Abstract
Copper (Cu) has a narrow range between optimal concentrations as a micronutrient critical for phytoplankton growth and concentrations potentially toxic to living organisms. This sensitivity indicates an ecosystem vulnerability that threatens not only nature but also human health due to bioaccumulation. An important source of elevated Cu concentrations in coastal environments are biocides used as antifouling protection on ships. A pilot study conducted in the Marine Protected Area (MPA) of the Krka Estuary (Croatia) over a period of 16 months investigated the relationship between ship traffic and Cu concentrations. The aim was to contribute to more informed environmental management by assessing the associated risks. In the study presented here, Cu concentrations were monitored, analyzed, and correlated with vessel traffic. Observations revealed that the seasonal increase in maritime traffic caused by nautical tourism was associated with an increase in Cu concentrations of more than five times, posing a toxicity risk to the environment. In order to understand the distribution of copper emissions, a mapping of maritime traffic was carried out by counting transits, radar imagery, and drone photography. This approach has proven sufficient to identify the potential risks to the marine environment and human health, thus providing an effective assessment tool for marine stakeholders.
1. Introduction
Over a 10-year period, tourist arrivals in the EU have increased by 60%, with coastal tourism accounting for a significant share of the EU maritime economy, with 86 billion EUR in gross value added [1]. As a result, maritime transport in the Mediterranean is increasing in all sectors and geographical areas. The seasonality of tourism leads to a disproportionate spatial and temporal distribution of maritime traffic, with pressure often concentrated in the most attractive and vulnerable locations, threatening their ecological status [2,3,4,5,6,7].
Nautical tourism causes environmental impacts through the discharge of effluents into the sea, the emission of air pollutants, and the degradation of marine organisms and their habitats [5,8,9]. The impacts vary depending on the size, type of vessel, and its use [5,6].
While some environmental aspects of nautical tourism, such as the discharge of effluents, chemicals or waste, the emission of air pollutants, and the impact on marine organisms and their habitats, are partially regulated, others are not [7]. A typical example of the latter is the pollutant emissions from antifouling paints, particularly copper (Cu), which was the focus of this study. Antifouling paint, also known as bottom paint, is a coating used to prevent the growth of marine organisms (biofouling) on the parts of the vessel immersed in water. Biofouling starts as soon as the surface is submerged in water [10,11,12]. This leads to a reduction in the ship’s speed and increased fuel consumption due to the increased frictional resistance [12]. In addition, the process of biofouling can lead to the introduction of new species into areas where they do not naturally occur, as they become attached to the hull of the vessel [9,13]. Antifouling biocides consist of metallic compounds that effectively kill marine organisms that attach to ship hulls, but also have serious environmental impacts due to their longevity and accumulation in the food web [14]. One of the most effective antifouling paints contains the organotin tributylin (TBT) [15,16,17]. Due to the serious negative effects, paints containing TBT are banned in the EU and many other countries [16]. Since the ban of tributyltin antifouling paints, copper-based antifouling paints now dominate [18]. Exposure to Cu can affect seagrass photosynthesis, inhibit the development of some larvae, cause genotypic shifts in polychaete populations, and bioaccumulate in marine fish, turtles, snails, barnacles, and mussels [19]. Recently, various non-toxic paints have been developed, mostly silicone-based, which do not allow fouling due to their very smooth surface [20]. These paints are still significantly more expensive and cannot be applied on uneven and unprepared surfaces, but the transition to their use is inevitable.
Copper pollution is a worldwide problem, especially in semi-enclosed areas, recreational marinas, and anchorages [21,22,23,24,25]. Research was initiated to relate shipping traffic and copper concentrations in order to develop a model for understanding environmental pollution and impacts through a pilot study on the influence of nautical tourism and associated copper (Cu) content. A highly stratified estuary of the Krka River on the eastern Adriatic coast (Croatia) was selected for the pilot study. This work aims primarily to assess the correlation between Cu concentration and nautical activities and to inform the discussion on policy structures related to Cu-related ecological and toxicological risks. The survey methodology used should also be applicable to other types of contaminants in order to estimate their ecotoxicological risk (hydrocarbons, fecal coliforms, marine litter, underwater noise, etc.). This work aims primarily to assess the relationship between Cu concentrations and nautical activities and to direct the discussion towards policy structures related to Cu-related ecological and toxicological risks, based on the literature data. The survey methodology used should also be applicable to other types of contaminants to estimate their ecotoxicological risk (hydrocarbons, fecal coliforms, marine litter, underwater noise, etc.).
Most antifouling research has focused on evaluating emissions from stationary sources [17]. The work presented here attempts to contribute by assessing the impact of antifouling on the dynamic aspects of maritime transport. In addition, the paper takes the first steps towards developing a model that maps the potential dissemination of toxic Cu through traffic counting and heat mapping. Policymakers are struggling to find effective approaches to mitigate the enormous pressures on the marine environment. The evidence presented here may prompt policy makers to bring the problem of marine pollution to the forefront and apply the precautionary principle using the methods employed here.
