Closer to Carrying Capacity: Analysis of the Internal Demographic Structure Associated with the Management and Density Dependence of a Controlled Wolf Population in Latvia
Abstract
:1. Introduction
2. Methods
2.1. Study Area and Culling Practice
2.2. Sample Collection
2.3. Estimating Abundance and Culling Impact
- (1)
- Despite increasing efforts of noninvasive monitoring by the SFS, harvest data are currently the only robust and comparable information on the wolf population. Neither countrywide surveys for dens nor counts of packs and their sizes have been regularly conducted.
- (2)
- Due to a considerably large quota, harvesting is believed to be the main source of wolf mortality. Natural wolf mortality at various age classes in Latvia is not known but is believed to be considerably lower than (and perhaps partially compensated for by) harvest mortality [33].
- (3)
- A lack of reliable data on local hunting efforts (e.g., duration and number of hunters involved) forbids using harvest-based population estimation methods that incorporate catch per unit of effort.
3. Results
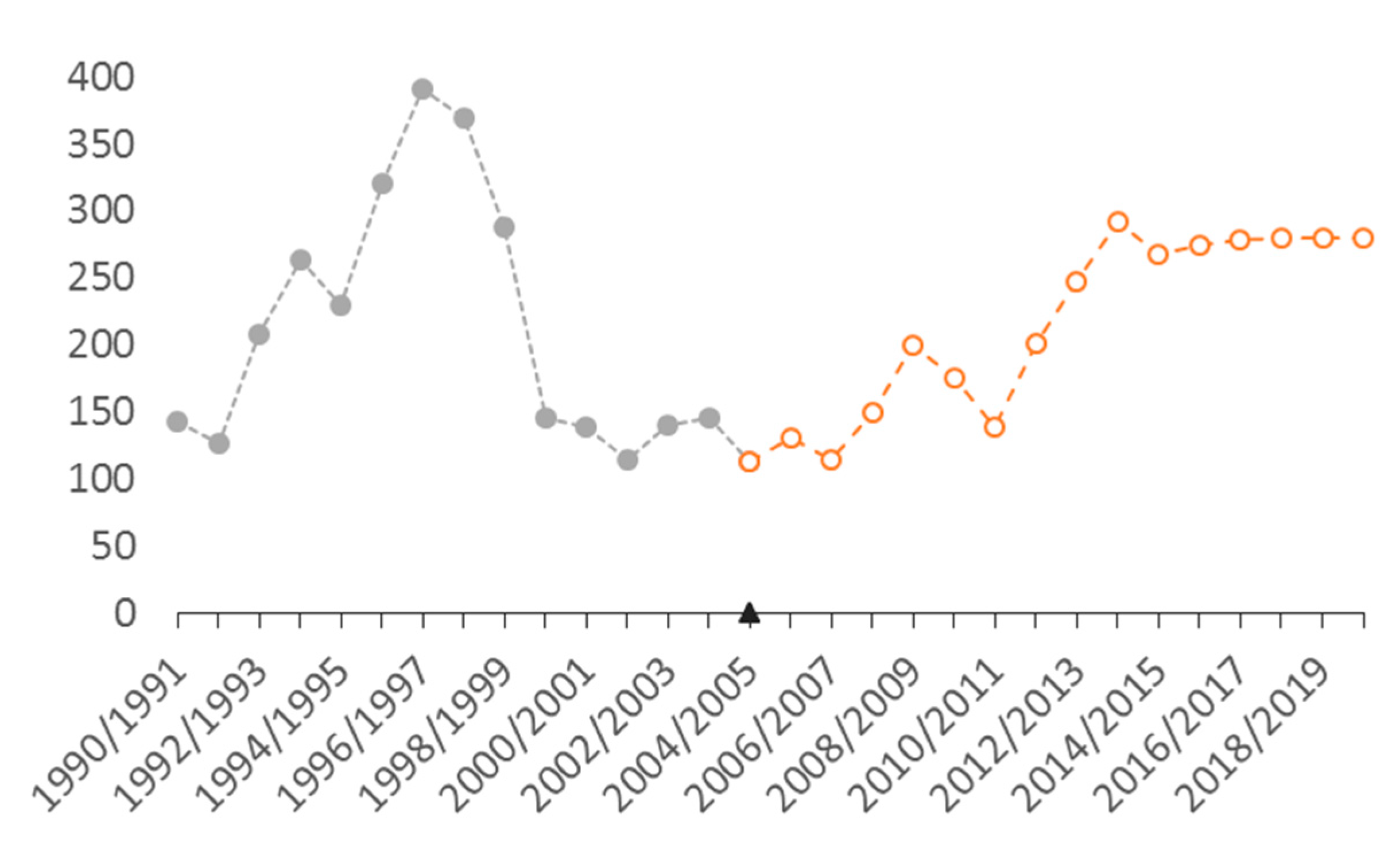
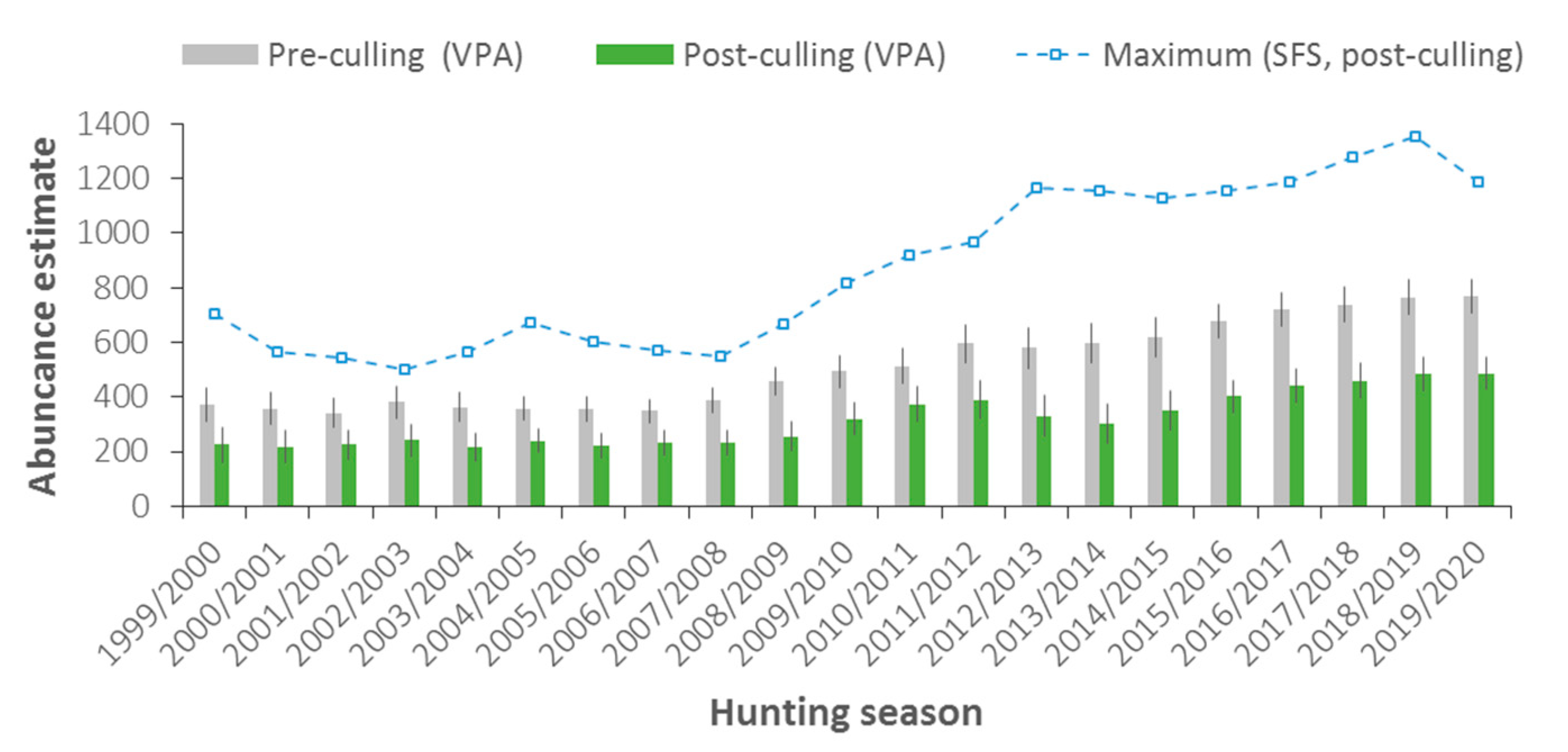
3.1. Abundance and Population Dynamics
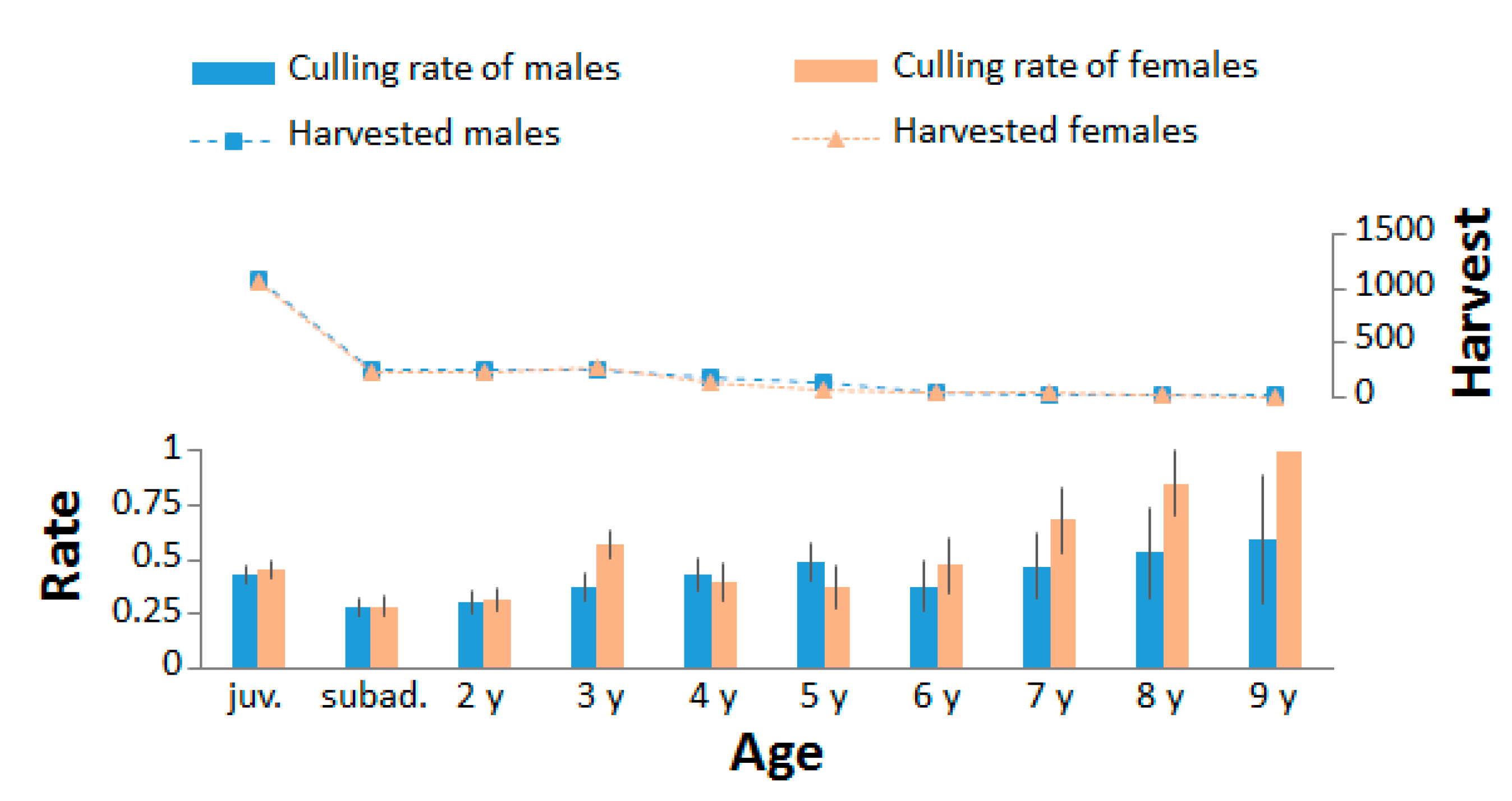
3.2. Sex and Age Structure and Culling Rate
3.3. Female Reproduction Rate and Fecundity
4. Discussion
4.1. Abundance, Population Dynamics, and Carrying Capacity
4.2. Culling Rate Assessment and Impact on Demography and Reproduction
4.3. Implications for Conservation and Management
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A. Data Used in the Study
| Hunting Season | Total Harvest (Number of Culled and Otherwise Recovered Individuals) | Ratio of Males to Females | Estimated Number of Individuals in the Harvest According to Age | |||
|---|---|---|---|---|---|---|
| Juveniles | Yearlings | 2 Years | 3 Years | |||
| 1999/2000 | 146 | 0.825 | 38.24 ± 19.52 | 20.86 ± 15.53 | 41.71 ± 20.05 | 24.33 ± 16.54 |
| 2000/2001 | 139 | 0.853 | 40.24 ± 20.44 | 21.95 ± 16.43 | 29.26 ± 18.37 | 21.95 ± 16.43 |
| 2001/2002 | 114 | 0.9 | 46.44 ± 12.81 | 33.78 ± 11.91 | 21.11 ± 10.13 | 2.11 ± 3.52 |
| 2002/2003 | 140 | 0.867 | 80 ± 23.87 | 20 ± 16.88 | 12 ± 13.5 | 16 ± 15.35 |
| 2003/2004 | 146 | 0.825 | 59.9 ± 23.05 | 18.72 ± 15.67 | 18.72 ± 15.67 | 18.72 ± 15.67 |
| 2004/2005 | 119 | 1.447 | 62.53 ± 12.69 | 10.08 ± 7.08 | 8.07 ± 6.39 | 8.07 ± 6.39 |
| 2005/2006 | 134 | 1.129 | 57.11 ± 14.45 | 21.97 ± 10.82 | 8.79 ± 7.23 | 17.57 ± 9.86 |
