Microplastics in Glaciers: First Results from the Vatnajökull Ice Cap
Abstract
1. Introduction
2. Methods
2.1. Sampling
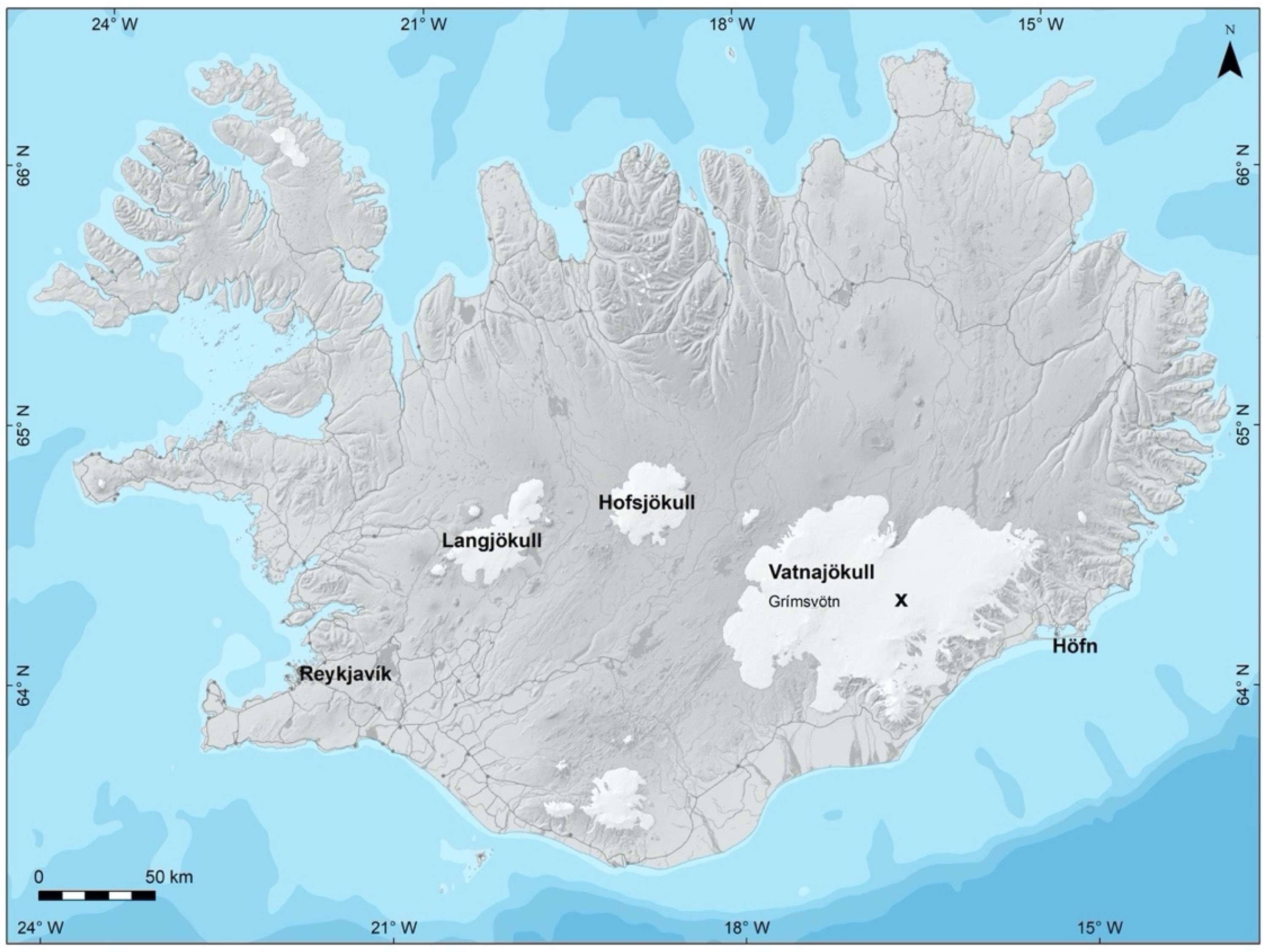
2.1.1. Location of Sampling
2.1.2. Collection of Samples
2.2. Methods for Processing and Analysing the Samples
2.2.1. Melting and Sieving of the Samples
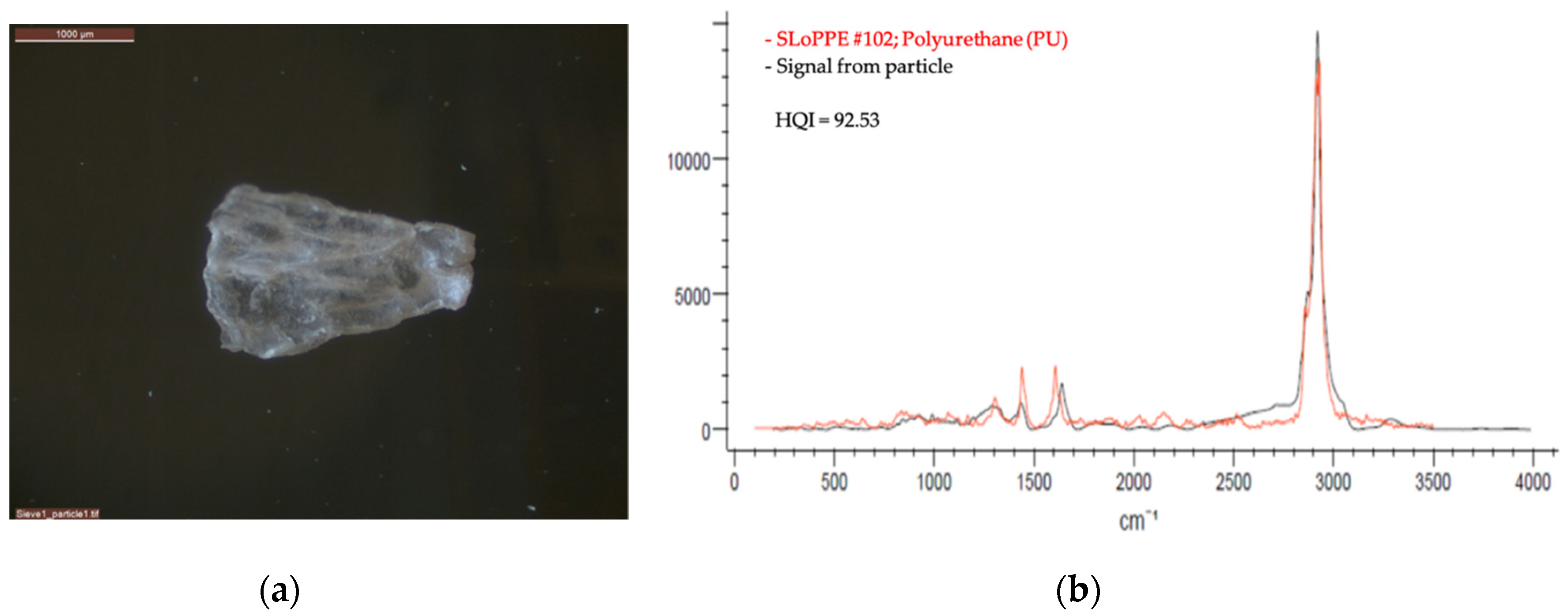
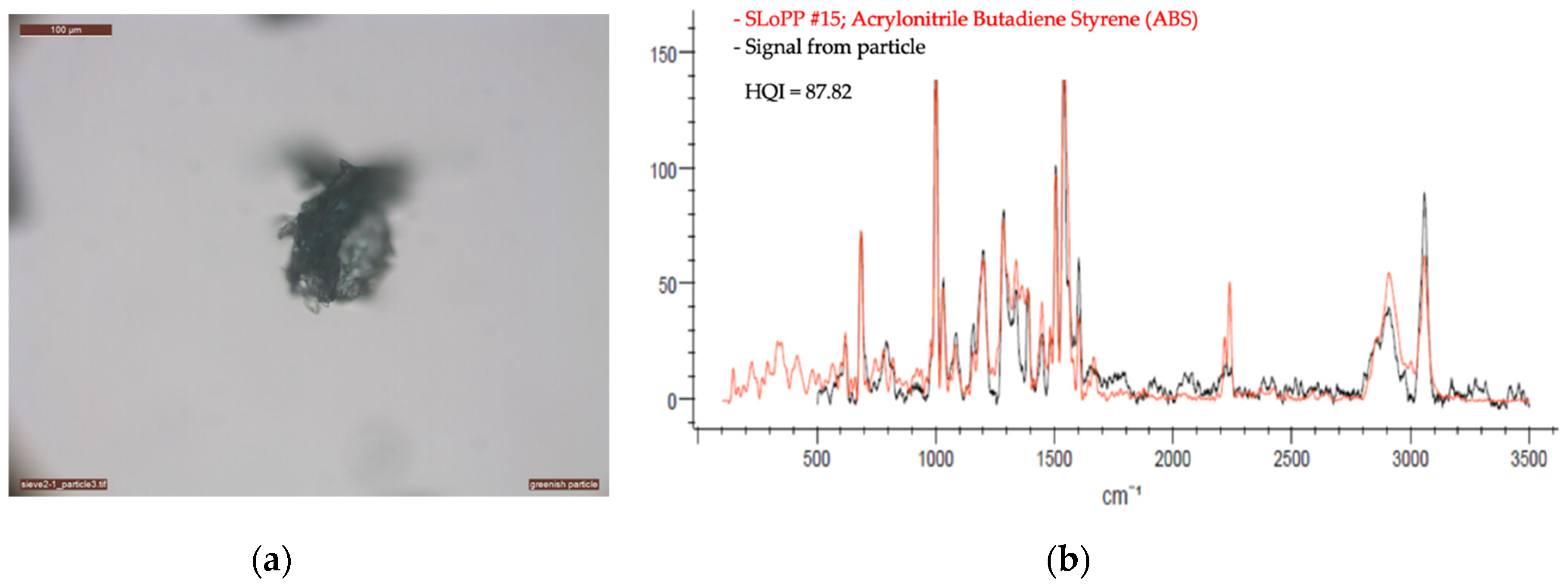
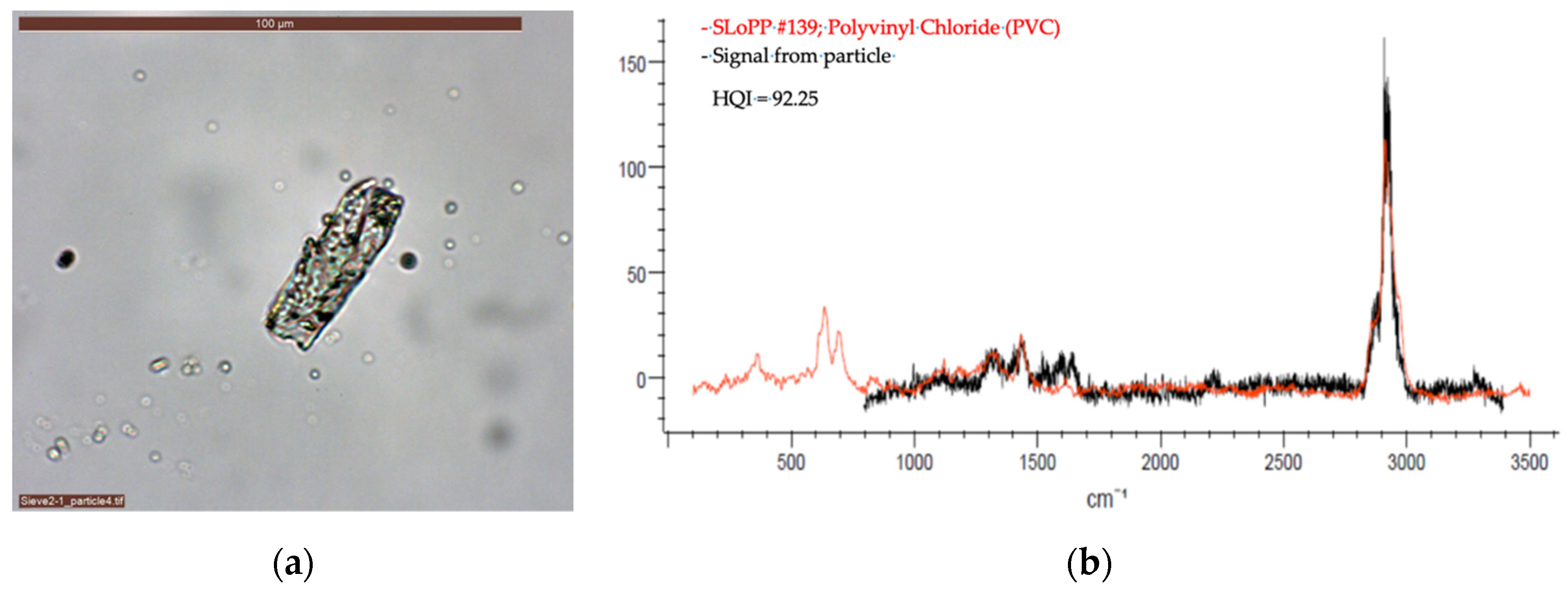
2.2.2. Optical Microscopy and Raman Spectroscopy
3. First Results from the Vatnajökull Ice Cap
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ritchie, H.; Roser, M. Plastic Pollution. In Our World Data; Oxford Martin School: Oxford, UK, 2018. [Google Scholar]
- Hartmann, N.B.; Hüffer, T.; Thompson, R.C.; Hassellöv, M.; Verschoor, A.; Daugaard, A.E.; Rist, S.; Karlsson, T.; Brennholt, N.; Cole, M. Are We Speaking the Same Language? Recommendations for a Definition and Categorization Framework for Plastic Debris; ACS Publications: Washington, DC, USA, 2019; ISBN 0013-936X. [Google Scholar]
- Wang, Y.-L.; Lee, Y.-H.; Chiu, I.-J.; Lin, Y.-F.; Chiu, H.-W. Potent Impact of Plastic Nanomaterials and Micromaterials on the Food Chain and Human Health. Int. J. Mol. Sci. 2020, 21, 1727. [Google Scholar] [CrossRef]
- Kershaw, P.; Rochman, C. Sources, Fate and Effects of Microplastics in the Marine Environment: Part 2 of a Global Assessment; GESAMP: London, UK, 2015. [Google Scholar]
- Dris, R.; Gasperi, J.; Mirande, C.; Mandin, C.; Guerrouache, M.; Langlois, V.; Tassin, B. A First Overview of Textile Fibers, Including Microplastics, in Indoor and Outdoor Environments. Environ. Pollut. 2017, 221, 453–458. [Google Scholar] [CrossRef] [PubMed]
- Gasperi, J.; Wright, S.L.; Dris, R.; Collard, F.; Mandin, C.; Guerrouache, M.; Langlois, V.; Kelly, F.J.; Tassin, B. Microplastics in Air: Are We Breathing It in? Curr. Opin. Environ. Sci. Health 2018, 1, 1–5. [Google Scholar] [CrossRef]