Study Site
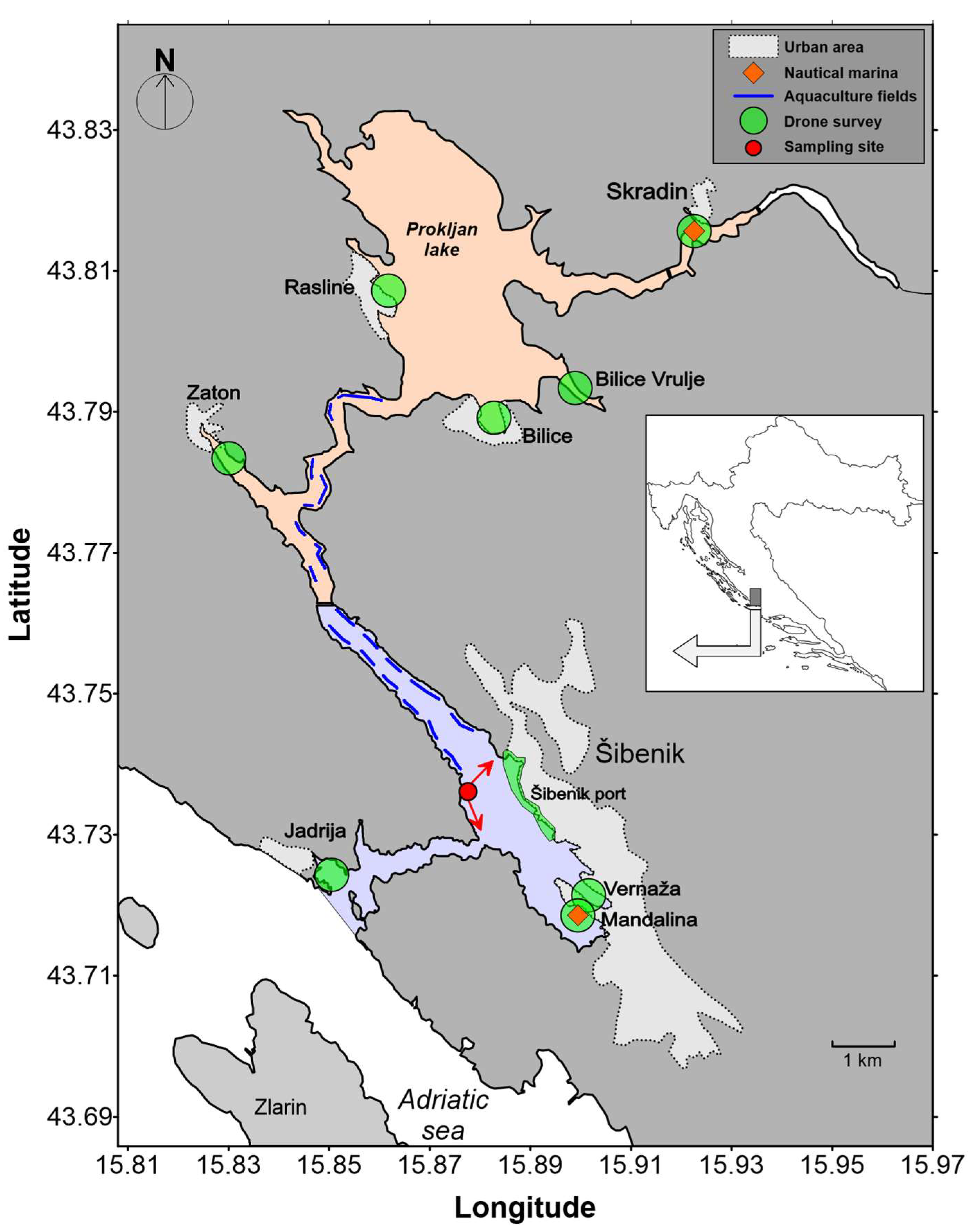
The estuary of the Krka River is a marine protected area. The length of the freshwater section is 49 km, while the estuary stretches for another 23.5 km. Since the largest part of the freshwater course, the Krka River, is protected as the Krka National Park, there is no significant pollution entering the Krka Estuary from this direction and the estuary is considered relatively unpolluted [26,27,28,29,30,31]. The estuary itself is formally protected as a NATURA 2000 site (“Krka Mouth”) and two protected regions categorized as “Significant Landscapes” by the County of Šibenik-Knin. The Krka River estuary is known as a mussel farming area (Figure 1). Numerous aquaculture facilities (20) for mussel (Mytilus galloprovincialis) farming are located mainly in the lower part of the estuary. The production of mussels in this area is about 500 tons per year.
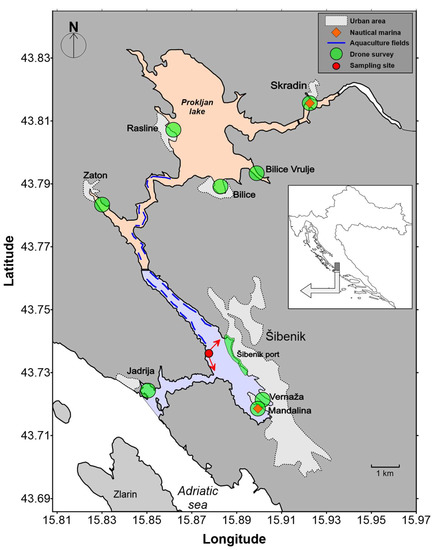
Figure 1.
Map of the study site: Red arrows show the directions of the video cameras for vessels monitoring. The colored parts represent two protected areas: the “Krka–lower part” (light orange) and the “Channel-Harbour” (light blue).
The estuary is a popular destination for nautical tourists, as the attractions of the aforementioned Krka National Park and the attractive coastal and estuarine landscapes are within easy reach. It is visited by more than 1 million people annually (before the COVID-19 pandemic). The environmental impacts of nautical and land-based tourism have not been adequately assessed due to a lack of data that would allow informed decision-making. Our preliminary “spot” count of maritime traffic (summer season 2014) showed that the number of transits during the nautical season increases by up to 1500 per day, which corresponds to a total number of almost 100,000 vessels during the summer. Although a considerable number of tourist boats pass through the area and anchor off the sensitive estuarine ecosystem of the Krka National Park, there was no reliable information on the actual movements and spatial and temporal distribution of the nautical vessels. The initial perceived risk was related to probable incidents and accidents due to seasonal intensity and overcrowding with unclear dimensions and aspects of potential impacts.
Research studies on the sources, distribution, and behavior of ecotoxic metals in the Krka River estuary have revealed an irregular “anomaly” in copper (Cu) concentrations: during the summer season, Cu concentrations are up to 20 times higher compared to the winter season [26]. The main source of the increased copper concentrations in the coastal environment is the release of copper from antifouling paints, for which it is used as a biocide.
2. Materials and Methods
2.1. Video Surveillance System for Real-Time Automatic Counting Off Vessel Passes
The camera model used for automatic vessel tracking was: DAHUA SD -59230U-HNI (Dahua Technology) with 30× optical zoom. Two cameras were used: one camera monitored the entrance to the estuary and captured vessels entering and leaving the channel, the other camera monitored vessels travelling upstream (red arrows in Figure 1).
The in-house software called: “Boats Monitoring” used for automatic counting of the vessels in real time was developed (on the Borland Delphi XE8 platform under the Windows OS environment). The description of the software can be found in the Supplementary Materials (Figures S1–S4). It has been estimated that the efficiency of automatic vessel detection is ~95% on average. The typical example of a daily “monitoring signal” is presented in Figure S5. Each peak in the figure corresponds to a passing vessel (in or out of the channel).
A separate software (“Check Boats”) was also developed to analyze the collected data in manual/automatic mode and to generate numerical and graphical reports for the selected period. The screenshots of the program are shown in Figures S3 and S4. The software was adapted to automatically exclude “false-positive” signals and to include signals for which no image was taken.
As it was not possible to find adequate non-commercial software that could automatically classify vessels from the “side view” images, the classification of vessel types into 6 different categories for 4 seasons was done manually (see Supplementary Materials for details). However, the size classification was done automatically based on the area of the vessel signal (Figure S6). The underwater surface of the vessels was classified into 3 classes (less than 7 m, 7–15 m, and more than 15 m).
2.2. Assessment of Nautical Status Using Drone Spot Monitoring and Heat Maps
To complement the video monitoring system for the defined positions within the estuary, which represents the dynamic aspect of traffic, we undertook an assessment of maritime traffic and permanent anchorages along the estuary using drones equipped with cameras. For flight planning and preparation, Litchi and Google Earth Pro software was used with the following drones and cameras:
- (a)
- DJI Phantom 3 Advanced with 12-megapixel camera HD Vide (DJI, Shenzhen, China).
- (b)
- DJI Inspire 1 v2 + Zenmuse X5 with 16-megapixel camera 2HD Video (DJI, China).
Images collected during the overflights of anchoring/docking sites were processed using Microsoft Image Composite Editor for image stitching and Agisoft Metashape for photogrammetry testing. The pixel-based image processing package ImageJ was used for object detection and measurement, allowing for the estimation of vessel dimensions (length, width) and calculation of total area for each unit.
