Conversion of Marine Litter from Venice Lagoon into Marine Fuels via Thermochemical Route: The Overview of Products, Their Yield, Quality and Environmental Impact
Abstract
:1. Introduction
1.1. Generalities on Marine Litter
1.2. Recycling of Marine Litter
1.3. Thermochemical Route to Recycle Marine Litter
2. Materials and Methods
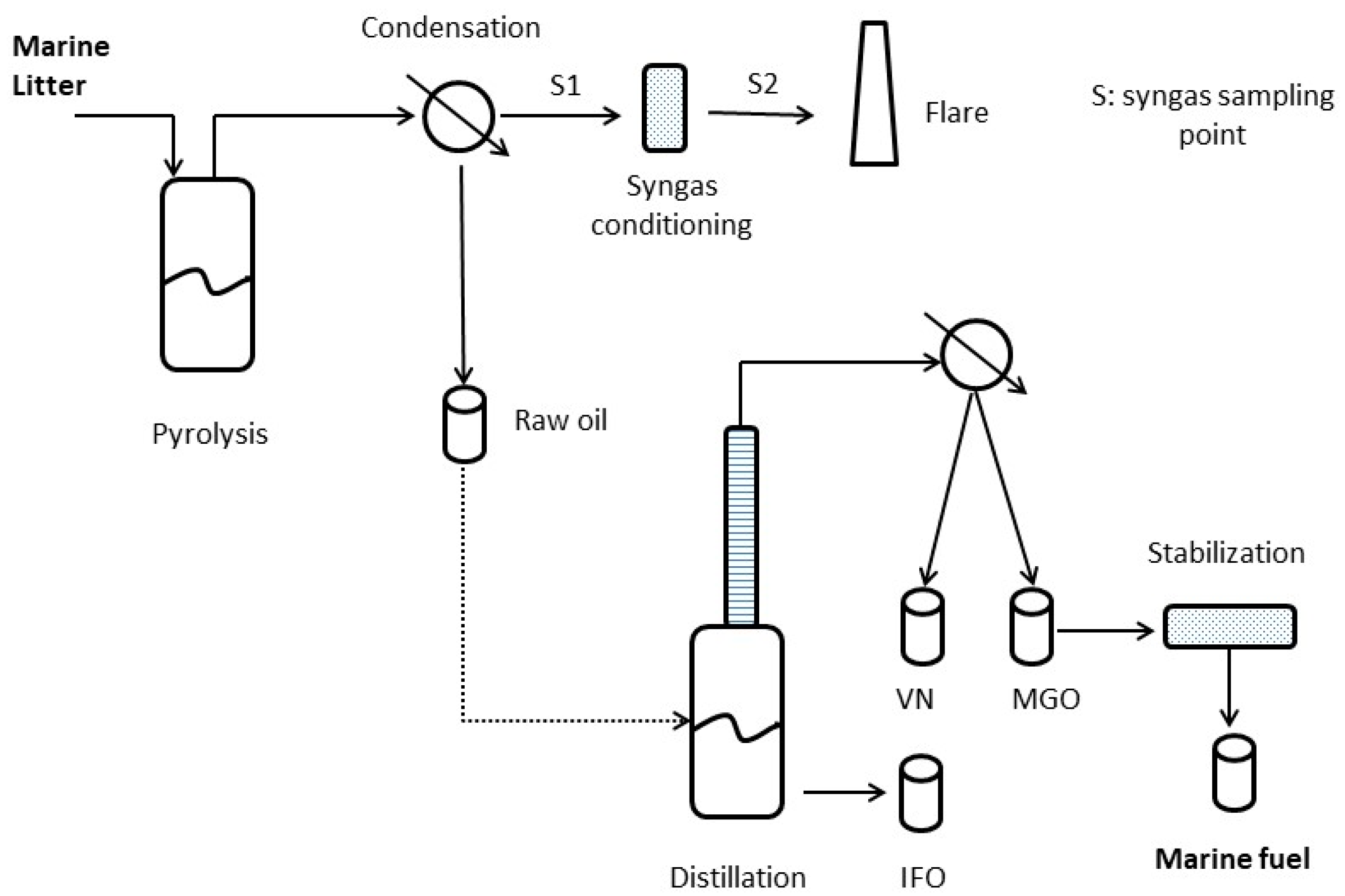
2.1. Pyrolysis and Distillation Setup
- A 100 L pyrolysis reactor, electrically heated and batch operated, directly connected with condensation train and raw pyrolysis oil collection tank. After the collection tank, the noncondensable syngas is conditioned before being flared adiabatically.
