Data Envelopment Analysis Applications on Primary Health Care Using Exogenous Variables and Health Outcomes
Abstract
1. Introduction
2. Materials and Methods
2.1. Data and Variables
2.2. Methodology
3. Results
3.1. Factor Analysis
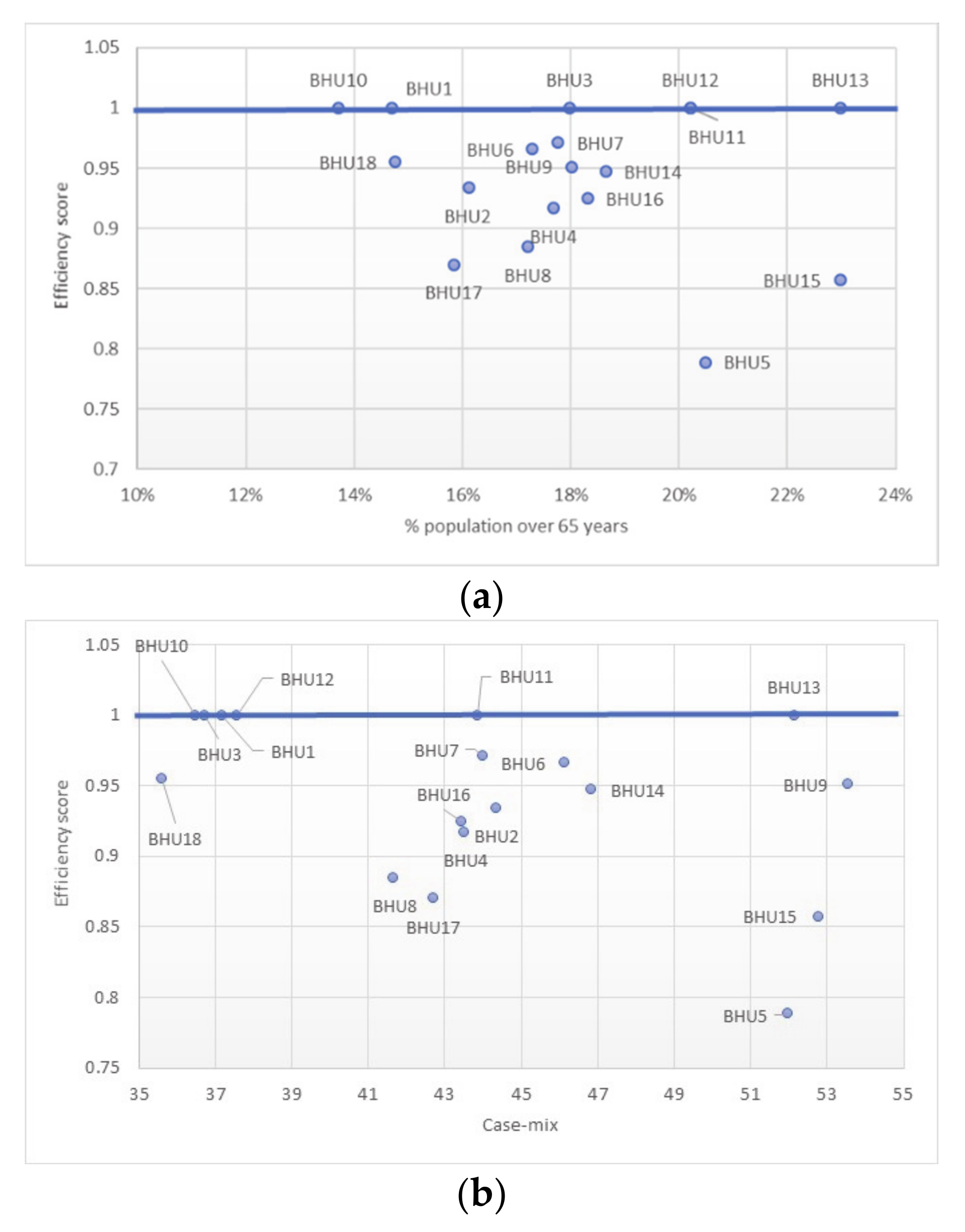
3.2. Data Envelopment Analysis
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
- Assigned population for each BHU.
- General practitioners and nursing personnel assigned to each BHU.