The survey was carried out in August 2018 by UAS Consulting. All locations along the estuary used by vessels as berths and/or anchoring sites were surveyed. The number and size of vessels were revealed, which allowed for the next step—the development of an algorithm that would use the traffic count data to more accurately predict Cu loading for each location. The typical examples of the drone surveys for Skradin marina/port and Zaton port are shown in Figure S7, and the table showing the number of anchoring sites within the estuary is included in Table S1.
In order to assess the spatial distribution of Cu concentrations in the sea surface at the regional level, a picture of the intensity of maritime traffic, spatial distribution, and seasonality of maritime traffic was heat mapped (Figure S8). The mapping was carried out by the Ministry of the Sea, Transport, and Infrastructure—Maritime Safety Directorate—Using Vessel Traffic Monitoring and Information System (VTMIS) and Croatian Integrated Maritime Information System (CIMIS-a). It is based on radar tracking signals and data from Automatic Identification System (AIS) and is illustrated with pixels where the traffic intensity gradually changes from yellow to red with increasing density.
2.3. Sampling, Sample Pre-Treatment and Measurement of Copper Content
The survey was conducted between June 2018 and October 2019. Surface water sampling was carried out regularly (every 2–5 days) in front of the marine station of the Ruđer Bošković Institute in the lower part of the estuary, while additional “transect” sampling was carried out across the Šibenik bay, essentially in the direction of the upper red arrow (Figure 1).
Surface samples (depth ~ 0.5 m) were collected using a telescopic sampler or by hand-grab technique from a rubber boat (Figure S9). The sampling and storage bottles for dissolved Cu measurement and Cu speciation analysis (fluorinated ethylene-propylene (FEP), Nalgene) were previously cleaned with 10% HNO3 (analytical reagent grade) and washed several times with ASTM Type I water. A total of 90 samples were taken for the analysis of dissolved Cu concentrations. The main physico-chemical parameters (salinity, temperature, pH, and dissolved oxygen) were measured in situ using the EXO2 CTD multi-parameter probe (YSI, Xylem).
For the determination of dissolved Cu concentrations, samples were filtered immediately after sampling using precleaned 0.22 μm cellulose acetate syringe filters (Minisart, Sartorius). For the analysis of total dissolved Cu concentration, the samples were acidified in the laboratory with trace metal grade nitric acid (TraceSelect, Fluka) to a pH of <2 and directly irradiated with UV light (150 W medium pressure Hg lamp, Hanau, Germany) for at least 24 h within the next few days in the FEP bottles to decompose natural organic matter [26].
Dissolved Cu concentrations were determined by differential pulse anodic stripping voltammetry (DPASV). Measurements were performed on a Metrohm Autolab (EcoChemie) potentiostat (PGSTAT128N) controlled by GPES 4.9 software in a three-electrode cell (663 VA Stand, Metrohm). Analyses were performed using a fully automated system consisting of the instrument, a home-made autosampler and five Cavro XE 1000 syringe pumps (Tecan, San Jose, CA, USA). The concentrations of trace metals were determined using the standard addition method. A certified “Nearshore seawater reference material for trace metals”—CASS-5 (NRC CNRC) was used to validate the Cu analysis. The value obtained (±standard deviation) was 0.370 ± 0.030 μg L−1 (the certified value was 0.380 ± 0.028 μg L−1).