| 2006/2007 | 116 | 1.5 | 48.65 ± 11.45 | 9.35 ± 6.32 | 14.97 ± 7.78 | 26.19 ± 9.7 |
| 2007/2008 | 155 | 0.987 | 66.2 ± 11.03 | 14.53 ± 6.5 | 16.15 ± 6.81 | 22.6 ± 7.87 |
| 2008/2009 | 202 | 0.922 | 120.11 ± 14.39 | 23.66 ± 9.43 | 16.38 ± 8 | 12.74 ± 7.13 |
| 2009/2010 | 172 | 1.25 | 103.55 ± 12.74 | 14.04 ± 7.12 | 14.04 ± 7.12 | 19.31 ± 8.21 |
| 2010/2011 | 141 | 0.986 | 76.31 ± 11 | 14.93 ± 6.79 | 14.93 ± 6.79 | 8.29 ± 5.19 |
| 2011/2012 | 206 | 1.138 | 123.6 ± 16.52 | 14.42 ± 8.6 | 28.84 ± 11.7 | 20.6 ± 10.11 |
| 2012/2013 | 248 | 1.157 | 110.22 ± 25.72 | 9.19 ± 9.77 | 24.49 ± 15.44 | 42.86 ± 19.57 |
| 2013/2014 | 294 | 1.014 | 162.21 ± 30.06 | 6.76 ± 9.06 | 27.03 ± 17.46 | 37.17 ± 20.09 |
| 2014/2015 | 267 | 1.153 | 176.32 ± 35.96 | 15.11 ± 17.55 | 20.15 ± 20.06 | 25.19 ± 22.19 |
| 2015/2016 | 275 | 1.116 | 173.68 ± 21.63 | 31.36 ± 14.26 | 24.12 ± 12.69 | 19.3 ± 11.46 |
| 2016/2017 | 279 | 1.182 | 173.43 ± 22.69 | 37.7 ± 15.99 | 25.14 ± 13.39 | 12.57 ± 9.7 |
| 2017/2018 | 280 | 1 | 127.77 ± 24.9 | 38.06 ± 17.13 | 35.34 ± 16.6 | 35.34 ± 16.6 |
| 2018/2019 | 280 | 1.345 | 146.54 ± 24.21 | 28.79 ± 14.72 | 23.55 ± 13.45 | 34.02 ± 15.83 |
| 2019/2020 | 280 | 1.5 | 118.17 ± 23.57 | 41.1 ± 16.89 | 43.67 ± 17.32 | 33.39 ± 15.47 |
| Hunting Season | Estimated Number of Individuals in the Harvest According to Age | |||||
| 4 Years | 5 Years | 6 Years | 7 Years | 8 Years | 9 Years | |
| 1999/2000 | 3.48 ± 6.77 | 6.95 ± 9.45 | 3.48 ± 6.77 | 3.48 ± 6.77 | 3.48 ± 6.77 | 0 ± 6.39 |
| 2000/2001 | 10.97 ± 12.15 | 3.66 ± 7.21 | 3.66 ± 7.21 | 7.32 ± 10.06 | 0 ± 6.77 | 0 ± 6.77 |
| 2001/2002 | 4.22 ± 4.92 | 0 ± 3.36 | 2.11 ± 3.52 | 4.22 ± 4.92 | 0 ± 3.36 | 0 ± 3.36 |
| 2002/2003 | 0 ± 7.51 | 8 ± 11.2 | 4 ± 8.04 | 0 ± 7.51 | 0 ± 7.51 | 0 ± 7.51 |
| 2003/2004 | 11.23 ± 12.49 | 3.74 ± 7.41 | 3.74 ± 7.41 | 7.49 ± 10.34 | 0 ± 6.97 | 3.74 ± 7.41 |
| 2004/2005 | 10.08 ± 7.08 | 6.05 ± 5.58 | 6.05 ± 5.58 | 0 ± 3.15 | 4.03 ± 4.6 | 0 ± 3.15 |
| 2005/2006 | 15.38 ± 9.31 | 8.79 ± 7.23 | 0 ± 3.57 | 2.2 ± 3.71 | 0 ± 3.57 | 2.2 ± 3.71 |
| 2006/2007 | 5.61 ± 4.98 | 5.61 ± 4.98 | 0 ± 2.81 | 1.87 ± 2.92 | 3.74 ± 4.1 | 0 ± 2.81 |
| 2007/2008 | 11.3 ± 5.8 | 6.46 ± 4.46 | 6.46 ± 4.46 | 6.46 ± 4.46 | 3.23 ± 3.19 | 1.61 ± 2.26 |
| 2008/2009 | 12.74 ± 7.13 | 5.46 ± 4.75 | 7.28 ± 5.46 | 1.82 ± 2.77 | 1.82 ± 2.77 | 0 ± 2.71 |
| 2009/2010 | 12.29 ± 6.7 | 1.76 ± 2.62 | 0 ± 2.55 | 7.02 ± 5.15 | 0 ± 2.55 | 0 ± 2.55 |
| 2010/2011 | 11.61 ± 6.07 | 6.64 ± 4.67 | 4.98 ± 4.07 | 0 ± 2.31 | 3.32 ± 3.35 | 0 ± 2.31 |