- Costs for personnel assigned to each BHU (personnel expenditure).
- Examinations expenditure, including costs of laboratory tests and imaging diagnoses performed on the assigned population of each BHU.
- Pharmaceutical expenditure. This includes the cost of medications prescribed and dispensed by general practitioners.
- Number of consultations with general practitioners and nursing staff.
- Number of referrals to specialised care.
- Number of emergencies attended for patients who are part of the assigned population.
- Number of avoidable hospitalisations. This refers to the number of hospital admissions of the population over 40 years old caused by pathologies that should be controlled from the primary health centres [48] and as such should not result in an admission.
- Mortality. Number of persons of each BHU dying in a year.
- Indicator of prescription efficiency. The quality of pharmaceutical prescription, or indicator of prescription efficiency, is measured with the prior development of other indicators which consider that, for a group of pathologies (which constitute a high percentage of the total pharmaceutical expenditure) there has been correct prescription of the drugs for the patient to be controlled and at the same time, these are the most economic and efficient in their therapeutic group.
- Percentage of population over 65 for each BHU.
- Case-mix, that is, a measure of the multimorbidity or the level of patients with multiple chronic conditions assigned to each BHU, is obtained from the Clinical Risk Group (CRG) classification of the population assigned to each BHU. The CRG assigns a weight to each health condition, related to the clinical complexity, in economic terms, of treatment.
References
- Organisation for Economic Co-operation and Development Health Resources. Health Spending—OECD. Available online: https://data.oecd.org/healthres/health-spending.htm (accessed on 20 July 2020).
- Ministerio de Sanidad Portal Estadístico del SNS. Sanidad en Datos. Available online: https://www.mscbs.gob.es/estadEstudios/sanidadDatos/home.htm (accessed on 20 July 2020).
- Ministerio de Sanidad. Servicios Sociales e Igualdad Sistema Nacional de Salud en España. In Monografía en Internet.; 2012. Available online: http://www.msssi.gob.es/organizacion/sns/docs/sns2012/SNS012__Espanol.pdf (accessed on 1 December 2020).
- Starfield, B.; Shi, L.; Macinko, J. Contribution of primary care to health systems and health. Milbank Q. 2005, 83, 457–502. [Google Scholar] [CrossRef]
- Caminal, J.; Starfield, B.; Sánchez, E.; Casanova, C.; Morales, M. The role of primary care in preventing ambulatory care sensitive conditions. Eur. J. Public Health 2004, 14, 246–251. [Google Scholar] [CrossRef]
- Sarti, T.D.; Lazarini, W.S.; Fontenelle, L.F.; Almeida, A.P.S.C. Qual o papel da Atenção Primária à Saúde diante da pandemia provocada pela COVID-19? Epidemiol. Serv. Rev. Sist. Unico Saude Bras. 2020, 29, e2020166. [Google Scholar] [CrossRef]
- Saint-Lary, O.; Gautier, S.; Le Breton, J.; Gilberg, S.; Frappé, P.; Schuers, M.; Bourgueil, Y.; Renard, V. How GPs adapted their practices and organisations at the beginning of COVID-19 outbreak: A French national observational survey. BMJ Open 2020, 10, e042119. [Google Scholar] [CrossRef]