2.4. Estimation of the Antifouling Emission
Larger estimates of biocide emissions were carried out for the German and world merchant fleets and were validated with theoretical biocide concentrations in the North Sea [32]. Several statistical models were developed to estimate the wetted surface area (WSA) for a given set of fleet data [33]. Initially, 190,000 vessels were identified representing a WSA of 571 × 106 m2 for the global fleet, but only 120,000 vessels could be confirmed to be active. This results in a global minimum WSA estimate of approximately 325 × 106 m2. The other approach to estimate emissions was used for the Galician coast, where the mass of cupric oxides in antifouling paints was calculated [34]. The correlation between the number of recreational boats at anchorages and Cu concentrations in the water column was investigated [35,36] and hypothetical Cu loadings were calculated for cruise ships based on port data and were confirmed by bioindicators and a re-survey of sediments in the field.
Once the dimensions of the ships are known, biocide leaching rates are the final component needed for the calculation. Some of the existing research has provided the following findings:
- -
- Leaching of Cu increases with increasing salinity and higher temperatures (19° vs. 4 °C) resulted in greater metal leaching under many conditions [37].
- -
- Emissions of 8.2 ± 2.7 µg/cm2/day were measured in recreational boats [38],
- -
- Antifouling paints with nano-additives showed a slow and steady release rate in the range of 20 to 25 µg/cm2/day [39].
- -
- The copper release rate along the salinity gradient varied from almost 0 in freshwater to 25 µg/cm2/day [40].
- -
- It was recommended that future environmental impact assessments of antifouling products [21] use site-specific release rates to estimate exposures to copper and other biocides.
The antifouling used in the Adriatic Sea has already been considered in research [35], and the estimated dissolution rate was 14.1 µg/cm2/day. For an even more accurate estimate, information should be obtained from the main charter fleet providers to reveal the main antifouling brands (i.e., chemical compounds) used.
3. Results and Discussion
3.1. Monitoring of Nautical Traffic and Copper Concentrations
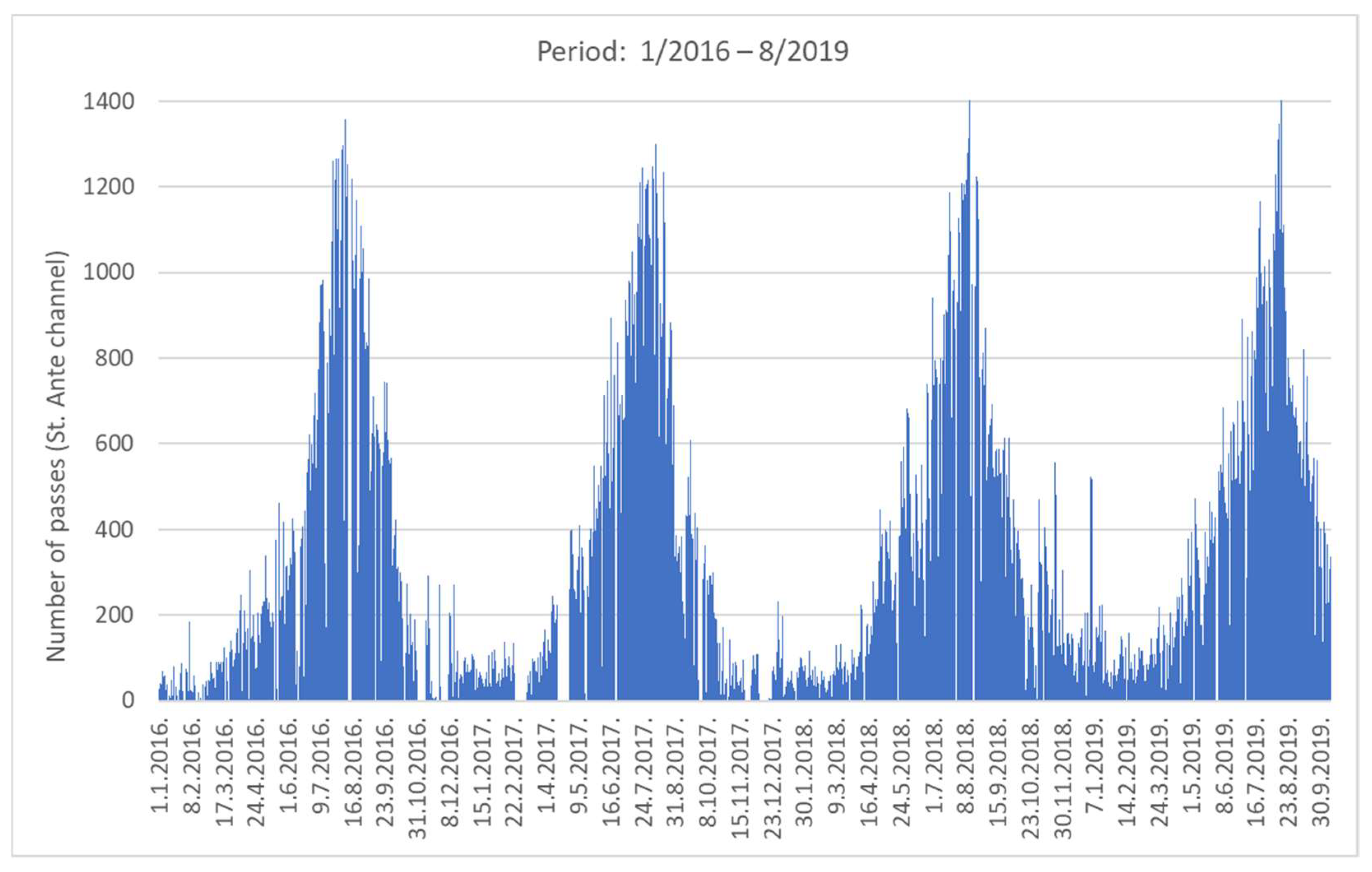
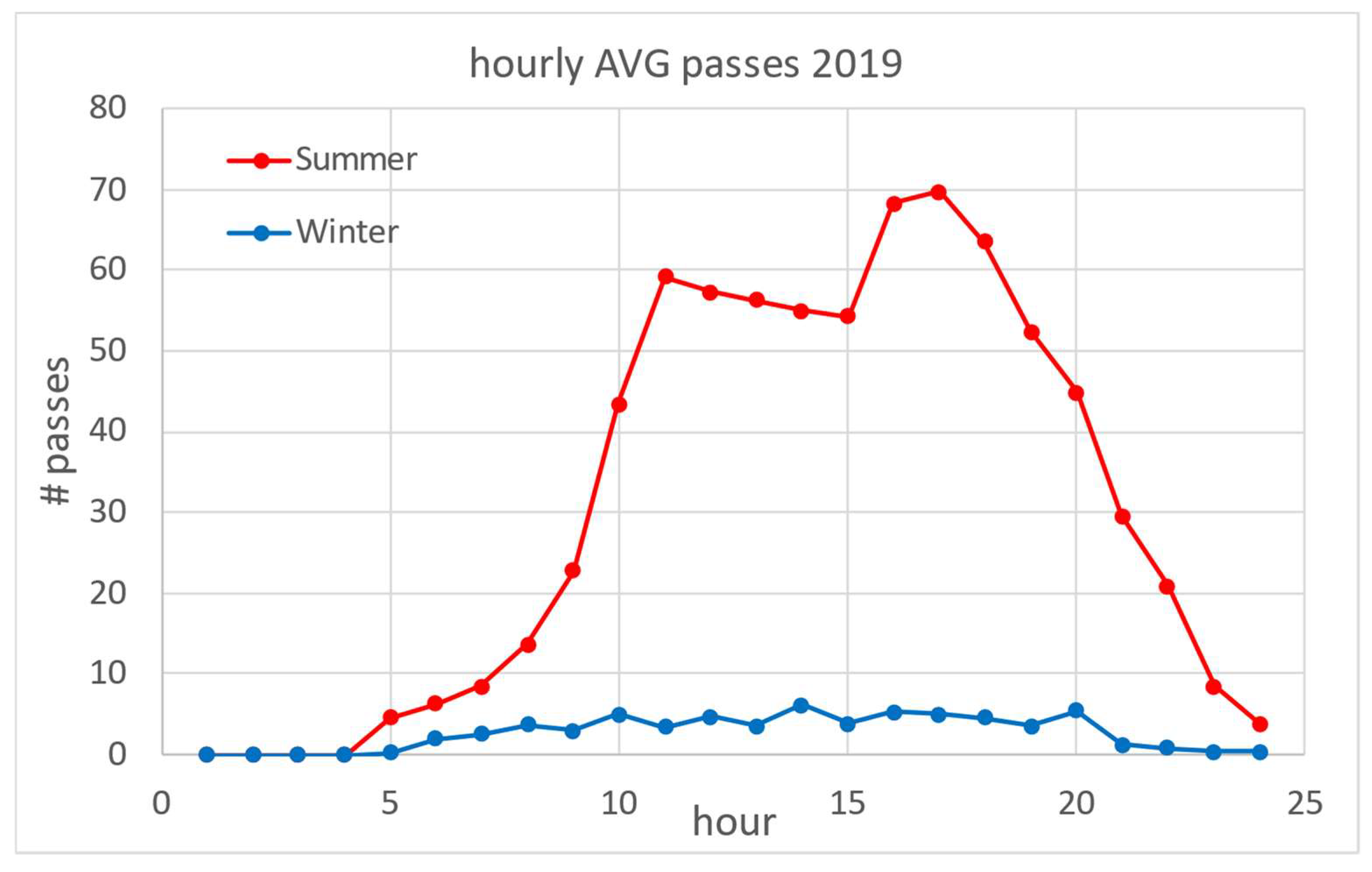
Figure 2 shows an overview of the number of vessel passages (NVP) for the 4-year period. As the estuary is a popular destination for nautical tourism, there is a clear difference in NVP between winter and summer periods: about 60 in the winter low season compared to 1400 in the summer high season. The daily distribution of averaged NVP based on a one-hour time frame is shown in Figure 3 for the winter and summer periods. In summer, the average NVP intensity increased 25-fold compared to the winter period, with the highest intensity at noon (around 11 h) and in the afternoon (around 17 h). Most vessels entered the estuary in the morning and left it in the afternoon. From the data in Figure 3, the estimated average daily NVP for the three summer months (June–August) was 750, while in winter (January–March) it was 65.
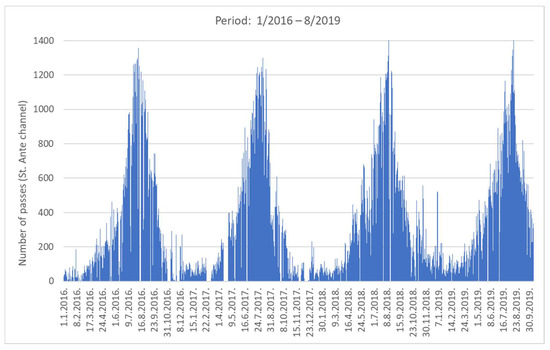
Figure 2.
Seasonality of maritime traffic: long-term maritime traffic counting through the channel (2016–2019).
