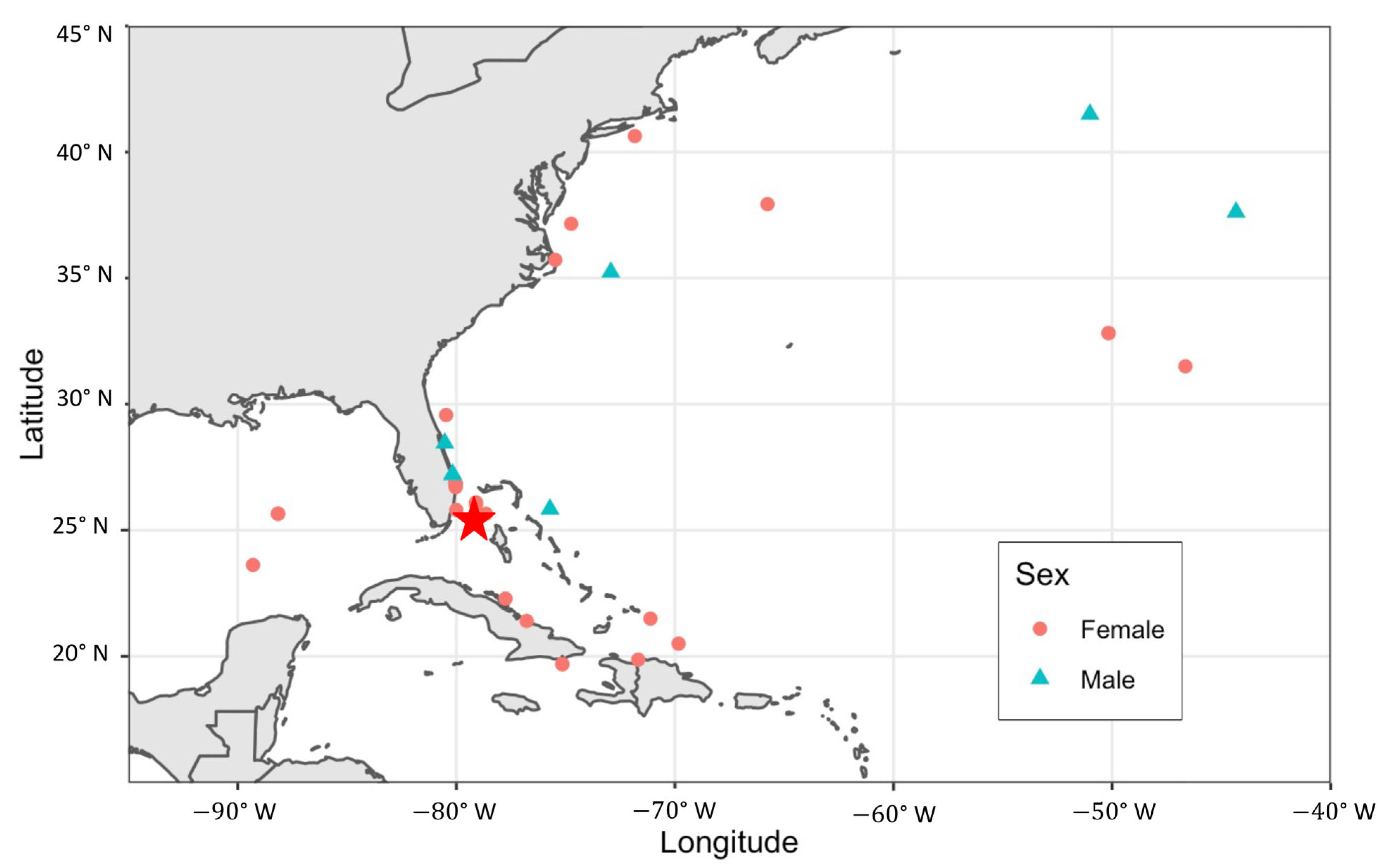
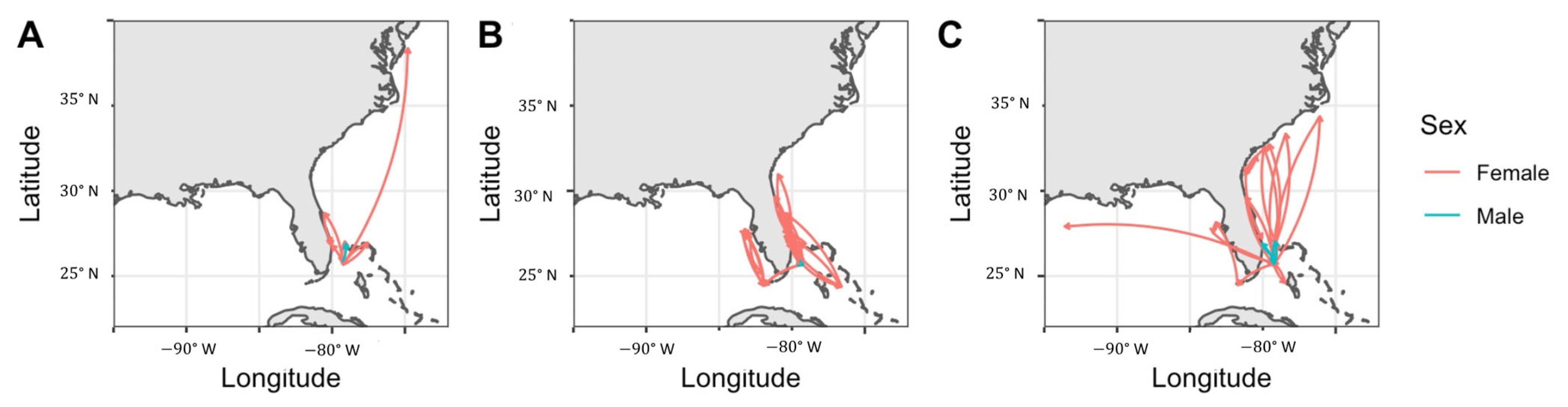
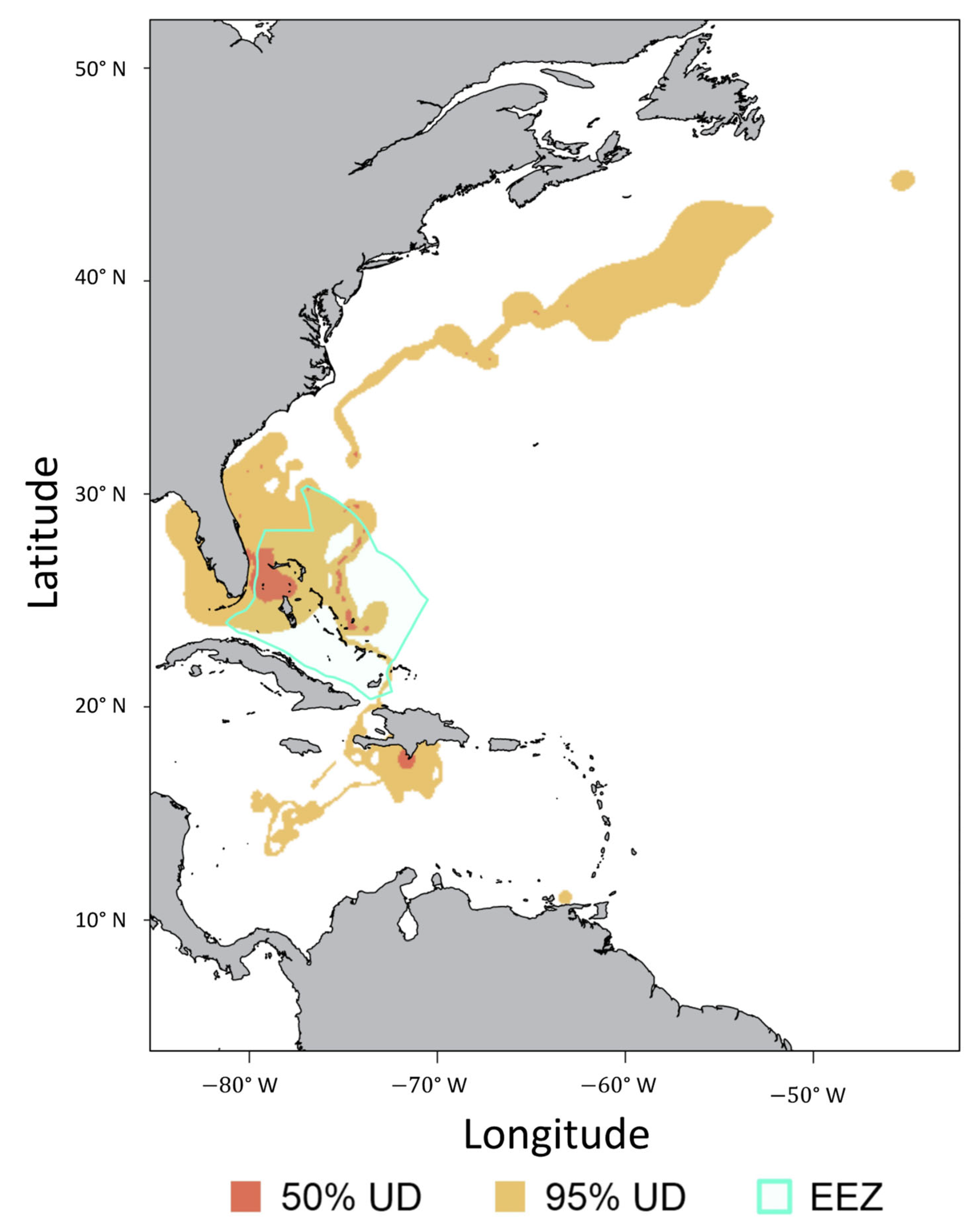
3.4. Regional Movements
4. Discussion
4.1. Overview
4.2. Evidence for a Pupping Location
4.3. Residency and Site Fidelity
4.4. Regional Movements and Connectivity
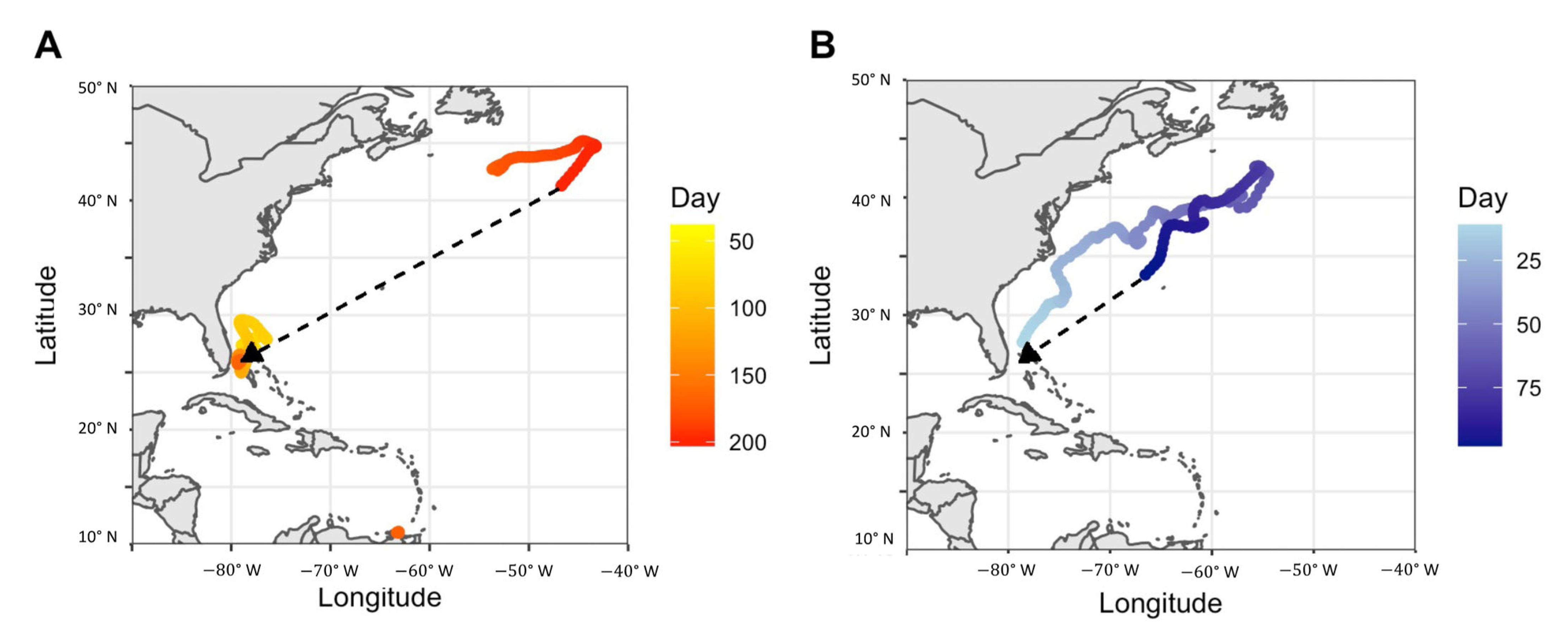
4.5. Notable Individual Movements
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Acknowledgments
Conflicts of Interest
References
- MacNeil, M.A.; Chapman, D.D.; Heupel, M.; Simpfendorfer, C.A.; Heithaus, M.; Meekan, M.; Harvey, E.; Goetze, J.; Kiszka, J.; Bond, M.E.; et al. Global Status and Conservation Potential of Reef Sharks. Nature 2020, 583, 801–806. [Google Scholar] [CrossRef]
- Dulvy, N.K.; Pacoureau, N.; Rigby, C.L.; Pollom, R.A.; Jabado, R.W.; Ebert, D.A.; Finucci, B.; Pollock, C.M.; Cheok, J.; Derrick, D.H.; et al. Overfishing Drives over One-Third of All Sharks and Rays toward a Global Extinction Crisis. Curr. Biol. 2021, 31, 4773–4787.e8. [Google Scholar] [CrossRef] [PubMed]
- Hays, G.C.; Bailey, H.; Bograd, S.J.; Bowen, W.D.; Campagna, C.; Carmichael, R.H.; Casale, P.; Chiaradia, A.; Costa, D.P.; Cuevas, E.; et al. Translating Marine Animal Tracking Data into Conservation Policy and Management. Trends Ecol. Evol. 2019, 34, 459–473. [Google Scholar] [CrossRef] [PubMed]
- Queiroz, N.; Humphries, N.E.; Couto, A.; Vedor, M.; Da Costa, I.; Sequeira, A.M.; Mucientes, G.; Santos, A.M.; Abascal, F.J.; Abercrombie, D.L.; et al. Global Spatial Risk Assessment of Sharks under the Footprint of Fisheries. Nature 2019, 572, 461–466. [Google Scholar] [CrossRef]
- Guttridge, T.L.; Bergmann, M.P.M.V.Z.; Bolte, C.; Howey, L.A.; Finger, J.S.; Kessel, S.T.; Brooks, J.L.; Winram, W.; Bond, M.E.; Jordan, L.K.B.; et al. Philopatry and Regional Connectivity of the Great Hammerhead Shark, Sphyrna Mokarran in the U.S. and Bahamas. Front. Mar. Sci. 2017, 4, 1–15. [Google Scholar] [CrossRef]
- Lennox, R.J.; Engler-Palma, C.; Kowarski, K.; Filous, A.; Whitlock, R.; Cooke, S.J.; Auger-Méthé, M. Optimizing Marine Spatial Plans with Animal Tracking Data. Can. J. Fish. Aquat. Sci. 2019, 76, 497–509. [Google Scholar] [CrossRef]