- Wynn, A.; Moore, K.M. Integration of primary health care and public health during a public health emergency. Am. J. Public Health 2012, 102, e9. [Google Scholar] [CrossRef]
- Joy, M.; Mcgagh, D.; Jones, N.; Liyanage, H.; Sherlock, J.; Parimalanathan, V.; Akinyemi, O.; Van Vlymen, J.; Howsam, G.; Marshall, M.; et al. Reorganisation of primary care for older adults during COVID-19: A cross-sectional database study in the UK. Br. J. Gen. Pr. 2020, 70, e540–e547. [Google Scholar] [CrossRef]
- Lim, W.H.; Wong, W.M. COVID-19: Notes from the front line, Singapore’s primary health care perspective. Ann. Fam. Med. 2020, 18, 259–261. [Google Scholar] [CrossRef] [PubMed]
- Charnes, A.; Cooper, W.W.; Rhodes, E. Measuring efficiency of decision-making units. Eur. J. Oper. Res. 1978, 2, 429–444. [Google Scholar] [CrossRef]
- Sherman, H.D.; Zhu, J. Service Productivity Management: Improving Service Performance Using Data Envelopment Analysis (DEA); Springer Science & Business Media: Berlin/Heidelberg, Germany, 2006. [Google Scholar]
- Del Rocío Moreno-Enguix, M.; Gómez-Gallego, J.C.; Gómez Gallego, M. Analysis and determination the efficiency of the European health systems. Int. J. Health Plann. Manag. 2018, 33, 136–154. [Google Scholar] [CrossRef] [PubMed]
- Dlouhý, M. Measuring geographic inequalities: Dealing with multiple health resources by data envelopment analysis. Front. Public Health 2018, 6, 53. [Google Scholar] [CrossRef] [PubMed]
- Sandiford, P.; Vivas Consuelo, D.; Rouse, P.; Bramley, D. The trade-off between equity and efficiency in population health gain: Making it real. Soc. Sci. Med. 2018, 212, 136–144. [Google Scholar] [CrossRef] [PubMed]
- Sandiford, P.; Consuelo, D.J.J.V.; Rouse, P. How efficient are New Zealand’s District Health Boards at producing life expectancy gains for Māori and Europeans? Aust. N. Z. J. Public Health 2017. [Google Scholar] [CrossRef] [PubMed]
- Caballer-Tarazona, M.; Moya-Clemente, I.; Vivas-Consuelo, D.; Barrachina-Martínez, I. A model to measure the efficiency of hospital performance. Math. Comput. Model. 2010, 52, 1095–1102. [Google Scholar] [CrossRef]
- Pérez-Romero, C.; Ortega-Díaz, M.I.; Ocaña-Riola, R.; Martín-Martín, J.J. Análisis de la eficiencia técnica en los hospitales del Sistema Nacional de Salud español. Gac. Sanit. 2017, 31, 108–115. [Google Scholar] [CrossRef] [PubMed]
- Tabanera, L.H.; Martín, J.J.; González, M.D.P.L.D.A. Eficiencia técnica de los hospitales públicos y de las empresas públicas hospitalarias de Andalucía. Gac. Sanit. 2015, 29, 274–281. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Siciliani, L. Estimating technical efficiency in the hospital sector with panel data: A comparison of parametric and non-parametric techniques. Appl. Health Econ. Health Policy 2006, 5, 99–116. [Google Scholar] [CrossRef]
- Martin Martin, J.J.; González, M.D.P.L.D.A. La medida de la eficiencia en las organizaciones sanitarias. Presup. Público 2007, 49, 139–161. [Google Scholar]