| 2011/2012 | 14.42 ± 8.6 | 2.06 ± 3.35 | 2.06 ± 3.35 | 0 ± 3.27 | 0 ± 3.27 | 0 ± 3.27 |
| 2012/2013 | 30.62 ± 17.03 | 21.43 ± 14.54 | 9.19 ± 9.77 | 0 ± 5.55 | 0 ± 5.55 | 0 ± 5.55 |
| 2013/2014 | 37.17 ± 20.09 | 13.52 ± 12.66 | 6.76 ± 9.06 | 3.38 ± 6.44 | 0 ± 6.26 | 0 ± 6.26 |
| 2014/2015 | 20.15 ± 20.06 | 5.04 ± 10.33 | 5.04 ± 10.33 | 0 ± 9.87 | 0 ± 9.87 | 0 ± 9.87 |
| 2015/2016 | 16.89 ± 10.77 | 7.24 ± 7.18 | 0 ± 4.09 | 0 ± 4.09 | 0 ± 4.09 | 2.41 ± 4.18 |
| 2016/2017 | 7.54 ± 7.59 | 15.08 ± 10.58 | 5.03 ± 6.22 | 2.51 ± 4.42 | 0 ± 4.32 | 0 ± 4.32 |
| 2017/2018 | 19.03 ± 12.58 | 10.87 ± 9.66 | 0 ± 4.79 | 5.44 ± 6.9 | 2.72 ± 4.9 | 5.44 ± 6.9 |
| 2018/2019 | 18.32 ± 11.98 | 10.47 ± 9.19 | 10.47 ± 9.19 | 5.23 ± 6.56 | 2.62 ± 4.66 | 0 ± 4.56 |
| 2019/2020 | 17.98 ± 11.7 | 12.84 ± 9.98 | 10.28 ± 8.97 | 2.57 ± 4.55 | 0 ± 4.45 | 0 ± 4.45 |
| Period | No Traces of Breeding | Confirmed Pregnancy | Mean Number of Placental Marks or Embryos |
|---|---|---|---|
| 1998–2001 | 9 | 22 | 6.1 (SD = 1.6, n = 16) |
| 2002−2004 | 7 | 15 | 6.3 (SD = 2.1, n = 16) |
| 2005–2007 | 14 | 21 | 6.6 (SD = 2.2, n = 23) |
| 2008–2010 | 7 | 15 | 6.1 (SD = 2.3, n = 13) |
| 2011–2013 | 7 | 14 | 5 (SD = 2.7, n = 31) |
| 2014–2016 | 18 | 17 | 6.5 (SD = 3, n = 27) |
| 2017–2019 | 14 | 26 | 6.5 (SD = 1.9, n = 29) |
| Total/mean | 76 | 130 | 6.1 (SD = 2.4, n = 155) |
References
- Breitenmoser, U. Large predators in the Alps: The fall and rise of man’s competitors. Biol. Conserv. 1998, 83, 279–289. [Google Scholar] [CrossRef]
- Linnell, J.D.C.; Andersen, R.; Kvam, T.O.R.; Andren, H.; Liberg, O.; Odden, J.; Moa, P.F. Home range size and choice of management strategy for lynx in Scandinavia. Environ. Manag. 2001, 27, 869–879. [Google Scholar] [CrossRef]
- Fritts, S.H.; Stephenson, R.O.; Hayes, R.D.; Boitani, L. Wolves and humans. In Wolves: Behavior, Ecology, and Conservation; Mech, D.L., Boitani, L., Eds.; University of Chicago Press: Chicago, IL, USA, 2003; pp. 289–316. [Google Scholar] [CrossRef]
- Chapron, G.; Kaczensky, P.; Linnell, J.D.C.; Von Arx, M.; Huber, D.; Andrén, H.; López-Bao, J.V.; Adamec, M.; Álvares, F.; Anders, O.; et al. Recovery of large carnivores in Europe’s modern human-dominated landscapes. Science 2014, 346, 1517–1519. [Google Scholar] [CrossRef] [Green Version]
- Pletscher, D.H.; Ream, R.R.; Boyd, D.K.; Fairchild, M.W.; Kunkel, K.E. Population dynamics of a recolonizing wolf population. J. Wildl. Manag. 1997, 61, 459–465. [Google Scholar] [CrossRef]