- A 50 L atmospheric distillation unit to fractionate pyrolysis oil into the useful target products: a light distillate boiling up to 170 °C (virgin naphtha: VN), the middle distillate boiling up to 320 °C (marine gasoil: MGO) and the intermediate fuel oil (IFO), which is collected as residue from the distillation flask.
2.2. Monitoring of Environmentally Critical Substances
2.3. Marine Litter Collection
2.4. Marine Litter Processing and Product Characterization
3. Results and Discussion
3.1. Product Yields
3.2. Product Quality
3.3. Pollutants in Noncondensed Gas
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- MacArthur, D.E.; Waughray, D.; Stuchtey, M.R. The New Plastics Economy, Rethinking the Future of Plastics; World Economic Forum: Cologny, Switzerland, 2016. [Google Scholar]
- Carson, H.S.; Lamson, M.R.; Nakashima, D.; Toloumu, D.; Hafner, J.; Maximenko, N.; McDermid, K.J. Tracking the sources and sinks of local marine debris in Hawaii. Mar. Environ. Res. 2013, 84, 76–83. [Google Scholar] [CrossRef]
- Barnes, D.K.; Galgani, F.; Thompson, R.C.; Barlaz, M. Accumulation and fragmentation of plastic debris in global environments. Philos. Trans. R. Soc. B Biol. Sci. 2009, 364, 1985–1998. [Google Scholar] [CrossRef] [Green Version]
- Obbard, R.W.; Sadri, S.; Wong, Y.Q.; Khitun, A.A.; Baker, I.; Thompson, R.C. Global warming releases microplastic legacy frozen in Arctic Sea ice. Earth’s Future 2014, 2, 315–320. [Google Scholar] [CrossRef]
- Lechner, A.; Keckeis, H.; Lumesberger-Loisl, F.; Zens, B.; Krusch, R.; Tritthart, M.; Glas, M.; Schludermann, E. The Danube so colourful: A potpourri of plastic litter outnumbers fish larvae in Europe’s second largest river. Environ. Pollut. 2014, 188, 177–181. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jambeck, J.R.; Geyer, R.; Wilcox, C.; Siegler, T.R.; Perryman, M.; Andrady, A.; Narayan, R.; Law, K.L. Plastic waste inputs from land into the ocean. Science 2015, 347, 768–771. [Google Scholar] [CrossRef] [PubMed]
- Hoornweg, D.; Bhada-Tata, P.; Kennedy, C. Environment: Waste production must peak this century. Nat. News 2013, 502, 615. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Eriksen, M.; Lebreton, L.C.; Carson, H.S.; Thiel, M.; Moore, C.J.; Borerro, J.C.; Galgani, F.; Ryan, P.G.; Reisser, J. Plastic pollution in the world’s oceans: More than 5 trillion plastic pieces weighing over 250,000 tons afloat at sea. PLoS ONE 2014, 9, e111913. [Google Scholar] [CrossRef] [Green Version]
- UNEP; Regional Seas Programme. Marine Litter: An Analytical Overview; United Nations Environment Programme: Nairobi, Kenya, 2005. [Google Scholar]
- Goldstein, M.C.; Goodwin, D.S. Gooseneck barnacles (Lepas spp.) ingest microplastic debris in the North Pacific Subtropical Gyre. PeerJ 2013, 1, e184. [Google Scholar] [CrossRef] [Green Version]
- Browne, M.A.; Niven, S.J.; Galloway, T.S.; Rowland, S.J.; Thompson, R.C. Microplastic moves pollutants and additives to worms, reducing functions linked to health and biodiversity. Curr. Biol. 2013, 23, 2388–2392. [Google Scholar] [CrossRef] [Green Version]
- Van Cauwenberghe, L.; Janssen, C.R. Microplastics in bivalves cultured for human consumption. Environ. Pollut. 2014, 193, 65–70. [Google Scholar] [CrossRef]
- Ragaert, K.; Delva, L.; Van Geem, K. Mechanical and chemical recycling of solid plastic waste. Waste Manag. 2017, 69, 24–58. [Google Scholar] [CrossRef] [PubMed]