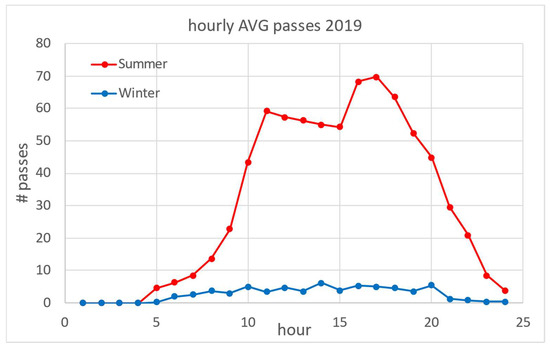
Figure 3.
Seasonal average daily vessel frequencies: distribution of average number of vessels for summer (June–August) and winter (January–March) periods.
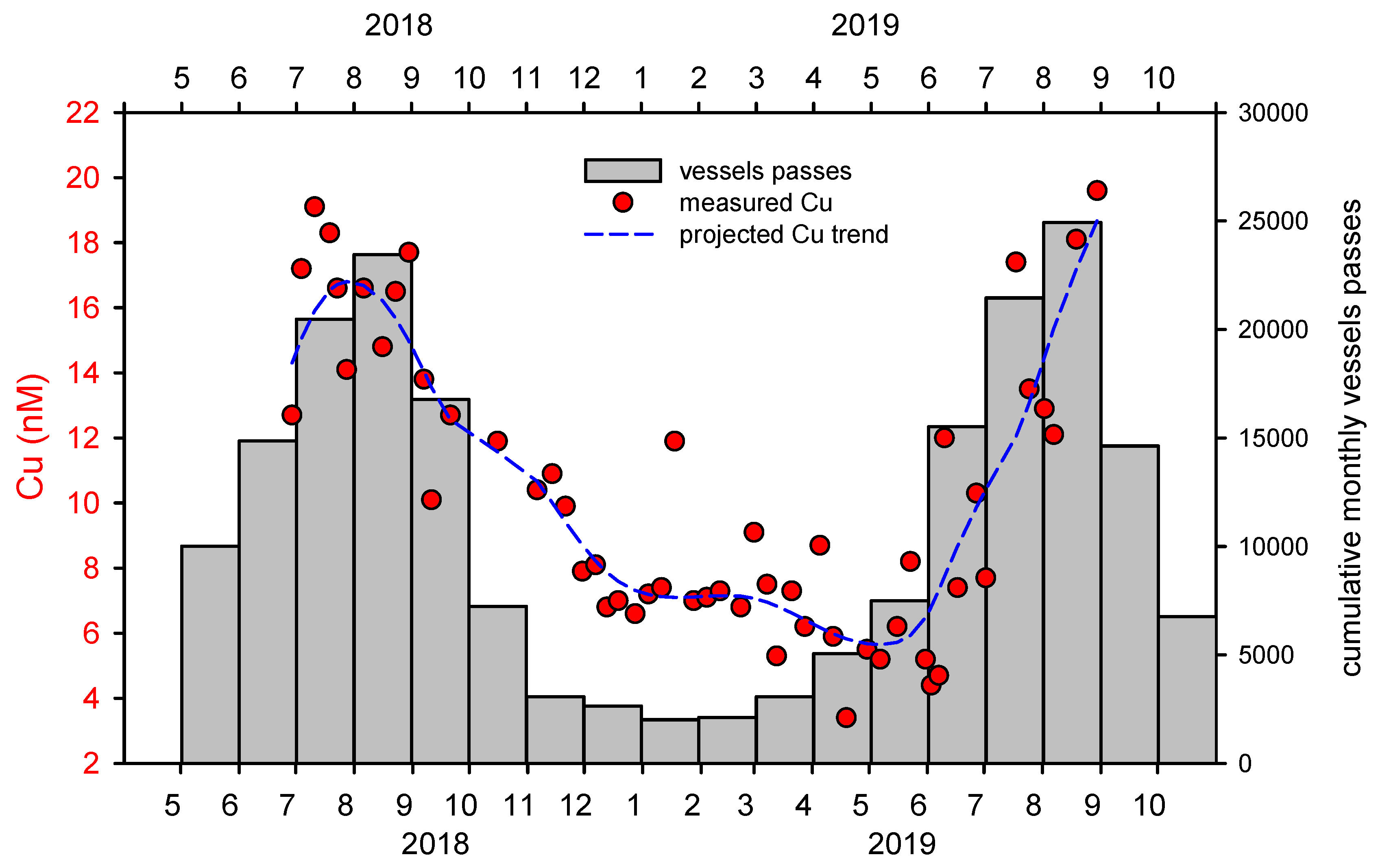
The cumulative monthly NVP for the period studied (May 2018–October 2019) is 110,000 (Figure 4). Since most boats pass in front of the camera twice (into and out of the estuary), with an average of 5 people per boat, this results in 275,000 visitors to the area for the study period. As more than 80% of the traffic in the sensitive ecosystem of the estuary takes place in a narrow summer period, this can be considered as having a very high impact and risk. The impact of the intensity of the NVP is clearly reflected in the copper concentration in the surface water at the studied site (Figure 4). In winter, dissolved copper concentrations were mostly between 3 and 7 nM. However, with the increase in NVP, the Cu concentration also increased and reached values around 20 nM. In the area of the marina in Šibenik Bay, Cu concentrations of up to 70 nM were found [26], which is more than a 20-fold increase compared to the lowest concentrations measured in our study. Note that the Cu concentration in the lower seawater layer changes only slightly throughout the year, i.e., the vertical transport down to deeper layers is insignificant (for more details see [26,27]).
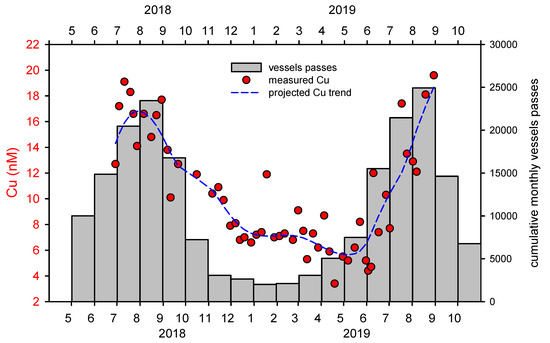
Figure 4.
Correlation of maritime traffic and Cu concentrations: Cumulative monthly vessel frequencies for the period May 2018 to October 2019 and the related concentrations of dissolved Cu.
3.2. Estimating Cu Emission from Stationary Vessels
The underwater surface area of boats in the Krka River estuary was calculated using the following equation:
In the equation, A is the submerged hull area (cm2), L is the length of the vessel, B (beam) is the width of the hull at water level, D (draft/draught) is the depth at which the vessel is below the water surface.
The size of the anchored boats was determined from images obtained from the aerial survey and the pixel-based image processing program ImageJ. Data on passing boats was obtained from a stationary camera at Naval Station Martinska, as described previously. The estimated submerged boats are listed in Table 1.

Table 1.
Estimated vessels’ underwater surface.