- Boitani, L. Action Plan for the Conservation of Wolves in Europe (Canis Lupus); Council of Europe: Strasbourg, France, 2000. [Google Scholar]
- Hayes, R.D.; Harestad, A.S. Demography of a recovering wolf population in the Yukon. Can. J. Zool. 2000, 78, 36–48. [Google Scholar] [CrossRef]
- Smith, D.W.; Peterson, R.O.; Houston, D.B. Yellowstone after wolves. BioScience 2003, 53, 330–340. [Google Scholar] [CrossRef]
- Vilà, C.; Sundqvist, A.K.; Flagstad, Ø.; Seddon, J.; Björnerfeldt, S.B.; Kojola, I.; Casulli, A.; Sand, H.; Wabakken, P.; Ellegren, H. Rescue of a severely bottlenecked wolf (Canis lupus) population by a single immigrant. Proc. R. Soc. Lond. B Biol. Sci. 2003, 270, 91–97. [Google Scholar] [CrossRef] [Green Version]
- Linnell, J.D.C.; Brøseth, H.; Solberg, E.J.; Brainerd, S.M. The origins of the southern Scandinavian wolf Canis lupus population: Potential for natural immigration in relation to dispersal distances, geography and Baltic ice. Wildl. Biol. 2005, 11, 383–391. [Google Scholar] [CrossRef]
- Reinhardt, I.; Kluth, G.; Nowak, C.; Szentiks, C.A.; Krone, O.; Ansorge, H.; Mueller, T. Military training areas facilitate the recolonization of wolves in Germany. Conserv. Lett. 2019, 12, e12635. [Google Scholar] [CrossRef]
- Szewczyk, M.; Nowak, S.; Niedźwiecka, N.; Hulva, P.; Špinkytė-Bačkaitienė, R.; Demjanovičová, K.; Bolfíková, B.Č.; Antal, V.; Fenchuk, V.; Figura, M.; et al. Dynamic range expansion leads to establishment of a new, genetically distinct wolf population in Central Europe. Sci. Rep. 2019, 9, 19003. [Google Scholar] [CrossRef]
- Kaczensky, P. Large carnivore depredation on livestock in Europe. Ursus 1999, 11, 59–71. [Google Scholar]
- Skogen, K. Adapting adaptive management to a cultural understanding of land use conflicts. Soc. Nat. Resour. 2003, 16, 435–450. [Google Scholar] [CrossRef]
- Mech, L.D. Where can wolves live and how can we live with them? Biol. Conserv. 2017, 210, 310–317. [Google Scholar] [CrossRef] [Green Version]
- Kouřilová, J.; Lososová, J.; Dohnalová, A. Discussion of the Issue of Wolf Expansion with an Impact on Landscape Management and Farming. Acta Univ. Bohem. Merid. 2019, 22, 1–12. [Google Scholar] [CrossRef] [Green Version]
- Linnell, J.; Salvatori, V.; Boitani, L. Guidelines for population level management plans for large carnivores in Europe. In A LCIE Report Prepared for the European Commission (Contract 070501/2005/424162/MAR/B2); Large Carnivore Initiative for Europe; IUCN/SSC Working Group: Rome, Italy, 2008. [Google Scholar]