- Bergmann, M.; Gutow, L.; Klages, M. (Eds.) Marine Anthropogenic Litter; Springer: Berlin/Heidelberg, Germany, 2015. [Google Scholar]
- Iñiguez, M.E.; Conesa, J.A.; Fullana, A. Marine debris occurrence and treatment: A review. Renew. Sustain. Energy Rev. 2016, 64, 394–402. [Google Scholar] [CrossRef] [Green Version]
- Jung, R.T.; Sung, H.G.; Chun, T.B.; Keel, S.I. Practical engineering approaches and infrastructure to address the problem of marine debris in Korea. Mar. Pollut. Bull. 2010, 60, 1523–1532. [Google Scholar] [CrossRef] [PubMed]
- Oosterhuis, F.; Papyrakis, E.; Boteler, B. Economic instruments and marine litter control. Ocean Coast. Manag. 2014, 102, 47–54. [Google Scholar] [CrossRef]
- Vince, J.; Stoett, P. From problem to crisis to interdisciplinary solutions: Plastic marine debris. Mar. Policy 2018, 96, 200–203. [Google Scholar] [CrossRef]
- Achilias, D.S.; Roupakias, C.; Megalokonomos, P.; Lappas, A.A.; Antonakou, Ε.V. Chemical recycling of plastic wastes made from polyethylene (LDPE and HDPE) and polypropylene (PP). J. Hazard. Mater. 2007, 149, 536–542. [Google Scholar] [CrossRef]
- Rahimi, A.; García, J. Chemical recycling of waste plastics for new materials production. Nat. Rev. Chem. 2017, 1, 0046. [Google Scholar] [CrossRef]
- Thiounn, T.; Smith, R.C. Advances and approaches for chemical recycling of plastic waste. J. Appl. Polym. Sci. 2020, 58, 1347–1364. [Google Scholar] [CrossRef] [Green Version]
- Ronchi, F.; Galgani, F.; Binda, F.; Mandić, M.; Peterlin, M.; Tutman, P.; Anastasopoulou, A.; Fortibuoni, T. Fishing for Litter in the Adriatic-Ionian macroregion (Mediterranean Sea): Strengths, weaknesses, opportunities and threats. Mar. Policy 2019, 100, 226–237. [Google Scholar] [CrossRef]
- Aguado, J.; Serrano, D.P.; Escola, J.M. Catalytic upgrading of plastic wastes. In Feedstock Recycling and Pyrolysis of Waste Plastics: Converting Waste Plastics into Diesel and Other Fuels; Scheirs, J., Kaminsky, W., Eds.; John Wiley Sons: Hoboken, NJ, USA, 2006; pp. 73–110. [Google Scholar]
- Marianne, B. Composition of liquid fuels derived from the pyrolysis of plastics. In Feedstock Recycling and Pyrolysis of Waste Plastics: Converting Waste Plastics into Diesel and Other Fuels; Scheirs, J., Kaminsky, W., Eds.; John Wiley Sons: Hoboken, NJ, USA, 2006; pp. 315–344. [Google Scholar]
- Buekens, A.G.; Huang, H. Catalytic plastics cracking for recovery of gasoline-range hydrocarbons from municipal plastic wastes. Resour. Conserv. Recycl. 1998, 23, 163–181. [Google Scholar] [CrossRef]
- Buekens, A. Introduction to feedstock recycling of plastics. In Feedstock Recycling and Pyrolysis of Waste Plastics: Converting Waste Plastics into Diesel and Other Fuels; Scheirs, J., Kaminsky, W., Eds.; John Wiley Sons: Hoboken, NJ, USA, 2006; pp. 1–41. [Google Scholar]
- Datta, J.; Kopczyńska, P. From polymer waste to potential main industrial products: Actual state of recycling and recovering. Crit. Rev. Environ. Sci. Technol. 2016, 46, 905–946. [Google Scholar] [CrossRef]
- Faussone, G.C. Transportation fuel from plastic: Two cases of study. Waste Manag. 2018, 73, 416–423. [Google Scholar] [CrossRef]
- Bezergianni, S.; Dimitriadis, A.; Faussone, G.C.; Karonis, D. Alternative diesel from waste plastics. Energies 2017, 10, 1750. [Google Scholar] [CrossRef] [Green Version]
- Fuels Europe Marine Fuel Consumption (Global and in The EU). Available online: https://www.fuelseurope.eu/dataroom/static-graphs/ (accessed on 16 November 2020).