- Mancuso, P.; Valdmanis, V.G. Care Appropriateness and health productivity evolution: A non-parametric analysis of the Italian regional health systems. Appl. Health Econ. Health Policy 2016, 14, 595–607. [Google Scholar] [CrossRef]
- Zakowska, I.; Godycki-Cwirko, M. Data envelopment analysis applications in primary health care: A systematic review. Fam. Pract. 2020, 37, 147–153. [Google Scholar] [CrossRef]
- Goñi-Legaz, S. El análisis envolvente de datos como sistema de evaluaclón de la eficiencia técnica de las organizaciones del sector público: Aplicación en los equipos de atención primaria. Rev. Esp. Financ. Contab. 1998, 27, 979–1004. [Google Scholar]
- Donabedian, A. The Definition of Quality and Approaches to Its Assessment and Monitoring; Health Administration Press: Chicago, IL, USA, 1980; Volume 1. [Google Scholar]
- Filipe Amado, C.A.; Dyson, R.G. On comparing the performance of primary care providers. Eur. J. Oper. Res. 2008, 185, 915–932. [Google Scholar] [CrossRef]
- Amado, C.A.; Santos, S.P. Challenges for performance assessment and improvement in primary health care: The case of the Portuguese health centres. Health Policy 2009, 91, 43–56. [Google Scholar] [CrossRef] [PubMed]
- Romano, J.; Choi, Á. Medida de la eficiencia de la atención primaria en Barcelona incorporando indicadores de calidad. Gac. Sanit. 2016, 30, 359–365. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Cordero, J.M.; Alonso-Morán, E.; Nuño-Solinis, R.; Orueta, J.F.; Arce, R.S. Efficiency assessment of primary care providers: A conditional nonparametric approach. Eur. J. Oper. Res. 2015, 240, 235–244. [Google Scholar] [CrossRef]
- Cordero-Ferrera, J.M.; Crespo-Cebada, E.; Murillo-Zamorano, L.R. Measuring technical efficiency in primary health care: The effect of exogenous variables on results. J. Med. Syst. 2011, 35, 545–554. [Google Scholar] [CrossRef]
- Pelone, F.; Kringos, D.S.; Romaniello, A.; Archibugi, M.; Salsiri, C.; Ricciardi, W. Primary care efficiency measurement using data envelopment analysis: A systematic review. J. Med. Syst. 2015, 39. [Google Scholar] [CrossRef]
- Cordero Ferrera, J.M.; Cebada, E.C.; Murillo Zamorano, L.R. The effect of quality and socio-demographic variables on efficiency measures in primary health care. Eur. J. Health Econ. 2014, 15, 289–302. [Google Scholar] [CrossRef]
- Oikonomou, N.; Tountas, Y.; Mariolis, A.; Souliotis, K.; Athanasakis, K.; Kyriopoulos, J. Measuring the efficiency of the Greek rural primary health care using a restricted DEA model; the case of southern and western Greece. Health Care Manag. Sci. 2016, 19, 313–325. [Google Scholar] [CrossRef]
- Cordero, J.M.; Nuño-Solinís, R.; Orueta, J.F.; Polo, C.; del Río-Cámara, M.; Alonso-Morán, E. Evaluación de la eficiencia técnica de la atención primaria pública en el País Vasco, 2010-2013. Gac. Sanit. 2016, 30, 104–109. [Google Scholar] [CrossRef]
- Ji, Y.; Lee, C. Data Envelopment analysis in stata. Stata J. 2010, 10, 267–280. [Google Scholar] [CrossRef]
- Simar, L.; Wilson, P. Sensitivity analysis of efficiency scores: How to bootstrap in nonparametric frontier models. Manag. Sci. 1998, 44, 49–61. [Google Scholar] [CrossRef]