- Frank, L.G.; Woodroffe, R. Behaviour of carnivores in exploited and controlled populations. In Carnivore Conservation; Gittleman, J.L., Funk, S.M., Macdonald, D.W., Wayne, R.K., Eds.; Cambridge University Press: Cambridge, UK, 2001; pp. 419–442. [Google Scholar]
- Ginsberg, J.R. Setting priorities for carnivore conservation: What makes carnivores different? In Carnivore Conservation; Gittleman, J.L., Funk, S.M., Macdonald, D.W., Wayne, R.K., Eds.; Cambridge University Press: Cambridge, UK, 2001; pp. 498–523. [Google Scholar]
- Ozoliņš, J.; Stepanova, A.; Žunna, A.; Bagrade, G.; Ornicāns, A. Wolf hunting in Latvia in the light of population continuity in the Baltics. In Beiträge zur Jagd-und Wildforschung; Bd., 36; Stubbe, M., Ed.; Gesellschaft für Wildtier- und Jagdforschung e.V.: Halle, Germany, 2011; pp. 93–104. [Google Scholar]
- Ozoliņš, J.; Männil, P.; Balčiauskas, L. Ecological, social and economic justification of wolf population management in the Baltic region. In Beiträge zur Jagd-und Wildforschung; Bd., 39; Stubbe, M., Ed.; Gesellschaft für Wildtier- und Jagdforschung e.V.: Halle, Germany, 2014; pp. 215–224. [Google Scholar]
- Kawata, Y. Estimation of carrying capacities of large carnivores in Latvia. Acta Zool. Litu. 2008, 18, 3–9. [Google Scholar] [CrossRef]
- Balčiauskas, L.; Kawata, Y. Estimation of carrying capacity and growth rate of wolf in Lithuania. Acta Zool. Litu. 2009, 19, 79–84. [Google Scholar] [CrossRef]
- Ozoliņš, J.; Andersone, Z.; Pupila, A. Status and management prospects of the wolf Canis lupus L. in Latvia. Balt. For. 2001, 7, 63–69. [Google Scholar]
- Ozoliņš, J.; Žunna, A.; Ornicāns, A.; Done, G.; Stepanova, A.; Pilāte, D.; Šuba, J.; Lūkins, M.; Howlett, S.J.; Bagrade, G. Action Plan for Grey Wolf Canis lupus Conservation and Management; LSFRI Silava: Salaspils, Latvia, 2017. [Google Scholar]
- Council Directive 92/43/EEC of 21 May 1992 on the Conservation of Natural Habitats and of Wild Fauna and Flora. Available online: https://eur-lex.europa.eu/legal-content/EN/TXT/?uri=CELEX:31992L0043 (accessed on 24 June 2021).
- Mills, L.S. Conservation of Wildlife Populations: Demography, Genetics, and Management, 2nd ed.; John Wiley & Sons, Ltd.: West Sussex, UK, 2013. [Google Scholar]
- Mason, N.; Ward, M.; Watson, J.E.M.; Venter, O.; Runting, R.K. Global opportunities and challenges for transboundary conservation. Nat. Ecol. Evol. 2020, 4, 694–701. [Google Scholar] [CrossRef]