- Lehner, R.; Weder, C.; Petri-Fink, A.; Rothen-Rutishauser, B. Emergence of Nanoplastic in the Environment and Possible Impact on Human Health. Environ. Sci. Technol. 2019, 53, 1748–1765. [Google Scholar] [CrossRef] [PubMed]
- Prüst, M.; Meijer, J.; Westerink, R.H. The Plastic Brain: Neurotoxicity of Micro-and Nanoplastics. Part. Fibre Toxicol. 2020, 17, 1–16. [Google Scholar] [CrossRef]
- Van Raamsdonk, L.W.; van der Zande, M.; Koelmans, A.A.; Hoogenboom, R.L.; Peters, R.J.; Groot, M.J.; Peijnenburg, A.A.; Weesepoel, Y.J. Current Insights into Monitoring, Bioaccumulation, and Potential Health Effects of Microplastics Present in the Food Chain. Foods 2020, 9, 72. [Google Scholar] [CrossRef] [PubMed]
- Prata, J.C.; da Costa, J.P.; Lopes, I.; Duarte, A.C.; Rocha-Santos, T. Environmental Exposure to Microplastics: An Overview on Possible Human Health Effects. Sci. Total Environ. 2020, 702, 134455. [Google Scholar] [CrossRef] [PubMed]
- Andrady, A.L. Microplastics in the Marine Environment. Mar. Pollut. Bull. 2011, 62, 1596–1605. [Google Scholar] [CrossRef]
- Law, K.L.; Thompson, R.C. Microplastics in the Seas. Science 2014, 345, 144–145. [Google Scholar] [CrossRef]
- Wang, C.; Zhao, J.; Xing, B. Environmental Source, Fate, and Toxicity of Microplastics. J. Hazard. Mater. 2020, 2020, 124357. [Google Scholar] [CrossRef]
- Rochman, C.M. Microplastics Research—From Sink to Source. Science 2018, 360, 28–29. [Google Scholar] [CrossRef]
- Novotna, K.; Cermakova, L.; Pivokonska, L.; Cajthaml, T.; Pivokonsky, M. Microplastics in Drinking Water Treatment—Current Knowledge and Research Needs. Sci. Total Environ. 2019, 667, 730–740. [Google Scholar] [CrossRef]
- Setälä, O.; Fleming-Lehtinen, V.; Lehtiniemi, M. Ingestion and Transfer of Microplastics in the Planktonic Food Web. Environ. Pollut. 2014, 185, 77–83. [Google Scholar] [CrossRef]
- Schwabl, P.; Köppel, S.; Königshofer, P.; Bucsics, T.; Trauner, M.; Reiberger, T.; Liebmann, B. Detection of Various Microplastics in Human Stool. Ann. Intern. Med. 2019, 171, 453–457. [Google Scholar] [CrossRef]