- Margnet. Available online: https://ec.europa.eu/easme/en/margnet-mapping-and-recycling-marine-litter-and-ghost-nets-sea-floor (accessed on 4 June 2019).
- Scott, D.S.; Czernik, S.R.; Piskorz, J.; Radlein, D.S.A. Fast pyrolysis of plastic wastes. Energy Fuels 1990, 4, 407–411. [Google Scholar] [CrossRef]
- Hartenstein, H.U.; Licata, A. Modern Technologies to Reduce Emissions of Dioxins and Furans from Waste Incineration; Babcock Borsig Power, DB Riley: Worcester, MA, USA, 2008. [Google Scholar]
- Nielsen, M.; Jurasek, P.; Hayashi, J.I.; Furimsky, E. Formation of toxic gases during pyrolysis of polyacrylonitrile and nylons. J. Anal. Appl. Pyrolysis 1995, 35, 43–51. [Google Scholar] [CrossRef]
- Galgani, F.; Hanke, G.; Maes, T. Global Distribution, Composition and Abundance of Marine Litter. In Marine Anthropogenic Litter; Bergmann, M., Gutow, L., Klages, M., Eds.; Springer: Cham, Switzerland, 2015. [Google Scholar] [CrossRef] [Green Version]
- Consoli, P.; Falautano, M.; Sinopoli, M.; Perzia, P.; Canese, S.; Esposito, V.; Battaglia, P.; Romeo, T.; Andaloro, F.; Galgani, F.; et al. Composition and abundance of benthic marine litter in a coastal area of the central Mediterranean Sea. Mar. Pollut. Bull. 2018, 136, 243–247. [Google Scholar] [CrossRef] [PubMed]
- Angiolillo, M.; di Lorenzo, B.; Farcomeni, A.; Bo, M.; Bavestrello, G.; Santangelo, G.; Cau, A.; Mastascusa, V.; Cau, A.; Sacco, F.; et al. Distribution and assessment of marine debris in the deep Tyrrhenian Sea (NW Mediterranean Sea, Italy). Mar. Pollut. Bull. 2015, 92, 149–159. [Google Scholar] [CrossRef]
- Plastic Europe “Plastics The Facts 2018”. Available online: https://www.plasticseurope.org/application/files/6315/4510/9658/Plastics_the_facts_2018_AF_web.pdf (accessed on 14 May 2019).
- de Marco Rodriguez, I.; Laresgoiti, M.F.; Cabrero, M.A.; Torres, A.; Chomon, M.J.; Caballero, B. Pyrolysis of scrap tyres. Fuel Process. Technol. 2001, 72, 9–22. [Google Scholar] [CrossRef]
- International Organization for Standardization. Petroleum Products—Fuels (Class F)—Specifications of Marine Fuels; International Organization for Standardization: Geneva, Switzerland, 2017; ISO8217:2017. [Google Scholar]