- Tapia, J.A.; Salvador, B.; Rodríguez, J.M. Data envelopment analysis with estimated output data: Confidence intervals efficiency. Meas. J. Int. Meas. Confed. 2020, 152. [Google Scholar] [CrossRef]
- Simar, L.; Wilson, P.W. Estimation and inference in two-stage, semi-parametric models of production processes. J. Econ. 2007, 136, 31–64. [Google Scholar] [CrossRef]
- Caballer-Tarazona, V.; Guadalajara-Olmeda, N.; Vivas-Consuelo, D. Predicting healthcare expenditure by multimorbidity groups. Health Policy 2019, 123, 427–434. [Google Scholar] [CrossRef]
- Vivas-Consuelo, D.; Usó-Talamantes, R.; Guadalajara-Olmeda, N.; Trillo-Mata, J.-L.; Sancho-Mestre, C.; Buigues-Pastor, L. Pharmaceutical cost management in an ambulatory setting using a risk adjustment tool. BMC Health Serv. Res. 2014, 14. [Google Scholar] [CrossRef] [PubMed]
- Caballer Tarazona, V.; Guadalajara Olmeda, N.; Vivas Consuelo, D.; Clemente Collado, A. Impact of Morbidity on Health Care Costs of a Department of Health through Clinical Risk Groups. Valencian Community, Spain. Rev. Esp. Salud Publica 2016, 90, e1–e15. [Google Scholar]
- Carreras, M.; Ibern, P.; Coderch, J.; Sánchez, I.; Inoriza, J.M. Estimating lifetime healthcare costs with morbidity data. BMC Health Serv. Res. 2013, 13. [Google Scholar] [CrossRef]
- Orueta, J.F.; García-Alvarez, A.; Grandes, G.; Nuño-Solinís, R. Variability in potentially preventable hospitalisations: An observational study of clinical practice patterns of general practitioners and care outcomes in the Basque Country (Spain). BMJ Open 2015, 5. [Google Scholar] [CrossRef]
- Gouveia, M.C.; Dias, L.C.; Antunes, C.H. Additive DEA based on MCDA with imprecise information. J. Oper. Res. Soc. 2008, 59, 54–63. [Google Scholar] [CrossRef]
- Jahanshahloo, G.R.; Vencheh, A.H.; Foroughi, A.A.; Matin, R.K. Inputs/outputs estimation in DEA when some factors are undesirable. Appl. Math. Comput. 2004, 156, 19–32. [Google Scholar] [CrossRef]
- Scheel, H. Undesirable outputs in efficiency valuations. Eur. J. Oper. Res. 2001, 132, 400–410. [Google Scholar] [CrossRef]
- Dyson, R.G.; Allen, R.; Camanho, A.S.; Podinovski, V.V.; Sarrico, C.S.; Shale, E.A. Pitfalls and protocols in DEA. Eur. J. Oper. Res. 2001, 132, 245–259. [Google Scholar] [CrossRef]
- Angulo-Pueyo, E.; Ridao-López, M.; Martínez-Lizaga, N.; García-Armesto, S.; Peiró, S.; Bernal-Delgado, E. Factors associated with hospitalisations in chronic conditions deemed avoidable: Ecological study in the Spanish healthcare system. BMJ Open 2017, 7, e011844. [Google Scholar] [CrossRef] [PubMed]
| Variable | Role | Average | Standard Dev. | Coef. Variation | Max. | Min. |
|---|---|---|---|---|---|---|
| % Population >65 years | Exogenous | 18.05% | 2.60% | 0.14 | 22.97% | 13.71% |
| Case mix | Exogenous | 43.91 | 5.86 | 0.13 | 53.53 | 35.62 |
| General practitioners * | Input | 7.11 | 0.97 | 0.14 | 9.58 | 5.93 |
| Nursing * | Input | 5.40 | 0.73 | 0.14 | 7.18 | 4.45 |
| Personnel expenditure (euros) * | Input | 1,165,648.20 | 245,940.11 | 0.21 | 1,982,827.01 | 901,336.29 |
| Examinations expenditure (euros) * | Input | 223,943.08 | 45,238.99 | 0.20 | 323,204.85 | 131,671.65 |
| Pharmaceutical expenditure (euros) * | Input | 1,953,897.53 | 272,025.09 | 0.14 | 2,389,896.30 | 1,445,721.18 |