- Bergmann, M.P.v.Z.; Guttridge, T.L.; Smukall, M.J.; Adams, V.M.; Bond, M.E.; Burke, P.J.; Fuentes, M.M.; Heinrich, D.D.; Huveneers, C.; Gruber, S.H.; et al. Using Movement Models and Systematic Conservation Planning to Inform Marine Protected Area Design for a Multi-Species Predator Community. Biol. Conserv. 2022, 266, 109469. [Google Scholar] [CrossRef]
- Bangley, C.W.; Curtis, T.H.; Secor, D.H.; Latour, R.J.; Ogburn, M.B. Identifying Important Juvenile Dusky Shark Habitat in the Northwest Atlantic Ocean Using Acoustic Telemetry and Spatial Modeling. Mar. Coast. Fish. 2020, 12, 348–363. [Google Scholar] [CrossRef]
- Heupel, M.R.; Kanno, S.; Martins, A.; Simpfendorfer, C.A. Advances in Understanding the Roles and Benefits of Nursery Areas for Elasmobranch Populations. Mar. Freshw. Res. 2019, 70, 897–907. [Google Scholar] [CrossRef]
- Chapman, D.D.; Babcock, E.A.; Gruber, S.H.; Dibattista, J.D.; Franks, B.R.; Kessel, S.A.; Guttridge, T.; Pikitch, E.K.; Feldheim, K.A. Long-Term Natal Site-Fidelity by Immature Lemon Sharks Negaprion brevirostris at a Subtropical Island. Mol. Ecol. 2009, 18, 3500–3507. [Google Scholar] [CrossRef]
- Grubbs, R.D.; Musick, J.A.; Conrath, C.L.; Romine, J.G. Long-Term Movements, Migration, and Temporal Delineation of a Summer Nursery for Juvenile Sandbar Sharks in the Chesapeake Bay Region. Am. Fish. Soc. Symp. 2007, 50, 87–107. [Google Scholar]
- Vandeperre, F.; Aires-Da-Silva, A.; Fontes, J.; Santos, M.; Santos, R.S.; Afonso, P. Movements of Blue Sharks Prionace Glauca across Their Life History. PLoS ONE 2014, 9, e103538. [Google Scholar] [CrossRef] [PubMed]
- Heupel, M.; Carlson, J.K.; Simpfendorfer, C.A. Shark Nursery Areas: Concepts, Definition, Characterization and Assumptions. Mar. Ecol. Prog. Ser. 2007, 337, 287–297. [Google Scholar] [CrossRef]
- Froeschke, J.; Stunz, G.; Sterba-Boatwright, B.; Wildhaber, M. An Empirical Test of the ‘Shark Nursery Area Concept’ in Texas Bays Using a Long-Term Fisheries-Independent Data Set. Aquat. Biol. 2010, 11, 65–76. [Google Scholar] [CrossRef]