- Evangeliou, N.; Grythe, H.; Klimont, Z.; Heyes, C.; Eckhardt, S.; Lopez-Aparicio, S.; Stohl, A. Atmospheric Transport Is a Major Pathway of Microplastics to Remote Regions. Nat. Commun. 2020, 11, 3381. [Google Scholar] [CrossRef]
- Zhang, Y.; Gao, T.; Kang, S.; Sillanpää, M. Importance of Atmospheric Transport for Microplastics Deposited in Remote Areas. Environ. Pollut. 2019, 254, 112953. [Google Scholar] [CrossRef]
- Zhang, Y.; Kang, S.; Allen, S.; Allen, D.; Gao, T.; Sillanpää, M. Atmospheric Microplastics: A Review on Current Status and Perspectives. Earth Sci. Rev. 2020, 203, 103118. [Google Scholar] [CrossRef]
- Allen, S.; Allen, D.; Phoenix, V.R.; Le Roux, G.; Durántez Jiménez, P.; Simonneau, A.; Binet, S.; Galop, D. Atmospheric Transport and Deposition of Microplastics in a Remote Mountain Catchment. Nat. Geosci. 2019, 12, 339–344. [Google Scholar] [CrossRef]
- Cózar, A.; Martí, E.; Duarte, C.M.; García-de-Lomas, J.; Van Sebille, E.; Ballatore, T.J.; Eguíluz, V.M.; González-Gordillo, J.I.; Pedrotti, M.L.; Echevarría, F. The Arctic Ocean as a Dead End for Floating Plastics in the North Atlantic Branch of the Thermohaline Circulation. Sci. Adv. 2017, 3, e1600582. [Google Scholar] [CrossRef] [PubMed]
- La Daana, K.K.; Gårdfeldt, K.; Lyashevska, O.; Hassellöv, M.; Thompson, R.C.; O’Connor, I. Microplastics in Sub-Surface Waters of the Arctic Central Basin. Mar. Pollut. Bull. 2018, 130, 8–18. [Google Scholar]
- La Daana, K.K.; Gardfeldt, K.; Krumpen, T.; Thompson, R.C.; O’Connor, I. Microplastics in Sea Ice and Seawater beneath Ice Floes from the Arctic Ocean. Sci. Rep. 2020, 10, 1–11. [Google Scholar] [CrossRef]
- Lusher, A.L.; Tirelli, V.; O’Connor, I.; Officer, R. Microplastics in Arctic Polar Waters: The First Reported Values of Particles in Surface and Sub-Surface Samples. Sci. Rep. 2015, 5, 14947. [Google Scholar] [CrossRef] [PubMed]