| General practitioners’ consultations * | Output | 46,346.89 | 6,036.59 | 0.13 | 56,779.43 | 34,659.60 |
| Nursing consultations * | Output | 25,261.07 | 4397.89 | 0.17 | 32,578.43 | 18,153.55 |
| Referrals * | Output | 3908.19 | 647.55 | 0.17 | 4947.35 | 2317.79 |
| Corrected emergencies * | Output | 6300.42 | 474.45 | 0.08 | 7403.66 | 5258.20 |
| Corrected avoidable hospitalisations * | Output | 76.64 | 6.46 | 0.08 | 85.87 | 59.43 |
| Corrected mortality * | Output | 932.35 | 16.48 | 0.02 | 950.04 | 899.27 |
| Prescription efficiency | Output | 47.33 | 30.62 | 0.65 | 91.70 | 8.30 |
| Component | Initial Eigenvalues | ||
|---|---|---|---|
| Total | % of Variance | % Accumulated | |
| 1 | 5.558 | 42.76% | 42.76% |
| 2 | 2.859 | 21.99% | 64.75% |
| 3 | 1.772 | 13.63% | 78.38% |
| 4 | 1.005 | 7.73% | 86.11% |
| 5 | 0.628 | 4.83% | 90.94% |
| 6 | 0.422 | 3.25% | 94.19% |
| 7 | 0.265 | 2.04% | 96.22% |
| 8 | 0.227 | 1.74% | 97.97% |
| 9 | 0.162 | 1.25% | 99.22% |
| 10 | 0.051 | 0.39% | 99.61% |
| 11 | 0.032 | 0.25% | 99.85% |
| 12 | 0.016 | 0.12% | 99.97% |
| 13 | 0.003 | 0.03% | 100.00% |
| Variable | Component | ||
|---|---|---|---|
| 1 | 2 | 3 | |
| Pharmaceutical expenditure * | 0.693 | 0.391 | 0.505 |
| Referrals * | 0.798 | −0.144 | −0.217 |
| Prescription efficiency | −0.650 | 0.582 | −0.186 |
| General practitioners’ consultations * | 0.877 | 0.386 | 0.126 |
| Nursing consultations * | 0.714 | 0.412 | −0.120 |
| Corrected emergencies * | −0.780 | −0.123 | −0.334 |
| General practitioners * | 0.383 | 0.745 | 0.222 |
| Nursing * | - | 0.821 | 0.227 |
| Personnel expenditure * | 0.264 | 0.873 | - |
| Examinations expenditure * | - | 0.702 | 0.216 |
| % Population >65 years | −0.362 | 0.253 | 0.776 |
| Case-mix | 0.252 | 0.585 | 0.745 |
| Corrected avoidable hospitalisations * | −0.234 | - | −0.868 |
| Role | Model 1 | Model 2 |
|---|---|---|
| Inputs | General practitioners * Nursing * Pharmaceutical expenditure * | General practitioners * Nursing * Pharmaceutical expenditure * |
| Outputs | Prescription efficiency Corrected emergencies * Corrected avoidable hospitalisations * | Prescription efficiency Consultations * Corrected avoidable hospitalisations * |
| DMU | VRS (without Exogenous Variables) | |||
|---|---|---|---|---|
| Model 1 | Model 2 | |||
| Rank | Efficiency Score | Rank | Efficiency Score | |
| Basic health unit (BHU)1 | 1 | 1 | 1 | 1 |
| BHU2 | 12 | 0.934 | 14 | 0.971 |
| BHU3 | 1 | 1 | 1 | 1 |
| BHU4 | 14 | 0.917 | 1 | 1 |
| BHU5 | 18 | 0.789 | 17 | 0.850 |
| BHU6 | 8 | 0.966 | 1 | 1 |
| BHU7 | 7 | 0.971 | 1 | 1 |
| BHU8 | 15 | 0.885 | 13 | 0.990 |
| BHU9 | 10 | 0.951 | 1 | 1 |
| BHU10 | 6 | 1 | 1 | 1 |
| BHU11 | 1 | 1 | 1 | 1 |
| BHU12 | 1 | 1 | 1 | 1 |
| BHU13 | 1 | 1 | 1 | 1 |
| BHU14 | 11 | 0.947 | 11 | 1 |
| BHU15 | 17 | 0.857 | 18 | 0.777 |
| BHU16 | 13 | 0.925 | 15 | 0.943 |
| BHU17 | 16 | 0.870 | 16 | 0.918 |
| BHU18 | 9 | 0.955 | 12 | 1 |
| DMU | Islacks | Oslacks | ||||
|---|---|---|---|---|---|---|
| General Practitioners * | Nursing * | Pharm. Expend. * | Corrected Emergencies * | Corrected Avoidable Hosp. * | Prescription Efficiency | |
| BHU1 | - | - | - | - | - | - |