- Hollensead, L.; Grubbs, R.; Carlson, J.; Bethea, D. Assessing Residency Time and Habitat Use of Juvenile Smalltooth Sawfish Using Acoustic Monitoring in a Nursery Habitat. Endanger. Species Res. 2018, 37, 119–131. [Google Scholar] [CrossRef]
- Macdonald, C.; Jerome, J.; Pankow, C.; Perni, N.; Black, K.; Shiffman, D.; Wester, J. First Identification of Probable Nursery Habitat for Critically Endangered Great Hammerhead Sphyrna mokarran on the Atlantic Coast of the United States. Conserv. Sci. Pract. 2021, 3, e418. [Google Scholar] [CrossRef]
- Ferreira, L.C.; Simpfendorfer, C.C. Tiger Shark: Galeocerdo cuvier. The IUCN Red List of Threatened Species 8235. 2019. Available online: http://www.iucnredlist.org/details/39378/0 (accessed on 25 April 2022).
- Heithaus, M.; Dill, L.; Marshall, G.; Buhleier, B. Habitat Use and Foraging Behavior of Tiger Sharks Galeocerdo cuvier in a Seagrass Ecosystem. Mar. Biol. 2002, 140, 237–248. [Google Scholar] [CrossRef]
- Burkholder, D.A.; Heithaus, M.R.; Fourqurean, J.; Wirsing, A.; Dill, L.M. Patterns of Top-down Control in a Seagrass Ecosystem: Could a Roving Apex Predator Induce a Behaviour-Mediated Trophic Cascade? J. Anim. Ecol. 2013, 82, 1192–1202. [Google Scholar] [CrossRef]
- Ferreira, L.C.; Thums, M.; Heithaus, M.R.; Barnett, A.; Abrantes, K.; Holmes, B.J.; Zamora, L.M.; Frisch, A.J.; Pepperell, J.G.; Burkholder, D.; et al. The Trophic Role of a Large Marine Predator, the Tiger Shark Galeocerdo cuvier. Sci. Rep. 2017, 7, 7641. [Google Scholar] [CrossRef]
- Meyer, C.; Clark, T.; Papastamatiou, Y.; Whitney, N.; Holland, K. Long-Term Movement Patterns of Tiger Sharks Galeocerdo cuvier in Hawaii. Mar. Ecol. Prog. Ser. 2009, 381, 223–235. [Google Scholar] [CrossRef]
- Lea, J.S.E.; Wetherbee, B.M.; Queiroz, N.; Burnie, N.; Aming, C.; Sousa, L.L.; Mucientes, G.R.; Humphries, N.; Harvey, G.M.; Sims, D.; et al. Repeated, Long-Distance Migrations by a Philopatric Predator Targeting Highly Contrasting Ecosystems. Sci. Rep. 2015, 5, 11202. [Google Scholar] [CrossRef] [PubMed]