- Central Statistical Bureau of Latvia. Latvia Statistics in Brief 2020; Central Statistical Bureau of Latvia: Riga, Latvia, 2020. [Google Scholar]
- State Forest Service. Population of Game Species. Available online: https://www.vmd.gov.lv/valsts-meza-dienests/statiskas-lapas/medibas/valsts-meza-dienests/statiskas-lapas/skaitli-un-fakti?id=766 (accessed on 13 June 2020).
- Klevezal, G.A. Age-Related Structures in Zoological Studies of Mammals; Nauka: Moscow, Russia, 1988. (In Russian) [Google Scholar]
- Kirkpatrick, R.L. Physiological indices in wildlife management. In Wildlife Management Techniques Manual, 4th ed.; Shemnitz, S.D., Ed.; The Wildlife Society: Washington, DC, USA, 1980; pp. 99–112. [Google Scholar]
- Fuller, T.K. Wolf population dynamics. In Wolves: Behavior, Ecology, and Conservation; Mech, D.L., Boitani, L., Eds.; University of Chicago Press: Chicago, IL, USA, 2003; pp. 161–191. [Google Scholar]
- Fry, F.E.J. Statistics of a lake trout fishery. Biometrics 1949, 5, 27–67. [Google Scholar] [CrossRef]
- Fry, F.E.J. Assessment of mortalities by use of the virtual population. In Proceedings of the Joint Scientific Meeting of the ICNAF (International Commission for Northwest Atlantic Fisheries), ICES (International Council for the Exploration of the Sea), and FAO (Food and Agriculture Organization of the United Nations) on Fishing Effort, the Effects of Fishing on Resources and the Selectivity of Fishing Gear, Lisbon, Portugal, 27 May–3 June 1957. [Google Scholar]
- Gulland, J.A. Estimation of Mortality Rates. In Annex to Arctic Fisheries Working Group Report; Document No. 3.; International Council for the Exploration of the Sea: Copenhagen, Denmark, 1965. [Google Scholar]
- Pope, J.G. An investigation of the accuracy of virtual population analysis using cohort analysis. ICNAF Res. Bull. 1972, 9, 65–74. [Google Scholar]
- MacCall, A.D. Virtual population analysis (VPA) equations for nonhomogeneous populations, and a family of approximations including improvements on Pope’s cohort analysis. Can. J. Fish. Aquat. Sci. 1986, 43, 2406–2409. [Google Scholar] [CrossRef]
- Skalski, J.R.; Ryding, K.E.; Millspaugh, J. Wildlife Demography: Analysis of Sex, Age, and Count Data; Elsevier Academic Press: Amsterdam, The Netherlands, 2005. [Google Scholar]
- R Core Team. A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2018; Available online: https://www.R-project.org/ (accessed on 23 April 2019).