| BHU2 | 1.03 | 1.54 | 601,265 | 17.89 | - | - |
| BHU3 | - | - | - | - | - | - |
| BHU4 | - | 0.65 | 576,808 | 59.75 | 229.78 | - |
| BHU5 | 1.46 | 0.45 | 624,615 | 50.58 | - | - |
| BHU6 | 0.89 | - | 751,928 | 48.16 | 759.55 | - |
| BHU7 | 1.62 | - | 460,540 | 16.82 | 1,148.08 | - |
| BHU8 | 0.26 | - | 311,815 | 36.85 | 206.18 | - |
| BHU9 | 2.45 | 2.02 | 708,310 | - | - | - |
| BHU10 | 0.88 | 0.26 | 526,033 | 44.05 | - | 2.52 |
| BHU11 | - | - | - | - | - | - |
| BHU12 | - | - | - | - | - | - |
| BHU13 | - | - | - | - | - | - |
| BHU14 | - | 0.73 | 428,381 | - | 165.46 | 10.06 |
| BHU15 | 0.88 | 0.71 | 660,667 | 24.80 | - | 13.54 |
| BHU16 | 0.35 | 0.56 | 310,626 | 24.27 | - | - |
| BHU17 | 1.80 | 0.30 | 573,162 | 44.63 | - | - |
| BHU18 | - | 0.07 | 93,651 | 24.23 | - | 1.31 |
| DMU | Islacks | Oslacks | ||||
|---|---|---|---|---|---|---|
| General Practitioners * | Nursing * | Pharm. Expend. * | Consultations * | Corrected Avoidable Hosp. * | Prescription Efficiency | |
| BHU1 | - | - | - | - | - | - |
| BHU2 | - | 1.59 | 173,732 | - | - | - |
| BHU3 | - | - | - | - | - | - |
| BHU4 | - | - | - | - | - | - |
| BHU5 | 1.33 | 0.04 | 155,593 | - | - | 5.54 |
| BHU6 | - | - | - | - | - | - |
| BHU7 | - | - | - | - | - | - |
| BHU8 | - | - | 51,971 | - | - | 10.80 |
| BHU9 | - | - | - | - | - | - |
| BHU10 | - | - | - | - | - | - |
| BHU11 | - | - | - | - | - | - |
| BHU12 | - | - | - | - | - | - |
| BHU13 | - | - | - | - | - | - |
| BHU14 | - | - | - | - | - | - |
| BHU15 | - | - | 206,793 | - | 1.45 | - |
| BHU16 | - | 0.23 | 200,120 | - | - | 19.16 |
| BHU17 | 1.30 | - | 97,912 | - | - | 6.64 |
| BHU18 | - | 0.04 | 93,415 | - | - | 20.52 |
| DMU | VRS Bootstrap (without Exogenous Variables) | |||
|---|---|---|---|---|
| Model 1 | Model 2 | |||
| Efficiency Score | Efficiency Score Bias-Corrected Bootstrap Confidence Intervals (BCI) 95% | Efficiency Score | Efficiency Score Bias-Corrected BCI 95% | |
| BHU1 | 1 | 0.934 [0.888–1.000] | 1 | 0.974 [0.948–1.000] |
| BHU2 | 0.934 | 0.895 [0.875–0.928] | 0.971 | 0.964 [0.957–0.981] |
| BHU3 | 1 | 0.920 [0.860–1.000] | 1 | 0.960 [0.920–1.000] |
| BHU4 | 0.917 | 0.883 [0.866–0.914] | 1 | 0.966 [0.933–1.000] |
| BHU5 | 0.789 | 0.764 [0.753–0.785] | 0.850 | 0.844 [0.837–0.870] |
| BHU6 | 0.966 | 0.943 [0.906–1.000] | 1 | 0.979 [0.958–1.000] |
| BHU7 | 0.971 | 0.919 [0.885–0.979] | 1 | 0.973 [0.946–1.000] |
| BHU8 | 0.885 | 0.841 [0.814–0.885] | 0.990 | 0.981 [0.972–1.000] |
| BHU9 | 0.951 | 0.907 [0.882–0.943] | 1 | 0.972 [0.944–1.000] |
| BHU10 | 1 | 0.915 [0.850–1.000] | 1 | 0.959 [0.919–1.000] |
| BHU11 | 1 | 0.923 [0.867–1.000] | 1 | 0.972 [0.945–1.000] |
| BHU12 | 1 | 0.912 [0.844–1.000] | 1 | 0.963 [0.926–1.000] |
| BHU13 | 1 | 0.927 [0.875–0.995] | 1 | 0.979 [0.958–1.000] |
| BHU14 | 0.947 | 0.900 [0.873–0.959] | 1 | 0.973 [0.946–1.000] |
| BHU15 | 0.857 | 0.812 [0.784–0.861] | 0.777 | 0.769 [0.761–0.806] |
| BHU16 | 0.925 | 0.881 [0.855–0.923] | 0.943 | 0.937 [0.932–0.948] |
| BHU17 | 0.870 | 0.839 [0.825–0.864] | 0.918 | 0.911 [0.905–0.932] |
| BHU18 | 0.955 | 0.917 [0.854–1.000] | 1 | 0.962 [0.923–1.000] |
| DMU | VRS with Exogenous Variables | |||