- Domingo, A.; Coelho, R.; Cortes, E.; Garcia-Cortes, B.; Mas, F.; Mejuto, J.; Miller, P.; Ramos-Cartelle, A.; Santos, M.N.; Yokawa, K. Is the Tiger Shark Galeocerdo cuvier a Coastal Species? Expanding Its Distribution Range in the Atlantic Ocean Using at-Sea Observer Data. J. Fish. Biol. 2016, 88, 1223–1228. [Google Scholar] [CrossRef] [PubMed]
- Lowe, C.G.; Wetherbee, B.M.; Meyer, C.G. Using Acoustic Telemetry Monitoring Techniques to Quantity Movement Patterns and Site Fidelity of Sharks and Giant Trevally around French Frigate Shoals and Midway Atoll. Atoll Res. Bull. 2006, 543, 281–303. [Google Scholar]
- Fitzpatrick, R.; Thums, M.; Bell, I.; Meekan, M.G.; Stevens, J.D.; Barnett, A. A Comparison of the Seasonal Movements of Tiger Sharks and Green Turtles Provides Insight into Their Predator-Prey Relationship. PLoS ONE 2012, 7, e051927. [Google Scholar] [CrossRef]
- Papastamatiou, Y.P.; Meyer, C.G.; Carvalho, F.; Dale, J.J.; Hutchinson, M.R.; Holland, K.N. Telemetry and Random-Walk Models Reveal Complex Patterns of Partial Migration in a Large Marine Predator. Ecology 2013, 94, 2595–2606. [Google Scholar] [CrossRef]
- Sulikowski, J.; Wheeler, C.; Gallagher, A.; Prohaska, B.; Langan, J.; Hammerschlag, N. Seasonal and Life-Stage Variation in the Reproductive Ecology of a Marine Apex Predator, the Tiger Shark Galeocerdo cuvier, at a Protected Female-Dominated Site. Aquat. Biol. 2016, 24, 175–184. [Google Scholar] [CrossRef]
- Hammerschlag, N.; McDonnell, L.H.; Rider, M.J.; Street, G.M.; Hazen, E.L.; Natanson, L.J.; McCandless, C.T.; Boudreau, M.R.; Gallagher, A.J.; Pinsky, M.L.; et al. Ocean Warming Alters the Distributional Range, Migratory Timing, and Spatial Protections of an Apex Predator, the Tiger Shark (Galeocerdo cuvier). Glob. Chang. Biol. 2022, 28, 1990–2005. [Google Scholar] [CrossRef]
- Holland, K.N.; Anderson, J.M.; Coffey, D.M.; Holmes, B.J.; Meyer, C.G.; Royer, M.A. A Perspective on Future Tiger Shark Research. Front. Mar. Sci. 2019, 6, 1–7. [Google Scholar] [CrossRef]
- Driggers, W.B., III; Ingram, G.W., Jr.; Grace, M.A.; Gledhill, C.T.; Henwood, T.A.; Horton, C.N.; Jones, C.M. Pupping Areas and Mortality Rates of Young Tiger Sharks Galeocerdo cuvier in the Western North Atlantic Ocean. Aquat. Biol. 2008, 2, 161–170. [Google Scholar] [CrossRef]