- Munari, C.; Infantini, V.; Scoponi, M.; Rastelli, E.; Corinaldesi, C.; Mistri, M. Microplastics in the Sediments of Terra Nova Bay (Ross Sea, Antarctica). Mar. Pollut. Bull. 2017, 122, 161–165. [Google Scholar] [CrossRef] [PubMed]
- Obbard, R.W.; Sadri, S.; Wong, Y.Q.; Khitun, A.A.; Baker, I.; Thompson, R.C. Global Warming Releases Microplastic Legacy Frozen in Arctic Sea Ice. Earths Future 2014, 2, 315–320. [Google Scholar] [CrossRef]
- Tirelli, V.; Suaria, G.; Lusher, A.L. Microplastics in Polar Samples. In Handbook of Microplastics in the Environment; Springer: Cham, Switzerland, 2020; pp. 1–42. [Google Scholar]
- Cuffey, K.M.; Paterson, W.S.B. The Physics of Glaciers; Academic Press: Cambridge, MA, USA, 2010; ISBN 0-08-091912-X. [Google Scholar]
- Allen, D.; Allen, S.; Sonke, J.; Phoenix, V. Atmospheric Transport and Our Planetary Boundary Layer as an Environmental Compartment of Microplastics Pollution. In Proceedings of the International Conference on Microplastic Pollution Inthe Mediterranean Sea. Springer Water; Springer: Cham, Switzerland, 2019; pp. 1–8. [Google Scholar]
- Ambrosini, R.; Azzoni, R.S.; Pittino, F.; Diolaiuti, G.; Franzetti, A.; Parolini, M. First Evidence of Microplastic Contamination in the Supraglacial Debris of an Alpine Glacier. Environ. Pollut. 2019, 253, 297–301. [Google Scholar] [CrossRef] [PubMed]
- Bergmann, M.; Mützel, S.; Primpke, S.; Tekman, M.B.; Trachsel, J.; Gerdts, G. White and Wonderful? Microplastics Prevail in Snow from the Alps to the Arctic. Sci. Adv. 2019, 5, eaax1157. [Google Scholar] [CrossRef]
- Cabrera, M.; Valencia, B.G.; Lucas-Solis, O.; Calero, J.L.; Maisincho, L.; Conicelli, B.; Moulatlet, G.M.; Capparelli, M.V. A New Method for Microplastic Sampling and Isolation in Mountain Glaciers: A Case Study of One Antisana Glacier, Ecuadorian Andes. Case Stud. Chem. Environ. Eng. 2020, 2, 100051. [Google Scholar] [CrossRef]
- Chen, X.; Zhang, X.; Church, J.A.; Watson, C.S.; King, M.A.; Monselesan, D.; Legresy, B.; Harig, C. The Increasing Rate of Global Mean Sea-Level Rise during 1993–2014. Nat. Clim. Chang. 2017, 7, 492–495. [Google Scholar] [CrossRef]