- Kang, M.J.; Yu, H.J.; Jegal, J.; Kim, H.S.; Gil Cha, H. Depolymerization of PET into Terephthalic Acid in Neutral Media Catalyzed by the ZSM-5 Acidic Catalyst. Chem. Eng. J. 2020, 398, 125655. [Google Scholar] [CrossRef]
- Levchik, S.V.; Weil, E.D.; Lewin, M. Thermal decomposition of aliphatic nylons. Polym. Int. 1999, 48, 532–557. [Google Scholar] [CrossRef]
- Czernik, S.; Elam, C.C.; Evans, R.J.; Meglen, R.R.; Moens, L.; Tatsumoto, K. Catalytic pyrolysis of nylon-6 to recover caprolactam. J. Anal. Appl. Pyrolysis 1998, 46, 51–64. [Google Scholar] [CrossRef]
- Kim, S.S.; Jeon, J.K.; Park, Y.K.; Kim, S. Thermal pyrolysis of fresh and waste fishing nets. Waste Manag. 2005, 25, 811–817. [Google Scholar] [CrossRef] [PubMed]
- American Bureau of Shipping (ABS). Notes on heavy fuel oil. In Handbook, H.F.O.; Rosendahls: Hørsholm, Denmark, 1984; Volume 31. [Google Scholar]
- International Maritime Organization (IMO). MARPOL Annex VI: Prevention of Air Pollution from Ships; International Maritime Organization: London, UK, 2010. [Google Scholar]
- Yoshioka, T.; Grause, G.; Eger, C.; Kaminsky, W.; Okuwaki, A. Pyrolysis of poly (ethylene terephthalate) in a fluidised bed plant. Polym. Degrad. Stab. 2004, 86, 499–504. [Google Scholar] [CrossRef]
- Artetxe, M.; Lopez, G.; Amutio, M.; Elordi, G.; Olazar, M.; Bilbao, J. Operating conditions for the pyrolysis of poly-(ethylene terephthalate) in a conical spouted-bed reactor. Ind. Eng. Chem. Res. 2010, 49, 2064–2069. [Google Scholar] [CrossRef]
- European Committee for Standardization. EN228:2012 Automotive Fuels-Unleaded Petrol-Requirement and Test Methods; European Committee for Standardization: Brussels, Belgium, 2012. [Google Scholar]


| Trial # | ML | Oil | Residue (a) | Gas (b) | Yield Oil | Yield Res. | Yield Gas |
|---|---|---|---|---|---|---|---|
| kg | kg | kg | kg | wt% | wt% | wt% | |
| 1 (ML + nets) | 9.0 | 5.7 | 1.7 | 1.6 | 63.3 | 18.9 | 17.8 |
| 2 (ML + nets) | 6.6 | 3.2 | 1.8 | 1.6 | 48.5 | 27.3 | 24.2 |
| 3 (largely nets) | 6.3 | 1.0 (c) | 2.7 | 2.6 | 15.9 | 42.9 | 41.3 |