- Hansell, A.C.; Kessel, S.T.; Brewster, L.R.; Cadrin, S.X.; Gruber, S.H.; Skomal, G.B.; Guttridge, T.L. Local Indicators of Abundance and Demographics for the Coastal Shark Assemblage of Bimini, Bahamas. Fish. Res. 2018, 197, 34–44. [Google Scholar] [CrossRef]
- Smukall, M.J.; Carlson, J.; Kessel, S.T.; Guttridge, T.L.; Dhellemmes, F.; Seitz, A.C.; Gruber, S. Thirty-five Years of Tiger Shark Galeocerdo cuvier Relative Abundance near Bimini, The Bahamas, and the Southeastern United States with a Comparison across Jurisdictional Bounds. J. Fish. Biol. 2022, 101, 13–25. [Google Scholar] [CrossRef] [PubMed]
- Gallagher, A.J.; Wagner, D.N.; Irschick, D.J.; Hammerschlag, N. Body Condition Predicts Energy Stores in Apex Predatory Sharks. Conserv. Physiol. 2014, 2, cou022. [Google Scholar] [CrossRef] [PubMed]
- Brooks, E.J.; Brooks, A.M.; Williams, S.; Jordan, L.K.; Abercrombie, D.; Chapman, D.D.; Howey-Jordan, L.A.; Grubbs, R.D. First Description of Deep-Water Elasmobranch Assemblages in the Exuma Sound, The Bahamas. Deep Sea Res. Part II Top. Stud. Oceanogr. 2015, 115, 81–91. [Google Scholar] [CrossRef]
- Branstetter, S.; Musick, J.A.; Colvocoresses, J.A. A Comparison of the Age and Growth of the Tiger Shark, Galeocerdo cuvier, From Off Virginia and From the Northwestern Gulf of Mexico. Fish. Bull. 1987, 85, 269–279. [Google Scholar]
- Kneebone, J.; Natanson, L.J.; Andrews, A.H.; Howell, W.H. Using Bomb Radiocarbon Analyses to Validate Age and Growth Estimates for the Tiger Shark, Galeocerdo cuvier, in the Western North Atlantic. Mar. Biol. 2008, 154, 423–434. [Google Scholar] [CrossRef]
- Kessel, S.T.; Hussey, N.E. Tonic immobility as an anesthetic for elasmobranchs during surgical implantation procedures. Can. J. Fish. Aqua. 2015, 72, 1–5. [Google Scholar]
- Hays, G.; Åkesson, S.; Godley, B.; Luschi, P.; Santidrian, P. The Implications of Location Accuracy for the Interpretation of Satellite-Tracking Data. Anim. Behav. 2001, 61, 1035–1040. [Google Scholar] [CrossRef]