- Aðalgeirsdóttir, G.; Pálsson, F.; Thorsteinsson, T.; Magnússon, E.; Belart, J.; Jóhannesson, T.; Hannesdóttir, H.; Sigurðsson, O.; Gunnarsson, A.; Einarsson, B. Glacier Changes in Iceland from ~1890 to 2019. Front. Earth Sci. 2020, 8, 520. [Google Scholar] [CrossRef]
- Talalay, P.G. (Ed.) Hand- and Power-Driven Portable Drills. In Mechanical Ice Drilling Technology; Springer Geophysics; Springer: Singapore, 2016; pp. 27–52. ISBN 978-981-10-0560-2. [Google Scholar]
- Pálsson, F.; Gunnarsson, A.; Skagfjörð Pálsson, H.; Steinþórsson, S. Vatnajökull: Mass Balance, Meltwater Drainage and Surface Velocity of the Glacial Year 2019–20; Landsvirkjun: Reykjavik, Iceland, 2021. [Google Scholar]
- Sobhani, Z.; Zhang, X.; Gibson, C.; Naidu, R.; Mallavarapu, M.; Fang, C. Identification and Visualisation of Microplastics/Nanoplastics by Raman Imaging (i): Down to 100 Nm. Water Res. 2020, 174, 115658. [Google Scholar] [CrossRef]
- Munno, K.; De Frond, H.; O’Donnell, B.; Rochman, C.M. Increasing the Accessibility for Characterizing Microplastics: Introducing New Application-Based and Spectral Libraries of Plastic Particles (SLoPP and SLoPP-E). Anal. Chem. 2020, 92, 2443–2451. [Google Scholar] [CrossRef]
- Peternell, M.; Wilson, C.J. Effect of Strain Rate Cycling on Microstructures and Crystallographic Preferred Orientation during High-Temperature Creep. Geology 2016, 44, 279–282. [Google Scholar] [CrossRef]
- Wilson, C.J.; Hunter, N.J.; Luzin, V.; Peternell, M.; Piazolo, S. The Influence of Strain Rate and Presence of Dispersed Second Phases on the Deformation Behaviour of Polycrystalline D2O Ice. J. Glaciol. 2019, 65, 101–122. [Google Scholar] [CrossRef]
- Gomiero, A.; Ásmundsdóttir, Á.M.; Þorsteinsson, Þ.; Oysæd, K.B. Preliminary Occurrence of Microplastics in a Remote Icelandic Glacier. 2021. Available online: https://pame.is/projects/arctic-marine-pollution (accessed on 3 March 2021).