| 4 (sunk ML) | 5.0 | 2.1 | 1.7 | 1.2 | 42.0 | 34.0 | 24.0 |
| 5 (sunk ML) | 5.0 | 2.4 | 1.0 | 1.6 | 48.0 | 20.0 | 32.0 |
| 6 (sunk ML) | 5.0 | 2.6 | 1.0 | 1.4 | 52.0 | 20.0 | 28.0 |
| 7 (sunk ML) | 7.5 | 4.0 | 3.2 | 0.3 | 53.3 | 42.7 | 4.0 |
| 8 (ecoleather) | 9.0 | 1.8 | 5.5 | 1.7 | 19.6 | 61.1 | 19.3 |
| 9 (sunk ML) | 7.5 | 3.6 | 1.0 | 2.9 | 48.0 | 13.3 | 38.7 |
| 10 (sunk ML) | 11 | 5.2 | 2.3 | 3.5 | 47.3 | 20.9 | 31.8 |
| 11 (ML) | 11.5 | 7.2 | 2.0 | 2.3 | 62.6 | 17.4 | 20.0 |
| 12 (rubbers) | 6.0 | 1.9 | 3.5 | 0.6 | 32.0 | 58.3 | 9.7 |
| 13 (ML + rubber) | 6.2 | 1.6 | 1.2 | 3.4 | 25.8 | 19.4 | 54.8 |
| 14 (floating) | 4.0 | 2.4 | 0.5 | 1.1 | 60.0 | 12.5 | 27.5 |
| TOT ML | 99.6 | 44.7 | 29.1 | 25.8 | 44.9 | 29.2 | 25.9 |
| Scrap tire | 3.0 | 0.8 | 1.3 | 0.9 | 26.7 | 43.3 | 30.0 |
| ML incl. tire | 102.6 | 45.5 | 30.4 | 26.7 | 44.3 | 29.6 | 26.1 |
| Feed | VN | MGO | IFO | H2O | Losses | |
|---|---|---|---|---|---|---|
| Volume (L) | 34 | 8 | 17.5 | 2 | 3.6 | 2.9 |
| Fraction (vol%) | 100.0% | 23.5% | 51.5% | 5.9% | 10.6% | 8.5% |
| Parameter | Value | Unit |
|---|---|---|
| Kinematic viscosity (50 °C) | 8.61 | mm2/s |
| Density (15 °C) | 841.2 | kg/m3 |
| Cetane index | 70.6 | - |
| Sulfur content | 34 | ppm |
| Flash point | >100 | °C |
| Hydrogen sulfide | <2 | ppm |
| Acid value | 0.04 | mg KOH/g |
| Sediment content | 1.58 | wt% (m/m) |
| Carbon residue | 1.86 | wt% (m/m) |
| Pour point | 34 | °C |
| Water content | 0.58 | vol% |
| Ash content (775 °C) | 0.667 | wt% |
| Vanadium (V) | <0.5 | mg/kg |
| Sodium (Na) | 959 | mg/kg |
| Aluminum (Al) | <0.5 | mg/kg |
| Silicon (Si) | 130 | mg/kg |
| Calcium (Ca) | 13.8 | mg/kg |
| Zinc (Zn) | 1.4 | mg/kg |
| Phosphorus (P) | 48.2 | mg/kg |
| Cloud point | >20 | °C |
| HFRR (Lubricity at 60 °C) | 210 | µm |
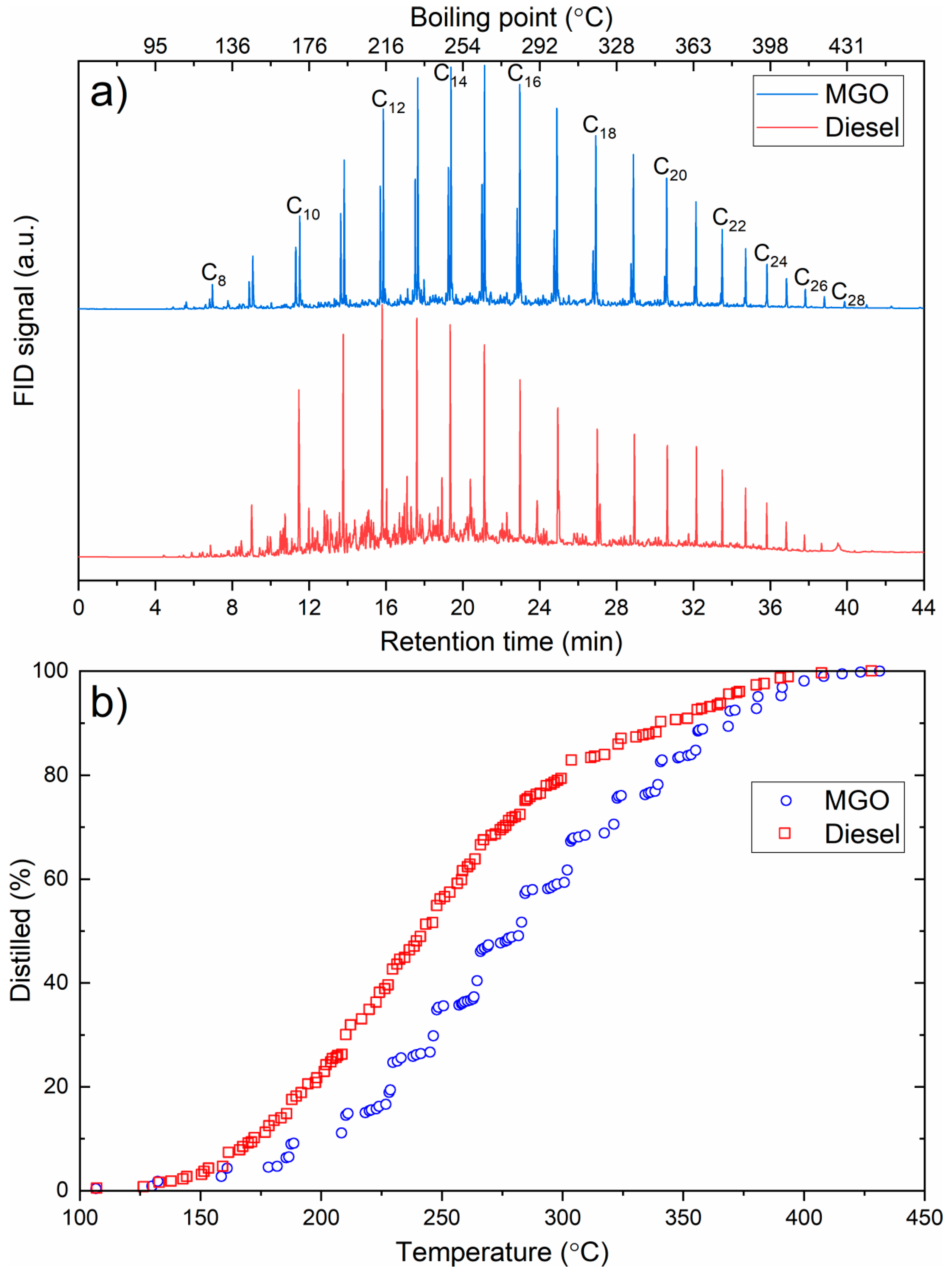
| Parameter | Result | Unit |
|---|---|---|
| Kinematic viscosity (50 °C) | 1.848 | mm2/s |
| Density (15 °C) | 802.9 | kg/m3 |
| Cetane index | 61.3 | - |
| 10% (V/V) recovery | 178.3 | °C |
| 50% (V/V) recovery | 257.1 | °C |
| 90% (V/V) recovery | 347.0 | °C |
| Sulfur content | 196 | ppm |
| Flash point | 35.0 | °C |