Copper emission from antifouling paints depends on salinity, which means that antifouling paints leach more Cu in seawater than in freshwater [41]. In the Krka River estuary, the salinity of the surface water at the study site varies from 5 in winter to almost seawater in midsummer. Most boats enter the estuary in summer, late spring, and early autumn, when salinity and copper leaching are higher. Copper emission was calculated for brackish and seawater using the estimates of Valkiris et al., 2003, and Lagerstroem et al., 2020. They estimated copper leaching to be between 0.05 and 0.25 g/m2/day. According to this estimate, between 5 and 27 kg Cu was washed out daily in the Krka River estuary during the summer season. During the winter season, the amount of copper released is much lower due to the smaller number of vessels and lower salinity.
3.3. Estimating the Cu Toxicity Risk
The notable increases in copper concentrations occur in spring, which coincides with the start of the nautical tourist season. At this time, charter yachts and private boats begin their maritime activities with freshly applied antifouling paint. Research has shown that the paint has more intense leaking at the early stages of application than at the end of the season [42]. At the same time, mussels Mytilus galoprovincialis from mussel farms and other wild mussels from the estuary release their eggs into the water column [43]. Due to this overlap of high Cu emission from the newly applied antifouling paints and the sensitive larval phase of mussels, the estuarine environment in spring can be considered toxic and therefore poses a high risk. The study on Cu toxicity to mussel embryos (48-h bioassay experiment, EC50) performed by Zitoun et al. (2019) found that Cu toxicity depends on dissolved organic carbon concentration (DOC) and salinity [44]. Furthermore, the same study found that estuarine DOC is more protective against Cu toxicity than pure marine (oceanic) DOC by buffering the effects of Cu. This is related to the higher input of humic substances by the rivers, which form strong Cu complexes, making it less bioavailable. Salinity was found to correlate negatively with the toxicity threshold, indicating a salinity-induced change in the physiology of exposed mussel embryos and/or Cu- DOC reactivity [44]. In this study, the 48 h EC50 Cu concentration was found to range from 82 to 250 nM. Cu concentrations measured in the pilot area during the summer season were up to ~ 20 nM, which is lower than the minimum Cu concentration estimated in the study by Zitoun et al. (2019). On the other hand, comparing the threshold for toxicity based on Species Sensitivity Distributions (SSD) curves, the estimated Hazardous Concertation at 5% (HC5) value for Cu is 21.9 nM [45], which is at the level we measured in the pilot area. However, we would like to emphasize that, in the study area, concentrations even more than twice as high Cu were measured [26]. Based on the above mentioned threshold literature values, these data indicate a clear toxicity risk for Mytilus galoprovincialis embryos.
In discussions with owners of mussel farms, we concluded that the first negative effects were already noticeable, as the number of larvae decreased and the farmed mussels were sometimes leaner.
In addition, many other animals besides mussels are in the larval stage, so there is a high probability of:
- Bioaccumulation;
- Reduction in food availability due to the negative effects of Cu on phytoplankton [46].
The above risks are sufficient to warrant caution.
4. Conclusions
The gaps in the implementation of the relevant policy are described in the Supplementary Materials and point to the overarching challenge—effective monitoring. Nevertheless, it should be emphasized here that environmental risks in the EU legal framework are grounded on the precautionary principle. As the toxicity of copper in the marine environment is generally neglected as a threat, it contributes to the existing unresolved problems in policy implementation. The EU’s ambitions to end emissions of hazardous substances are far from being achieved, and they are additionally compromised by new trends such as those described in this article. In other words, the research presented has provided evidence of significant ‘chemical’ threats that should be considered in marine management and planning.
Because of the vessel’s movement, Cu is emitted more rapidly than when it is at anchor or at berth [40]. In conjunction with the dramatic seasonal impacts, at least a five-fold increase in ecotoxic Cu has been found, which can harm not only marine ecosystems but also the people who depend on them.
The correlation between the seasonality of nautical tourism and copper concentrations was made possible by using existing techniques for monitoring nautical traffic (cameras, radar, drones). The data analysis system used was semi-automated and could be upgraded to a financially feasible, fully automated monitoring system.
The threats and opportunities described are an asset for informed and more competent management of marine and environmental risks, as well as for decision-making and policymaking. It also points to the need to advance research to abandon the use of heavy metal biocides as an antifouling method.
There are already alternatives to biofouling paints that can be considered as economically viable options (the initial investment is higher, but they are maintenance free for 5 years, e.g., MacGlide), although some of them are a cause for concern [47,48]. Other protective coatings against marine fouling have been proposed [49,50].
The work presented here offers a framework by relating traffic intensity to copper emissions and risks. With a closer monitoring of ship parameters, it is possible to consider other environmental impacts. The framework presented here could incorporate other parameters such as black and gray water inputs (bacteria, organic matter), bilge water (hydrocarbon emissions), marine debris (floating waste, plastics), air emissions, and underwater noise. The promotion of the ecological footprint in nautical tourism could be used with the best available methodologies for monitoring and mitigation such as: Copernicus, the EU Earth Observation Program, citizen science, real-time data sources (national and EU), artificial intelligence and Big Data. Once fully developed, the advantage could be its low cost and applicability in similar ecological areas that are understaffed, underfunded, and ineffectively monitored, such as most MPAs and NATURA 2000 marine areas in the Mediterranean.
As GDPs of EU Mediterranean member states are dependent on tourism in the range from 12 to 25%, the COVID-19 crisis has highlighted the harsh lesson of economic dependency on tourism, especially when it is positioned as an economic monoculture. As the main tourism sustainability discourse, at the moment, is centered around post-COVID-19 opportunities, with the central question being asked: is a shift away from the growth-only paradigm possible? The work presented here alarms that, not only is it possible, but past urgent.
Supplementary Materials
The following are available online at https://www.mdpi.com/article/10.3390/su132111897/s1, Figure S1: The screenshot of the boat tracking software, Figure S2: Problems associated with the correct recognition of the vessels passes: fast vessels passing caused waves, sun reflection and multiple vessels pass for the “entrance” camera, Figure S3: The screenshot of the program for analysis of collected data (pictures and signals), Figure S4: The screenshot of the program for classification and statistical analysis of boat passes, Figure S5: Typical intensity signal collected for daily monitoring of boats passes in summer period, Figure S6: Typical size classification of boat and their distribution for the June 2019, Figure S7: Examples of aerial images taken by the drone (August 2018 and August 2019), Figure S8: Representative examples of nautical traffic heatmaps for winter and summer period in wider area, Figure S9: Sampling of surface water in front of the marine station Martinska and “upstream” transversal profile sites, Figure S10: Examples of rubber boats, Figure S11: Examples of small motorboat (<10 m), Figure S12: Examples of motorboats (>10 m and <20 m), Figure S13: Examples of sailing boats, Figure S14: Examples of catamarans, Figure S15: Examples of big motorboats (>20 m), Table S1: Scanned area and the number of anchoring places with the MPA, Table S2: Classification of vessels according to types as explained above, Table S3: Overview of the EU’s legally binding policy objectives on the marine environment and chemical risks.
Author Contributions
Conceptualization, H.C., D.O. and N.C.; methodology, D.O. and N.C.; software, D.O.; validation, H.C., D.O. and N.C.; formal analysis, D.O.; data curation, D.O.; writing—original draft preparation, N.C. writing—review and editing, D.O. and H.C.; visualization, N.C. and D.O. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by MedPAN organization, organization under “NaTEF—Nautical Tourism Ecological footprint in MPAs” project.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
No public data available.