- Dris, R.; Gasperi, J.; Saad, M.; Mirande, C.; Tassin, B. Synthetic Fibers in Atmospheric Fallout: A Source of Microplastics in the Environment? Mar. Pollut. Bull. 2016, 104, 290–293. [Google Scholar] [CrossRef]
- Bianco, A.; Passananti, M. Atmospheric Micro and Nanoplastics: An Enormous Microscopic Problem. Sustainability 2020, 12, 7327. [Google Scholar] [CrossRef]
- Allen, S.; Allen, D.; Moss, K.; Le Roux, G.; Phoenix, V.R.; Sonke, J.E. Examination of the Ocean as a Source for Atmospheric Microplastics. PLoS ONE 2020, 15, e0232746. [Google Scholar] [CrossRef]
- Liu, K.; Wu, T.; Wang, X.; Song, Z.; Zong, C.; Wei, N.; Li, D. Consistent Transport of Terrestrial Microplastics to the Ocean through Atmosphere. Environ. Sci. Technol. 2019, 53, 10612–10619. [Google Scholar] [CrossRef]
- Draxler, R.; Rolph, G. HYSPLIT (HYbrid Single-Particle Lagrangian Integrated Trajectory) Model Access via NOAA ARL READY Website; NOAA Air Resources Laboratory: Silver Spring, MD, USA, 2010.