- Kawata, Y.; Ozoliņš, J.; Baumanis, J. Analysis of wildlife population dynamics using a connected scatter plot: Latvian wild animals as an example. Res. Opin Anim. Vet. Sci. 2013, 3, 50–59. [Google Scholar]
- Ozoliņš, J.; Žunna, A.; Howlett, S.J.; Bagrade, G.; Pilāte, D.; Ornicāns, A. Pēterhofs, Population dynamics of large mammals in Latvia with an emphasis on prey-predator interactions. In Beiträge zur Jagd- und Wildforschung; Bd., 41; Stubbe, M., Ed.; Gesellschaft für Wildtier- und Jagdforschung e.V.: Halle, Germany, 2016; pp. 59–73. [Google Scholar]
- Andersone, Ž. Wolves in Latvia: Past and present. Wolf Print 2003, 16, 13–14. [Google Scholar]
- Andersone-Lilley, Ž.; Balciauskas, L.; Ozolinš, J.; Valdmann, H. Would Baltic wolves vote for the EU? Wolf Print 2005, 23, 9. [Google Scholar]
- Andersone, Ž.; Ozolinš, J. Public perception of large carnivores in Latvia. Ursus 2004, 15, 181–187. [Google Scholar] [CrossRef]
- Žunna, A.; Bagrade, G.; Ozoliņš, J. Attitudes of the General Public and Hunters Towards Wolves in Latvia; Its Predictors and Changes Over Time. Proc. Latv. Acad. Sci. B Nat. Exact Appl. Sci. 2020, 74, 280–286. [Google Scholar] [CrossRef]
- Ballard, W.B.; Whitman, J.S.; Gardner, C.L. Ecology of an exploited wolf population in south-central Alaska. Wildl. Monogr. 1987, 98, 1–54. [Google Scholar]
- Andersone, Ž. Beaver: A new prey of wolves in Latvia? In Beaver Protection, Management, and Utilization in Europe and North America; Busher, P.E., Dzięciołowski, R.M., Eds.; Springer: Boston, MA, USA, 1999; pp. 103–108. [Google Scholar] [CrossRef]
- Žunna, A.; Ozoliņš, J.; Pupila, A. Food habits of the wolf Canis lupus in Latvia based on stomach analyses. Est. J. Ecol. 2009, 58, 141–152. [Google Scholar] [CrossRef] [Green Version]
- Mech, L.D. Canis lupus. Mamm. Species 1974, 37, 1–6. [Google Scholar] [CrossRef]
- Kojola, I. Status and development of wolf population in Finland. In Management Plan for the Wolf Population in Finland; Ministry of Agriculture and Forestry: Helsinki, Finland, 2005; pp. 8–14. [Google Scholar]
- Mech, L.D.; Barber-Meyer, S.M.; Erb, J. Wolf (Canis lupus) generation time and proportion of current breeding females by age. PLoS ONE 2016, 11, e0156682. [Google Scholar] [CrossRef]
- Creel, S.; Rotella, J.J. Meta-analysis of relationships between human offtake, total mortality and population dynamics of gray wolves (Canis lupus). PLoS ONE 2010, 5, e12918. [Google Scholar] [CrossRef]
- Haber, G.C. Biological, conservation, and ethical implications of exploiting and controlling wolves. Conserv. Biol. 1996, 10, 1068–1081. [Google Scholar] [CrossRef] [Green Version]
- Johnson, W.E.; Eizirik, E.; Roelke-Parker, M.; Brien, S.J.O. Applications of genetic concepts and molecular methods to carnivore conservation. In Conservation Biology Series; Gittleman, J.L., Funk, S.M., Macdonald, D.W., Wayne, R.K., Eds.; Cambridge University Press: Cambridge, UK, 2001; pp. 335–358. [Google Scholar]
- Rutledge, L.Y.; Patterson, B.R.; Mills, K.J.; Loveless, K.M.; Murray, D.L.; White, B.N. Protection from harvesting restores the natural social structure of eastern wolf packs. Biol. Conserv. 2010, 143, 332–339. [Google Scholar] [CrossRef]
- Young, S. The Wolves of North America, 1st ed.; American Wildlife Institute: Washington, DC, USA, 1944. [Google Scholar]
- Theberge, J.; Theberge, M. Wolf Country: Eleven Years Tracking the Algonquin Wolves; McClelland & Stewart: Toronto, ON, Canada, 1998. [Google Scholar]
- Jędrzejewska, B.; Jędrzejewski, W. Predation in Vertebrate Communities: The Białowieża Primeval Forest as a Case Study; Springer: Berlin/Heidelberg, Germany, 1998; Volume 135. [Google Scholar]
- Adams, L.G.; Stephenson, R.O.; Dale, B.W.; Ahgook, R.T.; Demma, D.J. Population dynamics and harvest characteristics of wolves in the central Brooks Range, Alaska. Wildl. Monogr. 2008, 170, 1–25. [Google Scholar] [CrossRef]
- Brainerd, S.M.; Andrén, H.; Bangs, E.E.; Bradley, E.H.; Fontaine, J.A.; Hall, W.; Iliopoulos, Y.; Jimenez, M.D.; Jozwiak, E.A.; Liberg, O. The effects of breeder loss on wolves. J. Wildl. Manag. 2008, 72, 89–98. [Google Scholar] [CrossRef]