- Kessel, S.T.; Cooke, S.; Heupel, M.; Hussey, N.E.; Simpfendorfer, C.A.; Vagle, S.; Fisk, A.T. A Review of Detection Range Testing in Aquatic Passive Acoustic Telemetry Studies. Rev. Fish. Biol. Fish. 2013, 24, 199–218. [Google Scholar] [CrossRef]
- Brooks, M.E.; Kristensen, K.; van Benthem, K.J.; Magnusson, A.; Berg, C.W.; Nielsen, A.; Skaug, H.J.; Maechler, M.; Bolker, B.M. GlmmTMB Balances Speed and Flexibility Among Packages for Zero-inflated Generalized Linear Mixed Modeling. R J. 2017, 9, 378–400. [Google Scholar] [CrossRef]
- Rider, M.J.; McDonnell, L.H.; Hammerschlag, N. Multi-Year Movements of Adult and Subadult Bull Sharks Carcharhinus Leucas: Philopatry, Connectivity, and Environmental Influences. Aquat. Ecol. 2021, 55, 559–577. [Google Scholar] [CrossRef]
- Freitas, C.; Lydersen, C.; Fedak, M.A.; Kovacs, K.M. A simple new algorithm to filter marine mammal Argos locations. Mar. Mammal Sci. 2008, 24, 315–325. [Google Scholar] [CrossRef]
- Jonsen, I.D.; Flemming, J.M.; Myers, R.A. Robust state-space modeling of animal movement data. Ecology 2005, 86, 2874–2880. [Google Scholar] [CrossRef]
- Kranstauber, B.; Smolla, M.; Scharf, A.K. Move: Visualizing and Analyzing Animal Tracking Data. R Package Version 3.2.2. 2019. Available online: http://cran.r.project.org/package=move (accessed on 17 June 2022).
- Kranstauber, B.; Kays, R.; Lapoint, S.D.; Wikelski, M.; Safi, K. A dynamic Brownian bridge movement model to estimate utilization distributions for heterogenous animal movements. J. Anim. Ecol. 2012, 81, 738–746. [Google Scholar] [CrossRef] [PubMed]
- Aspillaga, E.; Arlinghaus, R.; Martorell-Barcelo, M.; Follana-Berna, G.; Lana, A.; Campos-Candela, A.; Alos, J. Performance of a novel system for high-resolution tracking of marine fish societies. Anim. Biotelemetry 2021, 9, 1. [Google Scholar] [CrossRef]
- Whitney, N.M.; Crow, G.L. Reproductive Biology of the Tiger Shark Galeocerdo cuvier in Hawaii. Mar. Biol. 2007, 151, 63–70. [Google Scholar] [CrossRef]
- Afonso, A.S.; Hazin, F.H.V. Vertical Movement Patterns and Ontogenetic Niche Expansion in the Tiger Shark, Galeocerdo cuvier. PLoS ONE 2015, 10, e116720. [Google Scholar] [CrossRef]