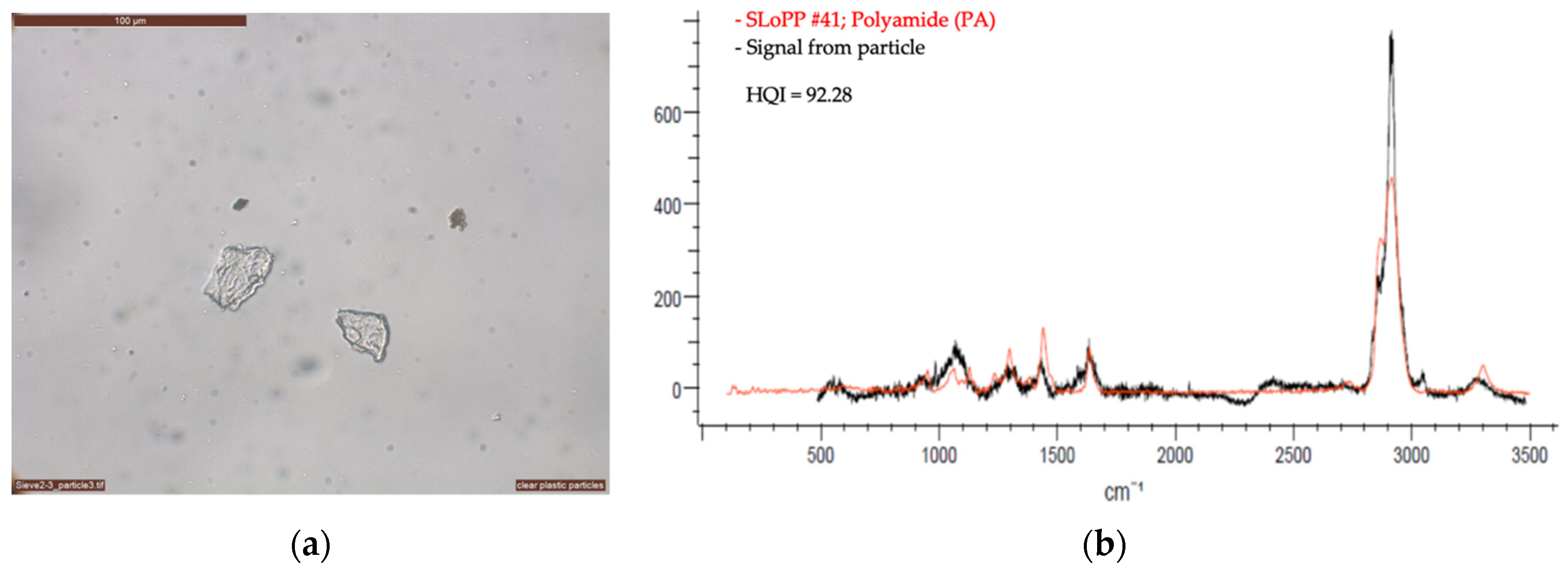
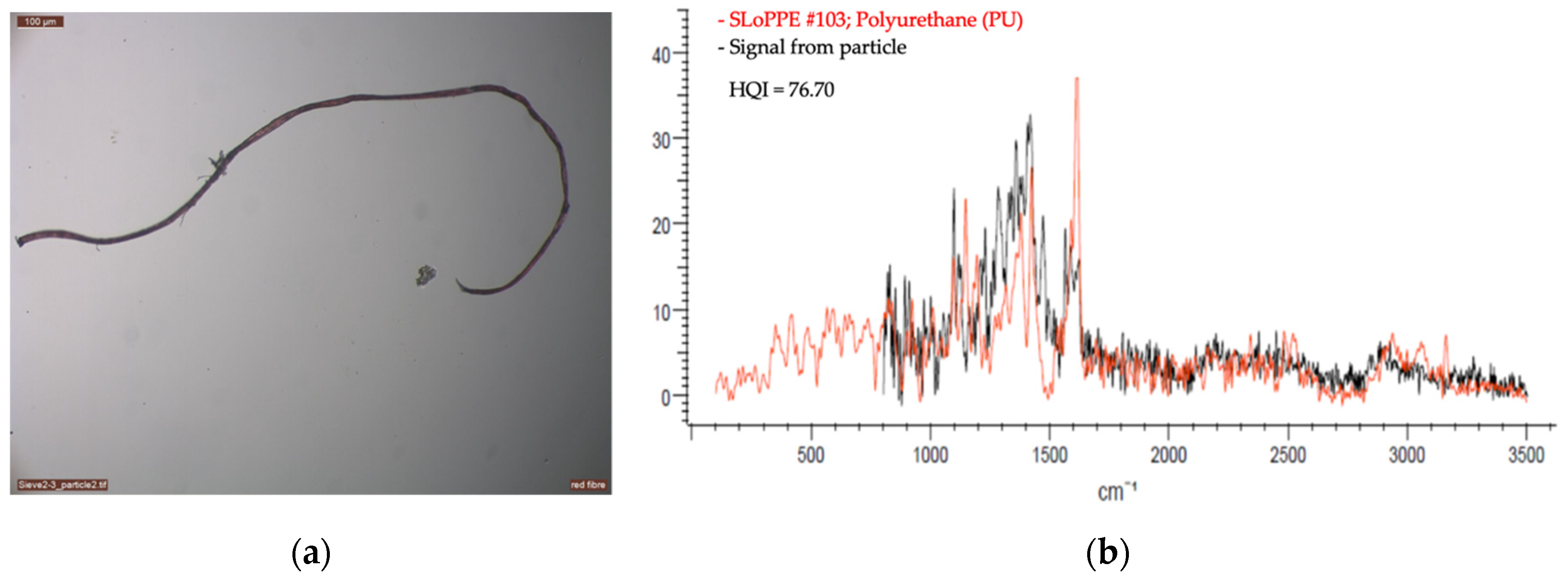
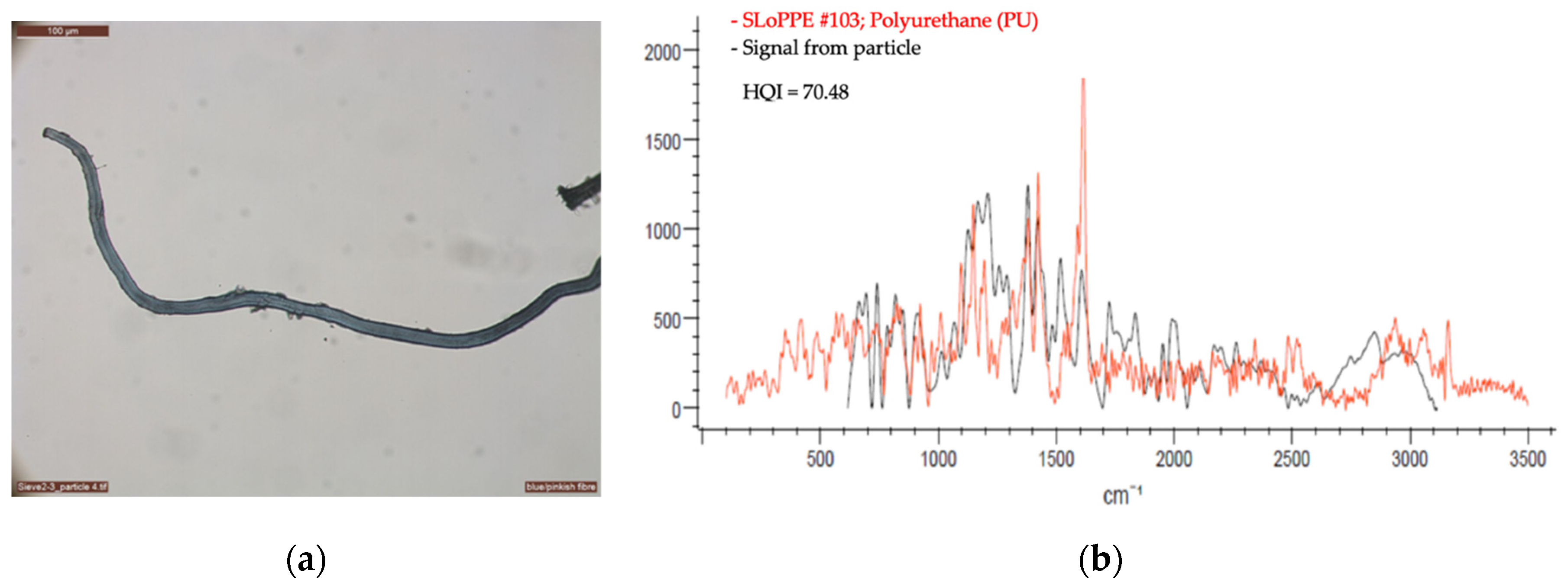
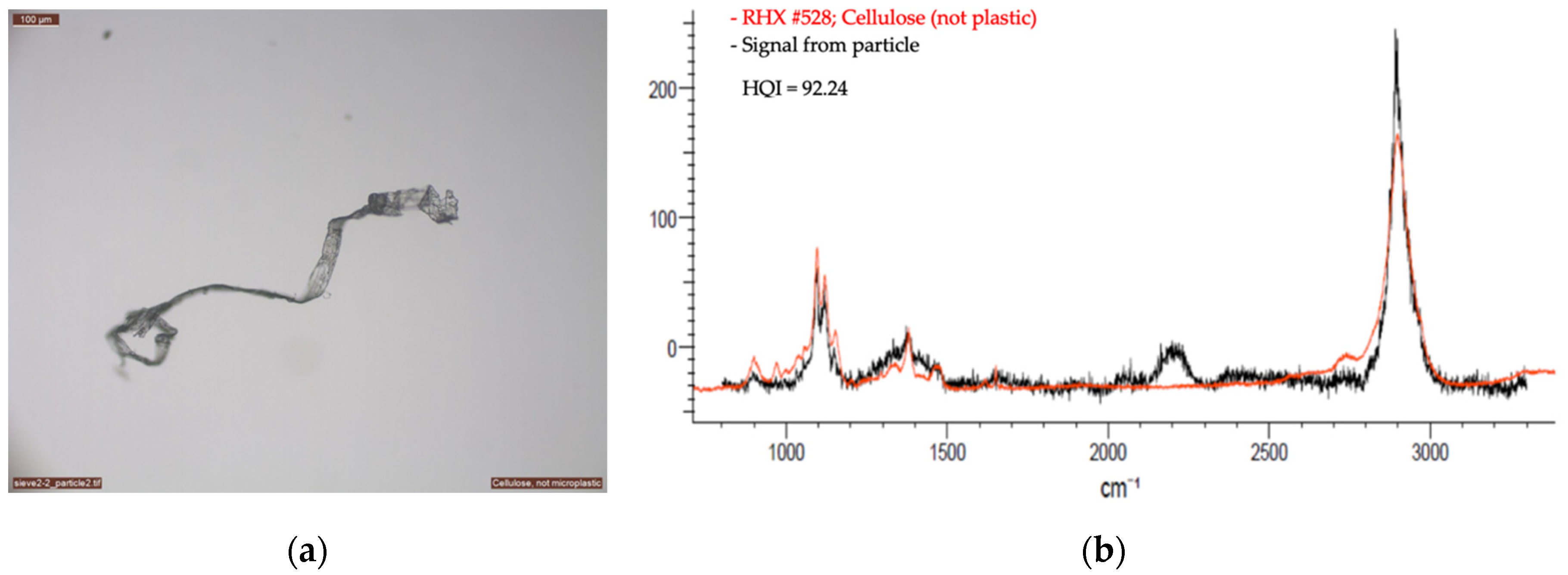
| Sample | Material | Equal Area Diameter, Particle Size (µm) | Laser (nm) | Laser Intensity (%) | Acquisition | Baseline Correction | Number of Measurement Per Sample | |
|---|---|---|---|---|---|---|---|---|
| Time | Cycles | |||||||
| VN01 | Polyurethane | 3000 | 785 | 10 | 120 | 10 | Yes | 5 |
| VN02 | Acrylonitrile butadiene styrene | 230 | 785 | 5 | 30 | 10 | Yes | 3 |
| VN03 | Polyvinyl chloride | 40 | 785 | 5 | 60 | 5 | Yes | 2 |
| VN04 | Clear polyamide | 30 | 785 | 10 | 60 | 3 | No | 3 |
| VN05 | Polyurethane fibre | 3000 | 532 | 100 | 60 | 3 | Yes | 5 |
| VN06 | Polyurethane fibre | 1300 | 532 | 100 | 240 | 3 | No | 4 |
| VN07 | Cellulose | 2400 | 785 | 5 | 30 | 5 | Yes | 2 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Stefánsson, H.; Peternell, M.; Konrad-Schmolke, M.; Hannesdóttir, H.; Ásbjörnsson, E.J.; Sturkell, E. Microplastics in Glaciers: First Results from the Vatnajökull Ice Cap. Sustainability 2021, 13, 4183. https://doi.org/10.3390/su13084183
Stefánsson H, Peternell M, Konrad-Schmolke M, Hannesdóttir H, Ásbjörnsson EJ, Sturkell E. Microplastics in Glaciers: First Results from the Vatnajökull Ice Cap. Sustainability. 2021; 13(8):4183. https://doi.org/10.3390/su13084183
Chicago/Turabian StyleStefánsson, Hlynur, Mark Peternell, Matthias Konrad-Schmolke, Hrafnhildur Hannesdóttir, Einar Jón Ásbjörnsson, and Erik Sturkell. 2021. "Microplastics in Glaciers: First Results from the Vatnajökull Ice Cap" Sustainability 13, no. 8: 4183. https://doi.org/10.3390/su13084183
APA StyleStefánsson, H., Peternell, M., Konrad-Schmolke, M., Hannesdóttir, H., Ásbjörnsson, E. J., & Sturkell, E. (2021). Microplastics in Glaciers: First Results from the Vatnajökull Ice Cap. Sustainability, 13(8), 4183. https://doi.org/10.3390/su13084183