Acknowledgments
Special thanks to the UAS Consulting SPRL for support and volunteering.
Conflicts of Interest
The authors declare no conflict of interest.
References
- COGEA. Study on the Establishment of a fraMework for Processing and Analysing Maritime Economic Data in Europe. Final Report for DG Maritime Affairs and Fisheries under Contract no EASME/EMFF/2014/1.3.1.13/SI2.718095. Accompanying Statistical Database. 2017. Available online: https://op.europa.eu/en/publication-detail/-/publication/9c132514-982d-11e7-b92d-01aa75ed71a1 (accessed on 20 September 2021). [CrossRef]
- Šulc, I.; Fuerst-Bjeliš, B. Changes of tourism trajectories in (post)covidian world: Croatian perspectives. Res. Glob. 2021, 3, 100052. [Google Scholar] [CrossRef]
- Bhuiyan, M.A.; Crovella, T.; Paiano, A.; Alves, H. A Review of Research on Tourism Industry, Economic Crisis and Mitigation Process of the Loss: Analysis on Pre, During and Post Pandemic Situation. Sustainability 2021, 13, 10314. [Google Scholar] [CrossRef]
- Martínez Vázquez, R.M.; Milán García, J.; De Pablo Valenciano, J. Analysis and Trends of Global Research on Nautical, Maritime and Marine Tourism. J. Mar. Sci. Eng. 2021, 9, 93. [Google Scholar] [CrossRef]
- Ukić Boljat, H.; Grubišić, N.; Slišković, M. The Impact of Nautical Activities on the Environment—A Systematic Review of Research. Sustainability 2021, 13, 10552. [Google Scholar] [CrossRef]
- Venturini, S.; Massa, F.; Castellano, M.; Fanciulli, G.; Povero, P. Recreational boating in the Portofino Marine Protected Area (MPA), Italy: Characterization and analysis in the last decade (2006–2016) and some considerations on management. Mar. Policy 2021, 127, 103178. [Google Scholar] [CrossRef]
- Lloret, J.; Carreño, A.; Carić, H.; San, J.; Fleming, L.E. Environmental and human health impacts of cruise tourism: A review. Mar. Pollut. Bull. 2021, 173, 112979. [Google Scholar] [CrossRef]
- Carić, H.; Mackelworth, P. Cruise tourism environmental impacts—The perspective from the Adriatic Sea. Ocean Coast. Manag. 2014, 102, 350–363. [Google Scholar] [CrossRef]
- Carreno, A.; Lloret, J. Environmental impacts of increasing leisure boating activity in Mediterranean coastal waters. Ocean Coast. Manag. 2021, 209, 105693. [Google Scholar] [CrossRef]
- Wahl, M. Marine Epibiosis. I. Fouling and Antifouling: Some Basic Aspects. In Marine Ecology Progress Series; 1998; Volume 58, pp. 175–189. Available online: http://www.int-res.com/articles/meps/58/m058p175.pdf (accessed on 20 September 2021).
- Dobretsov, S.; Al-Shibli, H.; Maharachchikumbura, S.S.N.; Al-Sadi, A.M. The Presence of Marine Filamentous Fungi on a Copper-Based Antifouling Paint. Appl. Sci. 2021, 11, 8277. [Google Scholar] [CrossRef]
- Hedgepeth, J. Marine Fouling and Its Prevention; Woods Hole Oceanographic Institution: Annapolis, MA, USA, 1952; p. 580. [Google Scholar]
- Lindgren, J.; Wilewska-Bien, M.; Granhag, L.; Andersson, K.; Eriksson, K. Discharges to the sea. In Shipping and the Environment; Springer: Berlin/Heidelberg, Germany, 2016; pp. 125–168. [Google Scholar]
- Torres, G.F.; De-la-Torre, G.E. Environmental pollution with antifouling paint particles: Distribution, ecotoxicology, and sustainable alternatives. Mar. Pollut. Bull. 2021, 169, 112529. [Google Scholar] [CrossRef]
- Dafforn, K.A.; Lewis, J.A.; Johnston, E.L. Antifouling strategies: History and regulation, ecological impacts and mitigation. Mar. Pollut. Bull. 2011, 62, 453–465. [Google Scholar] [CrossRef]
- Erdelez, A.; Furdek Turk, M.; Štambuk, A.; Župan, I.; Peharda, M. Ecological quality status of the Adriatic coastal waters evaluated by the organotin pollution biomonitoring. Mar. Pollut. Bull. 2017, 123, 313–323. [Google Scholar] [CrossRef]
- Ytreberg, E.; Bighiu, M.A.; Lundgren, L.; Eklund, B. XRF measurements of tin, copper and zinc in antifouling paints coated on leisure boats. Environ. Pollut. 2016, 213, 594–599. [Google Scholar] [CrossRef] [Green Version]
- Amara, I.; Miled, W.; Slama, R.B.; Ladhari, N. Antifouling processes and toxicity effects of antifouling paints on marine environment. A review. Environ. Toxicol. Pharmacol. 2018, 57, 115–130. [Google Scholar] [CrossRef]
- Leon, L.M.; Warnken, J. Copper and sewage inputs from recreational vessels at popular anchor sites in a semi-enclosed Bay (Qld, Australia): Estimates of potential annual loads. Mar. Pollut. Bull. 2008, 57, 838–845. [Google Scholar] [CrossRef]
- MACGlide. MACmar—MacGlide Foil. Available online: https://www.macglide.eu/ (accessed on 20 September 2021).