| Flash point (improved) | 58.0 | °C |
| Hydrogen sulfide | <2 | ppm |
| Acid value | 0.136 | mg KOH/g |
| Sediment content | 0.02 | % (m/m) |
| Carbon residue | <0.10 | % (m/m) |
| Pour point | −6 | °C |
| Water content | 0.01 | % (V/V) |
| Ash content (775 °C) | <0.001 | % (m/m) |
| Vanadium (V) | <0.5 | mg/kg |
| Sodium (Na) | <0.5 | mg/kg |
| Aluminum (Al) | 0.5 | mg/kg |
| Silicon (Si) | 113 | mg/kg |
| Calcium (Ca) | <0.5 | mg/kg |
| Zinc (Zn) | <0.5 | mg/kg |
| Phosphorus (P) | 4.2 | mg/kg |
| Cloud point | 14 | °C |
| HFRR (Lubricity at 60 °C) | 240 | µm |
| Oxidation stability | 15.46 | min |
| S1 | S2 | |
|---|---|---|
| Pollutant | ppm | ppm |
| Pentane C5H12 | >1500 | >1500 |
| Hydrogen sulfide H2S | <0.2 * | <0.2 * |
| Hydrochloric acid HCl | <0.2 * | <0.2 * |
| Sulfur dioxide SO2 | n.a. | <<0.5 |
| Ammonia NH3 | >600 | 100 |
| Hydrogen fluoride HF | <0.5 * | <0.5 * |
| Carbon monoxide CO | >700 | >700 |
| Hydrogen cyanide HCN | 5 | 0 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Faussone, G.C.; Kržan, A.; Grilc, M. Conversion of Marine Litter from Venice Lagoon into Marine Fuels via Thermochemical Route: The Overview of Products, Their Yield, Quality and Environmental Impact. Sustainability 2021, 13, 9481. https://doi.org/10.3390/su13169481
Faussone GC, Kržan A, Grilc M. Conversion of Marine Litter from Venice Lagoon into Marine Fuels via Thermochemical Route: The Overview of Products, Their Yield, Quality and Environmental Impact. Sustainability. 2021; 13(16):9481. https://doi.org/10.3390/su13169481
Chicago/Turabian StyleFaussone, Gian Claudio, Andrej Kržan, and Miha Grilc. 2021. "Conversion of Marine Litter from Venice Lagoon into Marine Fuels via Thermochemical Route: The Overview of Products, Their Yield, Quality and Environmental Impact" Sustainability 13, no. 16: 9481. https://doi.org/10.3390/su13169481
APA StyleFaussone, G. C., Kržan, A., & Grilc, M. (2021). Conversion of Marine Litter from Venice Lagoon into Marine Fuels via Thermochemical Route: The Overview of Products, Their Yield, Quality and Environmental Impact. Sustainability, 13(16), 9481. https://doi.org/10.3390/su13169481