|---|---|---|---|---|
| Model 1 | Model 2 | |||
| Percentage of Population Over 65 BCI 95% | Case-Mix BCI 95% | Percentage of Population Over 65 BCI 95% | Case-Mix BCI 95% | |
| BHU1 | 0.960 [0.924–1.000] | 0.955 [0.916–0.998] | 0.985 [0.970–1.000] | 0.989 [0.979–1.000] |
| BHU2 | 0.955 [0.916–1.000] | 0.950 [0.906–1.000] | 0.988 [0.977–1.000] | 0.991 [0.983–1.000] |
| BHU3 | 0.919 [0.857–0.996] | 0.944 [0.896–1.000] | 0.939 [0.889–0.995] | 0.980 [0.962–1.000] |
| BHU4 | 0.904 [0.831–1.000] | 0.948 [0.904–1.000] | 0.927 [0.873–1.000] | 0.988 [0.976–1.000] |
| BHU5 | 0.912 [0.876–0.956] | 0.935 [0.879–0.992] | 0.918 [0.889–0.937] | 0.938 [0.923–0.967] |
| BHU6 | 0.915 [0.889–0.957] | 0.912 [0.884–0.956] | 0.966 [0.936–1.000] | 0.954 [0.916–1.000] |
| BHU7 | 0.807 [0.767–0.846] | 0.825 [0.797–0.863] | 0.753 [0.736–0.783] | 0.752 [0.738–0.772] |
| BHU8 | 0.884 [0.853–0.917] | 0.898 [0.875–0.931] | 0.928 [0.914–0.949] | 0.936 [0.929–0.945] |
| BHU9 | 0.847 [0.827–0.872] | 0.853 [0.838–0.879] | 0.905 [0.893–0.927] | 0.905 [0.894–0.925] |
| BHU10 | 0.954 [0.913–1.000] | 0.955 [0.915–1.000] | 0.984 [0.969–1.000] | 0.991 [0.982–1.000] |
| BHU11 | 0.909 [0.886–0.941] | 0.911 [0.893–0.936] | 0.959 [0.947–0.978] | 0.960 [0.948–0.978] |
| BHU12 | 0.929 [0.872–1.000] | 0.954 [0.914–1.000] | 0.967 [0.937–1.000] | 0.989 [0.979–1.000] |
| BHU13 | 0.891 [0.870–0.921] | 0.898 [0.883–0.922] | 0.967 [0.938–1.000] | 0.973 [0.950–1.000] |
| BHU14 | 0.771 [0.757–0.793] | 0.775 [0.763–0.797] | 0.832 [0.816–0.854] | 0.832 [0.819–0.860] |
| BHU15 | 0.955 [0.923–0.995] | 0.959 [0.925–1.000] | 0.972 [0.948–1.000] | 0.965 [0.935–0.997] |
| BHU16 | 0.922 [0.883–0.974] | 0.937 [0.906–0.983] | 0.969 [0.941–1.000] | 0.977 [0.956–1.000] |
| BHU17 | 0.846 [0.815–0.883] | 0.860 [0.838–0.895] | 0.977 [0.965–0.995] | 0.981 [0.972–0.991] |
| BHU18 | 0.917 [0.892–0.965] | 0.919 [0.895–0.956] | 0.969 [0.942–1.000] | 0.888 [0.852–0.918] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
González-de-Julián, S.; Barrachina-Martínez, I.; Vivas-Consuelo, D.; Bonet-Pla, Á.; Usó-Talamantes, R. Data Envelopment Analysis Applications on Primary Health Care Using Exogenous Variables and Health Outcomes. Sustainability 2021, 13, 1337. https://doi.org/10.3390/su13031337
González-de-Julián S, Barrachina-Martínez I, Vivas-Consuelo D, Bonet-Pla Á, Usó-Talamantes R. Data Envelopment Analysis Applications on Primary Health Care Using Exogenous Variables and Health Outcomes. Sustainability. 2021; 13(3):1337. https://doi.org/10.3390/su13031337
Chicago/Turabian StyleGonzález-de-Julián, Silvia, Isabel Barrachina-Martínez, David Vivas-Consuelo, Álvaro Bonet-Pla, and Ruth Usó-Talamantes. 2021. "Data Envelopment Analysis Applications on Primary Health Care Using Exogenous Variables and Health Outcomes" Sustainability 13, no. 3: 1337. https://doi.org/10.3390/su13031337
APA StyleGonzález-de-Julián, S., Barrachina-Martínez, I., Vivas-Consuelo, D., Bonet-Pla, Á., & Usó-Talamantes, R. (2021). Data Envelopment Analysis Applications on Primary Health Care Using Exogenous Variables and Health Outcomes. Sustainability, 13(3), 1337. https://doi.org/10.3390/su13031337