- Kohler, N.E.; Turner, P.A. Distributions and Movements of Atlantic Shark Species: A 52-Year Retrospective Atlas of Mark and Recapture Data. Mar. Fish. Rev. 2019, 81, 1–93. [Google Scholar] [CrossRef]
- Ajemian, M.J.; Drymon, J.M.; Hammerschlag, N.; Wells, R.J.D.; Street, G.; Falterman, B.; McKinney, J.A.; Iii, W.B.D.; Hoffmayer, E.R.; Fischer, C.; et al. Movement Patterns and Habitat Use of Tiger Sharks Galeocerdo cuvier across Ontogeny in the Gulf of Mexico. PLoS ONE 2020, 15, e234868. [Google Scholar] [CrossRef]
- Hammerschlag, N.; Gallagher, A.J.; Wester, J.; Luo, J.; Ault, J.S. Don’t Bite the Hand That Feeds: Assessing Ecological Impacts of Provisioning Ecotourism on an Apex Marine Predator. Funct. Ecol. 2012, 26, 567–576. [Google Scholar] [CrossRef]
- Hammerschlag, N.; Gutowsky, L.; Gallagher, A.; Matich, P.; Cooke, S. Diel Habitat Use Patterns of a Marine Apex Predator Tiger Shark, Galeocerdo cuvier at a High Use Area Exposed to Dive Tourism. J. Exp. Mar. Biol. Ecol. 2017, 495, 24–34. [Google Scholar] [CrossRef]
- Lea, J.S.; Wetherbee, B.M.; Sousa, L.L.; Aming, C.; Burnie, N.; E Humphries, N.; Queiroz, N.; Harvey, G.M.; Sims, D.W.; Shivji, M.S. Ontogenetic Partial Migration Is Associated with Environmental Drivers and Influences Fisheries Interactions in a Marine Predator. ICES J. Mar. Sci. 2018, 75, 1383–1392. [Google Scholar] [CrossRef]
- Werry, J.M.; Planes, S.; Berumen, M.L.; Lee, K.A.; Braun, C.D.; Clua, E. Reef-Fidelity and Migration of Tiger Sharks, Galeocerdo cuvier, across the Coral Sea. PLoS ONE 2014, 9, e083249. [Google Scholar] [CrossRef] [PubMed]
- Feldheim, K.A.; Gruber, S.H.; Dibattista, J.D.; Babcock, E.A.; Kessel, S.T.; Hendry, A.P.; Pikitch, E.K.; Ashley, M.V.; Chapman, D.D. Two Decades of Genetic Profiling Yield First Evidence of Natal Philopatry and Long-Term Fidelity to Parturition Sites in Sharks. Mol. Ecol. 2013, 23, 110–117. [Google Scholar] [CrossRef] [PubMed]
| Estimate | Std. Error | z Value | PR(>|z|) | |
|---|---|---|---|---|
| Intercept | 2.81 | 0.88 | 3.19 | 0.001 * |
| Temperature | −0.42 | 0.02 | −17.41 | <0.001* |
| Sex (male) | −0.20 | 0.54 | −0.38 | 0.71 |
| Fork Length | 0.01 | 0.00 | 4.84 | <0.001* |