- Ytreberg, E.; Lagerstrom, M.; Nou, S.; Wiklund, A.E. Environmental risk assesment of using antifouling paints on pleasure crafts in European Union waters. J. Environ. Manag. 2021, 281, 111846. [Google Scholar] [CrossRef] [PubMed]
- Cukrov, N.; Frančišković-Bilinski, S.; Bogner, D. Metal contamination recorded in the sediment of the semi-closed Bakar Bay (Croatia). Environ. Geochem. Health 2014, 36, 195–208. [Google Scholar] [CrossRef] [PubMed]
- Adeleye, A.S.; Oranu, E.A.; Tao, M.; Keller, A.A. Release and detection of nanosized copper from a commercial antifouling paint. Water Res. 2016, 102, 374–382. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Deaehne, D.; Fürle, C.; Thomsen, A.; Watermann, B.; Feibicke, M. Antifouling Biocides in German Marinas: Exposure Assessment and Calculation of National Consumption and Emission. Integr. Environ. Assess. Manag. 2017, 13, 892–905. [Google Scholar] [CrossRef]
- Di Cesare, A.; Pjevac, P.; Eckert, E.; Cukrov, N.; Šparica, M.M.; Corno, G.; Orlić, S. The role of metal contamination in shaping microbial communities in heavily polluted marine sediments. Environ. Pollut. 2020, 265, 114823. [Google Scholar] [CrossRef]
- Cindrić, A.M.; Garnier, C.; Oursel, B.; Pižeta, I.; Omanović, D. Evidencing the natural and anthropogenic processes controlling trace metals dynamic in a highly stratified estuary: The Krka River estuary (Adriatic, Croatia). Mar. Pollut. Bull. 2015, 94, 199–216. [Google Scholar] [CrossRef] [Green Version]
- Cindrić, A.M.; Marcinek, S.; Garnier, C.; Salaun, P.; Cukrov, N.; Oursel, B.; Lenoble, V.; Omanović, D. Evaluation of diffusive gradients in thin films (DGT) technique for speciation of trace metals in estuarine waters—A multimethodological approach. Sci. Total Environ. 2020, 721, 137784. [Google Scholar] [CrossRef] [PubMed]
- Cukrov, N.; Frančišković-Bilinski, S.; Mikac, N.; Roje, V. Natural and anthropogenic influences recorded in sediments from the Krka river estuary (Eastern Adriatic coast), evaluated by statistical methods. Fresen. Environ. Bull. 2008, 17, 855–863. [Google Scholar]
- Cukrov, N.; Mlakar, M.; Cuculić, V.; Barišić, D. Origin and transport of U-238 and Ra-226 in riverine, estuarine and marine sediments of the Krka River, Croatia. J. Environ. Radioact. 2009, 100, 497–504. [Google Scholar] [CrossRef]
- Cukrov, N.; Tepić, N.; Omanović, D.; Lojen, S.; Bura-Nakić, E.; Vojvodić, V.; Pižeta, I. Qualitative interpretation of physico-chemical and isotopic parameters in the Krka River (Croatia) assessed by multivariate statistical analysis. Int. J. Environ. Anal. Chem. 2012, 92, 1187–1199. [Google Scholar] [CrossRef]
- Louis, Y.; Garnier, C.; Lenoble, V.; Mounier, S.; Cukrov, N.; Omanović, D.; Pižeta, I. Kinetic and equilibrium studies of copper-dissolved organic matter complexation in water column of the stratified Krka River estuary (Croatia). Mar. Chem. 2009, 114, 110–119. [Google Scholar] [CrossRef]
- Isensee, J.; Watermann, B.; Berger, H.-D. Emissions of antifouling-biocides into the north sea -an estimation. Dtsch. Hydrogr. Z. 1994, 46, 355–365. [Google Scholar] [CrossRef]
- Moser, C.S.; Wier, T.P.; Grant, J.F.; First, M.R.; Tamburri, M.N.; Ruiz, G.M.; Miller, A.W.; Drake, L.A. Quantifying the total wetted surface area of the world fleet: A first step in determining the potential extent of ships’ biofouling. Biol. Invasions 2016, 18, 265–277. [Google Scholar] [CrossRef]
- Rodriguez, M.J.; Fraguela, J.A.; Carral, L. Estimating Cupric Oxide Levels in Galician Sea Water (Nw Spain) from Antifouling Paint for Recreational Crafts. Thalassas 2015, 31, 51–56. [Google Scholar]
- Carić, H.; Klobučar, G.; Štambuk, A. Ecotoxicological risk assessment of antifouling emissions in a cruise ship port. J. Clean. Prod. 2016, 121, 159–168. [Google Scholar] [CrossRef]
- Warnken, J.; Dunn, R.J.K.; Teasdale, P.R. Investigation of recreational boats as a source of copper at anchorage sites using time-integrated diffusive gradients in thin film and sediment measurements. Mar. Pollut. Bull. 2004, 49, 833–843. [Google Scholar] [CrossRef]
- Singh, N.; Turner, A. Leaching of copper and zinc from spent antifouling paint particles. Environ. Pollut. 2009, 157, 371–376. [Google Scholar] [CrossRef]
- Valkirs, A.O.; Seligman, P.F.; Haslbeck, E.; Caso, J.S. Measurement of copper release rates from antifouling paint under laboratory and in situ conditions: Implications for loading estimation to marine water bodies. Mar. Pollut. Bull. 2003, 46, 763–779. [Google Scholar] [CrossRef]
- Zhao, X.; Fan, W.; Hou, B. A research on release of copper ions and panels in shallow submergence of antifouling paints with nano-additives. Adv. Mater. Res. 2013, 791, 179–182. [Google Scholar]
- Lagerstrom, M.; Ytreberg, E.; Wiklund, A.K.E.; Granhag, L. Antifouling paints leach copper in excess—Study of metal release rates and efficacy along a salinity gradient. Water Res. 2020, 186, 116383. [Google Scholar] [CrossRef]
- Lagerstrom, M.; Yngsell, D.; Eklund, B.; Ytreberg, E. Identification of commercial and recreational vessels coated with banned organotin paint through screening of tin by portable XRF. J. Hazard. Mater. 2019, 362, 107–114. [Google Scholar] [CrossRef] [PubMed]
- Earley, P.E.; Swope, B.L.; Barbeau, K.; Bundy, R.; McDonald, J.A.; Rivera-Duarte, I. Life cycle contributions of copper from vessel painting and maintenance activities. Biofouling 2014, 30, 51–68. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Marguš, D.; Teskeredžić, E. Settlement of Mussels (Mytilus-Galloprovincialis Lamarck) on Rope Collectors in the Estuary of the River Krka, Yugoslavia. Aquaculture 1986, 55, 285–296. [Google Scholar] [CrossRef]
- Zitoun, R.; Clearwater, S.J.; Hassler, C.; Thompson, K.J.; Albert, A.; Sander, S.G. Copper toxicity to blue mussel embryos (Mytilus galloprovincialis): The effect of natural dissolved organic matter on copper toxicity in estuarine waters. Sci. Total Environ. 2019, 653, 300–314. [Google Scholar] [CrossRef]
- Duran, I.; Beiras, R. Ecotoxicologically based marine acute water quality criteria for metals intended for protection of coastal areas. Sci. Total Environ. 2013, 463, 446–453. [Google Scholar] [CrossRef]
- Echeveste, P.; Croot, P.; von Dassow, P. Differences in the sensitivity to Cu and ligand production of coastal vs offshore strains of Emiliania huxleyi. Sci. Total Environ. 2018, 625, 1673–1680. [Google Scholar] [CrossRef] [PubMed]
- de Jesus, E.P.S.; de Figueiredo, L.; Pitombeira, M.F.; Martins, R.; Nilin, J. Acute and chronic effects of innovative antifouling nanostructured biocides on a tropical marine microcrustacean. Mar. Pollut. Bull. 2021, 164, 111970. [Google Scholar] [CrossRef] [PubMed]
- Watermann, B.; Eklund, B. Can the input of biocides and polymeric substances from antifouling paints into the sea be reduced by the use of non-toxic hard coatings? Mar. Pollut. Bull. 2019, 144, 146–151. [Google Scholar] [CrossRef]
- Gu, Y.; Yu, L.; Mou, J.; Wu, D.; Xu, M.; Zhou, P.; Ren, Y. Research Strategies to Develop Environmentally Friendly Marine Antifouling Coatings. Mar. Drugs 2020, 18, 371. [Google Scholar] [CrossRef]
- Pistone, A.; Scolaro, C.; Visco, A. Mechanical Properties of Protective Coatings against Marine Fouling: A Review. Polymers 2021, 13, 173. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).