- Rick, J.A.; Moen, R.A.; Erb, J.D.; Strasburg, J.L. Population structure and gene flow in a newly harvested gray wolf (Canis lupus) population. Conserv. Genet. 2017, 18, 1091–1104. [Google Scholar] [CrossRef]
- Allendorf, F.W.; Ryman, N. The role of genetics in population viability analysis. In Population Viability Analysis; Beissinger, S.R., McCullough, D.R., Eds.; The University of Chicago Press: Chicago, IL, USA, 2002; pp. 50–80. [Google Scholar]
- Smith, D.W.; Bangs, E.E.; Oakleaf, J.K.; Mack, C.; Fontaine, J.; Boyd, D.; Jimenez, M.; Pletscher, D.H.; Niemeyer, C.C.; Meier, T.J. Survival of colonizing wolves in the northern Rocky Mountains of the United States, 1982–2004. J. Wildl. Manag. 2010, 74, 620–634. [Google Scholar] [CrossRef]
- Sidorovich, V.E.; Stolyarov, V.P.; Vorobei, N.N.; Ivanova, N.V.; Jędrzejewska, B. Litter size, sex ratio, and age structure of gray wolves, Canis lupus, in relation to population fluctuations in northern Belarus. Can. J. Zool. 2007, 85, 295–300. [Google Scholar] [CrossRef]
- Webb, N.F.; Allen, J.R.; Merrill, E.H. Demography of a harvested population of wolves (Canis lupus) in west-central Alberta, Canada. Can. J. Zool. 2011, 89, 744–752. [Google Scholar] [CrossRef]
- Hiraiwa-Hasegawa, M. Skewed birth sex ratios in primates: Should high-ranking mothers have daughters or sons? Trends Ecol. Evol. 1993, 8, 395–2400. [Google Scholar] [CrossRef]
- Jędrzejewski, W.; Branicki, W.; Veit, C.; Međugorac, I.; Pilot, M.; Bunevich, A.N.; Jędrzejewska, B.; Schmidt, K.; Theuerkauf, J.; Okarma, H. Genetic diversity and relatedness within packs in an intensely hunted population of wolves Canis lupus. Acta Theriol. 2005, 50, 3–22. [Google Scholar] [CrossRef]
- Pavlov, M.P. Wolf; Agropomizdat: Moscow, Russia, 1990. (In Russian) [Google Scholar]
- Jędrzejewska, B.; Jędrzejewski, W.; Bunevich, A.N.; Minkowski, L.; Okarma, H. Population dynamics of Wolves Canis lupus in Białowieża Primeval Forest (Poland and Belarus) in relation to hunting by humans, 1847–1993. Mamm. Rev. 1996, 26, 103–126. [Google Scholar] [CrossRef]
- Ozoliņš, J.; Žunna, A.; (Latvian State Foresth Research Institute Silava, Salaspils, Latvia). Personal communication, 2021.
- Fernández-Gil, A.; Naves, J.; Ordiz, A.; Quevedo, M.; Revilla, E.; Delibes, M. Conflict Misleads Large Carnivore Management and Conservation: Brown Bears and Wolves in Spain. PLoS ONE 2016, 11, e0151541. [Google Scholar] [CrossRef] [PubMed] [Green Version]



Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Šuba, J.; Žunna, A.; Bagrade, G.; Done, G.; Lūkins, M.; Ornicāns, A.; Pilāte, D.; Stepanova, A.; Ozoliņš, J. Closer to Carrying Capacity: Analysis of the Internal Demographic Structure Associated with the Management and Density Dependence of a Controlled Wolf Population in Latvia. Sustainability 2021, 13, 9783. https://doi.org/10.3390/su13179783
Šuba J, Žunna A, Bagrade G, Done G, Lūkins M, Ornicāns A, Pilāte D, Stepanova A, Ozoliņš J. Closer to Carrying Capacity: Analysis of the Internal Demographic Structure Associated with the Management and Density Dependence of a Controlled Wolf Population in Latvia. Sustainability. 2021; 13(17):9783. https://doi.org/10.3390/su13179783
Chicago/Turabian StyleŠuba, Jurģis, Agrita Žunna, Guna Bagrade, Gundega Done, Mārtiņš Lūkins, Aivars Ornicāns, Digna Pilāte, Alda Stepanova, and Jānis Ozoliņš. 2021. "Closer to Carrying Capacity: Analysis of the Internal Demographic Structure Associated with the Management and Density Dependence of a Controlled Wolf Population in Latvia" Sustainability 13, no. 17: 9783. https://doi.org/10.3390/su13179783
APA StyleŠuba, J., Žunna, A., Bagrade, G., Done, G., Lūkins, M., Ornicāns, A., Pilāte, D., Stepanova, A., & Ozoliņš, J. (2021). Closer to Carrying Capacity: Analysis of the Internal Demographic Structure Associated with the Management and Density Dependence of a Controlled Wolf Population in Latvia. Sustainability, 13(17), 9783. https://doi.org/10.3390/su13179783