| PTT ID | Tag Date | Sex | FL | SPOT Model | Days Transmitting | Number of Locations | 50% UD | 95% UD | Time (%) in EEZ |
|---|---|---|---|---|---|---|---|---|---|
| 170335 | 13 February 2018 | F | 190 | 257 | 498 | 3369 | 22,000 | 378,800 | 4.2 |
| 173632 | 3 December 2017 | F | 240 | 258 | 72 | 190 | 19,700 | 102,600 | 48.6 |
| 174071 | 16 January 2019 | F | 303 | 257 | 153 | 434 | 22,000 | 301,800 | 40.5 |
| 174072 | 10 May 2018 | F | 310 | 257 | 540 | 121 | 91,800 | 1,796,300 | 38.7 |
| 174073 | 26 June 2018 | F | 285 | 257 | 97 | 804 | 89,300 | 637,000 | 2.1 |
| 174074 | 24 June 2018 | F | 302 | 258 | 31 | 307 | 4700 | 28,600 | 71 |
| 179469 | 30 September 2020 | F | 246 | 364 | 150 | 1020 | 1000 | 108,000 | 7.3 |
| 179470 | 9 December 2019 | F | 267 | 364 | 168 | 137 | 15,800 | 101,500 | 51.7 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Smukall, M.J.; Seitz, A.C.; Dhellemmes, F.; van Zinnicq Bergmann, M.P.M.; Heim, V.; Gruber, S.H.; Guttridge, T.L. Residency, Site Fidelity, and Regional Movement of Tiger Sharks (Galeocerdo cuvier) at a Pupping Location in the Bahamas. Sustainability 2022, 14, 10017. https://doi.org/10.3390/su141610017
Smukall MJ, Seitz AC, Dhellemmes F, van Zinnicq Bergmann MPM, Heim V, Gruber SH, Guttridge TL. Residency, Site Fidelity, and Regional Movement of Tiger Sharks (Galeocerdo cuvier) at a Pupping Location in the Bahamas. Sustainability. 2022; 14(16):10017. https://doi.org/10.3390/su141610017
Chicago/Turabian StyleSmukall, Matthew J., Andrew C. Seitz, Félicie Dhellemmes, Maurits P. M. van Zinnicq Bergmann, Vital Heim, Samuel H. Gruber, and Tristan L. Guttridge. 2022. "Residency, Site Fidelity, and Regional Movement of Tiger Sharks (Galeocerdo cuvier) at a Pupping Location in the Bahamas" Sustainability 14, no. 16: 10017. https://doi.org/10.3390/su141610017
APA StyleSmukall, M. J., Seitz, A. C., Dhellemmes, F., van Zinnicq Bergmann, M. P. M., Heim, V., Gruber, S. H., & Guttridge, T. L. (2022). Residency, Site Fidelity, and Regional Movement of Tiger Sharks (Galeocerdo cuvier) at a Pupping Location in the Bahamas. Sustainability, 14(16), 10017. https://doi.org/10.3390/su141610017






